Renal Effects of Graphite Oxide Sheets in Albino Rats (Rattus norvegicus): A Preliminary Study
1Department of Zoology, University of Sialkot, Punjab, Pakistan
2Government College University Faisalabad, Punjab, Pakistan
3Department of Mathematics, University of Sialkot, Punjab, Pakistan
4Department of Chemistry, University of Sialkot, Punjab, Pakistan
5Department of Biology, Faculty of Science and Technology, Universitas Airlangga, Surabaya, Indonesia
6Division of Research and Development, Jalan Tengah, Surabaya, Indonesia
7Virtual Research Center for Bioinformatics and Biotechnology, Surabaya, Indonesia
8Postgraduate School, Universitas Airlangga, Surabaya, Indonesia
9Uttaranchal Institute of Pharmaceutical Sciences, Uttaranchal University, Dehradun, India
*Correspondence to: Arif N. M. Ansori, E-mail: ansori.anm@gmail.com
Received: July 12 2024; Revised: August 13 2024; Accepted: September 2 2024; Published Online: November 14 2024
Cite this paper:
Muhammad AN, Ijaz A, Khalid A et al. Renal Effects of Graphite Oxide Sheets in Albino Rats (Rattus norvegicus): A Preliminary Study. BIO Integration 2024; 5: 1–9.
DOI: 10.15212/bioi-2024-0052. Available at: https://bio-integration.org/
Download citation
© 2024 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Although diamond-like carbon-coated joint replacements are widely recommended for full bone replacements in humans, their clinical application is substantially limited by contamination with wear particles, specifically graphite nanoparticles, which are cytotoxic. This study was aimed at assessing the adverse effects of graphite oxide (GO) sheets on the blood and renal tissues of adult male albino rats. A total of 25 albino rats were procured from the Government College University Faisalabad, Punjab, Pakistan, and acclimated for 7 days in well-ventilated enclosures, after approval from the ethical committee at the University of Sialkot. The rats were randomly assigned to the following groups: a control group receiving no treatment, a vehicle control group receiving normal saline, and three treatment groups (G1, G2, and G3, administered GO nanosheets at doses of 5, 6, or 7 mg/kg body weight, respectively). Treatments were delivered through intraperitoneal injection on alternating days over 28 days. Animal mortality, hematological parameters, and kidney histology were assessed. The control and vehicle control groups showed normal findings, whereas the groups exposed to GO exhibited highly significant pathological changes in renal function tests (p<0.05). Histological alterations were more severe in the moderate- and high-dose treatment groups than the low-dose group, which displayed typical histological features. The treatment groups exhibited various histological changes, including vacuolation, renal fibrosis, inflammation, and tubular damage, which were significantly more pronounced in the G2 and G3 groups than the G1 group. Thus, exposure to GO sheets resulted in detrimental effects on renal tissues in albino rats. The findings suggested that the investigated doses of GO have detrimental effects on the health of living organisms.
Keywords
Graphite oxide sheet, histopathology, kidney, medicine, Rattus norvegicus.
Introduction
Graphite oxide (GO) is a constituent of graphene oxide, a derivative of graphene. Graphene is a two-dimensional material consisting of a single layer of carbon atoms arranged in a hexagonal lattice [1]. GO is composed of stacked layers of graphene oxide sheets with oxygen-containing functional groups such as hydroxyl, epoxy, and carboxyl groups attached to the surface [2, 3]. GO sheets can induce inflammation and cell death in rats. However, owing to their high biocompatibility and low toxicity, carbon nanoparticles have substantial potential as a biomaterial for both diagnostic and therapeutic applications [4, 5].
GO sheets are biocompatible and hence suitable for use in biomedical applications such as drug delivery, imaging, and tissue engineering [6, 7]. Extensive research has been conducted both in vitro and in vivo to explore the properties and potential applications of GO. In vitro studies involve testing the effects of GO on cells and tissues outside a living organism [8, 9]. GO sheets have broad potential applications in fields including drug delivery, biosensors, and tissue engineering [10]. For instance, GO has been used as a drug carrier for cancer therapy, thus effectively delivering drugs to cancer cells while minimizing harm to healthy cells [11, 12].
The inflammation induced by graphene oxide in cells contributes to tissue damage and impaired kidney function. Although prior studies have not specifically addressed the effects of GO on the kidneys, they have suggested that graphene-based materials, including GO, might potentially cause kidney toxicity [5, 13]. A study published in the journal Nanotoxicology has investigated the potential toxicity of graphene oxide sheets in the kidneys in rats [14]. High doses of graphene oxide sheets have been found to result in significant kidney tissue damage, including inflammation [15–18].
Methods
Experimental setup
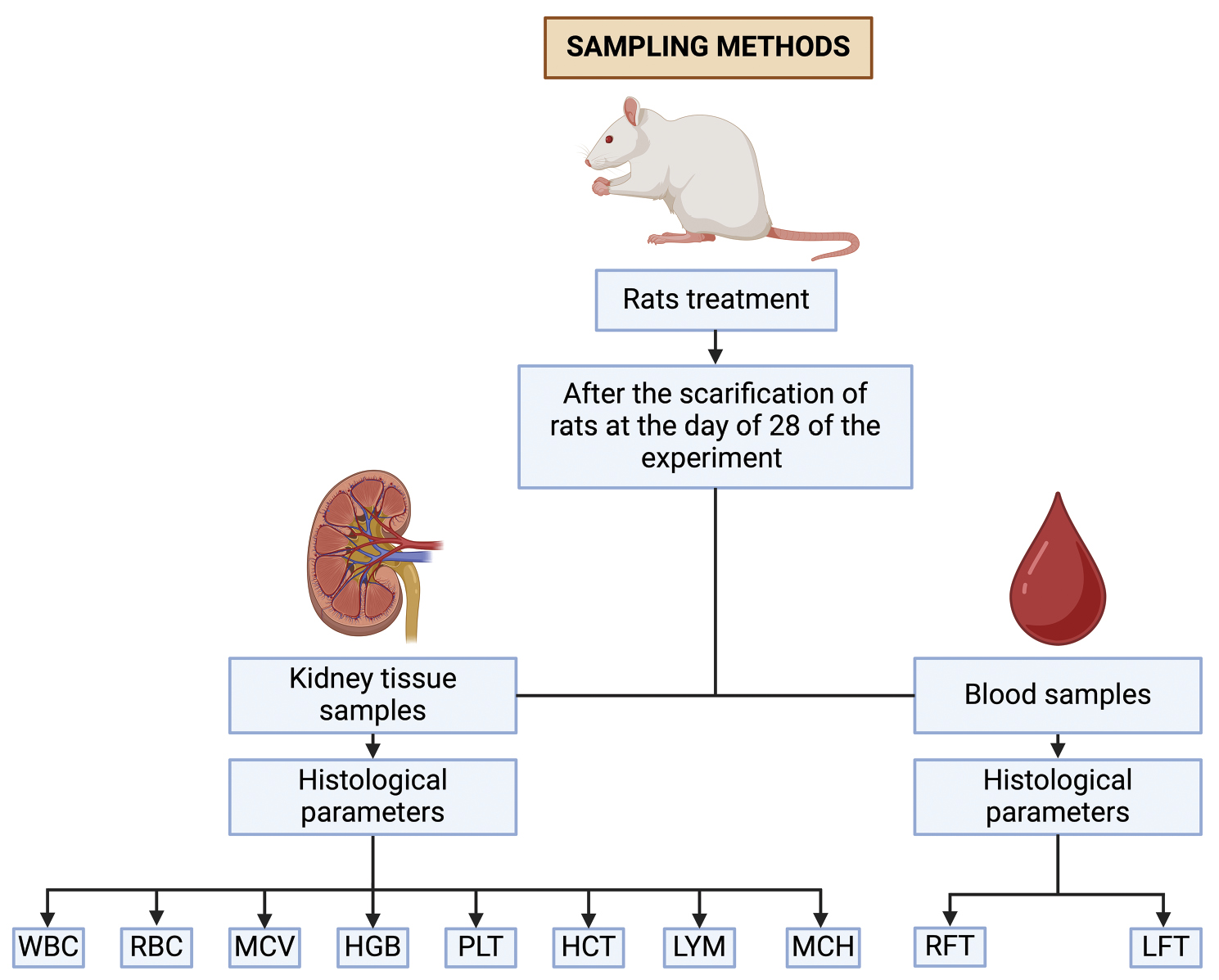
A total of 25 post-weaning albino rats, all of similar weight, were obtained from the animal facility at Government College University, Faisalabad, Punjab, Pakistan. Ethical approval was granted by the Ethics Committee of the University of Sialkot, Punjab, Pakistan (Certificate No. USKT/EIRB/02). The rats were divided into five groups of five animals each. The rats were housed in steel cages under standard lighting conditions and were given free access to water and food. Before the start of the experiment, the rats were acclimated for 7 days. During that period, they were maintained at a temperature of 25±2° C, with a humidity level of 35±5% and a 12:12 day-night cycle. The sampling procedure in albino rats is shown in a flowchart (Figure 1).
Figure 1 Flowchart of sampling methods.
Toxicity assessment
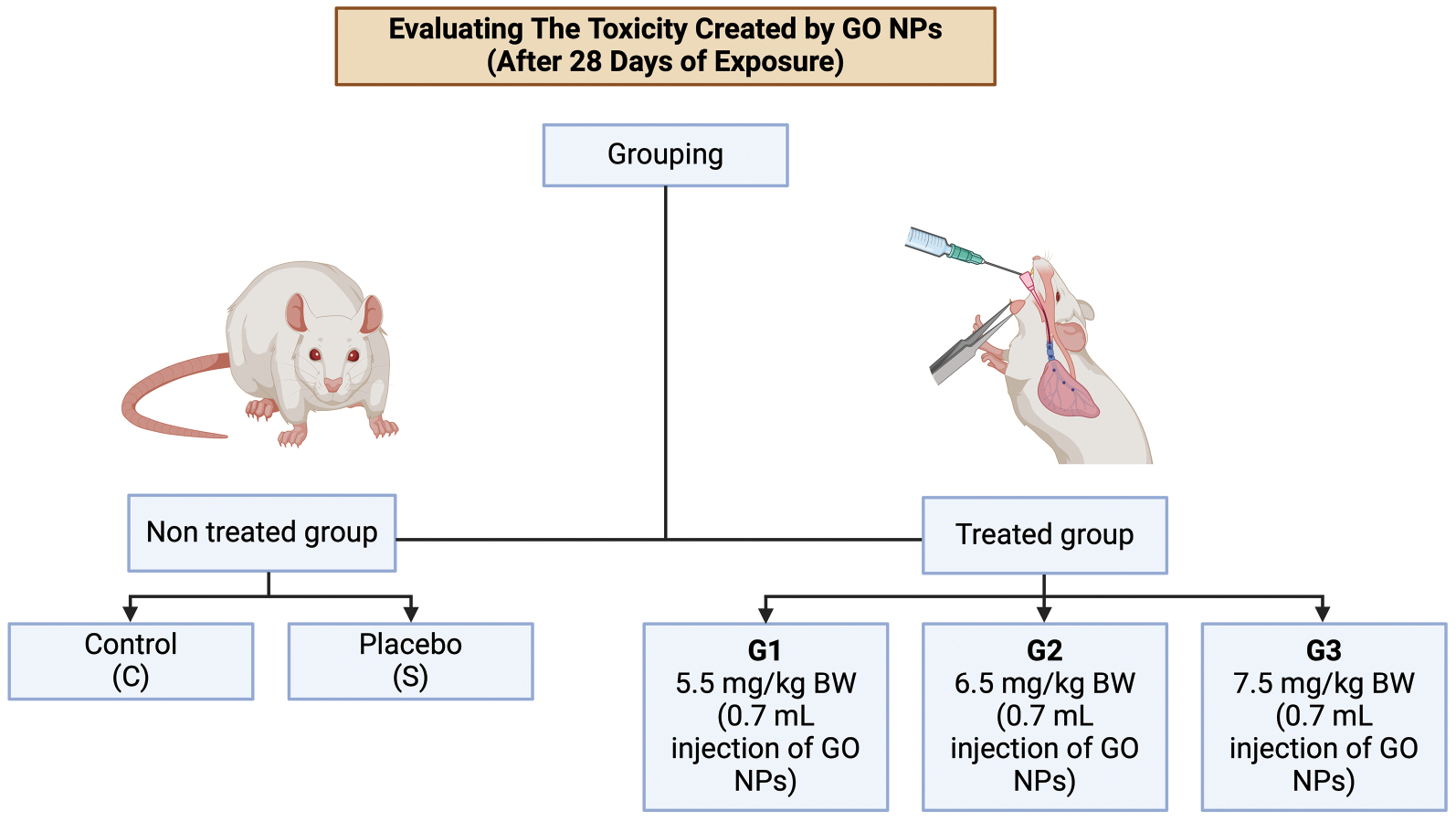
For assessment of toxicity, the rats were divided into five groups of five rats each (Table 1). The first group was the untreated control group. The second group received intraperitoneal injections of 1 ml normal saline (vehicle control). The remaining three groups, G1, G2, and G3, received intraperitoneal injections of GO nanoparticles (GOs) at doses of 5, 6, or 7 mg/kg body weight, respectively, every other day over a period of 28 days. Exposure to GO has been predicted to have cytotoxic effects [19–22]. The flowchart in Figure 2 shows the process of investigation of toxicity induced by GO sheets.
Figure 2 Flow chart of toxicity induced by GO sheets.
Table 1 Doses in Experimental Groups
| Group | Graphene oxide dose (mg/kg, intraperitoneal) |
|---|---|
| Control | 0.00 |
| Vehicle | 0.00 |
| G1 | 5.00 |
| G2 | 6.00 |
| G3 | 7.00 |
Blood sampling and sacrifice
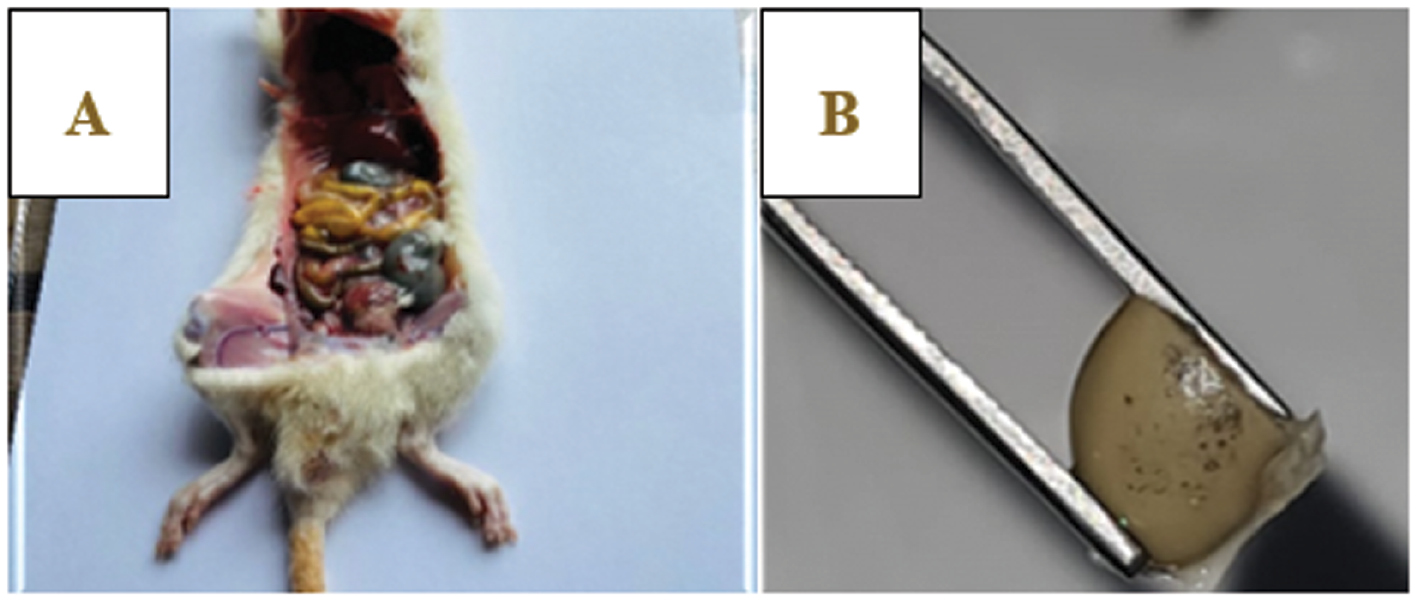
Blood samples were collected from all animals at the start of the trial and again after 28 days of treatment. These samples were used for various hematological analyses including CBC and RFT. On the 29th day, the rats were euthanized, and their kidneys were obtained for histological analysis with hematoxylin-eosin staining. The method of dissection for blood sampling is shown in Figure 3.
Figure 3 Dissection of albino rats: (A) rat dissection, (B) kidney view.
Sample collection
Blood samples for hematological examination were collected at the beginning of the study and after 28 days of exposure. On the 28th day, the animals were fasted overnight, and blood samples were collected from the caudal vein of individual rats into EDTA anticoagulant tubes. These samples were analyzed with a hematology autoanalyzer (Shanghaic Drawell Intelligent Instrument Co., Ltd, DW-TEK5000 robotized blood hematology analyzer) to assess hematological variables. The rats were then anesthetized with ketamine hydrochloride (30 mg/kg BW) and sacrificed. Their renal tissues were weighed with a Sartorius balance and fixed for histological processing [23, 24].
Hematological analysis of blood
Blood samples were collected from the animals’ hearts and transferred into two tubes containing calcium EDTA for subsequent CBC analysis. Hematology parameters, including RBC, HGB, WBC, LYM, MCH, MCHC, and PLT, were immediately analyzed with a hematology auto-analyzer.
Tissue preparation
For tissue fixation, small tissue portions were submerged in a solution of formaldehyde (30–35 ml), 100% alcohol (55–60 ml), and glacial acetic acid (10–15 ml). After fixation, the tissues were dehydrated with various grades of ethanol, then immersed in cedarwood oil until they became transparent. Subsequently, the tissues were embedded in benzene and paraplast, to form blocks for sectioning [25].
Hematoxylin and eosin staining
A hematoxylin stain was prepared by dissolving 2 g hematoxylin in 100 ml ethanol. An eosin stain was prepared by dissolving 1 g eosin in 100 ml 70% ethanol. The staining process involved hydration, deparaffinization, staining, and mounting.
Light microscopy
Tissue sections 5 μm thick were examined under a light microscope at 40× and 100× magnification. Slides from the control and treatment groups were thoroughly examined and documented.
Histological examination
After the animals were sacrificed, fresh kidney tissues were fixed, dehydrated, and embedded in benzene and paraplast. The tissues were cut into 5 μm sections with a microtome and mounted on clean albumenized glass slides. Hematoxylin and eosin staining were performed, and images were captured for histological examination [24, 26].
Statistical analysis
To assess the effects of GO sheets on various hematology parameters, we conducted statistical analysis (ANOVA with a linear model) in Minitab 17 software. A Tukey test was performed to compare the mean values of different groups, with a significance threshold set at p <0.05 [27].
Results
The study was conducted at the Exploration Research Center, Division of Zoology, Government College University Faisalabad, Punjab, Pakistan. The sublethal dose of GOs was determined on the basis of the histology, hematology, and mortality.
General health observations
Rats were carefully observed daily throughout the experiment. The rats in the control and GO sheet-treatment groups displayed good health and exhibited energetic behavior. Rats treated with a dose of 7 mg/kg body weight showed signs of irritability and diminished activity, but no mortality within 7 days. Rats treated with 6 mg/kg remained healthy and showed normal behavior. Weekly weight measurements over 4 weeks revealed significant weight loss in the treated rats by the end of the study. After 28 days, the control rats were thriving, whereas the GO sheet-treated rats exhibited symptoms such as dry skin, decreased coelomic fluid, shrunken body cavities, and organ fat depletion, thus indicating toxicity at a GO sheet dose of 7 mg/kg (Table 2).
Table 2 Descriptions of Experimental Treatments
| Group | Treatment |
|---|---|
| Control | No treatment |
| Vehicle | Normal saline (intraperitoneally injected) |
| G1 | GOS 5 mg/kg BW (intraperitoneally injected) |
| G2 | GOS 6 mg/kg BW (intraperitoneally injected) |
| G3 | GOS 7 mg/kg BW (intraperitoneally injected) |
Changes in body weight across groups
The animals in the vehicle and control groups behaved normally and typically gained weight during the study. During the first 2 weeks of exposure, no significant changes were observed in body weight in any groups (control and treatment) (p>0.05). However, during the third and fourth weeks of exposure, we observed significantly (p<0.05) lower body weight in the treatment groups receiving moderate (6 mg/kg BW) or high doses (7 mg/kg BW) than in the control, vehicle control, and low dose treatment groups (5 mg/kg BW) (Table 3).
Table 3 Weekly Body Weight (g) and Renal Somatic Index of Rats in Control and Treatment Groups (Mean±SE)
| Parameter | Group | ||||
|---|---|---|---|---|---|
| Control | G1 | G2 | G3 | Vehicle | |
| 0 week | 121.80±0.80a | 122.00±1.87a | 121.60±1.51a | 121.40±0.89a | 121.6±2.40a |
| 1st week | 132.40±1.10a | 133.40±1.14a | 132.40±1.14a | 133.40±1.14a | 132.2±1.00a |
| 2nd week | 143.40±0.80a | 143.20±1.30a | 143.00±1.58a | 143.00±1.87a | 142.8±1.00a |
| 3rd week | 153.60±1.10a | 150.40±0.54b | 146.20±0.83c | 141.80±2.28d ↓ | 153.4±1.10a |
| 4th week | 163.60±0.50a | 159.80±1.09b | 144.80±1.64c | 136.20±2.59d ↓ | 164.2±0.80a |
| R/S index | 1.73b | 1.73b | 1.77b | 2.33a | 2.34a |
a,b,c,dLevel of significant increase or decrease in body weight of treated and non-treated groups.
Exposure to GO caused dose-dependent toxicity to the kidney cells, including inflammation, and disruption of cellular processes. GO nanosheets were administered to the rats, and their effects on kidney function and histopathology were evaluated through social tests including open-field tests, maze tests, Morris water maze tests, and novel item acknowledgment tests (Table 4).
Table 4 Effects of GO Nanosheets on Hematological Parameters in Albino Rats
| Parameter | Group | ||||
|---|---|---|---|---|---|
| Control | G1 | G2 | G3 | Vehicle | |
| RBC | 6.71±0.15a | 5.64±0.11b | 4.27±0.05c | 3.2±0.24d ↓ | 6.67±0.21a |
| WBC | 13.36±0.43c | 13.80±0.51c | 17.53±0.37b | 22.25±0.56a ↑ | 13.82±0.62c |
| LYM% | 48.59±0.56d | 51.38±1.61c | 66.03±0.74b | 96.76±0.49a ↑ | 47.87±0.62d |
| MCH | 17.38±0.45cd | 17.99±0.38c | 20.75±0.61b | 24.60±0.40a ↑ | 17.05±0.48d |
| MCHC | 30.83±0.43d | 33.20±0.58c | 41.56±0.14b | 43.28±0.44a ↑ | 31.04±0.41d |
| MCV | 52.43±0.42d | 55.03±0.57c | 59.71±0.19b | 63.69±0.52a ↑ | 51.94±0.67d |
| HCV | 42.02±0.72d | 54.29±0.54c | 59.09±0.59b | 62.38±0.46a ↑ | 42.58±0.32d |
| PLT | 682.40±16.70a | 640.40±15.39b | 582.40±21.63c | 416.6±19.90d ↓ | 686.60±22.23a |
| GRA | 0.42±0.25a | 0.52±0.35a | 0.46±0.96a | 0.21±0.16a | 0.54±0.34a |
| GRA% | 3.90±1.15a | 4.72±0.22a | 5.16±0.83a | 5.45±0.41a | 4.41±0.81a |
| HGB | 13.68±0.20a | 12.79±0.16b | 6.62±0.38c | 4.75±0.24d ↓ | 13.55±0.13a |
| RDW% | 14.17±0.65a | 14.29±0.59a | 11.19±0.61b | 9.27±0.49c ↓ | 14.29±0.60a |
| MPV | 6.30±0.19b | 6.05±0.14b | 6.05±0.20b | 6.96±0.52a ↑ | 6.31±0.39b |
| PDW% | 43.91±0.77a | 31.26±0.99b | 19.98±0.58c | 6.52±0.23d ↓ | 44.87±3.24a |
| PCT | 0.24±0.01d | 0.39±0.01c | 0.59±0.02b | 0.97±0.03a ↑ | 0.25±0.01d |
| LPCR | 6.21±0.46b | 6.14±0.29b | 5.76±0.46b | 11.12±2.32a ↑ | 6.00±0.53b |
| MID | 0.54±0.22ab | 0.61±0.06ab | 0.63±0.05a | 0.35±0.19b | 0.63±0.04a |
| MID% | 8.49±1.54a | 7.24±.060a | 8.33±0.82a | 6.58±4.30a | 8.29±1.14a |
| Eosin | 3.82±0.20b | 1.80±0.44c | 1.80±0.44c | 6.00±0.70a ↑ | 1.80±0.44c |
| Mocyt | 0.90±0.37b | 2.60±0.54b | 4.10±1.81b | 8.40±0.54a ↑ | 2.60±0.54b |
| Nepils% | 69.60±6.11a | 32.60±4.39c | 43.00±8.69c | 27.60±1.67c ↓ | 32.60±4.39c |
a,b,c,dLevel of significant increase or decrease in body weight of treated and non-treated groups.
CRE, UA, and BUN were significantly lower (p>0.05) in the G2 and G3 groups (treated with moderate and high GO NP doses, respectively) than in the vehicle control group. The degree of urea and TBIL were fundamentally expanded in treated bunches in a portion subordinate way (p<0.05) (Table 5).
Table 5 Effects of GO Nanosheets on Hematological Parameters in Albino Rats
| Parameter | Group | ||||
|---|---|---|---|---|---|
| Control | G1 | G2 | G3 | Vehicle | |
| CRE | 1.22±0.07a | 1.07±0.03b | 0.85±0.03c | 0.37±0.04d | 1.20±0.07a |
| UA | 4.66±0.07a | 4.31±0.05b | 3.85±0.02c | 3.42±0.18d | 4.64±0.07a |
| Urea | 49.52±0.26c | 53.71±0.51b | 72.41±0.47a | 72.41±0.47a | 49.50±0.26c |
| TBIL | 0.46±0.05c | 0.60±0.10c | 1.90±0.09b | 2.54±0.30a ↓ | 0.44±0.05c |
| BUN | 21.42±0.45a | 15.86±0.66b | 9.64±0.18c | 6.89±0.60d ↓ | 21.40±0.45a |
a,b,c,dLevel of significant increase or decrease in body weight of treated and non-treated groups.
ALT, AST, LDH, and ALP were significantly higher (p<0.05) in the G2 and G3 treated groups than in the control, vehicle control, and low dose treatment groups (Table 6).
Table 6 Significantly Increased Parameters (ALT, AST, LDH, and ALP) after Exposure to GO Nanosheets (p<0.05)
| Parameter | Group | ||||
|---|---|---|---|---|---|
| Control | G1 | G2 | G3 | Vehicle | |
| ALT | 13.64±0.06d | 47.08±0.90c | 123.11±0.51b | 144.18±0.53a ↑ | 13.62±0.06d |
| AST | 40.63±9.30b | 48.90±0.66ab | 53.71±0.39a | 59.35±0.45a ↑ | 40.60±9.30b |
| ALP | 84.50±1.19d | 859.00±15.41c | 975.40±12.62b | 1172.20±17.90a ↑ | 85.52±0.72d |
| LDH | 1337.98±0.09d | 1551.15±7.92c | 2246.80±17.46b | 2769.20±24.00a ↑ | 1337.96±0.09d |
a,b,c,dLevel of significant increase or decrease in body weight of treated and non-treated groups.
Physiological changes
The body loads of the rodents were verified weekly for 28 days. Toward the end of the trial, the treated rodents in bunches 6, 7, and 8 exhibited a significant decrease in body weight. The decrease in body weight in the albino rats was associated with the kidney tissue histology. GO nanosheet treatment significantly increased ALT, ASP, ALP, and LDH, and significantly decreased CRE, UA, and BUN, thus leading to physiological changes in kidney structure.
Histopathological analysis
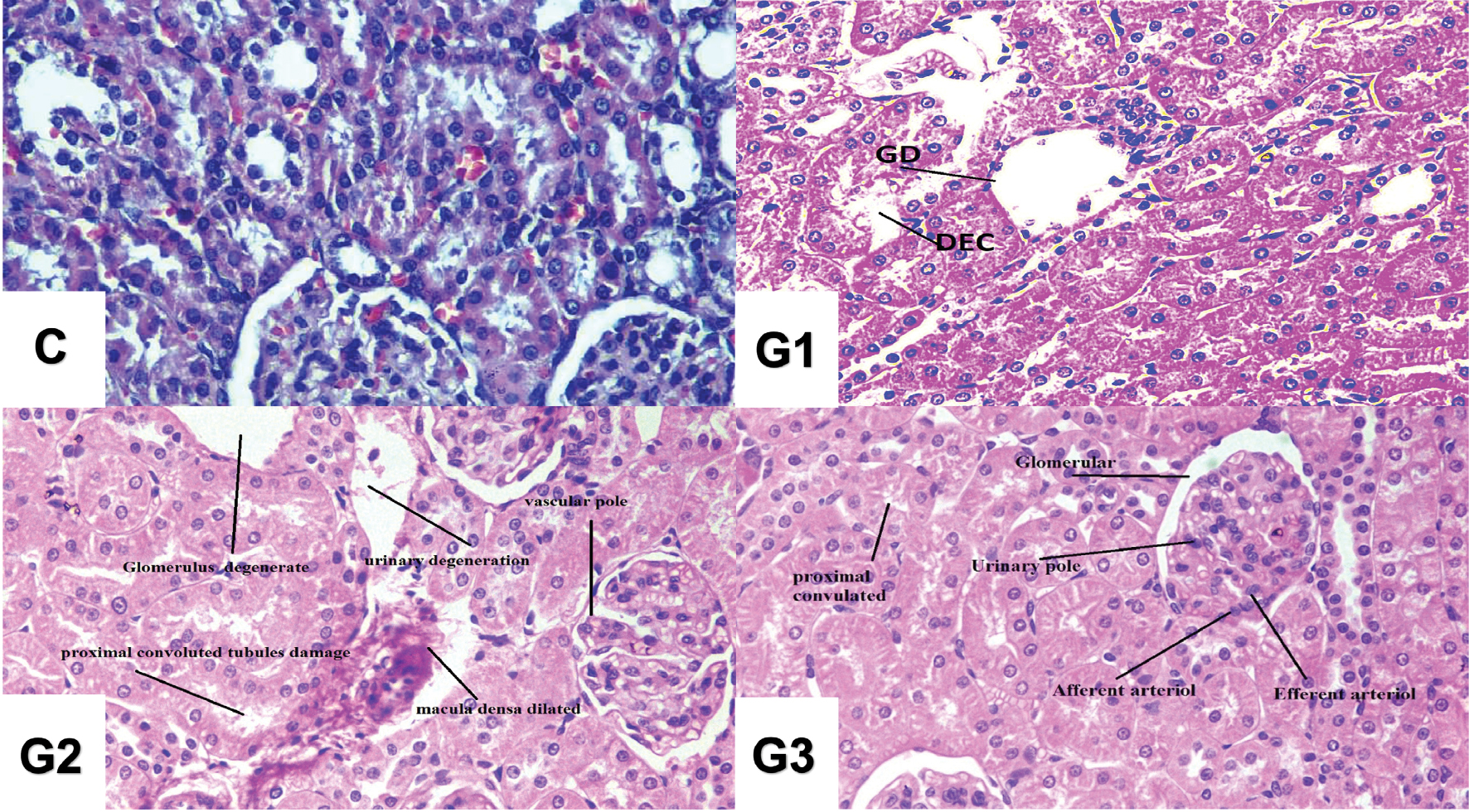
The histology analysis revealed histopathological changes in kidney tissue. Exposure of albino rats to GO sheets led to glomerulosclerosis, endothelial cell injury, and thickening of the glomerular basement membrane (Figure 4).
Figure 4 Histopathological analysis in albino rats (H&E, ×400 magnification). Control Group (C): No treatment; G1 Group (G1): GOS injection of 5 mg/kg BW (intraperitoneally injected); G2 Group (G2): GOS injection of 6 mg/kg BW (intraperitoneally injected); and G3 Group (G3): GOS injection of 7 mg/kg BW (intraperitoneally injected).
Discussion
GO sheets are economically important because of their widespread industrial use [28–30]. Assessment of their potential toxicity is crucial, because nanosheets can come into direct or indirect contact with the human body through various routes of exposure, such as cutaneous penetration, inhalation, oral consumption, or injection [31]. A recent study has investigated the toxicity of anatase GO Ns on histology and blood parameters in albino rats (liver function and CBC tests). For 28 days, rats were given GO N doses of 5 mg/kg, 6 mg/kg, or 7 mg/kg every other day. The rats in all groups showed no mortality but exhibited behavioral alterations. Sacrifice, appetite reduction, and violent behavior were observed in the G3 (7 mg/kg) and G2 (6 mg/kg) groups, but not in the G1 (5 mg/kg) group. Similar behavioral changes were observed by Gurunathan in 2012, in which animals treated with high doses of GO showed low appetites and diminished physical activity. Herein, we investigated the pathophysiological and physiological alterations caused by GO Ns in rats. For 28 days, rats were administered GO Ns at doses of 5 mg/kg, 6 mg/kg, and 7 mg/kg on alternating days. The rats in all groups died at the same rate. Although sacrifice, appetite reduction, and violent behavior were observed in the G2 (6 mg/kg) and G3 (7 mg/kg) groups, the G1 (5 mg/kg) group maintained normal behavior. Exposure of GO causes adverse impact on brain cells due to which they become unable to perform their activities properly [26, 32]. Physiological effects also occurred in the treated rodents, particularly in bunches 6, 7, and 8, which exhibited the body weight were significantly decrease the current result of research are not in accordance with [33, 34].
In liver function tests, a dose-dependent significant improvement in ALT, AST, and ALP enzymes was observed. Studies have shown that urea, creatinine, and uric acid levels increase in the presence of GO nanoparticles [35, 36]. GO Ns were administered to rats intravenously as a single injection (5 mg/kg BW) of GO N suspension in saline. The tissue content of GO Ns was measured after 1, 14, and 28 days. The kidneys showed a high GO nanosheet content. Liver function tests revealed dose-dependent increases in ALT, AST, and ALP enzymes—findings similar to those reported by Abbasi-Oshaghi et al. in 2019. The impacts of GO sheets on the kidney, GO sheet were found in renal cells, and glomeruli infection can produce pathological changes and nephron-like toxicity [37, 38]. Furthermore, as compared with the control group, 25 mg/kg GO N treatment markedly increases serum urea levels [39–42].
Renal toxicity induced by GO nanoparticles in albino rats can lead to various physiological changes within the kidneys and the overall organism; frequently observed physiological changes include renal function impairment, inflammation, tubular damage, distal tubular damage, histopathological changes, renal fibrosis, functional impairment, and other factors influencing renal toxicity. Research examining the effects of GO on kidney function impairment has indicated disruption of normal function [43], manifesting as changes in the glomerular filtration rate, impaired tubular reabsorption and secretion, and altered electrolyte and fluid balance [44–47]. Impaired kidney function may lead to disturbances in urine production and composition, and to inflammation in the body, particularly in the kidneys. The exposure of albino rats to GO can trigger an inflammatory response in the kidneys [48–51]. This response involves the release of pro-inflammatory cytokines and chemokines, recruitment of immune cells, and activation of inflammatory pathways. Sustained inflammation can exacerbate renal injury and contribute to further tissue damage [48, 51].
GO has been shown to accumulate in the renal tubules in albino rats, particularly the proximal tubules. This accumulation can result in tubular damage, including tubular dilation, vacuolization, and epithelial cell necrosis, thus leading to glomerular damage [52–54]. These changes can impair the filtration function of the glomeruli and lead to proteinuria, the presence of excessive protein in the urine [55]. Despite glomerular damage, GO exposure in the distal tubules can impair renal function, and contribute to electrolyte abnormalities and metabolic acidosis [56]. Renal fibrosis also occurred in albino rats after excessively long exposure to GO. Prolonged exposure to GO nanoparticles can lead to the development of renal fibrosis, characterized by aggregation of extracellular lattice components within renal tissue [57]. Renal fibrosis further impairs kidney function and contributes to the progression of renal damage [58, 59]. Furthermore, renal toxicity induced by GO leads to significant functional impairments in albino rats. The glomerular filtration rate is frequently affected, thus decreasing filtration efficiency [60–63]. Several factors influence the severity and extent of renal toxicity induced by GOs, including GO dose and concentration, exposure duration, administration route, and individual susceptibility. Higher doses and prolonged exposure to GOs tend to result in more severe renal damage [64, 65]. Exposure of albino rats to GO sheets resulted in observable kidney histopathological changes, observed through microscopic examination of kidney tissue. Glomerular damage is characterized by glomerulosclerosis, endothelial cell injury, and thickening of the glomerular basement membrane [66, 67].
Conclusion
GO sheets were observed to cause dose-dependent renal toxicity in albino rats. GOs decreased physical activity while increasing hematological and histological changes. The harmful effects of GOs on the blood and kidney tissues in male albino rats were investigated. Histological findings revealed necrosis and apoptosis in the GO sheet treatment groups, in contrast to the control and vehicle control groups. The observed histological alterations in the kidneys were caused by inflammation, tubular damage, renal fibrosis, and distal tubular damage. Our results indicated that GO sheet administration in rats had significant negative effects on blood parameters and kidney function.
Ethical approval
This study was approved by The University of Sialkot, Punjab, Pakistan.
Acknowledgments
We thank Jalan Tengah, Indonesia (www.jalantengah.site) for editing the manuscript.
Competing interests
All authors declare that there are no conflicts of interest in any capacity, including competing or financial.
Funding
This study received no external funding.
References
- Eigler S, Hirsch A. Chemistry with graphene and graphene oxide—challenges for synthetic chemists. Angew Chem Int Ed Engl 2014;53(30):7720-38. [PMID: 24962439 DOI: 10.1002/anie.201402780]
- Shalaby A, Nihtianova D, Markov P, Staneva AD, Iordanova RS, et al. Structural analysis of reduced graphene oxide by transmission electron microscopy. Bulg Chem Commun 2015;47(1):291-5.
- Shi J, Votruba AR, Farokhzad OC, Langer R. Nanotechnology in drug delivery and tissue engineering: from discovery to applications. Nano Lett 2010;10(9):3223-30. [PMID: 20726522 DOI: 10.1021/nl102184c]
- Chatterjee N, Kumar P, Kumar K, Misra SK. What makes carbon nanoparticles a potent material for biological application? Wiley Interdiscip Rev Nanomed Nanobiotechnol 2022;14(3):e1782. [PMID: 35194963 DOI: 10.1002/wnan.1782]
- Chen M, Yin J, Liang Y, Yuan S, Wang F. Oxidative stress and immunotoxicity induced by graphene oxide in zebrafish. Aquat Toxicol 2016;174:54-60. [DOI: 10.1016/j.aquatox.2016.02.015]
- Singh T, Shukla S, Kumar P, Wahla V, Bajpai VK. Application of nanotechnology in food science: perception and overview. Front Microbiol 2017;8:1501. [PMID: 28824605 DOI: 10.3389/fmicb.2017.01501]
- Strojny B, Sawosz E, Grodzik M, Jaworski S, Szczepaniak J, et al. Nanostructures of diamond, graphene oxide, and graphite inhibit CYP1A2, CYP2D6, and CYP3A4 enzymes and downregulate their genes in liver cells. Int J Nanomedicine 2018;13:8561. [PMID: 30587978 DOI: 10.2147/IJN.S188997]
- Coyne MJ, Schultze AE, McCrann DJ, Murphy RE, Cross J, et al. Evaluation of renal injury and function biomarkers, including symmetric dimethylarginine (SDMA), in the rat passive Heymann nephritis (PHN) model. PLoS One 2022;17(5):e0269085. [PMID: 35622875 DOI: 10.1371/journal.pone.0269085]
- Zahedi SM, Abolhassani M, Hadian-Deljou M, Feyzi H, Akbari A, et al. Proline-functionalized graphene oxide nanoparticles (GO-Pro NPs): a new engineered nanoparticle to ameliorate salinity stress on grape (Vitis vinifera L. cv Sultana). Plant Stress 2023;7:100128. [DOI: 10.1016/j.stress.2022.100128]
- Itoo AM, Vemula SL, Gupta MT, Giram MV, Kumar SA, et al. Multifunctional graphene oxide nanoparticles for drug delivery in cancer. J Control Release 2022;350:26-59. [PMID: 35964787 DOI: 10.1016/j.jconrel.2022.08.011]
- Jiang D, Ni D, Liu F, Zhang L, Liu L, et al. A fluorescent imaging assay of cast in renal disease based on graphene quantum dots and Fe3O4 nanoparticles. Clin Chim Acta 2016;454:94-101. [PMID: 26772724 DOI: 10.1016/j.cca.2016.01.001]
- Jiang ZY, Hunt JV, Wolff SP. Ferrous ion oxidation in the presence of xylenol orange for detection of lipid hydroperoxide in low density lipoprotein. Anal Biochem 1992;202(2):384-9. [PMID: 1519766 DOI: 10.1016/0003-2697(92)90122-n]
- Seifter JL, Chang HY. Disorders of acid-base balance: new perspectives. Kidney Dis (Basel) 2017;2(4):170-86. [PMID: 28232934 DOI: 10.1159/000453028]
- Patlolla AK, Randolph J, Kumari SA, Tchounwou PB. Toxicity evaluation of graphene oxide in kidneys of Sprague-Dawley rats. Int J Environ Res Public Health 2016;13(4):380. [PMID: 27043588 DOI: 10.3390/ijerph13040380]
- Acosta E. Bioavailability of nanoparticles in nutrient and nutraceutical delivery. Curr Opin Colloid Interface Sci 2009;14(1):3-15. [DOI: 10.1016/j.cocis.2008.01.002]
- Ahmad S, Tripathy DB, Mishra A. Sustainable nanomaterials. Sustainable Inorg Chem 2016;205. [DOI: 10.1002/9781119951438.eibc2430]
- Akbar K, Moretti E, Vomiero A. Carbon dots for photocatalytic degradation of aqueous pollutants: recent advancements. Adv Opt Mater 2021;9(17):2100532. [DOI: 10.1002/adom.202100532]
- Amrollahi-Sharifabadi M, Koohi MK, Zayerzadeh E, Hablolvarid MH, Hassan J, et al. In vivo toxicological evaluation of graphene oxide nanoplatelets for clinical application. Int J Nanomedicine 2018;13:4757. [PMID: 30174424 DOI: 10.2147/IJN.S168731]
- Hadizadeh N, Zeidi S, Khodabakhsh H, Zeidi S, Rezaei A, et al. An overview on the reproductive toxicity of graphene derivatives: highlighting the importance. Nanotechnol Rev 2022;11(1):1076-100. [DOI: 10.1515/ntrev-2022-0063]
- Hatane SE, Setiono FJ, Setiawan FF, Semuel H, Mangoting Y. Learning environment, students’ attitude and intention to enhance current knowledge in the context 79-97 of choosing accounting career. J Appl Res High Educ 2021;13(1):79-97. [DOI: 10.1108/JARHE-06-2019-0156]
- He C, Ruan F, Jiang S, Zeng J, Yin H, et al. Black phosphorus quantum dots cause nephrotoxicity in organoids, mice, and human cells. Small 2022;16(22):2001371. [PMID: 32338439 DOI: 10.1002/smll.202001371]
- Hola K, Markova Z, Zoppellaro G, Tucek J, Zboril R. Tailored functionalization of iron oxide nanoparticles for MRI, drug delivery, magnetic separation and immobilization of biosubstances. Biotechnol Adv 2015;33(6):1162-76. [PMID: 25689073 DOI: 10.1016/j.biotechadv.2015.02.003]
- Shakeel M, Jabeen F, Iqbal R, Chaudhry AS, Zafar S, et al. Assessment of titanium dioxide nanoparticles (TiO2 NPs) Induced hepatotoxicity and ameliorative effects of Cinnamomum cassia in Sprague-Dawley rats. Biol Trace Elem Res 2018;182(1):57-69. [PMID: 28631137 DOI: 10.1007/s12011-017-1074-3]
- Shakeel M, Jabeen F, Qureshi NA, Fakhr-E-Alam M. Toxic effects of titanium dioxide nanoparticles and titanium dioxide bulk salt in the liver and blood of male sprague-dawley rats assessed by different assays. Biol Trace Elem Res 2016;2(173):405-26. [PMID: 27008428 DOI: 10.1007/s12011-016-0677-4]
- Aebi H. Catalase. In: Packer L, editor. Methods in Enzymology, 105. Orlando, FL: Academic Press, 1984. pp. 121-6.
- Khanna P, Ong C, Bay BH, Baeg GH. Nanotoxicity: an interplay of oxidative stress, inflammation and cell death. Nanomaterials 2015;5(3):1163-80. [PMID: 28347058 DOI: 10.3390/nano5031163]
- Noureen A, Jabeen F, Tabish TA, Ali M, Iqbal R, et al. Histopathological changes and antioxidant responses in common carp (Cyprinus carpio) exposed to copper nanoparticles. Drug Chem Toxicol 2019;44(4):372-9. [PMID: 31088177 DOI: 10.1080/01480545.2019.1606233]
- Fu HB, Yao JN. Size effects on the optical properties of organic nanoparticles. J Am Chem Soc 2001;123(7):1434-9.[DOI: 10.1021/ja0026298]
- Pan S, Aksay IA. Factors controlling the size of graphene oxide sheets produced via the graphite oxide route. ACS Nano 2011;5(5):4073-83. [PMID: 21469697 DOI: 10.1021/nn200666r]
- Pratt EC. Multiplexed imaging using nanoparticles and rational isotope pairs. Weill Medical College of Cornell University; 2022.
- Yang L, Wei Y, Gao S, Wang Q, Chen J, et al. Effect of copper nanoparticles and ions on epididymis and spermatozoa viability of Chinese soft- shelled turtles Pelodiscus sinensis. Coatings 2022;12(2):110. [DOI: 10.3390/coatings12020110]
- Lin E, Fuda F, Luu HS, Cox AM, Fang F, et al. Digital pathology and artificial intelligence as the next chapter in diagnostic haematopathology. Semin Diagn Pathol 2023;40(2):88-94. [PMID: 36801182 DOI: 10.1053/j.semdp.2023.02.001]
- Rabiee N, Khatami M, Jamalipour Soufi G, Fatahi Y, Iravani S, et al. Diatoms with invaluable applications in nanotechnology, biotechnology, and biomedicine: recent advances. ACS Biomater Sci Eng 2021;7(7):3053-68. [PMID: 34152742 DOI: 10.1021/acsbiomaterials.1c00475]
- Raoof F, Munawar A, Ahmad M, Rizvi SFA, Ali Z, et al. Multifunctional iron oxide nanocarriers synthesis for drug delivery, diagnostic imaging, and biodistribution study. Appl Biochem Biotechnol 2023;1:1-16. [PMID: 36701093 DOI: 10.1007/s12010-023-04345-9]
- Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 1979;95(2):351-8. [PMID: 36810 DOI: 10.1016/0003-2697(79)90738-3]
- Singh B, Singh M, Kaur A, Kaur J, Prince . Asthma and chronic obstructive pulmonary disorder-phytomedicine. Int J Green Pharm 2017;11(3):S348-55. [DOI: 10.22377/IJGP.V11I03.1141]
- Bilisik K, Akter M. Polymer nanocomposites based on graphite nanoplatelets (GNPs): a review on thermal-electrical conductivity, mechanical and barrier properties. J Mater Sci 2022;57(15):7425-80. [DOI: 10.1007/s10853-022-07092-0]
- Schneider GF, Dekker C. DNA sequencing with nanopores. Nat Biotechnol 2012;30(4):326-8. [PMID: 22491281 DOI: 10.1038/nbt.2181]
- Lin H, Tan J, Wang J, Xie C, Chen B, et al. Subchronic oral toxicity study of food-related titanium dioxide nanoparticles in rats involved in Ti Biodistribution and Gut Microbiota. J Agric Food Chem 2023;71(3):1713-26. [PMID: 36634352 DOI: 10.1021/acs.jafc.2c05341]
- Lin Z, Monteiro-Riviere NA, Riviere JE. Pharmacokinetics of metallic nanoparticles. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2015;7(2):189-217. [PMID: 25316649 DOI: 10.1002/wnan.1304]
- Liu H, Hua M, Yang H. Magnetic resonance monitoring of focused ultrasound/magnetic nanoparticle targeting delivery of therapeutic agents to the brain. Proc Natl Acad Sci U S A 2010;107(34):15205-10. [PMID: 20696897 DOI: 10.1073/pnas.1003388107]
- Dkhil MA, Zrieq R, Al-Quraishy S, Abdel Moneim AE. Seleniumnanoparticles attenuate oxidative stress and testicular damage in streptozotocin-induced diabetic rats. Molecules 2016;21(11):1517. [PMID: 27869771 DOI: 10.3390/molecules21111517]
- Jasim DA, Murphy S, Newman L, Mironov A, Prestat E, et al. The effects of extensive glomerular filtration of thin graphene oxide sheets on kidney physiology. ACS Nano 2016;10(12):10753-67. [PMID: 27936585 DOI: 10.1021/acsnano.6b03358]
- Esmaeili Y, Yarjanli Z, Pakniya F, Bidram E, Łos MJ, et al. Targeting autophagy, oxidative stress, and ER stress for neurodegenerative disease treatment. J Controlled Release 2022;345:147-75. [PMID: 35248646 DOI: 10.1016/j.jconrel.2022.03.001]
- Fang Y, Lu Z, Li M, Qu J, Ye W, et al. An assessment of the reproductive toxicity of GONPs exposure to Bombyx mori. Ecotoxicol Environ Saf 2021;210:111888. [PMID: 33421719 DOI: 10.1016/j.ecoenv.2020.111888]
- Ghonimi WA, Alferah MA, Dahran N, El-Shetry ES. Hepatic and renal toxicity following the injection of copper oxide nanoparticles (CuO NPs) in mature male Westar rats: histochemical and caspase 3 immunohistochemical reactivities. Environ Sci Pollut Res Int 2022;29(54):81923-37. [PMID: 35739448 DOI: 10.1007/s11356-022-21521-2]
- Gupta P. Introduction and historical background. In: Nanotoxicology in Nanobiomedicine. Springer; 2023. Vol. 1. pp. 1-22. [DOI: 10.1007/978-3-031-24287-8_1]
- Rhazouani A, Gamrani H, El Achaby M, Aziz K, Gebrati L, et al. Synthesis and toxicity of graphene oxide nanoparticles: a literature review of in vitro and in vivo studies. Biomed Res Int 2021;10(2021):5518999. [PMID: 34222470 DOI: 10.1155/2021/5518999]
- Sarkar SK, Raul KK, Pradhan SS, Basu S, Nayak A. Magnetic properties of graphite oxide and reduced graphene oxide. Physica E Low Dimens Syst Nanostruct 2014;64:78-82. [DOI: 10.1016/j.physe.2014.07.014]
- Sasidharan A, Swaroop S, Koduri CK, Girish CM, Chandran P, et al. Comparative in vivo toxicity, organ biodistribution and immune response of pristine, carboxylated and PEGylated few-layer graphene sheets in Swiss albino mice: a three month study. Carbon 2015;95:511-24. [DOI: 10.1016/j.carbon.2015.08.074]
- Arora P, Sindhu A, Dilbaghi N, Chaudhury A, Rajakumar G, et al. Nano-regenerative medicine towards the clinical outcome of stem cell and tissue engineering in humans. J Cell Mol Med 2012;16(9):1991-2000. [PMID: 22260258 DOI: 10.1111/j.1582-4934.2012.01534.x]
- Banik S, Rahman MM, Sikder MT, Saito T, Kurasaki M. Protective effects of ajwain (Trachyspermum ammi L.) extract against cadmium-induced cytotoxicity and apoptosis in PC12 cells. J Herb Med 2021;26:100423. [DOI: 10.1016/j.hermed.2021.100423]
- Bechtold B, Clarke J. Multi-factorial pharmacokinetic interactions: unraveling complexities in precision drug therapy. Expert Opin Drug Metab Toxicol 2021;17(4):397-412. [PMID: 33339463 DOI: 10.1080/17425255.2021.1867105]
- Naz S, Gul A, Zia M. Toxicity of copper oxide nanoparticles: a review study. IET Nanobiotechnol 2020;14(1):1-13. [PMID: 31935671 DOI: 10.1049/iet-nbt.2019.0176]
- Shorter JA. Vernon harcourt: a founder of chemical kinetics and a friend of “Lewis Carroll”. J Chem Educ 1980;57(6):411. [DOI: 10.1021/ed057p411]
- Dumala N, Mangalampalli B, Kalyan KSS, Grover P. Biochemical alterations induced by nickel oxide nanoparticles in female Wistar albino rats after acute oral exposure. Biomarkers 2018;23(1):33-43. [PMID: 28748734 DOI: 10.1080/1354750X.2017.1360943]
- Araújo MJ, Sousa ML, Fonseca E, Felpeto AB, Martins JC, et al. Proteomics reveals multiple effects of titanium dioxide and silver nanoparticles in the metabolism of turbot, Scophthalmus maximus. Chemosphere 2022;308:136110. [PMID: 36007739 DOI: 10.1016/j.chemosphere.2022.136110]
- Huang KT, Wu CT, Huang KH, Lin WC, Chen CM, et al. Titanium nanoparticle inhalation induces renal fibrosis in mice via an oxidative stress upregulated transforming growth factor-β pathway. Chem Res Toxicol 2015;28(3):354-64. [PMID: 25406100 DOI: 10.1021/tx500287f]
- Ma R, Wang Y, Qi H, Shi C, Wei G, et al. Nanocomposite sponges of sodium alginate/graphene oxide/polyvinyl alcohol as potential wound dressing: in vitro and in vivo evaluation. Compos B Eng 2019;167:396-405. [DOI: 10.1016/j.compositesb.2019.03.006]
- Ma Y, Cai F, Li Y, Chen J, Han F, et al. A review of the application of nanoparticles in the diagnosis and treatment of chronic kidney disease. Bioact Mater 2020;5(3):732-43. [PMID: 32596555 DOI: 10.1016/j.bioactmat.2020.05.002]
- Makhdoumi P, Karimi H, Khazaei M. Review on metal-based nanoparticles: role of reactive oxygen species in renal toxicity. Chem Res Toxicol 2022;33(10):2503-14. [PMID: 32909744 DOI: 10.1021/acs.chemrestox.9b00438]
- Milosavljevic V, Mitrevska K, Adam V. Benefits of oxidation and size reduction of graphene/graphene oxide nanoparticles in biosensing application: classification of graphene/graphene oxide nanoparticles. Sensor Actuat B-Chem 2022;353:131122. [DOI: 10.1016/j.snb.2021.131122]
- Morais MGD, Martins VG, Steffens D, Pranke P, da Costa JA, et al. Biological applications of nanobiotechnology. J Nanosci Nanotechnol 2014;14(1):1007-17. [DOI: 10.1166/jnn.2014.8748]
- Krihariyani D, Haryanto E, Sasongkowati R. In silico analysis of antiviral activity and pharmacokinetic prediction of brazilein sappan wood (Caesalpinia sappan L.) against SARS-CoV-2 spike glycoproteins. Indones J Med Lab Sci Technol 2021;3(1):26-37. [DOI: 10.33086/ijmlst.v3i1.1854]
- Cholewińska E, Juśkiewicz J, Majewski M, Smagieł R, Listos P, et al. Effect of copper nanoparticles in the diet of WKY and SHR rats on the redox profile and histology of the heart, liver, kidney, and small intestine. Antioxidants 2022;11(5):910. [PMID: 35624774 DOI: 10.3390/antiox11050910]
- Clause BT. The wistar rat as a right choice: establishing mammalian standards and the ideal of a standardized mammal. J Hist Biol 1993;26(2):329-49. [PMID: 11623164 DOI: 10.1007/BF01061973]
- de Luna LAV, Loret T, Fordham A, Arshad A, Drummond M, et al. Lung recovery from DNA damage induced by graphene oxide is dependent on size, dose and inflammation profile. Part Fibre Toxicol 2022;19(1):1-21. [PMID: 36131347 DOI: 10.1186/s12989-022-00502-w]