Low-intensity Pulsed Ultrasound: A Promising Strategy for the Treatment of POI
1Key Laboratory of Medical Imaging Precision Theranostics and Radiation Protection, College of Hunan Province, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
2Institute of Medical Imaging, Hengyang Medical School, University of South China, Hengyang, China
3Department of Medical Imaging, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
4The Seventh Affiliated Hospital, Hunan Veterans Administration Hospital, Hengyang Medical School, University of South China, Changsha, Hunan, China
5School of Biomedical Engineering & Imaging Sciences, King’s College London, London, United Kingdom
aThese authors contributed equally to this work.
*Correspondence to: Weidong Liang, School of Biomedical Engineering and Imaging Sciences, King’s College London, United Kingdom, E-mail: weidong.liang@kcl.ac.uk
Received: July 16 2025; Revised: September 30 2025; Accepted: December 7 2025; Published Online: February 2 2026
Cite this paper:
Zhang Z, Kuang C, Guo Z et al. Low-intensity Pulsed Ultrasound: A Promising Strategy for the Treatment of POI. BIO Integration 2026; 7: 1–26.
DOI: 10.15212/bioi-2025-0131. Available at: https://bio-integration.org/
Download citation
© 2026 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Premature ovarian insufficiency (POI) is a complex endocrine disorder characterized by premature depletion of ovarian follicles, and resulting in ovarian failure and decreased fertility. Conventional hormone replacement therapy (HRT) alleviates menopausal symptoms but carries potential risks, such as breast malignancies, and does not restore ovarian endocrine function. As a noninvasive physical therapy, low-intensity pulsed ultrasound (LIPUS) regulates cell proliferation, apoptosis, inflammation, and angiogenesis through mechanical stress, cavitation effects, and microstreaming, thus providing a novel therapeutic avenue for POI. In this review, we systematically analyze recent advances in the application of LIPUS in POI, demonstrating its mechanism of regulating ovarian function. We also analyze the combination of LIPUS with other therapies for POI and discuss prospects for LIPUS-based treatments.
Keywords
Low-intensity pulsed ultrasound, premature ovarian insufficiency, stem cell therapy, tissue restoration, traditional Chinese medicine.
Introduction
Premature ovarian insufficiency (POI), a complex endocrine disorder characterized by the loss of ovarian function before the age of 40 years, is marked by elevated follicle-stimulating hormone (FSH) levels (>25 IU/L), and diminished anti-Müllerian hormone (AMH) and estradiol (E2) concentrations [1]. In 1–3% of women of reproductive age globally, POI leads to infertility, menopausal symptoms, and long-term health risks, such as osteoporosis and cardiovascular disease [2]. The pathogenesis of POI is multifactorial and involves genetic abnormalities, autoimmune disorders, iatrogenic interventions (e.g., chemotherapy or radiation), and idiopathic causes [3]. Early diagnosis and intervention are critical to mitigate irreversible ovarian atrophy and preserve fertility; however, delayed identification often exacerbates follicular depletion [4].
Current clinical management of POI relies primarily on hormone replacement therapy (HRT) to alleviate hypoestrogenic symptoms. Although HRT improves quality of life, it cannot restore ovarian endocrine function or fertility, and is associated with risks of thromboembolism, breast cancer, and hormone dependency with prolonged use [5, 6]. Alternative approaches, such as oocyte donation, offer fertility solutions but face ethical, genetic, and financial barriers. Ovarian cryopreservation is limited by age-dependent efficacy and tumor recurrence risk [7, 8]. Emerging therapies, including platelet-rich plasma (PRP) and mesenchymal stem cells (MSCs), have shown promise in preclinical studies but face translational challenges such as oncogenic risks, high costs, and standardization issues [6, 9]. Physical modalities such as lasers and electrical stimulation have been explored but are limited by poor tissue penetration or invasiveness [10]. These limitations underscore the need for noninvasive, safe, and effective therapies to address both endocrine dysfunction and follicular regeneration in patients with POI.
Low-intensity pulsed ultrasound (LIPUS), a noninvasive therapeutic modality, has demonstrated broad clinical utility across diverse medical fields. LIPUS operates at frequencies of 1–3 MHz and low intensities (<1 W/cm2), and applies mechanical stimuli through pulsed sound waves, thereby promoting tissue repair without thermal damage [11]. The clinical utility of LIPUS was recognized in its FDA approval for accelerating fracture healing, in which it promotes bone regeneration and decreases recovery time in nonunion fractures [12]. Its applications extend to cartilage repair, by mitigating degenerative joint damage. Owing to this clinical success, applications of LIPUS have expanded to other medical fields. For example, in musculoskeletal disorders, LIPUS has shown efficacy in mitigating degenerative joint damage through cartilage repair and facilitating tendon repair [13–15]. Similarly, its benefits extend to soft tissue regeneration, such as in chronic wound healing. These established successes across diverse tissues underscore the versatility of LIPUS in addressing structural and functional deficits via noninvasive mechanobiological interactions [16]. Given this broad regenerative potential, emerging research has explored LIPUS in reproductive medicine. Preclinical studies now suggest its ability to improve ovarian reserve, suppress apoptosis, and decrease fibrosis in models of ovarian dysfunction, including POI [17]. Because of its demonstrated safety profile and multitarget regenerative capabilities, LIPUS provides a novel, hormone-free strategy to counteract ovarian degeneration [15]. Its nonpharmacological nature and lack of systemic adverse effects make it a promising alternative to conventional therapies for POI.
This review systematically evaluates the therapeutic potential of LIPUS in POI. We first outline the clinical challenges and limitations of existing treatments. Next, we elucidate the mechanobiological mechanisms through which LIPUS regulates ovarian function. We further discuss synergistic strategies that combine LIPUS with stem cell therapy or traditional Chinese medicine (TCM) to improve therapeutic outcomes. Finally, we identify knowledge gaps and propose future directions for optimizing LIPUS parameters, validating clinical efficacy, and integrating multimodal therapies. By synthesizing preclinical evidence, this review is aimed at advancing LIPUS as a cornerstone for next-generation POI management.
POI clinical management and LIPUS use
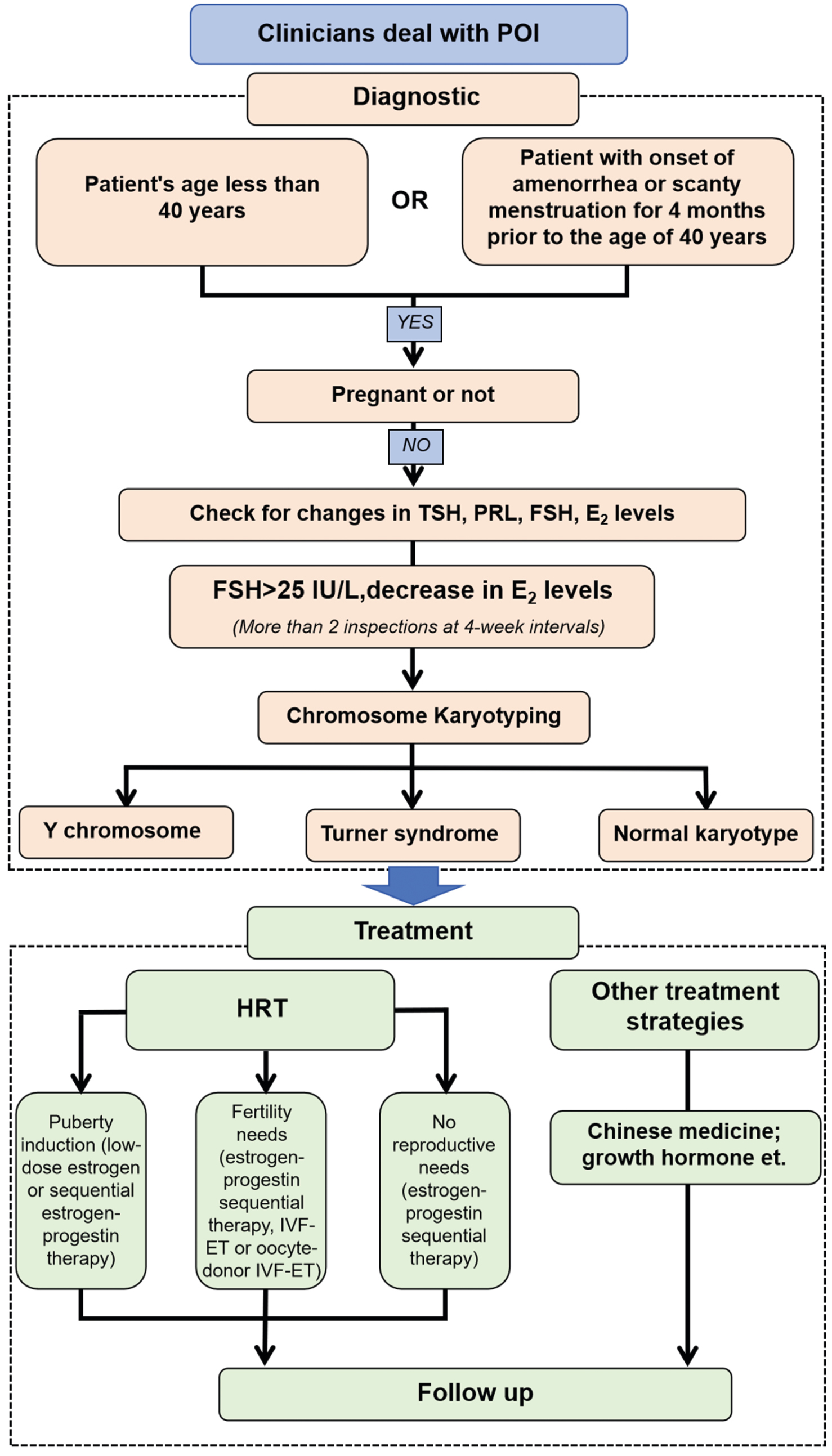
POI affects 1–3% of reproductive-aged women globally, and 1% population develop functional decline before age 30; the peak incidence occurs at 35–40 years [18, 19]. Its pathogenesis involves genetic (10–30%), iatrogenic (5–30%), and autoimmune (10–30%) factors [20]. This multifactorial etiology highlights the critical need for precision medicine approaches in POI treatment. These factors are also key determinants of the clinical treatment process for POI (Figure 1).
Figure 1 Clinical treatment POI flowchart. Source: Created by the authors.
Current clinical modalities
HRT remains the primary POI treatment. However, it cannot fully replicate ovarian endocrine function [21], and it carries risks of thromboembolism, stroke, and gynecological malignancies with prolonged use [22, 23]. More importantly, HRT does not effectively restore ovarian function or provide patients with hope for fertility. Although oocyte donation offers some patients the possibility of fertility, ethical controversies, genetic considerations, and high costs limit its application [24]. Ovarian cryopreservation technology has shown potential for treating drug-induced POI and addresses the ethical concerns associated with oocyte donation. Nevertheless, age-dependent effects and the risk of tumor cell recovery hinder its widespread application [25]. Although TCM has some regenerative potential, its clinical implementation is hindered by standardization issues [26]. Emerging biological therapies (platelet-rich plasma and stem cell treatments) face translational barriers of cost, oncogenic risks, and ethical controversies [27].
Emerging technologies
Innovative physical modalities are under investigation to address the limitations of conventional therapies and shift the treatment paradigm from symptom management to functional ovarian restoration. Several approaches have shown promise yet face considerable translational challenges. For example, laser therapy demonstrates tissue repair capabilities through photo-biomodulation, which can decrease oxidative stress and inflammation [13, 28]. However, its clinical application in POI is severely limited by its poor depth of tissue penetration, which renders it ineffective for targeting deeply located ovarian structures without invasive delivery systems [28]. Similarly, electrical stimulation modulates cellular activity and has been shown in models to mitigate ovarian damage by inhibiting primordial follicle loss and inducing antioxidant systems [29]. Nevertheless, this technique typically requires implanted electrodes to deliver current effectively and therefore is an invasive procedure. This invasiveness prompts biosafety concerns, including risks of infection, tissue damage, and fibrotic encapsulation, which are particularly undesirable for chronic conditions such as POI, which may require repeated interventions [10].
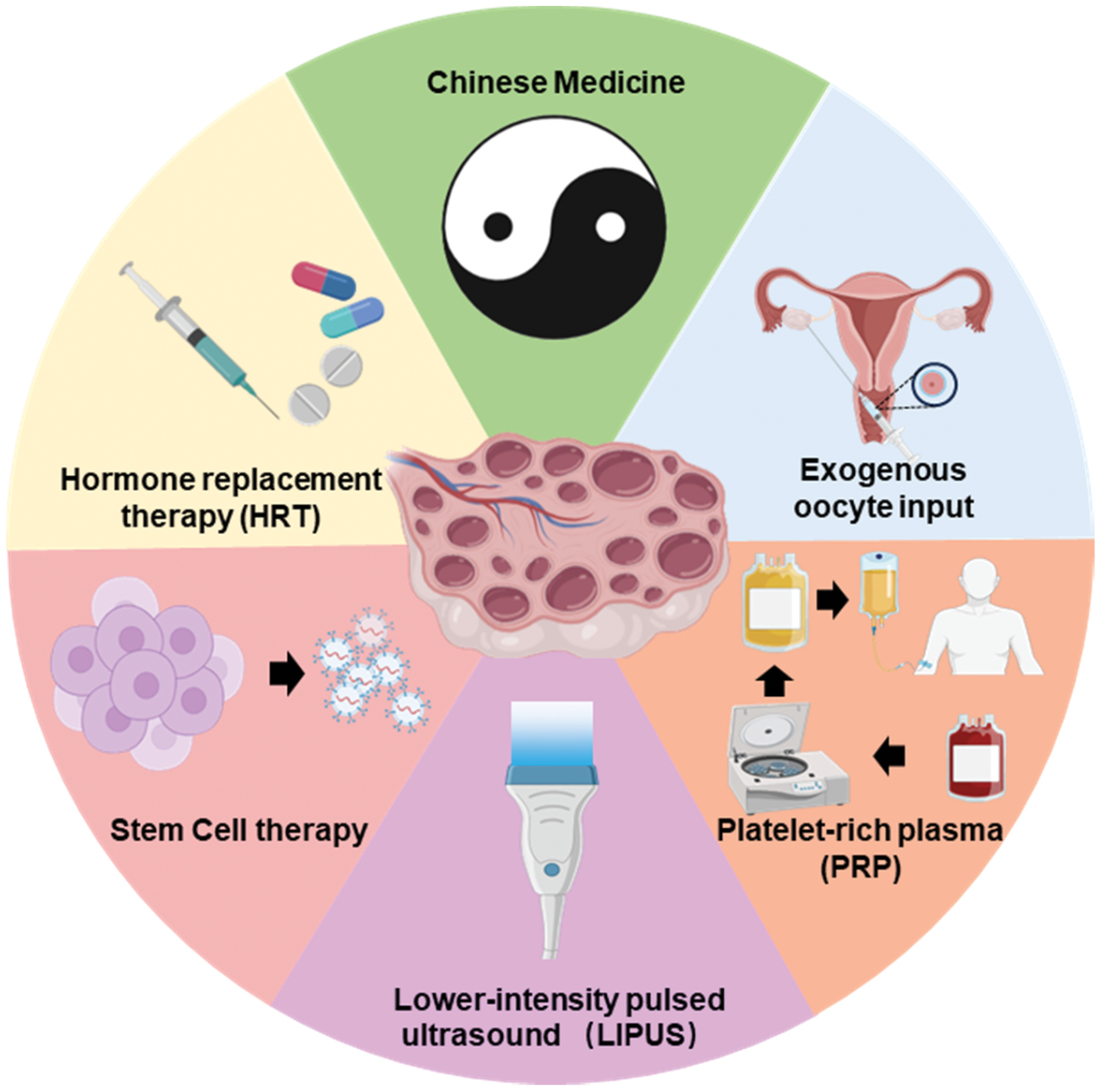
In contrast, LIPUS has emerged as a uniquely promising noninvasive alternative. This modality effectively addresses the key limitations of the aforementioned modalities, through its superior tissue penetration capabilities to reach the ovaries noninvasively, while eliminating the risks associated with implanted devices. LIPUS exerts therapeutic effects through mechanobiological interactions, including stable cavitation and acoustic streaming, that increase ovarian perfusion, suppress granulosa cell (GC) apoptosis, and decrease inflammation and fibrosis without thermal damage [9, 15]. This combination of deep penetration, non-invasiveness, and multi-faceted action on key pathological processes of POI positions LIPUS as a leading candidate among the physical modalities for achieving true ovarian rejuvenation.
The dichotomy between symptom management and functional restoration underscores the need for innovative solutions. Whereas current modalities prioritize symptom alleviation, emerging technologies such as LIPUS are aimed at rejuvenating ovarian microenvironments, thereby offering a paradigm shift toward curative interventions (Figure 2).
Figure 2 Treatment options for POI include HRT, TCM, exogenous oocyte input, PRP, LIPUS, and stem cell therapy. Source: Created by the authors via Biorender.com.
Mechanism of action of LIPUS
The therapeutic efficacy of LIPUS arises from its well-defined biophysical interactions with tissues. A key consideration for its clinical translation, particularly for deep-seated organs such as the ovaries, is its tissue penetration ability. As ultrasound waves propagate through the body, their energy is attenuated through mechanisms of reflection, scattering, and absorption, all of which are frequency-dependent [30]. A critical trade-off therefore exists: lower frequencies (e.g., 0.5–1 MHz) achieve greater penetration depth but offer lower spatial resolution, whereas higher frequencies (e.g., 1–3 MHz) provide finer resolution at the expense of shallower penetration [31, 32]. Consequently, the selection of LIPUS parameters, particularly their frequency, must be tailored to the target anatomy.
The safety and dosing rationale for LIPUS are based on an extensive clinical history. For example, the FDA-approved LIPUS device for accelerated fracture healing (Exogen® system) uses a frequency of 1.5 MHz, an intensity of 30 mW/cm2 (spatial average, temporal average), and a 20% duty cycle [33]. This specific set of low-intensity, pulsed parameters is deliberately chosen to maximize beneficial non-thermal bioeffects (e.g., stable cavitation and acoustic streaming) while rigorously avoiding thermal damage or tissue disruption. The success of this regimen in bone repair provides a strong foundation for exploring similar low-intensity parameters in other regenerative contexts, including POI.
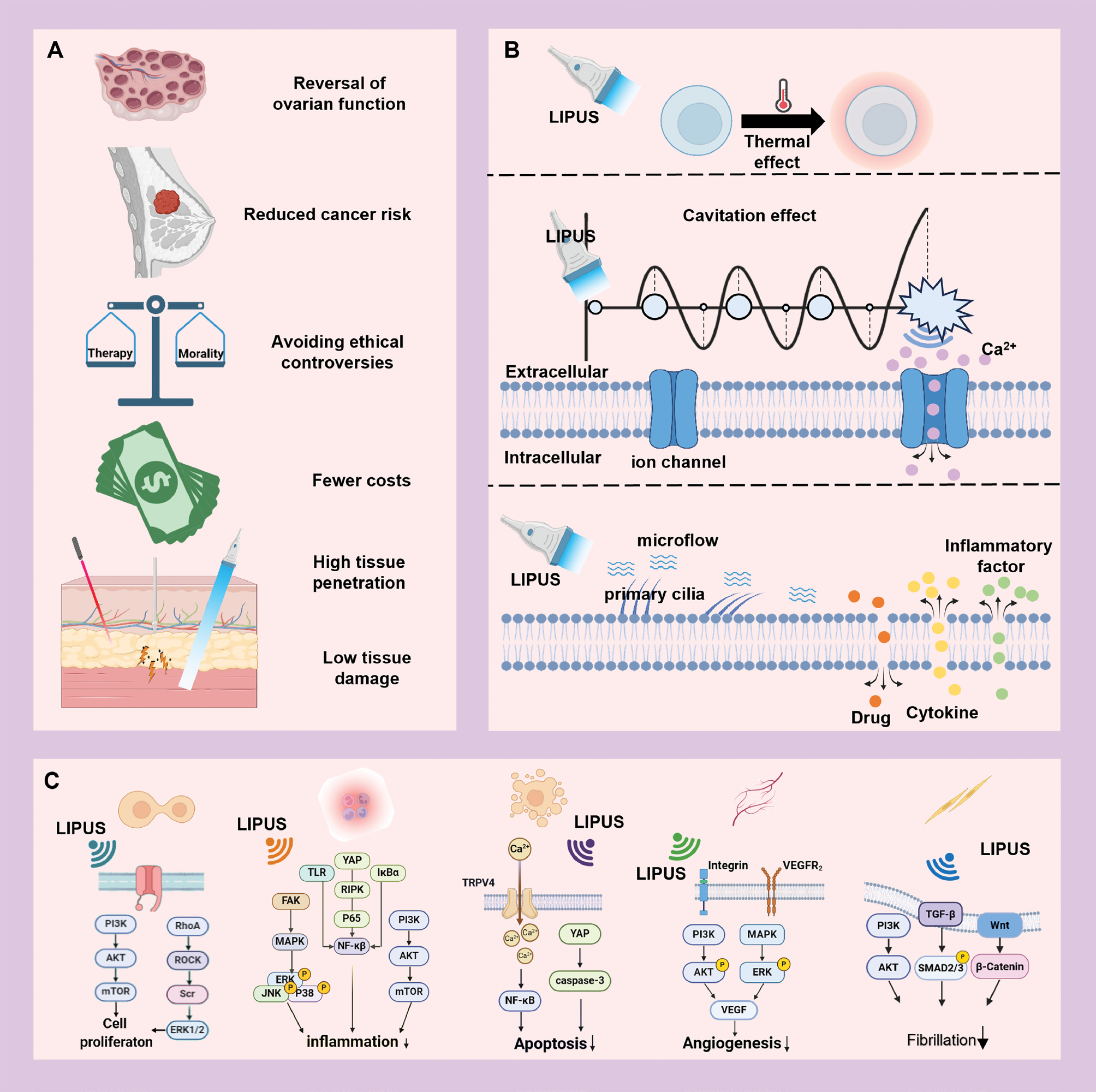
LIPUS delivers controlled mechanical stimuli through biomechanical interactions with cellular components, thus triggering intracellular biochemical responses that facilitate tissue repair and regeneration [16]. In recent years, LIPUS has garnered substantial attention in reproductive medicine, particularly its application in treating POI, in which it has demonstrated a unique combination of benefits. LIPUS offers substantial advantages over traditional therapies and innovative biotechnological approaches (Figure 3).
Figure 3 LIPUS in the treatment of POI. A. Advantages of LIPUS over other treatment modalities in POI treatment. B. LIPUS biological effects of ultrasound in regulating cellular activity. C. Mechanisms underlying the modulation of cellular activity by LIPUS. Source: Created by the authors via Biorender.com.
Potential biological effects of LIPUS for POI treatment
Ultrasonic modalities are broadly categorized into diagnostic and therapeutic applications. Within therapeutic ultrasound paradigms, the biological effects are mediated primarily through thermal mechanisms and nonthermal phenomena, the latter of which encompass acoustic cavitation and mechanical stress. Therapeutic applications of distinct ultrasound-induced biological effects demonstrate modality-specific implementation paradigms (Table 1). These dual mechanisms collectively enable LIPUS to regulate cellular homeostasis and facilitate tissue regeneration, thereby establishing a therapeutic foundation for POI management.
Table 1 Applications of the Biological Effects of Ultrasound
| Main Biological effects | Ultrasound Parameters | Application | Reference | |||
|---|---|---|---|---|---|---|
| Frequency (MHz) | Duty Cycle (%) | Intensity (W/cm2) | Time (Min) | |||
| Thermal effect | 3 | Unclear | 1 | 5 | Tissue repair | [125] |
| Unclear | 4 | 4.8 | 5 | Improved tissue perfusion | [126] | |
| 1.21 | Unclear | 99–102 | 0.5 | Tissue thermal ablation | [127] | |
| 3.3 | 2 | 60 | 1/3 | Tissue mechanical ablation | [128] | |
| Cavitation effect | 3 | 10 | 2.2 | 5 | Drug delivery | [129] |
| 1.5 | Unclear | 6.75 | 1 | Blood-brain barrier opening | [130] | |
| 1 | 20 | 0.8 | 1 | Sonoporation-enhanced gene delivery | [131] | |
| Mechanical effect | 1 | Unclear | 0.2 | 1/6 | Enhanced enhancing cell membrane permeability due to shear stress generated via microbubble oscillation | [132] |
| 0.4 | 7.5 | 5.3 | 10 | Acoustic streaming effect, promoting solute transport | [133] | |
| 3 | 50 | 0.1 | 15 | Osteogenesis promotion in BMMSCs, through cytoskeleton stimulation by mechanical stress | [134] | |
Thermal effect
LIPUS generates thermoacoustic effects by stimulating the formation, growth, and implosive collapse of bubbles in a liquid [30]. LIPUS achieves parameter-controlled thermogenic safety in POI treatment through the selective absorption and conversion of ultrasound energy in biological tissues [34]. This process results in tissue-specific bioeffects due to differential acoustic absorption coefficients across cellular components. The inherent safety of this modality derives from low-intensity pulsed delivery, which maintains nondamaging cellular responses through controlled thermal accumulation [35]. The ovarian parenchyma, which is anatomically enveloped within the adipose-rich stroma, exhibits a negligible thermal response to LIPUS exposure.
Cavitation effect
LIPUS modulates ovarian cell activity primarily through the nonthermal effects of ultrasound. The cavitation effect is an essential nonthermal effect of ultrasound. Cavitation can be categorized into two types: transient cavitation and steady-state cavitation. The type and mechanism of cavitation directly influence its biological effects and safety. When steady-state cavitation occurs, the bubbles oscillate steadily in the acoustic field, thus generating microfluidic shear and cyclic mechanical stresses; triggering the opening of calcium channels in the cell membrane; and activating repair signaling pathways [36]. Steady-state cavitation does not cause destructive thermal or chemical damage, and it promotes tissue regeneration through mechanical–biological coupling [37].
LIPUS precisely regulates the acoustic field energy through low-intensity and pulsed emission, and preferentially induces steady-state cavitation while suppressing the high-risk effects associated with inertial cavitation [38]. This approach enables LIPUS to avoid damaging cellular structures during treatment, because the mechanical stimulation from steady-state cavitation does not depend on bubble collapse, thereby circumventing potential high temperatures and free radical damage [39]. The parametric optimization inherent in LIPUS technology effectively confines energy deposition at the focal site and is particularly advantageous for the noninvasive regeneration of mechanosensitive tissues such as the ovarian parenchyma. Current experimental evidence has demonstrated that shear stress induced by stable cavitation regimes activates mechanotransductive ion channels in GCs and oocytes [40].
LIPUS-generated microstreaming and radiation forces act on the cell membrane by exerting mechanical forces that are transduced into biochemical signals, in a process known as mechanotransduction. A critical early event in this process is the influx of calcium ions (Ca2+), which can initiate downstream signaling pathways such as the PI3K/AKT/mTOR pathway [41, 42].
Although the specific mechanosensitive calcium channels (e.g., TRPV4 and Piezo1) that mediate this Ca2+ influx in ovarian cells in response to LIPUS have not yet been definitively identified, their fundamental roles in ovarian mechanobiology are increasingly recognized. For example, mechanical stress has been shown to accompany nuclear rotation in dormant mouse oocytes, and the biomechanical properties of ovarian tissue are acknowledged as key factors influencing follicular growth [43]. Furthermore, studies in non-ovarian cell types have provided compelling evidence that LIPUS activates TRPV4 channels in chondrocytes [44] and ASIC1a in neurons [45]. The precise modulation of stable cavitation thresholds by LIPUS is likely to initiate similar mechanotransductive cascades in ovarian cells and ultimately activate evolutionarily conserved pathways, including the PI3K/AKT/mTOR and Hippo/YAP pathway, that coordinately suppress follicular atresia and promote primordial follicle recruitment. Through this proposed mechanobiological axis, LIPUS achieves therapeutic efficacy in POI management while concurrently maintaining an exceptional biosafety profile. Simultaneously, mechanical forces are transmitted via integrins to focal adhesion complexes, thus triggering cytoskeletal reorganization, notably F-actin polymerization and increased tension. This remodeling inhibits Hippo pathway kinases, and results in YAP/TAZ dephosphorylation and nuclear translocation. Nuclear YAP/TAZ then binds transcription factors such as TEAD, thus activating target genes (e.g., CCND1, CTGF, and Bcl-2) that promote GC proliferation and inhibit apoptosis. The parallel activation of pathways such as PI3K/AKT synergizes with YAP/TAZ signaling in collectively mediating LIPUS-induced ovarian protection. Through these mechanisms, LIPUS activates evolutionarily conserved pathways, including PI3K/AKT/mTOR and Hippo/YAP, that coordinately suppress follicular atresia and promote primordial follicle recruitment [41, 42]. By precisely modulating stable cavitation thresholds, LIPUS achieves therapeutic efficacy in POI management through this mechanobiological axis and concurrently maintains exceptional biosafety profiles.
Mechanical effects
Acoustic streaming, another important nonthermal effect of ultrasound, also plays an important role in the modulation of ovarian cell activity by LIPUS. This fluid motion is induced by acoustic radiation forces, primarily through nonlinear wave propagation and bubble oscillation dynamics. Acoustic microstreaming, involving eddy currents adjacent to the source of oscillation, is a type of acoustic flow of sufficient intensity to alter membrane permeability and stimulate cellular activity at the interface between the cell membrane and tissue fluid. This alteration can modify diffusion rates, membrane permeability, protein synthesis, and secretory processes in cells [46]. The acoustic streaming phenomenon generated by LIPUS elicits multimodal therapeutic effects in early-onset POI through biomechanical modulation. This ultrasonic-induced microfluidic shear stress directly activates vascular endothelial mechanoreceptors, by triggering nitric oxide synthase-mediated vasodilation and consequently enhancing ovarian microcirculation—a mechanism with therapeutic potential for ameliorating follicular ischemia and hypoxia–reoxygenation injury [47, 48].
As a nonthermal therapeutic modality, LIPUS delivers low-intensity acoustic energy with minimal heat generation. LIPUS regulates cellular activity primarily through the mechanical stimulation of cells caused by the cavitation effect and acoustic flow. Through these mechanical stimuli, ultrasound can open the ion channels indicated by the cells and activate the signaling pathways inside the cells to achieve regulation of cellular activities, or can affect cells by stimulating force-sensitive elements on the cell surface and changing the biomechanics of the cells [49, 50].
Mechanisms underlying the modulation of cellular activity by LIPUS
LIPUS has demonstrated broad therapeutic efficacy across various biological systems, including musculoskeletal repair, wound healing, and neural regeneration [51]. Whereas mechanistic investigations have focused primarily on nonovarian contexts, emerging evidence underscores its translational potential for managing ovarian insufficiency. The mechanobiological effects of this approach and its transcriptional regulation of proliferative and apoptotic pathways are the underlying mechanisms [52, 53]. Additionally, LIPUS has a multitarget regulatory ability for modulating angiogenesis, exerting antifibrotic effects, and promoting inflammation resolution. These synergistic effects enhance ovarian perfusion and functional restoration, and position LIPUS as a novel noninvasive intervention for ovarian insufficiency.
Regulation of cell proliferation
Granulosa Cells (GCs), the primary functional cells that surround oocytes within the follicle, provide trophic factors to oocytes through gap junctions, support oocyte metabolism, and decrease oocyte apoptosis [54, 55]. Follicular atresia and ovarian failure occur when insufficient GC proliferation manifests. In contrast, promoting GC proliferation might potentially ameliorate POI [56].
LIPUS can promote cell proliferation effectively [57]. LIPUS has been found to increase cellular proliferation through irradiation of subcellular structures without inducing cytotoxicity or inflammation [58]. For example, Lim et al. have demonstrated that ASIC1a and cytoskeletal proteins are involved in low-intensity ultrasound-mediated mechanotransduction and neuronal activation [59]. Takeuchi et al. have reported that LIPUS exerts mechanical effects on the integrin/PI3K/Akt pathway in chondrocytes, thus increasing expression of type IX collagen, cyclin B1, and cyclin D1, and promoting cell proliferation [60]. Duan et al. have reported that LIPUS significantly increases the number of satellite cells, and facilitates repair and regeneration after muscle injury, by regulating the PGC-1α/AMPK/GLUT4 signaling pathway through mechanical effects [61]. Huang et al. have reported that LIPUS promotes Schwann cell proliferation by activating the NRG1/ErbB signaling pathway [62]. Additionally, LIPUS activates integrin receptors, which subsequently trigger the Rho/ROCK/ERK signaling pathway and further increase cell proliferation [63].
Regulation of apoptosis and inflammation
Mechanistically, POI pathogenesis is driven by reciprocally reinforced GC apoptosis and chronic inflammatory responses, which create self-perpetuating cascades that potentiate ovarian functional decline [64, 65]. GC apoptosis directly disrupts the follicular microenvironment; activates immune cells; and triggers the secretion of proinflammatory factors that further exacerbate GC apoptosis through oxidative stress and death receptor signaling [66–68]. Additionally, inflammation-induced autoimmune attacks further constrict the follicular survival space, and consequently worsen GC hypoxia and metabolic disorders [64, 69]. The observed link between markers of GC apoptosis and inflammatory factor levels highlights the therapeutic potential of targeting the apoptosis–inflammation axis. This approach might disrupt the cycle and offer new strategies for treatment [70].
LIPUS inhibits apoptosis through multiple pathways. Wu et al. have irradiated chondrocytes with LIPUS, thereby regulating the expression of primary cilia, activating TRPV4 mechanosensitive channels, inducing an inward flow of calcium, and facilitating the entry of NF-κB into the nucleus [44]. Jian et al. and Zhou et al. have reported that applying LIPUS inhibits the YAP/caspase-3 axis, a critical apoptotic signaling pathway, and consequently suppresses apoptosis [71, 72].
The anti-inflammatory effects of LIPUS arise from its activation of various immune signaling pathways through mechanical effects. LIPUS directly inhibits the inflammatory activation of the MAPK, NF-κB, and PI3K/AKT pathways, while enhancing the anti-inflammatory effects of other therapies, such as exosomes [73]. Sang et al. have reported that LIPUS inhibits MAPK-mediated inflammatory responses by activating the α-signaling pathway, thus significantly suppressing the expression of inflammatory factors such as IL-6, IL-8, and tumor necrosis factor (TNF)/FAK [74]. LIPUS significantly inhibits lipopolysaccharide-induced increases in NF-κB receptor activator and C-X-C motif chemokine ligands in mouse osteoblast cell lines, and therefore might attenuate inflammation by inhibiting the NF-κB pathway [75]. The multifunctional PI3K/Akt/mTOR signaling pathway promotes cell migration and proliferation, and inhibits inflammation and apoptosis. LIPUS can reprogram the fate of immune cells. Its ability to modulate macrophage polarization is particularly noteworthy: in an acute kidney injury model, LIPUS inhibits RIPK3/MLKL-dependent necrosis and ultimately promotes macrophage polarization toward the M2 phenotype by inhibiting NLRP3 inflammasomes [76]. In terms of T-cell subpopulation homeostasis, LIPUS activates the Mst1-TAZ axis; alters the interaction between the transcription factor FOXP3 and RORγt; and regulates the differentiation of regulatory T cells (Tregs) and helper T cells (Th17) [77]. In addition, transcutaneous electrical stimulation has been found to mitigate radiation-induced ovarian failure by inhibiting primordial follicle loss, increasing serum AMH secretion, and inducing antioxidant and anti-apoptotic systems [29].
Regulation of angiogenesis and antifibrosis
In the pathological process of POI, impaired angiogenesis and interstitial fibrosis are critical mechanisms that exacerbate the decline in ovarian function [78]. Decreased ovarian neovascularization results in insufficient blood supply to the perifollicular microcirculation, thus leading to accelerated follicular atresia due to hypoxia and nutrient deprivation. In addition, interstitial fibrosis gradually stiffens the ovarian tissue. This stiffening directly compresses the primordial follicles and restricts their developmental space, and additionally hinders signaling interactions between the follicles and the surrounding supportive cells, thereby inhibiting estrogen synthesis [79]. Decreased angiogenesis and fibrosis are interconnected in a vicious cycle: the ischemic microenvironment promotes fibrotic deposition, thereby exacerbating vascular torsion and occlusion, and ultimately leading to depletion of the ovarian reserve and endocrine failure. Interventions targeting these two critical components offer new strategies for improving the ovarian microenvironment in cases of POI.
LIPUS contributes to the regulation of fibrosis and angiogenesis, through its mechanical effects. Endothelial cells are sensitive to ultrasound-induced mechanical forces, and LIPUS has been found to alter endothelial cell morphology, proliferative ability, gene expression, and protein secretion [80]. The microacoustic flow and acoustic radiation forces generated by LIPUS can influence vesicle structures within endothelial cells, by mechanically converting sensors such as integrins and vesiculin-1 into intracellular biochemical signals. This conversion in turn upregulates the expression of related growth factors and promotes angiogenesis [81–83].
LIPUS generates sound pressure waves that penetrate living cells and induce biochemical events at the cellular level [16]. This process increases protein synthesis, promotes mast cell secretion, and alters the migratory function of fibroblasts [84]. LIPUS directly targets the phosphorylation process in the TGF-β1/Smad pathway, a central axis in fibrosis progression, and inhibits the differentiation of fibroblasts into myofibroblasts [85]. Concurrently, it suppresses profibrotic signaling pathways such as the Wnt/β-catenin and MAPK pathways, thereby decreasing excessive collagen secretion [86]. At the extracellular matrix level, LIPUS facilitates the structural remodeling of fibrotic tissue by increasing the enzymatic activity of MMPs while downregulating the expression of tissue inhibitor of metalloproteinase. These actions together enhance the degradation of pathological collagens [87].
Emerging preclinical evidence supports the concept that LIPUS has multitargeted tissue repair capabilities in various pathological scenarios and highlights its substantial translational potential for addressing POI. Mechanistically, LIPUS exerts its therapeutic effects by orchestrating antifibrotic, anti-inflammatory, and regeneration-promoting pathways (Table 2). These mechanisms are anticipated to reverse the hallmarks of POI pathogenesis, which are characterized by follicular atresia and ovarian stromal fibrosis. Collectively, these findings provide a compelling theoretical foundation for the use of LIPUS as a noninvasive strategy to combat ovarian dysfunction associated with POI.
Table 2 Potential Mechanisms and Corresponding Ultrasonic Power for POI Treatment with LIPUS
| Mechanism Category | Intensity (mW/cm2) | Target of Regulation | Signaling Pathway | Reference |
|---|---|---|---|---|
| Cell proliferation | 100 | Satellite cells | Activates the PGC-1α/AMPK/GLUT4 pathway | [61] |
| 30 | Chondrocytes | Activates the integrin receptor/PI3K/Akt pathway | [60] | |
| 20 | Schwann cells | Activates NRG1/ErbB signaling | [62] | |
| 30 | Skin fibroblasts | Activates integrin receptor and Rho/ROCK/ERK signaling pathways | [63] | |
| Inhibition of apoptosis | 30 | Articular chondrocytes | Modulates TRPV4 mechanosensitive channels, thereby activating NF-κB nuclear entry | [44] |
| 90 | Periodontal ligament cells | Suppresses the YAP/caspase-3 axis | [71] | |
| Anti-inflammatory | 30 | Osteoblasts | Inhibits MAPK, NF-κB, and PI3K/Akt-mediated inflammation | [75] |
| 40 | Chondrocytes | Decreases IL-6, IL-8, and TNF-α levels | ||
| 3×103 | M1 macrophages | Suppresses NLRP3 inflammasomes | [8] | |
| 250 | CD4+ T-cells | Activates the Mst1-TAZ axis | [77] | |
| Angiogenesis | 30±5 | Human umbilical vein endothelial cells | Activates the YAP/TAZ pathway | [82] |
| 300 | Follicles | Improves vascularization in transplanted ovarian tissue | [90] | |
| Antifibrosis | 60 | Kidney tissue | Inhibits the TGF-β/Smad pathway | [85] |
| 30 | Fibroblast-like synoviocytes | Modulates the Wnt/β-catenin and PI3K/AKT pathways | [87] |
Progress in LIPUS applications in POI
By harnessing the mechanobiological effects of acoustic cavitation and microstreaming, LIPUS has demonstrated postinjury regenerative capabilities across multiple tissue types and provided a robust theoretical foundation for POI intervention. Current preclinical evidence has confirmed the efficacy of LIPUS in three therapeutic areas: enhancement of ovarian reserve function, suppression of GC apoptosis, and immunomodulation through inflammatory pathway regulation mediated by tissue repair mechanisms. Furthermore, strategic integration of LIPUS with adjuvant therapies that exploit its bioeffects on cellular microenvironments enables a multitarget therapeutic approach to address the multifactorial pathophysiology of POI. This section systematically evaluates both the standalone therapeutic potential of LIPUS and its synergistic role within multimodal POI treatment frameworks.
Applications of LIPUS in POI therapy
Ultrasound has been studied in medicine for decades, and preclinical research has focused primarily on the effects of high-intensity focused ultrasound in tumor treatment. The past decade has seen a notable increase in preclinical studies investigating the use of LIPUS across various systems, particularly in the context of gynecological disorders [17]. Initially, research focused on how LIPUS might treat osteoporosis associated with POI, whereas less attention was paid to the changes in ovarian tissue cells influenced by LIPUS. POI models in preclinical studies are often established by surgical removal of the ovaries [88]. As research has progressed, scientists have begun to investigate the effects of LIPUS on the ovaries. As early as 1978, ultrasound at 3 MHz and 2 W/cm2 was demonstrated to influence uterine activity and promote endometrial metamorphosis; however, this finding was not further investigated [89].
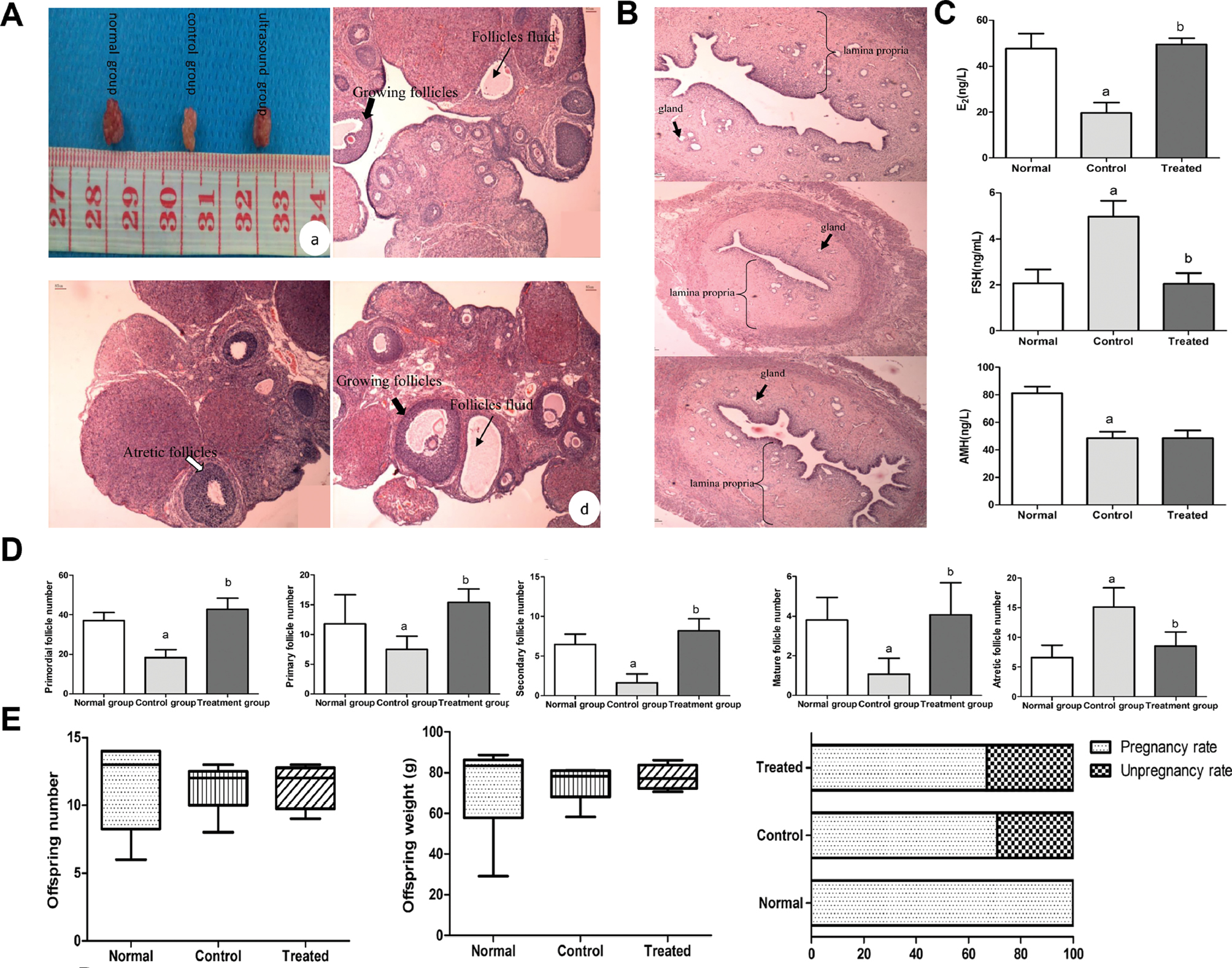
In 2014, Abtahi et al. developed an ovarian transplantation model by implanting ovarian tissue into the backs of mice. They discovered that LIPUS irradiation at 3 MHz, 0.3 W/cm2, and a 20% duty cycle enhanced folliculogenesis and angiogenesis in the transplanted ovaries [90]. In 2017, Tang et al. [91] established a model of POI using cyclophosphamide (CTX) and subjected it to 30 minutes of ultrasound irradiation at 0.9 MHz, 0.8 W/cm2, and a 20% duty cycle. LIPUS irradiation was associated with increase follicular proliferative activity in POI ovaries, thus resulting in an increase in follicle numbers at all stages except for atretic follicles and AMH levels. Specifically, the levels of E2 and FSH were restored to levels comparable to those in the control group [91]. In 2021, the same research group found that LIPUS treatment led to an amelioration of ovarian function in a rat model of CTX-induced POI [15]. The clearest effect was observed in the restoration of estrous cycles: 100% of treated rats (22/22) resumed normal cyclicity, whereas persistent disorder was observed in all untreated controls. This clinical improvement was underpinned by considerable histological recovery: LIPUS treatment resulted in a marked increase in the population of healthy follicles across all developmental stages, comprising primordial (from 18 ± 4 to 43 ± 5), primary (from 7 ± 2 to 15 ± 2), secondary (from 2 ± 1 to 8 ± 1), and mature follicles (from 1 ± 1 to 4 ± 2), while concurrently decreasing the number of atretic follicles (from 15 ± 3 to 9 ± 2). Concomitantly, the aberrant serum hormone profile was partially corrected, E2 levels rebounded from 20 ± 4 ng/L to 50 ± 3 ng/L, and elevated FSH levels normalized from 5 ± 1 ng/mL to 2 ± 1 ng/mL. However, LIPUS did not improve the serum level of AMH, which remained low (49 ± 5 ng/L vs. control 49 ± 5 ng/L). Crucially, these improvements did not translate into enhanced fertility, as evidenced by an absence of significant differences in pregnancy rates (66.7% vs. 71.4%), litter sizes (median 12 vs. 12), and pup weight (median 77 g vs. 78 g) with respect to those in untreated POI controls (Figure 4).
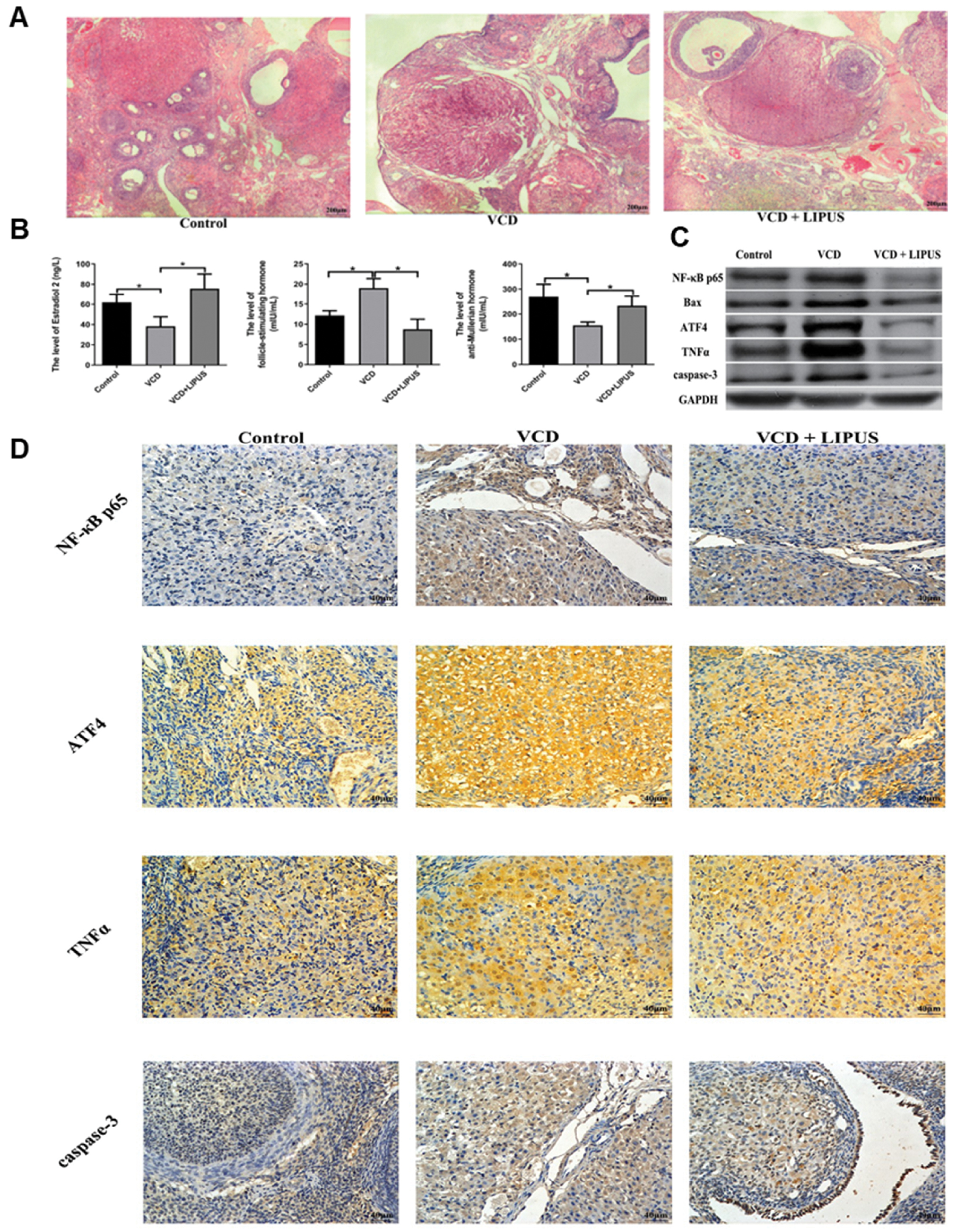
Figure 4 A. H&E staining of the ovaries and (a) rough observation. (b) The cortex of the normal group was usually normal. (c) The volume of the ovaries in the control group was atrophied, and the number of follicles was decreased. (d) In the ultrasound group, the number of follicles at different growth stages was higher, and the amount of follicular fluid was greater. B. Histological analysis of the uterus. C. Changes in follicle numbers among the three groups. D. Changes in levels of serum hormones among the three groups. E. Fertility outcomes among the three groups. Figure 4A–E was reproduced from Ref. [15] with permission from Elsevier. Copyright 2021, Reference: Tang et al., 2021 [15].
LIPUS can treat POI by inhibiting apoptosis and inflammation. In 2021, Xu et al. irradiated mice with 4-vinyl cyclohexene dioxide (VCD)-induced premature ovarian failure (POF) via LIPUS at a frequency of 0.3 MHz, an intensity of 200 mW/cm2, and a duty cycle of 20%. LIPUS irradiation increased the ovarian reserve ability in mice and decreased the apoptosis of GCs by decreasing the expression of BAX and increasing the expression of Bcl-2 [92]. After LIPUS treatment, the expression of biomarkers associated with inflammation and apoptosis, including NF-κB p65, TNF-α, Bax, ATF4, and caspase-3, significantly decreased in ovarian tissues, thereby leading to observed decreases in inflammation and apoptosis. Furthermore, as inflammation and apoptosis decreased, the ovarian microenvironment was restored, as evidenced by increased numbers of follicles, elevated levels of E2 and AMH, and decreased FSH levels (Figure 5). These findings suggest that LIPUS effectively mitigates ovarian damage [9].
Figure 5 A. Ovarian tissue sections of the rats in each group (hematoxylin and eosin staining, original magnification: 40×). B. Expression levels of inflammatory cytokines (E2, FSH, and AMH) in the ovaries of each group, detected via enzyme-linked immunosorbent assay. C. Expression levels of NF-κB p65, Bax, ATF4, TNFα, and caspase-3 in the ovaries of each group, analyzed via western blotting. D. Expression levels of NF-κB p65, ATF4, TNFα, and caspase-3, detected via immunohistochemistry (400×). Figure 5A–D was reproduced from Ref. [92] with permission from Oxford University Press. Copyright 2021, Reference: Xu et al., 2021 [92].
LIPUS mitigates fibrosis through pathways such as the TGF-β/Smad, Wnt/β-catenin, and PI3K/AKT pathways [87, 93, 94]. Zhou et al. have used cisplatin as a chemotherapeutic agent to establish a mouse model of premature ovarian failure and investigated the protective effects of LIPUS on ovarian function. The experimental results indicated that the ovarian volume in mice significantly decreased after cisplatin injection, and the number of follicles additionally decreased. In contrast, the LIPUS-treated group exhibited a marked decrease in follicular and GC apoptosis. Further studies demonstrated that LIPUS alleviated ovarian fibrosis by decreasing the deposition of the fibrosis markers α-SMA and collagen and downregulating the TGF-β1/Smad signaling pathway [95]. In summary, LIPUS effectively decreases chemotherapy-induced ovarian damage, protects ovarian function, and offers a novel approach for treating ovarian fibrosis. Deng et al. [96] have systematically investigated the long-term protective effects and molecular mechanisms of LIPUS in VCD-induced POI models. In vivo experiments using optimized LIPUS parameters (200 mW/cm2 intensity, 0.36 MHz frequency, 1 kHz pulse repetition frequency, and 20% duty cycle) with daily 20-minute exposure over 15 consecutive days demonstrated significant histological restoration of ovarian architecture and reactivation of folliculogenesis in POI rats, accompanied by normalized sex hormone secretion, restored estrous cyclicity, and improved fertility outcomes. Mechanistic analyses revealed that LIPUS conferred cytoprotection through modulation of the Daxx/ASK1/JNK signaling axis, thus effectively preserving mitochondrial membrane integrity while inhibiting apoptosis via downregulation of proapoptotic effectors (Bax, cytochrome c, and cleaved caspase-3) concomitantly with Bcl-2 upregulation. Furthermore, LIPUS alleviated menopausal symptoms associated with ovarian hypofunction, including depressive-like behaviors and motor dysfunction, through improvements in behavioral parameters such as locomotor distance, central zone retention time, and grip strength [96].
The divergent outcomes of AMH restoration between studies, such as the lack of improvement observed by Tang et al. [15] versus the significant increase observed by Qin et al. [9], can be rationalized by considering key experimental variables. First, the POI model pathophysiology differed: the CTX model used by Tang et al. [15] causes severe, widespread ovarian damage, which might overwhelm the ability to restore small, AMH-secreting follicles. In contrast, the VCD model used by Qin et al. [9] induces a more selective, apoptotic follicle loss, a process that might be more readily mitigated by LIPUS. Second, the LIPUS parameters are critical: the higher intensity and frequency used by Tang et al. [15] (0.8 W/cm2 and 0.9 MHz) might have elicited different bioeffects from the lower intensity and frequency used by Qin et al. [9] (0.03 W/cm2 and 0.25 MHz), and the latter might potentially be more favorable for preserving the primordial follicle pool. Therefore, these findings do not contradict but complement each other, by suggesting that LIPUS efficacy is context dependent. Low-intensity regimens might be optimal for protecting the ovarian reserve and AMH in degenerative models, whereas restoring function after severe injury might require alternative parameter sets or combinatorial approaches.
Although limited studies have investigated the direct application of LIPUS for treating POI, existing research indicates that LIPUS can effectively treat POI by promoting GC proliferation, enhancing angiogenesis, inhibiting apoptosis, and decreasing inflammatory responses and fibrosis.
LIPUS combined with other modalities to treat POI
LIPUS shares convergent therapeutic pathways with other modalities in POI management and achieves coordinated biological effects through analogous mechanotransduction principles. In emerging research paradigms, investigators are actively pursuing the strategic integration of LIPUS with adjuvant therapies. This approach leverages their complementary bioeffects to enable multitarget synergistic interventions aimed at improving clinical outcomes.
LIPUS affects the treatment of POI with MSCs
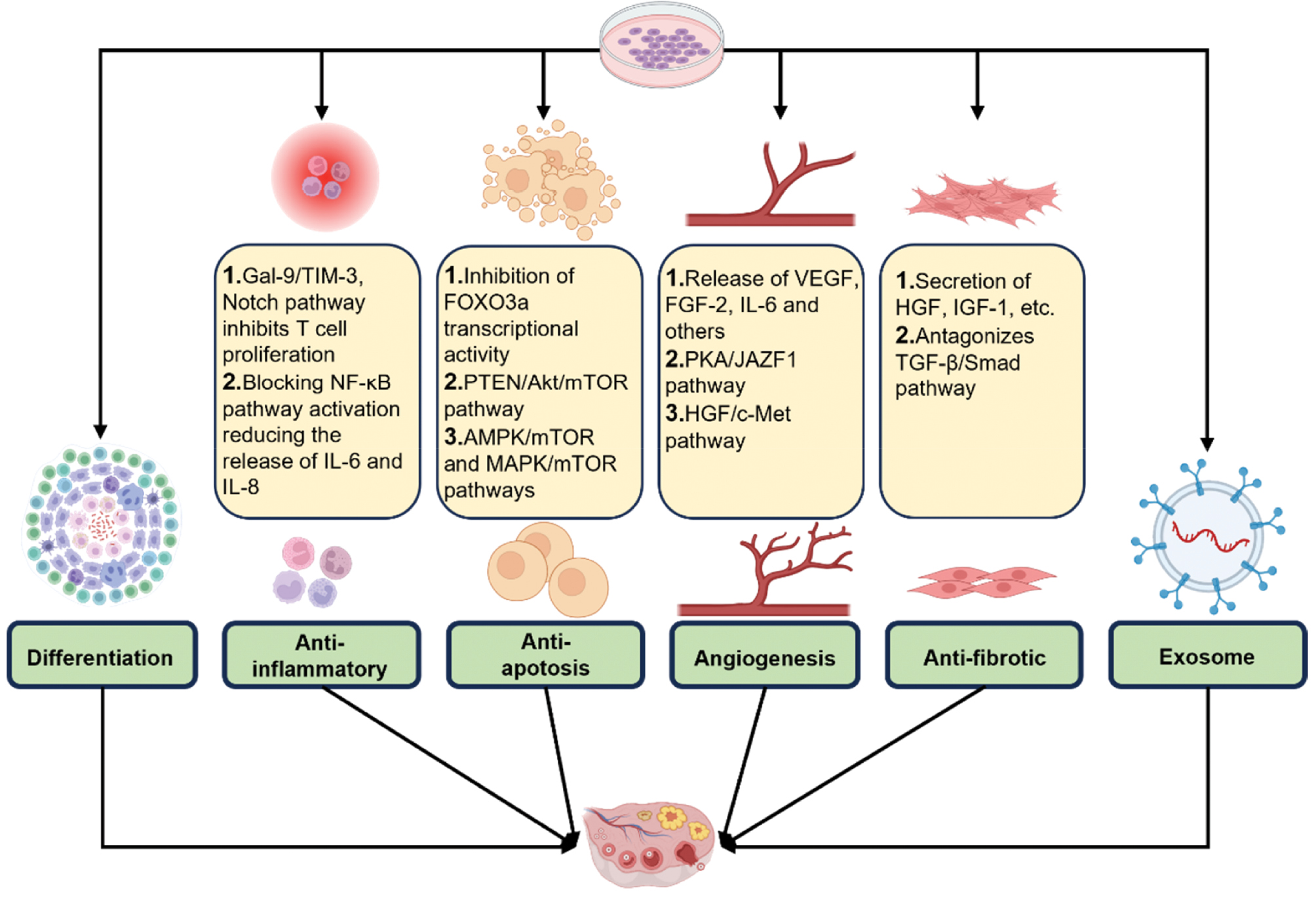
Stem cells differentiate in response to their environment and various signals, and activate endogenous repair mechanisms in the host through the secretion of growth factors and cytokines [97]. These cells have the potential to restore the ovarian reserve, improve hormone levels, and increase fertility through direct differentiation to replenish ovarian cells, as well as through antifibrotic, anti-inflammatory, antiapoptotic, and immunomodulatory mechanisms that promote angiogenesis (Figure 6) [98]. Currently, MSCs are the most advanced stem cell therapy for POI.
Figure 6 Mechanisms of stem cell therapy in POI: differentiation, anti-inflammatory differentiation, anti-inflammatory, antiapoptotic, antifibrotic, proangiogenic, and exosome release. Source: Created by the authors via Biorender.com.
MSC mechanisms for POI treatment
Similarly to the mechanism of LIPUS in treating POI, MSCs address POI primarily by modulating immune responses, inhibiting apoptosis, and promoting angiogenesis (Figure 3). Bone marrow mesenchymal stem cells (BMMSCs) ameliorate POI through multiple mechanisms [99]. In POI mouse models, BMMSCs contribute to arterial formation by producing and secreting VEGF, FGF-2, and IL-6 [100]. Additionally, BMMSCs upregulate Bcl-2, downregulate Bax expression, and exert antiapoptotic effects [100, 101]. Furthermore, BMMSCs play an immunomodulatory role by altering the immune phenotype of T cells and macrophages and consequently suppressing immune functions [102–104]. Exosomes secreted by BMMSCs have been demonstrated to decrease apoptosis in GCs by delivering microRNA-144-5p, which in turn stimulates the PTEN pathway [99, 102]. Additionally, microRNA-644-5p promotes angiogenesis through the AKT/mTOR signaling pathway [105].
Adipose-derived mesenchymal stem cells modulate dendritic cell activity via the Notch signaling pathway and diminish NF-κB activation through various mechanisms, including the Gal-9/TIM-3 pathway, thereby inhibiting T cells [106–108].
LIPUS enhances the effectiveness of MSCs in treating POI
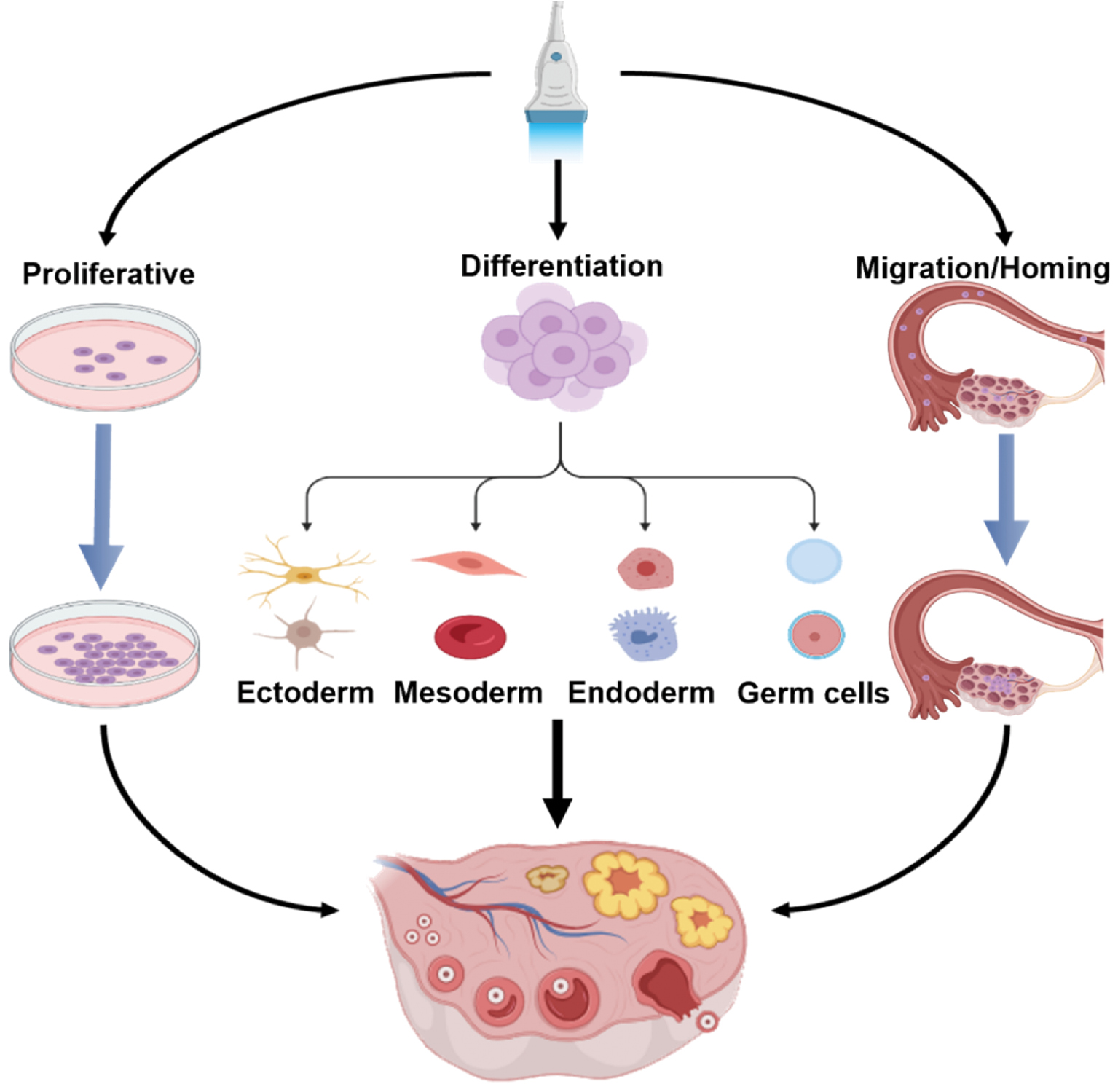
Although stem cell therapy is a promising approach for treating POI, its effectiveness is currently limited by insufficient transplanted MSCs within the affected tissue [109]. Specific LIPUS parameters have been shown to significantly increase the proliferation, differentiation, and homing ability of MSCs (Figure 7). Various LIPUS parameters have differing effects on stem cells (Table 3). Xie et al. have used LIPUS to stimulate human BMMSCs and have reported that LIPUS at intensities of 50 or 60 mW/cm2 increases PI3K/AKT pathway phosphorylation and significantly upregulates the expression of cyclin D1, thereby promoting BMMSC proliferation [110]. Kusuyama et al. have reported that LIPUS at 30 mW/cm2 inhibits adipogenic differentiation by downregulating the expression of PPAR-γ2 and FABP4, while simultaneously enhancing mineralization and the expression of osteogenic transcription factors such as Runx2 and OCN in BMMSCs [111]. Wei et al. have demonstrated that LIPUS at 30 mW/cm2 enhances the chemotactic migration of BMMSCs toward fracture sites in both in vitro and in vivo models [112].
Figure 7 LIPUS promotes stem cell proliferation, differentiation, and migration/homing. Source: Created by the authors via Biorender.com.
Table 3 Effects of LIPUS with Various Parameters on the Indicated Stem Cells
| Effect | Target of Regulation | Intensity (mW/cm2) | Mode of Action | References |
|---|---|---|---|---|
| Cell proliferation | BMMSCs | 50/60 | Activates the PI3K/Akt signaling pathway | [135] |
| ADSCs | 70 | Upregulates cell cycle proteins and promotes adipogenesis | [136] | |
| PDLSCs | 250–750 | Stimulates differentiation via PPAR-γ and adiponectin upregulation | [137] | |
| DPSCs | 750 | Modulates p38/MAPK phosphorylation | [138] | |
| AD-MSCs | 30 | Activates ERK1/2 or JNK signaling pathways | [139] | |
| UCMSCs | 25–35 | Promotes ERK1/2 phosphorylation via piezo ion channel activation | [140] | |
| Differentiation | BMMSCs (osteogenesis) |
100 | Activates ERK1/2 and PI3K/Akt pathways; upregulates cyclins (D1, E1, A2, and B1) | [141] |
| BMMSCs (osteogenesis) |
20–50 | Irradiation time affects cell viability but has no effect on proliferation | [111] | |
| BMMSCs (chondrogenesis) |
100 | Upregulates osteogenic markers (Runx2, ALP, and OCN) | [142, 143] | |
| BMMSCs (cytoskeletal) |
30 | Inhibits adipogenic genes (PPAR-γ and FABP4) and osteogenic differentiation via the ROCK-MEK-ERK pathway | [144] | |
| BMMSCs (osteointegration) |
45 | Upregulates COL2, SOX9, and aggrecan | [145] | |
| BMMSCs (osteointegration) |
30 | Suppresses COL1 via the integrin-mTOR-autophagy axis | [146] | |
| ADSCs (osteogenesis) |
30/100 | Induces YAP nuclear translocation via cytoskeletal mechanotransduction | [147, 148] | |
| PDLSCs | – | Enhances osteogenic differentiation via Runx2, OCN, and BSP upregulation | [149] | |
| UCMSCs | 30 | Promotes adipogenic differentiation via PPAR-γ and adiponectin upregulation | [150] | |
| ADSCs (angiogenesis) | 15 | Synergizes with chondrogenic medium in upregulating COL2 and GAG | [151] | |
| GMSCs (neural) |
30 | Enhances CD31 and VE-cadherin expression in cocultured endothelial cells | [152] | |
| BMMSCs (hepatic) |
500–1500 | Upregulates neural markers (neurofilament and vimentin); downregulates nucleostemin | [153] | |
| hUC-MSCs | 30–250 | Activates the Wnt/β-catenin pathway, thereby upregulating AFP, ALB, and CK18 under HGF induction | [150] | |
| Migration | BMMSCs | 30–50 | Inhibits TNF signaling, thus promoting chondrogenic differentiation | [154] |
| PDLSCs | 90 | Activates SDF-1/CXCR4 signaling; modulates FAK-ERK1/2 and autophagy pathways | [155] | |
| AD-MSCs | 30 | Promotes SDF-1/CXCR4 upregulation via TWIST1-mediated signaling | [156] |
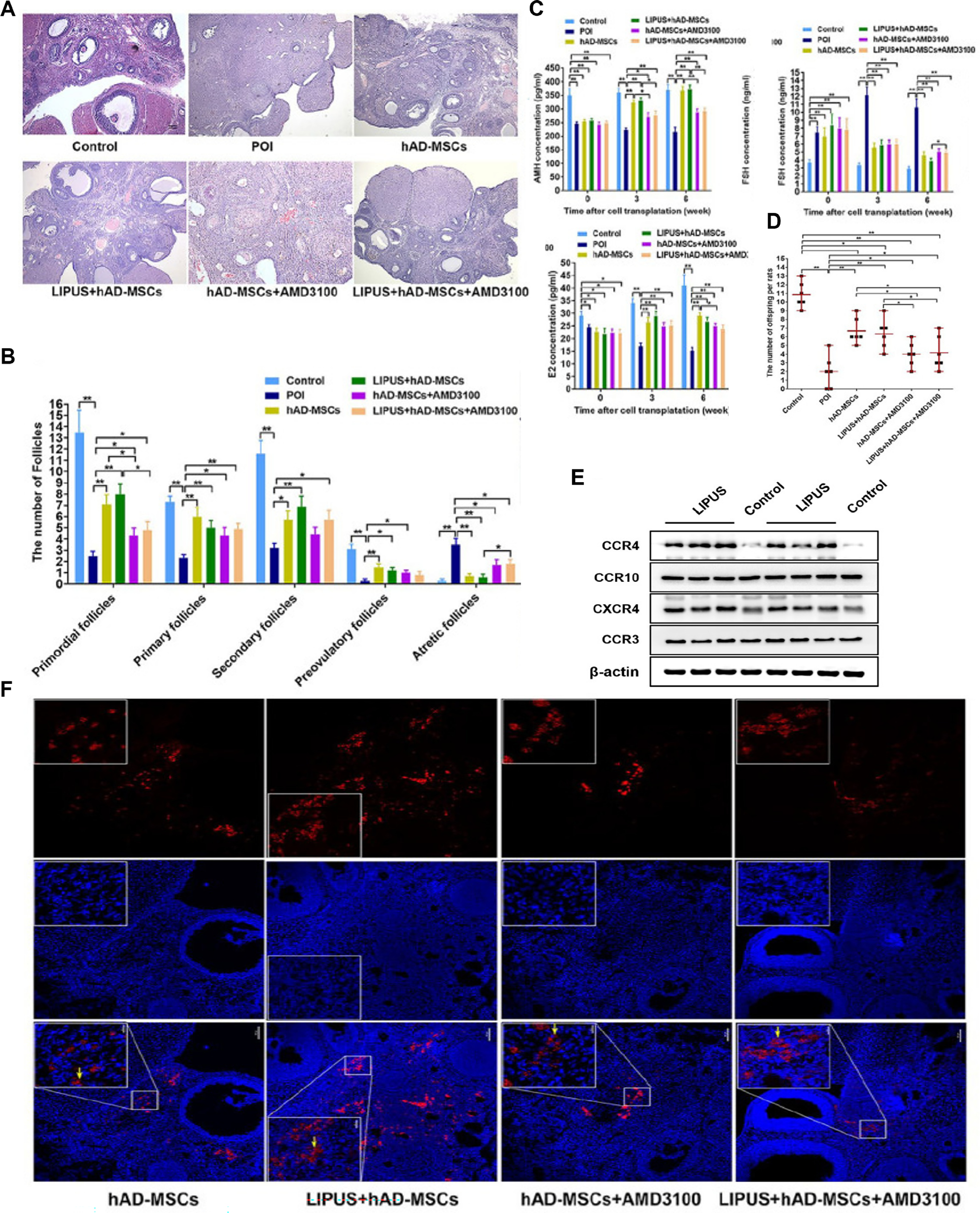
LIPUS enhances the proliferation, differentiation, and homing of stem cells. Therefore, combining LIPUS and stem cell therapy is anticipated to yield improved treatment outcomes for POI. Ling et al. [113] have used CTX to establish a rat model of POI. Human amniotic membrane mesenchymal stem cells (hAD-MSCs) were extracted from human amniotic membranes and transplanted into the POI model. The rat model was subsequently subjected to LIPUS irradiation at a frequency of 0.25 MHz, with a 20% duty cycle and an intensity of 30 mW/cm2 for 30 minutes. LIPUS pretreatment significantly improved the biological properties of hAD-MSCs. CCK-8 and EdU assays confirmed that LIPUS treatment markedly increased cell viability and proliferation ability (P < 0.05) without affecting apoptosis (P > 0.05). LIPUS treatment significantly enhanced the expression of various chemokine receptors in hAD-MSCs, particularly the mRNA and protein levels of CXCR4 (P < 0.01). Concurrently, the SDF-1 concentrations in the ovarian microenvironment in POI rats were significantly greater than those in the control group (P < 0.01), thus establishing a robust chemotactic gradient. In vitro experiments demonstrated that LIPUS pretreatment significantly enhanced SDF-1-induced migration of hAD-MSCs, as evidenced by Transwell and scratch assays, which revealed increased migrating cell counts and cell coverage ratios (P < 0.01). The marked inhibition of these responses by the CXCR4-specific antagonist AMD3100 confirmed the critical role of the SDF-1/CXCR4 axis. In vivo experiments revealed that, although the number of homing cells in the LIPUS-pretreated group (27.67 ± 23.78 cells/field) exceeded that in the conventional hAD-MSC group (24.67 ± 21.54 cells/field), the difference was not statistically significant (P > 0.05). However, AMD3100 treatment significantly decreased the numbers of homing cells in both groups (P < 0.05). These findings further validated the importance of CXCR4 in the homing process. In terms of therapeutic efficacy, hAD-MSC transplantation significantly improved ovarian function in POI rats, as manifested by a significant increase in primordial and growing follicles (P < 0.05), significantly elevated serum AMH and E2 levels (P < 0.05), a significant decrease in FSH levels (P < 0.05), a significant decrease in the proportion of abnormal estrous cycles, and markedly improved fertility (P < 0.05) (Figure 8) [113]. However, the study was limited to observations in animal models and involved few stem cell protocols. Future research is necessary to explore the effects of LIPUS on other types of stem cells, to validate the long-term safety of this approach in primate models, and to advance the clinical application of this strategy.
Figure 8 A. Histological changes in ovaries, analyzed with hematoxylin and eosin staining in the control, POI, hAD-MSCs, LIPUS + hAD-MSCs, hAD-MSCs + AMD3100, and LIPUS + hAD-MSCs + AMD3100 groups. B. Numbers of follicles at different stages, counted and compared at 6 weeks after cell transplantation in the six groups. C. Serum levels of AMH, FSH, and E2, tested at 0, 3, and 6 weeks after hAD-MSC transplantation in the six groups. D. Number of offspring per rat in each group. E. Effects of LIPUS on the protein expression levels of CCR3, CCR4, CCR10, and CXCR4 in hAD-MSCs, detected via western blotting analysis. F. Effects of LIPUS on hAD-MSC homing to chemotherapy-induced POI ovaries in vivo. Figure 8A–F was reproduced from Ref. [113] with permission from Sage Journals. Copyright 2022, Reference: Ling et al., 2022 [113].
LIPUS affects the treatment of POI with TCM
TCM has a long history of treating POI by enhancing ovarian reserve function and alleviating symptoms associated with low estrogen levels, through the holistic regulatory effects of multiple components and targets [114].
Treatment of POI with TCM
Studies have found that most prescriptions are based on the theory of “tonifying the kidney and activating the blood” and “regulating the liver and benefiting the kidney”, which improve granulocyte function and inhibit oxidative stress and fibrosis through multi-targeted regulation of the ovarian signaling pathway. Zuogui Wan, Yulinzhu, and Guiluoshi Anzang decoctions have been found to promote GC proliferation via the PI3K/AKT/mTOR pathway [115–117]. The Bushen Huoxue recipe and Yijing Hugui decoction also mitigate oxidative stress by regulating the Nrf2/HO-1 pathway [118, 119]. With respect to oxidative stress inhibition, the Huyang Yangkun formula regulates ovarian GC apoptosis through the FTO/m6A-P53 pathway [120]. Furthermore, Semen Cuscutae has been found to inhibit oxidative stress by activating the Keap1-Nrf2/HO-1 pathway and to modulate GC apoptosis by influencing the PI3K/AKT pathway [120].
LIPUS facilitates POI treatment with herbal medicine
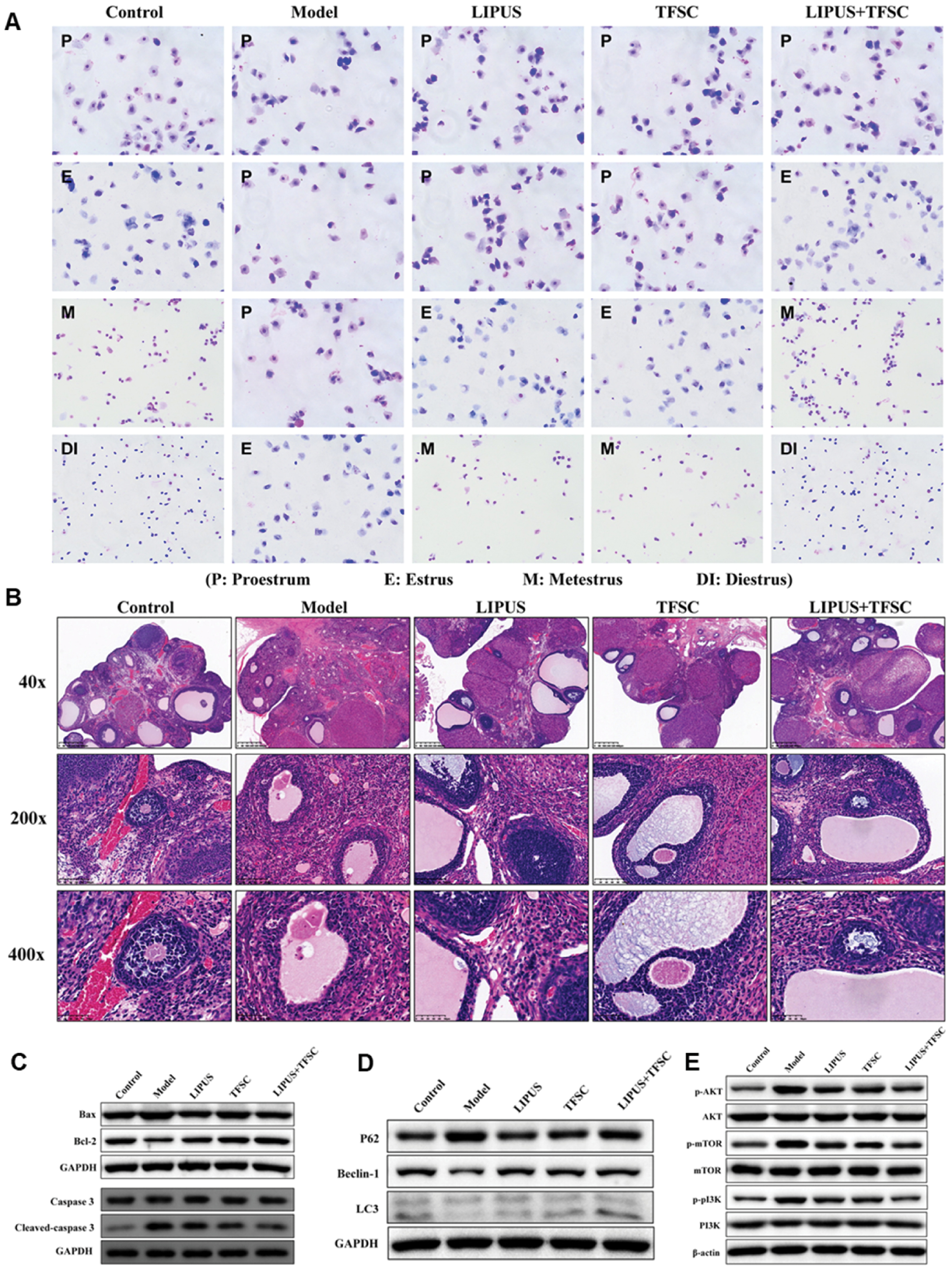
Traditional methods of enhancing drug action through ultrasound depend primarily on the temporal and spatial controllability of ultrasound to improve drug delivery efficiency. Zhou et al. [121] have used LIPUS with total flavonoids from Semen Cuscutae (TFSCs) to coregulate ovarian function in rats with POI. The combined application of LIPUS and total flavonoids from TFSCs demonstrated significant therapeutic efficacy in alleviating POF in both rat and cellular models. In vivo, POF model rats presented a disrupted estrous cycle, together with diminished body weight, ovarian indices, and uterine indices, all of which were markedly ameliorated by the combination treatment. Serum hormone analysis via ELISA revealed that the POF-induced decreases in AMH, E2, and progesterone levels, alongside the increase in FSH and LH, were effectively reversed by LIPUS and TFSC co-treatment, and achieved effects surpassing those of either treatment alone.
Histopathological examination revealed that the POF model led to ovarian atrophy, a significant decrease in primordial and primary follicles, and an increase in atretic follicles. These morphological disruptions were considerably ameliorated after the combined intervention. Furthermore, staining for senescence-associated β-galactosidase (SA-β-Gal) and the DNA damage marker γH2AX indicated a substantial increase in cellular senescence in POF ovaries, which was potently suppressed by the combination therapy.
At the molecular level, POF is characterized by inhibited autophagy and accelerated apoptosis. Quantitative PCR and western blot analyses of ovarian tissues and GCs indicated diminished expression of autophagy-associated genes and proteins (ATG5, Beclin-1, and the LC3-II/LC3-I ratio) and elevated levels of the autophagy substrate p62 in the model group. Concurrently, apoptosis was enhanced, as evidenced by elevated protein levels of Bax and cleaved caspase-3 and a decreased Bcl-2/Bax ratio. Treatment with LIPUS plus TFSC synergistically normalized these alterations, by promoting autophagic flux and suppressing apoptotic signaling. Transmission electron microscopy visually confirmed an increase in the number of autophagic vesicles in GCs, whereas flow cytometry quantitatively demonstrated a significant decrease in the apoptosis rate after combination treatment. This restoration of cellular function is consistent with the established principle that a cell’s physiological state, including its response to therapy, is directly reflected by its mechanical properties [122].
Mechanistically, these protective effects are mediated through the PI3K/AKT/mTOR pathway. The increased phosphorylation levels of PI3K, AKT, and mTOR observed in the POF group indicated pathway activation. The combined intervention significantly inhibited this activation, thereby relieving suppression of autophagy. In conclusion, the data strongly indicated that the co-administration of LIPUS and TFSC synergistically mitigates POF primarily by enhancing autophagy and inhibiting apoptosis in ovarian GCs via modulation of the PI3K/AKT/mTOR signaling pathway [121]. Although current investigations of LIPUS-TCM combination therapy for POI are limited, preliminary evidence suggests that their mechanistic complementarity might yield therapeutic synergy (Figure 9).
Figure 9 A. LIPUS combined with TFSC restores the estrus cycle in POI rats. B. LIPUS and TFSC inhibit ovarian tissue structural disorders in POI rats. C. Western blotting detection of protein levels of p62, Beclin 1, and LC3 II/I. D. Western blotting detection of protein levels of Bcl-2 and Bax in ovarian GCs. E. Protein expression levels of p-PI3K/PI3K, p-AKT/AKT, and p-mTOR/mTOR in ovarian GCs, detected via western blotting. Figure 9A–E was reproduced from Ref. [121] with permission from Taylor & Francis. Copyright 2023, Reference: Zhou et al., 2023 [121].
Sensitizing effects of LIPUS on other treatment modalities
In the therapeutic domain of POI, LIPUS is a noninvasive physical modality that establishes complementary therapeutic synergies through multiple mechanisms, including enhanced ovarian perfusion, GC functional restoration, and inflammatory cascade suppression. Beyond its synergy with conventional stem cell therapies and TCM, LIPUS has multidimensional cooperative potential with other treatment paradigms. Specifically, LIPUS intervention achieves estrogen level recovery and potentially decreases HRT dosage dependency in POI management, thus mitigating associated adverse effects [31]. Concurrently, its anti-inflammatory properties might potentiate the therapeutic efficacy of immunosuppressive regimens [32]. Furthermore, LIPUS-induced membrane permeability enhancement creates a biophysical adjuvant effect with potential to optimize gene-editing precision in POI-targeted genetic interventions [33].
LIPUS has demonstrated substantial potential as a standalone therapeutic intervention for POI, by promoting tissue repair and restoring ovarian function. When LIPUS is combined with adjunctive therapies such as stem cell transplantation or TCM, its efficacy can be further enhanced through synergistic effects. This integrated approach builds on LIPUS and enhances the effectiveness of adjuvant therapies. Current evidence indicates that LIPUS exerts powerful therapeutic effects independently and enhances the effectiveness of multimodal treatment strategies for POI (Table 4).
Table 4 LIPUS and LIPUS Combination Therapy in POI
| Treatment | Ultrasound Parameters | Mode of Action | Mechanisms | References | |||
|---|---|---|---|---|---|---|---|
| Frequency (MHz) | Duty cycle (%) | Intensity (W/cm2) | Time (min) | ||||
| LIPUS | 3 | – | 2 | – | Regulates smooth muscle activity | – | [89] |
| 3 | 20 | 0.3 | 20 | Promotes folliculogenesis and angiogenesis | Increases vascular growth factor expression | [90] | |
| 0.9 | 20 | 0.8 | 30 | Increases follicle number and restores hormone levels | – | [91] | |
| 0.3 | 20 | 0.2 | 20 | Increases ovarian reserve ability and decreases GC apoptosis | Decreases BAX expression and increases Bcl-2 expression | [92] | |
| 0.25 | 20 | 0.03 | 20 | Inhibits inflammatory response and apoptosis | Decreases expression of NF-κB p65, TNFα, Bax, ATF4, and caspase-3 | [9] | |
| 0.5 | 20 | 0.03 | 20 | Inhibits fibrosis | Downregulates TGF-β1/Smad | [95] | |
| LIPUS combined with MSCs | 0.25 | 20 | 0.03 | 30 | Inhibits inflammatory response and apoptosis | Increases secretion of growth factors | [156] |
| LIPUS combined with TCM | 30 | 20 | 0.25 | 30 | Inhibits GC apoptosis | Downregulates PI3K/AKT/mTOR | [121] |
Analysis of LIPUS parameter heterogeneity and rationale for selection
We observed significant heterogeneity in the LIPUS parameters used across different POI studies, such as frequency, intensity, duty cycle, and treatment duration (Table 4). This variability poses major challenges in comparing treatment outcomes, replicating study results, and establishing standardized clinical protocols. Therefore, a thorough analysis of the underlying rationale for parameter selection is crucial for advancing this field.
The diversity in applied parameters is not arbitrary but stems from several interrelated factors. Treatment choice is often guided by a specific therapeutic objective. For example, studies focused on angiogenesis and folliculogenesis in transplanted ovarian tissue have used parameters (3 MHz and 0.3 W/cm2) known to promote vascular endothelial growth factor expression [90]. In contrast, research targeting apoptosis inhibition and anti-inflammation has used settings (e.g., 0.25–0.3 MHz and 0.03–0.2 W/cm2) optimized for activating mechanosensitive signaling pathways such as the PI3K/Akt pathway and suppressing inflammatory mediators [9, 92]. Second, parameter selection is frequently influenced by precedents from other well-established applications of LIPUS (e.g., bone fracture healing) and the technical specifications of commercially available devices, which are then adapted for ovarian therapy.
The fundamental principle unifying these diverse parameters is the pursuit of a therapeutic window that maximizes beneficial non-thermal bioeffects while minimizing potential tissue damage. Key parameter selection is guided by this principle: frequency (typically 0.3–3 MHz) involves a trade-off, wherein lower frequencies (e.g., 0.3–1 MHz) offer deeper penetration for targeting the ovaries, whereas higher frequencies (e.g., 1–3 MHz) provide superior spatial resolution [32, 123]. This aspect is particularly relevant for ovarian targeting, because ovarian depth shows significant inter-individual variation. Although direct preclinical data correlating ovarian depth with metrics such as body mass index (BMI) are currently lacking, in clinical anatomy and medical imaging, increased abdominal adiposity in individuals with higher BMI has been well established to displace the ovaries to a deeper position and increase the overall attenuation of the ultrasound beam. Therefore, future clinical translation must account for patient-specific anthropometrics, and parameter optimization (probably favoring lower frequencies for patients with higher BMI) should be a key consideration in trial design.
The intensity is carefully maintained at a low level to ensure that the primary mechanisms of action are non-thermal mechanical effects, such as stable cavitation and acoustic streaming, thereby avoiding thermal injury. The use of a low duty cycle in pulsed mode is a critical safety feature allowing for effective heat dissipation between pulses [124]. Furthermore, the treatment duration and regimen (e.g., daily 20-minute sessions over 1–2 weeks) are designed to deliver a total acoustic energy dose that provides the sustained mechanical stimulation necessary to trigger cellular repair processes without causing overt damage [96].
Despite the empirical rationale for existing parameter choices, the optimal set for POI remains undefined. Future research must transition toward a mechanism-driven optimization strategy, which should involve computational modeling to predict energy deposition in ovarian tissue, systematic dose-response studies in preclinical models to establish causal links between parameters and outcomes, and ultimately international consensus efforts to standardize parameters for clinical trials. Addressing this heterogeneity will be a crucial step in translating LIPUS from a promising preclinical discovery into a reliable clinical therapy for POI.
Conclusion and future prospects
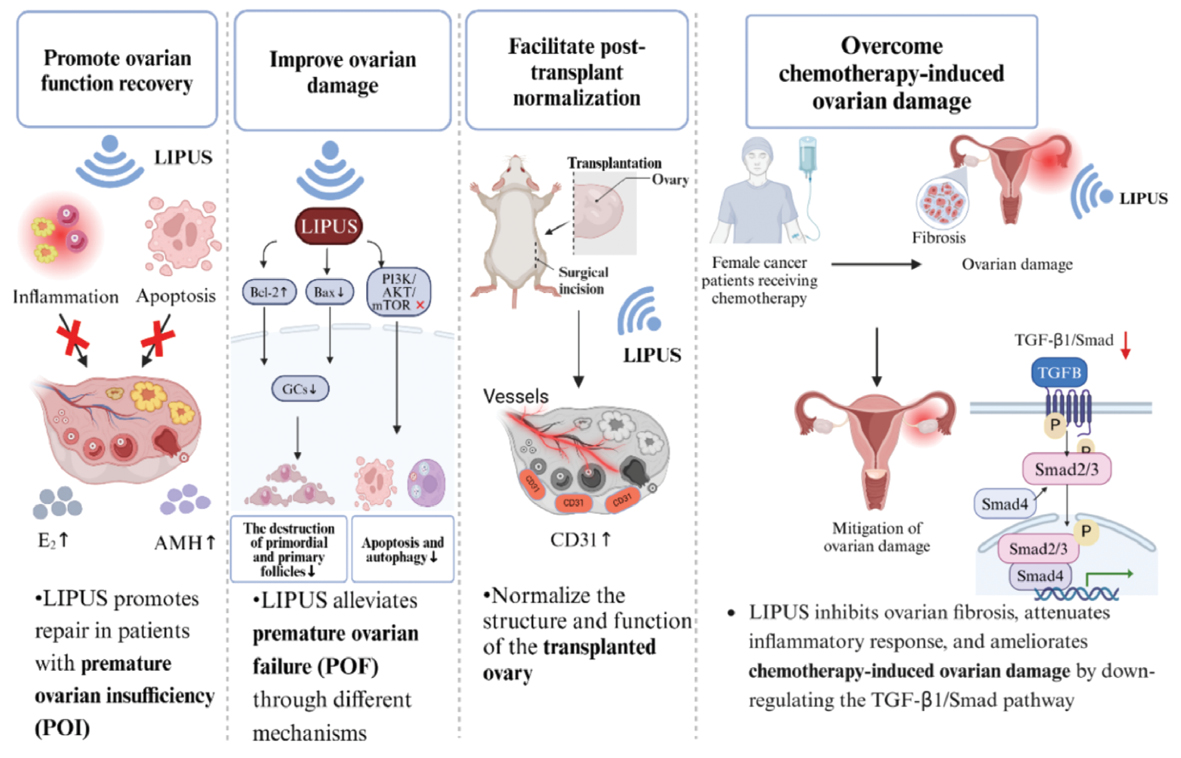
This review consolidated the growing body of evidence supporting LIPUS as a groundbreaking noninvasive therapy for POI. Preclinical studies have demonstrated that LIPUS uniquely addresses the multifactorial nature of POI through mechanical modulation of the ovarian microenvironment, thereby enhancing follicular survival, suppressing apoptosis, and mitigating fibrosis. Unlike conventional HRT, which merely alleviates symptoms, LIPUS targets the root causes of ovarian dysfunction without systemic adverse effects or hormonal dependency. Its demonstrated success in diverse areas, such as accelerating bone fracture healing, promoting cartilage repair, and enhancing soft tissue regeneration, underscores its translational potential. The mechanobiological principles of LIPUS have substantial promise for exploring the crosstalk between physical stimuli and immunomodulation in a wide range of other pathological contexts. This approach offers a paradigm shift from symptom management to functional restoration in reproductive medicine and beyond.
However, despite this promise, several limitations and potential risks of LIPUS therapy warrant consideration. First, the optimal therapeutic parameters (e.g., frequency, intensity, duty cycle, and exposure duration) remain undefined for ovarian tissue. Current preclinical studies have used heterogeneous settings (Table 4), thus complicating clinical translation and standardization. Second, the depth-dependent attenuation of ultrasound waves might limit energy delivery to deeply located ovaries, particularly in patients with high body mass indices. Third, long-term biosafety data are scarce; although LIPUS is nonthermal and noninvasive, repeated mechanical stimulation could theoretically induce unintended cellular stress or genomic instability. Moreover, the risk of cavitation-mediated tissue damage persists if parameters exceed safe thresholds (e.g., transient cavitation at high pressures) and has potential to exacerbate ovarian injury. Finally, potential synergistic interactions with concurrent therapies (e.g., chemotherapy agents) remain unexplored, and concerns have been raised regarding unanticipated interference or toxicity.
To address these challenges, future research must prioritize parameter optimization through computational modeling of tissue-specific dosimetry, alongside rigorous long-term safety assessments, including teratogenicity studies, in large-animal models. The development of real-time cavitation monitoring systems will be critical to mitigate the risk of tissue damage, and well-designed clinical trials should evaluate efficacy across diverse POI etiologies and patient phenotypes. Systematic bridging of these gaps might enable LIPUS to become a targeted and safe cornerstone for ovarian rejuvenation.
Importantly, despite the compelling preclinical evidence summarized in this review, the application of LIPUS for POI treatment remains in early stages. To our knowledge, no clinical trials of LIPUS for POI have been registered or conducted to date. The transition from robust animal models, as described in the previous sections, to human patients is a critical next step and current frontier in this field.
In addition, to achieve more efficient and safer applications, the clinical translation of LIPUS will require interdisciplinary collaboration. Integrating LIPUS with emerging technologies, such as stem cell therapy or ultrasound-responsive biomaterials, could amplify regenerative outcomes through synergistic mechanisms. Collaboration among engineers, clinicians, and regulatory bodies is essential to standardize protocols, optimize parameters, and validate safety in diverse populations. Concurrently, longitudinal studies are needed to map mechanosensitive biomarkers, monitor fertility outcomes, and ensure sustained efficacy. By bridging preclinical innovation with clinical rigor, LIPUS has the potential to redefine POI treatment and to restore not only hormonal balance but also fertility, as a transformative advancement for millions of women worldwide.
Data availability statement
Not applicable.
Ethics statement
This study did not involve human participants, their data, or biological material.
Author contributions
Zenan Zhang: writing—original draft preparation, investigation, visualization, and validation; Chenke Kuang: writing—review, visualization, and editing; Zhili Guo: visualization and validation; Weidong Liang: validation and supervision. All authors have read and agreed to the published version of the manuscript.
Funding
No funding or sponsorship was received for this study.
Acknowledgments
Not applicable.
Conflict of interest
The authors declare that there are no conflicts of interest.
Abbreviations
| AFMSC | Amniotic fluid mesenchymal stem cell |
| AMH | Anti-Müllerian hormone |
| Bax | BCL2-associated X |
| Bcl-2 | B-cell lymphoma-2 |
| BMMSC | Bone marrow mesenchymal stem cell |
| CTX | Cyclophosphamide |
| CXCR4 | CXC-chemokine receptor 4 |
| E2 | Estradiol |
| FAK | Focal adhesion kinase |
| FDA | Food and Drug Administration |
| FSH | Follicle-stimulating hormone |
| GC | Granulosa cell |
| hAD-MSC | Human amniotic membrane mesenchymal stem cell |
| HRT | Hormone replacement therapy |
| IVA | In vitro follicular activation |
| LIPUS | Low-intensity pulsed ultrasound |
| MMP | Matrix metalloproteinases |
| MSCs | Mesenchymal stem cells |
| POF | Premature ovarian failure |
| POI | Premature ovarian insufficiency |
| PRP | Platelet-rich plasma |
| SDF-1 | Stromal cell-derived factor-1 |
| TCM | Traditional Chinese medicine |
| TFSC | Total flavonoids from Semen Cuscutae |
| Th17 | Helper T cells |
Graphical abstract
Highlights
- LIPUS offers a noninvasive, hormone-free therapeutic strategy for POI.
- It enhances ovarian repair and decreases apoptosis through mechanobiological mechanisms.
- Synergy with stem cells or traditional medicine enhances its therapeutic potential.
In brief
This review evaluates LIPUS as a novel treatment for POI. LIPUS, through mechanobiological effects, improves ovarian function and follicle survival. The article also discusses its combination with stem cell therapy and traditional medicine, and outlines future directions for clinical translation.
References
- Nash Z, Davies M. Premature ovarian insufficiency. BMJ 2024;384:e077469. [PMID: 38508679 DOI: 10.1136/bmj-2023-077469]
- Ishizuka B. Current understanding of the etiology, symptomatology, and treatment options in premature ovarian insufficiency (POI). Front Endocrinol 2021;12:626924. [PMID: 33716979 DOI: 10.3389/fendo.2021.626924]
- Zhang Y, Zhao J, Han L, Zhang Z, Wang C, et al. Research progress of extracellular vesicles in the treatment of ovarian diseases (Review). Exp Ther Med 2024;27:15. [PMID: 38125352 DOI: 10.3892/etm.2023.12303]
- Ding X, Lv S, Guo Z, Gong X, Wang C, et al. Potential therapeutic options for premature ovarian insufficiency: experimental and clinical evidence. Reprod Sci 2023;30(12):3428-42. [PMID: 37460850 DOI: 10.1007/s43032-023-01300-1]
- Committee opinion No. 698 Summary: hormone therapy in primary ovarian insufficiency. Obstet Gynecol 2017;129(5):963-4. [PMID: 28426614 DOI: 10.1097/aog.0000000000002040]
- Zhang S, Zhu D, Mei X, Li Z, Li J, et al. Advances in biomaterials and regenerative medicine for primary ovarian insufficiency therapy. Bioact Mater 2021;6(7):1957-72. [PMID: 33426370 DOI: 10.1016/j.bioactmat.2020.12.008]
- Dai F, Liu H, He J, Wu J, Yuan C, et al. Model construction and drug therapy of primary ovarian insufficiency by ultrasound-guided injection. Stem Cell Res Ther 2024;15(1):49. [PMID: 38378684 DOI: 10.1186/s13287-024-03646-y]
- Pepin A, Chesnokova A, Pishko A, Gysler S, Martin C, et al. Hormone replacement therapy in patients with gynecologic cancer and radiation-induced premature ovarian insufficiency. Int J Radiat Oncol Biol Phys 2025;121(4):1042-52. [PMID: 39448037 DOI: 10.1016/j.ijrobp.2024.10.023]
- Qin J, Chen J, Xu H, Xia Y, Tang W, et al. Low-intensity pulsed ultrasound promotes repair of 4-vinylcyclohexene diepoxide-induced premature ovarian insufficiency in SD rats. J Gerontol A Biol Sci Med Sci 2021;77(2):221-7. [PMID: 34417809 DOI: 10.1093/gerona/glab242]
- Zangmo R, Suresh G, Sarkar A, Ramu S, Roy KK, et al. The effect of salpingectomy on ovarian reserve using two different electrosurgical instruments: ultrasonic shears versus bipolar electrocautery. Cureus 2024;16(5):e59434. [PMID: 38826994 DOI: 10.7759/cureus.59434]
- Xia P, Wang Q, Song J, Wang X, Wang X, et al. Low-intensity pulsed ultrasound enhances the efficacy of bone marrow-derived MSCs in osteoarthritis cartilage repair by regulating autophagy-mediated exosome release. Cartilage 2022;13(2):19476035221093060. [PMID: 35438034 DOI: 10.1177/19476035221093060]
- Palanisamy P, Alam M, Li S, Chow SKH, Zheng YP. Low-intensity pulsed ultrasound stimulation for bone fractures healing: a review. J Ultrasound Med 2022;41(3):547-63. [PMID: 33949710 DOI: 10.1002/jum.15738]
- He Y, Ye R, Peng Y, Pei Q, Wu L, et al. Photobiomodulation ameliorates ovarian aging by alleviating oxidative stress and inflammation damage and improving mitochondrial function. J Photochem Photobiol B 2024;260:113024. [PMID: 39276447 DOI: 10.1016/j.jphotobiol.2024.113024]
- Jiaman WU, Meng T, Yu L, Haimin Z, Tianqi Z, et al. Electroacupuncture enhances the mitophagy of granulosa cells in premature ovarian insufficiency model mice by inactivating the hippo-yes-associated protein/transcriptional co-activator with postsynaptic density protein, drosophila disc large tumor suppressor, and zonula occludens-1 protein binding motif pathway. J Tradit Chin Med 2025;45(1):13-21. [PMID: 39957154 DOI: 10.19852/j.cnki.jtcm.2025.01.002]
- Tang H, Liu Y, Fan Y, Li C. Therapeutic effects of low-intensity pulsed ultrasound on premature ovarian insufficiency. Ultrasound Med Biol 2021;47(8):2377-87. [PMID: 34088530 DOI: 10.1016/j.ultrasmedbio.2021.04.022]
- Jiang X, Savchenko O, Li Y, Qi S, Yang T, et al. A review of low-intensity pulsed ultrasound for therapeutic applications. IEEE Trans Biomed Eng 2019;66(10):2704-18. [PMID: 30596564 DOI: 10.1109/TBME.2018.2889669]
- Ji X, Duan H, Wang S, Chang Y. Low-intensity pulsed ultrasound in obstetrics and gynecology: advances in clinical application and research progress. Front Endocrinol 2023;14:1233187. [PMID: 37593351 DOI: 10.3389/fendo.2023.1233187]
- Ulin M, Cetin E, Hobeika E, Chugh RM, Park HS, et al. Human mesenchymal stem cell therapy and other novel treatment approaches for premature ovarian insufficiency. Reprod Sci 2021;28(6):1688-96. [PMID: 33956339 DOI: 10.1007/s43032-021-00528-z]
- Chae-Kim JJ, Gavrilova-Jordan L. Premature ovarian insufficiency: procreative management and preventive strategies. Biomedicines 2018;7(1):2. [PMID: 30597834 DOI: 10.3390/biomedicines7010002]
- Meng X, Peng L, Wei X, Li S. FOXO3 is a potential biomarker and therapeutic target for premature ovarian insufficiency (Review). Mol Med Rep 2023;27(2):34. [PMID: 36562359 DOI: 10.3892/mmr.2022.12921]
- Agarwal S, Alzahrani FA, Ahmed A. Hormone replacement therapy: would it be possible to replicate a functional ovary? Int J Mol Sci 2018;19(10):3160. [PMID: 30322209 DOI: 10.3390/ijms19103160]
- Anagnostis P, Paschou SA, Katsiki N, Krikidis D, Lambrinoudaki I, et al. Menopausal hormone therapy and cardiovascular risk: where are we now? Curr Vasc Pharmacol 2019;17(6):564-72. [PMID: 29984659 DOI: 10.2174/1570161116666180709095348]
- Baber R. Treating menopausal women: have we lost our way? Aust N Z J Obstet Gynaecol 2021;61(4):493-5. [PMID: 34378184 DOI: 10.1111/ajo.13381]
- Sun B, Li L, Zhang Y, Wang F, Sun Y. Pregnancy outcomes in women with primary ovarian insufficiency in assisted reproductive technology therapy: a retrospective study. Front Endocrinol (Lausanne) 2024;15:1343803. [PMID: 38745952 DOI: 10.3389/fendo.2024.1343803]
- Foster KL, Lee DJ, Witchel SF, Gordon CM. Ovarian insufficiency and fertility preservation during and after childhood cancer treatment. J Adolesc Young Adult Oncol 2024;13(3):377-88. [PMID: 38265460 DOI: 10.1089/jayao.2023.0111]
- Li HF, Zhang JX, Chen WJ. Dissecting the efficacy of the use of acupuncture and Chinese herbal medicine for the treatment of premature ovarian insufficiency (POI): a systematic review and metaanalysis. Heliyon 2023;9(10):e20498. [PMID: 37818004 DOI: 10.1016/j.heliyon.2023.e20498]
- Huang QY, Chen SR, Chen JM, Shi QY, Lin S. Therapeutic options for premature ovarian insufficiency: an updated review. Reprod Biol Endocrinol 2022;20(1):28. [PMID: 35120535 DOI: 10.1186/s12958-022-00892-8]
- Shariati BKB, Ansari MA, Khatami SS, Tuchin VV. Multimodal optical clearing to minimize light attenuation in biological tissues. Sci Rep 2023;13(1):21509. [PMID: 38057535 DOI: 10.1038/s41598-023-48876-x]
- Tan R, He Y, Zhang S, Pu D, Wu J. Effect of transcutaneous electrical acupoint stimulation on protecting against radiotherapy- induced ovarian damage in mice. J Ovarian Res 2019;12(1):65. [PMID: 31324205 DOI: 10.1186/s13048-019-0541-1]
- Liu J, Li B, Li L, Ming X, Xu ZP. Advances in nanomaterials for immunotherapeutic improvement of cancer chemotherapy. Small 2024;20(38):e2403024. [PMID: 38773882 DOI: 10.1002/smll.202403024]
- Li P, Li S, Li Z, Lu W, Shao W, et al. Ultrasound propagation characteristics within the bone tissue of miniature ultrasound probes: implications for the spinal navigation of pedicle screw placement. Quant Imaging Med Surg 2024;14(7):4878-92. [PMID: 39022289 DOI: 10.21037/qims-24-377]
- Sellyn GE, Lopez AA, Ghosh S, Topf MC, Chen H, et al. High-frequency ultrasound accuracy in preoperative cutaneous melanoma assessment: a meta-analysis. J Eur Acad Dermatol Venereol 2025;39(1):86-96. [PMID: 38967397 DOI: 10.1111/jdv.20179]
- Harrison A, Lin S, Pounder N, Mikuni-Takagaki Y. Mode & mechanism of low intensity pulsed ultrasound (LIPUS) in fracture repair. Ultrasonics 2016;70:45-52. [PMID: 27130989 DOI: 10.1016/j.ultras.2016.03.016]
- Quarato CMI, Lacedonia D, Salvemini M, Tuccari G, Mastrodonato G, et al. A review on biological effects of ultrasounds: key messages for clinicians. Diagnostics (Basel) 2023;13(5):855. [PMID: 36899998 DOI: 10.3390/diagnostics13050855]
- Ashar H, Ranjan A. Immunomodulation and targeted drug delivery with high intensity focused ultrasound (HIFU): principles and mechanisms. Pharmacol Ther 2023;244:108393. [PMID: 36965581 DOI: 10.1016/j.pharmthera.2023.108393]
- Ho YJ, Hsu HC, Wu BH, Lin YC, Liao LD, et al. Preventing ischemia-reperfusion injury by acousto-mechanical local oxygen delivery. J Control Release 2023;356:481-92. [PMID: 36921723 DOI: 10.1016/j.jconrel.2023.03.018]
- Furusawa Y, Kondo T, Tachibana K, Feril LB. Ultrasound-induced DNA damage and cellular response: historical review, mechanisms analysis, and therapeutic implications. Radiat Res 2022;197(6):662-72. [PMID: 35275998 DOI: 10.1667/RADE-21-00140.1.S1]
- Smith M, Khan S, Curiel L. Investigation of hardware and software techniques to enhance the characteristics of focused ultrasound (FUS) spectra. Phys Med Biol 2022;67(14):145015. [PMID: 35613564 DOI: 10.1088/1361-6560/ac7374]
- Zhang Q, Yuan Z, Song R, Xue H, Tu J, et al. Optimized acoustic streaming generated at oblique incident angles to improve ultrasound thrombolysis effect. Med Phys 2022;49(9):5728-41. [PMID: 35860901 DOI: 10.1002/mp.15874]
- Shah JS, Sabouni R, Cayton Vaught KC, Owen CM, Albertini DF, et al. Biomechanics and mechanical signaling in the ovary: a systematic review. J Assist Reprod Genet 2018;35(7):1135-48. [PMID: 29691711 DOI: 10.1007/s10815-018-1180-y]
- Nagamatsu G, Shimamoto S, Hamazaki N, Nishimura Y, Hayashi K. Mechanical stress accompanied with nuclear rotation is involved in the dormant state of mouse oocytes. Sci Adv 2019;5(6):eaav9960. [PMID: 31249869 DOI: 10.1126/sciadv.aav9960]
- Vasse J, Fiscus J, Fraison E, Salle B, David L, et al. Biomechanical properties of ovarian tissue and their impact on the activation of follicular growth: a narrative review. Reprod Biomed Online 2025;50(3):104450. [PMID: 39919556 DOI: 10.1016/j.rbmo.2024.104450]
- Wang H, Yang L. Ovarian mechanobiology: understanding the interplay between mechanics and follicular development. Cells 2025;14(5):355. [PMID: 40072084 DOI: 10.3390/cells14050355]
- Wu S, Zhou H, Ling H, Sun Y, Luo Z, et al. LIPUS regulates the progression of knee osteoarthritis in mice through primary cilia-mediated TRPV4 channels. Apoptosis 2024;29(5):785-98. [PMID: 38517601 DOI: 10.1007/s10495-024-01950-9]
- Han DS, Lee CH, Shieh YD, Chang KV, Lin SH, et al. Involvement of ASIC3 and substance P in therapeutic ultrasound-mediated analgesia in mouse models of fibromyalgia. J Pain 2023;24(8):1493-505. [PMID: 37054767 DOI: 10.1016/j.jpain.2023.04.003]
- Przystupski D, Baczyńska D, Rossowska J, Kulbacka J, Ussowicz M. Calcium ion delivery by microbubble-assisted sonoporation stimulates cell death in human gastrointestinal cancer cells. Biomed Pharmacother 2024;179:117339. [PMID: 39216448 DOI: 10.1016/j.biopha.2024.117339]
- Sahni J, McCue IS, Johnson AR, Schake MA, Sotelo LD, et al. Ultrasound induces similar temporal endothelial expression patterns of eNOS and KLF2 as normal flow. Ultrasound Med Biol 2024;50(12):1893-902. [PMID: 39306482 DOI: 10.1016/j.ultrasmedbio.2024.08.017]
- Huangfu Q, Zhang J, Xu J, Xu J, Yang Z, et al. Mechanosensitive Ca2+ channel TRPV1 activated by low-intensity pulsed ultrasound ameliorates acute kidney injury through Notch1-Akt-eNOS signaling. FASEB J 2025;39(1):e70304. [PMID: 39785696 DOI: 10.1096/fj.202401142RR]
- Xiao H, Yan A, Li M, Wang L, Xiang J. LIPUS accelerates bone regeneration via HDAC6-mediated ciliogenesis. Biochem Biophys Res Commun 2023;641:34-41. [PMID: 36521283 DOI: 10.1016/j.bbrc.2022.12.010]
- Chu YC, Lim J, Chien A, Chen CC, Wang JL. Activation of mechanosensitive ion channels by ultrasound. Ultrasound Med Biol 2022;48(10):1981-94. [PMID: 35945063 DOI: 10.1016/j.ultrasmedbio.2022.06.008]
- Rahman R, Panay N. Diagnosis and management of premature ovarian insufficiency. Best Pract Res Clin Endocrinol Metab 2021;35(6):101600. [PMID: 34823999 DOI: 10.1016/j.beem.2021.101600]
- Liu X, Zou D, Hu Y, He Y, Lu J. Research progress of low-intensity pulsed ultrasound in the repair of peripheral nerve injury. Tissue Eng Part B Rev 2023;29(4):414-28. [PMID: 36785967 DOI: 10.1089/ten.TEB.2022.0194]
- Puts R, Rikeit P, Ruschke K, Kadow-Romacker A, Hwang S, et al. Activation of mechanosensitive transcription factors in murine C2C12 mesenchymal precursors by focused low-intensity pulsed ultrasound (FLIPUS). IEEE Trans Ultrason Ferroelectr Freq Control 2016;63(10):1505-13. [PMID: 27392348 DOI: 10.1109/TUFFC.2016.2586972]
- Chen Y, Wang S, Zhang C. The differentiation fate of granulosa cells and the regulatory mechanism in ovary. Reprod Sci 2025;32(5):1414-26. [PMID: 39192066 DOI: 10.1007/s43032-024-01682-w]
- Zhu L, Yao X, Mo Y, Chen MW, Li SC, et al. miR-4433a-3p promotes granulosa cell apoptosis by targeting peroxisome proliferator-activated receptor alpha and inducing immune cell infiltration in polycystic ovarian syndrome. J Assist Reprod Genet 2023;40(6):1447-59. [PMID: 37204637 DOI: 10.1007/s10815-023-02815-x]
- di Clemente N, Racine C, Rey RA. Anti-müllerian hormone and polycystic ovary syndrome in women and its male equivalent. Biomedicines 2022;10(10):2506. [PMID: 36289767 DOI: 10.3390/biomedicines10102506]
- Lin Z, Gao L, Hou N, Zhi X, Zhang Y, et al. Application of low-intensity pulsed ultrasound on tissue resident stem cells: potential for ophthalmic diseases. Front Endocrinol 2023;14:1153793. [PMID: 37008913 DOI: 10.3389/fendo.2023.1153793]
- Fontana F, Iberite F, Cafarelli A, Aliperta A, Baldi G, et al. Development and validation of low-intensity pulsed ultrasound systems for highly controlled in vitro cell stimulation. Ultrasonics 2021;116:106495. [PMID: 34186322 DOI: 10.1016/j.ultras.2021.106495]
- Lim J, Tai HH, Liao WH, Chu YC, Hao CM, et al. ASIC1a is required for neuronal activation via low-intensity ultrasound stimulation in mouse brain. ELife 2021;10:e61660. [PMID: 34569932 DOI: 10.7554/eLife.61660]
- Takeuchi R, Ryo A, Komitsu N, Mikuni-Takagaki Y, Fukui A, et al. Low-intensity pulsed ultrasound activates the phosphatidylinositol 3 kinase/Akt pathway and stimulates the growth of chondrocytes in three-dimensional cultures: a basic science study. Arthritis Res Ther 2008;10(4):R77. [PMID: 18616830 DOI: 10.1186/ar2451]
- Duan H, Chen S, Mai X, Fu L, Huang L, et al. Low-intensity pulsed ultrasound (LIPUS) promotes skeletal muscle regeneration by regulating PGC-1α/AMPK/GLUT4 pathways in satellite cells/myoblasts. Cell Signal 2024;117:111097. [PMID: 38355078 DOI: 10.1016/j.cellsig.2024.111097]
- Huang B, Jiang Y, Zhang L, Yang B, Guo Y, et al. Low-intensity pulsed ultrasound promotes proliferation and myelinating genes expression of Schwann cells through NRG1/ErbB signaling pathway. Tissue Cell 2023;80:101985. [PMID: 36459840 DOI: 10.1016/j.tice.2022.101985]
- Zhou S, Schmelz A, Seufferlein T, Li Y, Zhao J, et al. Molecular mechanisms of low intensity pulsed ultrasound in human skin fibroblasts. J Biol Chem 2004;279(52):54463-9. [PMID: 15485877 DOI: 10.1074/jbc.M404786200]
- Ahmed M, Riaz U, Lv H, Amjad M, Ahmed S, et al. Nicotinamide mononucleotide restores NAD+ levels to alleviate LPS-induced inflammation via the TLR4/NF-κB/MAPK signaling pathway in mice granulosa cells. Antioxidants (Basel) 2024;14(1):39. [PMID: 39857373 DOI: 10.3390/antiox14010039]
- Orisaka M, Mizutani T, Miyazaki Y, Shirafuji A, Tamamura C, et al. Chronic low-grade inflammation and ovarian dysfunction in women with polycystic ovarian syndrome, endometriosis, and aging. Front Endocrinol (Lausanne) 2023;14:1324429. [PMID: 38192421 DOI: 10.3389/fendo.2023.1324429]
- Han Y, Yao R, Yang Z, Li S, Meng W, et al. Interleukin-4 activates the PI3K/AKT signaling to promote apoptosis and inhibit the proliferation of granulosa cells. Exp Cell Res 2022;412(1):113002. [PMID: 34973261 DOI: 10.1016/j.yexcr.2021.113002]
- Park SA, Joo NR, Park JH, Oh SM. Role of the SIRT1/p53 regulatory axis in oxidative stress-mediated granulosa cell apoptosis. Mol Med Rep 2021;23(1):20. [PMID: 33179088 DOI: 10.3892/mmr.2020.11658]
- Yuan X, Li Z, Kong Y, Zhong Y, He Y, et al. P65 targets FGFR1 to regulate the survival of ovarian granulosa cells. Cells 2019;8(11):1334. [PMID: 31671754 DOI: 10.3390/cells8111334]
- Yang Z, Hong W, Zheng K, Feng J, Hu C, et al. Chitosan oligosaccharides alleviate H2O2-stimulated granulosa cell damage via HIF-1α signaling pathway. Oxid Med Cell Longev 2022;2022:4247042. [PMID: 35401926 DOI: 10.1155/2022/4247042]
- Geng Z, Guo H, Li Y, Liu Y, Zhao Y. Stem cell-derived extracellular vesicles: a novel and potential remedy for primary ovarian insufficiency. Front Cell Dev Biol 2023;11:1090997. [PMID: 36875770 DOI: 10.3389/fcell.2023.1090997]
- Jian Z, Li Y, Zhang C, Zhong W, Ai D, et al. Low-intensity pulsed ultrasound attenuates periodontal ligament cells apoptosis by activating yes-associated protein-regulated autophagy. Ultrasound Med Biol 2023;49(5):1227-37. [PMID: 36878833 DOI: 10.1016/j.ultrasmedbio.2023.01.008]
- Zhou JX, Liu YJ, Chen X, Zhang X, Xu J, et al. Low-intensity pulsed ultrasound protects retinal ganglion cell from optic nerve injury induced apoptosis via yes associated protein. Front Cell Neurosci 2018;12:160. [PMID: 29950973 DOI: 10.3389/fncel.2018.00160]
- Li X, Zhong Y, Zhang L, Xie M. Recent advances in the molecular mechanisms of low-intensity pulsed ultrasound against inflammation. J Mol Med (Berl) 2023;101(4):361-74. [PMID: 36905436 DOI: 10.1007/s00109-023-02302-x]
- Sang F, Xu J, Chen Z, Liu Q, Jiang W. Low-intensity pulsed ultrasound alleviates osteoarthritis condition through focal adhesion kinase-mediated chondrocyte proliferation and differentiation. Cartilage 2021;13(suppl 2):196s-203s. [PMID: 32281401 DOI: 10.1177/1947603520912322]
- Nakao J, Fujii Y, Kusuyama J, Bandow K, Kakimoto K, et al. Low-intensity pulsed ultrasound (LIPUS) inhibits LPS-induced inflammatory responses of osteoblasts through TLR4-MyD88 dissociation. Bone 2014;58:17-25. [PMID: 24091132 DOI: 10.1016/j.bone.2013.09.018]
- Gouda SAA, Aboulhoda BE, Abdelwahed OM, Abdallah H, Rashed L, et al. Low-intensity pulsed ultrasound (LIPUS) switched macrophage into M2 phenotype and mitigated necroptosis and increased HSP 70 in gentamicin-induced nephrotoxicity. Life Sci 2023;314:121338. [PMID: 36592788 DOI: 10.1016/j.lfs.2022.121338]
- Fu S, Guo Z, Xu X, Li Y, Choi S, et al. Protective effect of low-intensity pulsed ultrasound on immune checkpoint inhibitor-related myocarditis via fine-tuning CD4+ T-cell differentiation. Cancer Immunol Immunother 2024;73(1):15. [PMID: 38236243 DOI: 10.1007/s00262-023-03590-5]
- Cho J, Kim TH, Seok J, Jun JH, Park H, et al. Vascular remodeling by placenta-derived mesenchymal stem cells restores ovarian function in ovariectomized rat model via the VEGF pathway. Lab Invest 2021;101(3):304-17. [PMID: 33303971 DOI: 10.1038/s41374-020-00513-1]
- Umehara T, Winstanley YE, Andreas E, Morimoto A, Williams EJ, et al. Female reproductive life span is extended by targeted removal of fibrotic collagen from the mouse ovary. Sci Adv 2022;8(24):eabn4564. [PMID: 35714185 DOI: 10.1126/sciadv.abn4564]
- Yu M, Bian Y, Wang L, Chen F. Low-intensity pulsed ultrasound enhances angiogenesis in rabbit capsule tissue that acts as a novel vascular bed in vivo. Adv Clin Exp Med 2021;30(6):581-9. [PMID: 34004084 DOI: 10.17219/acem/134115]
- Huang JJ, Shi YQ, Li RL, Hu A, Lu ZY, et al. Angiogenesis effect of therapeutic ultrasound on HUVECs through activation of the PI3K-Akt-eNOS signal pathway. Am J Transl Res 2015;7(6):1106-15. [PMID: 26279754]
- Xu XM, Xu TM, Wei YB, Gao XX, Sun JC, et al. Low-intensity pulsed ultrasound treatment accelerates angiogenesis by activating YAP/TAZ in human umbilical vein endothelial cells. Ultrasound Med Biol 2018;44(12):2655-61. [PMID: 30205992 DOI: 10.1016/j.ultrasmedbio.2018.07.007]
- Costa V, Carina V, Conigliaro A, Raimondi L, De Luca A, et al. miR-31-5p is a LIPUS-mechanosensitive microRNA that targets HIF-1α signaling and cytoskeletal proteins. Int J Mol Sci 2019;20(7):1569. [PMID: 30925808 DOI: 10.3390/ijms20071569]
- Zhao K, Zhang J, Xu T, Yang C, Weng L, et al. Erratum to: Low-intensity pulsed ultrasound ameliorates angiotensin II-induced cardiac fibrosis by alleviating inflammation via a caveolin-1-dependent pathway. J Zhejiang Univ Sci B 2024;25(9):800. [PMID: 39308070 DOI: 10.1631/jzus.B21e0130]
- Ouyang ZQ, Shao LS, Wang WP, Ke TF, Chen D, et al. Low intensity pulsed ultrasound ameliorates Adriamycin-induced chronic renal injury by inhibiting ferroptosis. Redox Rep 2023;28(1):2251237. [PMID: 37652897 DOI: 10.1080/13510002.2023.2251237]
- Hu L, Wang Y, Wan Y, Ma L, Zhao T, et al. Tangshen formula improves diabetes-associated myocardial fibrosis by inhibiting TGF-β/Smads and Wnt/β-catenin pathways. Front Med (Lausanne) 2021;8:732042. [PMID: 34938743 DOI: 10.3389/fmed.2021.732042]
- Liao B, Guan M, Tan Q, Wang G, Zhang R, et al. Low-intensity pulsed ultrasound inhibits fibroblast-like synoviocyte proliferation and reduces synovial fibrosis by regulating Wnt/β-catenin signaling. J Orthop Translat 2021;30:41-50. [PMID: 34611513 DOI: 10.1016/j.jot.2021.08.002]
- Cheung WH, Chin WC, Qin L, Leung KS. Low intensity pulsed ultrasound enhances fracture healing in both ovariectomy-induced osteoporotic and age-matched normal bones. J Orthop Res 2012;30(1):129-36. [PMID: 21688313 DOI: 10.1002/jor.21487]
- Ter Haar G, Dyson M, Talbert D. Ultrasonically induced contractions in mouse uterine smooth muscle in vivo. Ultrasonics 1978;16(6):275-6. [PMID: 568839 DOI: 10.1016/0041-624x(78)90055-0]
- Abtahi NS, Eimani H, Vosough A, Shahverdi A, Fathi R, et al. Effect of therapeutic ultrasound on folliculogenesis, angiogenesis and apoptosis after heterotopic mouse ovarian transplantation. Ultrasound Med Biol 2014;40(7):1535-44. [PMID: 24785439 DOI: 10.1016/j.ultrasmedbio.2014.02.006]
- Tang HJ, Yang H, Fan YJ, Li CZ. Low-intensity pulsed ultrasound promotes repair of cyclophosphamide-induced ovarian injury in rats. Nan Fang Yi Ke Da Xue Xue Bao 2017;37(12):1632-6. [PMID: 29292257 DOI: 10.3969/j.issn.1673-4254.2017.12.12]
- Xu H, Xia Y, Qin J, Xu J, Li C, et al. Effects of low intensity pulsed ultrasound on expression of B-cell lymphoma-2 and BCL2-associated X in premature ovarian failure mice induced by 4-vinylcyclohexene diepoxide. Reprod Biol Endocrinol 2021;19(1):113. [PMID: 34284777 DOI: 10.1186/s12958-021-00799-w]
- Liao Q, Chen J, Liu G. Low intensity pulsed ultrasound alleviates synovial fibrosis in osteoarthritis via the PI3K/AKT pathway. Sci Rep 2025;15(1):9644. [PMID: 40113833 DOI: 10.1038/s41598-025-92413-x]
- Zhou T, Zhou CX, Zhang QB, Wang F, Zhou Y. LIPUS alleviates knee joint capsule fibrosis in rabbits by regulating SOD/ROS dynamics and inhibiting the TGF-β1/Smad signaling pathway. Ultrasound Med Biol 2023;49(12):2510-8. [PMID: 37714800 DOI: 10.1016/j.ultrasmedbio.2023.08.014]
- Zhou Y, Zhu F, Zhou Y, Li X, Zhao S, et al. The value of low-intensity pulsed ultrasound in reducing ovarian injury caused by chemotherapy in mice. Reprod Biol Endocrinol 2024;22(1):51. [PMID: 38671458 DOI: 10.1186/s12958-024-01216-8]
- Deng J, Qin J, Song G, Li C, Tang W, et al. The potential of low-intensity pulsed ultrasound to apply the long-term ovary protection from injury induced by 4-vinylcyclohexene diepoxide through inhibiting granulosa cell apoptosis. Bioeng Transl Med 2024;10(3):e10744. [PMID: 40385545 DOI: 10.1002/btm2.10744].
- Cantaluppi V, Biancone L, Quercia A, Deregibus MC, Segoloni G, et al. Rationale of mesenchymal stem cell therapy in kidney injury. Am J Kidney Dis 2013;61(2):300-9. [PMID: 22938846 DOI: 10.1053/j.ajkd.2012.05.027]
- Na J, Kim GJ. Recent trends in stem cell therapy for premature ovarian insufficiency and its therapeutic potential: a review. J Ovarian Res 2020;13(1):74. [PMID: 32576209 DOI: 10.1186/s13048-020-00671-2]
- Fu YX, Ji J, Shan F, Li J, Hu R. Human mesenchymal stem cell treatment of premature ovarian failure: new challenges and opportunities. Stem Cell Res Ther 2021;12(1):161. [PMID: 33658073 DOI: 10.1186/s13287-021-02212-0]
- He Y, Chen D, Yang L, Hou Q, Ma H, et al. The therapeutic potential of bone marrow mesenchymal stem cells in premature ovarian failure. Stem Cell Res Ther 2018;9(1):263. [PMID: 30286808 DOI: 10.1186/s13287-018-1008-9]
- Wang L, Liu Y, Song Y, Mei Q, Mou H, et al. Enhancing oocyte quality in aging mice: insights from mesenchymal stem cell therapy and FOXO3a signaling pathway activation. Reprod Sci 2024;31(8):2392-408. [PMID: 38532230 DOI: 10.1007/s43032-024-01509-8]
- Chen H, Liu C, Zhu S, Li S, Zhang Q, et al. The therapeutic effect of stem cells on chemotherapy-induced premature ovarian failure. Curr Mol Med 2021;21(5):376-84. [PMID: 32888266 DOI: 10.2174/1566524020666200905113907]
- Geng L, Tang X, Wang S, Sun Y, Wang D, et al. Reduced let-7f in bone marrow-derived mesenchymal stem cells triggers Treg/Th17 imbalance in patients with systemic lupus erythematosus. Front Immunol 2020;11:233. [PMID: 32133007 DOI: 10.3389/fimmu.2020.00233]
- Liang Z, Yang Z, Xie H, Rao J, Xu X, et al. Small extracellular vesicles from hypoxia-preconditioned bone marrow mesenchymal stem cells attenuate spinal cord injury via miR-146a-5p-mediated regulation of macrophage polarization. Neural Regen Res 2024;19(10):2259-69. [PMID: 38488560 DOI: 10.4103/1673-5374.391194]
- Liang B, Liang JM, Ding JN, Xu J, Xu JG, et al. Dimethyloxaloylglycine-stimulated human bone marrow mesenchymal stem cell-derived exosomes enhance bone regeneration through angiogenesis by targeting the AKT/mTOR pathway. Stem Cell Res Ther 2019;10(1):335. [PMID: 31747933 DOI: 10.1186/s13287-019-1410-y]
- DelaRosa O, Lombardo E, Beraza A, Mancheño-Corvo P, Ramirez C, et al. Requirement of IFN-γ-mediated indoleamine 2,3-dioxygenase expression in the modulation of lymphocyte proliferation by human adipose-derived stem cells. Tissue Eng Part A 2009;15(10):2795-806. [PMID: 19231921 DOI: 10.1089/ten.TEA.2008.0630]
- Wang YC, Chen RF, Liu KF, Chen WY, Lee CC, et al. Adipose-derived stem cell modulate tolerogenic dendritic cell-induced T cell regulation is correlated with activation of Notch-NFκB signaling. Cytotherapy 2024;26(8):890-8. [PMID: 38625070 DOI: 10.1016/j.jcyt.2024.03.482]
- Zhou K, Guo S, Tong S, Sun Q, Li F, et al. Immunosuppression of human adipose-derived stem cells on T cell subsets via the reduction of NF-kappaB activation mediated by PD-L1/PD-1 and Gal-9/TIM-3 pathways. Stem Cells Dev 2018;27(17):1191-202. [PMID: 29978730 DOI: 10.1089/scd.2018.0033]
- Fu X, He Y, Xie C, Liu W. Bone marrow mesenchymal stem cell transplantation improves ovarian function and structure in rats with chemotherapy-induced ovarian damage. Cytotherapy 2008;10(4):353-63. [PMID: 18574768 DOI: 10.1080/14653240802035926]
- Xie S, Jiang X, Wang R, Xie S, Hua Y, et al. Low-intensity pulsed ultrasound promotes the proliferation of human bone mesenchymal stem cells by activating PI3K/AKt signaling pathways. J Cell Biochem 2019;120(9):15823-33. [PMID: 31090943 DOI: 10.1002/jcb.28853]
- Kusuyama J, Bandow K, Shamoto M, Kakimoto K, Ohnishi T, et al. Low intensity pulsed ultrasound (LIPUS) influences the multilineage differentiation of mesenchymal stem and progenitor cell lines through ROCK-Cot/Tpl2-MEK-ERK signaling pathway. J Biol Chem 2014;289(15):10330-44. [PMID: 24550383 DOI: 10.1074/jbc.M113.546382]
- Wei FY, Leung KS, Li G, Qin J, Chow SK, et al. Low intensity pulsed ultrasound enhanced mesenchymal stem cell recruitment through stromal derived factor-1 signaling in fracture healing. PLoS One 2014;9(9):e106722. [PMID: 25181476 DOI: 10.1371/journal.pone.0106722]
- Ling L, Hou J, Wang Y, Shu H, Huang Y. Effects of low-intensity pulsed ultrasound on the migration and homing of human amnion–derived mesenchymal stem cells to ovaries in rats with premature ovarian insufficiency. Cell Transplant 2022;31:9636897221129171. [PMID: 36282038 DOI: 10.1177/09636897221129171]
- Wang L, Liu J, Nie G, Li Y, Yang H. Danggui Buxue Tang rescues folliculogenesis and ovarian cell apoptosis in rats with premature ovarian insufficiency. Evid Based Complement Alternat Med 2021;2021(1):6614302. [PMID: 34035823 DOI: 10.1155/2021/6614302]
- Mei S, Yu C, Ding J, Cheng W. A network pharmacology study on the similarities and differences in the mechanisms of Zuo Gui Wan/You Gui Wan for the treatment of premature ovarian failure. Comb Chem High Throughput Screen 2023;26(6):1167-79. [PMID: 35657051 DOI: 10.2174/1386207325666220602114701]
- Wang Y, Teng X, Liu J. Research progress on the effect of traditional Chinese medicine on signal pathway related to premature ovarian insufficiency. Evid Based Complement Alternat Med 2022;2022(2):7012978. [PMID: 36159578 DOI: 10.1155/2022/7012978]
- Wu Y, Long Y, Su G, Fan X, He G, et al. Network pharmacology, molecular docking, and experimental validation on Guiluoshi Anzang decoction against premature ovarian insufficiency. Comb Chem High Throughput Screen 2025;28(4):724-36. [PMID: 38757315 DOI: 10.2174/0113862073291139240506114446]
- Chen L, Zeng L, Pan S, Zu L, Pan H, et al. β-sitosterol in Yijing Hugui decoction prevents cyclophosphamide-induced premature ovarian insufficiency via the AKT1/Nrf2 pathway. Cytotechnology 2025;77(2):76. [PMID: 40078376 DOI: 10.1007/s10616-025-00740-8]
- Chen S, Lu Y, Chen Y, Xu J, Chen L, et al. The effect of Bu Shen Huo Xue Tang on autoimmune premature ovarian insufficiency via modulation of the Nrf2/Keap1 signaling pathway in mice. J Ethnopharmacol 2021;273:113996. [PMID: 33684516 DOI: 10.1016/j.jep.2021.113996]
- Li Y, Wang L, Liu J, Nie G, Yang H. Huyang Yangkun formula regulates the mitochondria pathway of ovarian granulosa cell apoptosis through FTO/m6A-P53 pathway. Front Pharmacol 2024;15:1491546. [PMID: 39584135 DOI: 10.3389/fphar.2024.1491546]
- Zhou W, Chen A, Ye Y, Ren Y, Lu J, et al. LIPUS combined with TFSC alleviates premature ovarian failure by promoting autophagy and inhibiting apoptosis. Gynecol Endocrinol 2023;39(1):2258422. [PMID: 37855244 DOI: 10.1080/09513590.2023.2258422]
- Ma S, Wu J, Liu Z, He R, Wang Y, et al. Quantitative characterization of cell physiological state based on dynamical cell mechanics for drug efficacy indication. J Pharm Anal 2023;13(4):388-402. [PMID: 37181289 DOI: 10.1016/j.jpha.2023.03.002]
- Shi L, Jiang Y, Zhang Y, Lan L, Huang Y, et al. A fiber optoacoustic emitter with controlled ultrasound frequency for cell membrane sonoporation at submillimeter spatial resolution. Photoacoustics 2020;20:100208. [PMID: 33101926 DOI: 10.1016/j.pacs.2020.100208]
- Charbenny S, Huang Z. Brain thermal response to low intensity focused ultrasound at the action potential level and neuron response to various stimulus. Comput Biol Med 2025;197(Pt B):111094. [PMID: 40967140 DOI: 10.1016/j.compbiomed.2025.111094]
- Morales-Hernandez AG, Martinez-Aguilar V, Chavez-Gonzalez TM, Mendez-Avila JC, Frias-Becerril JV, et al. Short-term thermal effect of continuous ultrasound from 3 MHz to 1 and 0.5 W/cm2 applied to gastrocnemius muscle. Diagnostics (Basel) 2023;13(16):2644.[PMID: 37627903 DOI: 10.3390/diagnostics13162644]
- Zhang C, Wu Y, Zhang Q, Zhang M, Zhang D. The impact of ischemic vascular stenosis on LIPU hyperthermia efficacy investigated based on in vivo rabbit limb ischemia model. Ultrasonics 2024;138:107263. [PMID: 38350312 DOI: 10.1016/j.ultras.2024.107263]
- Wu D, Jin X, Wang X, Ma B, Lou C, et al. Engineering temperature-sensitive plateletsomes as a tailored chemotherapy platform in combination with HIFU ablation for cancer treatment. Theranostics 2019;9(13):3966-79. [PMID: 31281525 DOI: 10.7150/thno.32172]
- Huang X, Yuan F, Liang M, Lo HW, Shinohara ML, et al. M-HIFU inhibits tumor growth, suppresses STAT3 activity and enhances tumor specific immunity in a transplant tumor model of prostate cancer. PLoS One 2012;7(7):e41632. [PMID: 22911830 DOI: 10.1371/journal.pone.0041632]
- Nittayacharn P, Abenojar E, Cooley MB, Berg FM, Counil C, et al. Efficient ultrasound-mediated drug delivery to orthotopic liver tumors – Direct comparison of doxorubicin-loaded nanobubbles and microbubbles. J Control Release 2024;367:135-47. [PMID: 38237687 DOI: 10.1016/j.jconrel.2024.01.028]
- Noel RL, Batts AJ, Ji R, Pouliopoulos AN, Bae S, et al. Natural aging and Alzheimer’s disease pathology increase susceptibility to focused ultrasound-induced blood-brain barrier opening. Sci Rep 2023;13(1):6757. [PMID: 37185578 DOI: 10.1038/s41598-023-30466-6]
- Zhang L, Liu X, Gao L, Ji Y, Wang L, et al. Activation of Piezo1 by ultrasonic stimulation and its effect on the permeability of human umbilical vein endothelial cells. Biomed Pharmacother 2020;131:110796. [PMID: 33152952 DOI: 10.1016/j.biopha.2020.110796]
- Liufu C, Li Y, Tu J, Zhang H, Yu J, et al. Echogenic PEGylated PEI-loaded microbubble as efficient gene delivery system. Int J Nanomedicine 2019;14:8923-41. [PMID: 31814720 DOI: 10.2147/IJN.S217338]
- Van Reet J, Tunnell K, Anderson K, Kim HC, Kim E, et al. Evaluation of advective solute infiltration into porous media by pulsed focused ultrasound-induced acoustic streaming effects. Ultrasonography 2024;43(1):35-46. [PMID: 38029736 DOI: 10.14366/usg.23037]
- Yao H, Zhang L, Yan S, He Y, Zhu H, et al. Low-intensity pulsed ultrasound/nanomechanical force generators enhance osteogenesis of BMSCs through microfilaments and TRPM7. J Nanobiotechnology 2022;20(1):378. [PMID: 35964037 DOI: 10.1186/s12951-022-01587-3]
- Aliabouzar M, Lee SJ, Zhou X, Zhang GL, Sarkar K. Effects of scaffold microstructure and low intensity pulsed ultrasound on chondrogenic differentiation of human mesenchymal stem cells. Biotechnol Bioeng 2018;115(2):495-506. [PMID: 29064570 DOI: 10.1002/bit.26480]
- Wang Y, Jiang L, Xu T, Su Z, Guo X, et al. p38 MAPK signaling is a key mediator for low-intensity pulsed ultrasound (LIPUS) in cultured human omental adipose-derived mesenchymal stem cells. Am J Transl Res 2019;11(1):418-29. [PMID: 30787998]
- El-Bialy T, Alhadlaq A, Lam B. Effect of therapeutic ultrasound on human periodontal ligament cells for dental and periodontal tissue engineering. Open Dent J 2012;6:235-9. [PMID: 23308087 DOI: 10.2174/1874210601206010235]
- Gao Q, Walmsley AD, Cooper PR, Scheven BA. Ultrasound stimulation of different dental stem cell populations: role of mitogen-activated protein kinase signaling. J Endod 2016;42(3):425-31. [PMID: 26830427 DOI: 10.1016/j.joen.2015.12.019]
- Liu QW, Huang QM, Wu HY, Zuo GS, Gu HC, et al. Characteristics and therapeutic potential of human amnion-derived stem cells. Int J Mol Sci 2021;22(2):970. [PMID: 33478081 DOI: 10.3390/ijms22020970]
- Yoon JH, Roh EY, Shin S, Jung NH, Song EY, et al. Introducing pulsed low-intensity ultrasound to culturing human umbilical cord-derived mesenchymal stem cells. Biotechnol Lett 2009;31(3):329-35. [PMID: 18985278 DOI: 10.1007/s10529-008-9872-5]
- An Y, Song Y, Wang Z, Wang J, Wu G, et al. Effect of low-intensity pulsed ultrasound on the biological behaviors of bone marrow mesenchymal stem cells on titanium with different surface topographies. Am J Transl Res 2018;10(1):67-76. [PMID: 29422994]
- Wang X, Lin Q, Zhang T, Wang X, Cheng K, et al. Low-intensity pulsed ultrasound promotes chondrogenesis of mesenchymal stem cells via regulation of autophagy. Stem Cell Res Ther 2019;10(1):41. [PMID: 30670079 DOI: 10.1186/s13287-019-1142-z]
- Xia P, Wang X, Qu Y, Lin Q, Cheng K, et al. TGF-β1-induced chondrogenesis of bone marrow mesenchymal stem cells is promoted by low-intensity pulsed ultrasound through the integrin-mTOR signaling pathway. Stem Cell Res Ther 2017;8(1):281. [PMID: 29237506 DOI: 10.1186/s13287-017-0733-9]
- Yao H, Tang L, Wang D, Pang H, Yang K. F-actin microfilaments affect the LIPUS-promoted osteogenic differentiation of BMSCs through TRPM7. Biotechnol J 2024;19(8):e2400310. [PMID: 39212193 DOI: 10.1002/biot.202400310]
- Liang C, Zhang Y, Yan Y, Geng W, Li J, et al. LIPUS promotes osteogenic differentiation of rat BMSCs and osseointegration of dental implants by regulating ITGA11 and focal adhesion pathway. BMC Oral Health 2025;25(1):22. [PMID: 39755586 DOI: 10.1186/s12903-024-05411-2]
- Zhou J, Zhu Y, Ai D, Zhou M, Li H, et al. Low-intensity pulsed ultrasound regulates osteoblast-osteoclast crosstalk via EphrinB2/EphB4 signaling for orthodontic alveolar bone remodeling. Front Bioeng Biotechnol 2023;11:1192720. [PMID: 37425367 DOI: 10.3389/fbioe.2023.1192720]
- Yue Y, Yang X, Wei X, Chen J, Fu N, et al. Osteogenic differentiation of adipose-derived stem cells prompted by low-intensity pulsed ultrasound. Cell Prolif 2013;46(3):320-7. [PMID: 23692090 DOI: 10.1111/cpr.12035]
- Fu N, Yang X, Ba K, Fu Y, Wei X, et al. Low-intensity pulsed ultrasound induced enhanced adipogenesis of adipose-derived stem cells. Cell Prolif 2013;46(3):312-9. [PMID: 23692089 DOI: 10.1111/cpr.12031]
- Kusuyama J, Nakamura T, Ohnishi T, Eiraku N, Noguchi K, et al. Low-intensity pulsed ultrasound (LIPUS) promotes BMP9-induced osteogenesis and suppresses inflammatory responses in human periodontal ligament-derived stem cells. J Orthop Trauma 2017;31(7):S4. [PMID: 28632668 DOI: 10.1097/01.bot.0000520897.92470.70]
- Chen Y, Yang H, Wang Z, Zhu R, Cheng L, et al. Low-intensity pulsed ultrasound promotes mesenchymal stem cell transplantation-based articular cartilage regeneration via inhibiting the TNF signaling pathway. Stem Cell Res Ther 2023;14(1):93. [PMID: 37069673 DOI: 10.1186/s13287-023-03296-6]
- Kang PL, Huang HH, Chen T, Ju KC, Kuo SM. Angiogenesis-promoting effect of LIPUS on hADSCs and HUVECs cultured on collagen/hyaluronan scaffolds. Mater Sci Eng C Mater Biol Appl 2019;102:22-33. [PMID: 31146993 DOI: 10.1016/j.msec.2019.04.045]
- El-Bialy T, Alhadlaq A, Wong B, Kucharski C. Ultrasound effect on neural differentiation of gingival stem/progenitor cells. Ann Biomed Eng 2014;42(7):1406-12. [PMID: 24752635 DOI: 10.1007/s10439-014-1013-9]
- Li F, Liu Y, Cai Y, Li X, Bai M, et al. Ultrasound irradiation combined with hepatocyte growth factor accelerate the hepatic differentiation of human bone marrow mesenchymal stem cells. Ultrasound Med Biol 2018;44(5):1044-52. [PMID: 29499919 DOI: 10.1016/j.ultrasmedbio.2018.01.005]
- Wang Y, Li J, Zhou J, Qiu Y, Song J. Low-intensity pulsed ultrasound enhances bone marrow-derived stem cells-based periodontal regenerative therapies. Ultrasonics 2022;121:106678. [PMID: 35051693 DOI: 10.1016/j.ultras.2021.106678]
- Wang Y, Li J, Qiu Y, Hu B, Chen J, et al. Low-intensity pulsed ultrasound promotes periodontal ligament stem cell migration through TWIST1-mediated SDF-1 expression. Int J Mol Med 2018;42(1):322-30. [PMID: 29620151 DOI: 10.3892/ijmm.2018.3592]
- Ling L, Feng X, Wei T, Wang Y, Wang Y, et al. Effects of low-intensity pulsed ultrasound (LIPUS)-pretreated human amnion-derived mesenchymal stem cell (hAD-MSC) transplantation on primary ovarian insufficiency in rats. Stem Cell Res Ther 2017;8(1):283. [PMID: 29258619 DOI: 10.1186/s13287-017-0739-3]