Stability Indicating RP-HPLC Method for the Estimation of Impurities in Esomeprazole Gastro-Resistant Tablets by AQbD Approach
Announcing a new article publication for BIO Integration journal. Esomeprazole (ESO) gastro-resistant tablets (40 mg) are sold under the brand name, Zosa, which effectively manages conditions associated with the overproduction of gastric acid, including peptic ulcer disease and Zollinger-Ellison syndrome. This article quantifies impurities in esomeprazole using advanced analytical techniques known as analytical quality by design with high-performance liquid chromatography.
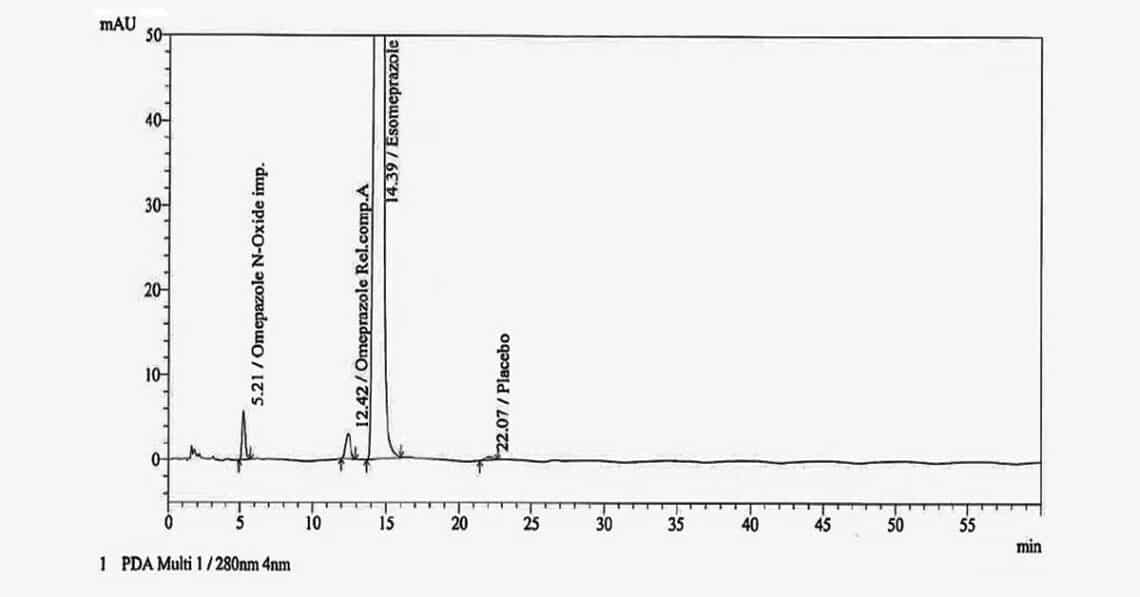
Buffer selection (pH 7.6) and mobile phase composition (75:25 v/v) were optimized utilizing a YMC C18 column (150 mm × 4.6 mm; particle size, 3 μm) with a flow rate of 1.0 mL/min. The analyte was monitored with a UV/PDA detector at a wavelength of 280 nm. The stability-indicating nature of the method was confirmed based on forced degradation studies. The method validation was performed per ICH guidelines. Linearity, specificity, limit of detection, limit of quantification, precision, accuracy, solution stability, and robustness parameters were validated.
All validation parameters were within an acceptable range. Excellent linearity with correlation coefficient values > 0.99 was achieved across the quantification limit. The solution stability study demonstrated no significant increase in percent impurity over a 24-h period. Analytical quality by design was instrumental in defining the design range for buffer pH and mobile phase composition, ensuring robust method performance. It was confirmed that 75% buffer solution, 25% acetonitrile, and pH 7.6 were the ideal conditions for determination of ESO impurities.
The validated method provides a reliable tool for accurately quantifying impurities in ESO tablet formulations.
https://www.scienceopen.com/hosted-document?doi=10.15212/bioi-2024-0018
BIO Integration is fully open access journal which will allow for the rapid dissemination of multidisciplinary views driving the progress of modern medicine. As part of its mandate to help bring interesting work and knowledge from around the world to a wider audience, BIOI will actively support authors through open access publishing and through waiving author fees in its first years. Also, publication support for authors whose first language is not English will be offered in areas such as manuscript development, English language editing and artwork assistance.
BIOI is now open for submissions; articles can be submitted online at: https://mc04.manuscriptcentral.com/bioi
There are no author submission or article processing fees.
Please visit www.bio-integration.org to learn more about the journal.
Editorial Board: https://bio-integration.org/editorial-board/
BIOI is available on the ScienceOpen platform.
Follow BIOI on Twitter @JournalBio; Facebook (https://www.facebook.com/BIO-Integration-Journal-108140854107716/) and LinkedIn (https://www.linkedin.com/company/bio-integration-journal/).
ISSN 2712-0074
eISSN 2712-0082
Vikram Gharge, Anil Gadhe and Vikas Mohite et al. Stability Indicating RP-HPLC Method for the Estimation of Impurities in Esomeprazole Gastro-Resistant Tablets by AQbD Approach. BIOI. 2024. Vol. 5(1). DOI: 10.15212/bioi-2024-0018