Digestive System Toxicities of Immune Checkpoint Inhibitors: Mechanisms and Clinical Implications
1Department of Oncology, Zhujiang Hospital, Southern Medical University, Guangzhou 510282, Guangdong, China
aJoint Authors. These authors have contributed equally to this work and share first authorship.
*Correspondence to: Dr. Peng Luo, Department of Oncology, Zhujiang Hospital, Southern Medical University, Guangzhou 510282, Guangdong, China. Tel: +86-18588447321. E-mail: luopeng@smu.edu.cn
Received: September 21 2025; Revised: November 25 2025; Accepted: December 14 2025; Published Online: January 8 2026
Cite this paper:
Lin A, Huang Q, Zhang J et al. Digestive System Toxicities of Immune Checkpoint Inhibitors: Mechanisms and Clinical Implications. BIO Integration 2026; 7: 1–22.
DOI: 10.15212/bioi-2025-0169. Available at: https://bio-integration.org/
Download citation
© 2026 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Immune checkpoint inhibitors (ICIs) have achieved remarkable efficacy in treating malignancies but may simultaneously induce immune-related adverse events (irAEs), among which ICI-related digestive system adverse events (ir-DSAEs) occur with relatively high frequency and present severe clinical manifestations. This review systematically examined ir-DSAEs, including clinical presentations, pathogenic mechanisms, risk factors, diagnostic approaches, and therapeutic strategies. It has been reported that ir-DSAEs primarily include colitis, hepatitis, and pancreatitis with underlying mechanisms involving T cell-mediated inflammatory responses, cytokine storm, activation of autoimmune reactions, and alterations in gut microbiota. The diagnosis of ir-DSAEs requires integration of multiple approaches. Standardized therapeutic strategies include treatment regimen adjustments, corticosteroid administration, use of selective immunosuppressants, and comprehensive supportive care. An in-depth understanding of ir-DSAEs has significant clinical implications for enhancing the safety and efficacy of ICI therapy, thereby providing a theoretical foundation for future precise prediction, prevention, and treatment of ir-DSAEs.
Keywords
Biological mechanisms, cancer, digestive system adverse events, immune checkpoint inhibitors.
Introduction
Immune checkpoint inhibitors (ICIs), as revolutionary cancer immunotherapeutic approaches, have achieved remarkable efficacy in the treatment of malignancies [1]. Multiple studies indicate that tumor cells evade immune surveillance by expressing multiple immunosuppressive proteins that inhibit the activation of immune effector cells [2, 3]. ICIs function by reversing the exhausted phenotype of immune cells [4, 5], thereby reactivating T cell immune responses against tumor cells and facilitating tumor cell destruction. Current evidence suggests that ICIs primarily target signaling pathways related to programmed cell death receptor-1 (PD-1), programmed cell death ligand 1 (PD-L1), and cytotoxic T lymphocyte-associated antigen 4 [CTLA-4] [6–9]. Furthermore, novel ICIs are under development, among which lymphocyte activation gene 3 (LAG-3) inhibitors have demonstrated promising clinical potential and are emerging as next-generation therapeutic agents [10]. The advent of ICIs has provided new treatment options for patients with unresectable tumors and elderly patients with malignancies. These agents have become important first-line therapeutic regimens in clinical oncology due to significant antitumor activity and favorable safety profiles [11].
However, the robust immune activation elicited by ICIs through reversal of immune suppression also triggers a spectrum of immune-related adverse events [irAEs] [12–14]. irAEs are a series of autoimmune-like reactions triggered by immune system dysregulation during ICI therapy. ICIs effectively reverse T cell exhaustion by alleviating immune suppression. However, hyperactivated T cells may aberrantly recognize and attack normal tissues, resulting in irAEs. Common irAEs include multi-system inflammatory reactions, such as rashes, hepatitis, colitis, pneumonitis, and myocarditis, as well as systemic adverse reactions, including diarrhea and fatigue [15–17]. Different ICIs classes exhibit distinct irAE profiles, as follows: PD-1 inhibitor-related irAEs predominantly manifest as skin reactions, fatigue, and diarrhea; PD-L1 inhibitor-related irAEs are similar to those of PD-1 inhibitors but generally of lesser severity [18–20]; and CTLA-4 inhibitor-related irAEs primarily present as digestive system toxicities [15]. Among multisystem irAEs, ICIs-related digestive system AEs (ir-DSAEs) are characterized by notably elevated incidence rates and severe clinical manifestations with an overall incidence ranging from 2%–40% [21]. The incidence suggests considerable organ-specific heterogeneity. Specifically, intestinal toxicity can reach 35%, the hepatotoxicity rate is approximately 14%, pancreatic toxicity is typically observed in 0.3%–3.9% of cases, and oral and esophageal toxicity is uncommon. ir-DSAEs can induce various complications with moderate-to-severe digestive system toxicity potentially leading to organ dysfunction or even failure [22], which may become life-threatening in severe cases. The occurrence of ir-DSAEs significantly reduces the quality of life with gastroenteritis-related diarrhea and vomiting frequently accompanied by decreased appetite and abdominal pain, potentially culminating in malnutrition and weight loss. The onset of ir-DSAEs frequently necessitates treatment interruption, significantly compromising treatment adherence and diminishing clinical benefit.
Despite the increasing recognition of ir-DSAEs as a prominent research focus, significant challenges remain regarding the specific pathophysiologic mechanisms, predictive factors, and optimal management strategies. Accumulating evidence indicates that the pathogenesis of ir-DSAEs involves multi-level and -pathway immune dysregulation mechanisms, including immune cell-mediated tissue injury, cytokine-driven inflammatory responses, and intestinal microbiota dysbiosis [22–24]. Early identification of ir-DSAEs not only contributes to improved quality of life but may also lead to enhanced therapeutic outcomes [13]. However, existing biomarkers and predictive models remain insufficient to accurately forecast toxicity risk for individual patients in clinical practice. Current prediction of ir-DSAEs primarily relies on peripheral hematologic parameters, including the absolute lymphocyte count (ALC), absolute eosinophil count (AEC), platelet count, neutrophil-to-lymphocyte ratio (NLR), and platelet-to-lymphocyte ratio [PLR] [25]. Nevertheless, these conventional indicators demonstrate significant limitations in predicting the onset timing, severity, and long-term prognosis of ir-DSAEs. Developing precision treatment protocols for established ir-DSAEs based on individual patient characteristics represents a critical clinical imperative. However, the development of individualized treatment regimens faces significant challenges due to an incomplete understanding of the molecular mechanisms and immunopathologic characteristics underlying ir-DSAEs. Furthermore, patient heterogeneity with respect to genetic background, immune status, co-morbidities, and concomitant medications further complicates the standardization of treatment protocols.
This review comprehensively synthesized the current research advances and clinical implications of ir-DSAEs. The article comprehensively examined the clinical manifestations, molecular pathogenesis (T cell-mediated inflammatory responses, cytokine storm, autoimmune reaction activation, and gut microbiota dysbiosis), risk factors (drug type, history of autoimmune diseases, and underlying liver conditions), diagnostic methods (clinical assessment, laboratory testing, imaging, and endoscopic examinations), and standardized therapeutic strategies for ir-DSAEs. Current evidence has demonstrated significant differences in the risk of ir-DSAEs induced by different ICIs agents, with anti-CTLA-4 inhibitors and combination immunotherapy regimens carrying the highest risk. This review not only comprehensively synthesized clinical and preclinical research evidence but also developed an integrated “mechanism-prediction-intervention-translation” analytical framework by proposing novel pathologic models, including compartmentalized cytokine dysregulation and the bidirectional intestinal immunity-microbiota axis, thereby elucidating the immunologic basis of ir-DSAEs. Furthermore, this review provided, for the first time, a systematic synthesis and comparative analysis from the digestive system perspective, thereby establishing a comprehensive framework for organ-specific immune toxicity, and examined precision prediction models integrating multi-omics and artificial intelligence approaches alongside intervention strategies for immune homeostasis restoration. A comprehensive understanding of ir-DSAEs and the effective management is critical for optimizing the safety of ICI therapy, enhancing quality of life, and maximizing clinical efficacy. This review was intended to furnish a solid theoretical foundation and mechanistic insights and identify future translational research opportunities for clinical practice.
Clinical manifestations of digestive system adverse events
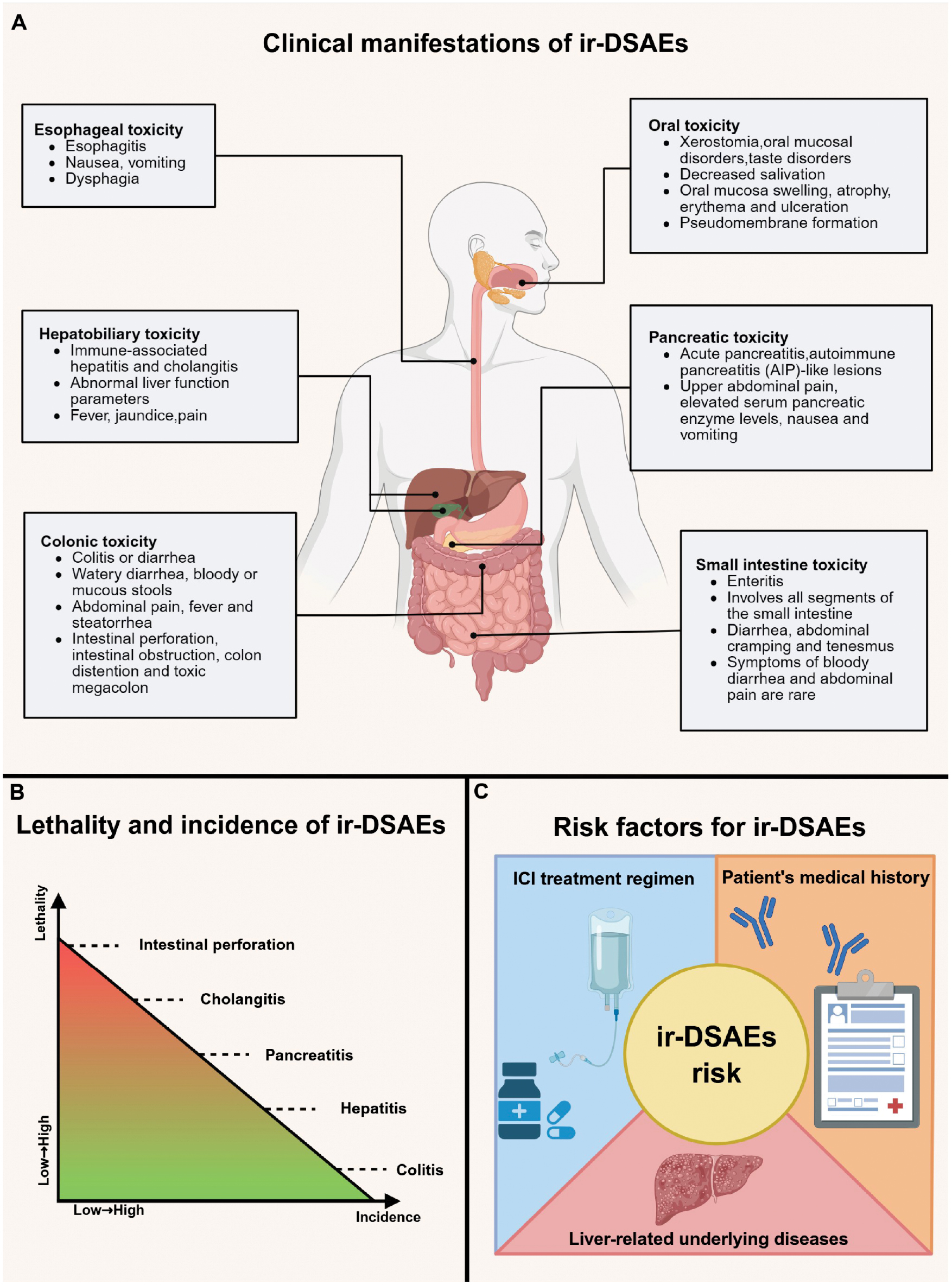
ir-DSAEs represent one of the most prevalent and clinically consequential types of irAEs. The overall incidence of ir-DSAEs ranges from 2%–40% across multiple studies, representing approximately 32.4% of all grade ≥ 3 irAEs, thereby underscoring a substantial clinical burden and severity within the spectrum of irAEs [21, 26]. ir-DSAEs can involve multiple organ systems, including the gastrointestinal tract, hepatobiliary system, pancreas, oral cavity, and esophagus (Figure 1). Specifically, gastrointestinal toxicity characteristically presents as colitis and diarrhea. Hepatobiliary toxicity occurs in approximately 14% of patients with clinical presentations primarily comprising autoimmune hepatitis and cholangitis, among which cholestatic liver injury is the most common. Patients with ICI-related hepatitis characteristically present with markedly elevated transaminases and other deranged liver function parameters, whereas ICI-related cholangitis exhibits a greater propensity for severe disease and increased mortality despite a lower incidence. Pancreatic toxicity characteristically manifests as pancreatitis, occurring in 0.3%–3.9% of patients. Pancreatic toxicity is comprised of two distinct entities: acute pancreatitis; and autoimmune pancreatitis (AIP)-like presentation. The clinical symptoms of pancreatic toxicity typically include epigastric tenderness, elevated serum amylase, nausea, and vomiting (Table 1). Oral and esophageal toxicities are uncommon with clinical manifestations typically including xerostomia and esophagitis, which are generally relatively mild. Early recognition, ongoing monitoring, and standardized management of ir-DSAEs are critical for optimizing cancer patient outcomes and preserving quality of life.
Figure 1 Clinical characteristics, fatality, and risk factors of immune-related digestive system adverse events (ir-DSAEs). This figure systematically illustrates the clinical manifestation spectrum, organ involvement patterns, fatality risk, and high-risk population characteristics of ir-DSAEs, which are divided into three sections (panels A–C). Panel A: An overview of the major organ systems affected by ir-DSAEs and the characteristic clinical syndromes. The illustration encompasses stomatitis (xerostomia, dysgeusia, and oral ulceration), esophagitis (dysphagia and retrosternal pain), hepatobiliary toxicity (autoimmune hepatitis and cholangitis), pancreatitis (epigastric pain, nausea, and elevated serum amylase), and enterocolitis (diarrhea, hematochezia, and abdominal pain). This panel highlights the “multi-organ, multi-phenotype” nature of ir-DSAEs. Panel B: A comparison of the incidence and fatality rates among different types of ir-DSAEs. Colitis/diarrhea is the most common (30%–35%, predominantly associated with CTLA-4 inhibitors) but exhibits low fatality, whereas intestinal perforation, cholangitis, and pancreatitis, though rare (≤5%), demonstrate extremely high fatality rates. A color gradient is used to depict the incidence-fatality bidimensional risk spectrum across different organs, thereby intuitively demonstrating data magnitude and clinical risk gradients. Panel C: A summary of key risk factors for ir-DSAEs, including treatment regimens (combination therapy > anti-CTLA-4 monotherapy > anti-PD-1/PD-L1), host factors (pre-existing hepatobiliary diseases), and patient medical history (prior autoimmune diseases). The figure provides a reference framework for clinical risk stratification by highlighting these risk factors. This figure was created using tools provided by Biorender.com (accessed on August 4, 2025).
Table 1 Summary of Incidence, Clinical Manifestations, and Therapeutic Strategies of ir-DSAEs Across Different ICI Regimens and Tumor Types
| Category of ir-DSAEs | Tumor Type | Anti-CTLA-4 (Incidence %) | Anti-PD-1/PD-L1 (Incidence %) | Combination Therapy (Incidence %) | Clinical Manifestations | Therapeutic Strategies | References |
|---|---|---|---|---|---|---|---|
| Diarrhea/colitis | Pan-cancer | 9.1% (colitis) | 1.3% (colitis) | 13.6% (colitis) | Watery diarrhea, which may be accompanied by abdominal pain and tenesmus; some patients present with mucoid or bloody stools; fever, weight loss, and dehydration may occur. Severe cases may develop intestinal perforation, toxic megacolon, and intestinal obstruction. | General principles: Should follow CTCAE criteria. Grade 1 (increase in stool frequency < 4 times per day above baseline; mild abdominal discomfort): Symptomatic support (hydration, electrolyte correction, antidiarrheal/analgesic agents); close follow-up. Grade 2 (increase in stool frequency of 4–6 times per day with moderate increase in ostomy output): Withhold ICIs; initiate systemic corticosteroids; hospitalization if necessary. Grade ≥ 3 (increase in stool frequency ≥ 7 times per day; severe abdominal pain, bloody stools, or abdominal distension): Continue or escalate immunosuppression, with biologic agents as first-line therapy; surgical consultation and intervention are required for perforation/toxic megacolon. Steroid-refractory/recurrent cases: Consider second-line/third-line strategies (vedolizumab, infliximab, JAK or IL-6 inhibitors, fecal microbiota transplantation). | [32] |
| Pan-cancer | 33% (diarrhea); 7% (colitis) | 10% (diarrhea); 2% (colitis) | 21%–37% (diarrhea) and 4%–8% (colitis) | [27] | |||
| Non-small cell lung cancer | 27%–30% (diarrhea); 4% (colitis) | 5%–24% (diarrhea); <4% (colitis) | 6.4%–20% (diarrhea); 1%–6% (colitis) | [156–159] | |||
| Melanoma | 4% (grade ≥ 3 diarrhea); 2% (grade ≥ 3 colitis) | — | 5% (grade ≥ 3 diarrhea); 11% (grade ≥ 3 colitis) | [160] | |||
| Melanoma | 30.4%–40.5% (diarrhea); 6.1%–11.5% (colitis) | 10.1%–17.2% (diarrhea); 0.7%–2.4% (colitis) | — | [161] | |||
| Hepatitis | Pan-cancer | 15% | 5%–10% | 25%–30% | Can be classified into cholestatic liver injury, hepatocellular liver injury, and mixed liver injury; often insidious, primarily manifesting as abnormal liver function parameters, fatigue, poor appetite, jaundice, fever, and right upper quadrant discomfort/pain. Patients with cholangitis are more prone to severe disease progression. | General principles: Should follow CTCAE criteria. Grade 1 (asymptomatic; mild elevation of liver enzymes): Monitor and may continue treatment/brief interruption; close follow-up of liver function. Grade 2 (mild fatigue or right upper quadrant discomfort): Withhold ICIs and initiate systemic corticosteroids; closely monitor biochemical parameters. Grade ≥ 3 (significant fatigue, jaundice, right upper quadrant pain, fever, liver failure, coagulopathy, altered consciousness): Discontinue ICIs and administer corticosteroids; MMF is preferred for steroid-refractory cases; intensified immunosuppression with hepatology specialist intervention when necessary. Cholangitis/cholangiopathic injury: Radiologic assessment, early intervention, and further management based on pathology/imaging findings; although cholangitis is rare, its high severity rate warrants close attention. |
[7] |
| Non-small cell lung cancer | 5%–42% | 1%–23% | — | [156, 162–164] | |||
| Renal cell carcinoma | 3%–9% | 0.7%–1.8% | 29% | [98] | |||
| Melanoma | 3.9% | 3.8% | 17.6% | [156] | |||
| Small cell lung cancer | — | 4.8% | 10.4% | [165] | |||
| Colorectal cancer | — | 2.6% (PD-1), 2.2% (PD-L1) | 1.7% | [166] | |||
| Pancreatitis | Pan-cancer | Approximately 3%–4% | Approximately 1.5%–2% | 5%–7% | Can be classified into acute pancreatitis and AIP-like lesions; primarily manifests as epigastric tenderness, nausea and vomiting, diarrhea, fever, and elevated serum pancreatic enzyme levels. | General principles: Should follow CTCAE criteria. Grade 1 (mild/enzyme elevation without symptoms): Symptomatic treatment and monitoring, assess risk of continuing medication. Grade 2 (mild-to-moderate abdominal pain or enzyme elevation < 3 × ULN): Withhold ICIs, routine supportive care. Grade ≥ 3 (significant symptoms or enzyme elevation ≥ 3 × ULN): In addition to routine supportive care, systemic corticosteroids may be used; intensified immunosuppressive support may be considered for steroid-refractory or severe cases. | [167] |
| Pan-cancer | 0.99%–8.65% | 1.86%–4.87% (PD-L1); 1.67%–2.39% (PD-1) | 1.84%–7.67% | [168] |
Abbreviations: ir-DSAEs: ICIs-related digestive system AEs; ICIs: immune checkpoint inhibitors; PD-1: programmed cell death receptor-1; PD-L1: programmed cell death ligand 1; CTLA-4: cytotoxic T lymphocyte-associated antigen 4; CTCAE: Common Terminology Criteria for Adverse Events; JAK: Janus kinase; IL-6: interleukin-6; MMF: mycophenolate mofetil; AIP: autoimmune pancreatitis; ULN: upper limit of normal.
Gastrointestinal toxicity
Among ICI-related adverse events, ir-DSAEs are most frequently manifested as colitis, diarrhea, and enteritis with clinical presentations ranging from asymptomatic-to-severe intestinal perforation. Epidemiologic data demonstrated that the overall incidence of anti-CTLA-4 antibody-associated diarrhea is 30%–35% with colitis occurring in 5%–10% of patients, exhibiting a clear dose-dependent relationship [27–31]. In contrast, the incidence of anti-PD-1/PD-L1 antibody-associated diarrhea is approximately 10% with colitis occurring in 1%–2% of patients [27, 32, 33]. The primary clinical manifestation of ICI-related colitis is watery diarrhea, although some patients may present with bloody or mucous stools [28, 34]. In addition, patients may experience systemic symptoms, such as abdominal pain, fever, steatorrhea, weight loss, vomiting, and nausea [33–36]. Severe cases may develop intestinal perforation with an incidence of approximately 1%, which requires urgent surgical intervention [37]. Other severe complications include intestinal obstruction, colonic distension, and toxic megacolon [38], which not only significantly impact the quality of life but may also be life-threatening. ICI-induced diarrhea, classified according to the Common Terminology Criteria for Adverse Events (CTCAE) grading criteria, ranges from mild-to-life-threatening and may cause complications, such as weight loss, vomiting, abdominal pain, severe dehydration, neutropenia, electrolyte disturbances, and fever [34, 35, 39]. It has been reported that some patients may present with melena [40], which suggests potential upper gastrointestinal bleeding. It should be emphasized that when patients present with mucous bloody stools accompanied by abdominal pain, clinicians should be vigilant for the possibility of colitis rather than simple diarrhea [28].
Patients may develop ICI-related enteritis when ir-DSAEs involve the small intestine. ICI-related enteritis frequently occurs concurrent with colitis, while isolated enteritis is rarely reported. Consequently, the independent incidence of ICI-related enteritis is relatively low and in clinical practice ICI-related enteritis is typically considered in conjunction with colitis. ICI-related enteritis primarily affects various segments of the small intestine (duodenum, jejunum, and ileum) with characteristic clinical manifestations of watery diarrhea, and less frequently, bloody diarrhea [41]. Common symptoms of enteritis caused by ir-DSAEs include diarrhea, abdominal cramping, and tenesmus, whereas severe abdominal pain and hemodynamic instability are relatively uncommon [41]. Overall, ICI-related enteritis exhibits clinical features very similar to ICI-related colitis. However, ICI-related enteritis is less commonly associated with hematochezia and severe abdominal pain, suggesting a relatively lower degree of mucosal inflammation and disruption.
The severity assessment of ICI-related diarrhea and (entero)colitis is primarily based on the CTCAE, as established by the National Cancer Institute. Diarrhea severity is classified into the following 5 grades: grade 1 is characterized by an increase in stool frequency (<4 stools per day) with mild symptoms; grade 2 is characterized by an increase in stool frequency (4–6 stools per day) accompanied by a moderate increase in ostomy output; grade ≥ 3 are categorized as severe diarrhea requiring urgent medical intervention and defined as an increase in stool frequency (≥7 stools per day) above baseline, necessitating heightened clinical attention; and grades 4 and 5 diarrhea are potentially life-threatening and associated with a high mortality risk. (Entero)colitis severity is similarly classified into 5 grades, as follows: grade 1 is defined by patients with no apparent symptoms, warranting diagnostic observation; grade 2 patients manifest abdominal pain and mucous or bloody stools; grade 3 is characterized by severe or persistent abdominal pain, fever, and intestinal obstruction; and grades 4 and 5 represent life-threatening conditions that can potentially lead to death.
Hepatobiliary toxicity
Hepatobiliary toxicity among ir-DSAEs constitutes one of the current foci of research. Although the incidence of hepatobiliary toxicity is lower than intestinal toxicity, the complex pathologic mechanisms and diverse injury patterns pose significant challenges to clinical diagnosis and management. Hepatobiliary toxicity can be categorized into two types (immune-related hepatitis and cholangitis), which exhibit significant differences in clinical manifestations, therapeutic strategies, and prognosis. Data from recent studies indicated that the overall incidence of ICI-related hepatotoxicity is 14%, with severe (grade ≥3) hepatic adverse events occurring in 7% of patients, potentially leading to fatal outcomes [42]. In clinical practice common liver injuries include cholestatic, hepatocellular, and mixed pattern liver injuries [43]. A study conducted at Cochin University Hospital between 2010 and 2017 found that cholestatic liver injury occurred at the highest rate (60.3%), while mixed pattern liver injury occurred at the lowest rate [10.3%] [43], which aligns with data from a Japanese survey that indicated 60.7% of patients experienced cholestatic or mixed pattern liver injury [44]. Notably, medications exert varying effects on hepatotoxicity. It has been reported that patients receiving CTLA-4 inhibitors are more susceptible to hepatotoxicity, which is characterized clinically by fever, while patients receiving PD-1 or PD-L1 inhibitors have a slightly lower probability of developing hepatotoxicity with milder symptoms [7, 45]. The onset of ICI-related hepatotoxicity is typically occult with mild hepatitis generally manifesting as abnormal liver function parameters, such as elevated aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels [46], with some patients also exhibiting elevated bilirubin or alkaline phosphatase (ALP) levels [47]. More severe cases may present with systemic symptoms, such as fever, jaundice, and peri-hepatic pain [45]. One study further noted that patients with ICI-related hepatitis may experience changes in stool color [22]. ICI-related cholangitis, although rare, is associated with substantially more severe clinical consequences compared to ICI-related hepatotoxicity and is characterized by refractory cholestasis and progressive biliary tract injury with a poor prognosis. The incidence of cholangitis has not been established. According to relevant reports, among 91 patients receiving nivolumab for non-small cell lung cancer, only 3.3% (3 cases) developed ICI-related cholangitis [48]. ICI-related cholangitis can be classified into three categories: extrahepatic; intrahepatic; and diffuse cholangitis [17]. Extrahepatic bile duct abnormalities occur at a higher proportion with an incidence of 76.9%, presenting as localized extrahepatic bile duct dilation and bile duct wall thickening with the absence of biliary stricture as the distinctive clinical characteristic [49]. The intrahepatic type commonly presents with multiple irregular strictures of intrahepatic bile ducts without extrahepatic biliary enlargement, occurring at a rate of 30.4% [17, 49]. Patients with diffuse biliary tract thickening accompanied by multiple strictures of intra- and extra-hepatic bile ducts can be diagnosed with the diffuse type [49].
Pancreatic toxicity
The pancreas is one of the commonly affected organs during ICI therapy with ir-DSAEs primarily manifesting as pancreatitis. ICI-related pancreatitis, which is similar to cholangitis, is a rare adverse event with a reported incidence rate of 0.3%–3.9% [50] and in severe cases may lead to fatal outcomes [51]. Current research indicates that ICI-related pancreatitis can be primarily categorized into two distinct types (acute pancreatitis and AIP-like lesions [52]. Patients with acute pancreatitis present clinically with fever, abdominal distension, upper abdominal pain, and elevated serum pancreatic enzyme levels [53], whereas patients with AIP-like lesions demonstrate abdominal tenderness and upper limb erythema on physical examination with imaging studies revealing pancreatic enlargement and peripancreatic fluid collection accompanied by elevated serum pancreatic enzyme levels and recurrent episodes [54]. The typical clinical manifestations of ICI-related pancreatitis include upper abdominal tenderness, nausea and vomiting, diarrhea, and fever [54, 55]. It should be noted that a subset of patients may present asymptomatically [56].
Oral and esophageal toxicity
ICIs can induce upper digestive tract toxicity, which primarily manifests as oral and esophageal toxicities. Epidemiologic data have indicated that ir-DSAEs affecting the oral cavity occur at a rate of 6.8% [57] with the most common manifestations being xerostomia (68.5%), oral mucosal disorders (33.4%), and taste disorders (24.0%) [57, 58]. Patients with xerostomia characteristically present with reduced salivary secretion [58], while oral mucosal disorders manifest as swelling, atrophy, erythema, and ulceration of the oral mucosa [59]. The severity of these mucosal disorders can range from mild erythema-to-severe ulceration with pseudomembrane formation, potentially progressing to death in extreme cases. ir-DSAEs affecting the esophagus are rare and typically present as esophagitis. It has been reported that ICIs-induced esophagitis occurs at a rate of 1%–2.7% and is predominantly of mild-to-moderate severity [60]. The symptoms of esophagitis are generally mild, primarily presenting as nausea and vomiting, often accompanied by dysphagia, hematemesis, indigestion, and melena [61].
Mechanisms underlying digestive system adverse events
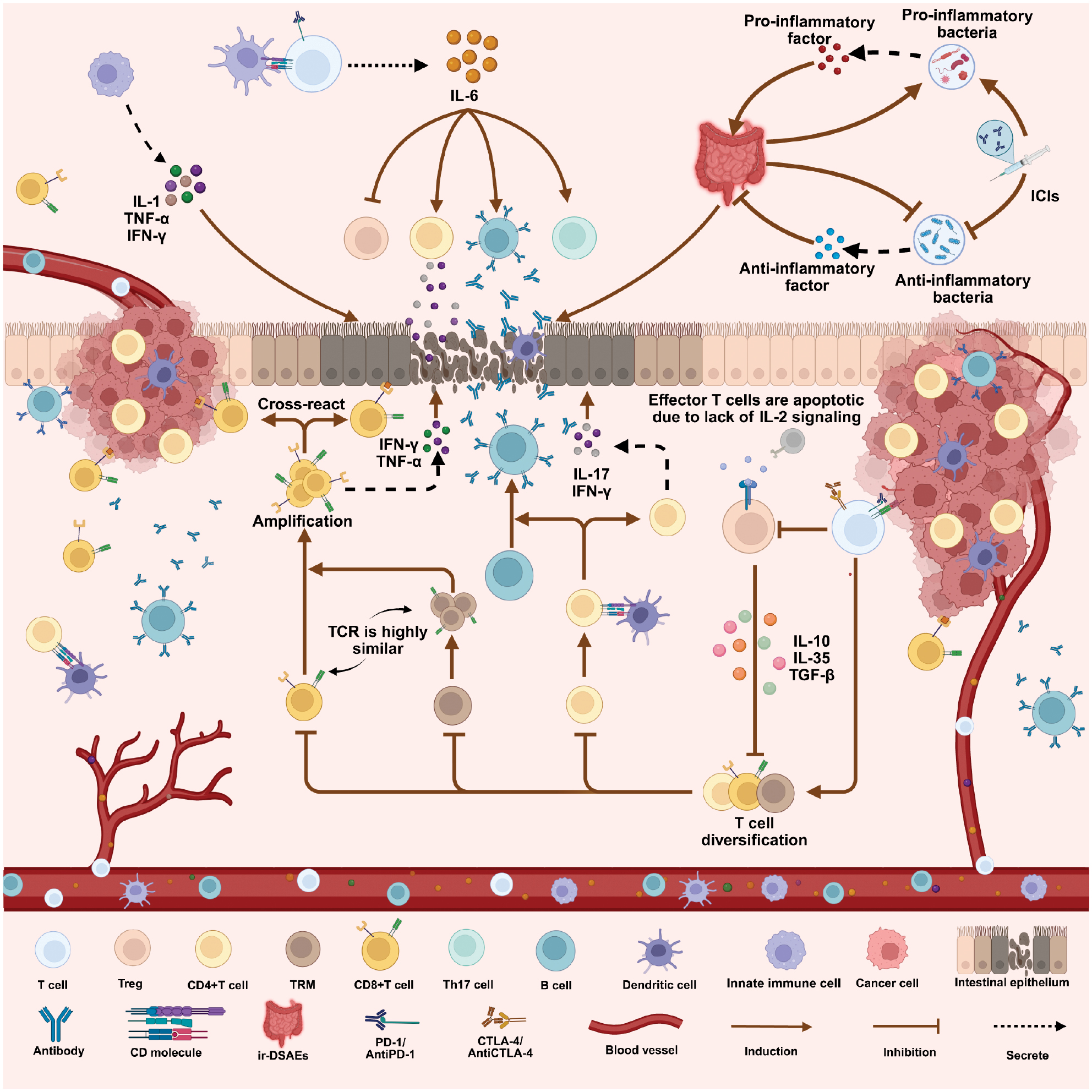
Current evidence suggests that there are four pathogenic mechanisms underlying ir-DSAEs, as follows: T cell-mediated inflammatory responses; compartmentalized cytokine dysregulation; activation of autoimmune responses; and compositional alterations in the gut microbiota (Figure 2). Specifically, ICIs activate multiple T cell subsets, including regulatory T cells (Tregs), CD4+ T cells, CD8+ T cells, and tissue-resident memory T cells (TRM) to orchestrate inflammatory responses. The accompanying compartmentalized cytokine dysregulation, particularly IL-6-mediated inflammatory cascades, further amplifies immune-mediated tissue injury. Concurrently, blockade of immune checkpoint signaling compromises self-tolerance mechanisms, thereby eliciting immune responses against intestinal autoantigens. Furthermore, disruption of gut microbiota homeostasis, especially reduced abundance of commensal bacteria and expansion of pro-inflammatory bacterial taxa, may further intensify inflammatory responses. These pathologic mechanisms demonstrate intricate mutual interactions and causal relationships, collectively establishing a complex regulatory network that governs the pathogenesis and progression of ir-DSAEs.
Figure 2 Pathophysiologic mechanisms of ir-DSAEs. This figure systematically illustrates the multi-layered immune dysregulation network triggered within the digestive system following ICI treatment. These mechanisms can be categorized into four major pathologic pathways: T cell-mediated inflammatory cascades; compartmentalized cytokine dysregulation; activation of autoimmune responses; and disruption of gut microbiota composition. ICIs lead to impaired immunosuppressive function of Tregs, accompanied by the activation and clonal expansion of CD4⁺, CD8⁺, and tissue-resident memory T cells (TRMs). Activated effector T cells and Th17 cells secrete pro-inflammatory mediators and synergistically promote mucosal inflammation, epithelial cell damage, and tissue destruction. Impaired Treg function further weakens the negative regulation of effector T cells, establishing an endogenous basis for sustained inflammation. Concurrently, IL-6-driven compartmentalized cytokine dysregulation promotes Th17, B cell, and CD4⁺ T cell activation, while suppressing Treg function, resulting in persistent cytokine imbalance and inflammatory cascades. Disruption of immune tolerance and antigen cross-recognition triggers autoreactive T cell infiltration and autoimmune attacks against intestinal tissues. Conversely ICI treatment disrupts the gut micro-ecologic balance by reducing anti-inflammatory bacteria, such as Lactobacillus and Bifidobacterium, while increasing the abundance of pro-inflammatory species, thereby weakening the mucosal barrier and inducing the further release of inflammatory mediators. The aforementioned immune cell dysregulation, compartmentalized cytokine imbalance, autoimmune activation, and microbiota disruption interact with and amplify one another through positive feedback loops, ultimately constituting the immunopathologic network underlying the development and progression of ir-DSAEs. This figure was created using tools provided by Biorender.com (accessed on October 25, 2025).
T Cell-mediated inflammatory response
ICIs enhance anti-tumor immunity by blocking signaling pathways, such as PD-1 and CTLA-4, thereby alleviating the negative regulation of T cell activation. The PD-1/PD-L1 signaling axis restricts T lymphocyte proliferation and cytokine production under physiologic conditions [62]. Blockade of this axis can lead to excessive T cell activation and tissue toxicity. In addition, CTLA-4 is a critical molecule involved in regulating T cell activation [63]. As a co-inhibitory checkpoint molecule on the T cell surface, CTLA-4 maintains immune homeostasis by inhibiting the CD28-B7 co-stimulatory pathway [64]. Blockade of the CTLA-4 pathway results in uncontrolled clonal expansion of T cells and functional impairment of regulatory Tregs [65]. Tregs have a critical role in constraining inflammatory responses [66], functioning not only by suppressing inflammation through secretion of anti-inflammatory cytokines (IL-10, IL-35, and TGF-β), but also by inhibiting effector T cell expansion through competitive IL-2 binding via the surface receptor, CD25 [67]. Upon suppression of Treg function increased CD8+ T cell activation may result in exacerbated inflammatory responses [68, 69]. Furthermore, tissue-resident memory T cells (TRMs) also contribute to the pathogenesis of ir-DSAEs. Luoma et al. demonstrated that TRMs harbor the same T cell receptor (TCR) repertoire as colitis-associated T cells, and notably in patients with colitis, 56.62% of clonally expanded TCRs derived from TRM clusters overlap with colitis-associated T cell clusters, indicating that TRM activation and expansion contribute substantially to the inflammatory amplification of ir-DSAEs [70]. Beyond the aforementioned cell subsets, multiple additional T cell populations collectively orchestrate inflammatory responses. Antigen-specific T cells are primed through CD28-CD80/CD86 interaction upon antigen presentation, whereby antigen-presenting cells present major histocompatibility complex (MHC)-peptide complexes. Activated antigen-specific T cells promote B cell differentiation into plasma cells, facilitating autoantibody production [65]. CD4+ T cells amplify tissue inflammation by secreting pro-inflammatory cytokines [IL-17 and IFN-γ] [71]. Collectively, patients with ir-DSAEs have elevated frequencies of multiple T cell subsets (Tregs, CD4+, CD8+ T cells, and antigen-specific T cells), which perpetuate the pathogenesis of ir-DSAEs through synergistic interactions with excessive T cell activation sustaining chronic inflammatory progression.
Compartmentalized cytokine dysregulation
Compartmentalized cytokine dysregulation represents a critical factor in the development of ir-DSAEs. Specifically, ICIs exert antitumor effects through enhanced immune system activity. However, this immune activation may lead to excessive cytokine release from immune cells under sustained hyperactivation, subsequently triggering compartmentalized uncontrolled inflammatory cascade reactions in the host [72, 73]. ir-DSAEs represent the clinical manifestation of compartmentalized cytokine dysregulation, which essentially results from the massive release of cytokines by various immune cells, including T cells, B cells, natural killer cells, macrophages, and endothelial cells, due to non-specific immune activation during immunotherapy [74]. Innate immune cells recognize pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs) upon ICI stimulation through pattern recognition receptors (such as TLRs), subsequently activating downstream signaling pathways and inducing excessive production of proinflammatory cytokines and chemokines, including IL-1, IL-6, and TNF-α [75]. These cytokines not only recruit additional immune cells to migrate to inflammatory sites but also act on distant organs through humoral pathways, triggering severe regional inflammatory responses in multiple systems, including the digestive system, potentially culminating in serious clinical manifestations, such as gastrointestinal bleeding and perforation. Notably, among numerous factors IL-6 is considered a pivotal molecular hub in ir-DSAEs. As a crucial proinflammatory cytokine, IL-6 not only promotes Th17 cell proliferation and enhances Th17 function and inhibits Treg-mediated regulation of CD4+ T cells but also induces trans-differentiation of Tregs into proinflammatory CD4+ T cells, while facilitating B cell and plasma cell proliferation [76]. Animal model studies further validated this central role. Serum IL-6 levels in mice with ir-DSAEs were significantly elevated compared to control groups [77]. Furthermore, different types of ICIs may trigger a cytokine storm through distinct molecular mechanisms. For example, atezolizumab binding to hepatocyte PD-L1 activates the RIP1/RIP3/MLKL complex, promoting necrosome formation and cell membrane lysis, thereby inducing hepatocyte toxicity [78]. In summary, ICI-induced compartmentalized cytokine dysregulation has a central role in the pathogenesis of ir-DSAEs, wherein excessive activation of the proinflammatory cytokine network, particularly the IL-6-driven inflammatory amplification loop, constitutes a critical pathologic foundation of ir-DSAEs.
Activation of autoimmune responses
Activation of autoimmune responses represents one of the key mechanisms underlying ir-DSAEs. Immune checkpoints prevent autoimmunity and inflammatory diseases by maintaining immune tolerance under physiologic conditions [79]. However, ICIs disrupt immune homeostasis by inhibiting immune checkpoint signaling pathways, including PD-1/PD-L1 and CTLA-4, thereby obscuring the distinction between antitumor and autoimmune responses. Studies using transgenic mouse models with small intestine-specific OVA expression have confirmed that PD-1/PD-L1 inhibitors disrupt CD8+ T cell tolerance to intestinal self-antigens, resulting in OVA-specific autoreactive CD8+ T cell infiltration and excessive release of IFN-γ and TNF-α, ultimately inducing severe tissue inflammation [80]. Furthermore, antigen cross-reactivity constitutes one of the critical mechanisms contributing to autoimmune ir-DSAEs. Antitumor T cells may mistakenly attack normal self-tissues When epitopes homologous to tumor antigens are recognized in healthy tissues. A growing body of research has demonstrated that this cross-reactivity is closely associated with the occurrence of ir-DSAEs [81] and has a particularly important role in the pathologic process of digestive system ir-DSAEs. Therefore, ICI therapy disrupts the homeostatic balance of lymphocytes and cytokines in vivo through immune checkpoint inhibition, ultimately triggering immune-mediated damage to normal digestive system tissues and representing a critical pathogenic mechanism underlying ir-DSAEs.
Alterations in the gut microbiome
In recent years accumulating evidence has shown that the gut microbiota serves as a critical link between immune homeostasis and ir-DSAEs. ICI therapy modulates the composition and metabolic functions of the gut microbiome through multiple mechanisms, consequently affecting mucosal immune responses and inflammatory thresholds [82–88]. Clinical studies have established that fecal microbiota transplantation (FMT) can effectively ameliorate the clinical manifestations of ir-DSAE-related refractory colitis, indicating that microbial homeostasis has a critical role in disease pathogenesis [24, 89]. B cell-mediated immune dysregulation may impair mucosal barrier function, thereby promoting the translocation of diverse bacterial species and triggering subsequent inflammatory cascades. Probiotics, such as Lactobacillus and Bifidobacterium species, exert protective effects in sustaining an anti-inflammatory microenvironment. Wang et al. reported that anti-CTLA-4 treatment leads to a significantly reduced relative abundance of Lactobacillus in IMC mouse models [90]. Lactobacillus exerts anti-inflammatory effects by inhibiting pro-inflammatory factor expression and promoting anti-inflammatory factor production. Consequently, this protective effect is significantly weakened when the abundance of Lactobacillus decreases. Bifidobacterium promotes inflammation resolution by regulating the gut microbiota network composition [91]. During ICI treatment patients experience significantly reduced gut microbial diversity, decreased abundance of anti-inflammatory bacterial populations, and increased proliferation of pro-inflammatory bacterial populations, including Proteobacteria, Pasteurellaceae, Veillonellaceae, Clostridium, and Ruminococcus [92]. These pro-inflammatory microbial populations not only secrete diverse pro-inflammatory metabolites that amplify intestinal inflammatory responses but may also compromise the intestinal epithelial barrier, thereby increasing the risk of pathogenic colonization and facilitating secondary infections. Gut microbial dysbiosis constitutes a consequence of excessive immune activation and a potent amplifier of sustained and disseminated inflammatory responses, thereby serving as a critical amplification loop in the pathogenesis of ir-DSAEs.
Risk factors for digestive system adverse reactions
Systematic evidence has demonstrated that the risk of ir-DSAEs is modulated by multiple determinants, including drug class, treatment regimen, and patient-related characteristics. In aggregate, CTLA-4 inhibitors and combination immunotherapy exhibit substantially greater incidence and severity of ir-DSAEs relative to PD-1/PD-L1 monotherapy inhibitors. Among patient-intrinsic factors, a history of autoimmune disease and pre-existing hepatic disease, including cirrhosis and hepatitis C virus (HCV) infection, substantially elevate the risk of ir-DSAEs. Thus, clinical practice requires careful assessment of these determinants and the adoption of personalized treatment approaches and enhanced surveillance strategies for high-risk patient populations.
ICI treatment regimens
Epidemiologic studies have indicated significant differences in the incidence of ir-DSAEs among different classes of ICIs, providing important evidence for selecting safer treatment regimens in clinical practice. Current clinical data have demonstrated that the incidence of ir-DSAEs caused by CTLA-4 inhibitors is significantly higher than that observed with PD-1/PD-L1 inhibitors [23, 93]. Clinical data have indicated that CTLA-4 inhibitor-related diarrhea occurs in up to 54% of patients [94]. In contrast, PD-1/PD-L1 inhibitors demonstrate a substantially lower incidence of ir-DSAEs with milder clinical presentations, including a 4.4% diarrhea rate [95]. Hepatic injury is observed in approximately 20% of patients [96, 97]. Notably, CTLA-4 inhibitor-induced immune-related hepatitis has an incidence ranging from 3%–9% in renal cell carcinoma patients, whereas PD-1 inhibitor-associated immune-related hepatitis occurs in only 0.7%–1.8% of renal cell carcinoma patients [98]. Notably, the incidence of ir-DSAEs significantly increases when patients receive combination therapy with CTLA-4 and PD-1 inhibitors and is accompanied by more severe clinical symptoms and earlier onset of these adverse events. The incidence of hepatotoxicity after combination therapy can reach 29% with 14% of patients experiencing severe grade 3–4 toxicity [98]. Approximately 20.7% of patients receiving combination ICI therapy develop severe hepatitis [99]. However, recent studies have suggested that although combination ICI therapy significantly increases the risk of high-grade hepatotoxicity, combination ICI therapy may simultaneously reduce the incidence of severe ir-DSAEs [100]. These observations indicate that PD-1 and PD-L1 inhibitors demonstrate superior gastrointestinal tolerability compared to CTLA-4 inhibitors, whereas combination ICI strategies frequently result in more severe ir-DSAEs.
Patient medical history
Epidemiologic evidence indicates that patients with a history of autoimmune diseases are more susceptible to ir-DSAEs when receiving ICI therapy. A large-scale prospective study demonstrated that among 45 patients with a prior history of autoimmune disease, the overall incidence of ir-DSAEs reached 44%, which was significantly higher than the 29% observed in patients without such a history [101]. ICI therapy may induce recurrence or exacerbation of pre-existing autoimmune diseases by potentiating immune system activity. Therefore, clinicians should comprehensively assess a history of autoimmune diseases when formulating treatment strategies and conduct rigorous risk-benefit evaluations.
Liver-related underlying diseases
Underlying hepatic diseases, including cirrhosis, chronic hepatitis, and hepatocellular carcinoma, are considered important risk factors for ir-DSAEs. Clinicians should closely monitor changes in liver function in patients with chronic liver disease and hepatocellular carcinoma receiving ICI therapy [102] to prevent immune-related liver injury. Studies have shown that underlying hepatic diseases, particularly cirrhosis, significantly increase the risk of hepatic toxicity in patients with hepatocellular carcinoma [103]. The incidence of grade 3–4 hepatic injury was 25% among patients with hepatocellular carcinoma treated with atezolizumab and concomitant HCV infection, which is substantially greater than the 3% rate in HCV-negative patients [104]. Based on the aforementioned evidence, clinicians should monitor hepatic function changes and gastrointestinal symptoms in patients with underlying hepatic disease closely and promptly adjust immunotherapy regimens or implement hepatoprotective and immunosuppressive interventions as appropriate.
Diagnosis and monitoring of ir-DSAEs
The diagnosis of ir-DSAEs necessitates a multidimensional assessment, including clinical manifestations, laboratory testing, imaging studies, and an endoscopic pathologic examination. Clinical manifestations of ir-DSAEs primarily include diarrhea and abdominal pain, and may also involve nausea, vomiting, hematochezia, and abdominal distension, with severe cases potentially progressing to intestinal perforation. Laboratory examinations, including hematological parameters, fecal tests, and immunostaining, offer valuable auxiliary diagnostic information. Imaging examinations, including ultrasonography and computed tomography(CT), are essential for determining the location and extent of lesions. Endoscopic examination remains the gold standard for diagnosis, enabling direct observation of mucosal lesions, while pathologic biopsies reveal characteristic inflammatory cell infiltration patterns, thus providing crucial evidence for clinical diagnosis and treatment decisions.
Clinical examination
The clinical manifestations of ir-DSAEs are heterogeneous and frequently insidious in presentation. The most common gastrointestinal manifestation is watery diarrhea, which is frequently accompanied by abdominal pain, hematochezia, nausea, vomiting, anorexia, weight loss, and fever. In severe cases, intestinal perforation may develop, necessitating emergency surgical intervention. On physical examination, patients typically present with abdominal tenderness, hyperactive bowel sounds, and abdominal distension. Clinical symptoms of hepatic and pancreatic toxicity tend to be relatively subtle, primarily manifesting as abdominal pain with pancreatic toxicity often presenting with steatorrhea and glycemic abnormalities. Patients with hepatic toxicity frequently present with jaundice on physical examination [105], facilitating rapid clinical diagnosis.
Laboratory testing
The diagnosis of ir-DSAEs requires objective evidence from laboratory testing. Hematologic testing represents the most fundamental and widely utilized examination method in this context. Elevated levels of liver enzymes, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and bilirubin, are frequently observed in patients presenting with hepatic toxicity, indicating hepatocellular injury or bile duct congestion [7, 17]. Seronegative antinuclear antibodies (ANAs) with elevated immunoglobulin G (IgG) can be detected in some patients, which may aid in the differential diagnosis of autoimmune hepatitis [106]. Fecal calprotectin testing can function as an initial screening tool in patients with ICI-related colitis, although the specificity and sensitivity parameters require further clinical validation [7]. Immunohistochemical analysis has revealed an increased proportion of CD8+ cytotoxic T lymphocytes, resulting in altered CD3+/CD20+ and CD4+/CD8+ ratios, which can function as significant diagnostic indicators for irAEs [107]. ir-DSAEs can be effectively differentiated from other clinically similar conditions through systematic functional examinations, infectious marker screening, autoimmune marker detection, and complementary diagnostic modalities, thereby providing a robust scientific basis for developing appropriate clinical treatment strategies.
Imaging studies
Imaging findings are essential for the diagnosis and severity grading of ir-DSAEs in clinical practice. Ultrasonography has become the preferred diagnostic approach among various imaging modalities because of cost-effectiveness, safety, and diagnostic efficiency. ICI-related hepatotoxicity may be present when ultrasonographic evaluation reveals abnormal liver volume and echogenicity, ascites formation, and periportal edema. Conversely, diffuse pancreatic enlargement accompanied by pancreatic duct dilation suggests potential ICI-related pancreatic toxicity. CT, serving as a crucial imaging modality for ir-DSAE diagnosis, demonstrates high sensitivity and positive predictive value in identifying these adverse events [108]. CT demonstrates that patients with ICI-related colonic toxicity frequently present with diffuse colonic wall thickening [22]. ICI-related pancreatic and biliary toxicity is primarily characterized by diffuse pancreatic enlargement, bile duct dilation, and intrahepatic bile duct strictures [17]. Periportal lymphadenopathy may be observed in patients with severe hepatotoxicity [7]. Comprehensive imaging examinations can precisely quantify the extent of digestive system involvement, thereby providing objective evidence essential for formulating appropriate clinical management strategies.
Endoscopic examination and pathologic biopsy
Gastrointestinal endoscopy remains the gold standard diagnostic method for ir-DSAEs. Endoscopic examinations can reveal characteristic manifestations of ir-DSAEs, including mucosal hyperemia, vascular pattern erosion, mucosal erythema formation, and increased mucosal friability [22, 109–111]. Studies have demonstrated significant differences in the severity of lesions across different anatomic sites of the digestive tract with research indicating that lesions in the descending colon are typically less severe than the ascending colon, while the actual severity of lesions in the sigmoid colon may be significantly higher than what is apparent during endoscopic examination [22]. Histopathologic examinations have a crucial role in diagnosis. The predominant histopathologic features of ir-DSAEs include acute inflammatory changes, characterized by marked inflammatory cell infiltration and crypt abscess formation on microscopic examination [22, 46, 112]. Chronic inflammation is less frequently observed and typically manifests as crypt distortion and granuloma formation, primarily involving the lamina propria [22]. Pathologic findings provide essential evidence for the classification and severity assessment of ir-DSAEs.
Management strategies for ir-DSAEs
Therapeutic and pharmacologic management strategies
Comprehensive treatment strategies primarily include the following for ir-DSAEs. First, appropriate treatment regimen adjustments should be implemented based on established adverse event grading criteria, including dose modification, temporary interruption, or permanent discontinuation of ICI therapy [7, 46, 113]. Second, systemic corticosteroids remain the first-line standard treatment for ir-DSAEs with dosing regimens requiring individualized optimization based on the grade and clinical presentation of adverse events [46]. Selective immunosuppressants, such as mycophenolate mofetil and infliximab, should be considered as salvage second-line treatment options for patients with inadequate response to corticosteroids or steroid-refractory cases [114]. Furthermore, appropriate comprehensive supportive care measures, including nutritional support, maintenance of fluid and electrolyte balance, and prophylactic anti-infection strategies, have a crucial role in alleviating patient symptoms, preventing complications, and enhancing the quality of life. The standardized application of these treatment strategies, coupled with individualized comprehensive interventions, can effectively control and manage ir-DSAEs, thereby ensuring the safety and continuity of cancer immunotherapy and ultimately improving patient outcomes and overall clinical benefit.
Therapeutic implications of ir-DSAEs on antitumor efficacy
Although immunosuppressive treatment for severe ir-DSAEs may raise concerns regarding potential attenuation of immunotherapy efficacy, accumulating clinical evidence suggests that standardized management generally does not significantly compromise antitumor responses. A retrospective analysis demonstrated that the use of immunomodulatory agents following any-grade ir-DSAEs does not negatively impact the rate or quality of antitumor responses [115]. Multiple studies have further confirmed that early, short-term, and appropriately dosed immunosuppressive interventions can effectively control toxicity while preserving the durability of antitumor immune responses [116–118]. Furthermore, judicious ICI discontinuation-rechallenge strategies for mitigating ir-DSAEs do not significantly diminish the antitumor efficacy of ICI therapy [119, 120]. Collectively, these findings suggest that proactive management of ir-DSAEs do not merely represent a remedial measure at the expense of antitumor efficacy but rather a therapeutic strategy that facilitates restoration of patient tolerance to immunotherapy and ensures treatment continuity, thereby preserving overall therapeutic benefit.
Impact of ir-DSAEs on anticancer efficacy
Correlation between ir-DSAEs and treatment response
The emergence of ir-DSAEs signifies activation of the host immune system, which may in turn promote antitumor responses. Numerous studies demonstrate that the occurrence of irAEs reflects the degree of immune system activation and frequently correlates positively with antitumor efficacy. Specifically, patients with non-small cell lung cancer who develop mild-to-moderate ir-DSAEs commonly derive therapeutic benefit, thus suggesting that moderate immune-related toxicity may serve as a clinical biomarker of treatment response [121]. In a study enrolling 576 patients with melanoma receiving nivolumab therapy, the treatment response rate was significantly higher in patients who developed ir-DSAEs than in patients without ir-DSAEs [115]. In contrast, patients with digestive system malignancies, such as colorectal cancer and primary hepatocellular carcinoma, are often associated with a shorter overall survival (OS) and progression-free survival (PFS), higher grades of ir-DSAEs, and frequently require treatment interruption, dose reduction, or permanent discontinuation, thereby impairing treatment adherence and reducing long-term efficacy [122, 123]. This discrepancy may be explained by the fact that the relationship between ir-DSAEs and treatment response is modulated by multiple factors, including drug and tumor type; further investigation is warranted to elucidate the underlying mechanisms. Furthermore, emerging evidence suggests that the gut microbiota composition may modulate the balance between ir-DSAEs and anticancer efficacy. Specifically, enrichment of specific bacterial taxa, such as Faecalibacterium prausnitzii, is associated with a lower risk of severe intestinal toxicity and higher treatment response rates [124].
Impact of ir-DSAEs on overall survival and prognosis
Current evidence indicates heterogeneous associations between irAEs in different organ systems and clinical prognosis. Cutaneous and endocrine irAEs may be associated with superior therapeutic efficacy, whereas the relationship between ir-DSAEs and survival outcomes remains unclear. Although multiple studies have demonstrated that irAEs are associated with prolonged OS and PFS, hepatic ir-DSAEs have not shown similar favorable prognostic outcomes. Clinical evidence has demonstrated that patients with ICI-induced hepatitis exhibit no statistically significant differences in median OS or median PFS compared to patients without hepatitis, suggesting that hepatic toxicity may not be substantially associated with patient prognosis [99, 125, 126]. Studies have demonstrated that gastrointestinal toxicity is associated with prolonged recurrence-free survival [RFS] [21], a finding that may be related to enhanced antitumor immune responses resulting from increased immune system activation. At present, evidence regarding the prognostic impact of ICI-associated pancreatic toxicity is limited and the clinical significance requires further investigation.
Future perspectives
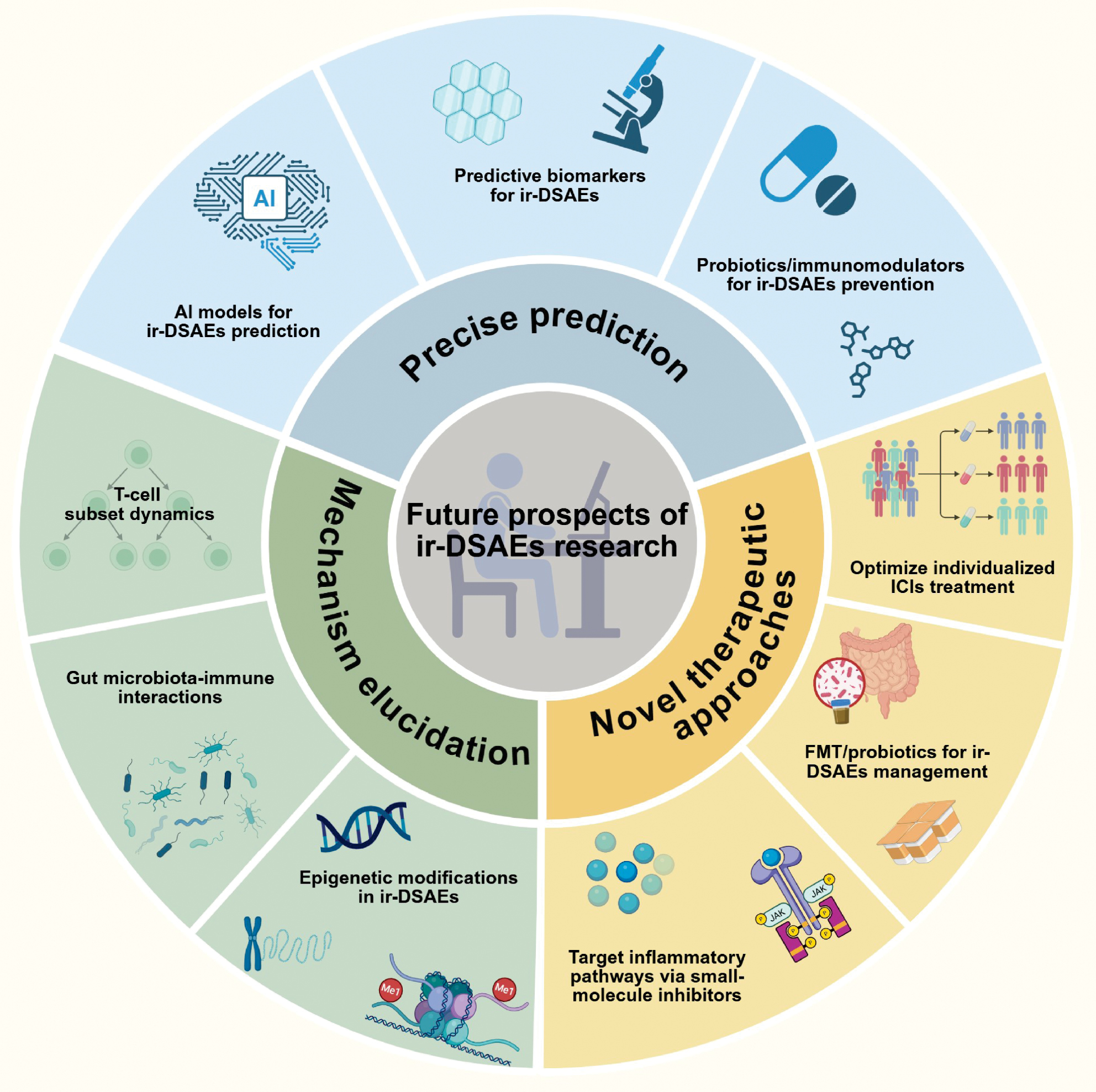
In recent years research involving ir-DSAEs has primarily focused on T cell-mediated inflammatory responses and gut microbiota influences, which has yielded significant progress. However, elucidation of more detailed molecular mechanisms and development of more effective management strategies still require continuous investigation. Future research should focus on three main directions: precise prediction; mechanism elucidation; and novel therapeutic approaches (Figure 3). Specifically, researchers can integrate emerging technologies, such as artificial intelligence, to build effective ir-DSAEs prediction platforms. At the mechanistic level, investigations can explore the dynamics of T cell populations, microbiota-immune interactions, and epigenetic influences on ir-DSAEs development. Studies may identify new immune pathway inhibitors and probiotic-assisted treatments to alleviate symptoms in ir-DSAEs patients. Future studies are expected to advance our understanding of the underlying mechanisms and facilitate the development of more comprehensive management approaches to address ir-DSAEs in clinical practice. Furthermore, advances in modern antibody engineering may enhance precision oncologic approaches to reduce the occurrence of ir-DSAEs. Future research is expected to advance our mechanistic understanding and clinical management of ir-DSAEs through investigation of underlying biological pathways and implementation of improved management strategies.
Figure 3 Frontiers in ir-DSAEs research. This figure systematically illustrates the key research frontiers and translational trends in the field of ir-DSAEs. Future research is anticipated to focus on three major directions: precision prediction and prevention; mechanistic elucidation; and innovative therapeutic strategies. Artificial intelligence and machine learning will be seamlessly integrated with clinical data, imaging, and multi-omics information to construct multidimensional risk prediction models. The identification of candidate biomarkers for high-risk patients, such as IL-17, TNFSF9, IFIT2, and IL-6, coupled with exploration of the preventive potential of probiotics or immunomodulatory agents, will also contribute to controlling the occurrence of ir-DSAEs. At the mechanistic level, research will focus on T cell population dynamics, gut–liver–immune axis interactions, and epigenetic regulation, thereby revealing the association between CD4⁺/CD8⁺ and tissue-resident memory (TRM) cell clonal expansion and inflammatory amplification, as well as elucidating the roles of gut microbiota dysbiosis, DNA methylation, and histone modifications in immune dysregulation. From a therapeutic perspective, small-molecule inhibitors targeting inflammatory pathways, such as JAK and IL-6R inhibitors, have shown promising prospects in managing steroid-refractory irAEs, while fecal microbiota transplantation (FMT) and personalized microbiome therapies have also demonstrated potential for alleviating refractory cases. Furthermore, modern antibody engineering, through Fc structural optimization, bispecific design, and conditional masking technologies, is expected to enhance tumor specificity and reduce off-target immune activation, thereby mitigating the risk of ir-DSAEs. This figure was created using tools provided by Biorender.com (accessed April 18, 2025).
Development of precise prediction and prevention strategies for ir-DSAEs
With advances in artificial intelligence (AI) and machine learning technologies, machine learning–based and deep learning–based approaches, including multimodal data integration models, have been progressively applied to integrate clinical, imaging, and genomic data to construct multidimensional risk prediction models that enable more precise identification of patients susceptible to ir-DSAEs [127, 128]. Recently developed AI models and natural language processing pipeline systems can automatically detect ICI toxicity from clinical text and these technologies may be further extended for early detection of ir-DSAEs [129]. Moreover, candidate biomarkers for ir-DSAEs have been proposed. Specifically, elevated titers of anti-integrin α v β 6 autoantibodies, increased IL-17, and elevated calprotectin levels are associated with an increased risk of colitis [130, 131]. Low expression of TNFSF9, TGFB2, LILRA3, IFIT2, and EIF2AK2 in whole blood is significantly associated with the development of colitis [132]. Decreased baseline IL-6 levels are associated with the occurrence of grade ≥3 hepatitis [133]. Elevated ALT and ALT:AST ratios may be associated with irAEs [134]. The gut microbiota may serve as a potential biomarker for ICI-related hepatotoxicity [135]. Although validation of these candidate biomarkers in large-scale cohorts or prospective trials is limited, candidate biomarkers may provide valuable insights for the prediction of ir-DSAEs. The administration of probiotics and immunomodulatory agents demonstrates considerable promise for the prevention of ir-DSAEs and multiple pilot clinical studies and registered clinical trials are currently evaluating the efficacy of these preventive strategies in ir-DSAEs [NCT06508034 and NCT06470971] [136, 137].
In-depth molecular mechanisms underlying ir-DSAEs
Further elucidation of the molecular mechanisms underlying ir-DSAEs will provide essential evidence for the development of targeted therapeutic strategies. An accumulating body of evidence from single-cell and multi-omics studies has demonstrated that T cell subset composition, clonal dynamics, and tissue localization have pivotal roles in the development of ir-DSAEs. The abundance of activated CD4+ memory T cells, TCR clonality, and TCR diversity in peripheral blood prior to treatment is associated with the development of severe irAEs [138]. Single-cell and spatial transcriptomic analyses further demonstrated that CD4+ TRM cells, CD8+ TRM cells, and cytotoxic CD8+ T cells share the same TCR clonotypes, suggesting that the latter two populations may serve as progenitors of cytotoxic effector cells [139]. Furthermore, CD4+ T cells serve as initiators of adaptive immune responses, promoting the differentiation of CD8+ TRM and circulating CD8+ T cells toward IFN γ + CD8+ T cells, thereby contributing to the pathogenesis of ir-DSAEs [68, 140]. The gut microbiota, as a key component of the host immune microenvironment, exhibits compositional and functional alterations that are closely associated with ir-DSAEs risk. Evidence has demonstrated that specific microorganisms, such as Bacteroides, Clostridium, and Faecalibacterium species, can promote Treg cell differentiation and enhance the production of anti-inflammatory cytokines, thereby reducing the incidence of ir-DSAEs [141]. Enterococcus gallinarum can activate systemic immune responses, causing TLR8 dysfunction that leads to attacks on normal self-tissues [142]. In addition, epigenetic modifications may contribute to the pathogenesis of ir-DSAEs. DNA methylation can influence gene expression in immune cells, thus regulating immune responses and inflammatory processes. Histone modifications can regulate inflammatory factor production as well as immune cell differentiation and function. Studying the role of epigenetic modifications in irAEs occurrence not only provides a theoretical foundation for potential epigenetic interventions but also potentially offers novel research perspectives on ir-DSAEs pathophysiology.
Development of novel therapeutic strategies for ir-DSAEs
Based on an in-depth understanding of ir-DSAE mechanisms, the development of more effective and precise treatment modalities represents a critical direction for future research. Recently, small-molecule inhibitors targeting specific inflammatory pathways have increasingly emerged as a research focus for treating refractory ir-DSAEs with JAK and IL-6 demonstrating the greatest therapeutic potential. JAK inhibitors, such as tofacitinib, improve ir-DSAEs through suppression of the JAK-STAT pathway and reduction of inflammatory cytokine secretion [143, 144]. IL-6, as a critical immunoregulatory cytokine, mediates immune regulation. IL-6 receptor antagonists, such as tocilizumab, have demonstrated efficacy in a recent phase II clinical trial [NCT05428007] [145] in reducing ICI-related toxicity [146]. IL-6 receptor antagonism should be considered for patients with steroid-refractory colitis refractory to conventional therapy [147]. Furthermore, FMT represents a promising therapeutic approach for ir-DSAEs. Preliminary clinical evidence demonstrates that FMT ameliorates refractory ir-DSAEs through significant alterations in baseline microbial diversity and composition [148, 149]. Prospective clinical trials suggest that FMT may serve as an effective first-line therapeutic option for ir-DSAEs [150] with ongoing clinical trials [NCT06206707] [151] evaluating this approach. However, FMT efficacy exhibits substantial interindividual variability and the stability and safety of microbiota reconstitution remain critical areas of future investigation. Advances in antibody engineering offer innovative strategies for mitigating ir-DSAE risk. Existing ICIs may elicit non-specific inflammation via Fc-mediated effector cell recruitment. Going forward, antibody engineering approaches, such as optimization of the antibody Fc region and bispecific antibody engineering, are anticipated to substantially attenuate ir-DSAE risk while preserving antitumor efficacy [152, 153]. Notably, conditionally masked antibodies have been shown to restrict immune activation to the tumor microenvironment, thereby attenuating potential ir-DSAE risk with clinical studies currently in progress [154].
Optimization of individualized ICI treatment regimens
Establishing precision treatment regimens based on individual patient characteristics is a critical factor in improving the efficacy and safety of ICI therapy. First, developing treatment decision support systems based on patient genotypes, tumor characteristics, and microbiome features is of paramount importance [155]. ir-DSAEs are closely associated with epigenetics, tumor location, and gut microbiota. Therefore, a significant approach to formulating individualized treatment plans involves the clinical stratification of patients. Developing corresponding treatment decision systems based on this classification enables physicians to develop scientifically sound and intuitive treatment plans for patients, thereby facilitating individualized therapy. Second, investigating individualized adjustment strategies for ICI dosage and administration methods is also crucial. The risk of ir-DSAEs can be minimized while ensuring optimal therapeutic effects by proactively modifying drug types and doses, thus achieving a balance between antitumor efficacy and ir-DSAE risk. In addition, actively exploring individualized combination therapy regimens incorporating other immunomodulators, such as IDO inhibitors or STING agonists, can further enhance ICI therapeutic efficacy, thereby improving quality of life, reducing various adverse events, and ultimately leading to improved prognosis and clinical outcomes.
Conclusions
As revolutionary antitumor therapeutic approaches, ICIs have significantly improved patient prognosis. However, ICI-induced ir-DSAEs continue to pose significant clinical management challenges. This review systematically summarized recent research advances in clinical characteristics, pathogenesis mechanisms, associated risk factors, and diagnostic and therapeutic strategies for ir-DSAEs. Based on existing research evidence, ICIs primarily cause digestive system toxicity through multiple mechanisms, including mediating T cell infiltration, inducing cytokine release syndrome, activating autoimmune responses, and modulating gut microbiota. Studies have indicated that multiple factors, including ICI drug type, patient history of autoimmune diseases, baseline liver function status, age, and gender, may influence the risk and severity of ir-DSAEs. Future research directions should focus on developing precise prediction models, elucidating molecular pathologic mechanisms, and exploring innovative treatment strategies, aimed at providing more accurate diagnostic and therapeutic guidance for clinical practice, effectively reducing the incidence and severity of ir-DSAEs, and ultimately improving patient prognosis and quality of life.
Abbreviations
| ICIs: | Immune checkpoint inhibitors |
| irAEs: | Immune-related adverse events |
| ir-DSAEs: | Immune checkpoint inhibitor-related digestive system adverse events |
| PD-1: | Programmed cell death receptor-1 |
| PD-L1: | Programmed cell death ligand 1 |
| CTLA-4: | Cytotoxic T lymphocyte-associated antigen 4 |
| ALC: | Absolute lymphocyte count |
| AEC: | Absolute eosinophil count |
| NLR: | Neutrophil-to-lymphocyte ratio |
| PLR: | Platelet-to-lymphocyte ratio |
| AST: | Aspartate aminotransferase |
| ALT: | Alanine aminotransferase |
| ALP: | Alkaline phosphatase |
| CTCAE: | Common terminology criteria for adverse events |
| AIP: | Autoimmune pancreatitis |
| TRM: | Tissue-resident memory T cells |
| IL-6: | Interleukin-6 |
| HCV: | Hepatitis C virus |
| CT: | Computed tomography |
| ORR: | Objective response rate |
| OS: | Overall survival |
| PFS: | Progression-free survival |
| JAK: | Janus kinase |
| STAT: | Signal transducer and activator of transcription |
| PAMPs: | Pathogen-associated molecular patterns |
| DAMPs: | Damage-associated molecular patterns |
| FMT: | Fecal microbiota transplantation |
Data availability statement
Not applicable.
Ethics statement
No direct interactions with human or animal subjects were involved. Therefore, ethical approval and informed consent were not required.
Author contributions
Writing-original draft, A.Q.L, Q.L.H, J.Z.; Conceptualization, P.L.; Investigation, A.Q.L, Q.L.H; Writing-review and editing, A.Q.L, Q.L.H, J.Z., and P.L.; Visualization, A.Q.L, Q.L.H. All authors have read and agreed to the published version of the manuscript.
Funding
No funding or sponsorship was received for this study.
Acknowledgements
We acknowledge Sogen Biotechnology, Zhengzhou, Henan Province, and the SolvingLab team for their technical support and assistance.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Consent for publication
Not applicable
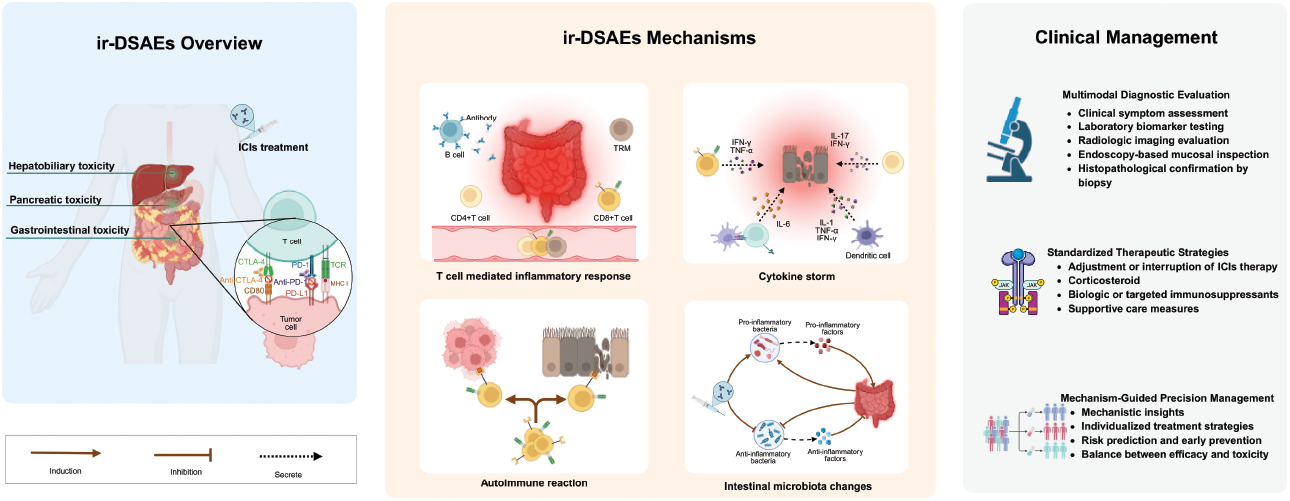
Graphical abstract
Highlights
- This review summarized the clinical spectrum, mechanisms, diagnostic workflow, and management strategies of digestive system irAEs (ir-DSAEs) induced by ICIs.
- A unified mechanistic model links T-cell activation, cytokine signaling, autoimmunity, and gut microbiome disruption to the development of ir-DSAEs.
- Drug-specific toxicity patterns of different ICI classes are compared with detailed characterization of pancreatic and hepatobiliary ir-DSAEs.
- A future research framework is proposed integrating AI-based risk prediction, single-cell analysis, and microbiome- or target-directed therapeutic strategies.
In brief
This review summarized the risk factors, immunopathogenic mechanisms, and diagnostic–therapeutic strategies of immune-related digestive adverse events induced by ICIs, highlighting the role of T-cell dysregulation, cytokine signaling, and microbiota alterations, and underscoring the need for early evaluation and mechanism-guided management.
References
- Lin A, Ye P, Li Z, Jiang A, Liu Z, et al. Natural killer cell immune checkpoints and their therapeutic targeting in cancer treatment. Research (Wash D C) 2025;8:0723. [PMID: 40463500 DOI: 10.34133/resaerch.0723]
- Kostecki KL, Iida M, Crossman BE, Salgia R, Harari PM, et al. Immune escape strategies in head and neck cancer: evade, resist, inhibit, recruit. Cancers 2024;16(2):312. [PMID: 38254801 DOI: 10.3390/cancers16020312]
- Dermani FK, Samadi P, Rahmani G, Kohlan AK, Najafi R. PD-1/PD-L1 immune checkpoint: potential target for cancer therapy. J Cell Physiol 2019;234(2):1313-25. [PMID: 30191996 DOI: 10.1002/jcp.27172]
- Sangro B, Chan SL, Meyer T, Reig M, El-Khoueiry A, et al. Diagnosis and management of toxicities of immune checkpoint inhibitors in hepatocellular carcinoma. J Hepatol 2020;72(2):320-41. [PMID: 31954495 DOI: 10.1016/j.jhep.2019.10.021]
- Zeng M, Lin A, Jiang A, Qiu Z, Zhang H, et al. Decoding the mechanisms behind second primary cancers. J Transl Med 2025;23(1):115. [PMID: 39856672 DOI: 10.1186/s12967-025-06151-9]
- Cheng AL, Hsu C, Chan SL, Choo SP, Kudo M. Challenges of combination therapy with immune checkpoint inhibitors for hepatocellular carcinoma. J Hepatol 2020;72(2):307-19. [PMID: 31954494 DOI: 10.1016/j.jhep.2019.09.025]
- Li Y, Kang X, Wang H, Guo X, Zhou J, et al. Clinical diagnosis and treatment of immune checkpoint inhibitor-associated adverse events in the digestive system. Thorac Cancer 2020;11(4):829-34. [PMID: 32107847 DOI: 10.1111/1759-7714.13338]
- Liu L, Xie Y, Yang H, Lin A, Dong M, et al. HPVTIMER: a shiny web application for tumor immune estimation in human papillomavirus-associated cancers. iMeta 2023;2(3):e130. [PMID: 38867938 DOI: 10.1002/imt2.130]
- Shi X, Cheng X, Jiang A, Shi W, Zhu L, et al. Immune checkpoints in B cells: unlocking new potentials in cancer treatment. Adv Sci (Weinh) 2024;11(47):e2403423. [PMID: 39509319 DOI: 10.1002/advs.202403423]
- Aggarwal V, Workman CJ, Vignali DAA. LAG-3 as the third checkpoint inhibitor. Nat Immunol 2023;24(9):1415-22. [PMID: 37488429 DOI: 10.1038/s41590-023-01569-z]
- Lin A, Qi C, Wei T, Li M, Cheng Q, et al. CAMOIP: a web server for comprehensive analysis on multi-omics of immunotherapy in pan-cancer. Brief Bioinform 2022;23(3):bbac129. [PMID: 35395670 DOI: 10.1093/bib/bbac129]
- Gu T, Jiang A, Zhou C, Lin A, Cheng Q, et al. Adverse reactions associated with immune checkpoint inhibitors and bevacizumab: a pharmacovigilance analysis. Int J Cancer 2023;152(3):480-95. [PMID: 36274626 DOI: 10.1002/ijc.34332]
- Zhou C, Peng S, Lin A, Jiang A, Peng Y, Gu T, et al. Psychiatric disorders associated with immune checkpoint inhibitors: a pharmacovigilance analysis of the FDA Adverse Event Reporting System (FAERS) database. EClinicalMedicine 2023;59:101967. [PMID: 37131541 DOI: 10.1016/j.eclinm.2023.101967]
- Lin A, Zheng K, Jiang A, Huang X, Wang Q, et al. The evolving landscape of immunotoxicity: charting mechanisms and future strategies for immune checkpoint inhibitor adverse events. Med Res 2025;1(3):322-58. [DOI: 10.1002/mdr2.70019]
- Frey C, Etminan M. Adverse events of PD-1, PD-L1, CTLA-4, and LAG-3 immune checkpoint inhibitors: an analysis of the FDA adverse events database. Antibodies (Basel) Switz 2024;13(3):59. [PMID: 39051335 DOI: 10.3390/antib13030059]
- Regev A, Avigan MI, Kiazand A, Vierling JM, Lewis JH, et al. Best practices for detection, assessment and management of suspected immune-mediated liver injury caused by immune checkpoint inhibitors during drug development. J Autoimmun 2020;114:102514. [PMID: 32768244 DOI: 10.1016/j.jaut.2020.102514]
- Kataoka S, Moriguchi M, Okishio S, Takahashi A, Okuda K, et al. Re-administration of nivolumab after immune checkpoint inhibitor-induced cholangitis: the first reported case. Clin J Gastroenterol 2022;15(2):467-74. [PMID: 35201598 DOI: 10.1007/s12328-022-01590-z]
- Autio KA, Boni V, Humphrey RW, Naing A. Probody therapeutics: an emerging class of therapies designed to enhance on-target effects with reduced off-tumor toxicity for use in immuno-oncology. Clin Cancer Res 2020;26(5):984-9. [PMID: 31601568 DOI: 10.1158/1078-0432.CCR-19-1457]
- Bhardwaj M, Chiu MN, Pilkhwal Sah S. Adverse cutaneous toxicities by PD-1/PD-L1 immune checkpoint inhibitors: pathogenesis, treatment, and surveillance. Cutan Ocul Toxicol 2022;41(1):73-90. [PMID: 35107396 DOI: 10.1080/15569527.2022.2034842]
- Wang Y, Zhou S, Yang F, Qi X, Wang X, et al. Treatment-related adverse events of PD-1 and PD-L1 inhibitors in clinical trials: a systematic review and meta-analysis. JAMA Oncol 2019;5(7):1008-19.
- Riveiro-Barciela M, Carballal S, Díaz-González Á, Mañosa M, Gallego-Plazas J, et al. Management of liver and gastrointestinal toxicity induced by immune checkpoint inhibitors: position statement of the AEEH-AEG-SEPD-SEOM-GETECCU. Gastroenterol Hepatol 2024;47(4):401-32. [PMID: 38228461 DOI: 10.1016/j.gastrohep.2023.10.009]
- Tan B, Li Y, Xu Y, Chen M, Wang M, et al. Recognition and management of the gastrointestinal and hepatic immune-related adverse events. Asia Pac J Clin Oncol 2020;16(3):95-102. [PMID: 32077625 DOI: 10.1111/ajco.13317]
- Miao K, Zhang L. Incidence rate and treatment strategy of immune checkpoint inhibitor mediated hepatotoxicity: a systematic review. Cancer Pathog Ther 2023;1(1):46-55. [PMID: 38328612 DOI: 10.1016/j.cpt.2022.11.003]
- Lin A, Jiang A, Huang L, Li Y, Zhang C, et al. From chaos to order: optimizing fecal microbiota transplantation for enhanced immune checkpoint inhibitors efficacy. Gut Microbes 2025;17(1):2452277. [PMID: 39826104 DOI: 10.1080/19490976.2025.2452277]
- Chennamadhavuni A, Abushahin L, Jin N, Presley CJ, Manne A. Risk factors and biomarkers for immune-related adverse events: a practical guide to identifying high-risk patients and rechallenging immune checkpoint inhibitors. Front Immunol 2022;13:779691. [PMID: 35558065 DOI: 10.3389/fimmu.2022.779691]
- Auch LAM, Sieber C, Lehnick D, Hug BL. Adverse drug events of immune checkpoint inhibitors – a retrospective, descriptive real-world data analysis. BMC Cancer 2025;25(1):1303. [PMID: 40790180 DOI: 10.1186/s12885-025-14733-5]
- Nielsen DL, Juhl CB, Chen IM, Kellermann L, Nielsen OH. Immune checkpoint inhibitor-induced diarrhea and colitis: incidence and management. A systematic review and meta-analysis. Cancer Treat Rev 2022;109:102440. [PMID: 35917654 DOI: 10.1016/j.ctrv.2022.102440]
- Gupta A, De Felice KM, Loftus EV Jr, Khanna S. Systematic review: colitis associated with anti-CTLA-4 therapy. Aliment Pharmacol Ther 2015;42(4):406-17. [PMID: 26079306 DOI: 10.1111/apt.13281]
- Tian Y, Abu-Sbeih H, Wang Y. Immune checkpoint inhibitors-induced colitis. Adv Exp Med Biol 2018;995:151-7. [PMID: 30539510 DOI: 10.1007/978-3-030-02505-2_7]
- Tang L, Wang J, Lin N, Zhou Y, He W, et al. Immune checkpoint inhibitor-associated colitis: from mechanism to management. Front Immunol 2021;12:800879. [PMID: 34992611 DOI: 10.3389/fimmu.2021.800879]
- Weber JS, Kähler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol 2012;30(21):2691-7. [PMID: 22614989 DOI: 10.1200/JCO.2012.41.6750]
- Wang DY, Ye F, Zhao S, Johnson DB. Incidence of immune checkpoint inhibitor-related colitis in solid tumor patients: a systematic review and meta-analysis. Oncoimmunology 2017;6(10):e1344805. [PMID: 29123955 DOI: 10.1080/2162402X.2017.1344805]
- Zoghbi M, Burk KJ, Haroun E, Saade M, Carreras MTC. Immune checkpoint inhibitor-induced diarrhea and colitis: an overview. Support Care Cancer 2024;32(10):680. [PMID: 39311981 DOI: 10.1007/s00520-024-08889-2]
- Yoshino K, Nakayama T, Ito A, Sato E, Kitano S. Severe colitis after PD-1 blockade with nivolumab in advanced melanoma patients: potential role of Th1-dominant immune response in immune-related adverse events: two case reports. BMC Cancer 2019;19(1):1019. [PMID: 31664934 DOI: 10.1186/s12885-019-6138-7]
- Ni CX, Zhao Y, Qian H, Fu H, Yan YY, et al. Long survival in a pancreatic carcinoma patient with multi-organ toxicities after sintilimab treatment: a case report. Front Pharmacol 2023;14:1121122. [PMID: 36744247 DOI: 10.3389/fphar.2023.1121122]
- Dougan M. Checkpoint blockade toxicity and immune homeostasis in the gastrointestinal tract. Front Immunol 2017;8:1547. [PMID: 29230210 DOI: 10.3389/fimmu.2017.01547]
- Soularue E, Lepage P, Colombel JF, Coutzac C, Faleck D, et al. Enterocolitis due to immune checkpoint inhibitors: a systematic review. Gut 2018;67(11):2056-67. [PMID: 30131322 DOI: 10.1136/gutjnl-2018-316948]
- Som A, Mandaliya R, Alsaadi D, Farshidpour M, Charabaty A, et al. Immune checkpoint inhibitor-induced colitis: a comprehensive review. World J Clin Cases 2019;7(4):405-18. [PMID: 30842952 DOI: 10.12998/wjcc.v7.i4.405]
- Benson AB 3rd, Ajani JA, Catalano RB, Engelking C, Kornblau SM, et al. Recommended guidelines for the treatment of cancer treatment-induced diarrhea. J Clin Oncol 2004;22(14):2918-26. [PMID: 15254061 DOI: 10.1200/JCO.2004.04.132]
- Kubo T, Hirohashi Y, Keira Y, Akimoto M, Ikeda T, et al. Identification of characteristic subepithelial surface granulomatosis in immune-related adverse event-associated enterocolitis. Cancer Sci 2021;112(3):1320-5. [PMID: 33459466 DOI: 10.1111/cas.14773]
- Dougan M, Blidner AG, Choi J, Cooksley T, Glezerman I, et al. Multinational Association of Supportive Care in Cancer (MASCC) 2020 clinical practice recommendations for the management of severe gastrointestinal and hepatic toxicities from checkpoint inhibitors. Support Care Cancer 2020;28(12):6129-43. [PMID: 32856210 DOI: 10.1007/s00520-020-05707-3]
- Jiang Y, Li R, Li X, Zhang N. Risk factors of immune-mediated hepatotoxicity induced by immune checkpoint inhibitors in cancer patients: a systematic review and meta-analysis. Curr Oncol 2024;31(11):7129-43. [PMID: 39590156 DOI: 10.3390/curroncol31110525]
- Parlati L, Vallet-Pichard A, Batista R, Hernvann A, Sogni P, et al. Incidence of grade 3-4 liver injury under immune checkpoints inhibitors: a retrospective study. J Hepatol 2018;69(6):1396-7. [PMID: 30292476 DOI: 10.1016/j.jhep.2018.08.014]
- Imoto K, Kohjima M, Hioki T, Kurashige T, Kurokawa M, et al. Clinical features of liver injury induced by immune checkpoint inhibitors in Japanese patients. Can J Gastroenterol Hepatol 2019;2019:6391712. [PMID: 31929981 DOI: 10.1155/2019/6391712]
- Remash D, Prince DS, McKenzie C, Strasser SI, Kao S, et al. Immune checkpoint inhibitor-related hepatotoxicity: a review. World J Gastroenterol 2021;27(32):5376-91. [PMID: 34539139 DOI: 10.3748/wjg.v27.i32.5376]
- Cui TM, Liu Y, Wang JB, Liu LX. Adverse effects of immune-checkpoint inhibitors in hepatocellular carcinoma. OncoTargets Ther 2020;13:11725-40. [PMID: 33235462 DOI: 10.2147/OTT.S279858]
- Coukos A, Vionnet J, Obeid M, Bouchaab H, Peters S, et al. Systematic comparison with autoimmune liver disease identifies specific histological features of immune checkpoint inhibitor-related adverse events. J Immunother Cancer 2022;10(10):e005635. [PMID: 36283734 DOI: 10.1136/jitc-2022-005635]
- Kawakami H, Tanizaki J, Tanaka K, Haratani K, Hayashi H, et al. Imaging and clinicopathological features of nivolumab-related cholangitis in patients with non-small cell lung cancer. Invest New Drugs 2017;35(4):529-36. [PMID: 28317087 DOI: 10.1007/s10637-017-0453-0]
- Onoyama T, Takeda Y, Yamashita T, Hamamoto W, Sakamoto Y, et al. Programmed cell death-1 inhibitor-related sclerosing cholangitis: a systematic review. World J Gastroenterol 2020;26(3):353-65. [PMID: 31988594 DOI: 10.3748/wjg.v26.i3.353]
- Nakano R, Shiomi H, Fujiwara A, Yoshihara K, Yoshioka R, et al. Clinical characteristics of ICI-related pancreatitis and cholangitis including radiographic and endoscopic findings. Healthcare (Basel) 2022;10(5):763. [PMID: 35627900 DOI: 10.3390/healthcare10050763]
- Tanaka T, Sakai A, Shiomi H, Masuda A, Kobayashi T, et al. An autopsy case of severe acute pancreatitis induced by administration of pazopanib following nivolumab. Pancreatology 2021;21(1):21-4. [PMID: 33184007 DOI: 10.1016/j.pan.2020.11.002]
- Ichiki T, Hori Y, Tsuji Y. Two types of immune checkpoint inhibitor-related pancreatitis. Intern Med Tokyo Jpn 2022;61(10):1477-8. [PMID: 34744111 DOI: 10.2169/internalmedicine.8452-21]
- Yazaki T, Moriyama I, Tobita H, Sonoyama H, Okimoto E, et al. The simultaneous onset of pancreatitis and colitis as immune-related adverse events in a patient receiving nivolumab treatment for renal cell carcinoma. Intern Med 2022;61(10):1485-90. [PMID: 34744104 DOI: 10.2169/internalmedicine.7911-21]
- Ueno M, Tsuji Y, Yokoyama T, Koyama T, Uenishi Y, et al. Fatal immune checkpoint inhibitor-related pancreatitis. Intern Med 2021;60(24):3905-11. [PMID: 34121010 DOI: 10.2169/internalmedicine.7366-21]
- Di Giacomo AM, Danielli R, Guidoboni M, Calabrò L, Carlucci D, et al. Therapeutic efficacy of ipilimumab, an anti-CTLA-4 monoclonal antibody, in patients with metastatic melanoma unresponsive to prior systemic treatments: clinical and immunological evidence from three patient cases. Cancer Immunol Immunother 2009;58(8):1297-306. [PMID: 19139884 DOI: 10.1007/s00262-008-0642-y]
- Abu-Sbeih H, Tang T, Lu Y, Thirumurthi S, Altan M, et al. Clinical characteristics and outcomes of immune checkpoint inhibitor-induced pancreatic injury. J Immunother Cancer 2019;7(1):31. [PMID: 30728076 DOI: 10.1186/s40425-019-0502-7]
- Xu Y, Wen N, Sonis ST, Villa A. Oral side effects of immune checkpoint inhibitor therapy (ICIT): an analysis of 4683 patients receiving ICIT for malignancies at Massachusetts General Hospital, Brigham & Women’s Hospital, and the Dana-Farber Cancer Institute, 2011 to 2019. Cancer 2021;127(11):1796-804. [PMID: 33595843 DOI: 10.1002/cncr.33436]
- Asan MF, Castelino RL, Babu SG, Rao K, Pandita V. Oral immune-related adverse events – current concepts and their management. Asia Pac J Oncol Nurs 2021;8(6):604-9. [PMID: 34790844 DOI: 10.4103/apjon.apjon-2136]
- Raber-Durlacher JE, Elad S, Barasch A. Oral mucositis. Oral Oncol 2010;46(6):452-6. [PMID: 20403721 DOI: 10.1016/j.oraloncology.2010.03.012]
- Xu T, Liu Y, Lu X, Liang J. Toxicity profile of combined immune checkpoint inhibitors and thoracic radiotherapy in esophageal cancer: a meta-analysis and systematic review. Front Immunol 2022;13:1039020. [PMID: 36439117 DOI: 10.3389/fimmu.2022.1039020]
- Shatila M, Zhang HC, Shirwaikar Thomas A, Machado AP, Naz S, et al. Systematic review of immune checkpoint inhibitor-related gastrointestinal, hepatobiliary, and pancreatic adverse events. J Immunother Cancer 2024;12(11):e009742. [PMID: 39542654 DOI: 10.1136/jitc-2024-009742]
- Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 2000;192(7):1027-34. [PMID: 39542654 DOI: 10.1136/jitc-2024-009742]
- Brunet JF, Denizot F, Luciani MF, Roux-Dosseto M, Suzan M, et al. A new member of the immunoglobulin superfamily–CTLA-4. Nature 1987;328(6127):267-70. [PMID: 3496540 DOI: 10.1038/328267a0]
- Robert L, Tsoi J, Wang X, Emerson R, Homet B, et al. CTLA4 blockade broadens the peripheral T-cell receptor repertoire. Clin Cancer Res 2014;20(9):2424-32. [PMID: 24583799 DOI: 10.1158/1078-0432.CCR-13-2648]
- Sullivan RJ, Weber JS. Immune-related toxicities of checkpoint inhibitors: mechanisms and mitigation strategies. Nat Rev Drug Discov 2022;21(7):495-508. [PMID: 34316029 DOI: 10.1038/s41573-021-00259-5]
- Li Y, Zhang C, Jiang A, Lin A, Liu Z, et al. Potential anti-tumor effects of regulatory T cells in the tumor microenvironment: a review. J Transl Med 2024;22(1):293. [PMID: 38509593 DOI: 10.1186/s12967-024-05104-y]
- Wing JB, Tanaka A, Sakaguchi S. Human FOXP3+ regulatory T cell heterogeneity and function in autoimmunity and cancer. Immunity 2019;50(2):302-16. [PMID: 30784578 DOI: 10.1016/j.immuni.2019.01.020]
- Gupta T, Antanaviciute A, Hyun-Jung Lee C, Ottakandathil Babu R, Aulicino A, et al. Tracking in situ checkpoint inhibitor-bound target T cells in patients with checkpoint-induced colitis. Cancer Cell 2024;42(5):797-814.e15. [PMID: 38744246 DOI: 10.1016/j.ccell.2024.04.010]
- Peng S, Lin A, Jiang A, Zhang C, Zhang J, et al. CTLs heterogeneity and plasticity: implications for cancer immunotherapy. Mol Cancer 2024;23(1):58. [PMID: 38515134 DOI: 10.1186/s12943-024-01972-6]
- Luoma AM, Suo S, Williams HL, Sharova T, Sullivan K, et al. Molecular pathways of colon inflammation induced by cancer immunotherapy. Cell 2020;182(3):655-71.e22. [PMID: 32603654 DOI: 10.1016/j.cell.2020.06.001]
- Bamias G, Delladetsima I, Perdiki M, Siakavellas SI, Goukos D, et al. Immunological characteristics of colitis associated with anti-CTLA-4 antibody therapy. Cancer Invest 2017;35(7):443-55. [PMID: 28548891 DOI: 10.1080/07357907.2017.1324032]
- Lee DW, Gardner R, Porter DL, Louis CU, Ahmed N, et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood 2014;124(2):188-95. [PMID: 24876563 DOI: 10.1182/blood-2014-05-552729]
- Jarczak D, Nierhaus A. Cytokine storm-definition, causes, and implications. Int J Mol Sci 2022;23(19):11740. [PMID: 36233040 DOI: 10.3390/ijms231911740]
- Ceschi A, Noseda R, Palin K, Verhamme K. Immune checkpoint inhibitor-related cytokine release syndrome: analysis of WHO global pharmacovigilance database. Front Pharmacol 2020;11:557. [PMID: 32425791 DOI: 10.3389/fphar.2020.00557]
- He D, Xu Y, Liu M, Cui L. The inflammatory puzzle: piecing together the links between neuroinflammation and amyotrophic lateral sclerosis. Aging Dis 2024;15(1):96-114. [PMID: 37307819 DOI: 10.14336/AD.2023.0519]
- Jones BE, Maerz MD, Buckner JH. IL-6: a cytokine at the crossroads of autoimmunity. Curr Opin Immunol 2018;55:9-14. [PMID: 30248523 DOI: 10.1016/j.coi.2018.09.002]
- Meng Y, Lv Y, Shen M, Yu W, Liu Y, et al. Establishment of an animal model of immune-related adverse events induced by immune checkpoint inhibitors. Cancer Med 2024;13(14):e70011. [PMID: 39001676 DOI: 10.1002/cam4.70011]
- Endo Y, Winarski KL, Sajib MS, Ju A, Wu WJ. Atezolizumab induces necroptosis and contributes to hepatotoxicity of human hepatocytes. Int J Mol Sci 2023;24(14):11694. [PMID: 37511454 DOI: 10.3390/ijms241411694]
- Yang H, Yao Z, Zhou X, Zhang W, Zhang X, et al. Immune-related adverse events of checkpoint inhibitors: insights into immunological dysregulation. Clin Immunol 2020;213:108377. [PMID: 32135278 DOI: 10.1016/j.clim.2020.108377]
- Reynoso ED, Elpek KG, Francisco L, Bronson R, Bellemare-Pelletier A, et al. Intestinal tolerance is converted to autoimmune enteritis upon PD-1 ligand blockade. J Immunol 2009;182(4):2102-12. [PMID: 19201863 DOI: 10.4049/jimmunol.0802769]
- Michot JM, Bigenwald C, Champiat S, Collins M, Carbonnel F, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer 2016;54:139-48. [PMID: 26765102 DOI: 10.1016/j.ejca.2015.11.016]
- Lin A, Xiong M, Jiang A, Huang L, Wong HZH, et al. The microbiome in cancer. iMeta 2025;4(5):e70070. [PMID: 41112042 DOI: 10.1002/imt2.70070]
- Li D, Wu R, Yu Q, Tuo Z, Wang J, et al. Microbiota and urinary tumor immunity: mechanisms, therapeutic implications, and future perspectives. Chin J Cancer Res 2024;36(6):596-615. [PMID: 39802902 DOI: 10.21147/j.issn.1000-9604.2024.06.03]
- Zhang M, Xie F, Zhou F. The impact of intestinal microbiota-derived metabolites on cancer and their potential application in tumor immunotherapy. MedComm – Futur Med 2024;3(3):e92. [DOI: 10.1002/mef2.92]
- Zhao B, Zhou B, Li Q, Su C, Ma J, et al. Harnessing the gut microbiome to enhance cancer immunotherapy: current advances and future directions in microbiota-based therapeutic strategies. MedComm – Futur Med 2024;3(4):e70006. [DOI: 10.1002/mef2.70006]
- Huang L, Li Y, Zhang C, Jiang A, Zhu L, et al. Microbiome meets immunotherapy: unlocking the hidden predictors of immune checkpoint inhibitors. NPJ Biofilms Microbiomes 2025;11(1):180. [PMID: 40897718 DOI: 10.1038/s41522-025-00819-2]
- Lin A, Huang L, Jiang A, Zhu L, Mou W, et al. Microbiota boost immunotherapy? A meta-analysis dives into fecal microbiota transplantation and immune checkpoint inhibitors. BMC Med 2025;23(1):341. [DOI: 10.1186/s12916-025-04183-y]
- Situ Y, Zhang P, Zhang C, Jiang A, Zhang N, et al. The metabolic dialogue between intratumoural microbes and cancer: implications for immunotherapy. EBioMedicine 2025;115:105708. [PMID: 40267755 DOI: 10.1016/j.ebiom.2025.105708]
- Wang Y, Wiesnoski DH, Helmink BA, Gopalakrishnan V, Choi K, et al. Fecal microbiota transplantation for refractory immune checkpoint inhibitor-associated colitis. Nat Med 2018;24(12):1804-8. [PMID: 30420754 DOI: 10.1038/s41591-018-0238-9]
- Wang T, Zheng N, Luo Q, Jiang L, He B, et al. Probiotics Lactobacillus reuteri abrogates immune checkpoint blockade-associated colitis by inhibiting group 3 innate lymphoid cells. Front Immunol 2019;10:1235. [PMID: 31214189 DOI: 10.3389/fimmu.2019.01235]
- Sun S, Luo L, Liang W, Yin Q, Guo J, et al. Bifidobacterium alters the gut microbiota and modulates the functional metabolism of T regulatory cells in the context of immune checkpoint blockade. Proc Natl Acad Sci U S A 2020;117(44):27509-15. [PMID: 33077598 DOI: 10.1073/pnas.1921223117]
- Knox NC, Forbes JD, Peterson CL, Van Domselaar G, Bernstein CN. The gut microbiome in inflammatory bowel disease: lessons learned from other immune-mediated inflammatory diseases. Am J Gastroenterol 2019;114(7):1051-70. [PMID: 31232832 DOI: 10.14309/ajg.0000000000000305]
- Zheng C, Huang S, Lin M, Hong B, Ni R, et al. Hepatotoxicity of immune checkpoint inhibitors: what is currently known. Hepatol Commun 2023;7(3):e0063. [PMID: 36802366 DOI: 10.1097/HC9.0000000000000063]
- Schneider BJ, Naidoo J, Santomasso BD, Lacchetti C, Adkins S, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO Guideline Update. J Clin Oncol 2021;39(36):4073-126. [PMID: 34724392 DOI: 10.1200/JCO.21.01440]
- Yamamoto N, Nakanishi Y, Gemma A, Nakagawa K, Sakamoto T, et al. Real-world safety of nivolumab in patients with non-small-cell lung cancer in Japan: postmarketing surveillance. Cancer Sci 2021;112(11):4692-701. [PMID: 34431585 DOI: 10.1111/cas.15117]
- Hao Y, Lin G, Xiang J, Wang W, Xu C, et al. Analysis of the efficacy and safety of immunotherapy in advanced thymoma patients. Cancer Med 2023;12(5):5649-55. [PMID: 36394097 DOI: 10.1002/cam4.5357]
- Ge X, Jiang W, Li H, Wu Y, Li X, et al. Immune-related adverse events and outcomes among pan-cancer patients receiving immune checkpoint inhibitors: a monocentric real-world observational study. Cancer Med 2023;12(18):18491-502. [PMID: 37564011 DOI: 10.1002/cam4.6449]
- Denaro N, Garrone O, Occelli M, Fea E, Granetto C, et al. Unusual fatal outcome following administration of a combination of anti-PD1 and anti-CTLA4 in metastatic renal cell carcinoma: liver toxicity case report and a literature review. Eur J Case Rep Intern Med 2021;8(7):002639. [PMID: 34268267 DOI: 10.12890/2021_002639]
- Biewenga M, van der Kooij MK, Wouters MWJM, Aarts MJB, van den Berkmortel FWPJ, et al. Checkpoint inhibitor induced hepatitis and the relation with liver metastasis and outcome in advanced melanoma patients. Hepatol Int 2021;15(2):510-9. [PMID: 33634373 DOI: 10.1007/s12072-021-10151-4]
- Yang W, Li X, He J, Xuan Q, Si H, et al. Efficacy and safety of immune checkpoint inhibitors combined with anti-VEGF therapy in the treatment of unresectable or advanced liver cancer: a systematic review. Immunopharmacol Immunotoxicol 2023;45(6):770-9. [PMID: 37228221 DOI: 10.1080/08923973.2023.2215404]
- Danlos FX, Voisin AL, Dyevre V, Michot JM, Routier E, et al. Safety and efficacy of anti-programmed death 1 antibodies in patients with cancer and pre-existing autoimmune or inflammatory disease. Eur J Cancer 2018;91:21-9. [PMID: 29331748 DOI: 10.1016/j.ejca.2017.12.008]
- Honma Y, Shibata M, Gohda T, Matsumiya H, Kumamoto K, et al. Rapid progression of liver fibrosis induced by acute liver injury due to immune-related adverse events of atezolizumab. Intern Med 2021;60(12):1847-53. [PMID: 33456046 DOI: 10.2169/internalmedicine.6535-20]
- De Martin E, Michot JM, Rosmorduc O, Guettier C, Samuel D. Liver toxicity as a limiting factor to the increasing use of immune checkpoint inhibitors. JHEP Rep 2020;2(6):100170. [PMID: 33205034 DOI: 10.1016/j.jhepr.2020.100170]
- El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017;389(10088):2492-502. [PMID: 28434648 DOI: 10.1016/S0140-6736(17)31046-2]
- Mok K, Wu C, Chan S, Wong G, Wong VWS, et al. Clinical management of gastrointestinal and liver toxicities of immune checkpoint inhibitors. Clin Colorectal Cancer 2024;23(1):4-13. [PMID: 38172003 DOI: 10.1016/j.clcc.2023.12.003]
- Zen Y, Yeh MM. Hepatotoxicity of immune checkpoint inhibitors: a histology study of seven cases in comparison with autoimmune hepatitis and idiosyncratic drug-induced liver injury. Mod Pathol 2018;31(6):965-73. [PMID: 29403081 DOI: 10.1038/s41379-018-0013-y]
- Zen Y, Yeh MM. Checkpoint inhibitor-induced liver injury: a novel form of liver disease emerging in the era of cancer immunotherapy. Semin Diagn Pathol 2019;36(6):434-40. [PMID: 31358424 DOI: 10.1053/j.semdp.2019.07.009]
- Garcia-Neuer M, Marmarelis ME, Jangi SR, Luke JJ, Ibrahim N, et al. Diagnostic comparison of CT scans and colonoscopy for immune-related colitis in ipilimumab-treated advanced melanoma patients. Cancer Immunol Res 2017;5(4):286-91. [PMID: 28373217 DOI: 10.1158/2326-6066.CIR-16-0302]
- Goel N, Levine MD, Chambers LM, Nagel CI. Gastritis as an immunotherapy-related toxicity in the treatment of endometrial cancer: a case report. Gynecol Oncol Rep 2023;47:101174. [PMID: 37102082 DOI: 10.1016/j.gore.2023.101174]
- Haryal A, Townsend MJ, Baskaran V, Srivoleti P, Giobbie-Hurder A, et al. Immune checkpoint inhibitor gastritis is often associated with concomitant enterocolitis, which impacts the clinical course. Cancer 2023;129(3):367-75. [PMID: 36377339 DOI: 10.1002/cncr.34543]
- Nakai M, Kai Y, Suzuki K, Matsuda M, Kikukawa S, et al. A case of perforated immune-related colitis complicated by cytomegalovirus infection during treatment of immune-related adverse effect in lung cancer immunotherapy. Respir Med Case Rep 2023;41:101794. [PMID: 36583058 DOI: 10.1016/j.rmcr.2022.101794]
- Karamchandani DM, Chetty R. Immune checkpoint inhibitor-induced gastrointestinal and hepatic injury: pathologists’ perspective. J Clin Pathol 2018;71(8):665-71. [PMID: 29703758 DOI: 10.1136/jclinpath-2018-205143]
- Jiang C, Tang W, Yang X, Li H. Immune checkpoint inhibitor-related pancreatitis: what is known and what is not. Open Med 2023;18(1):20230713. [PMID: 37273919 DOI: 10.1515/med-2023-0713]
- de La Rochefoucauld J, Noël N, Lambotte O. Management of immune-related adverse events associated with immune checkpoint inhibitors in cancer patients: a patient-centred approach. Intern Emerg Med 2020;15(4):587-98. [PMID: 32144552 DOI: 10.1007/s11739-020-02295-2]
- Weber JS, Hodi FS, Wolchok JD, Topalian SL, Schadendorf D, et al. Safety profile of nivolumab monotherapy: a pooled analysis of patients with advanced melanoma. J Clin Oncol 2017;35(7):785-92. [PMID: 28068177 DOI: 10.1200/JCO.2015.66.1389]
- Amin A, DePril V, Hamid O, Wolchock J, Maio M, et al. Evaluation of the effect of systemic corticosteroids for the treatment of immune-related adverse events (irAEs) on the development or maintenance of ipilimumab clinical activity. J Clin Oncol 2009;27(15 suppl):9037. [DOI: 10.1200/jco.2009.27.15_suppl.9037]
- Downey SG, Klapper JA, Smith FO, Yang JC, Sherry RM, et al. Prognostic factors related to clinical response in patients with metastatic melanoma treated by CTL-associated antigen-4 blockade. Clin Cancer Res 2007;13(22 Pt 1):6681-8. [PMID: 17982122 DOI: 10.1158/1078-0432.CCR-07-0187]
- Harmankaya K, Erasim C, Koelblinger C, Ibrahim R, Hoos A, et al. Continuous systemic corticosteroids do not affect the ongoing regression of metastatic melanoma for more than two years following ipilimumab therapy. Med Oncol 2011;28(4):1140-4. [PMID: 20593249 DOI: 10.1007/s12032-010-9606-0]
- Liu SJ, Yan LJ, Wang HC, Ding ZN, Liu H, et al. Safety, efficacy, and survival outcomes of immune checkpoint inhibitors rechallenge in patients with cancer: a systematic review and meta-analysis. Oncologist 2024;29(11):e1425-34. [PMID: 38940446 DOI: 10.1093/oncolo/oyae134]
- Ueno K, Suzuki K, Saito K, Shinohara H, Miura A. Nivolumab rechallenge after immune-related adverse events in patients with unresectable advanced or recurrent esophageal cancer. In Vivo 2024;38(2):794-9. [PMID: 38418136 DOI: 10.21873/invivo.13503]
- Socinski MA, Jotte RM, Cappuzzo F, Nishio M, Mok TSK, et al. Association of immune-related adverse events with efficacy of atezolizumab in patients with non-small cell lung cancer: pooled analyses of the phase 3 IMpower130, IMpower132, and IMpower150 randomized clinical trials. JAMA Oncol 2023;9(4):527-35. [PMID: 36795388 DOI: 10.1001/jamaoncol.2022.7711]
- Nasca V, Barretta F, Corti F, Lonardi S, Niger M, et al. Association of immune-related adverse events with the outcomes of immune checkpoint inhibitors in patients with dMMR/MSI-H metastatic colorectal cancer. J Immunother Cancer 2023;11(1):e005493. [PMID: 36593068 DOI: 10.1136/jitc-2022-005493]
- Fu J, Li WZ, McGrath NA, Lai CW, Brar G, et al. Immune checkpoint inhibitor associated hepatotoxicity in primary liver cancer versus other cancers: a systematic review and meta-analysis. Front Oncol 2021;11:650292. [PMID: 33968750 DOI: 10.3389/fonc.2021.650292]
- Gao Y, Xu P, Sun D, Jiang Y, Lin XL, et al. Faecalibacterium prausnitzii abrogates intestinal toxicity and promotes tumor immunity to increase the efficacy of dual CTLA4 and PD-1 checkpoint blockade. Cancer Res 2023;83(22):3710-25. [PMID: 37602831 DOI: 10.1158/0008-5472.CAN-23-0605]
- Kawano M, Yano Y, Yamamoto A, Yasutomi E, Inoue Y, et al. Risk factors for immune checkpoint inhibitor-induced liver injury and the significance of liver biopsy. Diagnostics (Basel) 2024;14(8):815. [PMID: 38667461 DOI: 10.3390/diagnostics14080815]
- Weissman S, Saleem S, Sharma S, Krupka M, Inayat F, et al. Incidence, mortality, and risk factors of immunotherapy-associated hepatotoxicity: a nationwide hospitalization analysis. Liver Res 2021;5(1):28-32. [PMID: 33828870 DOI: 10.1016/j.livres.2021.01.003]
- Chen J, Lin A, Luo P. Advancing pharmaceutical research: a comprehensive review of cutting-edge tools and technologies. Curr Pharm Anal 2024;21(1):1-19. [DOI: 10.1016/j.cpan.2024.11.001]
- Saleem M, Watson AE, Anwaar A, Jasser AO, Yusuf N. Optimizing immunotherapy: the synergy of immune checkpoint inhibitors with artificial intelligence in melanoma treatment. Biomolecules 2025;15(4):589. [PMID: 40305346 DOI: 10.3390/biom15040589]
- Zitu MM, Gatti-Mays ME, Johnson KC, Zhang S, Shendre A, et al. Detection of patient-level immunotherapy-related adverse events (irAEs) from clinical narratives of electronic health records: a high-sensitivity artificial intelligence model. Pragmatic Obs Res 2024;15:243-52. [PMID: 39720010 DOI: 10.2147/POR.S468253]
- Yokode M, Shiokawa M, Kawakami H, Kuwada T, Nishikawa Y, et al. Anti-integrin αvβ6 autoantibodies are a potential biomarker for ulcerative colitis-like immune checkpoint inhibitor-induced colitis. Br J Cancer 2024;130(9):1552-60. [PMID: 38461170 DOI: 10.1038/s41416-024-02647-1]
- Wang D, Zhao Y, Zeng Y, Hu L, Xu C. Update on immunotherapy-mediated colitis: clinical features, mechanisms, and management. Malig Spectr 2024;1(4):225-42. [DOI: 10.1002/msp2.50]
- Mallardo D, Fordellone M, Bailey M, White A, Simeone E, et al. Gene-expression signature predicts autoimmune toxicity in metastatic melanoma. J Immunother Cancer 2025;13(7):e011315. [PMID: 40633934 DOI: 10.1136/jitc-2024-011315]
- Nobusawa T, Kaneko S, Murakawa M, Tsuchiya J, Miyoshi M, et al. Immune-related adverse events in patients with low baseline serum IL-6 treated with durvalumab plus tremelimumab for hepatocellular carcinoma: a case series. Clin J Gastroenterol 2025;18(5):899-904. [PMID: 40632185 DOI: 10.1007/s12328-025-02176-1]
- Cunningham M, Iafolla M, Kanjanapan Y, Cerocchi O, Butler M, et al. Evaluation of liver enzyme elevations and hepatotoxicity in patients treated with checkpoint inhibitor immunotherapy. PloS One 2021;16(6):e0253070. [PMID: 34115819 DOI: 10.1371/journal.pone.0253070]
- Lee SK, Choi JY, Jung ES, Kwon JH, Jang JW, et al. An immunological perspective on the mechanism of drug induced liver injury: focused on drugs for treatment of hepatocellular carcinoma and liver transplantation. Int J Mol Sci 2023;24(5):5002. [PMID: 36902432 DOI: 10.3390/ijms24055002]
- Mayo Clinic. Probiotics preventing immune checkpoint inhibitor-induced colitis. clinicaltrials.gov; 2025 Aug. Report No.: NCT06508034. Available from: https://clinicaltrials.gov/study/NCT06508034
- Yang Y. Immune checkpoint inhibitor rechallenge in combination with siltuximab prophylaxis for patients who had prior immune-related adverse event (CIRES Trial). clinicaltrials.gov; 2025 Jan. Report No.: NCT06470971. Available from: https://clinicaltrials.gov/study/NCT06470971
- Lozano AX, Chaudhuri AA, Nene A, Bacchiocchi A, Earland N, et al. T cell characteristics associated with toxicity to immune checkpoint blockade in patients with melanoma. Nat Med 2022;28(2):353-62. [PMID: 35027754 DOI: 10.1038/s41591-021-01623-z]
- He JY, Kim YJ, Mennillo E, Rusu I, Bain J, et al. Dysregulation of CD4+ and CD8+ resident memory T, myeloid, and stromal cells in steroid-experienced, checkpoint inhibitor colitis. J Immunother Cancer 2024;12(4):e008628. [PMID: 38642938 DOI: 10.1136/jitc-2023-008628]
- Zhai Y, Liang X, Deng M. Myeloid cells meet CD8+ T cell exhaustion in cancer: what, why and how. Chin J Cancer Res 2024;36(6):616-51. [PMID: 39802897 DOI: 10.21147/j.issn.1000-9604.2024.06.04]
- Dubin K, Callahan MK, Ren B, Khanin R, Viale A, et al. Intestinal microbiome analyses identify melanoma patients at risk for checkpoint-blockade-induced colitis. Nat Commun 2016;7:10391. [PMID: 26837003 DOI: 10.1038/ncomms10391]
- Gronke K, Nguyen M, Fuhrmann H, Santamaria de Souza N, Schumacher J, et al. Translocating gut pathobiont Enterococcus gallinarum induces TH17 and IgG3 anti-RNA-directed autoimmunity in mouse and human. Sci Transl Med 2025;17(784):eadj6294. [PMID: 39908347 DOI: 10.1126/scitranslmed.adj6294]
- Raychaudhuri SP, Raychaudhuri SK. JAK inhibitor: introduction. Indian J Dermatol Venereol Leprol 2023;89(5):688-90. [PMID: 37609754 DOI: 10.25259/IJDVL_8_2023]
- Liu Q, Liu M, Zou Z, Lin J, Zhang N, et al. Tofacitinib for the treatment of immune-related adverse events in cancer immunotherapy: a multi-center observational study. J Transl Med 2024;22(1):803. [PMID: 39210332 DOI: 10.1186/s12967-024-05617-6]
- NYU Langone Health. A phase II study of the interleukin-6 receptor inhibitor sarilumab in combination with ipilimumab, nivolumab and relatlimab in patients with unresectable stage III or stage IV melanoma. clinicaltrials.gov; 2025 Feb. Report No.: NCT05428007. Available from: https://clinicaltrials.gov/study/NCT05428007
- Mehnert JM, Mehmi I, Goldberg JD, Dimitrova M, Arriola P, et al. A phase II study of the interleukin-6 (IL-6) receptor blocking antibody sarilumab (Sari) in combination with ipilimumab (Ipi), nivolumab (Nivo) and relatlimab (Rela) in patients with unresectable stage III or stage IV melanoma. J Clin Oncol 2025;43(16 suppl):9510. [DOI: 10.1200/JCO.2025.43.16_suppl.9510]
- Holmstroem RB, Nielsen OH, Jacobsen S, Riis LB, Theile S, et al. COLAR: open-label clinical study of IL-6 blockade with tocilizumab for the treatment of immune checkpoint inhibitor-induced colitis and arthritis. J Immunother Cancer 2022;10(9):e005111. [PMID: 36096534 DOI: 10.1136/jitc-2022-005111]
- Halsey TM, Thomas AS, Hayase T, Ma W, Abu-Sbeih H, et al. Microbiome alteration via fecal microbiota transplantation is effective for refractory immune checkpoint inhibitor–induced colitis. Sci Transl Med 2023;15(700):eabq4006. [PMID: 37315113 DOI: 10.1126/scitranslmed.abq4006]
- Elkrief A, Waters NR, Smith N, Dai A, Slingerland J, et al. Immune-related colitis is associated with fecal microbial dysbiosis and can be mitigated by fecal microbiota transplantation. Cancer Immunol Res 2024;12(3):308-21. [PMID: 38108398 DOI: 10.1158/2326-6066.CIR-23-0498]
- Wang Y, Varatharajalu K, Shatila M, Campbell MT, Msaouel P, et al. First-line treatment of fecal microbiota transplantation for immune-mediated colitis. J Clin Oncol 2024;42(16 suppl):2660. [DOI: 10.14309/01.ajg.0001030568.48967.db]
- University of Aarhus. FMT in checkpoint inhibitor-mediated diarrhea and colitis (Immunobiome). clinicaltrials.gov; 2024 Jan. Report No.: NCT06206707. Available from: https://clinicaltrials.gov/study/NCT06206707
- Herrera M, Pretelli G, Desai J, Garralda E, Siu LL, et al. Bispecific antibodies: advancing precision oncology. Trends Cancer 2024;10(10):893-919. [PMID: 39214782 DOI: 10.1016/j.trecan.2024.07.002]
- Dahan R, Korman AJ. Fc-optimized checkpoint antibodies for cancer immunotherapy. Trends Mol Med 2025;31(6):501-5. [PMID: 39487064 DOI: 10.1016/j.molmed.2024.10.008]
- Naing A, Thistlethwaite F, De Vries EGE, Eskens FALM, Uboha N, et al. CX-072 (pacmilimab), a Probody ® PD-L1 inhibitor, in advanced or recurrent solid tumors (PROCLAIM-CX-072): an open-label dose-finding and first-in-human study. J Immunother Cancer 2021;9(7):e002447.
- Ahmed LA, Al-Massri KF. Gut microbiota modulation for therapeutic management of various diseases: a new perspective using stem cell therapy. Curr Mol Pharmacol 2023;16(1):43-59. [PMID: 35196976 DOI: 10.2174/1874467215666220222105004]
- Govindan R, Szczesna A, Ahn MJ, Schneider CP, Gonzalez Mella PF, et al. Phase III trial of ipilimumab combined with paclitaxel and carboplatin in advanced squamous non-small-cell lung cancer. J Clin Oncol 2017;35(30):3449-57.
- Baas P, Scherpereel A, Nowak AK, Fujimoto N, Peters S, et al. First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): a multicentre, randomised, open-label, phase 3 trial. Lancet 2021;397(10272):375-86. [PMID: 33485464 DOI: 10.1016/S0140-6736(20)32714-8]
- Hellmann MD, Ciuleanu TE, Pluzanski A, Lee JS, Otterson GA, et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med 2018;378(22):2093-104. [PMID: 29658845 DOI: 10.1056/NEJMoa1801946]
- O’Byrne KJ, Lee KH, Kim SW, Park K, Nishio M, et al. First-line nivolumab + ipilimumab in advanced NSCLC: CheckMate 227 subpopulation analyses in Asian patients. ESMO Open 2022;7(1):100394. [PMID: 35158207 DOI: 10.1016/j.esmoop.2022.100394]
- Hodi FS, Chesney J, Pavlick AC, Robert C, Grossmann KF, et al. Combined nivolumab and ipilimumab versus ipilimumab alone in patients with advanced melanoma: 2-year overall survival outcomes in a multicentre, randomised, controlled, phase 2 trial. Lancet Oncol 2016;17(11):1558-68. [PMID: 27622997 DOI: 10.1016/S1470-2045(16)30366-7]
- Tandon P, Bourassa-Blanchette S, Bishay K, Parlow S, Laurie SA, et al. The risk of diarrhea and colitis in patients with advanced melanoma undergoing immune checkpoint inhibitor therapy: a systematic review and meta-analysis. J Immunother 2018;41(3):101-8. [PMID: 29401166 DOI: 10.1097/CJI.0000000000000213]
- Felip E, Altorki N, Zhou C, Csőszi T, Vynnychenko I, et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB-IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase 3 trial. Lancet 2021;398(10308):1344-57. [PMID: 34555333 DOI: 10.1016/S0140-6736(21)02098-5]
- Pujol JL, Greillier L, Audigier-Valette C, Moro-Sibilot D, Uwer L, et al. A randomized non-comparative phase II study of anti-programmed cell death-ligand 1 atezolizumab or chemotherapy as second-line therapy in patients with small cell lung cancer: results from the IFCT-1603 trial. J Thorac Oncol Off Publ Int Assoc Study Lung Cancer 2019;14(5):903-13. [PMID: 30664989 DOI: 10.1016/j.jtho.2019.01.008]
- Seto T, Nosaki K, Shimokawa M, Toyozawa R, Sugawara S, et al. Phase II study of atezolizumab with bevacizumab for non-squamous non-small cell lung cancer with high PD-L1 expression (@Be Study). J Immunother Cancer 2022;10(2):e004025. [PMID: 35105689 DOI: 10.1136/jitc-2021-004025]
- Ready NE, Ott PA, Hellmann MD, Zugazagoitia J, Hann CL, et al. Nivolumab monotherapy and nivolumab plus ipilimumab in recurrent small cell lung cancer: results from the CheckMate 032 randomized cohort. J Thorac Oncol 2020;15(3):426-35. [PMID: 31629915 DOI: 10.1016/j.jtho.2019.10.004]
- Saowapa S, Polpichai N, Tanariyakul M, Wannaphut C, Wattanachayakul P, et al. Immunotherapy-induced hepatitis in metastatic colorectal cancer: a systematic review and meta-analysis. Proc Bayl Univ Med Cent 2024;37(5):841-50. [PMID: 39165807 DOI: 10.1080/08998280.2024.2374161]
- Koseki M, Nishimura Y, Elias E, Estaris J, Chesta F, et al. Pancreatitis in patients with cancer receiving immune checkpoint inhibitors: a systematic review and meta-analysis. Target Oncol 2024;19(6):867-77. [PMID: 39392549 DOI: 10.1007/s11523-024-01098-1]
- Zhang T, Wang Y, Shi C, Liu X, Lv S, et al. Pancreatic injury following immune checkpoint inhibitors: a systematic review and meta-analysis. Front Pharmacol 2022;13:955701. [PMID: 36133806 DOI: 10.3389/fphar.2022.955701]