Establishment of Breast Phyllodes Tumor Cell Lines Preserving the Features of Phyllodes Tumors
1Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation, Guangdong-Hong Kong Joint Laboratory for RNA Medicine, Medical Research Center, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
2Breast Tumor Center, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
3Department of Ultrasound, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
4Department of Breast Surgery, Zhujiang Hospital, Southern Medical University, Guangzhou 510282, China
5Department of Anesthesiology, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
6Department of Pathology, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
aShishi He and Xiaoyun Xiao contributed equally to this article.
*Correspondence to: Yan Nie, E-mail: nieyan7@mail.sysu.edu.cn; Phei Er Saw, E-mail: caipeie@mail.sysu.edu.cn
Received: September 30 2022; Revised: October 19 2022; Accepted: October 24 2022; Published Online: November 2 2022
Cite this paper:
He S, Xiao X, Lei R et al. Establishment of Breast Phyllodes Tumor Cell Lines Preserving the Features of Phyllodes Tumors. BIO Integration 2023; 4(1): 7–17.
DOI: 10.15212/bioi-2022-0025. Available at: https://bio-integration.org/
Download citation
© 2023 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Breast phyllodes tumors (PTs) are biphasic, with epithelial and stromal components. Although the PT incidence is low (approximately 1% of all breast tumors), its clinical outcomes are unpredictable, and malignant PTs often progress rapidly. No effective treatment is currently available, thus resulting a high mortality rate from malignant PTs. PT cell lines must be established to facilitate the study of PTs. Herein, we established six PT cell lines through continuous passage or cell immortalization. We characterized these PT cell lines through in vitro functional assays, malignant PT marker detection and short tandem repeat identification. Benign PT cell lines (SYSH-BPT-01 and SYSH-BPT-02) were transfected with human papillomavirus 16 E6/E7, and two malignant PT cell lines (SYSH-MPT-01 and SYSH-MPT-02) were transfected with Simian virus 40 large T antigen. Two malignant PT cell lines (SYSH-MPT-03 and SYSH-MPT-04) were established through continuous passage. All malignant PT cell lines showed greater proliferation, colony formation, migration, invasion and collagen contraction ability than the benign PT cell lines. Moreover, the expression levels of malignant PT markers (α-smooth muscle actin and fibroblast activation protein) and short tandem repeat identification indicated that each PT cell line was identical to the parental primary cells. We successfully established PT cell lines that preserved the features of primary cells. These cell lines may serve as ideal experimental models for studying the function of breast PTs, thus opening new possibilities for PT drug screening and therapeutic target validation.
Keywords
Phyllodes tumor, benign, malignant, cell line, immortalization
Introduction
Breast phyllodes tumors (PTs) are fibroepithelial tumors accounting for approximately 1% of all breast tumors [1]. The World Health Organization classifies PTs into benign, borderline or malignant, on the basis of stromal cellularity, stromal atypia, stromal overgrowth, mitotic activity and tumor-border characteristics [2]. The clinical outcomes of PTs are difficult to predict, local relapse is frequent, and distant metastasis may occur. The current approach to prevent recurrence and metastasis is surgical resection with a wide margin, given that adjuvant chemotherapy or radiotherapy is ineffective against PTs [2, 3]. However, even with wide surgical resection, PTs remain prone to recurrence. The recurrence rates of benign, borderline and malignant PTs have been reported to be 3.4%–20.8%, 14.1%–41.7% and 42.3%–65%, respectively [4–6]. Furthermore, recurrent PTs progress toward more malignant phenotypes [7]. Of note, patients with malignant PTs may have distant metastasis and may die within short time periods [8–10]. The metastasis rate is 21%–43.1% [11, 12], and the 5-year overall survival is 61%–66.1% for patients with malignant PTs [13, 14]. In our clinical experience, some patients with malignant PTs experience frequent recurrence and distant metastasis even after receiving radiotherapy and chemotherapy. The shortest interval of recurrence that we have observed was only 3 weeks, thus placing great psychological and economic pressure on patients [15]. However, owing to the low incidence of PTs, few studies have focused on PT pathological mechanisms and therapeutic targets. Moreover, a lack of PT cell lines further limits research on PTs. Our previous research on the malignant progression of PTs [12, 16, 17] has relied on PT primary cells (PTPCs), whose acquisition and culture are time consuming and difficult. Therefore, we sought to establish PT cell lines.
Cell lines generally refer to cells that can be successively passaged in vitro. Cell lines that cannot be passaged continuously are called finite cell lines, whereas those that can be passaged continuously and proliferate infinitely are called continuous cell lines [18]. Cell immortalization is a method to obtain continuous cell lines [19, 20]. Human papillomavirus 16 E6/E7 (HPV-16 E6/E7) and simian virus 40 large T antigen (SV40-T) are commonly used viral genes for immortalization [21–23]. HPV-16 E6/E7 has successfully immortalized preadipocytes [24], keratinized epithelium [25] and mesenchymal stem cells [26]. SV40-T has successfully immortalized mesenchymal cells [27], fibroblasts [28] and endothelial cells [29]. The growth of benign PTPCs usually decreases after ten passages; among malignant PTPCs, half can be infinitely passaged (to more than 50 passages), whereas the other half shows a decline in proliferation rate after 15 passages [16]. Therefore, transfecting benign or malignant PTPCs with immortalized virus genes may help establish immortalized cell lines.
In this study, we successfully established two benign PT cell lines (SYSH-BPT-01 and SYSH-BPT-02) and four malignant PT cell lines (SYSH-MPT-01, SYSH-MPT-02, SYSH-MPT-03 and SYSH-MPT-04) through continuous passage or cell immortalization. We verified the functions and features of these PT cell lines through conducting proliferation, colony formation, migration, invasion and collagen contraction assays; detecting the expression levels of α-smooth muscle actin (α-SMA) and fibroblast activation protein (FAP); and performing short tandem repeat (STR) identification. PT cell lines stably preserved the features of parental PTPCs and thus may serve as ideal models for studying the biological functions of PTs in vitro. These cell lines may enable consistent and repeatable experimental results to be obtained in future PT research.
Materials and methods
Patients and tissue samples
Breast PT tissues were obtained from two patients with benign PT and four patients with malignant PT treated at the Breast Tumor Center, Sun Yat-sen Memorial Hospital, Sun Yat-sen University (Guangzhou, China). Informed consent was obtained from all patients.
Isolation and culture of PTPCs
PTPCs were isolated from fresh breast PT tissues under aseptic conditions. Adipose tissues and necrotic tissues were removed, and the tissues were minced into a paste comprising 1–2 mm pieces. The minced tissues were transferred to a 50 mL centrifuge tube with 1 mg/mL collagenase III (LS004182, Worthington) and digested at 37°C for 1.5–2 h. The cell suspension was filtered through a 70 μm cell strainer, and the filtrate was collected and centrifuged at 2000 rcf for 10 min to obtain cell precipitate. PTPCs were cultured in DMEM/F12 (C11330500BT, GIBCO) containing 15% fetal bovine serum (10270106, GIBCO), 20 ng/mL epidermal growth factor (AF-100-15, PeproTech), 0.5 mg/mL hydrocortisone (B1951, APExBio) and 10 μg/mL insulin (I-1882, Sigma). The culture medium was routinely replaced every 2–3 days. All PTPCs were tested for mycoplasma, and the results were negative.
Proliferation and colony formation assays
In cell proliferation assays, 2000 cells were seeded into 96-well plates. CCK8 reagent (K1018, APExBio) was added and then incubated at 37°C for 2 h, and the OD values were detected at 450 nm. In colony formation assays, 100 cells were seeded into six-well plates. After 10-day culture, the cells were fixed with 4% paraformaldehyde and stained with crystal violet, and the colonies were counted.
Cell migration and invasion assays
For migration assays, 2×104 cells were seeded into Transwell inserts (upper chambers) (353097, Corning) containing serum-free culture medium, whereas the lower chambers contained complete medium. After 24 h culture, the upper chambers were fixed with 4% paraformaldehyde and stained with crystal violet. For invasion assays, the upper chambers were first coated with 20% Matrigel (356234, BD), and the remaining steps were as described above. Migrated and invasive cells were counted in five random fields (200×).
Collagen contraction assays
Cells were suspended in acid-soluble collagen type I (Corning, USA) solution. The collagen/cell mixture was seeded into 24-well plates coated with bovine serum albumin, and the mixture was allowed to polymerize at 37°C for 30 min. Immediately after polymerization, 1 mL of culture medium was added to each plate. After 36 h, the collagen gels were imaged, and the contraction length of the gels was measured as the distance from the collagen gel edge to the edge of the culture plate. Mean values were calculated from four distances measured for each collagen gel.
Cell morphology
Cells were cultured to 60–90% confluence, fixed with 4% paraformaldehyde and stained with crystal violet. Cells were imaged under 200× magnification with an upright microscope (Nikon Ni-U).
Establishment of PT cell lines
PTPCs were continuously cultured in vitro for more than 50 passages. Cells maintaining normal morphology and rapid proliferation rate were considered established. The immortalization of breast PT cell lines was conducted by Applied Biological Materials Inc. (Canada). PTPCs were infected with Lenti-HPV-16 E6/E7 lentivirus (LV617, abm) or Lenti-SV40-T lentivirus (LV613, abm). After 24 h, the culture medium was replaced with fresh culture medium containing puromycin. Cells were cultured in vitro for more than 50 passages, and the expression of HPV-16 E6/E7 or SV40-T was detected. The cells that successfully expressed HPV-16 E6/E7 or SV40-T were considered established.
RNA extraction and quantitative real-time PCR
Total RNA was extracted with an RNA Quick Purification kit (RN001, ESscience). Reverse transcription was performed with HiScript®II Q RT SuperMix (R223-01, Vazyme). Quantitative real-time PCR (qRT-PCR) assays were performed with ChamQ SYBR® qPCR Master Mix (Q311-02, Vazyme). The primer sequences are displayed in Table S1.
Immunofluorescence assays
Cells were fixed with 4% paraformaldehyde and permeabilized with 0.1% Triton X-100. HPV-16 E7 (sc-6981, Santa Cruz Biotechnology) and α-SMA (MAB1420, R&D Systems) were stained with the corresponding antibodies at 1:50 dilution, then incubated with Alexa Fluor® 488 goat anti-mouse secondary antibody (ZF-0512, ZSGB-BIO) at 1:50 dilution. SV40-T (ab234426, Abcam) and FAP (ab28244, Abcam) were stained with the corresponding antibodies at 1:50 dilution, then incubated with Alexa Fluor® 594 goat anti-rabbit secondary antibody (ZF-0516, ZSGB-BIO) at 1:50 dilution. Cells were imaged under 400× magnification with a confocal laser scanning microscope (Zeiss LSM 710).
STR identification
PT cell lines and parental PTPCs were digested with trypsin (12604021, Gibco) and collected in centrifuge tubes. STR identification was performed by Guangzhou IGE Biotechnology Co. Ltd. (Guangdong, China).
Statistical analysis
Statistical analysis was performed in GraphPad Prism 6 software (GraphPad Software Inc., USA). Statistical differences were evaluated with unpaired two-tailed Student’s t-test. The values are reported as mean ± SEM. Statistical significance was defined as *P < 0.05, **P < 0.01 or ***P < 0.001.
Results
Phyllodes tumor primary cells have similar biological characteristics to those of the corresponding tumors
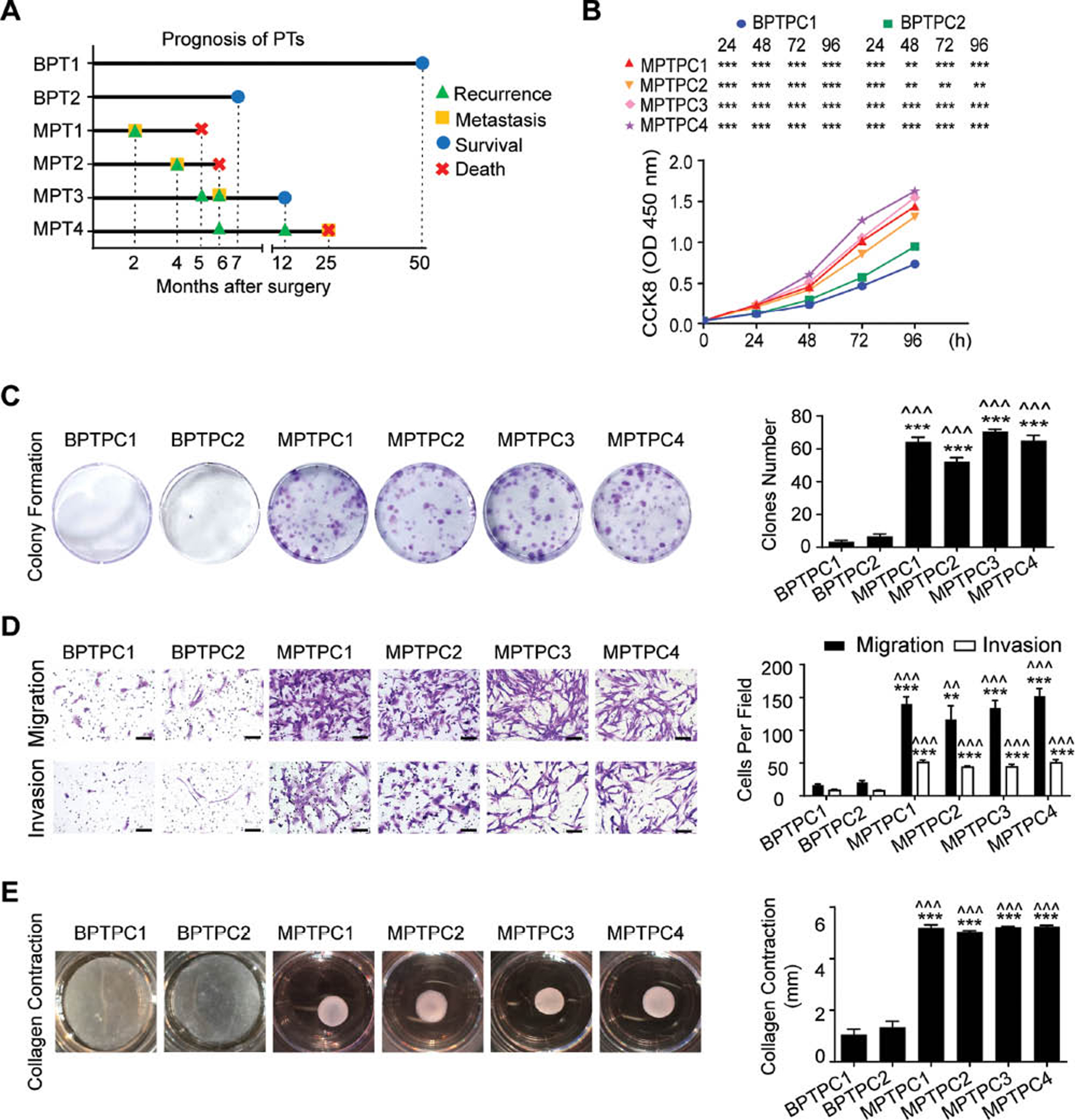
To obtain PT cell lines, we first successfully isolated six PTPCs from PT tissues through collagenase III digestion according to a previous protocol [16], including two benign PTPCs (BPTPC1 and BPTPC2) and four malignant PTPCs (MPTPC1, MPTPC2, MPTPC3 and MPTPC4). BPTPC1 was derived from a 4 cm×2.5 cm×2 cm benign PT, and BPTPC2 was derived from a 3 cm×2 cm×2 cm benign PT. Both benign PT donors (BPT1 and BPT2) had no tumor recurrence after surgery. In contrast, malignant PTPCs were derived from larger and more aggressive malignant PTs. MPTPC1 was derived from a 20 cm×15 cm×7 cm malignant PT that progressed rapidly. The donor (patient MPT1) of MPTPC1 experienced recurrence and distant metastasis to abdominal wall 2 months after surgery. Although patient MPT1 received chemotherapy immediately after surgery, she died 5 months after surgery. MPTPC2 was derived from a 11.3 cm×8.7 cm×7.9 cm malignant PT. The donor (patient MPT2) experienced recurrence and distant metastasis to the lungs, abdominal wall and pancreas 4 months after surgery, and died within 6 months after surgery. MPTPC3 was derived from a 10 cm×10 cm×7.2 cm malignant PT. The donor (patient MPT3) experienced tumor recurrence twice, with a recurrence interval of only 1 month, and distant metastasis to the lungs 7 months after surgery. MPTPC4 was obtained from a 9 cm×8.2 cm×6.1 cm malignant PT. The corresponding donor (patient MPT4) experienced tumor recurrence twice, and the recurrence interval was 6 months (Table 1 and Figure 1A). Clinicopathological information of the PT tissues from donors showed that the two benign PTs displayed typical benign PT features, including well-defined tumor borders, inactive mitotic activity, low stromal atypia and good prognosis. In contrast, the four malignant PTs displayed typical malignant PT features, including a permeative tumor border, active mitotic activity, high stromal atypia and poor prognosis (Table 1).
Table 1 Clinical Information on the Breast Phyllodes Tumors
| Patients | BPT1 | BPT2 | MPT1 | MPT2 | MPT3 | MPT4 |
|---|---|---|---|---|---|---|
| Age (years) | 46 | 23 | 47 | 54 | 25 | 51 |
| Tumor size (cm) | 4×2.5×2 | 3×2×2 | 20×15×7 | 11.3×8.7×7.9 | 10×10×7.2 | 9×8.2×6.1 |
| Tumor bordera | Well defined | Well defined | Permeative | Permeative | Permeative | Permeative |
| Mitotic activity (per 10 HPF)b | 2 | – | 12 | 22 | 35 | 17 |
| Stromal cellularityc | Mild | Mild | Marked | Marked | Marked | Marked |
| Stromal atypiad | Mild | Mild | Moderate | Marked | Marked | Marked |
| Stromal overgrowthe | Absent | Absent | Present | Present | Present | Present |
| Recurrence | No | No | Yes | Yes | Yes | Yes |
| Metastasis | No | No | Yes | Yes | Yes | No |
| Death | No | No | Yes | Yes | No | No |
a. Tumor border: The pattern of growth at the edge of the phyllodes tumors. Well defined: Tumor penetrating into the surrounding tissue, and the boundary between tumor and normal tissue was clear. Permeative: Tumor showing invasive growth, and an unclear boundary between tumor and normal tissue. b. Mitotic activity: Quantified per 10 high-power field (HPF, 400×). c. Stromal cellularity: Evaluated in the most cellular areas. Mild: Nuclei evenly spaced without overlapping. Marked: Nuclei touching or overlapping. d. Stromal atypia: Differences in tumor nuclear morphology. Mild: Small and uniform nuclei with inconspicuous nucleoli. Marked: Clearly differing shape and size of nuclei, with irregular nuclear membranes and clear nucleoli. Moderate: Intermediate between mildly and marked. e. Stromal overgrowth: Stromal proliferation without accompanying epithelial elements in at least one low-power field (40×). Absent: Epithelial component seen in a low-power field. Present: Tumor lacking epithelial components in a low-power field [1].
Figure 1 Phyllodes tumor primary cells have similar biological characteristics to those of the corresponding tumors. A Schematic diagram of PT patients’ prognosis. BPT1, BPT2, MPT1, MPT2, MPT3 and MPT4 are donors of BPTPC1, BPTPC2, MPTPC1, MPTPC2, MPTPC3 and MPTPC4, respectively. B CCK8 cell proliferation assay showed the proliferation rate of PTPCs. C (Left) Images of colony formation assay of PTPCs. (Right) Bar chart showed the clones number of each group. D (Left) Images of migration and invasion assay of PTPCs. Scale bar, 100 µm. (Right) Bar chart showed the number of migration or invasion cells under 200 × field of each group. E Images of collagen contraction assay of PTPCs. (Right) Bar chart showed the contraction length of each group. Data in (B – E) were shown as mean ± SEM, n=3. (* displayed the significance between BPTPC1 and MPTPCs; ^ displayed the significance between BPTPC2 and MPTPCs. *, P < 0.05; **, P < 0.01; ***, P < 0.001).
Next, we tested the biological functions of these PTPCs. Proliferation assays indicated that malignant PTPCs proliferated significantly faster than benign PTPCs (Figure 1B). Colony formation assays demonstrated that all malignant PTPCs formed more and larger clones than benign PTPCs (Figure 1C). Migration and invasion assays revealed that malignant PTPCs migrated and invaded more cells than benign PTPCs within the same time period (Figure 1D). Moreover, our previous studies have indicated that myofibroblasts are the major malignant component of PTs, and the proportion of myofibroblasts increases with the malignant progression of PT [16]. Because myofibroblasts are known to markedly induce collagen gel contraction [30], we conducted collagen contraction assays to test whether these PTPCs had myofibroblast function. All malignant PTPCs contracted collagen gels to a much greater extent than benign PTPCs (Figure 1E). Together, these results indicated that the biological behaviors of PTPCs were similar to those of the parental PTs and could feasibly be used to establish PT cell lines.
Establishment of breast phyllodes tumor cell lines by gene transfection or continuous passage
Cell immortalization
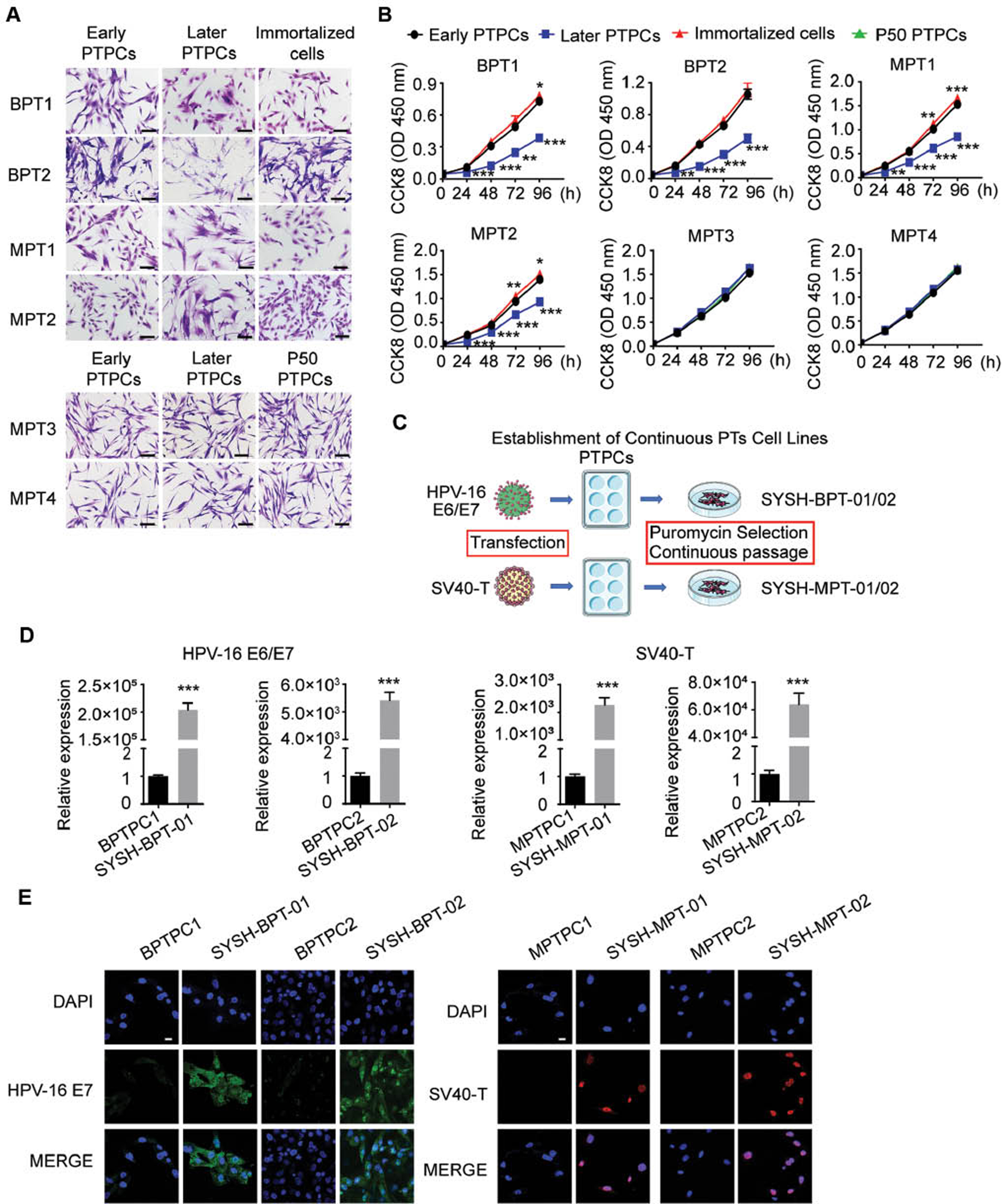
To obtain PT cell lines to examine the pathological mechanism and potential therapeutic target of PTs viably, we routinely passaged the PTPCs in vitro. However, cultured BPTPC1, BPTPC2, MPTPC1 and MPTPC2 showed altered cell morphology, and cells grew slowly after 15 passages. A comparison of the cell morphology of PTPCs that were cultured to passages 3–5 (early PTPCs) and those were cultured to passages 10–15 (later PTPCs) indicated that late-passage BPTPC1, BPTPC2, MPTPC1 and MPTPC2 underwent a phenotypic change from spindle-shaped cells toward hypertrophic, polygonal and irregular forms (Figure 2A). Moreover, cell proliferation assays indicated that the late-passage BPTPC1, BPTPC2, MPTPC1 and MPTPC2 showed a lower proliferation rate than early-passage cells (Figure 2B). These results indicated that BPTPC1, BPTPC2, MPTPC1 and MPTPC2 could not be cultured to more than 15 passages and therefore could be defined as finite cell lines.
Figure 2 Establishment of breast phyllodes tumor cell lines by gene transfection or continuous passage. A The cell morphology of PT cells under light microscope. Scale bar, 100 µm. (Early PTPCs: PTPCs that were cultured to passages 3–5; Later PTPCs: PTPCs that were cultured to passages 10–15; Immortalized cells: BPTPC1, BPTPC2, MPTPC1 and MPTPC2 that were transfected with immortalization gene; P50 PTPCs: MPTPC3 and MPTPC4 that were cultured to more than 50 passages.) B CCK8 cell proliferation assay showed the proliferation rate of PT cells. The early PTPCs served as a control. C Graphical illustration of the continuous PT cell lines establishment through cell immortalization. D The expression level of HPV-16 E6/E7 or SV40-T mRNA was determined by qRT-PCR in PTPCs (control) and parental PT cell lines. RNA expression normalized to GAPDH. E Immunofluorescence assay showed the expression of HPV-16 E7 (Green) in BPTPC1, BPTPC2, SYSH-BPT-01, SYSH-BPT-02, and SV40-T (Red) in MPTPC1, MPTPC2, SYSH-MPT-01, SYSH-MPT-02. Scale bar, 20 µm. Data in (B, D) were shown as mean ± SEM, n=3. (*, P < 0.05; **, P < 0.01; ***, P < 0.001).
To successfully immortalize these finite cell lines, we transduced HPV-16 E6/E7 or SV40-T genes into BPTPC1, BPTPC2, MPTPC1 and MPTPC2 through lentiviral infection, then added puromycin to the culture medium to select successfully transfected cells (Figure 2C). We then routinely passaged these selected PTPCs in vitro. BPTPC1 and BPTPC2 transfected with HPV-16 E6/E7, and MPTPC1 and MPTPC2 transfected with SV40-T, were successfully cultured to more than 50 passages in vitro. Because the expression of immortalization genes was essential for cell immortalization, we compared the expression levels of HPV-16 E6/E7 or SV40-T in paired transfected and non-transfected BPTPC1, BPTPC2, MPTPC1 and MPTPC2 PT cells by qRT-PCR and immunofluorescence assays. The transfected BPTPC1 and BPTPC2 cells expressed much higher levels of HPV-16 E6/E7 than the non-transfected cells (Figure 2D, E), and the transfected MPTPC1 and MPTPC2 cells expressed much higher levels of SV40-T than their controls (Figure 2D, E), thus suggesting that the viral immortalization genes were successfully transfected into BPTPC1, BPTPC2, MPTPC1 and MPTPC2. Furthermore, the transfected BPTPC1, BPTPC2, MPTPC1 and MPTPC2 cells were continuously cultured to more than 50 passages while maintaining their original spindle cell morphology (Figure 2A). After HPV-16 E6/E7 transfection, the doubling time of BPTPC1 and BPTPC2 changed from 96 hours to 72 hours. After SV40-T transfection, the doubling time of MPTPC1 and MPTPC2 changed from 72 hours to 48 hours. In cell proliferation assays, we also observed that the transfected BPTPC1, MPTPC1 and MPTPC2 proliferated more rapidly than parental early PTPCs (Figure 2B).
The BPTPC1 and BPTPC2 cells successfully expressing HPV-16 E6/E7 were considered benign PT continuous cell lines, and the MPTPC1 and MPTPC2 cells successfully expressing SV40-T were considered malignant PT continuous cell lines; these cell lines were denoted SYSH-BPT-01, SYSH-BPT-02, SYSH-MPT-01 and SYSH-MPT-02, respectively.
Continuous passage
Under stimulation by external physical, chemical and biological factors, cells can spontaneously gain immortalization; one example is human cervical cancer cell line HeLa [22]. Cells that can be cultured continuously to more than 50 passages and proliferate infinitely are called continuous cell lines. During the routine passage of PTPCs, we found that MPTPC3 and MPTPC4 could be cultured to more than 50 passages in vitro with a rapid proliferation rate. We compared the cell morphology among early PTPCs, (later PTPCs) and PTPCs cultured to more than 50 passages (P50 PTPCs). The P50 PTPCs of both MPTPC3 and MPTPC4 maintained an elongated spindle shape similar to that of parental early and (later PTPCs) (Figure 2A). Proliferation assays indicated that MPTPC3 and MPTPC4 maintained rapid proliferation rates throughout continuous passage. No significant difference was found in the proliferation rates of early PTPCs, (later PTPCs) and P50 PTPCs (Figure 2B). Hence, we considered the P50 PTPCs of MPTPC3 and MPTPC4 to be malignant PT continuous cell lines, which were denoted SYSH-MPT-03 and SYSH-MPT-04, respectively.
The established phyllodes tumor cell lines display biological functions consistent with those of parental primary cells
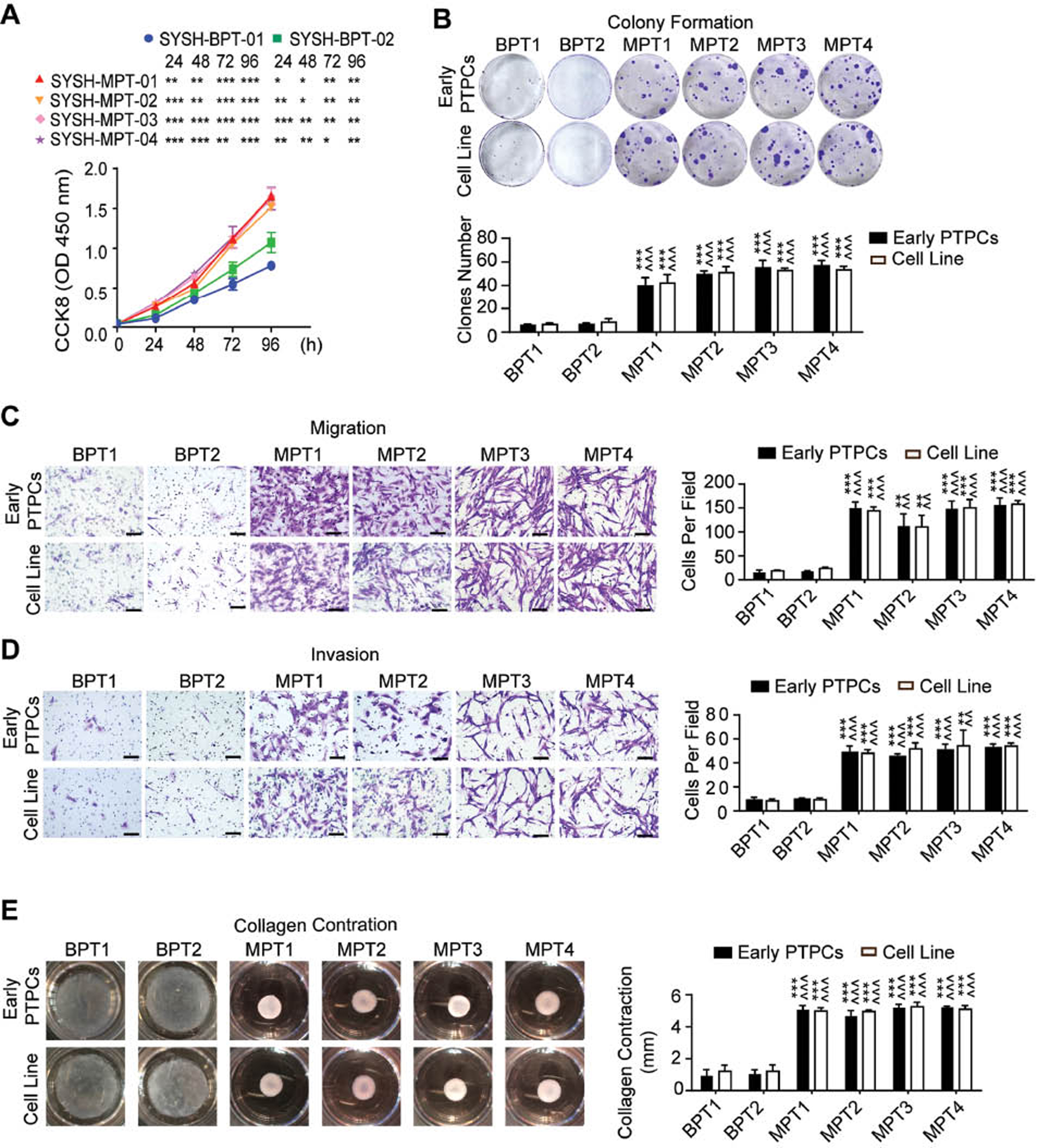
To investigate whether the functions of PT cell lines might be identical to those of the parental PTPCs, we conducted a series of functional assays. In proliferation assays, all malignant PT cell lines proliferated more rapidly than the two benign PT cell lines (Figure 3A). Furthermore, we compared the difference between PT cell lines and parental PTPCs, and found that the SYSH-BPT-01, SYSH-MPT-01 and SYSH-MPT-02 cells proliferated more rapidly than the parental early PTPCs, whereas SYSH-BPT-02, SYSH-MPT-03 and SYSH-MPT-04 did not (Figure 2B). In colony formation assays, SYSH-BPT-01, SYSH-BPT-02 formed only several small clones, whereas SYSH-MPT-01, SYSH-MPT-02, SYSH-MPT-03 and SYSH-MPT-04 formed more and larger clones, which were identical to those of parental early PTPCs (Figure 3B). Moreover, migration and invasion assays indicated that malignant PT cell lines showed greater migration and invasion ability than benign PT cell lines. Of note, all PT cell lines displayed similar migration and invasion ability to those of their parental early PTPCs (Figure 3C, D). Consistently, collagen contraction assays revealed that SYSH-BPT-01 and SYSH-BPT-02 scarcely contracted collagen gels, whereas SYSH-MPT-01, SYSH-MPT-02, SYSH-MPT-03 and SYSH-MPT-04 contracted collagen gels to a much greater extent similar to that of parental early PTPCs (Figure 3E).
Figure 3 The established phyllodes tumor cell lines display biological functions consistent with those of parental primary cells. A CCK8 cell proliferation assay showed the proliferation rate of PT cell lines. B (Upper) Images of colony formation assay of PTPCs and parental PT cell lines. (Below) Bar chart showed the clones number of each group. C (Left) Images of migration assay of PTPCs and parental PT cell lines. Scale bar, 100 µm. (Right) Bar chart showed the number of migration cells under 200 × field of each group. D (Left) Images of invasion assay of PTPCs and parental PT cell lines. Scale bar, 100 µm. (Right) Bar chart showed the number of invasion cells under 200 × field of each group. E (Left) Images of collagen contraction assay of PTPCs and parental PT cell lines. (Right) Bar chart showed the contraction length of each group. Data in (B – E) were shown as mean ± SEM, n=3. (* displayed the significance between malignant PT cells and BPTPC1/SYSH-BPT-01; ^ displayed the significance between malignant PT cells and BPTPC2/SYSH-BPT-02. *, P < 0.05; **, P < 0.01; ***, P < 0.001).
These results indicated that the malignant PT cell lines appeared more aggressive than benign PT cell lines, with stronger proliferation, colony formation, migration, invasion and collagen contraction ability, in agreement with the results for early PTPCs. Furthermore, all PT cell lines had similar biological functions to those of their corresponding parental early PTPCs. Therefore, the established PT cell lines displayed similar behaviors to those of the parental PTPCs and therefore may be ideal for PT in vitro functional assays.
Established phyllodes tumor cell lines preserve the features of parental primary cells
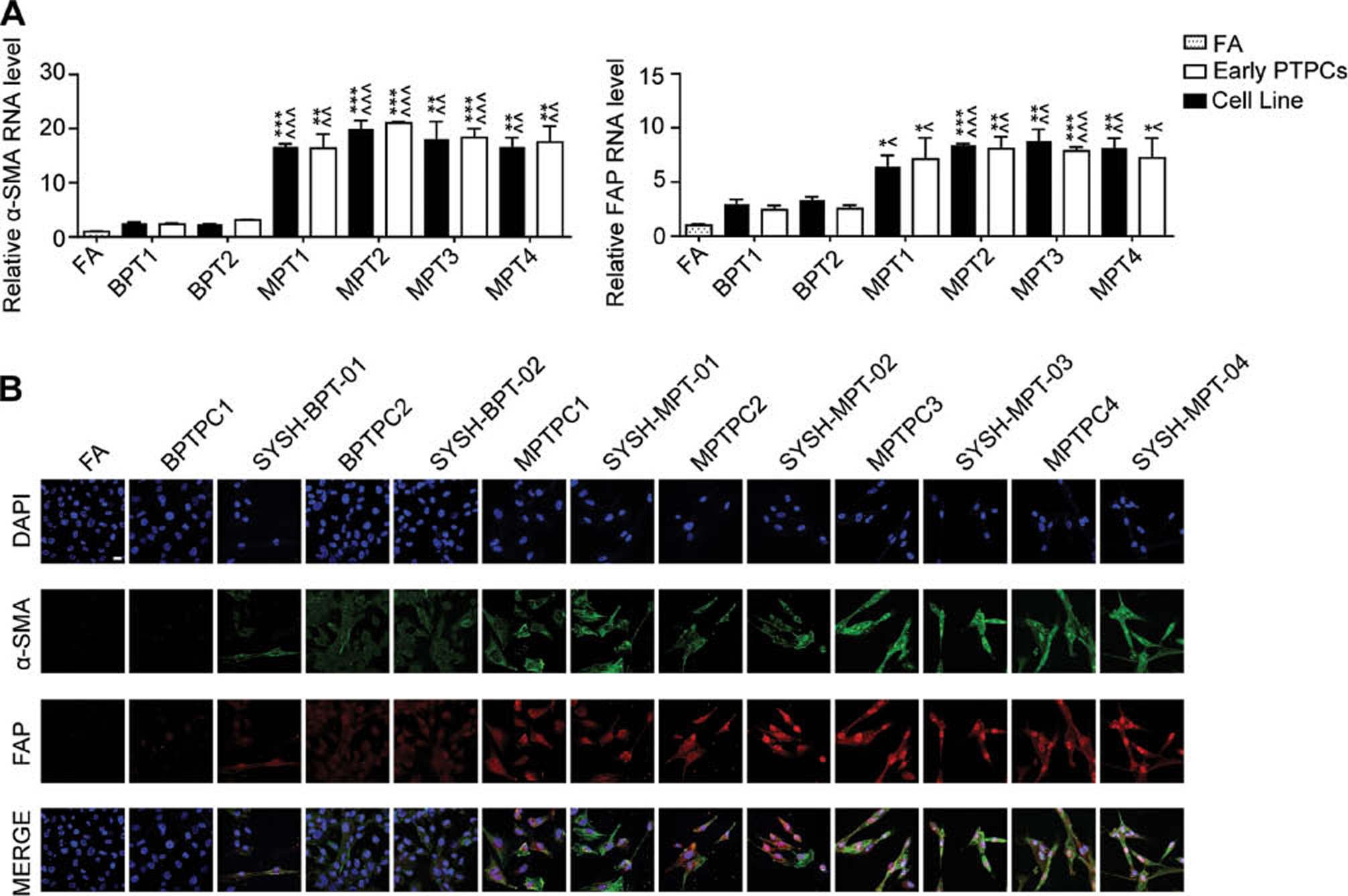
To verify whether these PT cell lines preserved the features of PTs after undergoing continuous passage in vitro, we detected PT markers and performed STR identification. Our previous studies have indicated that α-SMA and FAP, hallmarks of myofibroblasts, progressively increased during the malignant progression of PTs [16]. Therefore, we detected their expression levels in paired PT cell lines and PTPCs through qRT-PCR. Breast fibroadenoma cells, used as a negative control, had low α-SMA and FAP expression, whereas the expression of α-SMA and FAP in malignant PT cell lines was significantly higher than that in benign PT cell lines. Each PT cell line had similar expression levels of α-SMA and FAP to those in parental PTPCs (Figure 4A). Moreover, the malignant PTPCs and corresponding cell lines had stronger α-SMA and FAP fluorescence signals than the benign PT cells in immunofluorescence assays (Figure 4B).
Figure 4 Established phyllodes tumor cell lines preserve the features of parental primary cells. A The expression levels of α-SMA and FAP mRNA were determined by qRT-PCR in PTPCs and parental PT cell lines. The FA served as a negative control, RNA expression normalized to GAPDH. All data were shown as mean ± SEM, n=3. (* displayed the significance between malignant PT cells and BPTPC1/SYSH-BPT-01; ^ displayed the significance between malignant PT cells and BPTPC2/SYSH-BPT-02. *, P < 0.05; **, P < 0.01; ***, P < 0.001). B Immunofluorescence assay showed the expression levels of α-SMA (Green) and FAP (Red) in PTPCs and parental PT cell lines. Scale bar, 20 µm
STR identification was used to verify that the cell lines had not been cross-contaminated or incorrectly identified. Newly established cell lines require STR identification for confirmation of their identity. We performed the STR identification on PT cell lines cultured to more than 50 passages and parental PTPCs. The copy numbers of the 21 STR loci in each PT cell line matched those of the parental PTPCs (Table S2), thus clearly indicating that all PT cell lines were identical to the corresponding parental PTPCs and were not cross-contaminated by other cells. Together, these findings suggested that the PT cell lines established herein were stable and preserved the genetic features of PTPCs throughout continuous passaging in vitro. Therefore, these cells may be suitable for research on PT pathological mechanisms in future studies.
Discussion
In this study, we established two benign PT and four malignant PT cell lines through continuous passage or cell immortalization. Furthermore, through a series of experiments, we verified that the cell lines were morphologically and functionally consistent with the parental PTPCs. STR identification also verified that the PT cell lines were identical to the parental PTPCs.
Breast PTs are typically large and fast-growing tumors. Patients with benign PTs can experience local recurrence, and recurrent PTs show progression toward malignant phenotypes [4–6]. Patients with malignant PTs may experience repeated recurrence, rapid metastasis and death due to a lack of effective treatment, thus placing patients under great psychological pressure [6, 8]. The treatments differ between benign PTs and malignant PTs. Whereas wide tumor resection is sufficient for benign PTs, malignant PTs require additional adjuvant therapy to decrease the high rate of recurrence and metastasis. However, current adjuvant radiotherapy and chemotherapy are overall ineffective against malignant PTs [3]. Of note, no effective targeted therapy against PTs is available, because of deficiencies in tools for PT research. Our prior studies on PTs have relied on fresh frozen tissues from patients with PTs and PTPCs. However, the source of PT tissue is limited by the low incidence of PTs. Moreover, even at low temperatures, nucleic acids and proteins in tissues may degrade over time. Furthermore, culturing PTPCs is difficult, because the in vitro culture conditions cannot easily meet the requirements of PTPCs. Repeated isolation of PTPCs is costly and time consuming, thus hindering research. Therefore, established PT cell lines are much needed.
Cell lines are routinely used to screen drugs, validate therapeutic targets and develop vaccines. Monoclonal antibodies to human epidermal growth factor receptor 2 (HER2) are an effective treatment for breast cancer with high HER2 expression, on the basis of in vitro studies on cell lines [31]. Trastuzumab and pertuzumab are representative drugs used in clinical treatments. Moreover, HeLa, HEK-293 and Vero cell lines have been used to develop vaccines for rabies, poliovirus, measles virus, rubella virus and SARS-CoV-2 [32–35]. Cancer vaccines are designed to promote the immune system’s elimination of tumors, and cell lines could be used to validate the efficacy of cancer vaccines. Thus, to better study new therapeutic targets for malignant PTs, herein we selected representative benign and malignant PTs for establishing PT cell lines.
HPV-16 E6/E7 and SV40-T are common viral genes used for cell immortalization [21–23]. However, some cells may acquire immortalization ability spontaneously. For example, the oldest and widely used cell line, HeLa, can be passaged in vitro infinitely, owing to its telomerase activity [22, 36]. In this study, SYSH-BPT-01 and SYSH-BPT-02 were benign PT cell lines transfected with HPV-16 E6/E7; SYSH-MPT-01 and SYSH-MPT-02 were malignant PT cell lines transfected with SV40-T; and SYSH-MPT-03 and SYSH-MPT-04 were malignant PT cells lines with inherent immortalization. The PT cell lines established herein preserve the functions and features of their parental PTPCs, thus demonstrating their potential for use in PT in vitro functional assays. These cells may be highly important for screening potential drugs and vaccines against PTs. However, PT cell lines that could be used in animal models are lacking. Further work on establishing more PT cell lines is needed to accelerate the future study of PTs.
In conclusion, we established two benign PT cell lines and four malignant PT cell lines displaying biological functions and features consistent with those of the parental PTPCs. These PT cell lines may serve as promising tools for research on PTs, thus contributing to the development of effective therapeutic strategies for malignant PTs in the future.
Abbreviations
| PTs: | Phyllodes tumors |
| BPT: | Benign phyllodes tumor |
| MPT: | Malignant phyllodes tumor |
| PTPCs: | Phyllodes tumor primary cells |
| BPTPC: | Benign phyllodes tumor primary cells |
| MPTPC: | Malignant phyllodes tumor primary cell |
| HPV: | Human papillomavirus |
| SV40-T: | Simian virus 40 large T antigen |
| qRT-PCR: | Quantitative real-time PCR |
| α-SMA: | α-Smooth muscle actin |
| FAP: | Fibroblast activation protein |
| STR: | Short tandem repeat |
| HER2: | Human epidermal growth factor receptor 2 |
Acknowledgments
The authors thank the patients who participated in the trial and their families, and acknowledge assistance from the Disease Registry Department of Sun Yat-Sen Memorial Hospital, Sun Yat-sen University.
Statements and Declarations
Funding
This work was supported by the National Natural Science Foundation of China (82173054, 81872158 and 82001822), Guangdong Science and Technology Department (2022B1515020048 and 2018A030313097), and Guangzhou Science, Technology and Innovation Commission (202102010148).
Competing Interests
The authors declare that they have no competing interests.
Author Contributions
Shishi He and Xiaoyun Xiao contributed equally to this article. Conception and design were performed by Yan Nie and Phei Er Saw. Data collection and analysis were performed by Shishi He and Xiaoyun Xiao. PT primary cell isolation was performed by Shishi He, Rong Lei, Jiewen Chen, Hongyan Huang and Ailifeire Yilihamu. Material preparation and clinical information collection were performed by Rong Lei, Mingyan Guo, Cui Tan, Xun Li and Zilin Zhuang. The first draft of the manuscript was written by Shishi He. All authors read and approved the manuscript.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval
The study was approved by Sun Yat-sen Memorial Hospital, Sun Yat-sen University ethics Committee, under approval number 2018-229.
Consent to Participate
Informed consent was obtained from all patients in this study.
Consent to Publish
The authors affirm that the human research participants provided informed consent for publication.
Supplementary Tables
Click here to download Supplementary Tables.
References
- Zhang Y, Kleer CG. Phyllodes tumor of the breast: histopathologic features, differential diagnosis, and molecular/genetic updates. Arch Pathol Lab Med 2016;140:665-71. [PMID: 27362571 DOI: 10.5858/arpa.2016-0042-RA]
- Cserni G. Histological type and typing of breast carcinomas and the WHO classification changes over time. Pathologica 2020;112:25-41. [PMID: 32202537 DOI: 10.32074/1591-951X-1-20]
- Chao X, Chen K, Zeng J, Bi Z, Guo M, et al. Adjuvant radiotherapy and chemotherapy for patients with breast phyllodes tumors: a systematic review and meta-analysis. BMC Cancer 2019;19:372. [PMID: 31014268 DOI: 10.1186/s12885-019-5585-5]
- Zhou ZR, Wang CC, Sun XJ, Yang ZZ, Chen XX, et al. Prognostic factors in breast phyllodes tumors: a nomogram based on a retrospective cohort study of 404 patients. Cancer Med 2018;7:1030-42. [PMID: 29479819 DOI: 10.1002/cam4.1327]
- Ramakant P, Chakravarthy S, Cherian JA, Abraham DT, Paul MJ. Challenges in management of phyllodes tumors of the breast: a retrospective analysis of 150 patients. Indian J Cancer 2013;50:345-8. [PMID: 24369215 DOI: 10.4103/0019-509X.123625]
- Kim S, Kim JY, Kim DH, Jung WH, Koo JS. Analysis of phyllodes tumor recurrence according to the histologic grade. Breast Cancer Res Treat 2013;141:353-63. [PMID: 24062207 DOI: 10.1007/s10549-013-2684-x]
- Wang ZC, Buraimoh A, Iglehart JD, Richardson AL. Genome-wide analysis for loss of heterozygosity in primary and recurrent phyllodes tumor and fibroadenoma of breast using single nucleotide polymorphism arrays. Breast Cancer Res Treat 2006;97:301-9. [PMID: 16791486 DOI: 10.1007/s10549-005-9124-5]
- Strode M, Khoury T, Mangieri C, Takabe K. Update on the diagnosis and management of malignant phyllodes tumors of the breast. Breast 2017;33:91-6. [PMID: 28327352 DOI: 10.1016/j.breast.2017.03.001]
- Chaney AW, Pollack A, McNeese MD, Zagars GK, Pisters PW, et al. Primary treatment of cystosarcoma phyllodes of the breast. Cancer 2000;89:1502-11. [PMID: 11013364 DOI: 10.1002/1097-0142(20001001)89:7<1502::aid-cncr13>3.0.co;2-p]
- Tan BY, Acs G, Apple SK, Badve S, Bleiweiss IJ, et al. Phyllodes tumours of the breast: a consensus review. Histopathology 2016;68:5-21. [PMID: 26768026 DOI: 10.1111/his.12876]
- Moffat CJ, Pinder SE, Dixon AR, Elston CW, Blamey RW, et al. Phyllodes tumours of the breast: a clinicopathological review of thirty-two cases. Histopathology 1995;27:205-18. [PMID: 8522284 DOI: 10.1111/j.1365-2559.1995.tb00212.x]
- Nie Y, Chen J, Huang D, Yao Y, Chen J, et al. Tumor-associated macrophages promote malignant progression of breast phyllodes tumors by inducing myofibroblast differentiation. Cancer Res 2017;77:3605-18. [PMID: 28512246 DOI: 10.1158/0008-5472.CAN-16-2709]
- Reinfuss M, Mitus J, Duda K, Stelmach A, Rys J, et al. The treatment and prognosis of patients with phyllodes tumor of the breast: an analysis of 170 cases. Cancer 1996;77:910-6. [PMID: 8608483 DOI: 10.1002/(sici)1097-0142(19960301)77:5<910::aid-cncr16>3.0.co;2-6]
- Abdalla HM, Sakr MA. Predictive factors of local recurrence and survival following primary surgical treatment of phyllodes tumors of the breast. J Egypt Natl Canc Inst 2006;18:125-33. [PMID: 17496937]
- Chao X, Jin X, Tan C, Sun P, Cui J, et al. Re-excision or “wait and watch”-a prediction model in breast phyllodes tumors after surgery. Ann Transl Med 2020;8:371. [PMID: 32355815 DOI: 10.21037/atm.2020.02.26]
- Gong C, Nie Y, Qu S, Liao JY, Cui X, et al. miR-21 induces myofibroblast differentiation and promotes the malignant progression of breast phyllodes tumors. Cancer Res 2014;74:4341-52. [PMID: 24980553 DOI: 10.1158/0008-5472.CAN-14-0125]
- Nie Y, Huang H, Guo M, Chen J, Wu W, et al. Breast phyllodes tumors recruit and repolarize tumor-associated macrophages via secreting cCL5 to promote malignant progression, which can be inhibited by CCR5 inhibition therapy. Clin Cancer Res 2019;25:3873-86. [PMID: 30890553 DOI: 10.1158/1078-0432.CCR-18-3421]
- Hayflick L. A brief history of the mortality and immortality of cultured cells. Keio J Med 1998;47:174-82. [PMID: 9785764 DOI: 10.2302/kjm.47.174]
- Duncan EL, Wadhwa R, Kaul SC. Senescence and immortalization of human cells. Biogerontology 2000;1:103-21. [PMID: 11707927 DOI: 10.1023/a:1010000132671]
- Gudjonsson T, Villadsen R, Ronnov-Jessen L, Petersen OW. Immortalization protocols used in cell culture models of human breast morphogenesis. Cell Mol Life Sci 2004;61:2523-34. [PMID: 15526159 DOI: 10.1007/s00018-004-4167-z]
- Hubbard K, Ozer HL. Mechanism of immortalization. Age (Omaha) 1999;22:65-9. [PMID: 23604398 DOI: 10.1007/s11357-999-0008-1]
- Jeong Seo E, Jung Kim H, Jae Lee C, Tae Kang H, Seong Hwang E. The role of HPV oncoproteins and cellular factors in maintenance of hTERT expression in cervical carcinoma cells. Gynecol Oncol 2004;94:40-7. [PMID: 15262117 DOI: 10.1016/j.ygyno.2004.03.041]
- Jha KK, Banga S, Palejwala V, Ozer HL. SV40-Mediated immortalization. Exp Cell Res 1998;245:1-7. [PMID: 9828095 DOI: 10.1006/excr.1998.4272]
- Darimont C, Mace K. Immortalization of human preadipocytes. Biochimie 2003;85:1231-3. [PMID: 14739075 DOI: 10.1016/j.biochi.2003.10.015]
- Liu X, Disbrow GL, Yuan H, Tomaic V, Schlegel R. Myc and human papillomavirus type 16 E7 genes cooperate to immortalize human keratinocytes. J Virol 2007;81:12689-95. [PMID: 17804506 DOI: 10.1128/JVI.00669-07]
- Hung SC, Yang DM, Chang CF, Lin RJ, Wang JS, et al. Immortalization without neoplastic transformation of human mesenchymal stem cells by transduction with HPV16 E6/E7 genes. Int J Cancer 2004;110:313-9. [PMID: 15095294 DOI: 10.1002/ijc.20126]
- Daya-Grosjean L, Azzarone B, Maunoury R, Zaech P, Elia G, et al. SV40 immortalization of adult human mesenchymal cells from neuroretina. Biological, functional and molecular characterization. Int J Cancer 1984;33:319-29. [PMID: 6321361 DOI: 10.1002/ijc.2910330308]
- Ozer HL, Banga SS, Dasgupta T, Houghton J, Hubbard K, et al. SV40-mediated immortalization of human fibroblasts. Exp Gerontol 1996;31:303-10. [PMID: 8706800 DOI: 10.1016/0531-5565(95)00024-0]
- Maishi N, Kikuchi H, Sato M, Nagao-Kitamoto H, Annan DA, et al. Development of Immortalized human tumor endothelial cells from renal cancer. Int J Mol Sci 2019;20:4595. [PMID: 31533313 DOI: 10.3390/ijms20184595]
- Liu G, Friggeri A, Yang Y, Milosevic J, Ding Q, et al. miR-21 mediates fibrogenic activation of pulmonary fibroblasts and lung fibrosis. J Exp Med 2010;207:1589-97. [PMID: 20643828 DOI: 10.1084/jem.20100035]
- Hudziak RM, Lewis GD, Winget M, Fendly BM, Shepard HM, et al. p185HER2 monoclonal antibody has antiproliferative effects in vitro and sensitizes human breast tumor cells to tumor necrosis factor. Mol Cell Biol 1989;9:1165-72. [PMID: 2566907 DOI: 10.1128/mcb.9.3.1165-1172.1989]
- Scherer WF, Syverton JT, Gey GO. Studies on the propagation in vitro of poliomyelitis viruses. IV. Viral multiplication in a stable strain of human malignant epithelial cells (strain HeLa) derived from an epidermoid carcinoma of the cervix. J Exp Med 1953;97:695-710. [PMID: 13052828 DOI: 10.1084/jem.97.5.695]
- Petiot E, Cuperlovic-Culf M, Shen CF, Kamen A. Influence of HEK293 metabolism on the production of viral vectors and vaccine. Vaccine 2015;33:5974-81. [PMID: 26073013 DOI: 10.1016/j.vaccine.2015.05.097]
- Aubrit F, Perugi F, Leon A, Guehenneux F, Champion-Arnaud P, et al. Cell substrates for the production of viral vaccines. Vaccine 2015;33:5905-12. [PMID: 26187258 DOI: 10.1016/j.vaccine.2015.06.110]
- Gao Q, Bao L, Mao H, Wang L, Xu K, et al. Development of an inactivated vaccine candidate for SARS-CoV-2. Science 2020;369:77-81. [PMID: 32376603 DOI: 10.1126/science.abc1932]
- Harley CB, Sherwood SW. Telomerase, checkpoints and cancer. Cancer Surv 1997;29:263-84. [PMID: 9338104]