Engineering Extracellular Vesicles for Osteoarthritis Treatment: From Therapeutic Strategies-to-Clinical Translation
1Department of Orthopedic Surgery, Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200233, China
2Department of Hand and Foot Surgery, The First Hospital of Jilin University, Changchun, Jilin 130021, China
3School of Biomedical Engineering, Shanghai Jiao Tong University, Shanghai 200030, China
*Correspondence to: Kai Feng, Department of Orthopedic Surgery, Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200233, China, E-mail: jsycfk@126.com; Xuetao Xie, Department of Orthopedic Surgery, Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200233, China, E-mail: xuetaoxie@163.com
Received: October 8 2025; Revised: December 29 2025; Accepted: January 6 2026; Published Online: February 3 2026
Cite this paper:
Liu J, Luo L, Feng K et al. Engineering Extracellular Vesicles for Osteoarthritis Treatment: From Therapeutic Strategies-to-Clinical Translation. BIO Integration 2026; 7: 1–22.
DOI: 10.15212/bioi-2025-0177. Available at: https://bio-integration.org/
Download citation
© 2026 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Extracellular vesicles (EVs) are membrane-bound nanoparticles secreted by cells and have emerged as promising therapeutic agents for osteoarthritis (OA). These vesicles exhibit high biocompatibility and the intrinsic ability to traverse physiologic barriers, delivering bioactive molecular cargo to recipient cells to exert regulatory effects. To enhance therapeutic efficacy, EVs can be bioengineered to improve biological activity and targeting capacity toward specific cells or tissues. Strategies for EV engineering primarily involve modifications to cargo, membranes, or the pretreatment of parental cells. In recent years advances in EV engineering have led to growing interest in the application of engineered EVs for OA treatment, offering new possibilities for clinical translation. In this review we summarize the fundamental characteristics of EVs and examine the factors limiting the current therapeutic efficacy in OA. We also provide an overview of existing engineering strategies and discuss the therapeutic applications of engineered EVs in OA models. Finally, we outline the remaining challenges and future directions necessary to advance EV-based therapies toward clinical use in OA.
Keywords
Bioengineering strategies; extracellular vesicles; osteoarthritis; therapeutic delivery.
Introduction
Osteoarthritis (OA) is a common degenerative joint disease and has become a major public health concern worldwide, particularly with the accelerating trend in population aging [1]. Various non-conventional therapeutic strategies, including cell-based therapies, have been explored for OA treatment over the past several decades [2]. However, several challenges, such as the low survival rate of transplanted cells [3], high cell culture costs, and the limited control over stem cell differentiation, have limited the large-scale clinical application of cell-based therapies [4]. Moreover, given the complexity of the host environment, it remains uncertain whether cell-based therapies may trigger safety issues, such as immune rejection or inflammatory responses [5]. These potential risks require long-term clinical validation. Although cell-based therapies have demonstrated promising effects in promoting cartilage repair and exerting anti-inflammatory functions in experimental studies, the underlying mechanisms remain poorly understood. With advances in research, increasing evidence has shown that extracellular vesicles (EVs) secreted by therapeutic cells also have a significant role in mediating these beneficial effects [6–8]. Accordingly, EV-based therapy has been further investigated as a cell-free alternative for the treatment of OA.
According to the minimal information for studies of extracellular vesicles (MISEV) guidelines, EVs are defined as particles that are released from cells, enclosed by a lipid bilayer, and lack the capacity for self-replication [9]. In previous studies the term “exosome” was frequently used as a general label for EVs. However, in a strict sense, exosomes refer specifically to EVs that originate from intracellular compartments and are released via a multivesicular body. However, due to the complexity of EV biogenesis and the difficulty in determining whether isolated EVs originate from the endosomal system, the blanket use of the term “exosome” for all isolated EVs is not appropriate [10]. Because most existing studies have not clearly identified the specific origin of the EVs used, we follow the MISEV2023 recommendation and uniformly adopt the term “EVs” throughout this review.
EVs contain a variety of biologically active molecules, such as nucleic acids, proteins, and lipids [11]. These components enable EVs to participate in intercellular material transport and signal transmission, thereby regulating the physiologic activities of recipient cells [12, 13]. Several studies have reported that the application of EVs contributes to the protection of cartilage tissue and alleviation of clinical symptoms, such as pain, in the context of OA treatment [14–16]. EVs may therefore represent a promising therapeutic option for the clinical management of OA based on a combination of low immunogenicity and favorable biocompatibility [17]. However, several issues need to be addressed before EVs-based therapies can be translated into clinical practice. Natural EVs are available only in limited yields and achieving the doses required for therapeutic use may substantially increase production costs [18]. The molecular cargo within EVs is highly complex [19, 20], yet only a limited number of these molecules are likely to contribute to the therapeutic effects [21, 22], resulting in constrained treatment efficacy. In addition, the targeting ability of natural EVs is limited because EVs lack the capacity to specifically home to desired tissues or cell types [23–25]. To address these limitations, various engineering strategies have been developed to modify EVs and enhance the suitability for OA therapy [26–29].
This review aimed to highlight engineered EVs as a promising therapeutic option for the clinical treatment of OA. We summarize the fundamental characteristics of EVs and discuss why native EVs may not fully meet the requirements for clinical application in OA. We then introduce current engineering strategies and corresponding technical approaches used to modify EVs. Finally, we review the therapeutic applications of engineered EVs in OA and discuss the remaining challenges that need to be addressed to advance the clinical translation.
Properties of EVs and limitations in OA therapy
Biogenesis and isolation of EVs
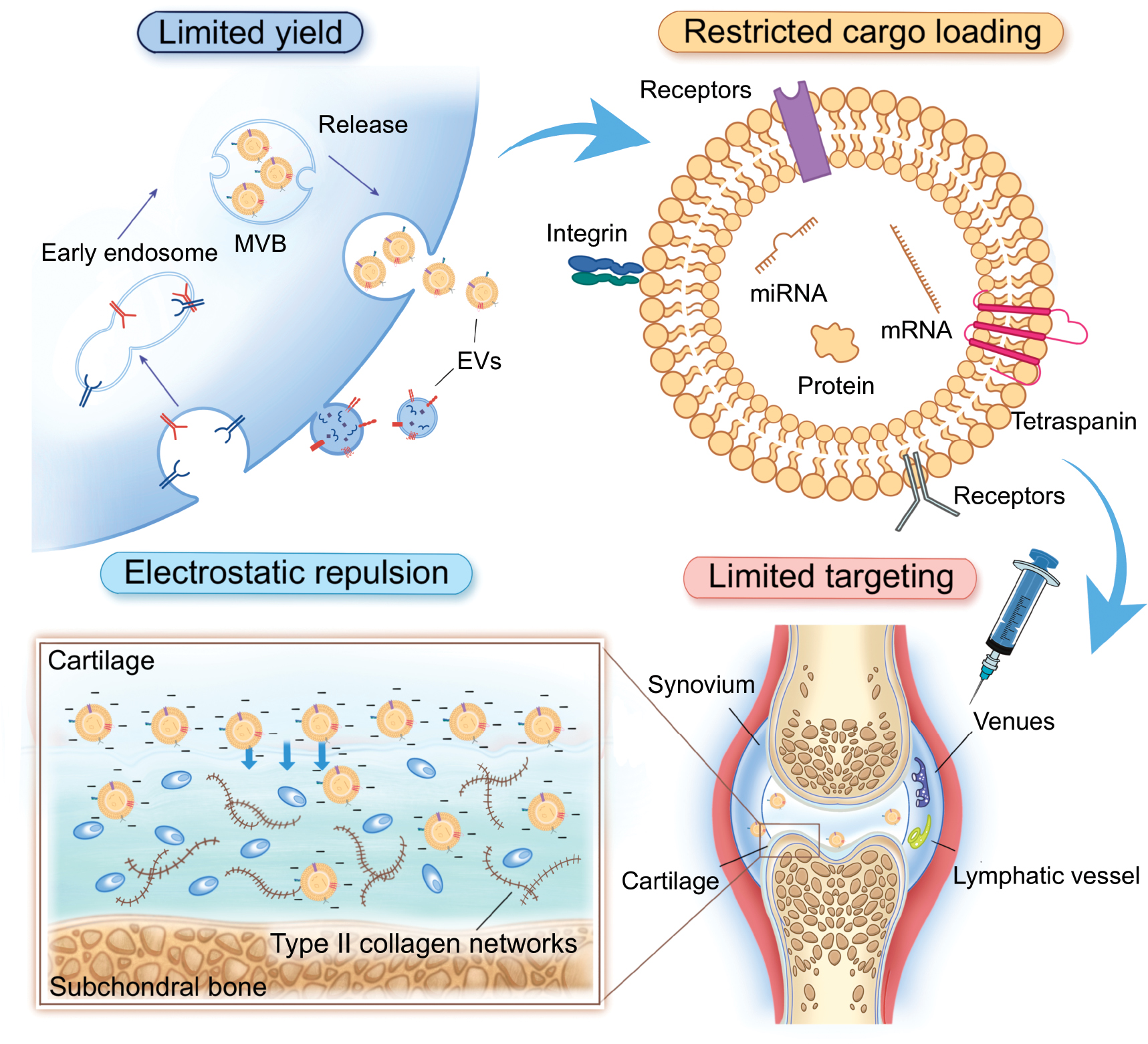
The biogenesis of EVs has been extensively studied and is generally classified based on the origin from the endosomal system or the plasma membrane [30–32]. In brief, EVs derived from the endosomal pathway are formed through the inward budding of the plasma membrane to generate endocytic vesicles, which subsequently fuse to form early endosomes. Mediated by systems, such as the endosomal sorting complex required for transport (ESCRT) [32], early endosomes encapsulate intracellular components to generate multiple intraluminal vesicles (ILVs). During this process, early endosomes mature into late endosomes, also known as multivesicular bodies (MVBs). Finally, MVBs fuse with the plasma membrane and release EVs into the extracellular space. In contrast, EVs originating from the plasma membrane are formed by direct outward budding (Figure 1). Commonly used methods for EV isolation include ultracentrifugation, ultrafiltration, and size-exclusion chromatography [33–36]. These approaches have been widely discussed in previous reviews and are not the focus of this article.
Figure 1 Limitations of native EVs in OA therapy. Native EVs face multiple barriers in OA treatment, including limited yield, restricted therapeutic cargo, electrostatic repulsion from the negatively charged cartilage matrix, and insufficient cartilage targeting, which together constrain the therapeutic effectiveness.
Although EV biogenesis and isolation have been well-studied, the EV production yield remains low. This is a major limitation for therapeutic applications, especially for degenerative diseases, like OA [37]. In most studies the number of EVs collected is ≤ 109 particles/mL of culture medium. However, effective doses in animal studies often exceed 1010 particles/g of mouse tissue [18]. This production gap limits the possibility of using EVs for clinical treatment of OA.
EV size and surface charge
Size is one of the most fundamental properties of EVs. According to MISEV2023 recommendations, EVs with a diameter > 200 nm are commonly referred to as large EVs (lEVs), while EVs < 200 nm in size are designated small EVs (sEVs) [9]. Although MISEV2023 does not define strict upper or lower size limits, observations have shown that sEVs and lEVs typically fall within the ranges of 30–200 nm and 200–300 nm, respectively, due to structural stability. Because of the higher biological penetrability, especially the ability to cross the blood–brain barrier, sEVs have been widely used in disease-related therapeutic research [38–40]. In contrast, lEVs are often taken up by the lungs, liver, or spleen when administered systemically, cleared by macrophages, or fail to penetrate vascular barriers, thereby limiting the biological effects [41–43]. However, lEVs under local injection remain of great interest and have potential for further exploration [44].
Surface charge is another basic characteristic of EVs. During biogenesis, whether derived from the endosomal pathway or directly from the plasma membrane, EVs inherit membrane components from cellular organelles or the cell surface [30, 45]. As a result, most EVs carry a negative surface charge. A stronger negative charge increases electrostatic repulsion between EVs, making EVs less likely to aggregate [46]. This feature improves EV stability during application and storage.
Most EVs are administered through intra-articular injection during OA therapy, which does not require crossing systemic biological barriers. However, efficient therapeutic delivery remains difficult due to absorption by the synovium and the dense cartilage structure [47, 48]. The cartilage matrix is composed of type II collagen networks filled with dense and negatively charged aggrecan proteoglycans [49]. The matrix contains only very small pores (<6 nm in diameter) for molecular diffusion [50]. Although some studies have shown that EVs may pass through the extracellular matrix by undergoing deformation mediated by aquaporins [51], the ability to do so is limited. The cartilage matrix is dense and negatively charged; EVs also carry a negative surface charge (Figure 1). This charge similarity leads to electrostatic repulsion, which further reduces the efficiency of EV penetration [52, 53]. As a result, EVs often fail to reach deep-layer chondrocytes and cannot exert therapeutic effects effectively in these regions.
Membrane
The EV membrane has a critical role in preserving vesicle integrity, shielding internal cargo, and enabling communication with recipient cells. Structurally, the EV membrane is composed of a lipid bilayer enriched with components, such as cholesterol, sphingomyelin, and phosphatidylserine, along with a variety of membrane-associated proteins [45]. These constituents provide mechanical rigidity and chemical stability, allowing EVs to remain intact in the extracellular environment and resist degradation by enzymes present in biological fluids [54]. In addition to serving as a protective barrier, the EV membrane also participates actively in biological signaling. The EV membrane displays multiple surface markers, including tetraspanins (CD9, CD63, and CD81), integrins, and adhesion molecules, which contribute to processes, such as cellular recognition, membrane docking, and internalization [55–58]. These surface features are essential for determining how EVs interact with different cell types and tissues.
However, the native membrane composition of EVs often lacks precise targeting ability. In the context of OA, the lack of precise targeting ability represents a major limitation (Figure 1). EVs may be taken up by non-target cells within the joint cavity after intra-articular injection or diffuse away from the joint due to the absence of specific affinity for some cells or tissues [47]. Notably, it has been reported that EVs secreted by stem cells and other anti-inflammatory cell types may inherit adhesive molecules and exhibit some degree of tropism toward inflamed regions [59]; this effect remains limited in OA. In most cases, multiple joint tissues are simultaneously inflamed and EVs entering the joint cavity cannot selectively target articular cartilage. This lack of specificity reduces the overall therapeutic efficacy of native EVs in OA treatment.
Cargo
The cargo carried by EVs is the key to the biological activity. EVs can transport a wide variety of bioactive molecules, including proteins, lipids, mRNAs, microRNAs, and other non-coding RNAs. These components reflect the physiologic state of the parental cells and can influence gene expression, signal transduction, and cellular behavior in recipient cells [11]. The incorporation of cargo into EVs is not a random process. During EV biogenesis, specific sorting mechanisms selectively package molecules into vesicles. In the endosomal pathway, cargo loading is regulated by protein complexes, such as the ESCRT machinery, which recognizes ubiquitinated proteins and guides inclusion into ILVs. In addition to ESCRT-dependent pathways, other mechanisms involving tetraspanins, lipid rafts, and RNA-binding proteins also contribute to selective cargo sorting [32]. EVs formed by direct budding from the plasma membrane may incorporate cytosolic components or membrane proteins based on local lipid and protein interactions.
In the context of OA therapy, EV cargo has a central role in regulating inflammation, cartilage metabolism, and tissue repair. For example, some microRNAs within EVs have been shown to inhibit catabolic enzymes or promote the expression of anabolic factors in chondrocytes [60]. Proteins may also support cartilage protection and regeneration [29]. However, the therapeutic potential of native EVs is limited by the heterogeneity and complexity of the cargo. Most EVs carry a broad mixture of molecules but only a small portion may contribute to the treatment of OA. Moreover, EVs lack the ability to actively sort or enrich specific therapeutic molecules (Figure 1). This attribute limits the precision and efficiency of natural EVs as delivery vehicles. A more controlled and targeted cargo composition is desirable for clinical application in OA to improve treatment efficacy and safety.
Methodologies for EV engineering
Although many studies have demonstrated that native EVs exert beneficial effects in the treatment of OA, several challenges remain before clinical application. As summarized above, these challenges include insufficient yield, electrostatic repulsion, poor targeting ability, and a limited number of therapeutic molecules within the vesicles. Such general limitations in clinical translation have stimulated extensive research on EV modification and given birth to the field of EVs engineering.
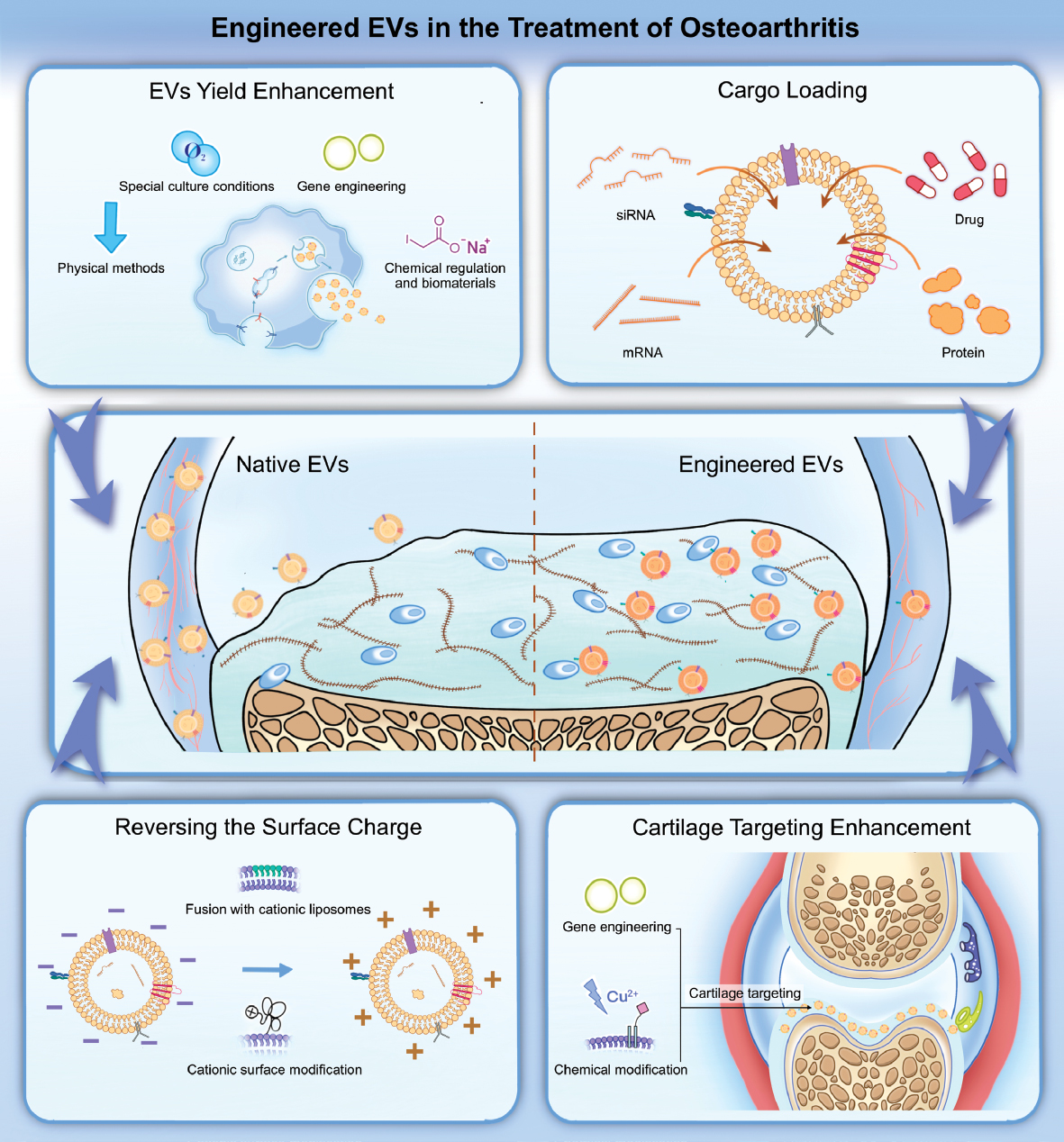
EV engineering refers to the artificial modification of naturally secreted EVs using biological, chemical, or physical methods to enhance or endow the EVs with new functions required for therapeutic use [61]. Importantly, these engineering approaches typically do not disrupt the fundamental structure of EVs, such as the nanoscale size and lipid bilayer membrane. The inherent properties, including low immunogenicity and barrier penetration capability, are usually preserved after modification. Based on the major challenges that EVs face before clinical use in OA, EV engineering strategies are classified into four main categories: enhancing EV yield altering surface charge; modifying the EV membrane; and loading therapeutic cargo.
EV yield enhancement
Physical methods
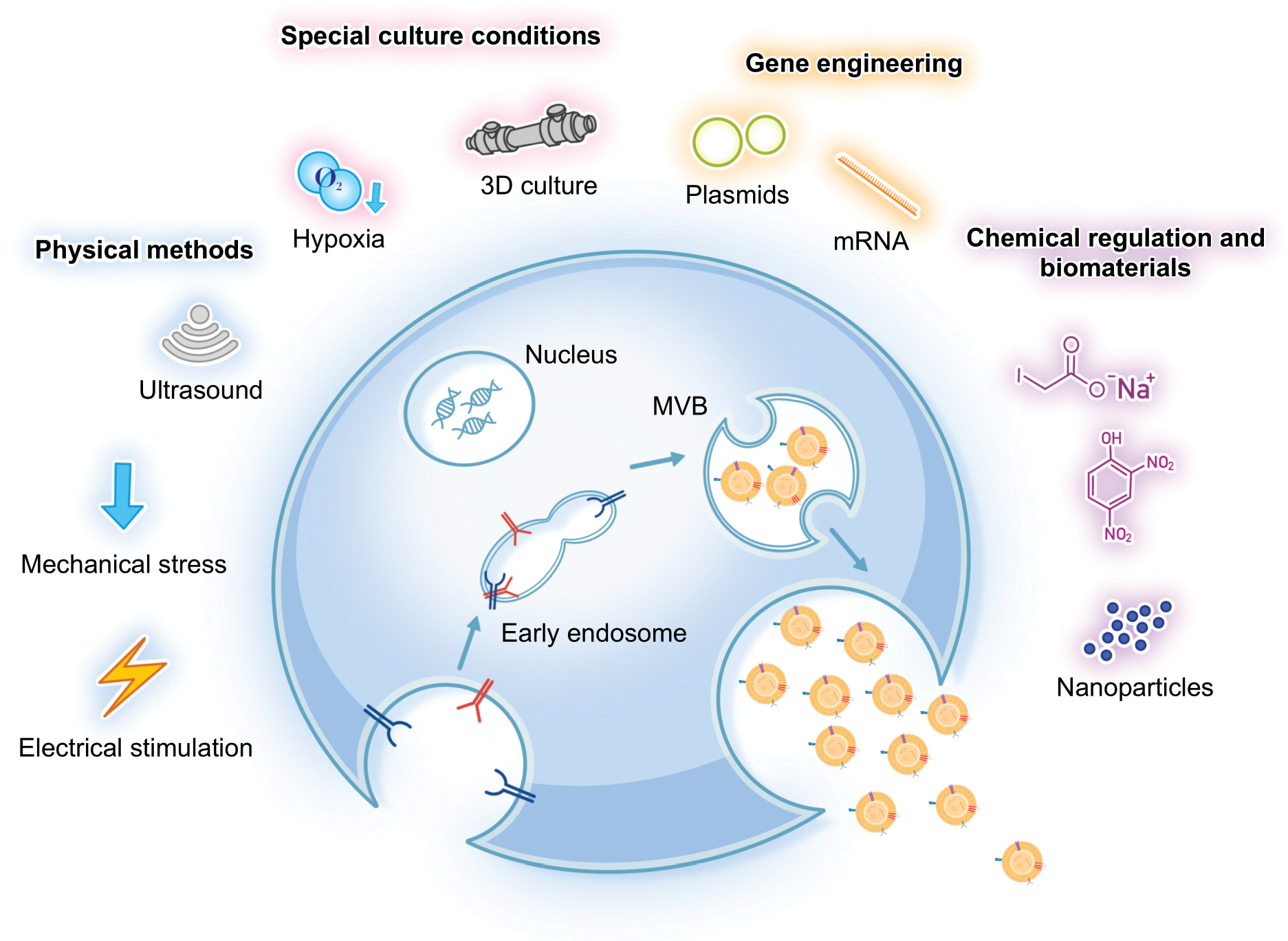
Physical stimulation methods, such as mechanical stress, ultrasound treatment, and electrical stimulation, have been shown to influence EV yield (Figure 2). Mechanical stress can affect receptors and integrins on the plasma membrane that respond to tension, thereby regulating gene expression in cells [62]. Similar effects have been observed in EV secretion, in which mechanical stimulation acts as a promoting factor. For example, cyclic tensile stress has been reported to increase EV secretion from periodontal ligament cells by > 20-fold [63]. Guo et al. also reported that stretching skeletal muscle cells increases EV yield by 11-fold and identified yes-associated protein (YAP) as a key mechanosensitive mediator in this process [64].
Figure 2 Strategies for yield enhancement of EVs. This figure illustrates major approaches to increase EV production, including physical stimulation (mechanical stress, ultrasound, and electrical stimulation), special culture conditions (hypoxia and 3D culture), gene engineering, and chemical regulation or biomaterials. These strategies enhance EV biogenesis and secretion by modulating cellular signaling pathways and vesicle release.
Ultrasound, a technique widely applied in medical practice, has more recently been implicated in promoting EV production. The principle is similar to mechanical stimulation but ultrasound generates multiple physical effects, including heat, pressure, shockwaves, microjets, and shear stress, making the underlying mechanisms more complex [65]. Yang et al. reported that low-intensity pulsed ultrasound (LIPUS) significantly induces the expression of EVs-associated proteins, such as Rab11 and STX6. Following LIPUS treatment, myeloid-derived suppressor cells, mesenchymal stem cells, and immunosuppressive dendritic cells exhibited increased EV secretion [66].
Electrical stimulation, a common approach used in cell fusion and nucleic acid transfection, has also been shown to promote EV release. Fukuta et al. reported that low-level electrical stimulation (0.3–0.5 mA/cm2) could activate intracellular signaling pathways and enhance subsequent endocytosis of foreign molecules, thereby promoting EVs secretion [67]. Yang et al. developed a cellular nanoporation (CNP) biochip, which transfected plasmid DNA into donor cells and applied brief electrical pulses to stimulate EV release [68]. Using this method, exosome production was increased up to 50-fold and the generated EVs contained target nucleotide sequences.
Taken together, these findings indicate that physical methods, such as mechanical stress, ultrasound, and electrical stimulation, can significantly enhance EV production. Each approach relies on different cellular responses, ranging from mechanosensitive signaling pathways-to-changes induced by thermal and pressure effects or electrical activation of intracellular processes. While these strategies demonstrate clear potential for boosting EV yield, the underlying mechanisms are not fully understood and may vary depending on cell type and stimulation parameters. Further studies are needed to determine how to optimize these methods for large-scale and standardized EV production. Importantly, the ability of physical stimulation to increase yield without substantially altering vesicle properties must be carefully evaluated because this will determine the safety and reliability for therapeutic applications, including OA treatment.
Special culture conditions
Special culture conditions, such as hypoxia and three-dimensional (3D) culture, have been shown to significantly promote EV yield. Hypoxic culture increases the release of EVs compared to normoxia and this effect has been reported in a wide range of cell types [69]. Hypoxia can upregulate EV-associated proteins involved in vesicle biogenesis and secretion, leading to enhanced production [70]. Although the exact mechanisms remain to be fully clarified, most studies consistently support hypoxia as a strong inducer of EV release.
Similarly, 3D-culture systems, including spheroid models, hydrogel scaffolds, and bioreactors, provide a more physiologically relevant microenvironment that promotes vesicle secretion [71]. 3D conditions mimic in vivo cell–cell and cell–matrix interactions compared to conventional two-dimensional (2D)-culture systems, which stimulate cellular communication and EV biogenesis. Studies have demonstrated that stem cells in 3D-culture systems produce a significantly higher number of EVs than 2D-cultures systems [72]. This enhancement is thought to result from changes in mechanical stress, nutrient gradients, and metabolic activity that more closely resemble the natural tissue environment. Together, hypoxic culture and 3D-culture systems represent promising approaches to increase EV yield. These strategies offer valuable tools for overcoming the limitation of low EV production and provide potential avenues for large-scale preparation of EVs for therapeutic applications.
Gene engineering for yield enhancement
Gene engineering has been explored as a strategy to enhance EV yield by modulating genes involved in vesicle biogenesis and release. Regulatory factors associated with EV biogenesis, including components of the endosomal sorting machinery and membrane remodeling pathways, such as HRS [73], STAM1 [74], TSG101 [75], CHMP4 [76], and STEAP3 [77], as well as regulators of EV release, including Rab2b [78], Rab5a [78], Rab27b [79], and Syndecan-4 [77], have been suggested to participate in these processes and are therefore considered candidate targets for genetic manipulation.
Experimental evidence supports the feasibility of this approach. For example, Kojima et al. reported that simultaneous overexpression of STEAP3 and Syndecan-4 markedly enhance EV secretion, resulting in an approximately 40-fold increase in EV yield [77]. However, these effects are often highly dependent on cell type and experimental context. The same gene may exert divergent or even opposing effects on EV production across different cellular systems. Such variability reflects the complexity and heterogeneity of EV biogenesis, in which many of these regulators also participate in broader cellular processes in addition to vesicle formation and trafficking [18]. Consequently, gene engineering has not yet provided a universal solution for achieving consistently high EV output, and most current strategies influence EV-associated pathways indirectly rather than through precise and predictable modulation of specific targets.
Chemical regulation and biomaterials
Chemical regulators modulate EV secretion through several interconnected mechanisms. Metabolic inhibitors, such as sodium iodoacetate (IAA) and 2,4-dinitrophenol (DNP), trigger cellular stress by simultaneously blocking glycolysis and oxidative phosphorylation, which depletes intracellular energy and activates adenosine–A2B receptor signaling, ultimately leading to a several-fold increase in vesicle release [80]. Small molecules identified through high-throughput screening, such as norepinephrine and N-methyldopamine, promote EV production via upregulation of ESCRT-independent regulators, such as nSMase2 [81]. Collectively, these compounds enhance vesicle yield by altering energy metabolism, intracellular trafficking, receptor-mediated signaling, and gene expression.
Biomaterials have also emerged as promising tools for boosting EV production. Engineered nanoparticles represent a key example. Specifically, positively charged iron oxide nanoparticles internalized by stem cells accumulate in lysosomes, reduce vesicle degradation, and increase secretion through Rab-dependent pathways [82]. Structural and mechanical properties of biomaterials also contribute significantly. Titanium surfaces engineered with micro–nano features enhance vesicle release through Rab and sphingomyelinase activation [83]. Furthermore, bioglass-derived ionic extracts upregulate regulators, such as nSMase2 and Rab27a, resulting in more than a 2-fold increase in EV yield from MSCs [84]. Together, these findings highlight that biomaterial cues, including surface charge, stiffness, and structural architecture, offer effective strategies to overcome low EV production and support scalable preparation for therapeutic applications.
Although chemical regulators and biomaterials have shown promise in increasing EV yield, the effects are often cell type–specific and sometimes inconsistent. Most studies also emphasize short-term outcomes, leaving uncertainties about long-term stability and reproducibility. These limitations suggest that further optimization and standardization are needed before these strategies can be applied in large-scale EV production.
Surface charge modification
As summarized earlier, the membrane potential of native EVs is typically negative. This intrinsic surface charge can reduce cellular uptake in some tissues due to electrostatic repulsion, thereby limiting the overall utilization. To overcome this limitation, several strategies have been developed to shift the surface potential of EVs toward positive values, thereby reducing repulsive interactions and improving delivery efficiency (Figure 3). Current methods mainly include the introduction of cationic groups and fusion with positively charged liposomes [85–87].
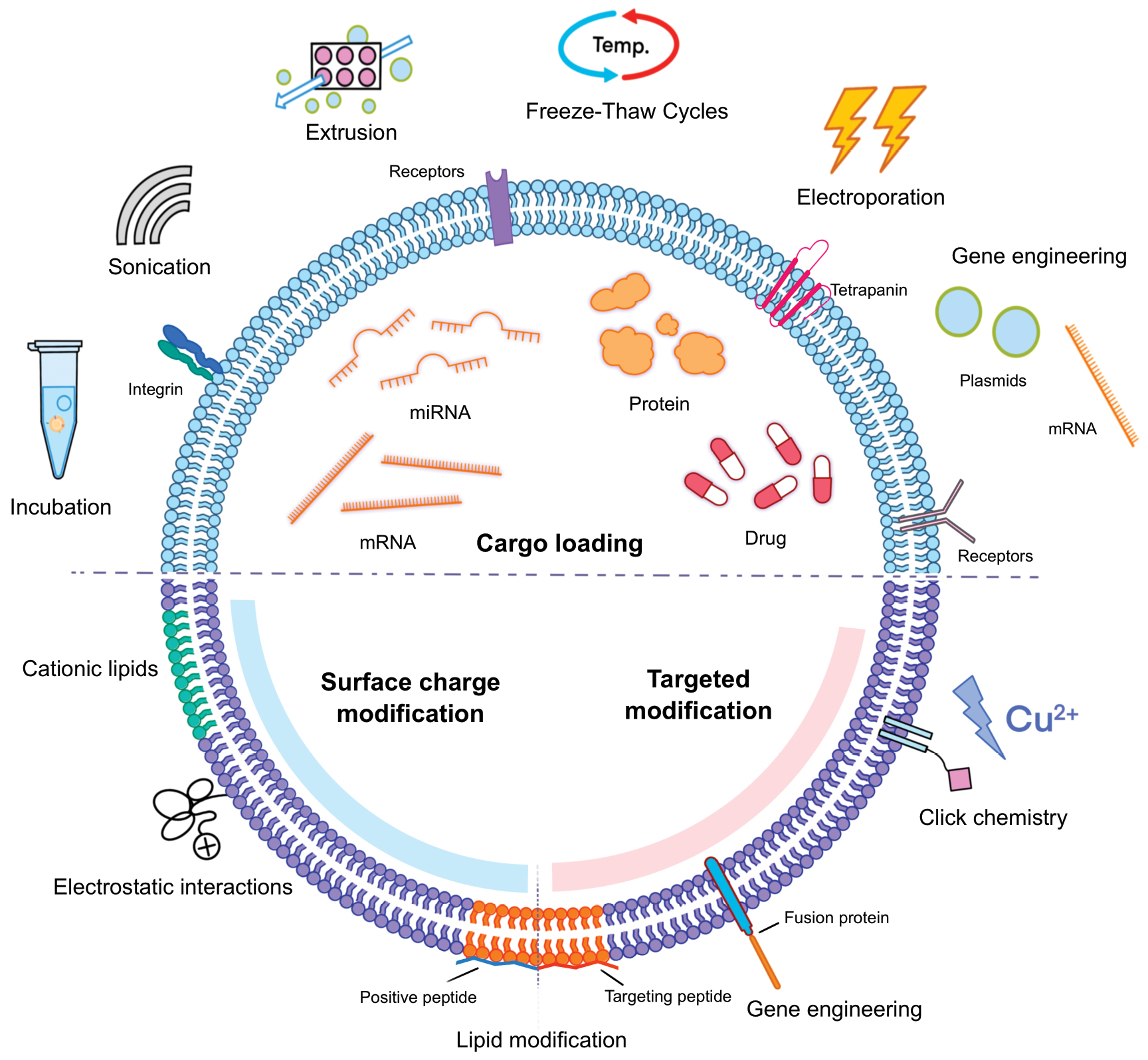
Figure 3 Strategies for EVs cargo loading, membrane engineering, and surface charge modification. This figure summarizes representative approaches for EVs engineering, including cargo loading, membrane engineering, and surface charge modification. Cargo loading strategies include incubation, sonication, extrusion, freeze-thaw cycles, electroporation, and gene engineering to incorporate nucleic acids, proteins, or drugs into EVs. Membrane engineering focuses on modifying EVs membranes through gene engineering or chemical modification, such as lipid insertion, fusion proteins, and click chemistry, to display targeting ligands or receptors and enhance delivery specificity. Surface charge modification aims to regulate EV surface potential by introducing cationic lipids, positively charged peptides, or electrostatic interactions, thereby reducing electrostatic repulsion and improving cellular uptake.
Cationic surface modification
One approach involves introducing positively charged groups directly onto the EV membrane. This can be achieved by inserting cationic moieties into the lipid bilayer or by coating EVs with cationic polymers. For example, distearoyl phosphatidylethanolamine (DSPE) has been conjugated with ε-polylysine or cationic peptides and incubation with EVs produces engineered vesicles with a positive surface potential [52, 86]. Chen et al. developed an esterase-responsive charge-reversal cationic polymer that when incubated with EVs generates vesicles with a zeta potential of approximately +20 mV [88]. Unlike direct insertion, this approach relies on polymer adsorption to the EV surface, which effectively alters the overall surface charge. However, the reversibility of polymer binding raises concerns about stability in complex biological environments and further evaluation is required to ensure consistency.
Fusion with cationic liposomes
Another strategy uses cationic liposomes to adjust the EV surface charge. Sun et al. reported that incubating EVs with positively charged liposomes shifts the zeta potential from −6.2 mV to +0.5 mV. Although this method is relatively simple, fusion with liposomes significantly alters vesicle size and cargo composition [85]. Whether these changes affect the therapeutic performance or biosafety of engineered EVs remains unclear and requires further investigation.
Although surface charge modification offers a direct and effective means to improve EV uptake, several challenges remain. Both polymer-based adsorption and liposome fusion approaches can successfully shift the surface charge but may also compromise vesicle stability, reproducibility, or structural integrity. Specifically, polymer binding may be unstable in vivo, while liposome fusion can introduce undesirable alterations to EV size and content. Therefore, while this strategy holds promise, future studies must focus on developing stable, biocompatible, and reproducible charge-modification techniques that enhance uptake without compromising the safety or reliability of EVs for therapeutic applications.
Membrane engineering
Like cellular membranes, the EV membrane consists of a phospholipid bilayer enriched with lipids, proteins, and glycans. These components provide the structural basis for membrane modification. The primary aim of engineering EV membranes is to enhance or impart specific targeting capabilities toward desired cells or organs [89]. Current strategies for membrane engineering can be broadly divided into two categories (gene engineering and chemical modification; Figure 3).
Gene engineering for membrane functionalization
Gene engineering provides an effective strategy for customizing EV membranes to enhance targeting capacity and therapeutic potential. The general principle involves introducing genetic constructs into donor cells so that specific peptides, receptors, or fusion proteins are expressed on the EV surface [90]. For example, Ben et al. used lentiviral transduction to overexpress CD47 in MSCs, thereby generating EVs enriched in CD47 [91].
However, the process of protein expression is complex and it is difficult to ensure that proteins other than membrane-associated proteins can be stably displayed on the EV surface. To address this limitation, many studies have fused the coding sequences of target proteins or peptides with the coding sequences of endogenous membrane proteins, thereby achieving stable expression on EV membranes [89, 92]. For example, Alvarez-Erviti et al. fused the sequence of the RVG peptide with the sequence of the membrane protein, Lamp2b, and transfected the construct into donor cells via plasmids [93]. The resulting EVs carried abundant targeting peptides on the surface and exhibited strong tropism for the hippocampal region in animal experiments, highlighting the potential as a therapeutic option for Alzheimer’s disease.
Together, these examples demonstrated that genetic engineering provides a versatile toolkit for customizing EV membranes. By selecting appropriate ligands and membrane anchors, donor cells can be programmed to release EVs with improved tissue tropism, enhanced stability, or reduced immune clearance. However, this approach also has inherent limitations because only proteins or peptides can be displayed on the EV surface, restricting applicability in broader contexts.
Chemical modification
Chemical modification provides a practical approach to engineer EV membranes after isolation, enabling the addition of new properties without altering donor cells. This strategy relies on covalent or non-covalent interactions between functional molecules and components of the EV membrane [94–96]. A wide range of moieties can be introduced, including peptides, antibodies, polymers, and small chemical groups, to enhance targeting capacity or prolong circulation time.
Click chemistry has become one of the most widely applied covalent methods. This bio-orthogonal reaction forms stable triazole linkages between azide and alkyne groups and can proceed rapidly under mild, aqueous conditions with high efficiency and selectivity, causing minimal disruption to the membrane [97]. Click chemistry is particularly suited for EV surface functionalization owing to the biocompatibility [98, 99]. For example, Hao et al. used this technique to conjugate a collagen-binding peptide (SILY) to MSC EVs, which markedly improved retention at injury sites and demonstrated promise in tissue repair [100].
Non-covalent modification represents a complementary approach. Hydrophobic insertion allows lipid-conjugated ligands to spontaneously embed into the phospholipid bilayer of EVs, providing a simple and efficient means of surface engineering [101–103]. Similarly, electrostatic adsorption of cationic polymers or peptides can decorate the EV surface, although the stability of such modifications may be influenced by physiologic conditions [88]. Chemical modification offers greater flexibility compared to genetic engineering because chemical modification can be applied to a broad range of EV sources and adjusted according to therapeutic needs. However, potential drawbacks include reduced stability, alteration of vesicle integrity, and variability in modification efficiency, all of which require further optimization.
Cargo loading
Engineering EV cargo is an essential strategy to broaden therapeutic applications. Enhancing delivery efficiency and tailoring biological functions become possible by incorporating drugs, nucleic acids, proteins, or other bioactive molecules into EVs (Figure 3). Several loading methods have been developed, each with distinct mechanisms and efficiencies. These loading methods include incubation, extrusion, sonication, freeze–thaw cycles, electroporation, and gene engineering [104]. The choice of method depends on the physicochemical characteristics of the intended cargo, as well as the balance between efficiency, integrity, and practicality [105].
Incubation
The incubation method is one of the most straightforward techniques for EV cargo loading. The incubation method relies on passive diffusion and hydrophobic interactions between cargo molecules and the EV lipid bilayer. This approach is particularly suitable for small hydrophobic drugs. For example, curcumin and paclitaxel have been successfully incorporated into EVs using simple co-incubation, enabling effective delivery [106, 107]. While the method is easy to implement and preserves EV integrity, the loading efficiency is generally low for hydrophilic molecules and optimization of incubation conditions, such as temperature and concentration, is often necessary.
Sonication
Sonication uses ultrasonic waves to generate transient pores in EV membranes, enabling cargo molecules to enter the vesicle interior [108]. Sonication typically achieves higher loading efficiency compared to incubation, making sonication particularly useful for drugs and nucleic acids. However, overexposure to ultrasonic energy can damage vesicle integrity and affect the biological properties. Careful optimization of sonication intensity and duration is therefore required to maximize efficiency while maintaining vesicle stability [109].
Extrusion
Extrusion involves repeatedly forcing a mixture of EVs and cargo molecules through membranes with defined pore sizes. This mechanical process temporarily disrupts the vesicle membrane, allowing external molecules to be encapsulated as the membrane reforms [110]. Extrusion provides relatively uniform cargo distribution and is suitable for a broad range of molecules. However, the high shear forces involved may compromise EV stability or alter surface proteins [111]. Despite these drawbacks, extrusion remains an important option for achieving efficient incorporation of larger or more structurally complex molecules.
Freeze–thaw cycles
The freeze–thaw method is a simple mechanical strategy for loading cargo into EVs. The vesicle membrane is temporarily disrupted and allows molecules to enter before resealing by repeatedly freezing mixtures of EVs and target molecules, often in liquid nitrogen, then thawing at room or physiologic temperature [112]. Studies have shown that this technique can achieve acceptable encapsulation while maintaining vesicle integrity, such as in loading of human chorionic gonadotropin (hCG) [113]. However, the efficiency is generally lower than sonication or extrusion and repeated cycles may cause protein denaturation, aggregation, or size heterogeneity.
Electroporation
Electroporation is a widely used technique for loading hydrophilic molecules into EVs. Transient pores are formed in the EVs membrane by applying short electrical pulses, which allows nucleic acids or other water-soluble molecules to diffuse into the vesicle lumen. This approach has been frequently used to encapsulate siRNA, miRNA, and other nucleic acids, enabling efficient delivery for therapeutic applications [114–116]. For example, Liu et al. successfully used electroporation to load BCL6 siRNA into EVs modified with iRGD peptides [117]. The engineered vesicles inhibited the proliferation of diffuse large B-cell lymphoma cells in vitro, highlighting the potential for targeted cancer therapy. Although electroporation achieves high loading efficiency and is relatively straightforward to perform, challenges remain, including vesicle aggregation, partial loss of surface protein activity, and the need for specialized equipment [118].
Gene engineering for cargo loading
Gene engineering provides a unique strategy for cargo loading by programming donor cells to incorporate specific molecules into EVs during natural biogenesis. This approach enables stable and sustained incorporation of therapeutic proteins or nucleic acids into vesicles, unlike physical or chemical methods that introduce cargo after EVs isolation [119, 120]. This strategy underscores the potential of gene engineering to generate customized EVs with precise molecular payloads. However, several challenges remain, such as ensuring biosafety, achieving scalable production, and maintaining reliable control of expression [121]. Overcoming these barriers will be essential for successful clinical translation.
Applications of engineered EVs in OA therapy
The major strategies currently used for EVs engineering were summarized in the previous sections, with an emphasis on the general principles and technical characteristics. While these approaches provide a broad methodologic toolbox, the relevance and effectiveness can vary substantially depending on the disease context. Therefore, to bridge engineering concepts with clinical relevance, the following section focuses specifically on how engineered EVs have been applied in OA therapy. Representative preclinical studies are highlighted to illustrate how different engineering strategies are tailored to address OA-specific pathologic features and therapeutic demands (Figure 4).
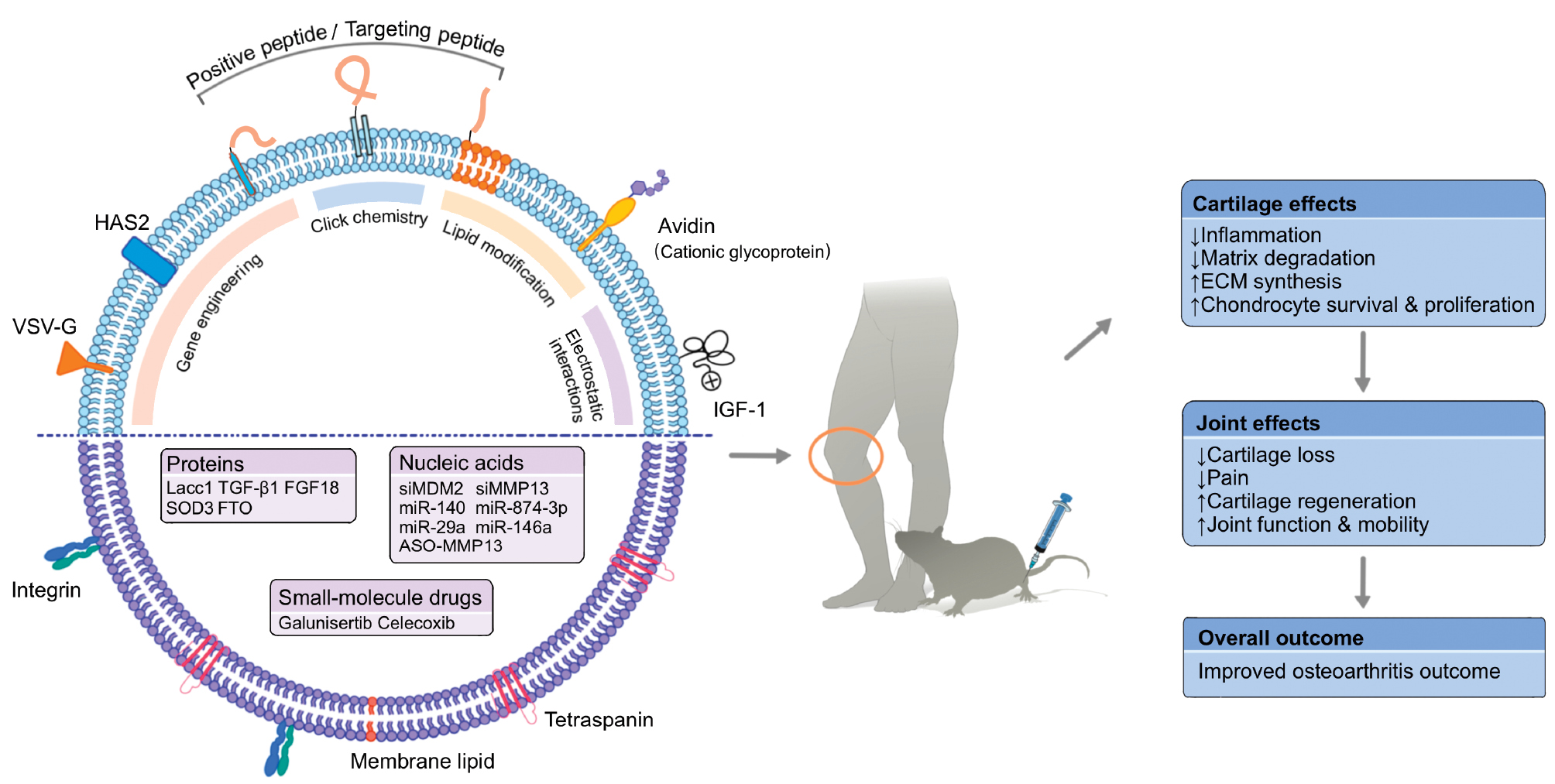
Figure 4 Applications of engineered EVs and the therapeutic outcomes in OA. This schematic illustrates how engineered EVs are tailored to address OA-specific pathologic features and therapeutic needs. EVs are modified through cargo loading, membrane engineering, and surface charge modification to incorporate proteins, nucleic acids, or small-molecule drugs and to enhance cartilage targeting via peptides, fusion proteins, or chemical conjugation. Following intra-articular delivery, engineered EVs act on cartilage and joint tissues to reduce inflammation, inhibit matrix degradation, promote extracellular matrix synthesis, and support chondrocyte survival and proliferation. Collectively, these effects alleviate cartilage loss and pain, enhance cartilage regeneration and joint function, and ultimately improve OA outcomes, highlighting the translational potential of EV engineering strategies in OA therapy.
EVs yield optimization in OA therapy
As described earlier, the basal yield of EVs is often insufficient to fully meet the requirements of clinical therapy. Various strategies have been developed to address this limitation, including physical stimulation, specialized culture conditions, gene engineering, chemical regulation, and biomaterial-based approaches. However, only some these methods have been broadly adopted in the context of OA. Among the methods, 3D culture and hypoxic conditioning are the most widely applied, while some physical and chemical approaches have also been explored for the potential to increase EV yield in OA therapy.
3D systems provide a spatial environment that better mimics in vivo conditions compared to conventional 2D culture and supports robust cell proliferation. Fu et al. reported that MSCs proliferate extensively under 3D culture, reaching a cell count of 6.14 × 108 after 10 d [122]. The corresponding EV yield, obtained by ultracentrifugation, was approximately 1010 particles/mL, providing a substantial source for therapeutic applications or further engineering. Importantly, subsequent studies revealed that 3D culture not only enhances EV production but also alters the biological functions. Yang et al. demonstrated that MSC-EVs produced under 3D conditions exhibit a 2.5-fold increase in yield and exhibit superior effects in promoting cartilage repair and suppressing inflammation [123]. They further suggested that these effects may be associated with the differential accumulation of non-coding RNAs, including miR-181b-5p, miR-664a-5p, and miR-4491, although the underlying mechanisms remain unclear.
Similarly, hypoxic treatment, a commonly used method to promote paracrine activity, has also been shown to increase EV yield and therapeutic function. Zhang et al. observed that EVs derived from hypoxia-preconditioned MSCs promote cartilage repair more effectively in OA models [124]. Transcriptomic analysis further suggested that enrichment of miR-122-5p in hypoxic MSC-EVs may mediate this effect by modulating autophagy through DUSP2 in chondrocytes. Although the selective enrichment of miRNAs in hypoxia-derived EVs has been reported by other studies as well, the exact mechanism driving this phenomenon remains poorly understood.
In addition to these widely used methods, physical stimulation, such as electromagnetic or electrical fields, has also been investigated. Lin et al. applied nanosecond-pulsed electric fields (nsPEFs) to MSCs, resulting in a 5.77-fold increase in EV production [125]. Wang et al. treated MSCs with an electromagnetic field (EMF) combined with ultrasmall superparamagnetic iron oxide (USPIO) particles, which also promoted EV release [126]. Interestingly, like EVs produced under 3D or hypoxic conditions, these electrically stimulated EVs also exhibited enhanced therapeutic effects in OA models, potentially mediated by the miRNA-99b/MFG-E8/NF-κB signaling axis.
Collectively, current evidence indicates that methods designed to increase EV yield often alters the biological activity. This effect is frequently attributed to changes in cellular metabolism and molecular cargo induced by stimulation, although different studies have reported inconsistent molecular signatures (Table 1). The precise mechanisms underlying this selective enrichment remain to be elucidated. Nevertheless, these findings suggest that systematic evaluation of the therapeutic performance of EVs generated through yield-enhancing strategies may uncover new opportunities for OA treatment.
Table 1 Research Examples of Source Cell Processing Strategies in Osteoarthritis Therapy
| Source of EVs | Processing Method | Observed Changes in EVs | Biological Functions | References |
|---|---|---|---|---|
| MSC | Cytokine priming with IFN-γ, IL-6, and TNF-α | Altered protein composition with enhanced immunoregulatory profile | Promoted macrophage polarization toward M2 phenotype and suppressed OA-related inflammation via mitochondrial metabolism modulation | [154] |
| MSC | Mechanical loading priming | Enriched with miR-27b-3p | Attenuated chondrocyte senescence and ECM degradation by regulating the ROR1/NF-κB signaling pathway, thereby mitigating OA progression | [136] |
| MSC | Inflammatory/fibrotic + oxytocin priming | Enrichment of miRNAs linked to immune regulation, tissue repair, and anabolic signaling | Promoted M2 macrophage polarization and exerted chondroprotective effects in inflammatory OA models | [155] |
| MSC | TNF-α preconditioning | Increased EVs secretion via PI3K/AKT pathway activation and ATG16L1 upregulation; elevated LRP1 expression in EVs | Ameliorated OA pathology and gait abnormalities in vivo; exerted chondroprotective effects by maintaining cartilage homeostasis | [156] |
| MSC | EMF stimulation combined with USPIO particles | Increased EVs yield; enrichment of miR-99b | Promoted anabolic activity and inhibited catabolic processes; enhanced M2 macrophage polarization; alleviated OA progression and supported cartilage regeneration | [126] |
| MSC | Nanosecond-pulsed electric field pretreatment | Increased EVs yield | Inhibited OA-like chondrocyte development, enhanced MSC viability and migration, and prevented OA progression in rat models | [125] |
| MSC | Hypoxia preconditioning | Enrichment of miR-122-5p | Promoted chondrocyte proliferation and migration via autophagy, regulated by the miR-122-5p/DUSP2 pathway, reduced OA progression | [124] |
| BMSC | PTH | Enrichment of miRNA let-7a-5p in EVs | Promoted chondrocyte proliferation and migration, inhibited apoptosis, and delayed OA progression by suppressing IL-6 expression | [157] |
| BMSC | Quercetin precondition | Enrichment of chondroprotective and anti-inflammatory cargo | Reduced IL-1β-induced inflammation, restored cartilage repair genes, and preserved cartilage integrity in OA models | [158] |
| UCMSC | IL-1β priming | Increased EVs secretion; enrichment of anti-inflammatory miRNAs and signaling pathway modulation | Promoted chondrocyte function and cartilage matrix production, enhanced macrophage polarization, improved inflammatory microenvironment, and supported cartilage regeneration in OA models | [159] |
| Synovial MSCs | Strontium pretreatment | Increased EVs yield; altered miRNA profile with higher enrichment of protective miRNAs (e.g., miR-143-3p) and reduction of harmful ones | Mitigated chondrocyte ferroptosis; reduced osteoclast-mediated joint pain; improved therapeutic efficacy | [160] |
Charge-engineered EVs in OA therapy
In the context of OA therapy, modification of EV surface charge is primarily achieved by introducing positively charged groups. Arginine and lysine, two natural polar amino acids carrying positive charges, are ideal candidates for this purpose. Zhang et al. used lipid insertion combined with click chemistry to anchor arginine- and lysine-rich short cartilage-targeting peptides onto milk-derived EVs [127]. This strategy shifted the zeta potential of EVs from −25.4 ± 1.1 mV to −2.5 ± 1.0 mV, enabling the vesicles to penetrate the full depth of mouse cartilage. Similarly, Pathrikar et al. applied lipid insertion to modify milk-derived EVs with the cationic glycoprotein, avidin (Av), or an arginine-rich peptide carrier (CPC) [128]. Both approaches successfully increased the surface potential from −27.9 ± 3.0 mV to −2.3 ± 0.8 mV and −1.6 ± 1.1 mV, respectively. Feng et al. further reported that the cartilage-targeting peptide WYRGRL exhibits a net positive charge [86]. By inserting this peptide into MSC-derived EV membranes, they shifted the surface potential from −17.23 ± 2.42 mV to +9.13 ± 1.31 mV. By acquiring a positive surface potential, these charge-engineered EVs reduced electrostatic repulsion with cartilage tissue and improved targeting as well as therapeutic performance.
In addition to lipid insertion of positively charged peptides, electrostatic adsorption has also been explored. Kim et al. co-incubated insulin-like growth factor-1 (IGF-1), a positively charged protein, with EVs derived from umbilical cord MSCs [87]. The modification was achieved through natural electrostatic attraction, resulting in EVs with a positive surface potential and improved affinity for damaged cartilage tissue. Although this non-covalent approach effectively altered surface charge, the long-term stability has not been systematically evaluated, which remains a critical consideration for preclinical applications.
Together, these studies demonstrated that engineering the surface charge of EVs is an effective strategy to enhance the therapeutic potential in OA. Positively charged groups are most often introduced through cationic amino acids, peptides, or proteins. Among the positively charged groups, peptides are particularly attractive due to the ease of synthesis and flexibility of application. However, only a small subset of potential peptide candidates has been explored to date, leaving significant opportunities for discovery. Further systematic screening of peptide-based carriers may help expand the utility of charge-engineered EVs. At the same time, the intra-articular environment in OA is complex, with many negatively charged macromolecules [129]. It is therefore important to determine if positively modified EVs can be non-specifically sequestered. Addressing this issue will be essential to fully assess the therapeutic value.
Membrane-engineered EVs in OA therapy
EVs are often administered by intra-articular injection in OA therapy. Although this approach avoids non-specific capture in distant organs, as occurs with systemic delivery, retention and utilization within the joint remain limited due to rapid clearance by the synovium [48]. Researchers have explored membrane engineering strategies to enhance targeting toward specific tissues or cells given the modifiable nature of the EV membrane, thereby improving therapeutic efficacy (Table 2). As summarized earlier, approaches to EV membrane modification can be broadly divided into genetic engineering and chemical modification.
Table 2 Research Examples of Membrane-Engineered EVs in OA Therapy
| Source of EVs | Engineering Methods | Modified Molecule | Modification Strategy | Biological Functions | References |
|---|---|---|---|---|---|
| DC | Genetic engineering | CAP | Genetic fusion of CAP with Lamp2b on EV membrane | Enhanced cartilage targeting, improved therapeutic efficiency in OA | [131] |
| HEK293T | Genetic engineering | VSV-G, HAS2 | Overexpression of VSV-G and HAS2 on EV membrane | Promoted membrane fusion, increased therapeutic efficacy in OA models | [132] |
| UCMSC | Genetic engineering | WYRGRL | Genetic fusion of WYRGRL with Lamp2b on EV membrane | Enhanced cartilage affinity and prolonged intra-articular retention | [161] |
| UCMSC | Chemical modification | CAP | Click chemistry coupling | Stronger cartilage targeting, prolonged intra-articular retention | [133] |
| Expi293F | Chemical modification | CAP | Sortase A enzyme-mediated coupling | Improved binding affinity to chondrocytes | [134] |
| Expi293F | Chemical modification | CAP | Lipid insertion (DSPE-PEG-MAL anchor) | Enhanced cartilage affinity and improved intra-articular retention | [162] |
| UCMSC | Chemical modification | CAP, CWYRGRL | Lipid insertion (DSPE-PEG/cholesterol anchor) | High-efficiency dual cartilage targeting | [135] |
| BMSC | Chemical modification | WYRGRL | Covalent conjugation (methacrylate linkage) | Improved cartilage targeting and prolonged intra-articular retention | [163] |
Genetic engineering-based strategies take advantage of the natural expression of target proteins on EV membranes or rely on the fusion of target sequences with highly expressed membrane proteins [130]. These methods are often used to confer cartilage-targeting ability in OA-related studies. For example, Liang et al. fused the cartilage-affinity peptide (CAP) DWRVIIPPRPSA sequence with the EV membrane protein, Lamp2b, followed by transfection into dendritic cells [131]. The engineered EVs displayed high levels of CAP on the surface and achieved efficient cartilage targeting, thereby enhancing therapeutic performance. Another approach involves vesicular stomatitis virus glycoprotein (VSV-G), which facilitates membrane fusion. Wu et al. generated EVs with enriched VSV-G expression, achieving improved therapeutic effects in OA models [132]. Unlike cartilage-specific strategies, this approach promotes non-specific binding and fusion, which could potentially benefit the entire joint. However, the effects on other intra-articular tissues have not been evaluated. Further studies are needed to confirm the therapeutic potential of this method.
Chemical modification is one of the major strategies used in EV membrane engineering. Click chemistry is the most frequently applied covalent conjugation approach. Sun et al. used this method to couple CAP to MSC-derived EVs [133]. The modified EVs showed stronger targeting ability toward chondrocytes and remained longer in the joint cavity. Other covalent methods have also been reported. For example, Yan et al. applied the enzyme, Sortase A, to link CAP to EVs membranes, demonstrating an alternative strategy [134]. In addition to covalent modification, non-covalent approaches are also widely explored. Lipid insertion is the most common non-covalent approach, in which molecules, such as DSPE-PEG or cholesterol derivatives, are embedded into the EV membrane to anchor functional ligands. Deng et al. used lipid insertion to attach CAP and a type II collagen–binding peptide (CWYRGRL) to EVs, which resulted in efficient targeting to cartilage tissue [135]. These studies showed that chemical modification can effectively endow EVs with cartilage specificity, thereby enhancing the therapeutic potential in OA.
Although membrane modification has proven to be an effective strategy for OA treatment, the application of membrane modification remains limited by the availability of cartilage-targeting molecules [136]. Currently used ligands include CAP [134], type II collagen–binding peptides [135], and type II collagen antibodies [137], but the range of options is still narrow. Future studies should focus on identifying and validating additional cartilage-targeting peptides, such as through phage display screening [138], to further advance EV-based strategies for OA therapy.
Cargo-engineered EVs in OA therapy
Cargo modification provides another important strategy to enhance the therapeutic potential of EVs in OA therapy. Researchers can endow bioactive molecules, such as nucleic acids, proteins, or small-molecule drugs, with specific regulatory functions that directly target pathologic processes of OA by loading the bioactive molecules into EVs [29]. These engineered vesicles offer improved precision in delivering therapeutic cargo to chondrocytes or other disease-relevant cells within the joint compared to native EVs. Recent studies have explored multiple approaches, including incubation, electroporation, sonication, and genetic engineering, to generate EVs carrying customized therapeutic payloads (Table 3). These methods have enabled the development of EV-based systems capable of modulating inflammation, promoting cartilage repair, and alleviating OA symptoms.
Table 3 Research Examples of Cargo-Engineered EVs in OA Therapy
| Source of EVs | Engineering Methods | Cargo Molecule | Biological Functions | References |
|---|---|---|---|---|
| MSC | Genetic engineering | TGF-β1 | Reduced inflammation and provided chondroprotective effects, alleviating OA progression | [137] |
| BMSC | Genetic engineering | miR874-3p | Suppressed NF-κB–mediated inflammation, reduced oxidative stress damage, promoted chondrogenesis, and alleviated cartilage degeneration | [139] |
| BMSC | Genetic engineering | FTO | Suppressed senescence and apoptosis, promoted autophagy, reduced oxidative stress, and alleviated OA progression through FTO-mediated m6A demethylation | [164] |
| hADSC | Genetic engineering | miR-146a | Rebalanced cytokine profile; reduced cartilage inflammation and degeneration in OA | [165] |
| hUSC | Genetic engineering | miR140 | Promoted mitophagy via the miR-140/CAPN1 axis, improved mitochondrial structure and function, reduced ROS levels | [166] |
| HEK293 | Genetic engineering | FGF18 | Activated FGF18 signaling in chondrocytes, promoted cartilage regeneration, reduced inflammation, and prevented ECM degradation in OA models | [167] |
| RAW 264.7 | Genetic engineering | Lacc1 | Suppressed IL-1β–induced inflammation, inhibited glycolysis, improved mitochondrial function, and promoted cartilage repair with enhanced matrix integrity | [168] |
| Synovial fibroblast | Genetic engineering | SOD3 | Reduced oxidative stress, stabilized extracellular matrix metabolism, protected chondrocytes, and alleviated OA progression in vivo | [169] |
| Expi293F | Incubation | ASO-MMP13 | Decreased MMP13, increased COL2A1, suppressed IL-17/TNF signaling, alleviated OA progression | [134] |
| RAW264.7 | Incubation | Galunisertib | Restored coupled bone remodeling via inhibition of pSmad2/3-dependent TGF-β signaling, enhanced BMSC activity, and alleviated OA progression with low toxicity | [141] |
| MSC | Electroporation | siMDM2 | Eliminated senescent chondrocytes via the MDM2–P53 pathway, restored cartilage matrix homeostasis, and alleviated OA progression in both animal models and human cartilage explants | [86] |
| mADSC | Electroporation | Celecoxib | Inhibited M1 macrophage polarization, maintained chondrocyte homeostasis, promoted BMSC chondrogenic differentiation | [122] |
| Expi293F | Electroporation | siMMP13 | Silenced MMP13 expression in chondrocytes, restored ECM protein balance, and alleviated cartilage degeneration in OA models | [162] |
| MSC | Sonication | miR-29a | Reduced inflammation, stabilized ECM metabolism, and protected cartilage, thereby slowing OA progression | [170] |
Gene engineering represents a powerful tool for customizing EV cargo. By manipulating donor cells, therapeutic molecules, such as siRNA, miRNA, or proteins, can be selectively enriched in EVs, offering advantages in stability and long-term efficacy for the treatment of OA. Wu et al. applied this approach by modifying BMSCs to generate EVs highly enriched with miRNA-874-3p [139]. These engineered vesicles alleviate chondrocyte damage by suppressing the NF-κB signaling pathway and promote chondrogenesis under inflammatory conditions, thereby reducing cartilage degeneration in OA models. Similarly, Kim et al. engineered mesenchymal stem cells to overexpress transforming growth factor (TGF)-β1 [137], producing EVs that exhibited stronger chondroprotective and anti-inflammatory effects and effectively attenuated cartilage damage in experimental OA. These findings demonstrated that gene engineering is an effective strategy for producing EVs with defined therapeutic molecules. However, the intrinsic sorting machinery of EVs does not guarantee efficient incorporation of every molecule expressed in donor cells [140]. As a result, the loading efficiency of this method remains variable and systematic evaluation is still required. Addressing this limitation will be essential for translating gene-engineered EVs into reliable and reproducible therapies for OA.
In addition to genetic programming, therapeutic molecules can be directly loaded into EVs after isolation. Two commonly used techniques are incubation and electroporation, each of which is suited to different types of cargos. Incubation relies on passive diffusion through hydrophobic interactions and is generally applied to lipophilic small drugs. Jing et al. used this method to incorporate a TGF-β inhibitor into EVs, which effectively suppressed pSmad2/3-dependent signaling in subchondral bone and reduced cartilage degeneration [141]. Electroporation, by contrast, is better suited for hydrophilic cargos, such as siRNA or miRNA. Electrical pulses generate transient pores in the vesicle membrane, allowing nucleic acids to enter efficiently. Feng et al. applied this technique to load siMDM2 into MSC-derived EVs [86]. The engineered vesicles eliminated senescent chondrocytes via the MDM2–P53 signaling pathway and preserved cartilage matrix homeostasis in both post-traumatic and aged OA models. Together, these studies confirmed that direct loading approaches endow EVs with potent therapeutic functions in OA. Incubation is simple and gentle but often suffers from low encapsulation efficiency, whereas electroporation achieves higher efficiency for nucleic acids yet risks vesicle aggregation or membrane disruption [118]. Despite these trade-offs, both techniques significantly expand the range of molecules that can be packaged into EVs and underscore the promise for OA therapy.
In summary, loading therapeutic molecules into EVs is an effective strategy to enhance the application in OA therapy. The efficacy of this approach is determined by the therapeutic relevance of the selected cargo and the compatibility of the loading technique with the physicochemical properties [142]. Ultimately, progress in this field will rely not only on refining loading methodologies but also on advancing mechanistic insights into OA pathogenesis, which will guide rational cargo selection and support clinical translation.
Challenges and future prospects
Research on EVs has progressed rapidly in recent decades, deepening our understanding of the biological roles of EVs. Once considered simple mediators of intercellular communication, EVs are now recognized as promising therapeutic tools [143, 144]. Preclinical studies have confirmed the beneficial effects of EVs in OA, yet multiple limitations remain. As summarized in this review, native EVs face challenges, including limited yield, electrostatic repulsion with cartilage, poor targeting, and restricted loading of therapeutic molecules. To address these shortcomings, engineering strategies have been developed to optimize EV production, cargo incorporation, and surface modification.
Despite these advances, technical constraints persist across different EVs engineering strategies, particularly with respect to cargo loading efficiency, stability, and translational feasibility. Gene engineering enables donor cells to package therapeutic nucleic acids or proteins into EVs during biogenesis and generally achieves higher and more sustained cargo enrichment compared to post-isolation methods. However, the intrinsic and tightly regulated sorting mechanisms of EVs limit precise and fully predictable loading, and excessive overexpression of target genes may perturb donor-cell homeostasis [140]. In contrast, post-isolation loading approaches, such as incubation, electroporation, and freeze-thaw cycles offer greater operational simplicity and flexibility in cargo selection. Electroporation and incubation approaches assisted by external stimuli have frequently been adopted for in vivo cargo delivery based on accumulated evidence from prior studies, suggesting the potential feasibility in specific contexts. Nevertheless, the outcomes are strongly influenced by cargo characteristics, loading conditions, and EV sources, and further systematic evaluation is required before definitive conclusions can be drawn. Passive incubation preserves EV membrane integrity but is mainly suitable for small hydrophobic molecules and suffers from low loading efficiency [106]. Electroporation improves the encapsulation of hydrophilic cargos, such as nucleic acids, but may induce EV aggregation or compromise surface protein function [68]. Freeze-thaw cycling is technically straightforward and maintains overall vesicle structure, yet typically yields modest cargo loading and requires repeated processing [112]. These trade-offs highlight that no single loading strategy is universally optimal and method selection must balance efficiency, vesicle integrity, and scalability.
Similarly, surface engineering strategies aimed at improving cartilage targeting, including both gene-based membrane modification and chemical conjugation approaches, have demonstrated encouraging therapeutic outcomes in multiple in vivo OA models. By enabling the display of targeting peptides or proteins on the EV membrane, these strategies have been shown to enhance tissue specificity and improve retention at disease sites. However, despite these promising in vivo results, important limitations remain. The stability and reproducibility of membrane protein presentation continue to pose challenges, particularly due to variable expression levels in gene-engineered systems and the potential loss of functionality during EV isolation, purification, and storage [145]. Collectively, these considerations indicate that current EV engineering approaches remain constrained by instability and context-dependent performance. A systematic and comparative evaluation of cargo loading and surface modification strategies, guided by specific therapeutic goals and translational requirements, will be essential. Continued progress will depend on advances in molecular engineering, delivery technologies, and standardized assessment frameworks to enable rational selection of EV modification strategies for clinical applications.
Another critical factor is disease-specific understanding. As discussed in the cargo-engineering section, the selection of therapeutic molecules must be guided by mechanistic insights into OA pathogenesis. Molecules that modulate inflammation, senescence, or matrix degradation represent promising cargos but the identification depends on ongoing advances in OA biology. Likewise, the repertoire of cartilage-targeting ligands is limited to CAP, type II collagen binding peptides, and related antibodies, which constrain membrane modification strategies. For example, expanding this toolkit through phage display screening will be necessary to discover novel targeting motifs [138].
Current therapeutic strategies for OA primarily focus on symptomatic relief and remain limited in the ability to modify disease progression. Non-pharmacologic interventions and pharmacologic treatments can provide partial pain relief but the effects are often variable, transient, and insufficient for long-term disease control [146]. Consequently, a considerable proportion of patients continue to experience persistent symptoms and ultimately require surgical intervention. In this context, engineered EVs have emerged as a promising alternative with several advantages over conventional therapies. EVs can achieve enhanced tissue penetrability, improved targeting capability, and more efficient delivery of therapeutic molecules through surface modification and cargo engineering, enabling sustained biological activity at disease sites. These features distinguish engineered EVs from traditional small-molecule or protein-based treatments and underscore the potential to address unmet clinical needs in OA management. The clinical translation of engineered EVs also faces safety concerns. Although native EVs have demonstrated encouraging safety in preclinical and early clinical studies [14, 15], engineering inevitably alters the intrinsic properties, including charge, ligand composition, and molecular content. These changes may influence biodistribution, immunogenicity, and long-term stability. Comprehensive evaluation is therefore essential, encompassing toxicology, immune response, and long-term monitoring to ensure biocompatibility in humans. Only with robust safety data can engineered EVs progress toward reliable clinical use.
Methodologic limitations further hinder translation. Current isolation and purification techniques lack standardization, leading to variability in yield, purity, and functional activity across studies [147]. The development of scalable cell-culture processes suitable for large-volume EV production is equally important for obtaining high-quality EVs for clinical applications in OA. Most current approaches rely on 2D-culture systems, which limit the surface area available for cell growth and restrict overall yield. Multilayer cell factories can support small-to-medium-scale expansion but require considerable labor and physical space, highlighting the need for more efficient platforms for large-scale production. Bioreactor-based expansion offers a practical solution by enabling the generation of EVs at clinically relevant scales while reducing consumable use, labor demands, and processing time. The ability to collect EVs in a single batch also helps improve batch consistency and enhances the stability of the final product [148]. A remaining challenge lies in the downstream isolation of bioactive EVs from large volumes of conditioned medium. Laboratory methods, such as ultracentrifugation, polymer precipitation, ultrafiltration, size-exclusion chromatography, immunoaffinity-based approaches, and microfluidic devices, often lack the throughput or robustness needed for GMP-compatible manufacturing. Tangential flow filtration and polymer-based precipitation show promise for large-scale purification, yet further optimization is required to achieve rapid, reliable, and high-yield recovery of high-purity EVs from protein-rich and compositionally complex supernatants [149]. Recent studies provide an interesting alternative by showing that cell lysis followed by freeze-thaw processing can generate large quantities of artificial vesicles that resemble natural EVs in composition and biological activity. This approach has been reported to increase vesicle output by as much as 16-fold from the same number of cells, suggesting a potential route for overcoming current limitations in scalable EVs production [150, 151]. In addition, key parameters, such as optimal dosing, frequency of intra-articular injection, and long-term pharmacokinetics, remain undefined [152]. In vivo imaging and tracking tools are still underdeveloped, restricting precise evaluation of EV biodistribution, therapeutic persistence, and potential off-target effects [153]. Addressing these gaps will be essential to improve reproducibility and comparability across studies.
In summary, the future of engineered EVs for OA therapy relies on integrating mechanistic insights with technological innovation. Effective therapies will require cargos that intervene in core disease mechanisms, paired with engineering methods that enhance stability, efficiency, and scalability. At the same time, biosafety, manufacturing consistency, and regulatory pathways must be systematically addressed. With these advances, engineered EVs hold the potential to evolve from experimental tools into clinically viable therapies for OA.
Conclusion
Research on engineered EVs has opened new opportunities for OA therapy by addressing the limitations of native EVs, such as low yield, limited cargo content, and poor targeting efficiency. Advances in genetic modification, and chemical and physical engineering have provided diverse strategies to enhance the therapeutic properties of EVs. Preclinical studies have confirmed that engineered EVs can modulate inflammation, reduce chondrocyte senescence, and promote cartilage repair, thereby offering promising alternatives to conventional treatments. However, challenges remain regarding biosafety, production scalability, and the selection of effective therapeutic cargos. Future work should integrate mechanistic insights into OA pathogenesis with technological innovations to refine engineering methods and ensure reproducibility, safety, and clinical applicability. With continued progress, engineered EVs hold strong potential to evolve into reliable and effective therapies for OA.
List of abbreviations
(FGF18) Fibroblast growth factor 18, (Lacc1) Laccase domain-containing 1, (FTO) Obesity-associated protein, (SOD3) Superoxide dismutase 3, (hADSCs) Human adipose-derived MSCs, (ASO-MMP13) Antisense oligonucleotides targeting matrix metalloproteinase-13, (mADSC) Mouse adipose-derived mesenchymal stem cell, (PTH) Parathyroid hormone, (EMF) Electromagnetic field, (USPIO) Ultrasmall superparamagnetic iron oxide, (nsPEF) Nanosecond pulsed electric field, (VSV-G) Vesicular stomatitis virus glycoprotein, (HAS2) Hyaluronan synthase type 2, (DC) Dendritic cells, (CAP) Chondrocyte-affinity peptide, (MAL) Maleimide, (hUCMSCs) Human umbilical-cord-derived mesenchymal stem cells, (DSPE-PEG) Polyethylene glycol-grafted 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (82072419 and 82402879).
Conflict of interest
The authors declare that there are no conflicts of interest.
Graphical abstract
Highlights
- This review outlines the fundamental characteristics of extracellular vesicles (EVs) and the limitations in osteoarthritis (OA) therapy.
- Current engineering strategies, including yield enhancement, charge modification, cargo loading, and membrane modification, are summarized with applications in OA models.
- Future directions emphasize integrating OA mechanisms with engineering advances, while ensuring safety and standardization for clinical translation.
In brief
Osteoarthritis (OA) remains a leading cause of disability and current therapies fail to effectively restore joint homeostasis. Extracellular vesicles (EVs) have emerged as promising therapeutic candidates owing to the natural biocompatibility and regenerative potential. However, the therapeutic efficacy is constrained by low production yield, electrostatic repulsion from cartilage tissue, limited loading capacity of therapeutic molecules, and insufficient targeting efficiency. This review provides an overview of recent engineering strategies to improve EV performance, summarizes the therapeutic applications in OA models, and further discusses the remaining challenges and future prospects for translating engineered EVs into clinically applicable therapies.
References
- Pigeolet M, Jayaram A, Park KB, Meara JG. Osteoarthritis in 2020 and beyond. Lancet 2021;397(10279):1059-60. [PMID: 33743863 DOI: 10.1016/S0140-6736(21)00208-7]
- Barry F, Murphy M. Mesenchymal stem cells in joint disease and repair. Nat Rev Rheumatol 2013;9(10):584-94. [PMID: 23881068 DOI: 10.1038/nrrheum.2013.109]
- Xia B, Deng Y, Lv Y, Chen G. Stem cell recruitment based on scaffold features for bone tissue engineering. Biomater Sci 2021;9(4):1189-203. [PMID: 33355545 DOI: 10.1039/d0bm01591a]
- Agung M, Ochi M, Yanada S, Adachi N, Izuta Y, et al. Mobilization of bone marrow-derived mesenchymal stem cells into the injured tissues after intraarticular injection and their contribution to tissue regeneration. Knee Surg Sports Traumatol Arthrosc 2006;14(12):1307-14. [PMID: 16788809 DOI: 10.1007/s00167-006-0124-8]
- Baranovskii DS, Klabukov ID, Arguchinskaya NV, Yakimova AO, Kisel AA, et al. Adverse events, side effects and complications in mesenchymal stromal cell-based therapies. Stem Cell Investig 2022;9:7. [PMID: 36393919 DOI: 10.21037/sci-2022-025]
- You B, Zhou C, Yang Y. MSC-EVs alleviate osteoarthritis by regulating microenvironmental cells in the articular cavity and maintaining cartilage matrix homeostasis. Ageing Res Rev 2023;85:101864. [PMID: 36707035 DOI: 10.1016/j.arr.2023.101864]
- Yin B, Ni J, Witherel CE, Yang M, Burdick JA, et al. Harnessing tissue-derived extracellular vesicles for osteoarthritis theranostics. Theranostics 2022;12(1):207-31. [PMID: 34987642 DOI: 10.7150/thno.62708]
- Boulestreau J, Maumus M, Jorgensen C, Noël D. Extracellular vesicles from mesenchymal stromal cells: therapeutic perspectives for targeting senescence in osteoarthritis. Adv Drug Deliv Rev 2021;175:113836. [PMID: 34166759 DOI: 10.1016/j.addr.2021.113836]
- Welsh JA, Goberdhan DCI, O’Driscoll L, Buzas EI, Blenkiron C, et al. Minimal information for studies of extracellular vesicles (MISEV2023): from basic to advanced approaches. J Extracell Vesicles 2024;13(2):e12404. [PMID: 38326288 DOI: 10.1002/jev2.12404]
- Witwer KW, Théry C. Extracellular vesicles or exosomes? On primacy, precision, and popularity influencing a choice of nomenclature. J Extracell Vesicles 2019;8(1):1648167. [PMID: 31489144 DOI: 10.1080/20013078.2019.1648167]
- Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science 2020;367(6478):eaau6977. [PMID: 32029601 DOI: 10.1126/science.aau6977]
- Pitt JM, Kroemer G, Zitvogel L. Extracellular vesicles: masters of intercellular communication and potential clinical interventions. J Clin Invest 2016;126(4):1139-43. [PMID: 27035805 DOI: 10.1172/jci87316]
- Zappulli V, Friis KP, Fitzpatrick Z, Maguire CA, Breakefield XO. Extracellular vesicles and intercellular communication within the nervous system. J Clin Invest 2016;126(4):1198-207. [PMID: 27035811 DOI: 10.1172/jci81134]
- Bolandnazar NS, Raeissadat SA, Haghighatkhah H, Rayegani SM, Oshnari RS, et al. Safety and efficacy of placental mesenchymal stromal cells-derived extracellular vesicles in knee osteoarthritis: a randomized, triple-blind, placebo-controlled clinical trial. BMC Musculoskelet Disord 2024;25(1):856. [PMID: 39465400 DOI: 10.1186/s12891-024-07979-w]
- Kong Y, Wang Y, Yang Y, Hou Y, Yu J, et al. Intra-articular injection of exosomes derived from different stem cells in animal models of osteoarthritis: a systematic review and meta- analysis. J Orthop Surg Res 2024;19(1):834. [PMID: 39696589 DOI: 10.1186/s13018-024-05227-4]
- Łabędź-Masłowska A, Wieczorek J, Mierzwiński M, Sekuła-Stryjewska M, Noga S, et al. Evaluation of the safety and regenerative potential of human mesenchymal stem cells and their extracellular vesicles in a transgenic pig model of cartilage-bone injury in vivo – preclinical study. Stem Cell Rev Rep 2025;21(4):1075-95. [PMID: 40380984 DOI: 10.1007/s12015-025-10853-4]
- Teng F, Fussenegger M. Shedding light on extracellular vesicle biogenesis and bioengineering. Adv Sci (Weinh) 2020;8(1):2003505. [PMID: 33437589 DOI: 10.1002/advs.202003505]
- Luo L, Wu Z, Wang Y, Li H. Regulating the production and biological function of small extracellular vesicles: current strategies, applications and prospects. J Nanobiotechnology 2021;19(1):422. [PMID: 34906146 DOI: 10.1186/s12951-021-01171-1]
- Villarroya-Beltri C, Baixauli F, Gutiérrez-Vázquez C, Sánchez-Madrid F, Mittelbrunn M. Sorting it out: regulation of exosome loading. Semin Cancer Biol 2014;28:3-13. [PMID: 24769058 DOI: 10.1016/j.semcancer.2014.04.009]
- Lee YJ, Shin KJ, Chae YC. Regulation of cargo selection in exosome biogenesis and its biomedical applications in cancer. Exp Mol Med 2024;56(4):877-89.[PMID: 38580812 DOI: 10.1038/s12276-024-01209-y]
- Chevillet JR, Kang Q, Ruf IK, Briggs HA, Vojtech LN, et al. Quantitative and stoichiometric analysis of the microRNA content of exosomes. Proc Natl Acad Sci U S A 2014;111(41):14888-93. [PMID: 25267620 DOI: 10.1073/pnas.1408301111]
- Turchinovich A, Tonevitsky AG, Cho WC, Burwinkel B. Check and mate to exosomal extracellular miRNA: new lesson from a new approach. Front Mol Biosci 2015;2:11. [PMID: 25988178 DOI: 10.3389/fmolb.2015.00011]
- Frolova L, Li ITS. Targeting capabilities of native and bioengineered extracellular vesicles for drug delivery. Bioengineering (Basel) 2022;9(10):496. [PMID: 36290464 DOI: 10.3390/bioengineering9100496]
- Murphy DE, de Jong OG, Brouwer M, Wood MJ, Lavieu G, et al. Extracellular vesicle-based therapeutics: natural versus engineered targeting and trafficking. Exp Mol Med 2019;51(3):1-12. [PMID: 30872574 DOI: 10.1038/s12276-019-0223-5]
- Gurung S, Perocheau D, Touramanidou L, Baruteau J. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal 2021;19(1):47. [PMID: 33892745 DOI: 10.1186/s12964-021-00730-1]
- Zhuang Y, Jiang S, Yuan C, Lin K. The potential therapeutic role of extracellular vesicles in osteoarthritis. Front Bioeng Biotechnol 2022;10:1022368. [PMID: 36185451 DOI: 10.3389/fbioe.2022.1022368]
- Drohat P, Baron M, Kaplan LD, Best TM, Kouroupis D. Long-acting extracellular vesicle-based biologics in osteoarthritis immunotherapy. Bioengineering (Basel) 2025;12(5):525. [PMID: 40428144 DOI: 10.3390/bioengineering12050525]
- Abreu H, Canciani E, Raineri D, Cappellano G, Rimondini L, et al. Extracellular vesicles in musculoskeletal regeneration: modulating the therapy of the future. Cells 2021;11(1):43. [PMID: 35011605 DOI: 10.3390/cells11010043]
- Ehab S, Gaser OA, Oyouni AAA, Kameli N, Alzahrani F, et al. Engineered extracellular vesicles in arthritic diseases: therapeutic applications & challenges. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2025;17(4):e70031. [PMID: 40830968 DOI: 10.1002/wnan.70031]
- van Niel G, D’Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol 2018;19(4):213-28. [PMID: 29339798 DOI: 10.1038/nrm.2017.125]
- Jadli AS, Ballasy N, Edalat P, Patel VB. Inside(sight) of tiny communicator: exosome biogenesis, secretion, and uptake. Mol Cell Biochem 2020;467(1-2):77-94. [PMID: 32088833 DOI: 10.1007/s11010-020-03703-z]
- Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol 2014;30:255-89. [PMID: 25288114 DOI: 10.1146/annurev-cellbio-101512-122326]
- Gurunathan S, Kang MH, Jeyaraj M, Qasim M, Kim JH. Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes. Cells 2019;8(4):307. [PMID: 30987213 DOI: 10.3390/cells8040307]
- Li P, Kaslan M, Lee SH, Yao J, Gao Z. Progress in exosome isolation techniques. Theranostics 2017;7(3):789-804. [PMID: 28255367 DOI: 10.7150/thno.18133]
- Livshits MA, Khomyakova E, Evtushenko EG, Lazarev VN, Kulemin NA, et al. Isolation of exosomes by differential centrifugation: theoretical analysis of a commonly used protocol. Sci Rep 2015;5:17319. [PMID: 26616523 DOI: 10.1038/srep17319]
- Chen BY, Sung CW, Chen C, Cheng CM, Lin DP, et al. Advances in exosomes technology. Clin Chim Acta 2019;493:14-9. [PMID: 30797770 DOI: 10.1016/j.cca.2019.02.021]
- Yamashita T, Takahashi Y, Takakura Y. Possibility of exosome-based therapeutics and challenges in production of exosomes eligible for therapeutic application. Biol Pharm Bull 2018;41(6):835-42. [PMID: 29863072 DOI: 10.1248/bpb.b18-00133]
- Hu G, Xia Y, Chen B, Zhang J, Gong L, et al. ESC-sEVs rejuvenate aging hippocampal NSCs by transferring SMADs to regulate the MYT1-Egln3-Sirt1 axis. Mol Ther 2021;29(1):103-20. [PMID: 33038325 DOI: 10.1016/j.ymthe.2020.09.037]
- Zhu Q, Ling X, Yang Y, Zhang J, Li Q, et al. Embryonic stem cells-derived exosomes endowed with targeting properties as chemotherapeutics delivery vehicles for glioblastoma therapy. Adv Sci (Weinh) 2019;6(6):1801899. [PMID: 30937268 DOI: 10.1002/advs.201801899]
- Xia Y, Hu G, Chen Y, Yuan J, Zhang J, et al. Embryonic stem cell derived small extracellular vesicles modulate regulatory T cells to protect against ischemic stroke. ACS Nano 2021;15(4):7370-85. [PMID: 33733738 DOI: 10.1021/acsnano.1c00672]
- Nordin JZ, Lee Y, Vader P, Mäger I, Johansson HJ, et al. Ultrafiltration with size-exclusion liquid chromatography for high yield isolation of extracellular vesicles preserving intact biophysical and functional properties. Nanomedicine 2015;11(4):879-83. [PMID: 25659648 DOI: 10.1016/j.nano.2015.01.003]
- Driedonks T, Jiang L, Carlson B, Han Z, Liu G, et al. Pharmacokinetics and biodistribution of extracellular vesicles administered intravenously and intranasally to Macaca nemestrina. J Extracell Biol 2022;1(10):e59. [PMID: 36591537 DOI: 10.1002/jex2.59]
- Kang M, Jordan V, Blenkiron C, Chamley LW. Biodistribution of extracellular vesicles following administration into animals: a systematic review. J Extracell Vesicles 2021;10(8):e12085. [PMID: 34194679 DOI: 10.1002/jev2.12085]
- Ye T, Chen Z, Zhang J, Luo L, Gao R, et al. Large extracellular vesicles secreted by human iPSC-derived MSCs ameliorate tendinopathy via regulating macrophage heterogeneity. Bioact Mater 2023;21:194-208. [PMID: 36101856 DOI: 10.1016/j.bioactmat.2022.08.007]
- Klumperman J, Raposo G. The complex ultrastructure of the endolysosomal system. Cold Spring Harb Perspect Biol 2014;6(10):a016857. [PMID: 24851870 DOI: 10.1101/cshperspect.a016857]
- Yao C, Wang P, Zhou L, Wang R, Li X, et al. Highly biocompatible zwitterionic phospholipids coated upconversion nanoparticles for efficient bioimaging. Anal Chem 2014;86(19):9749-57. [PMID: 25075628 DOI: 10.1021/ac5023259]
- Wehling P, Evans C, Wehling J, Maixner W. Effectiveness of intra-articular therapies in osteoarthritis: a literature review. Ther Adv Musculoskelet Dis 2017;9(8):183-96. [PMID: 28835778 DOI: 10.1177/1759720×17712695]
- Pradal J, Maudens P, Gabay C, Seemayer CA, Jordan O, et al. Effect of particle size on the biodistribution of nano- and microparticles following intra-articular injection in mice. Int J Pharm 2016;498(1-2):119-29. [PMID: 26685724 DOI: 10.1016/j.ijpharm.2015.12.015]
- Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet 2019;393(10182):1745-59. [PMID: 31034380 DOI: 10.1016/s0140-6736(19)30417-9]
- Kiani C, Chen L, Wu YJ, Yee AJ, Yang BB. Structure and function of aggrecan. Cell Res 2002;12(1):19-32. [PMID: 11942407 DOI: 10.1038/sj.cr.7290106]
- Lenzini S, Bargi R, Chung G, Shin JW. Matrix mechanics and water permeation regulate extracellular vesicle transport. Nat Nanotechnol 2020;15(3):217-23. [PMID: 32066904 DOI: 10.1038/s41565-020-0636-2]
- Feng K, Xie X, Yuan J, Gong L, Zhu Z, et al. Reversing the surface charge of MSC-derived small extracellular vesicles by εPL-PEG-DSPE for enhanced osteoarthritis treatment. J Extracell Vesicles 2021;10(13):e12160. [PMID: 34724347 DOI: 10.1002/jev2.12160]
- Stell G, Joslin CG. The donnan equilibrium: a theoretical study of the effects of interionic forces. Biophys J 1986;50(5):855-9. [PMID: 19431690 DOI: 10.1016/s0006-3495(86)83526-3]
- Skotland T, Sandvig K, Llorente A. Lipids in exosomes: current knowledge and the way forward. Prog Lipid Res 2017;66:30-41. [PMID: 28342835 DOI: 10.1016/j.plipres.2017.03.001]
- Luo L, Chen ZS, Yuan J, Niu X, Liu JS, et al. Identification of a specific marker panel for human mesenchymal stem cell-derived small extracellular vesicles. Interdiscip Med 2025;3(6):e70065. [DOI: 10.1002/inmd.70065]
- Chen Z, Luo L, Ye T, Zhou J, Niu X, et al. Identification of specific markers for human pluripotent stem cell-derived small extracellular vesicles. J Extracell Vesicles 2024;13(2):e12409. [PMID: 38321535 DOI: 10.1002/jev2.12409]
- Christianson HC, Svensson KJ, van Kuppevelt TH, Li JP, Belting M. Cancer cell exosomes depend on cell-surface heparan sulfate proteoglycans for their internalization and functional activity. Proc Natl Acad Sci U S A 2013;110(43):17380-5. [PMID: 24101524 DOI: 10.1073/pnas.1304266110]
- Viñas JL, Spence M, Gutsol A, Knoll W, Burger D, et al. Receptor-ligand interaction mediates targeting of endothelial colony forming cell-derived exosomes to the kidney after ischemic injury. Sci Rep 2018;8(1):16320. [PMID: 30397255 DOI: 10.1038/s41598-018-34557-7]
- Ivosevic Z, Ljujic B, Pavlovic D, Matovic V, Gazdic Jankovic M. Mesenchymal stem cell-derived extracellular vesicles: new soldiers in the war on immune-mediated diseases. Cell Transplant 2023;32:9636897231207194. [PMID: 37882092 DOI: 10.1177/09636897231207194]
- Sun Y, Zhao J, Wu Q, Zhang Y, You Y, et al. Chondrogenic primed extracellular vesicles activate miR-455/SOX11/FOXO axis for cartilage regeneration and osteoarthritis treatment. npj Regen Med 2022;7(1):53. [PMID: 36114225 DOI: 10.1038/s41536-022-00250-7]
- Joo HS, Suh JH, Lee HJ, Bang ES, Lee JM. Current knowledge and future perspectives on mesenchymal stem cell-derived exosomes as a new therapeutic agent. Int J Mol Sci 2020;21(3):727. [PMID: 31979113 DOI: 10.3390/ijms21030727]
- Shah N, Morsi Y, Manasseh R. From mechanical stimulation to biological pathways in the regulation of stem cell fate. Cell Biochem Funct 2014;32(4):309-25. [PMID: 24574137 DOI: 10.1002/cbf.3027]
- Wang Z, Maruyama K, Sakisaka Y, Suzuki S, Tada H, et al. Cyclic stretch force induces periodontal ligament cells to secrete exosomes that suppress IL-1β production through the inhibition of the NF-κB signaling pathway in macrophages. Front Immunol 2019;10:1310. [PMID: 31281309 DOI: 10.3389/fimmu.2019.01310]
- Guo S, Debbi L, Zohar B, Samuel R, Arzi RS, et al. Stimulating extracellular vesicles production from engineered tissues by mechanical forces. Nano Lett 2021;21(6):2497-504. [PMID: 33709717 DOI: 10.1021/acs.nanolett.0c04834]
- de Lucas B, Pérez LM, Bernal A, Gálvez BG. Ultrasound therapy: experiences and perspectives for regenerative medicine. Genes (Basel) 2020;11(9):1086. [PMID: 32957737 DOI: 10.3390/genes11091086]
- Yang Q, Nanayakkara GK, Drummer C, Sun Y, Johnson C, et al. Low-intensity ultrasound-induced anti-inflammatory effects are mediated by several new mechanisms including gene induction, immunosuppressor cell promotion, and enhancement of exosome biogenesis and docking. Front Physiol 2017;8:818. [PMID: 29109687 DOI: 10.3389/fphys.2017.00818]
- Fukuta T, Nishikawa A, Kogure K. Low level electricity increases the secretion of extracellular vesicles from cultured cells. Biochem Biophys Rep 2020;21:100713. [PMID: 31828227 DOI: 10.1016/j.bbrep.2019.100713]
- Yang Z, Shi J, Xie J, Wang Y, Sun J, et al. Large-scale generation of functional mRNA-encapsulating exosomes via cellular nanoporation. Nat Biomed Eng 2020;4(1):69-83. [PMID: 31844155 DOI: 10.1038/s41551-019-0485-1]
- Bister N, Pistono C, Huremagic B, Jolkkonen J, Giugno R, et al. Hypoxia and extracellular vesicles: a review on methods, vesicular cargo and functions. J Extracell Vesicles 2020;10(1):e12002. [PMID: 33304471 DOI: 10.1002/jev2.12002]
- King HW, Michael MZ, Gleadle JM. Hypoxic enhancement of exosome release by breast cancer cells. BMC Cancer 2012;12:421. [PMID: 22998595 DOI: 10.1186/1471-2407-12-421]
- Haraszti RA, Miller R, Stoppato M, Sere YY, Coles A, et al. Exosomes produced from 3D cultures of MSCs by tangential flow filtration show higher yield and improved activity. Mol Ther 2018;26(12):2838-47. [PMID: 30341012 DOI: 10.1016/j.ymthe.2018.09.015]
- Yan L, Wu X. Exosomes produced from 3D cultures of umbilical cord mesenchymal stem cells in a hollow-fiber bioreactor show improved osteochondral regeneration activity. Cell Biol Toxicol 2020;36(2):165-78. [PMID: 31820164 DOI: 10.1007/s10565-019-09504-5]
- Hoshino D, Kirkbride KC, Costello K, Clark ES, Sinha S, et al. Exosome secretion is enhanced by invadopodia and drives invasive behavior. Cell Rep 2013;5(5):1159-68. [PMID: 24290760 DOI: 10.1016/j.celrep.2013.10.050]
- Colombo M, Moita C, van Niel G, Kowal J, Vigneron J, et al. Analysis of ESCRT functions in exosome biogenesis, composition and secretion highlights the heterogeneity of extracellular vesicles. J Cell Sci 2013;126(Pt 24):5553-65. [PMID: 24105262 DOI: 10.1242/jcs.128868]
- Abrami L, Brandi L, Moayeri M, Brown MJ, Krantz BA, et al. Hijacking multivesicular bodies enables long-term and exosome-mediated long-distance action of anthrax toxin. Cell Rep 2013;5(4):986-96. [PMID: 24239351 DOI: 10.1016/j.celrep.2013.10.019]
- Baietti MF, Zhang Z, Mortier E, Melchior A, Degeest G, et al. Syndecan-syntenin-ALIX regulates the biogenesis of exosomes. Nat Cell Biol 2012;14(7):677-85. [PMID: 22660413 DOI: 10.1038/ncb2502]
- Kojima R, Bojar D, Rizzi G, Hamri GC, El-Baba MD, et al. Designer exosomes produced by implanted cells intracerebrally deliver therapeutic cargo for Parkinson’s disease treatment. Nat Commun 2018;9(1):1305. [PMID: 29610454 DOI: 10.1038/s41467-018-03733-8]
- Ostrowski M, Carmo NB, Krumeich S, Fanget I, Raposo G, et al. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat Cell Biol 2010;12(1):19-30; sup pp 1-13. [PMID: 19966785 DOI: 10.1038/ncb2000]
- Peinado H, Alečković M, Lavotshkin S, Matei I, Costa-Silva B, et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat Med 2012;18(6):883-91. [PMID: 22635005 DOI: 10.1038/nm.2753]
- Ludwig N, Yerneni SS, Menshikova EV, Gillespie DG, Jackson EK, et al. Simultaneous inhibition of glycolysis and oxidative phosphorylation triggers a multi-fold increase in secretion of exosomes: possible role of 2’3’-cAMP. Sci Rep 2020;10(1):6948. [PMID: 32332778 DOI: 10.1038/s41598-020-63658-5]
- Datta A, Kim H, McGee L, Johnson AE, Talwar S, et al. High-throughput screening identified selective inhibitors of exosome biogenesis and secretion: a drug repurposing strategy for advanced cancer. Sci Rep 2018;8(1):8161. [PMID: 29802284 DOI: 10.1038/s41598-018-26411-7]
- Park DJ, Yun WS, Kim WC, Park JE, Lee SH, et al. Improvement of stem cell-derived exosome release efficiency by surface-modified nanoparticles. J Nanobiotechnology 2020;18(1):178. [PMID: 33287848 DOI: 10.1186/s12951-020-00739-7]
- Zhang Z, Xu R, Yang Y, Liang C, Yu X, et al. Micro/nano-textured hierarchical titanium topography promotes exosome biogenesis and secretion to improve osseointegration. J Nanobiotechnology 2021;19(1):78. [PMID: 33741002 DOI: 10.1186/s12951-021-00826-3]
- Wu Z, He D, Li H. Bioglass enhances the production of exosomes and improves their capability of promoting vascularization. Bioact Mater 2021;6(3):823-35. [PMID: 33024902 DOI: 10.1016/j.bioactmat.2020.09.011]
- Sun Z, Chen X, Niu R, Chen H, Zhu Y, et al. Liposome fusogenic enzyme-free circuit enables high-fidelity determination of single exosomal RNA. Mater Today Bio 2023;19:100613. [PMID: 37009069 DOI: 10.1016/j.mtbio.2023.100613]
- Feng K, Liu J, Gong L, Ye T, Chen Z, et al. Engineered MSC-sEVs as a versatile nanoplatform for enhanced osteoarthritis treatment via targeted elimination of senescent chondrocytes and maintenance of cartilage matrix metabolic homeostasis. Adv Sci (Weinh) 2025;12(8):e2413759. [PMID: 39755936 DOI: 10.1002/advs.202413759]
- Kim JY, Lee SY, Cha SG, Park JM, Song DH, et al. Combinatory nanovesicle with siRNA-loaded extracellular vesicle and IGF-1 for osteoarthritis treatments. Int J Mol Sci 2024;25(10):5242. [PMID: 38791285 DOI: 10.3390/ijms25105242]
- Chen Y, Li B, Christelle M, Eugene N, Han W, et al. Modifying MSCs-derived EVs with esterase-responsive and charge-reversal cationic polymers enhances bone regeneration. iScience 2024;27(9):110801. [PMID: 39310777 DOI: 10.1016/j.isci.2024.110801]
- Vader P, Mol EA, Pasterkamp G, Schiffelers RM. Extracellular vesicles for drug delivery. Adv Drug Deliv Rev 2016;106(Pt A):148-56. [PMID: 26928656 DOI: 10.1016/j.addr.2016.02.006]
- Richter M, Vader P, Fuhrmann G. Approaches to surface engineering of extracellular vesicles. Adv Drug Deliv Rev 2021;173:416-26. [PMID: 33831479 DOI: 10.1016/j.addr.2021.03.020]
- Ben XY, Wang YR, Zheng HH, Li DX, Ren R, et al. Construction of exosomes that overexpress CD47 and evaluation of their immune escape. Front Bioeng Biotechnol 2022;10:936951. [PMID: 35845399 DOI: 10.3389/fbioe.2022.936951]
- Ohno S, Takanashi M, Sudo K, Ueda S, Ishikawa A, et al. Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol Ther 2013;21(1):185-91. [PMID: 23032975 DOI: 10.1038/mt.2012.180]
- Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, et al. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol 2011;29(4):341-5. [PMID: 21423189 DOI: 10.1038/nbt.1807]
- Tian T, Zhang HX, He CP, Fan S, Zhu YL, et al. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials 2018;150:137-49. [PMID: 29040874 DOI: 10.1016/j.biomaterials.2017.10.012]
- Huang X, Wu W, Jing D, Yang L, Guo H, et al. Engineered exosome as targeted lncRNA MEG3 delivery vehicles for osteosarcoma therapy. J Control Release 2022;343:107-17. [PMID: 35077741 DOI: 10.1016/j.jconrel.2022.01.026]
- Tamura R, Uemoto S, Tabata Y. Augmented liver targeting of exosomes by surface modification with cationized pullulan. Acta Biomater 2017;57:274-84. [PMID: 28483695 DOI: 10.1016/j.actbio.2017.05.013]
- N’Diaye ER, Orefice NS, Ghezzi C, Boumendjel A. Chemically modified extracellular vesicles and applications in radiolabeling and drug delivery. Pharmaceutics 2022;14(3):653. [PMID: 35336027 DOI: 10.3390/pharmaceutics14030653]
- Raghav A, Jeong GB. A systematic review on the modifications of extracellular vesicles: a revolutionized tool of nano-biotechnology. J Nanobiotechnology 2021;19(1):459. [PMID: 34965878 DOI: 10.1186/s12951-021-01219-2]
- Smyth T, Petrova K, Payton NM, Persaud I, Redzic JS, et al. Surface functionalization of exosomes using click chemistry. Bioconjug Chem 2014;25(10):1777-84. [PMID: 25220352 DOI: 10.1021/bc500291r]
- Hao D, Lu L, Song H, Duan Y, Chen J, et al. Engineered extracellular vesicles with high collagen-binding affinity present superior in situ retention and therapeutic efficacy in tissue repair. Theranostics 2022;12(13):6021-37. [PMID: 35966577 DOI: 10.7150/thno.70448]
- Liu Q, Li D, Pan X, Liang Y. Targeted therapy using engineered extracellular vesicles: principles and strategies for membrane modification. J Nanobiotechnology 2023;21(1):334. [PMID: 37717008 DOI: 10.1186/s12951-023-02081-0]
- Li L, Wang F, Zhu D, Hu S, Cheng K, et al. Engineering exosomes and exosome-like nanovesicles for improving tissue targeting and retention. Fundam Res 2025;5(2):851-67. [PMID: 40242543 DOI: 10.1016/j.fmre.2024.03.025]
- Komuro H, Aminova S, Lauro K, Harada M. Advances of engineered extracellular vesicles-based therapeutics strategy. Sci Technol Adv Mater 2022;23(1):655-81. [PMID: 36277506 DOI: 10.1080/14686996.2022.2133342]
- Qian F, Huang Z, Zhong H, Lei Q, Ai Y, et al. Analysis and biomedical applications of functional cargo in extracellular vesicles. ACS Nano 2022;16(12):19980-20001. [PMID: 36475625 DOI: 10.1021/acsnano.2c11298]
- Dave KM, Pinky PP, S Manickam D. Molecular engineering of extracellular vesicles for drug delivery: strategies, challenges, and perspectives. J Control Release 2025;386:114068. [PMID: 40721069 DOI: 10.1016/j.jconrel.2025.114068]
- Pascucci L, Coccè V, Bonomi A, Ami D, Ceccarelli P, et al. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J Control Release 2014;192:262-70. [PMID: 25084218 DOI: 10.1016/j.jconrel.2014.07.042]
- Sun D, Zhuang X, Xiang X, Liu Y, Zhang S, et al. A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol Ther 2010;18(9):1606-14. [PMID: 20571541 DOI: 10.1038/mt.2010.105]
- Zeng H, Guo S, Ren X, Wu Z, Liu S, Yao X. Current strategies for exosome cargo loading and targeting delivery. Cells 2023;12(10):1416. [PMID: 37408250 DOI: 10.3390/cells12101416]
- Han Y, Jones TW, Dutta S, Zhu Y, Wang X, et al. Overview and update on methods for cargo loading into extracellular vesicles. Processes (Basel) 2021;9(2):356. [PMID: 33954091 DOI: 10.3390/pr9020356]
- Donoso-Quezada J, Ayala-Mar S, González-Valdez J. State-of-the-art exosome loading and functionalization techniques for enhanced therapeutics: a review. Crit Rev Biotechnol 2020;40(6):804-20. [PMID: 32605394 DOI: 10.1080/07388551.2020.1785385]
- García-Manrique P, Matos M, Gutiérrez G, Pazos C, Blanco-López MC. Therapeutic biomaterials based on extracellular vesicles: classification of bio-engineering and mimetic preparation routes. J Extracell Vesicles 2018;7(1):1422676. [PMID: 29372017 DOI: 10.1080/20013078.2017.1422676]
- Won Lee G, Thangavelu M, Joung Choi M, Yeong Shin E, Sol Kim H, et al. Exosome mediated transfer of miRNA-140 promotes enhanced chondrogenic differentiation of bone marrow stem cells for enhanced cartilage repair and regeneration. J Cell Biochem 2020;121(7):3642-52. [PMID: 32091634 DOI: 10.1002/jcb.29657]
- Hajipour H, Farzadi L, Roshangar L, Latifi Z, Kahroba H, et al. A human chorionic gonadotropin (hCG) delivery platform using engineered uterine exosomes to improve endometrial receptivity. Life Sci 2021;275:119351. [PMID: 33737084 DOI: 10.1016/j.lfs.2021.119351]
- Liang G, Zhu Y, Ali DJ, Tian T, Xu H, et al. Engineered exosomes for targeted co-delivery of miR-21 inhibitor and chemotherapeutics to reverse drug resistance in colon cancer. J Nanobiotechnology 2020;18(1):10. [PMID: 31918721 DOI: 10.1186/s12951-019-0563-2]
- Zhao Z, Shuang T, Gao Y, Lu F, Zhang J, et al. Targeted delivery of exosomal miR-484 reprograms tumor vasculature for chemotherapy sensitization. Cancer Lett 2022;530:45-58. [PMID: 35051533 DOI: 10.1016/j.canlet.2022.01.011]
- Zhou W, Zhou Y, Chen X, Ning T, Chen H, et al. Pancreatic cancer-targeting exosomes for enhancing immunotherapy and reprogramming tumor microenvironment. Biomaterials 2021;268:120546. [PMID: 33253966 DOI: 10.1016/j.biomaterials.2020.120546]
- Liu Q, Dai G, Wu Y, Zhang M, Yang M, et al. iRGD-modified exosomes-delivered BCL6 siRNA inhibit the progression of diffuse large B-cell lymphoma. Front Oncol 2022;12:822805. [PMID: 35982974 DOI: 10.3389/fonc.2022.822805]
- Usman WM, Pham TC, Kwok YY, Vu LT, Ma V, et al. Efficient RNA drug delivery using red blood cell extracellular vesicles. Nat Commun 2018;9(1):2359. [PMID: 29907766 DOI: 10.1038/s41467-018-04791-8]
- Liu Y, Zeng Y, Si HB, Tang L, Xie HQ, et al. Exosomes derived from human urine-derived stem cells overexpressing miR-140-5p alleviate knee osteoarthritis through downregulation of VEGFA in a rat model. Am J Sports Med 2022;50(4):1088-105. [PMID: 35179989 DOI: 10.1177/03635465221073991]
- Chen J, Huang T, Liu R, Wang C, Jiang H, et al. Congenital microtia patients: the genetically engineered exosomes released from porous gelatin methacryloyl hydrogel for downstream small RNA profiling, functional modulation of microtia chondrocytes and tissue-engineered ear cartilage regeneration. J Nanobiotechnology 2022;20(1):164. [PMID: 35346221 DOI: 10.1186/s12951-022-01352-6]
- Xu M, Yang Q, Sun X, Wang Y. Recent advancements in the loading and modification of therapeutic exosomes. Front Bioeng Biotechnol 2020;8:586130. [PMID: 33262977 DOI: 10.3389/fbioe.2020.586130]
- Fu S, Wang Y, Zhou J, Liu Y, Wan J, et al. Engineered EVs from 3D-cultured MSCs for synergistic modulation of inflammatory microenvironment and cartilage regeneration in osteoarthritis. ACS Appl Mater Interfaces 2025;17(30):42658-73. [PMID: 40673894 DOI: 10.1021/acsami.5c06750]
- Yang Z, Li X, Lin Q, Zhou F, Liang K, et al. Cortical actin depolymerisation in 3D cell culture enhances extracellular vesicle secretion and therapeutic effects. J Extracell Vesicles 2025;14(6):e70109. [PMID: 40560785 DOI: 10.1002/jev2.70109]
- Zhang H, Yang Y, Wu Y, Denslin V, Koh YWJ, et al. Unveiling the potential of MSC extracellular vesicles: MiR-122-5p enhancing chondrocyte regeneration in osteoarthritis via autophagy mechanism. Stem Cell Res Ther 2025;16(1):289. [PMID: 40483498 DOI: 10.1186/s13287-025-04412-4]
- Lin J, Li K, Yang Z, Cao F, Gao L, et al. Functionally improved mesenchymal stem cells via nanosecond pulsed electric fields for better treatment of osteoarthritis. J Orthop Translat 2024;47:235-48. [PMID: 39161657 DOI: 10.1016/j.jot.2024.03.006]
- Wang T, Zhao H, Zhang Y, Liu Y, Liu J, et al. A novel extracellular vesicles production system harnessing matrix homeostasis and macrophage reprogramming mitigates osteoarthritis. J Nanobiotechnology 2024;22(1):79. [PMID: 38419097 DOI: 10.1186/s12951-024-02324-8]
- Zhang C, Pathrikar TV, Baby HM, Li J, Zhang H, et al. Charge-reversed exosomes for targeted gene delivery to cartilage for osteoarthritis treatment. Small Methods 2024;8(9):e2301443. [PMID: 38607953 DOI: 10.1002/smtd.202301443]
- Pathrikar TV, Baby HM, Hakim B, Zhang H, Millán Cotto HA, et al. Cartilage-targeting exosomes for delivery of receptor antagonist of interleukin-1 in osteoarthritis treatment. Osteoarthritis Cartilage 2025;33(7):835-47. [PMID: 40158651 DOI: 10.1016/j.joca.2025.02.785]
- Kulkarni P, Deshpande S, Koppikar S, Patil S, Ingale D, et al. Glycosaminoglycan measured from synovial fluid serves as a useful indicator for progression of osteoarthritis and complements Kellgren-Lawrence score. BBA Clin 2016;6:1-4. [PMID: 27331021 DOI: 10.1016/j.bbacli.2016.05.002]
- Cheng W, Xu C, Su Y, Shen Y, Yang Q, et al. Engineered extracellular vesicles: a potential treatment for regeneration. iScience 2023;26(11):108282. [PMID: 38026170 DOI: 10.1016/j.isci.2023.108282]
- Liang Y, Xu X, Li X, Xiong J, Li B, et al. Chondrocyte-targeted microRNA delivery by engineered exosomes toward a cell-free osteoarthritis therapy. ACS Appl Mater Interfaces 2020;12(33):36938-47. [PMID: 32814390 DOI: 10.1021/acsami.0c10458]
- Wu X, Tao W, Lan Z, Tian Y, Zhong Z, et al. pH-responsive engineered exosomes enhance endogenous hyaluronan production by reprogramming chondrocytes for cartilage repair. Adv Healthc Mater 2025;14(10):e2405126. [PMID: 40042438 DOI: 10.1002/adhm.202405126]
- Sun Y, Wu H, Chen J, Xue C, Luo J, et al. Surface-engineered umbilical cord mesenchymal stem cell-derived sEVs for targeted therapy of osteoarthritis. ACS Biomater Sci Eng 2025;11(8):4725-36. [PMID: 40644710 DOI: 10.1021/acsbiomaterials.5c00306]
- Yan W, Li Y, Xie S, Tao WA, Hu J, et al. Chondrocyte-targeted delivery system of sortase A-engineered extracellular vesicles silencing MMP13 for osteoarthritis therapy. Adv Healthc Mater 2024;13(16):e2303510. [PMID: 38545904 DOI: 10.1002/adhm.202303510]
- Deng S, Chen X, Cao H, Chen M, Shi W, et al. Self-assembled bifunctional exosomes simplify chondrocyte-targeted sustained-release systems in decellularized cartilage microgels for synergistic osteoarthritis therapy. Small 2025;21(38):e03492. [PMID: 40765307 DOI: 10.1002/smll.202503492]
- Wang P, Zhao H, Chen W, Guo Y, Zhang S, et al. Cell-free osteoarthritis treatment with dual-engineered chondrocyte-targeted extracellular vesicles derived from mechanical loading primed mesenchymal stem cells. J Tissue Eng 2025;16:20417314241312563. [PMID: 39926048 DOI: 10.1177/20417314241312563]
- Kim JY, Rhim WK, Lee SY, Park JM, Song DH, et al. Hybrid nanoparticle engineered with transforming growth factor -β1-overexpressed extracellular vesicle and cartilage-targeted anti-inflammatory liposome for osteoarthritis. ACS Nano 2024;18(50):33937-52. [PMID: 39648484 DOI: 10.1021/acsnano.4c07992]
- Andrieu J, Re F, Russo L, Nicotra F. Phage-displayed peptides targeting specific tissues and organs. J Drug Target 2019;27(5-6):555-65. [PMID: 30281393 DOI: 10.1080/1061186x.2018.1531419]
- Wu Y, Feng Y, Hu F, Zheng X, Ding Y, et al. Engineered stem cell clusters for extracellular vesicles-mediated gene delivery to rejuvenate chondrocytes and facilitate chondrogenesis in osteoarthritis therapy. Adv Sci (Weinh) 2025;12(25):e2500964. [PMID: 40278049 DOI: 10.1002/advs.202500964]
- Xu G, Jin J, Fu Z, Wang G, Lei X, et al. Extracellular vesicle-based drug overview: research landscape, quality control and nonclinical evaluation strategies. Signal Transduct Target Ther 2025;10(1):255. [PMID: 40804047 DOI: 10.1038/s41392-025-02312-w]
- Jing Z, Zhang G, Cai Y, Liang J, Lv L, et al. Engineered extracellular vesicle-delivered TGF-β inhibitor for attenuating osteoarthritis by targeting subchondral bone. J Tissue Eng 2024;15:20417314241257781. [PMID: 39071897 DOI: 10.1177/20417314241257781]
- Peruzzi JA, Gunnels TF, Edelstein HI, Lu P, Baker D, et al. Enhancing extracellular vesicle cargo loading and functional delivery by engineering protein-lipid interactions. Nat Commun 2024;15(1):5618. [PMID: 38965227 DOI: 10.1038/s41467-024-49678-z]
- O’Brien TJ, Hollinshead F, Goodrich LR. Extracellular vesicles in the treatment and prevention of osteoarthritis: can horses help us translate this therapy to humans? Extracell Vesicles Circ Nucl Acids 2023;4(2):151-69. [PMID: 37829144 DOI: 10.20517/evcna.2023.11]
- Zhang X, Che X, Zhang S, Wang R, Li M, et al. Mesenchymal stem cell-derived extracellular vesicles for human diseases. Extracell Vesicles Circ Nucl Acids 2024;5(1):64-82. [PMID: 39698413 DOI: 10.20517/evcna.2023.47]
- Goo J, Lee Y, Lee J, Kim IS, Jeong C. Extracellular vesicles in therapeutics: a comprehensive review on applications, challenges, and clinical progress. Pharmaceutics 2024;16(3):311. [PMID: 38543204 DOI: 10.3390/pharmaceutics16030311]
- Martel-Pelletier J, Barr AJ, Cicuttini FM, Conaghan PG, Cooper C, et al. Osteoarthritis. Nat Rev Dis Primers 2016;2:16072. [PMID: 27734845 DOI: 10.1038/nrdp.2016.72]
- Gandham S, Su X, Wood J, Nocera AL, Alli SC, et al. Technologies and standardization in research on extracellular vesicles. Trends Biotechnol 2020;38(10):1066-98. [PMID: 32564882 DOI: 10.1016/j.tibtech.2020.05.012]
- Phan J, Kumar P, Hao D, Gao K, Farmer D, et al. Engineering mesenchymal stem cells to improve their exosome efficacy and yield for cell-free therapy. J Extracell Vesicles 2018;7(1):1522236. [PMID: 30275938 DOI: 10.1080/20013078.2018.1522236]
- Luo L, Du Y, Niu X, Liu J, Yuan J, et al. Optimized tangential flow filtration for efficient isolation of highly pure and bioactive sEVs. Anal Chem 2025;97(31):17261-74. [PMID: 40746027 DOI: 10.1021/acs.analchem.5c03781]
- Duan X, Zhang R, Feng H, Zhou H, Luo Y, et al. A new subtype of artificial cell-derived vesicles from dental pulp stem cells with the bioequivalence and higher acquisition efficiency compared to extracellular vesicles. J Extracell Vesicles 2024;13(7):e12473. [PMID: 38965648 DOI: 10.1002/jev2.12473]
- Ye Q, Zhang R. Intracellular vesicles: novel nanovesicles superior to extracellular vesicles in translational medicine and clinical applications. Nano TransMed 2024;3:100044. [DOI: 10.1016/j.ntm.2024.100044]
- Gupta D, Zickler AM, El Andaloussi S. Dosing extracellular vesicles. Adv Drug Deliv Rev 2021;178:113961. [PMID: 34481030 DOI: 10.1016/j.addr.2021.113961]
- Aimaletdinov AM, Gomzikova MO. Tracking of extracellular vesicles’ biodistribution: new methods and approaches. Int J Mol Sci 2022;23(19):11312. [PMID: 36232613 DOI: 10.3390/ijms231911312]
- Zhan J, Zou J, Pang Q, Chen Z, Liu J, et al. MSCs-EVs harboring OA immune memory reprogram macrophage phenotype via modulation of the mt-ND3/NADH-CoQ axis for OA treatment. J Nanobiotechnology 2025;23(1):140. [PMID: 40001168 DOI: 10.1186/s12951-025-03216-1]
- Philippon M Jr, Labib R, Ley MBRG, Kaplan LD, Mendez AJ, et al. Characterization of extracellular vesicles from infrapatellar fat pad mesenchymal stem/stromal cells expanded using regulatory-compliant media and inflammatory/hormonal priming. Cells 2025;14(10):706. [PMID: 40422209 DOI: 10.3390/cells14100706]
- Wu J, Wu J, Xiang W, Gong Y, Feng D, et al. Engineering exosomes derived from TNF-α preconditioned IPFP-MSCs enhance both yield and therapeutic efficacy for osteoarthritis. J Nanobiotechnology 2024;22(1):555. [PMID: 39261846 DOI: 10.1186/s12951-024-02795-9]
- Shao L, Ding L, Li W, Zhang C, Xia Y, et al. Let-7a-5p derived from parathyroid hormone (1-34)-preconditioned BMSCs exosomes delays the progression of osteoarthritis by promoting chondrocyte proliferation and migration. Stem Cell Res Ther 2025;16(1):299. [PMID: 40490830 DOI: 10.1186/s13287-025-04416-0]
- Lu M, Lou A, Gao J, Li S, He L, et al. Quercetin-primed MSC exosomes synergistically attenuate osteoarthritis progression. J Orthop Surg Res 2025;20(1):373. [PMID: 40229791 DOI: 10.1186/s13018-025-05785-1]
- Chen M, Liu Y, Cao Y, Zhao C, Liu Q, et al. Remodeling the proinflammatory microenvironment in osteoarthritis through interleukin-1 beta tailored exosome cargo for inflammatory regulation and cartilage regeneration. ACS Nano 2025;19(4):4924-41. [PMID: 39848926 DOI: 10.1021/acsnano.4c16785]
- Yuan W, Liu J, Zhang Z, Ye C, Zhou X, et al. Strontium-Alix interaction enhances exosomal miRNA selectively loading in synovial MSCs for temporomandibular joint osteoarthritis treatment. Int J Oral Sci 2025;17(1):6. [PMID: 39890774 DOI: 10.1038/s41368-024-00329-5]
- Liu W, Liu A, Li X, Sun Z, Sun Z, et al. Dual-engineered cartilage-targeting extracellular vesicles derived from mesenchymal stem cells enhance osteoarthritis treatment via miR-223/NLRP3/pyroptosis axis: toward a precision therapy. Bioact Mater 2023;30:169-83. [PMID: 37593145 DOI: 10.1016/j.bioactmat.2023.06.012]
- Zhang H, Yan W, Wang J, Xie S, Tao WA, et al. Surface functionalization of exosomes for chondrocyte-targeted siRNA delivery and cartilage regeneration. J Control Release 2024;369:493-505. [PMID: 38582335 DOI: 10.1016/j.jconrel.2024.04.009]
- Wan J, He Z, Peng R, Wu X, Zhu Z, et al. Injectable photocrosslinking spherical hydrogel-encapsulated targeting peptide-modified engineered exosomes for osteoarthritis therapy. J Nanobiotechnology 2023;21(1):284. [PMID: 37605203 DOI: 10.1186/s12951-023-02050-7]
- Chen L, Liu J, Rao Z. FTO-overexpressing extracellular vesicles from BM-MSCs reverse cellular senescence and aging to ameliorate osteoarthritis by modulating METTL3/YTHDF2-mediated RNA m6A modifications. Int J Biol Macromol 2024;278(Pt 1):134600. [PMID: 39122063 DOI: 10.1016/j.ijbiomac.2024.134600]
- Cao B, Liu G, Gao K, Fan W, Zhao W, et al. The role of extracellular vesicles derived from microRNA-146a-modified mesenchymal stem cells in modulating inflammation in experimental glenohumeral osteoarthritis. Iran J Allergy Asthma Immunol 2024;23(5):578-87. [PMID: 39586750 DOI: 10.18502/ijaai.v23i5.16752]
- Liu Y, Huang K, Zhou SL, Li S, Si HB, et al. Engineered exosomal miR140 modulates mitophagy of chondrocytes through targeting CAPN1 to alleviate osteoarthritis. Sci China Life Sci 2025;68(9):2617-34. [PMID: 40506565 DOI: 10.1007/s11427-024-2843-7]
- Chen M, Lu Y, Liu Y, Liu Q, Deng S, et al. Injectable microgels with hybrid exosomes of chondrocyte-targeted FGF18 gene-editing and self-renewable lubrication for osteoarthritis therapy. Adv Mater 2024;36(16):e2312559. [PMID: 38266145 DOI: 10.1002/adma.202312559]
- Hu X, Xie J, Su J. Lacc1-engineered extracellular vesicles reprogram mitochondrial metabolism to alleviate inflammation and cartilage degeneration in TMJ osteoarthritis. J Nanobiotechnology 2025;23(1):276. [PMID: 40186254 DOI: 10.1186/s12951-025-03355-5]
- Cao H, Li W, Zhang H, Hong L, Feng X, et al. Bio-nanoparticles loaded with synovial-derived exosomes ameliorate osteoarthritis progression by modifying the oxidative microenvironment. J Nanobiotechnology 2024;22(1):271. [PMID: 38769545 DOI: 10.1186/s12951-024-02538-w]
- Yang F, Xiong WQ, Li CZ, Wu MJ, Zhang XZ, et al. Extracellular vesicles derived from mesenchymal stem cells mediate extracellular matrix remodeling in osteoarthritis through the transport of microRNA-29a. World J Stem Cells 2024;16(2):191-206. [PMID: 38455098 DOI: 10.4252/wjsc.v16.i2.191]