Understanding of the Entry Mechanism of Nanoparticles into Tumors Determines the Future Direction of Nanomedicine Development
1Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
2Medical Research Center, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
3Department of Biological Sciences, Korea Advanced Institute of Science and Technology (KAIST), 291 Daehak-ro, Daejeon 34141, Korea
4Center for Precision Bio-Nanomedicine, Korea Advanced Institute of Science and Technology (KAIST), 291 Daehak-ro, Daejeon 34141, Korea
*Correspondence to: Phei Er Saw, E-mail: caipeie@mail.sysu.edu.cn; Sangyong Jon, E-mail: syjon@kaist.ac.kr
Received: August 9 2020; Revised: September 1 2020; Accepted: September 7 2020; Published Online: December 22 2020
Cite this paper:
Phei Er Saw and Sangyong Jon. Understanding of the Entry Mechanism of Nanoparticles into Tumors Determines the Future Direction of Nanomedicine Development. BIO Integration 2020; 1(4): 193–195.
DOI: 10.15212/bioi-2020-0033. Available at: https://bio-integration.org/
Download citation
© 2020 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
According to the prevalent concept of enhanced permeability and retention (EPR) effect proposed by Matsumura and Maeda in 1986 [1], nanoparticles can passively accumulate at the tumor site via gaps between endothelial cells (inter-endothelial gaps) in tumor-associated blood vessels that have abnormal structural integrity with pores ranging in size from submicron to micron. According to the paper published very recently by Sindhwani et al. [2], however, this classic concept needs to be modified or replaced by a new concept on the entry of nanoparticles into solid tumors. In their paper, Sindhwani and his team focused on how nanoparticles – represented by gold nanoparticles – enter tumors, by carrying out detailed visualization and data quantification on specific areas such as blood vessel gaps in tumors, accumulation and localization of nanoparticles in tumors, and also the comparison of transendothelially transported rate of nanoparticles to the rate of diffusion across blood vessel gaps. They analyzed 313 blood vessels across several tumor models, such as U87-MG glioma, 4T1 breast cancer, MMTV-PyMT breast cancer, PDX breast cancer and so on, and found only 26 gaps, which implied that gaps occurred at a very low frequency in all the aforementioned tumor types, which is consistent with the knowledge that gaps are also rare in tumor vessels from cancer patients. Furthermore, they demonstrated that only seven out of 26 openings were intercellular gaps (26.9%), while the rest were transendothelial pores. Through this study, they claimed that the main mechanism of nanoparticle tumor entry is through transendothelial openings. In fact, in 1997, researchers at Harvard University had proposed that the endothelial cell gaps that were induced by vasoactive mediators were mostly transcellular pores [3]. Feng et al. defined a novel endothelial cell organelle in the endothelial cells of both humans and animals, termed the vesiculovacuolar organelle (VVO) [3]. It participates in the regulation of intracellular transportation pathways during the transendothelial transport of soluble macromolecules. In this regard, there is a structural basis for nanoparticles to enter tumors through transendothelial pathways. According to this article, the extravasation of nanoparticles shows no significant relevance with tight junctions, fenestra, vesicles or endothelial gaps in blood vessels, which infers that nanoparticles primarily enter tumors by active transportation. Based on the aforementioned fact, there are two possible ends when nanoparticles enter the circulation. The first one is that nanoparticles are also capable of entering other organs by crossing the endothelial cells. Feng and coworkers showed that colloidal carbon could traverse endothelial cells via the development of pores; this mechanism does not require communication with the intercellular gap junction, nor do they need to disrupt intercellular junctions by gap formation in these vessels [4]. If this hypothesis of “nanoparticles entering normal vessels” proved to be true, this may explain why the efficiency of nanoparticle delivery to tumors is mostly lower than 1% in clinical experiments [5].
Hida et al. reported that endothelial cells in tumors exhibit different characteristics from normal vascular endothelial cells in terms of cell proliferation, migration ability, gene expression and response to growth factors and drugs [6]. Akino et al. showed that in human clear cell renal cell carcinoma (CCRCC), 22–58% (median, 33%) of tumor-associated endothelial cells were aneuploid, while normal endothelial cells were diploid [7]. Furthermore, tumor endothelial cells expressed the epidermal growth factor receptor (EGFR), which activates the mitogen-activated protein kinase (MAPK) signaling pathway and accelerates the proliferation rate of tumor cells. On the other hand, tumor endothelial cells did not express ErbB3, a gene that encodes for a protein that inhibits endothelial cell growth by coupling with neuregulin (NRG) ligands. Therefore, they concluded that tumor endothelial cells could proliferate faster than normal vascular endothelial cells [8]. In this regard, it could be inferred that the tumor-associated endothelial cells might have a higher active transportation capacity due to the enhanced proliferation than those in normal vessels.
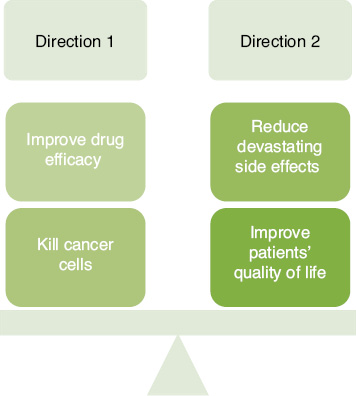
On December 4, 2019, an editorial titled “The Two Directions of Cancer Nanomedicine” was published in Nature Nanotechnology [9]. This editorial discussed the two future directions of nanomedicine development, that is, (i) to accelerate clinical transformation of nanomedicine to improve cancer treatment regimes or (ii) to boost the research on the mechanism of action of nanomedicine by reducing devastating side effects (Figure 1). The million dollar question now is: how do we balance the two sides? The ideal approach would be for targeted therapeutics, such as nanoparticle encapsulating chemotherapeutic drugs, to efficiently and specifically target only tumor cells upon systemic injection. This could be enhanced by attaching specific targeting ligands onto the nanoparticle that could bind to overexpressing receptors on tumor cells [10–12]. In this way, we could not only reduce the detrimental side effects of non-targeted chemotherapy but also significantly enhance the drug efficacy and the ability to kill tumor cells [13, 14]. Hence, understanding the nanoparticle cellular entry mechanism could indeed pave the way for future therapeutic modality for solid tumors.
Figure 1 The balancing of both the directions of cancer nanomedicine is crucial in creating an optimal treatment with the highest efficacy in combating cancer cells while reducing devastating side effects and improving the quality of life (QoL) of patients.
One reason that scientists are advocating for the latter is that the delivery efficiency of nanomedicine into tumors is still very low. It is suggested that research and development in the field of nanomedicine ought to focus on understanding of the mechanism of action first on the tumor entry of nanomedicine, followed by pursuing the acceleration of clinical translation. For example, a better understanding of the differences between normal and tumor-associated endothelial cells will give a clue or hint for a better design of the nanomedicine, enabling greater accumulation or entry into the tumor. In this regard, nanomedicine able to respond to various stimuli present in the tumor has been developed as a strategy to increase the tumor accumulation efficiency. Hida et al. showed that high levels of reactive oxygen species (ROS) accumulated in blood vessels of human tumor tissues, including hepatocellular carcinoma, colorectal cancer and renal cell carcinoma, compared with normal counterparts and showed that contact with cancer cells induced a rapid increase in ROS in human umbilical vascular endothelial cells [8, 15, 16]. Apart from that, there are also various reports that have shown that pH, glutathione (GSH) and other substances can trigger the disruption of nanoparticles [12, 17–19]. Overall, it is necessary for us to explore the dynamics of nanoparticles in tumor and normal endothelial cells.
This article is of great significance as it provides a profound horizon for future directions of nanomedicine development, leading us to focus research on the mechanism of nanoparticle entry into tumor instead of clinical transformation itself. It reminds us that we can design better nanoparticles to achieve efficient clinical transformation only when we have a deep understanding of the mechanism or the mode of action. Surely, some questions remain. Although a thorough investigation on the entry of gold nanoparticles into tumors was made in the article, nanocarriers come in many forms including high-molecular-weight polymeric nanoparticles, liposomes, virus-like particles, iron oxide nanoparticles and water-based nanogels, just to name a few. The entry mechanism of other nanocarriers remains unclear and thus requires further studies. Do other nanoparticles also enter the tumor via transendothelial openings? Why were the nanoparticles released polarly from the opposite end of endothelial cells after entering from one end? Why were they not released adjacently or retained in the endothelial cells? All these unanswered questions demand further in-depth research.
References
- Matsumura Y, Maeda H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res 1986;46:6387-92. [PMID: 2946403]
- Sindhwani S, Syed AM, Ngai J, Kingston BR, Maiorino L, et al. The entry of nanoparticles into solid tumours. Nat Mater 2020;19:566-75. [PMID: 31932672 DOI: 10.1038/s41563-019-0566-2]
- Feng D, Nagy JA, Hipp J, Pyne K, Dvorak HF, et al. Reinterpretation of endothelial cell gaps induced by vasoactive mediators in guinea-pig, mouse and rat: many are transcellular pores. J Physiol 1997;504: 747-61. [PMID: 9401980 DOI: 10.1111/j.1469-7793.1997.747bd.x]
- Feng D, Nagy JA, Dvorak HF, Dvorak AM. Ultrastructural studies define soluble macromolecular, particulate, and cellular transendothelial cell pathways in venules, lymphatic vessels, and tumor-associated microvessels in man and animals. Microsc Res Tech 2002;57:289-326. [PMID: 12112440 DOI: 10.1002/jemt.10087]
- Wilhelm S, Tavares AJ, Dai Q, Ohta S, Audet J, et al. Analysis of nanoparticle delivery to tumours. Nat Rev Mater 2016;1:16014. [DOI: 10.1038/natrevmats.2016.14]
- Hida K, Maishi N, Sakurai Y, Hida Y, Harashima H. Heterogeneity of tumor endothelial cells and drug delivery. Adv Drug Deliv Rev 2016;99:140-7. [PMID: 26626622 DOI: 10.1016/j.addr.2015.11.008]
- Akino T, Hida K, Hida Y, Tsuchiya K, Freedman D, et al. Cytogenetic abnormalities of tumor-associated endothelial cells in human malignant tumors. Am J Pathol 2009;175:2657-67. [PMID: 19875502 DOI: 10.2353/ajpath.2009.090202]
- Amin DN, Hida K, Bielenberg DR, Klagsbrun M. Tumor endothelial cells express epidermal growth factor receptor (EGFR) but not ErbB3 and are responsive to EGF and to EGFR kinase inhibitors. Cancer Res 2006;66:2173-80. [PMID: 16489018 DOI: 10.1158/0008-5472.CAN-05-3387]
- The two directions of cancer nanomedicine. Nat Nanotechnol 2019;14:1083. [PMID: 31802029 DOI: 10.1038/s41565-019-0597-5]
- Ding J, Chen J, Gao L, Jiang Z, Zhang Y, et al. Engineered nanomedicines with enhanced tumor penetration. Nano Today 2019;29:100800. [DOI: 10.1016/j.nantod.2019.100800]
- Saw PE, Park J, Lee E, Ahn S, Lee J, et al. Effect of PEG pairing on the efficiency of cancer-targeting liposomes. Theranostics 2015;5:746-54. [PMID: 25897339 DOI: 10.7150/thno.10732]
- Lin C, Tao Y, Saw PE, Cao M, Huang H, et al. A polyprodrug-based nanoplatform for cisplatin prodrug delivery and combination cancer therapy. Chem Commun 2019;55:13987-90. [PMID: 31687673 DOI: 10.1039/c9cc06567a]
- Kim H, Lee Y, Lee IH, Kim S, Kim D, et al. Synthesis and therapeutic evaluation of an aptide-docetaxel conjugate targeting tumor-associated fibronectin. J Control Release 2014;178:118-24. [PMID: 24462899 DOI: 10.1016/j.jconrel.2014.01.015]
- Yu M, Amengual J, Menon A, Kamaly N, Zhou F, et al. Targeted nanotherapeutics encapsulating liver X receptor agonist GW3965 enhance antiatherogenic effects without adverse effects on hepatic lipid metabolism in Ldlr(-/-) mice. Adv Healthc Mater 2017;6. [PMID: 28730752 DOI: 10.1002/adhm.201700313]
- Haidari M, Zhang W, Willerson JT, Dixon RA. Disruption of endothelial adherens junctions by high glucose is mediated by protein kinase C-beta-dependent vascular endothelial cadherin tyrosine phosphorylation. Cardiovasc Diabetol 2014;13:105. [PMID: 25927959 DOI: 10.1186/1475-2840-13-105]
- Lin RZ, Wang TP, Hung RJ, Chuang YJ, Chien CC, et al. Tumor-induced endothelial cell apoptosis: roles of NAD(P)H oxidase-derived reactive oxygen species. J Cell Physiol 2011;226:1750-62. [PMID: 21506107 DOI: 10.1002/jcp.22504]
- Li S, Saw PE, Lin C, Nie Y, Tao W, et al. Redox-responsive polyprodrug nanoparticles for targeted siRNA delivery and synergistic liver cancer therapy. Biomaterials 2020;234:119760. [PMID: 31945619 DOI: 10.1016/j.biomaterials.2020.119760]
- Saw PE, Yao H, Lin C, Tao W, Farokhzad OC, et al. Stimuli-responsive polymer-prodrug hybrid nanoplatform for multistage siRNA delivery and combination cancer therapy. Nano Lett 2019;19: 5967-74. [PMID: 31381852 DOI: 10.1021/acs.nanolett.9b01660]
- Xu X, Wu J, Liu S, Saw PE, Tao W, et al. Redox-responsive nanoparticle-mediated systemic RNAi for effective cancer therapy. Small 2018;14:e1802565. [PMID: 30230235 DOI: 10.1002/smll.201802565]