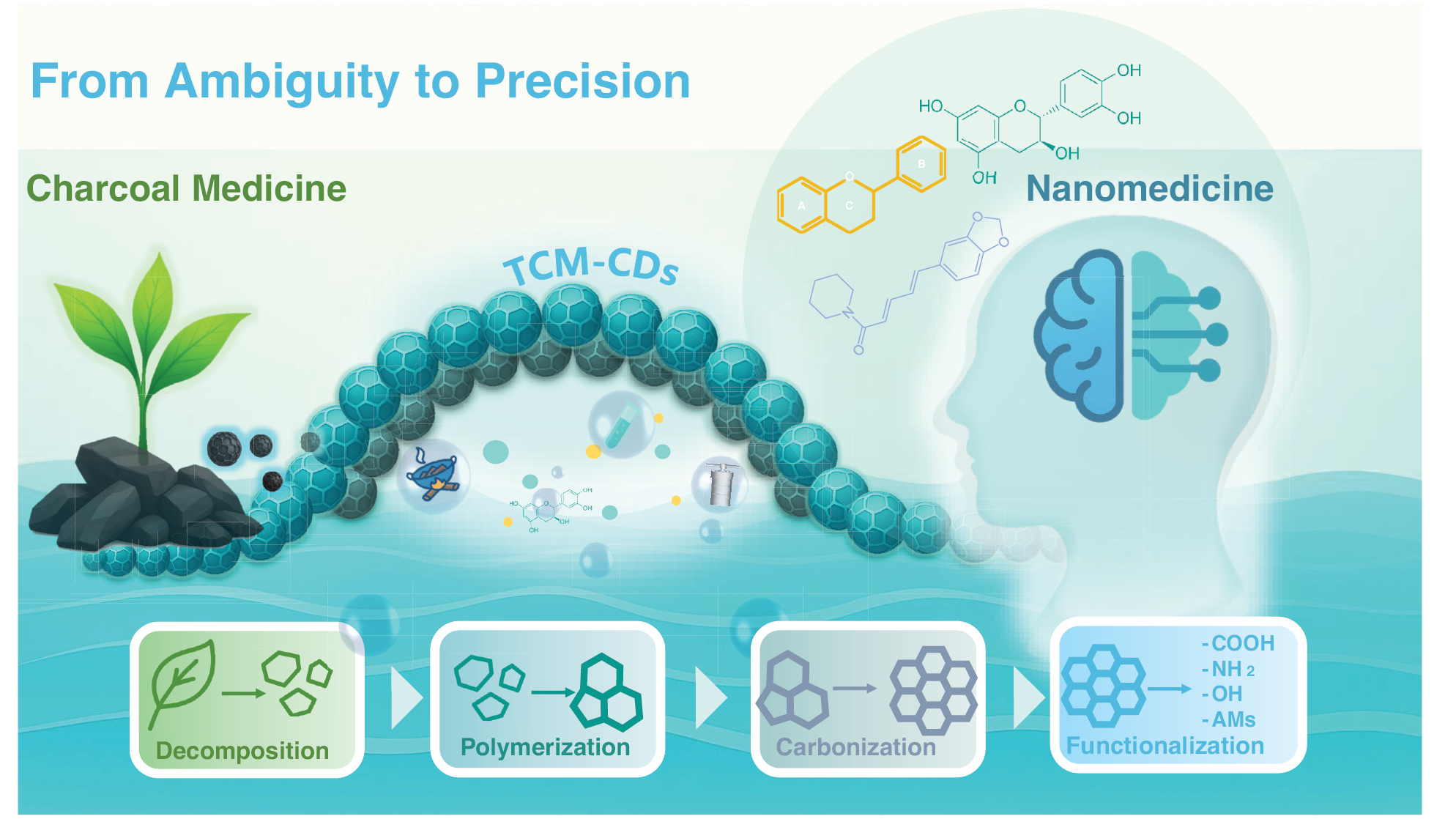
From Ambiguity to Precision: Carbon Dots Bridge Traditional Charcoal Medicine and Modern Nanomedicine
1Nanjing Hospital of Chinese Medicine, Nanjing University of Chinese Medicine, Nanjing 210000, China
2State Key Laboratory of Tree Genetics and Breeding, College of Life Sciences, Nanjing Forestry University, Nanjing 210037, China
3School of Chinese Medicine, The University of Hong Kong, Pokfulam, Hong Kong, China
aThese authors contributed equally to this work and share first authorship.
*Correspondence to: Yibin Feng, School of Chinese Medicine, The University of Hong Kong, Pokfulam, Hong Kong, China. E-mail: yfeng@hku.hk; Kang Ding, Nanjing Hospital of Chinese Medicine, Nanjing University of Chinese Medicine, Nanjing 210000, China. E-mail: fsyy00237@njucm.edu.cn; Tao Luo, Nanjing Hospital of Chinese Medicine, Nanjing University of Chinese Medicine, Nanjing 210000, China; School of Chinese Medicine, The University of Hong Kong, Pokfulam, Hong Kong, China. E-mail: luotao@hnu.edu.cn
Received: November 23 2025; Revised: December 23 2025; Accepted: January 12 2026; Published Online: January 28 2026
Cite this paper:
Zhang A, Sun Y, Zhang S et al. From Ambiguity to Precision: Carbon Dots Bridge Traditional Charcoal Medicine and Modern Nanomedicine. BIO Integration 2026; 7: 1–7.
DOI: 10.15212/bioi-2025-0212. Available at: https://bio-integration.org/
Download citation
© 2026 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Traditional Chinese Medicine (TCM) provides rich pharmacological resources, among which charcoal medicine is a distinctive product of carbonization techniques. Renowned for its anti-inflammatory, hemostatic, and antidiarrheal properties, charcoal medicine has faced developmental limitations because of its unclear pharmacological mechanisms. The emergence of carbon dots (CDs), a nanomaterial with excellent biocompatibility and biochemical versatility, has provided novel perspectives in charcoal medicine. TCM-CDs, derived from TCM precursors such as charcoal medicine, serve as a bridge between traditional practices and modern nanomedicine. Combining high-temperature processing and advanced synthesis enables TCM-CDs to retain therapeutic properties while gaining nanoscale features. These molecules have shown potential in the treatment of neurological disorders, inflammation, and metabolic diseases, and in applications such as biomedical imaging and diagnostics. However, challenges such as incomplete structural characterization, inconsistent synthesis, and limited clinical validation remain. Future research should use advanced analytical methods, artificial intelligence (AI), and standardized protocols to achieve scalable production and quality control. Guided by TCM’s holistic philosophy, multifunctional TCM-CDs have potential to enhance therapeutic effects. By bridging traditional practices and modern nanomedicine, TCM-CDs highlight the modernization of charcoal medicine, and may open avenues for innovation and advancement in TCM research.
Keywords
Carbon dots, charcoal medicine, nanomedicine, pharmacological mechanisms, traditional chinese medicine.
Traditional carbonization techniques have been used in Traditional Chinese medicine (TCM) processing for millennia, yet their underlying material basis has remained unclear, thus hindering the precise elucidation of their mechanisms of action. The emergence of TCM carbon dots (CDs) has enabled the transformation of long-standing, experience-based charcoal medicine into quantifiable, characterizable, and designable nanoscale systems, which offer new possibilities for defining the material basis of TCM. This perspective traces the historical roots of charcoal medicine and discusses TCM-CD formation mechanisms, synthesis strategies, and applications to reveal how these nanostructures can facilitate the transition of charcoal medicine from ambiguity to precision (Figure 1).
Figure 1 By bridging ancient charcoal medicine with modern nanomedicine, carbon dots offer a scientific foundation for TCM modernization and mechanistic studies.
The ambiguity of charcoal medicine: history and limitations
TCM provides an enormous yet still partially untapped reservoir of pharmacological resources. Originating from natural sources, TCM encompasses a wide diversity of substances, including medicinal plants (roots, rhizomes, leaves, flowers, pollen, fruits, peels, and seeds), minerals, and animal-derived materials [1–3]. Depending on the physicochemical properties of each medicine and its intended therapeutic purpose, TCM formulas undergo various processing techniques, such as soaking, steaming, and carbonization, to modify bioactivity and clinical effects [4, 5]. Charcoal medicine, produced through controlled carbonization, is a distinctive outcome of such processing methods. The earliest record of this technique dates back to the Fifty-Two Prescriptions unearthed from the Mawangdui tombs, which documented 31 charcoal-based preparations prescribed for conditions including urinary retention, wounds, abscesses, and epilepsy [6]. Over centuries of practice, more than 200 TCM materials have undergone carbonization, and approximately 70 are currently in clinical use. Modern pharmacological investigations have demonstrated that carbonization alters the chemical composition of TCM, thus leading to functional transformations and expanded therapeutic potential. For example, whereas fresh ginger exhibits anticoagulant activity, ginger charcoal displays an opposite hemostatic effect [7]. Similarly, rhubarb, which is commonly prescribed for fever and constipation, acquires new functions associated with hemostasis and wound healing after carbonization through stir-frying [8].
Although charcoal medicine has been extensively applied in clinical practice within TCM, several factors continue to hinder its modernization. First, the evaluation criteria for charcoal medicine have remained ambiguous for millennia. Quality has traditionally been judged by external appearance, color, and texture, through an empirical approach that lacks standardized preparation and quality control protocols [9]. Second, historical records often provide inconsistent or even contradictory descriptions. Some texts define the desired endpoint as complete blackening, whereas others emphasize moderate carbonization, warning that “overheating produces dead ash with no medicinal value.” Third, the pharmacological efficacy and material basis of charcoal medicine remain poorly understood [10]. This reliance on experiential knowledge and qualitative descriptions has substantially hindered systematic exploration of its bioactivity and industrial scalability. To overcome these limitations, modern preparation and characterization technologies are urgently needed to clarify its chemical and biological foundations. Against this backdrop, the emergence of CDs offers a novel conceptual and technological framework for reinterpreting and advancing research on charcoal medicine.
In summary, despite its long history and clinical relevance, charcoal medicine remains constrained by ambiguous quality standards, poorly defined material compositions, and an unclear mechanistic basis. These limitations underscore the need for modern analytical and technological frameworks to provide a foundation for introducing CDs as a means of reinterpreting and advancing charcoal medicine research.
The rise of carbon dots: rapid development and challenges
CDs are a class of zero-dimensional carbon-based nanomaterials with particle sizes smaller than 10 nm. CDs were first reported by Xu et al. [11] in 2004 during the arc-discharge synthesis of single-walled carbon nanotubes. They typically consist of an amorphous carbon core composed of mixed sp2/sp3 hybridized carbon atoms, surrounded by a shell enriched with diverse functional groups or polymer chains [12]. According to differences in their carbon core structure, surface chemistry, and physicochemical properties, CDs can be further categorized into carbon quantum dots, graphene quantum dots, carbon nanodots, and carbonized polymer dots [13, 14]. CDs exhibit unique optical properties, as well as excellent bioimaging and biosensing capabilities [15, 16]. Their size-dependent drug delivery and targeting potential further position them as promising candidates for precision therapeutics and real-time diagnostics.
However, the structural characterization of this emerging class of nanomaterials remains incomplete; therefore, the relationships between CDs’ complex compositions and biological functions remain poorly defined. The diversity of synthesis methods and the lack of standardized fabrication protocols further contribute to variability in performance. Moreover, early studies frequently used chemical organic precursors such as citric acid, methanol, and aniline [17–20]. Because the related reactions can generate toxic byproducts during synthesis, concerns regarding sustainability and biosafety ultimately limited their biomedical application. In recent years, the pursuit of environmentally friendly and renewable alternatives has driven rapid progress in the development of green CDs [21, 22]. Notably, TCM has emerged as a distinctive natural carbon source that combines structural diversity with intrinsic pharmacological potential.
The rapid expansion of CD research highlights substantial biomedical promise, as well as persistent challenges in structural definition, standardization, and biosafety. These considerations have stimulated growing interest in green and biologically derived precursors, and provided a clear rationale for the development of TCM-derived CDs.
The emergence of TCM-CDs: a promising paradigm for nanomedicine
Charcoal medicine and CDs share comparable carbonization temperatures (approximately 250°C) and a carbon-centered composition, and both retain the pharmacological activities of their source materials [23]. Consequently, CDs are considered active nanostructures that form spontaneously during the high-temperature preparation of charcoal medicine and therefore represent a nanoscale extension of TCM [24]. Since their first synthesis from medicinal precursors in 2013, TCM-CDs have gained attention for their low toxicity, high biocompatibility, and pharmacological properties inherited from their precursors [25]. This research trend has continued to grow over the past decade. To date, 107 TCM precursors have been converted into TCM-CDs. Herbs account for the majority and are followed by animal-derived sources, whereas minerals appear primarily as doped elements within the structures.
Synthesis methods
To understand the formation mechanisms of TCM-CDs, the preparation conditions of traditional charcoal medicine versus modern CDs must be compared. Traditionally, charcoal medicine is produced through the decomposition of herbal materials under high temperature and limited oxygen. This process generally involves two main approaches: stir-frying carbonization and calcination carbonization. The former is commonly applied to rhizomatous herbs such as Rheum palmatum and Zingiber officinale, whereas the latter is used for loose or lightweight materials and solid drugs unsuitable for stir-frying, such as Nelumbinis Folium and Crinis.
Modern TCM-CDs synthesis methods include high-temperature pyrolysis, hydrothermal, solvothermal, and microwave techniques. All provide high-temperature environments conducive to carbonization, but they differ in their energy input and reaction control; therefore, they are suitable for various synthesis scenarios. Because high-temperature pyrolysis applies stronger heat for rapid decomposition, it is suitable for thermally stable or structurally dense precursors [26, 27]. The hydrothermal method uses water as the solvent for mild, prolonged carbonization, thus decreasing toxicity and improving uniformity [28, 29]. The solvothermal method uses various non-aqueous solvents to enhance solubility and control surface chemistry; however, concerns regarding solvent toxicity, cost, and post-treatment may restrict its large-scale biomedical application. The microwave approach transfers energy efficiently through electromagnetic waves, thereby completing carbonization within minutes and markedly improving synthesis efficiency. Nevertheless, challenges in reaction homogeneity and scale-up remain to be addressed [30].
Formation mechanisms
The carbonization pathway for preparing CDs from conventional small-molecule precursors is relatively simple and compositionally uniform, and generally involves dehydration and condensation followed by carbon core formation. However, the incorporation of heteroatoms and the construction of surface functional groups require further manual adjustment during synthesis. In contrast, TCM-CDs benefit from the unique compositions of their precursors, including polysaccharides, flavonoids, alkaloids, and various natural bioactive molecules. Because of the pronounced differences in thermal stability, structural heterogeneity, and reactivity among these molecules, the formation mechanisms during pyrolysis follow a stepwise reconstruction process.
After the evaporation of water from TCM, the first stage predominantly involves the thermal decomposition of polysaccharides, alkaloids, and flavonoids at relatively low temperatures (approximately 100–200°C) [31]. Polysaccharides undergo hydrolysis into monosaccharides, which are further transformed into furan derivatives. Alkaloids, depending on their varying sensitivity to high temperatures, may partially decompose, and some retain aromatic heterocyclic structures. Flavonoids (C6-C3-C6) undergo demethylation, thereby preserving the active phenolic hydroxyl groups essential for pharmacological activity [32]. Subsequently, during the polymerization stage (approximately 200–300°C), covalent cross-linking between molecules, through aldol condensation and etherification reactions, leads to the formation of polycyclic aromatic frameworks. These frameworks constitute the core TCM-CD structure. In the carbonization stage (above 300°C), elevated temperatures promote structural ordering within the carbon core and drive the aggregation of smaller carbon domains into larger ones. Amorphous carbon gradually restructures into sp2/sp3 hybridized graphite-like domains, thereby further optimizing the internal structure [33]. Meanwhile, thermally stable natural active molecules such as saponins and polyphenols remain attached to the surfaces or embedded within the CDs through physical adsorption, π-π stacking, or metal coordination; consequently, their intrinsic antioxidant and antimicrobial activities are preserved [24, 34]. In the surface functionalization stage, components that are incompletely deoxygenated or deaminated during carbonization are anchored to the defect sites on the surfaces of TCM-CDs and form stable surface functional groups [12]. These functional groups enhance the water solubility of TCM-CDs and contribute to their excellent pharmacological activity.
Across the four stages of decomposition, polymerization, carbonization, and surface functionalization, TCM-CDs preserve a portion of TCM biological activity while acquiring new physicochemical characteristics through structural reconstruction [35]. This evolution enhances their potential biomedical applications.
Applications
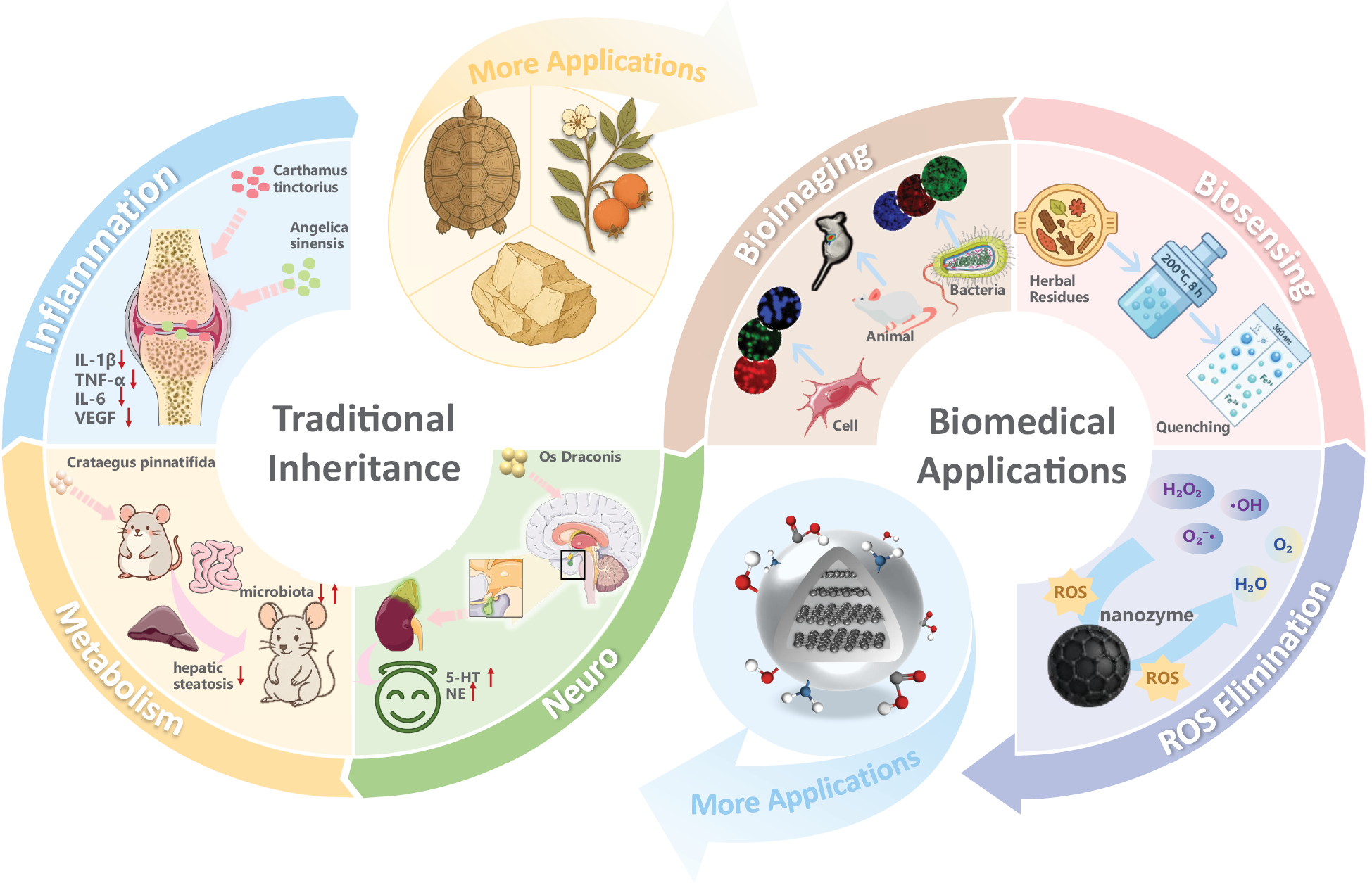
Overall, the therapeutic value of TCM-CDs involves two complementary dimensions. First, they inherit the disease-targeting pharmacological activities of their herbal precursors. Second, their unique optical features and nanozyme-like properties further enable modern biomedical applications that traditional TCM alone cannot achieve (Figure 2).
Figure 2 Applications of TCM-CDs.
Guided by TCM theory, researchers have drawn inspiration from traditional medical practices to design diverse TCM-CDs with disease-specific functions (Figure 1). Inspired by the calming and sedative effects of Os Draconis, Chen et al. [36] have developed TCM-CDs capable of modulating 5-HT, NE, and the HPA axis, thereby restoring neuroendocrine balance in neurological disorders. In inflammatory diseases, Carthamus tinctorius and Angelica sinensis are a classic herbal pair known to promote blood circulation and remove stasis Qiang et al. [37] using these herbs as carbon sources, have synthesized TCM-CDs that suppress pro-inflammatory cytokines such as IL-1β and TNF-α, and downregulate the NF-κB and MAPK pathways. These findings have provided molecular evidence that TCM-CDs can retain the anti-inflammatory effects of their parent herbs. Moreover, in the field of metabolic disorders, Lin et al. [38] have prepared Crataegus pinnatifida-derived TCM-CDs that markedly ameliorate insulin resistance, prevent hepatic steatosis, and modulate the gut microbiota in mice fed a high-fat diet. The preparation therefore effectively inherited the traditional effects of Crataegus in digesting stagnation and lowering lipids.
TCM-CDs, given their superior optical properties, abundant surface functional groups, and nanozyme-like activity, have applications extending beyond traditional pharmacological uses (Figure 1). For bioimaging applications, Yuan et al. [39] have prepared green TCM-CDs from Poria cocos that exhibit multicolor fluorescence and enable clear cytoplasmic imaging under different excitation wavelengths. Their low toxicity, high stability, and low cost make them promising materials for future biomedical imaging. Building on their biosensing responsiveness, Zhang et al. [40] have synthesized boron-doped CDs derived from herbal residues. The rich surface functional groups endow the CDs with strong coordination capacity and high selectivity toward Fe3+, thus leading to fluorescence quenching. Beyond bioimaging and biosensing, TCM-CDs have also demonstrated remarkable antioxidant and reactive oxygen species (ROS) scavenging abilities. Various examples, such as Gardenia fructus and Curcuma zedoaria–derived TCM-CDs, have shown excellent free-radical elimination both in vitro and in vivo [41, 42]. Mechanistically, their activity is likely to be associated with the carbon-based nanozyme behavior of CDs. Gao et al. [43] have designed CDs with nanozyme activity by introducing surface –OH, –COOH, and C=O groups onto graphite-derived carbon frameworks. These engineered CDs exhibit SOD-like catalytic behavior and mitochondrial targeting ability, and effectively scavenge reactive oxygen species and repair oxidative damage in cells. Notably, their surface chemistry and intrinsic antioxidant activity closely resemble those of TCM-CDs, thereby underscoring the similar mechanistic basis between systems. Designing TCM-CDs with enhanced antioxidant and targeted nanozyme activity may therefore provide a key direction for future research and a foundation for their precise functional development.
The evolution toward precision: future directions of TCM-CDs
The continued advancement of TCM-CDs will rely on synergistic progress in technological innovation, safety validation, mechanistic elucidation, system standardization, and integration with TCM theory. Together, these efforts have potential to transform the field of TCM-CDs from empirical exploration into a precise and controllable form of nanomedicine.
Technological innovation
With rapid advancements, the integration of artificial intelligence (AI) into the full workflow of TCM-CD preparation is becoming increasingly feasible. AI may aid in precursor design by mining the TCM literature to extract information on herb properties and carbonization practices, while also enabling real-time optimization of synthesis parameters to improve yield and batch consistency. By linking physicochemical characteristics with pharmacological outcomes, AI has potential to facilitate data-driven structure–function analysis and support the development of more systematic and automated research pipelines.
Safety and clinical translation
To advance the safety and clinical translation of TCM-CDs, strict standardization of quality control will be essential, including parameters such as particle size, surface chemistry, and purity. Variations in the quality of TCM precursors caused by geography, species, and harvest period must also be addressed. Scalable and reproducible production processes, such as SOPs, validated purification methods, and large-scale manufacturing techniques, must be established. Moreover, challenges including production costs, equipment requirements, and batch-to-batch consistency remain to be resolved.
Comprehensive toxicological studies should be conducted, including acute toxicity, chronic toxicity, immunotoxicity, genotoxicity, and biodistribution assessments of representative batches. Stability and shelf-life investigations, and the development of reference standards, will also be crucial. To strengthen quality control, chemical and biological multi-marker fingerprinting methods should be used for batch release and consistency testing. Early engagement with regulatory authorities to clarify non-clinical research requirements and clinical trial designs will be critical. In addition, multidisciplinary teams of experts in natural product chemistry, nanomaterials, analytical techniques, toxicology, and regulatory affairs should be assembled to ensure the safety, efficacy, and compliance of TCM-CDs. Key challenges such as toxicity, safety, reproducibility, and scalable production must be addressed to facilitate the clinical and commercial application of TCM-CDs.
Mechanistic elucidation
The structural complexity of TCM-CDs is closely associated with their pharmacological activity and optical properties. Subtle variations in synthesis conditions, such as temperature or reaction time, can markedly alter particle size, heteroatom content, and surface functional groups. However, the conventional characterization tools currently applied to TCM-CDs (e.g., TEM, AFM, XPS, and FT-IR) lack the resolution to capture these fine structural differences. To better understand the relationships between structure and function, advanced techniques such as HPLC–MS and ultra-high field NMR are necessary. For example, high-resolution mass spectrometry (e.g., HPLC-MS) could be used to identify residual or reconstructed small-molecule species and heteroatom-containing fragments formed during charcoal processing, whereas in situ spectroscopic techniques might help track the evolution of functional groups and carbon domains in real time.
System standardization
Variations in growth regions, soil composition, and climate conditions lead to notable differences in TCM precursors. Accurate evaluation of precursor quality is therefore essential for reproducible downstream synthesis. In the preparation of traditional charcoal medicine, core parameters such as temperature and duration have long been overlooked or have been vaguely described, although these conditions strongly influence the physicochemical features and therapeutic efficacy of the final product. Standardized synthesis protocols developed in CDs research have the potential to inform the modernization of charcoal medicine processing by linking processing parameters with material properties and therapeutic effects.
Theoretical integration
Holism, a foundational principle of TCM, governs not only the dynamic interactions between humans and nature or among internal organ systems but also the underlying rationale for herbal combinations. In clinical practice, single-drug prescriptions are rarely used; instead, therapeutic effects are optimized through complementary herbal pairings. Guided by this theory, the interplay among various TCM-CDs and their formulations, according to the classical TCM principle of Jun–Chen–Zuo–Shi (sovereign–minister–assistant–courier), may offer a new paradigm for understanding the material basis of TCM. Such an approach might also enable new directions in the design and application of multifunctional TCM-CDs.
Conclusion
From traditional charcoal medicine to structurally characterizable nanomaterials, the development of TCM-CDs is a key step in bridging traditional practices with modern nanomedicine. This transition reflects a shift from empirical processing toward mechanism-informed and designable nanoscale systems.
In the future, advances in analytical techniques, standardization strategies, and data-driven approaches may further enable precise structure–function understanding and clinical translation of TCM-CDs. Overall, TCM-CDs offer a promising framework for modernizing and systematizing charcoal medicine, by providing a material and conceptual basis for the quantifiable understanding and nanoscale innovation of TCM.
Abbreviations
| TCM | Traditional Chinese Medicine |
| CDs | Carbon Dots |
| TCM-CDs | Traditional Chinese Medicine–Derived Carbon Dots |
| AI | Artificial Intelligence. |
Credit author statement
Ao Zhang and Yichen Sun contributed to writing—original draft and visualization; Shijie Zhang engaged in writing—review & editing; Yibin Feng, Kang Ding, and Tao Luo led conceptualization, secured funding acquisition, and provided supervision. All authors have read and agreed to the published version of the manuscript.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (82400186), Natural Science Foundation of Jiangsu Province (BK20240269), Jiangsu Province Young Scientific and Technological Talents Promotion Programme (JSYJ-2025–728), and Feihong Program of Nanjing Hospital of Chinese Medicine (FHJH202402).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Wang DQH, Carey MC. Therapeutic uses of animal biles in traditional Chinese medicine: an ethnopharmacological, biophysical chemical and medicinal review. World J Gastroenterol 2014;20(29):9952-75. [PMID: 25110425 DOI: 10.3748/wjg.v20.i29.9952]
- Shan F, Li L, Bi Y, Wang W, Amu G, et al. Exotic medicinal plants from the Silk Road promote the diversification of traditional Chinese medicines. Pharmacol Res 2024;205:107243. [PMID: 38815881 DOI: 10.1016/j.phrs.2024.107243]
- Liu Y, Li X, Chen C, Leng A, Qu J. Effect of mineral excipients on processing traditional Chinese medicines: an insight into the components, pharmacodynamics and mechanism. Chin Med 2021;16(1):143. [PMID: 34952619 DOI: 10.1186/s13020-021-00554-8]
- Zhai C, Zhao J, Chittiboyina AG, Meng Y, Wang M, et al. Newly generated atractylon derivatives in processed rhizomes of Atractylodes macrocephala Koidz. Molecules 2020;25(24):5904. [PMID: 33322214 DOI: 10.3390/molecules25245904]
- Zhang W, Zhang C, Cao L, Liang F, Xie W, et al. Application of digital-intelligence technology in the processing of Chinese materia medica. Front Pharmacol 2023;14:1208055. [PMID: 37693890 DOI: 10.3389/fphar.2023.1208055]
- Zhao Y, Li Y, Li D, Yuan H, Shen C. Eco-friendly synthesized carbon dots from Chinese herbal medicine: a review. Int J Nanomedicine 2025;20:3045-65. [PMID: 40098722 DOI: 10.2147/IJN.S497892]
- Zhang C, Rao A, Chen C, Li Y, Tan X, et al. Pharmacological activity and clinical application analysis of traditional Chinese medicine ginger from the perspective of one source and multiple substances. Chin Med 2024;19(1):97. [PMID: 38997763 DOI: 10.1186/s13020-024-00969-z]
- Wang S, Zhang Y, Shi Y, He Q, Tan Q, et al. Rhubarb charcoal-crosslinked chitosan/silk fibroin sponge scaffold with efficient hemostasis, inflammation, and angiogenesis for promoting diabetic wound healing. Int J Biol Macromol 2023;253(Pt 2):126796. [PMID: 37689294 DOI: 10.1016/j.ijbiomac.2023.126796]
- Chen Z, Ye SY, Yang Y, Li ZY. A review on charred traditional Chinese herbs: carbonization to yield a haemostatic effect. Pharm Biol 2019;57(1):498-506. [PMID: 31401925 DOI: 10.1080/13880209.2019.1645700]
- Guo K, Cao YY, Qian LC, Daniels MJ, Tian Y, et al. Dihydroartemisinin induces ferroptosis in pancreatic cancer cells by the regulation of survival prediction-related genes. Tradit Med Res. 2023;8(12):67. [DOI: 10.53388/TMR20230618001]
- Xu X, Ray R, Gu Y, Ploehn HJ, Gearheart L, et al. Electrophoretic analysis and purification of fluorescent single-walled carbon nanotube fragments. J Am Chem Soc 2004;126(40):12736-7. [PMID: 15469243 DOI: 10.1021/ja040082h]
- Liu Y, Zhang L, Cai H, Qu X, Chang J, et al. Biomass-derived carbon dots with pharmacological activity for biomedicine: recent advances and future perspectives. Sci Bull (Beijing) 2024;69(19):3127-49. [PMID: 39183109 DOI: 10.1016/j.scib.2024.08.011]
- Giordano MG, Seganti G, Bartoli M, Tagliaferro A. An overview on carbon quantum dots optical and chemical features. Molecules 2023;28(6):2772. [DOI: 10.3390/molecules28062772]
- Guo F, Li Q, Zhang X, Liu Y, Jiang J, et al. Applications of carbon dots for the treatment of Alzheimer’s disease. Int J Nanomedicine 2022;17:6621-38. [PMID: 36582459 DOI: 10.2147/IJN.S388030]
- Ansari L, Hallaj S, Hallaj T, Amjadi M. Doped-carbon dots: recent advances in their biosensing, bioimaging and therapy applications. Colloids Surf B Biointerfaces 2021;203:111743. [PMID: 33872828 DOI: 10.1016/j.colsurfb.2021.111743]
- Molaei MJ. A review on nanostructured carbon quantum dots and their applications in biotechnology, sensors, and chemiluminescence. Talanta 2019;196:456-78. [PMID: 30683392 DOI: 10.1016/j.talanta.2018.12.042]
- Li X, Wang H, Shimizu Y, Pyatenko A, Kawaguchi K, et al. Preparation of carbon quantum dots with tunable photoluminescence by rapid laser passivation in ordinary organic solvents. Chem Commun 2011;47(3):932-4. [DOI: 10.1039/C0CC03552A]
- Shan D, Hsieh JT, Bai X, Yang J. Citrate-based fluorescent biomaterials. Adv Healthc Mater 2018;7(18):e1800532. [PMID: 30047618 DOI: 10.1002/adhm.201800532]
- Yu H, Li X, Zeng X, Lu Y. Preparation of carbon dots by non-focusing pulsed laser irradiation in toluene. Chem Commun 2015;52(4):819-22. [DOI: 10.1039/C5CC08384B]
- Wang B, Song H, Tang Z, Yang B, Lu S. Ethanol-derived white emissive carbon dots: the formation process investigation and multi-color/white LEDs preparation. Nano Res 2022;15(2):942-9. [DOI: 10.1007/s12274-021-3579-5]
- Bosu S, Rajamohan N, Sagadevan S, Raut N. Biomass derived green carbon dots for sensing applications of effective detection of metallic contaminants in the environment. Chemosphere 2023;345:140471. [PMID: 37871875 DOI: 10.1016/j.chemosphere.2023.140471]
- Fang M, Wang B, Qu X, Li S, Huang J, et al. State-of-the-art of biomass-derived carbon dots: preparation, properties, and applications. Chin Chem Lett 2024;35(1):108423. [DOI: 10.1016/j.cclet.2023.108423]
- Tian S, Hu Z, Zhang W. Modernization of charcoal drugs: integrating research paradigms of carbon dots to gain new perspectives. Chin Herb Med 2025;17(2):292-5. [PMID: 40256721 DOI: 10.1016/j.chmed.2025.01.004]
- Li M, Li H, Zeng K, Lu Z. From ancient charred herbs to modern nanomedicine: carbon dots as key bioactive components in carbonized traditional Chinese medicine. World J Integr Trad West Med 2025;11(3):145-54. [DOI: 10.70976/j.2096-0964.wjim-2025-0015]
- Wu D, Huang X, Deng X, Wang K, Liu Q. Preparation of photoluminescent carbon nanodots by traditional Chinese medicine and application as a probe for Hg2+. Anal Methods 2013;5(12):3023-7. [DOI: 10.1039/C3AY40337H]
- Zeng M, Wang Y, Liu M, Wei Y, Wen J, et al. Potential efficacy of herbal medicine-derived carbon dots in the treatment of diseases: from mechanism to clinic. Int J Nanomedicine 2023;18:6503-25. [PMID: 37965279 DOI: 10.2147/IJN.S431061]
- Ngoc LTN, Moon JY, Lee YC. Plant extract-derived carbon dots as cosmetic ingredients. Nanomaterials 2023;13(19):2654. [DOI: 10.3390/nano13192654]
- Jung H, Sapner VS, Adhikari A, Sathe BR, Patel R. Recent progress on carbon quantum dots based photocatalysis. Front Chem 2022;10:881495. [PMID: 35548671 DOI: 10.3389/fchem.2022.881495]
- Tejwan N, Saha SK, Das J. Multifaceted applications of green carbon dots synthesized from renewable sources. Adv Colloid Interface Sci 2020;275:102046. [PMID: 31757388 DOI: 10.1016/j.cis.2019.102046]
- Jaiswal A, Ghsoh SS, Chattopadhyay A. Quantum dot impregnated-chitosan film for heavy metal ion sensing and removal. Langmuir 2012;28(44):15687-96. [PMID: 23020689 DOI: 10.1021/la3027573]
- Zhang J, Zou L, Li Q, Wu H, Sun Z, et al. Carbon dots derived from traditional Chinese medicines with bioactivities: a rising star in clinical treatment. ACS Appl Bio Mater 2023;6(10):3984-4001. [PMID: 37707491 DOI: 10.1021/acsabm.3c00462]
- Zhang X, Jiang M, Niu N, Chen Z, Li S, et al. Natural-product-derived carbon dots: from natural products to functional materials. ChemSusChem 2018;11(1):11-24. [PMID: 29072348 DOI: 10.1002/cssc.201701847]
- Zhang M, Cheng J, Hu J, Luo J, Zhang Y, et al. Green Phellodendri Chinensis Cortex-based carbon dots for ameliorating imiquimod-induced psoriasis-like inflammation in mice. J Nanobiotechnology 2021;19(1):105. [PMID: 33858431 DOI: 10.1186/s12951-021-00847-y]
- Yang M, Deng Z, Zhu Y, Xu C, Ding C, et al. Advancements in herbal medicine-based nanozymes for biomedical applications. Chin Med J (Engl) 2025;138(9):1037-49. [PMID: 40169370 DOI: 10.1097/CM9.0000000000003584]
- Singh H, Razzaghi M, Ghorbanpoor H, Ebrahimi A, Avci H, et al. Carbon dots in drug delivery and therapeutic applications. Adv Drug Deliv Rev 2025;224:115644. [PMID: 40581049 DOI: 10.1016/j.addr.2025.115644]
- Chen Y, Xiong W, Zhang Y, Bai X, Cheng G, et al. Carbon dots derived from Os draconis and their anxiolytic effect. Int J Nanomedicine 2022;17:4975-88. [PMID: 36275482 DOI: 10.2147/IJN.S382112]
- Qiang R, Huang H, Chen J, Shi X, Fan Z, et al. Carbon quantum dots derived from herbal medicine as therapeutic nanoagents for rheumatoid arthritis with ultrahigh lubrication and anti-inflammation. ACS Appl Mater Interfaces 2023;15(32):38653-64. [PMID: 37535012 DOI: 10.1021/acsami.3c06188]
- Lin S, Zheng YJ, Xu YZ, Zhou Y, He X, et al. Hawthorn carbon dots: a novel therapeutic agent for modulating body weight and hepatic lipid profiles in high-fat diet-fed mice. Nanoscale 2025;17(5):2668-81. [PMID: 39820263 DOI: 10.1039/d4nr04486j]
- Yuan S, Luo Y, Jiang Y, Xu W, Cheng B, et al. Poria cocos-derived carbon dots for cellular imaging, free radical scavenging and pH sensing. Diam Relat Mater 2023;137:110121. [DOI: 10.1016/j.diamond.2023.110121]
- Zhang L, Luo W, Chen Y, Zheng J, Cao L, et al. Green synthesis of boron-doped carbon dots from Chinese herbal residues for Fe3+ sensing, anti-counterfeiting, and photodegradation applications. J Clean Prod 2023;422:138577. [DOI: 10.1016/j.jclepro.2023.138577]
- Chen BL, Zang XY, Mo JR, Zhang RY, Wang H, et al. Preparation, characterization, and protective effects of Gardenia fructus carbon dots against oxidative damage induced by LPS in IPEC-J2 cells. Front Cell Infect Microbiol 2024;14:1423760. [PMID: 39744157 DOI: 10.3389/fcimb.2024.1423760]
- Zhang Y, Li P, Yan H, Guo Q, Xu Q, et al. Green synthesis and multifunctional applications of nitrogen-doped carbon quantum dots via one-step hydrothermal carbonization of Curcuma zedoaria. Anal Bioanal Chem 2023;415(10):1917-31. [PMID: 36864311 DOI: 10.1007/s00216-023-04603-z]
- Gao W, He J, Chen L, Meng X, Ma Y, et al. Deciphering the catalytic mechanism of superoxide dismutase activity of carbon dot nanozyme. Nat Commun 2023;14(1):160. [PMID: 36631476 DOI: 10.1038/s41467-023-35828-2]