Harnessing Cell Membrane-Derived Nanovesicles for Enhanced Nanoprobes in Multimodal Imaging: Progress and Perspectives
1Key Laboratory of Medical Imaging Precision Theranostics and Radiation Protection, College of Hunan Province, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
2Department of Medical Imaging, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
3Institute of Medical Imaging, Hengyang Medical School, University of South China, Hengyang 710127, China
4Key Laboratory of Synthetic and Natural Functional Molecule Chemistry of Ministry of Education, Key Lab of Modern Separation Science in Shaanxi Province, College of Chemistry and Materials Science, Northwest University, Xi’an, Shaanxi, China
5School of Biological Sciences, Nanyang Technological University, Singapore 637551, Singapore
*Correspondence: Meng Du, Changsha Central Hospital, 161 Shaoshan South Road, Changsha, China. E-mail: dumeng_work@126.com, Tel: +86-13570919798; Jianjian Zhang, Key Laboratory of Synthetic and Natural Functional Molecule Chemistry of Ministry of Education, Key Lab of Modern Separation Science in Shaanxi Province, College of Chemistry and Materials Science, Northwest University, Xi’an, Shaanxi 710127, China. E-mail: zhangjj@nwu.edu.cn; Yuguang Mu, School of Biological Sciences, Nanyang Technological University, 60 Nanyang Drive, Singapore 637551, Singapore. E-mail: ygmu@ntu.edu.sg
Received: March 5 2025; Revised: June 19 2025; Accepted: August 14 2025; Published Online: October 17 2025
Cite this paper:
Lei L, Du M, Zhang J et al. Harnessing Cell Membrane-Derived Nanovesicles for Enhanced Nanoprobes in Multimodal Imaging: Progress and Perspectives. BIO Integration 2025; 6: 1–17.
DOI: 10.15212/bioi-2025-0047. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Cell membrane-derived nanovesicles (CMNVs) are natural nanocarriers efficiently encapsulating bioactive molecules and imaging probes. Conventional nanoprobes face challenges, like rapid immune clearance, off-target accumulation, and long-term toxicity. CMNVs overcome these challenges by leveraging inherent biofunctionalization. CMNVs enhance encapsulated nanoprobe targeting, prolong circulation, boost specific tissue accumulation, and improve imaging precision through inherited membrane proteins or minimally disruptive exogenous modifications. CMNVs show significant promise across multiple imaging modalities, including fluorescence, bioluminescence, photoacoustic, and MRI. This review systematically evaluates the limitations of traditional nanoprobes, highlights the unique advantages of CMNVs in enhancing diverse probe performance, discusses current challenges in harnessing CMNVs for imaging, and provides insights for developing future high-precision imaging strategies.
Keywords
Bioluminescence, cell membrane-derived nanovesicles, MRI, nanoprobes, fluorescence, photoacoustic imaging.
Introduction
Nanoprobes, as nanoscale sensors for probing individual living cells, encompass organic and inorganic variants, such as fluorescent dyes, magnetic nanoparticles, quantum dots (QDs), and noble metal nanoparticles [1]. While the minimal serum protein coverage of nanoprobes enhances surface functionalization and cellular uptake, conventional nanoprobes face critical limitations in biomedical imaging [2, 3]. Challenges include rapid clearance by the reticuloendothelial system (RES), accumulation in off-target tissues (e.g., liver and spleen), and resultant low signal-to-noise ratios [SNRs] [4–7]. Moreover, persistent biocompatibility concerns, such as the slow clearance of QDs and the long-term toxicity, remain unresolved despite strategies, like PEGylation or antibody modifications [8–11]. These challenges highlight the pressing need for biocompatible platforms that can evade the RES to propel advances in nanoprobe-based imaging. Significantly, these vesicles function as self-contained imaging platforms, which allows for the efficient encapsulation of reporter molecules and enhances the versatility and effectiveness of imaging applications [e.g., luciferase [12] and tyrosinase [13]] through genetic engineering of parent cells, thus eliminating manual cargo loading.
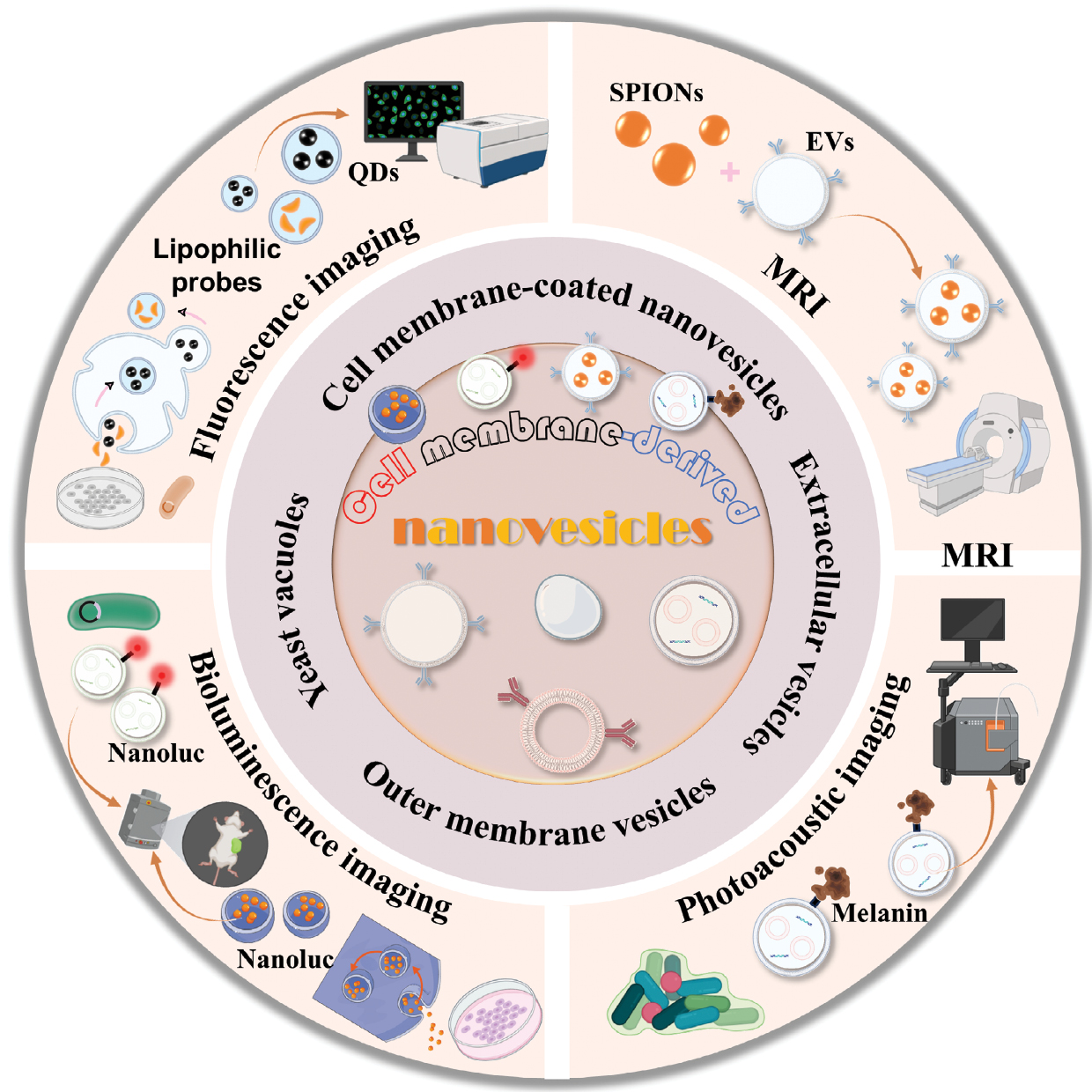
Cell membrane-derived nanovesicles (CMNVs) have emerged as a promising class of natural nanocarriers to overcome these barriers. CMNVs, including cell membrane-coated nanovesicles (CMCNVs), extracellular vesicles (EVs), bacterial outer membrane vesicles (OMVs), and yeast vesicles, serve as natural engineering substrates for nanoprobe design. CMNVs leverage inherent biofunctionalization, which is achieved through inherited membrane proteins or minimally disruptive exogenous modifications, to fundamentally redesign nanoprobe delivery [14–17]. This biomimetic strategy enhances targeting capability, prolongs systemic circulation, boosts specific tissue accumulation, and improves imaging precision across diverse pathologic contexts.
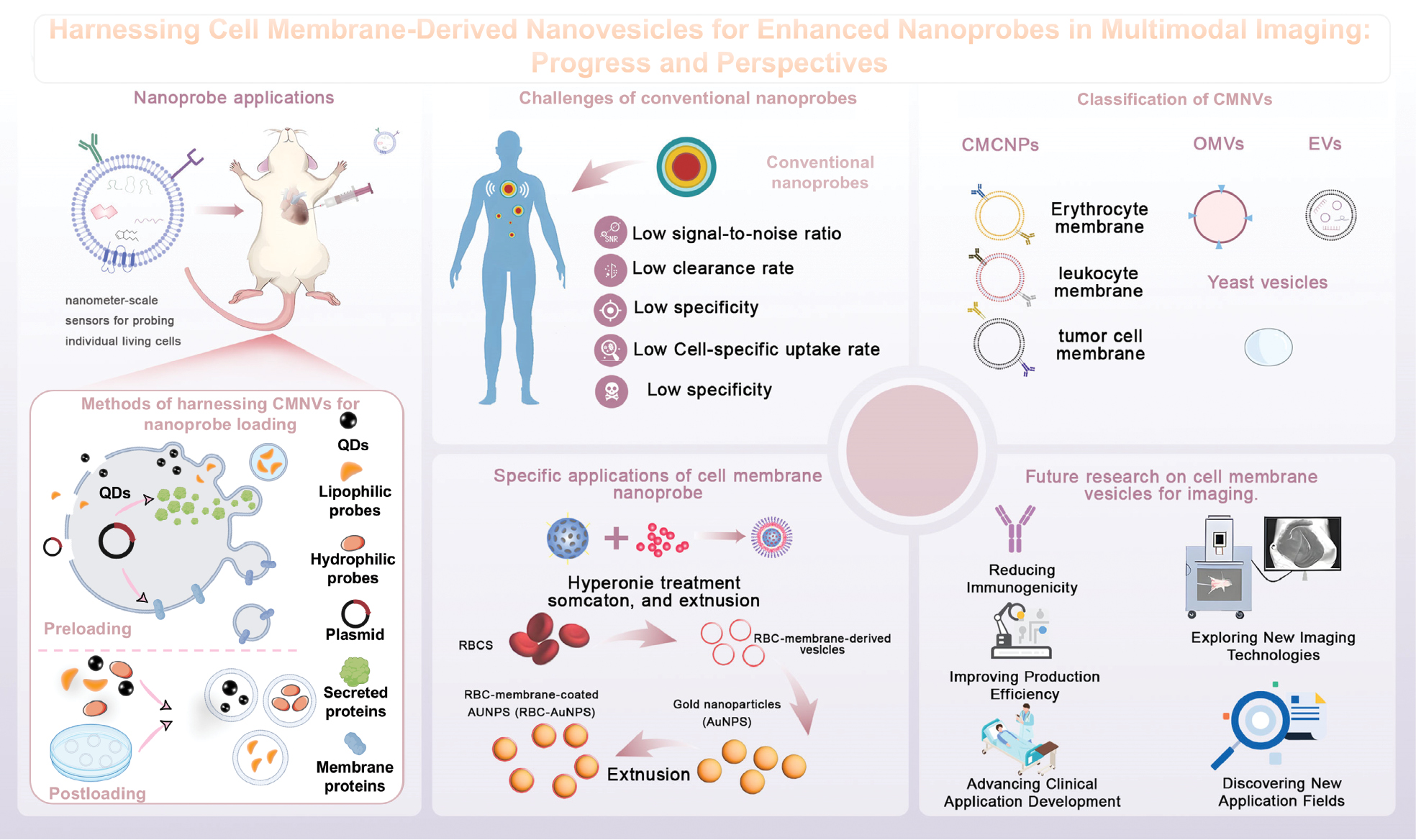
CMNVs demonstrate significant promise as versatile platforms for multiple imaging modalities, most notably fluorescence imaging, bioluminescence imaging, photoacoustic imaging, and magnetic resonance imaging (MRI). The ability of CMNVs to integrate seamlessly with various contrast mechanisms positions CMNVs at the forefront of next-generation diagnostic tools. This review systematically evaluates the fundamental constraints of existing traditional nanoprobes and highlights the unique advantages of harnessing CMNVs for enhanced nanoprobes in multimodal imaging. Furthermore, we critically discuss current challenges in CMNVs-based imaging (spanning scalable production, standardization, and in vivo stability) and provide insights into future translational pathways. By synthesizing recent advances and unresolved questions, this work aims to catalyze the development of high-precision imaging strategies leveraging CMNV nanotechnology (Figure 1).
Figure 1 A summary of harnessing CMNVs in multimodal imaging, showing the design, applications, and future prospects of CMNVs as advanced platforms for enhancing nanoprobes in multimodal imaging.
Challenges of conventional nanoprobes
Despite advantages, like enhanced brightness and stability, the clinical translation of nanoprobes faces the following critical barriers: low SNR; suboptimal clearance rates; and material toxicity. Based on these critical barriers, researchers have conducted numerous investigations.
Low SNR
SNR determines imaging clarity by measuring target-specific signals against background noise. Traditional nanoprobes suffer from non-specific biodistribution and poor specific cellular uptake, which compromises imaging efficacy.
Non-specific biodistribution
Non-specific biodistribution of nanoprobes amplifies off-target signals. To address this issue, conjugating antibodies, ligands, or peptides enable selective tumor binding, leveraging biomarkers, like EGFR or folate receptors overexpressed in cancers. For example, gold nanorods conjugated to UM-A9 antibodies selectively bind squamous cell carcinoma biomarkers, enabling tumor differentiation via X-ray signal amplification [18]. Khademi et al. attached gold nanoparticles to folate-conjugated cysteamines (FA-Cys-AuNPs), which exploit folate receptor overexpression in tumors to detect lesions < 1 mm in size that are undetectable by conventional CT [19]. Polysaccharides have good water solubility and flexibility and can be used as standalone macromolecular nanocarriers. Ma et al. reported efficient targeting of visceral adipose tissue macrophages in obesity with polysaccharide nanocarriers for multimodal imaging or delivery of anti-inflammatory drugs [20].
Poor specific cellular uptake
Exogenous nanoprobes face rapid opsonization and sequestration by the RES (liver and spleen), which compromises the target tissue SNR. Consequently, it is crucial to simultaneously mitigate RES clearance, while promoting targeted cellular internalization. PEGylation enhances circulation time through steric hindrance [21] but is limited by immunogenicity [pre-existing anti-PEG antibodies accelerate clearance and induce hypersensitivity] [22, 23]. Dextran coating clinically validated for iron oxide nanoparticles, provides dual protection against aggregation and macrophage uptake [24]. Chitosan-based systems can also evade clearance by the RES, prolong drug circulation in the bloodstream, and improve drug targeting and bioavailability [25].
Low clearance rate
Conventional nanoprobes face a critical clearance paradox. Specifically, excessive systemic retention induces potential toxicity, while rapid renal elimination compromises imaging efficacy through insufficient target accumulation. This dilemma stems from the static physicochemical properties that cannot dynamically balance circulation, persistence, and clearance efficiency. Size-variable designs resolve this conflict through stimulus-responsive structural transitions, maintaining > 100 nm hydrodynamic diameters for prolonged blood circulation, then disassembling into < 6 nm subunits (i.e., below the glomerular filtration threshold) post-targeting for prompt renal clearance [26]. Light/pH/enzyme-activated systems exemplify this spatiotemporal control [27–31], as demonstrated by Huang’s oxygen-sensitive activatable polymeric nanoprobe for superoxide anion (APNso) [160 nm → 4 nm transition, achieving a 12-d clearance] [32]. Ruan et al. also created ADN1 and ADN2, size-variable nano-emitters, that can be activated by superoxide anions (O2−) at disease sites, which enable dual imaging/urinalysis via renal-excreted fluorophores [33].
Material toxicity
Nanoprobe-induced oxidative damage constitutes a major clinical safety barrier with reactive oxygen species (ROS) generation triggering cascade biological destruction through DNA strand breaks, membrane lipid peroxidation, and chronic inflammation [34]. Current mitigation strategies adopt dual approaches, as follows: accelerated clearance via renal-excretable designs; and intrinsic biocompatibility engineering. Yang’s magnetic ions coordinated nanoplatform (MICN) exemplifies a synergistic theranostic system. Fe/Mn/Gd-coordinated nanoparticles undergo programmed biodegradation into renal-clearable metal ions (< 5 nm), avoiding persistent bioaccumulation risks, while maintaining multimodal imaging capacity (photoacoustic/MRI SNR enhancement) through controlled metal coordination dynamics [35].
Harnessing CMNVs for multimodal imaging
The biomimetic camouflage strategy addresses the critical limitations of conventional surface modification approaches [e.g., PEG-induced accelerated blood clearance] [36], while resolving the dual challenges of nanoprobe biocompatibility and functionality. Natural cell membrane encapsulation establishes the following three critical biological interfaces: antigenic camouflage for RES evasion and prolonged circulation via native surface markers [37]; toxicity isolation through biomaterial-membrane separation; and tissue-specific homing through membrane-inherited targeting motifs, like integrins/chemokine receptors. To facilitate a clear comparison of harnessing different types of CMNVs for imaging, the key characteristics of various CMNVs are systematically summarized in Table 1.
Table 1 Summary of Cell Membrane-derived Vesicles as Nanoprobe Modification Strategies
| CMNVs | Cargo Loading Strategies | Advantages | Disadvantages | References |
|---|---|---|---|---|
| CMCNVs | Co-extrusion Ultrasonication Microfluidic electroporation infiltration |
Ease of production Highly biocompatible Multi-functional modifications possible |
Functions only as nanoparticle surface modification and cannot replace artificial nanocarriers Potential for disease transmission Immunocompatibility concerns the possibility of immune reactions occurring |
[38, 42] |
| EVs | Preloading: co-incubation, transfection Post-loading: co-extrusion, electroporation, sonication, co-incubation, saponins, and transfection |
Ease of storage Mature cargo loading technology High biostability for long-distance transfer Homologous targeting capability Low immunogenicity |
Production stability Low yield of EVs High difficulty and high cost of EV isolation and purification technology |
[65, 68, 110, 173] |
| OMVS | Direct loading: co-incubation, electroporation, and extrusion Indirect loading: transfection |
Intrinsic immunomodulatory properties Ease of industrialization Ease of customization |
Lack of standardization Potential off-target effects Potential biosafety Ambiguous contents |
[78, 79] |
| Yeast vacuoles | Preloading: transfection | Good tissue penetration and easy amplification | Ambiguous long-term stability, immunogenicity, and toxicity | [101, 104] |
Classification of CMNVs
CMNVs, which encompass entities, like EVs, OMVs, and yeast vesicles, have been harnessed for enhancing nanoprobes in multimodal imaging and studied in prokaryotic and eukaryotic cells [13].
Cell membrane-coated nanovesicles (CMCNVs)
CMCNVs are synthesized through three-stage precision engineering, encompassing membrane extraction, nanocarrier core fabrication, and membrane-core fusion via extrusion, sonication, or microfluidic electroporation [38] with classification based on membrane origin, including red blood cells (RBCs), white blood cells (WBCs), platelets, mesenchymal stem cells (MSCs), cancer cells, and hybrid membranes [39]. The membrane extraction process for anucleate mammalian cells, like RBCs and platelets, utilizes hypotonic lysis or freeze-thaw cycles, followed by differential centrifugation and extrusion [40, 41]. RBC membranes leverage CD47-mediated SIRPα signaling for immune evasion and prolonged circulation [16, 42], while early RBC-camouflaged nanoprobes rely on passive EPR targeting and are later enhanced by anti-EGFR modifications for active tumor targeting [43]. Platelet membranes offer low immunogenicity [44] and intrinsic affinity for circulating tumor cells, enabling pathologic site-specific delivery [45].
Eukaryotic cell membranes (WBCs, cancer cells, and MSCs) require complex isolation techniques, like sucrose gradient centrifugation and extrusion [46]. WBC membranes utilize endogenous chemotaxis for ligand-free tumor homing [42, 47], which has been demonstrated by therapeutic protein delivery using tumor-associated macrophage membranes [48]. MSC membranes offer scalable platforms due to expansion stability and tumor tropism, making MSC membrane-modified nanoprobes suitable for large-scale production [49–51].
Cancer cells exhibit homotypic adhesion, unlimited proliferation, and immune escape [52]. Cancer cell surface adhesion molecules (N-cadherin, galectin-3, and EpCAM) enable multicellular aggregation and can be exploited for nanoparticle functionalization. Cancer cell membrane-encapsulated nanoprobes are therefore promising for homologous targeting and specific imaging [53]. Lv et al. further utilized the ability of cancer cell membranes to evade phagocytosis and enable homotypic targeting for nanoprobe modification, resulting in excellent biocompatibility and imaging performance [15]. These probes show high recognition specificity and selective aggregation in homologous cancer cells, exhibiting a 16-fold increase in intracellular fluorescence intensity and strong in vivo signals.
While red blood cell membrane-camouflaged nanoparticles (RBCNPs) have a long circulation, RBCNPs often require added targeting ligands for tumor localization [54]. Fusing membranes from different sources provides multifunctionality, adapting CMCNVs to complex environments. Current hybrid-coated nanoparticle development focuses on RBC-based hybrids, particularly RBC-platelet fusion membranes. Thermodynamic treatment achieves membrane fusion [55], retaining dual functions and enhancing circulation and offering a natural alternative for post-synthesis functionalization.
Although harnessing CMCNVs offers advantages, such as prolonged circulation time, immune evasion, and active targeting, the clinical translation of CMCNVs still faces several challenges. For example, the extraction of cancer cell membranes requires thorough removal of the nucleus and genetic material to reduce carcinogenic risks; related clinical trials are still under investigation [42]. Furthermore, developing standardized protocols is crucial for the large-scale production and clinical translation of cell membrane-modified nanoprobes. Membrane isolation typically requires at least 100 million cells and these cells must maintain phenotype, purity, and quality during passage. Developing standardized and mature cell culture protocols for each cell type is essential for large-scale production. In this regard, existing mature biomanufacturing platforms, such as the use of 3D bioreactors (e.g., stirred-tank and WAVE bioreactors), can be used for large-scale cell expansion [56]. Additionally, extrusion methods commonly used in laboratories have low production efficiency and are challenging to scale up for industrial manufacturing, which often requires good reproducibility and stable technical parameters [57]. Moreover, for some cell types, such as WBCs, surface membrane proteins exhibit heterogeneity, posing a risk of triggering hemolysis in vivo. Autologous cells are the most suitable option in this case and strict donor cell screening protocols must be established to prevent the use of allogeneic cells as membrane sources.
EVs
EVs are nanosized vesicles released from various cells, including reticulocytes, platelets, stem cells, dendritic cells, and tumor cells [58], and can be detected in body fluids, such as blood, cerebrospinal fluid, and urine [59, 60]. EVs were considered “waste material” released by cells until 1973 [61] but recent studies have shown that EVs are key messengers in intercellular communication [62–64]. EVs can be classified into apoptotic bodies (500 nm–5 μm), microvesicles (150–500 nm), and EVs (40–150 nm) based on size and origin with EVs being the smallest and most suitable for nanoprobe modification [65].
EVs are composed of a lipid bilayer membrane encapsulating a hydrophilic core and carry ligands and receptors from parent cells. EVs offer lower cytotoxicity and immunogenicity, better resistance to degradation in circulation, and enhanced ability to evade phagocytosis by the mononuclear phagocyte system when harnessed for nanoprobe loading compared to synthetic nanocarriers [66–68]. Tumor-derived EVs can target specific tissues and protect nanoprobes from blood degradation [17] and can cross the blood-brain barrier, providing new strategies for drug delivery and diagnostic imaging in neurologic diseases [66]. Given the immense research potential of harness EVs to load nanoprobes for precious imaging, understanding the in vivo biodistribution of EVs is crucial for accelerating clinical translation. Relevant studies mainly use fluorescent probe labeling, transgenic parent cells expressing luciferase on the vesicle surface, and radioactive labeling to analyze EV behavior [69–71].
A key prerequisite for the widespread use of EVs in nanoprobe loading is the efficient preparation of high-purity vesicles with maintained structural integrity. Currently, common EV isolation methods include ultracentrifugation, ultrafiltration, immunoaffinity separation, and polymer-based precipitation. Among these modalities, ultracentrifugation is the gold standard but faces challenges, such as being time-consuming, requiring expensive equipment, resulting in non-specific purification, and reducing biological activity, making ultracentrifugation unsuitable for large-scale production [72]. Both ultrafiltration and precipitation methods face the dilemma of low purity recovery, which hinders subsequent experiments [73]. Immunoaffinity separation is based on antibody recognition of unique surface markers of EVs, such as CD9, CD63, CD81, and Alix, and involves separating the EVs using antibodies fixed to magnetic beads, chromatographic column resins, porous plates, or microfluidic devices. Although this method effectively improves the purity and sensitivity of EV isolation, high cost makes immunoaffinity separation unsuitable for large-scale separation [74]. With the development of microfluidic chips and micro-nanofabrication technologies, new tools for high-sensitivity, high-accuracy EV separation, and detection are emerging [75].
Microfluidic technology, with its advantages of low sample consumption, high throughput, and easy integration, can precisely control micro-scale reaction conditions and enhance EV operation efficiency. In particular, integrated microfluidic systems combining two or more separation technologies, such as the integrated magnetic-electrochemical EV device, which combines electrochemical EV detection methods with magnetic-based EV separation methods to achieve high-throughput EVs detection, better meet the needs of clinical research [76]. Nanoplasmonic biosensors, such as surface plasmon resonance, localized surface plasmon resonance, and surface-enhanced Raman scattering, enable label-free, high-sensitivity detection of EVs through the optical properties of metal nanostructures [75].
OMVs
OMVs are natural vesicles secreted by Gram-negative bacteria [77]. Some studies in recent years have shown that some Gram-positive bacteria also produce vesicles. Only OMVs produced by Gram-negative bacteria will be discussed herein. An important role of OMVs in nature is to deliver biomolecules to specific distant sites for parent bacteria [78]. OMVs have been developed as bacterial vaccines, adjuvants, tumor immunotherapeutic reagents, drug delivery carriers, and anti-bacterial adherence reagents [79], which are nanolipid bilayer vesicles with a particle size between 50 and 200 nm, and are mainly composed of lipids, proteins, and various pathogen-associated molecular patterns (PAMPs), including LPS, lipoproteins, peptidoglycan, DNA, and RNA [80]. Unlike the above-mentioned modifications, these PAMPs make OMVs easily detectable and internalized by phagocytes. Li et al. demonstrated that 83.4% of attenuated OMVs-loaded nanoparticles are internalized by neutrophils compared to 9.6% internalization by monocytes and by piggybacking on neutrophils, OMVs can efficiently target the nanoprobe to the deeper parts of inflamed tumor tissue [81].
Harnessing OMVs to load nanoprobes offers several advantages. Genetic engineering of the parent bacterium enables facile targeted delivery. Chen et al. developed a modular method embedding original cell-expressed luciferase within OMVs, while displaying specific antibodies externally that bypassed complex purification or attachment steps for versatile applications, like live-cell imaging [82]. Furthermore, the intrinsic OMV composition promotes efficient neutrophil uptake and “piggybacking” transport to deeper inflammatory sites, like tumors, with rapid neutrophil release contrasting slower erythrocyte and monocyte release involving digestion, which facilitates specific neutrophil imaging. The rigid OMV membrane also protects encapsulated nanoprobes from degradation en route to targets. Additionally, OMVs possess inherent anti-tumor activity by inducing anti-tumor cytokines (CXCL10 and interferon [IFN]-γ), inhibiting tumor growth [83]. Thus, harnessing OMVs to load nanoprobes is expected to achieve theranostics.
Despite several successful applications, the safety of OMVs requires ongoing scrutiny. Surface LPS activates macrophages, endothelial cells, and inflammatory cascades, potentially causing tissue damage or septic shock [78, 84]. The toxicity resides primarily in lipid A, biosynthesized through nine essential enzymes [85]. Genetic engineering mitigates this risk. Deleting lpxL or lpxM/msbB genes or introducing Hp0021 generates OMVs containing exclusively monophosphorylated lipid A, which significantly reduces toxicity and inflammation [86, 87].
Low production efficiency further impedes OMV applications. OMV production efficiency is dynamically regulated by environmental signals, growth phase, and genetic factors [88]. Peak production occurs during late-exponential to stationary phases, even though harvesting during the late-exponential phase prevents contamination from bacterial lysis [89]. Environmental modulation is equally crucial. Iron limitation boosts production in Mycobacterium tuberculosis and Helicobacter pylori, while suppressing VacA virulence factor expression [90, 91]. Hypoxia increases Pseudomonas aeruginosa vesicle output sixfold [92]. Detergents, metal chelators, or H2O2 induce oxidative stress to promote vesiculation [93–95]. Mutations in any gene of the Escherichia coli Tol-Pal membrane complex (tolQ, tolR, tolA, tolB, and pal) significantly increase yields [86, 96, 97]. Periplasmic expression of colicin A/E3 or phage G3p translocation domains similarly enhances vesiculation [86]. Nevertheless, OMV purification remains a bottleneck due to reliance on time-consuming ultracentrifugation and filtration. Advanced technologies are therefore imperative for scalable production.
Yeast vacuoles
With the discovery of biologically derived, nano-sized vesicles, an increasing number of researchers are focusing on the study of biologically derived nanocarriers. Yeast vacuoles, as one of the biologically derived nanocarriers, are derived from yeast cells and are highly dynamic degradative single-membrane organelles used to break down abnormal intra- or extra-cellular substrates and to recover the stored nutrients [98]. Yeast vacuoles have lower immunogenicity and stronger tissue penetration compared to the uncertain immunogenicity of bacterial exocysts from genetically engineered bacteria and the high technical difficulty of purifying EVs from mammalian cell sources and yeast vacuole production technology is simpler than EVs [99]. Moreover, yeast vacuoles possess a lipid bilayer similar to the composition of mammalian cell membranes, which improves the fusion efficiency with the plasma membrane or endolysosomes and ultimately facilitates the release of the drug into the target cells or tissues [100]. In general, obtaining yeast vesicles for use as bio-nanocarriers requires genetically engineered yeast cells for modification. There are usually 2–5 large vesicles with a diameter of approximately 1 μm inside the yeast cells. Size is a key cycle that determines the vesicle drug encapsulation rate and half-life with smaller vesicles having more chances to evade phagocytosis for uptake [101]. Rab GTPase is a prerequisite for homotypic vesicle fusion. The major Rab GTPase that promotes fusion of yeast vesicles is yeast protein transfer protein7 (YPT7), the overexpression of which causes membrane expansion of yeast vesicles. Thus, setting up YPT7 protein knockdown results in many fragmented, nanoscale vesicles [102, 103]. Engineering to increase the specific targeting of yeast vesicles has also garnered interest in transfecting some tissue-specific ligands into yeast cells so that the surface of yeast vesicles carries tissue-specific targeting ligands [104].
Yeast cells that have been used as vectors for testing include Saccharomyces cerevisiae, a non-polluting, non-pathogenic yeast that has been used in fermentation products for thousands of years [105]. Unlike bacteria, the lipid composition of S. cerevisiae is like that of mammalian cell membranes, potentially increasing the efficiency of fusion with the plasma membrane or endolysosomes and thereby facilitating drug release into targeted cells or tissues [100]. Taking advantage of these properties, Gujrati et al. genetically engineered S. cerevisiae to express human epidermal growth factor receptor 2 (HER2)-specific affinities on vesicular membranes, which target the HER2 receptor that is expressed on a variety of cancers [99]. The anti-HER2 affinity-expressing vacuole (AffiHER2vacuole) was further loaded with the chemotherapeutic drug, adriamycin (Dox), which is widely used for the treatment of solid tumors. Active administration of AffiHER2 vacuoleDox had greater accumulation and penetration in tumor tissues, reflecting significant tumor growth inhibition. Although vacuoleDox exhibited relatively low cellular uptake due to passive targeting, the enhanced distribution, like that of AffiHER2 vacuoleDox, ensured the effectiveness of the yeast vesicle biocarrier for deep tissue penetration. The above-mentioned results, as well as the low toxicity of the yeast vesicle vector, suggest that the bioengineered vesicles have the potential to be used as drug delivery carriers for the treatment of cancer.
Methods of harnessing CMNVs for nanoprobe loading
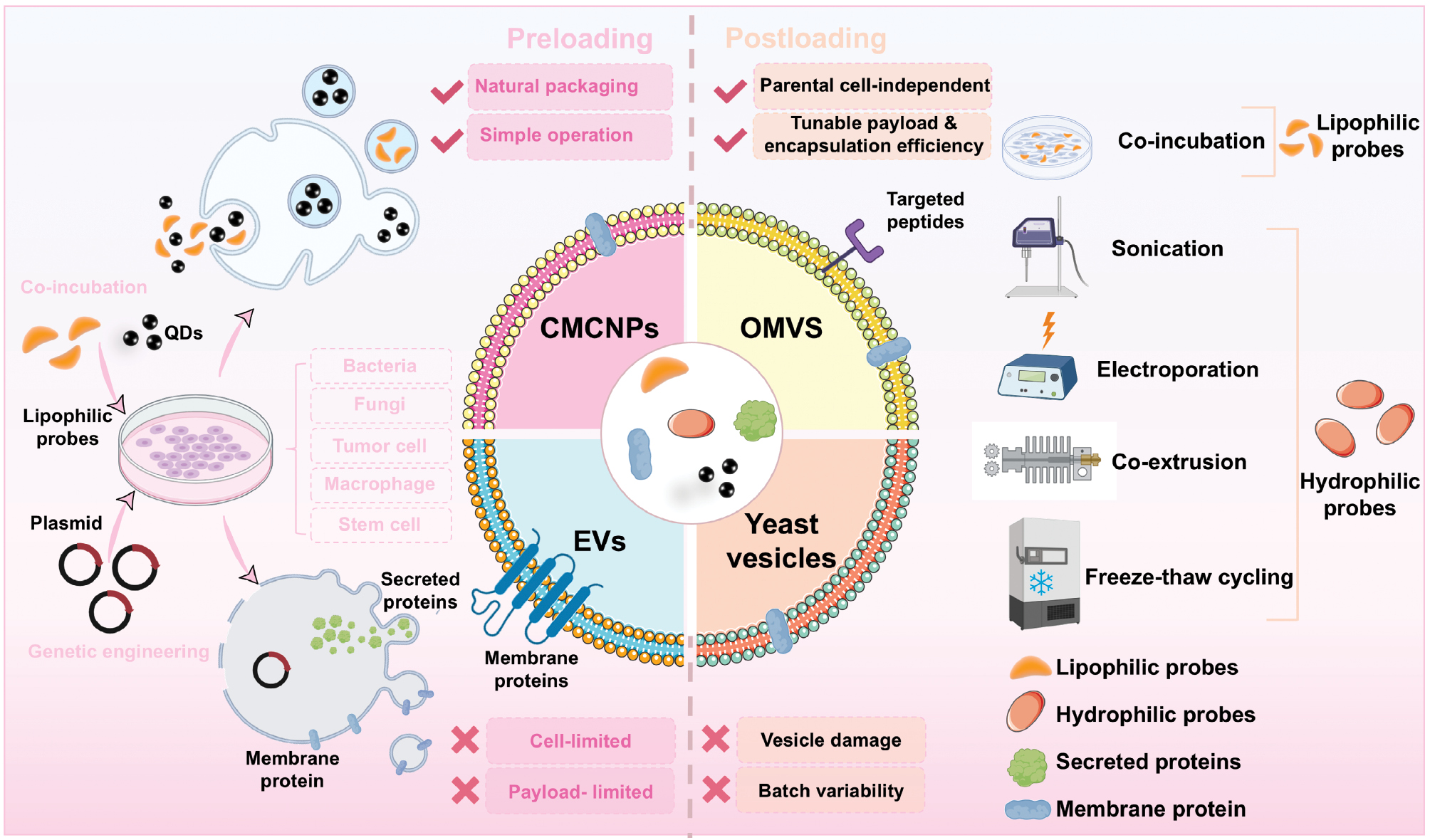
The methodologies for nanoprobe loading into CMNVs are fundamentally classified into pre- and post-loading strategies [106]. Pre-loading approaches accomplish probe incorporation during CMNV biogenesis through two primary mechanisms [genetic engineering involving transfection of plasmids encoding reporter molecules (e.g., fluorescent proteins and luciferases) into parental cells (bacteria, fungi, tumor cells, macrophages, or stem cells) for endogenous expression and subsequent packaging into secreted vesicles [107] or cellular uptake, where lipophilic probes (organic dyes, quantum dots) internalize via co-incubation and integrate during membrane assembly] [108, 109]. Conversely, post-loading strategies use physicochemical techniques, including co-incubation, electroporation, sonication, extrusion, and freeze-thaw cycling, to modify isolated CMNVs, which offers enhanced flexibility in controlling encapsulation efficiency (EE%) and nanoprobe payload [110] without requiring parental cell modification. This strategy renders post-loading particularly suitable for hard-to-transfect cells (erythrocytes and primary cells) or biofluid-derived vesicles. Lipophilic probes (dialkylcarbocyanines and cyanine dyes) spontaneously embed into lipid bilayers through co-incubation, as exemplified by DSPE-PEG2000-Cy5/DSPE-PEG2000-N3 incubation with EVs forming Cy5-EV-N3 within 30 min at room temperature [111, 112]. However, hydrophilic probes (polar dyes and ultrasmall superparamagnetic iron oxide nanoparticles) require assisted loading methods, such as electroporation or freeze-thaw cycling, although these probes may compromise vesicle integrity by altering physicochemical properties or morphology [113, 114] (Figure 2).
Figure 2 Schematic overview of pre- and post-loading strategies for integrating nanoprobes into CMNVs. Nanoprobes (QDs and lipophilic probes) and plasmids encoding fluorescent proteins or luciferase can be pre-loaded into parental cells via co-incubation or genetic engineering. Post-isolation, lipophilic probes are incorporated into purified CMNVs via co-incubation, whereas hydrophilic probes require active loading methods (e.g., sonication, electroporation, co-extrusion, or freeze-thaw cycling).
Harnessing CMNVs advances in multimodal imaging
Harnessing CMNVs in fluorescence imaging
Fluorescence imaging, recognized for operational simplicity and cost-effectiveness, has demonstrated significant advancements in sensitivity, spatiotemporal resolution, and rapid response capabilities [115]. Driven by growing clinical demand for enhanced clarity and deeper penetration in in vivo imaging, which is crucial for early diagnosis and prognosis, researchers are actively exploring novel methodologies with superior bioimaging performance. The near-infrared-II (NIR-II [1000–1700 nm]) spectral window offers substantial advantages over conventional NIR-I wavelengths, enabling greater tissue penetration depth, a SNR, and improved spatiotemporal resolution [116, 117]. Consequently, inorganic and carbon nanomaterial-based NIR-II probes have been developed, including single-walled carbon nanotubes [SWCNTs] [118, 119], QDs [120, 121], and rare earth-doped nanoparticles [122, 123].
QDs exhibit broad absorption spectra, high quantum yields (QYs), exceptional photostability, and strong fluorescence intensity, which facilitates application in disease imaging and diagnosis [124, 125]. However, inadequate targeting often leads to off-site accumulation and rapid clearance by the RES, which compromises tumor-specific imaging efficacy. For example, intravenous administration of Ag2S QDs results in predominant fluorescence signals in the bladder and urine within 1 h with negligible tumor detection, indicating rapid renal clearance. By 12 h, only weak fluorescence appears at tumor peripheries without deep tissue penetration. Free QDs become undetectable in blood after 24 h and bladder fluorescence nearly disappears by 48 h [126].
Surface modification strategies to enhance QD hydrophilicity and targeting may reduce fluorescence QY and stability, which diminish imaging signals [127]. To address this issue, macrophage-secreted EVs were used to deliver Ag2S QDs (MVs@QDs&DOX). MVs@QDs&DOX generates intense tumor fluorescence 12 h post-injection, peaking at 24 h with sustained detection until 48 h compared to free QDs. Near-infrared light-triggered EV disruption releases QDs, yielding uniform signal distribution throughout tumors with an 85-μm penetration depth and full coverage of 9-mm diameter lesions. This biomimetic carrier prolongs circulation, enhances tumor accumulation, and improves penetration via light-responsive release, effectively overcoming the limitations of free QDs and offering a promising strategy for efficient, low-toxicity theranostics [126]. Nevertheless, the targeting mechanism relies on macrophage-derived exosomal membrane proteins, while cumbersome purification and low yield hinder scalability. Consequently, alternative biomimetic vesicular carriers for QD delivery are being explored. Choi et al. engineered erythrocyte membrane-derived nanoparticles (EDNs) incorporating QDs within the lipid bilayers and conjugated the EDNs with tumor-specific anti-EGFR antibodies. The natural erythrocyte membrane structure significantly improved circulatory stability with detectable QD signals in blood 48 h post-administration [43].
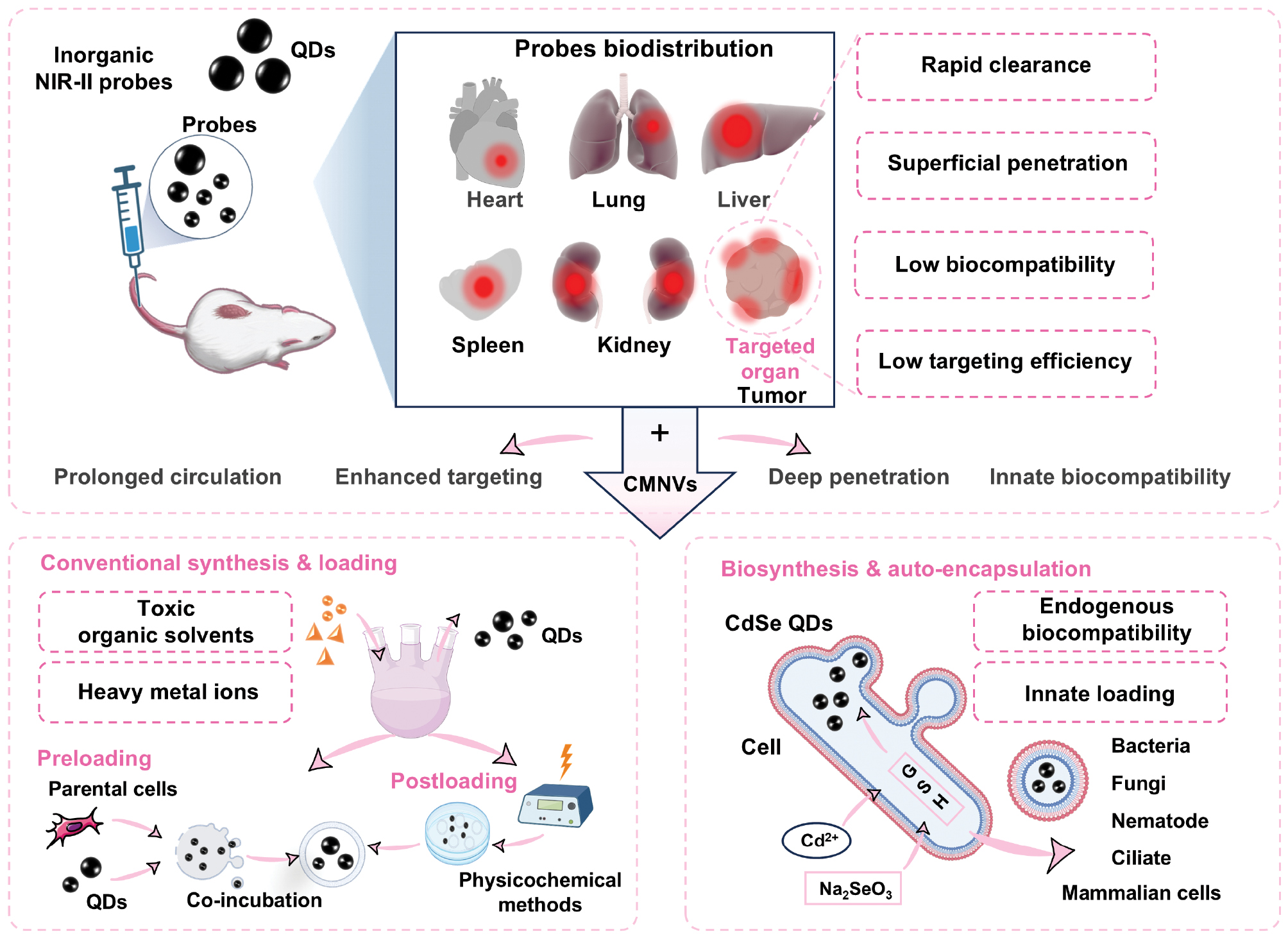
Conventional chemical synthesis of QDs often involves toxic organic solvents and heavy metal ions, compromising biocompatibility and clinical translation [127]. Emerging biosynthesis approaches leverage endogenous metabolic pathways, such as glutathione (GSH) and thioredoxin (TRX) systems in S. cerevisiae, Staphylococcus aureus, and MCF-7 cells. to synthesize CdSe QDs under mild conditions. This strategy confers intrinsic biostability and biocompatibility without requiring additional ligand exchange or encapsulation, establishing a green synthesis route for biocompatible QDs [128, 129]. Intracellularly synthesized QDs, exemplified by CdSe QDs in MCF-7 cells, can integrate into EVs via natural secretory pathways, enabling autonomous vesicular loading [130]. Expanding this approach to macrophages could facilitate mass production of fluorescent probes combining prolonged circulation, high targeting specificity, and exceptional biocompatibility (Figure 3).
Figure 3 Application of CMNVs to overcome inherent limitations of conventional QDs in biomedical fluorescence imaging. Conventionally synthesized QDs loaded into CMNVs via pre- or post-loading strategies exhibit prolonged circulation, enhanced targeting, and deeper penetration. Biosynthetic QDs, generated via intracellular biogenesis, further improve biocompatibility when integrated into this framework.
Organic small molecule fluorophores represent promising NIR-II imaging agents due to excellent biocompatibility and pharmacokinetic profiles. However, most small molecule fluorophores exhibit low QYs and brightness. To overcome this issue, researchers have developed innovative strategies to enhance fluorescence QY and spatiotemporal resolution. One effective approach utilizes natural nanoplatforms to integrate multiple NIR-II fluorophores with tumor-targeting moieties, creating targeted fluorescent nanoprobes with optimal biocompatibility and emissive properties. For example, PEGylated CH1055, which is noted for high water solubility, was incorporated by Hong et al. into small extracellular vesicles (sEVs [140 nm diameter]) along with EGFR-targeting GE11 peptides to construct the sEV-CH1055-GE11 nanoprobe. This assembly preserved the characteristic absorption (720 nm) and emission (1050 nm) profiles of CH1055-PEG, while delivering high NIR-II brightness and enhanced tumor targeting. The tumor-to-background ratio exceeded the Rose criterion of 5 at 48 h and reached 10.43 by 72 h post-injection. Cell viability, hematologic analysis, and hematoxylin and eosin (H&E) staining confirmed excellent biocompatibility. This platform enables non-invasive, high-contrast imaging of EGFR-overexpressing tumors, demonstrating how natural nanocarriers can optimize precision and reliability in fluorescence imaging [131].
Harnessing CMNVs in bioluminescence imaging
Bioluminescence is a type of optical imaging that utilizes luciferase enzymes to react with substrates. Bioluminescence offers high sensitivity, resolution, and a low background signal. Unlike fluorescence imaging, bioluminescence does not require external light excitation because animals lack autoluminescence [132]. Luciferase reporter imaging is the representative technique in bioluminescence imaging with bacterial luciferase, firefly luciferase, and flower bug luciferase being commonly used reporter genes. Recently, a new platform (NanoLuc [NLuc]) has emerged, which is more stable, smaller in size, > 150 times more efficient in luminescence, and unaffected by ATP. NLuc also has substrates with higher stability and lower background activity, opening new possibilities for bioluminescence imaging [133]. Another advantage of NLuc is that NLuc does not require post-translational modification in mammalian cells, which allows for low-level expression of reporter genes that better mimic physiologic conditions [134]. Mammals can be made bioluminescent by introducing luciferase genes or via infection with a recombinant adeno-associated virus (rAAV) to enable stable expression of the required enzymes for bioluminescence imaging [135]. Bioluminescence imaging is non-toxic, making bioluminescence suitable for live cell applications that do not require high spatial and temporal resolution, particularly for long-term studies involving slow kinetics or photosensitive components [136].
Intercellular communication is a dynamic process involving cellular activity. Tumor-derived EVs can induce various biological responses, including oncogenic responses, proliferation, cell movement, and stemness, ultimately leading to metastasis [137–139]. Studies have shown that melanomas can utilize an efficient exosomal messenger mechanism to prepare for metastasis through tumor exosomal precursors to pre-metastatic lymph nodes [140]. Therefore, monitoring the distribution of EVs in tumor cells would be valuable for studying tumor metastasis. Tracking the in vivo distribution of EVs is crucial and several studies have attempted to engineer EVs to express luciferase for tracing purposes. Gangadaran et al. stably transduced the Rluc gene into a human undifferentiated thyroid cancer cell line (CAL-62 cells [CAL-62/Rluc]) and a human breast cancer cell line (MDA-MB-231) by lentivirus [141]. EVs with Rluc were successfully prepared and the reaction with the substrate, luminalin, produced optical radiation at wavelengths of 475–480 nm, which was used to trace the distribution of EVs in vivo. This study also demonstrated for the first time that the transduction of BLI reporter protein expression into CAL-62 and MDA-MB-231 cells did not significantly alter EV properties. This study also demonstrated for the first time that transduction of BLI reporter protein expression in CAL-62 and MDA-MB-231 cells did not significantly alter EV properties. However, this approach, which requires in vitro gene transfection of tumor cells, isolation, and purification, and then import into mice for tracing of the EVs, may not faithfully reflect the concentration, location, and transport dynamics of endogenous EVs.
Therefore, Luo et al. developed a mouse model that can be non-invasively and quantitatively labeled to monitor endogenous EVs [12]. A mouse model expressing TG-α MHC-STOP-CD63NanoLuc was successfully established by performing prokaryotic microinjection. Upstream of the CD63NanoLuc reporter gene is not expressed due to the STOP sequence but can be driven by the cardiomyocyte-specific α-MHC promoter, and for the purpose of controllable labeling, this is the first genetic mouse model capable of tracking endogenous EVs. Compared to fluorescently labeled EVs, CD63-NanoLuc-labeled EVs are more specific and show a strong bioluminescent signal only in the cardiac region of transgenic animals. In addition, CD63NanoLuc mRNA and protein were not induced to be expressed in cardiac fibroblasts, indicating that this EV tracking system is highly cell-specific and strictly controllable. NanoLuc bioluminescence is highly sensitive, stable, and non-invasive in vivo, and the fusion of NanoLuc and CD63 has no deleterious effect on cardiac EV biosynthesis. This mouse model provides a useful tool for tracking the dynamics of cardiac EVs in vivo.
Hikita et al. previously designed CD63NanoLuc for EV tracking in a cancer cell line to directly quantify EVs without purification [142]. However, only purified exotics could be tracked but by adding a tumor cell-specific promoter upstream of CD63NanoLuc, the exosomal behavior of tumor cells in vivo could be monitored.
The synthesis of NanoLuc-expressing OMVs by genetically engineered and edited E. coli became a good choice due to the difficulty of introducing the luciferase gene into mammalian eukaryotic cells and the low yield of EVs. Chen et al. reported a one-pot synthesis method to design multifunctional sensors based on OMVs, where the lipoprotein, Sly B, specifically directs the bacterial in-house expressed nanoluciferase into the interior of OMVs. The ice nucleation protein (INP) was used to decorate the vesicle surface with the Z-domain via a trifunctional scaffold composed of cohesion domains from Clostridium cellulolyticum (CC), C. thermocellum (CT), and Ruminococcus flavefaciens (RF). This approach facilitates vesicle binding to the antibody. This engineered OMV emitted > 105 times as many photons as control OMVs after co-incubation with substrate, displaying highly sensitive luminescent signals and creating a virtually infinite combination of capture and reporter sections [82].
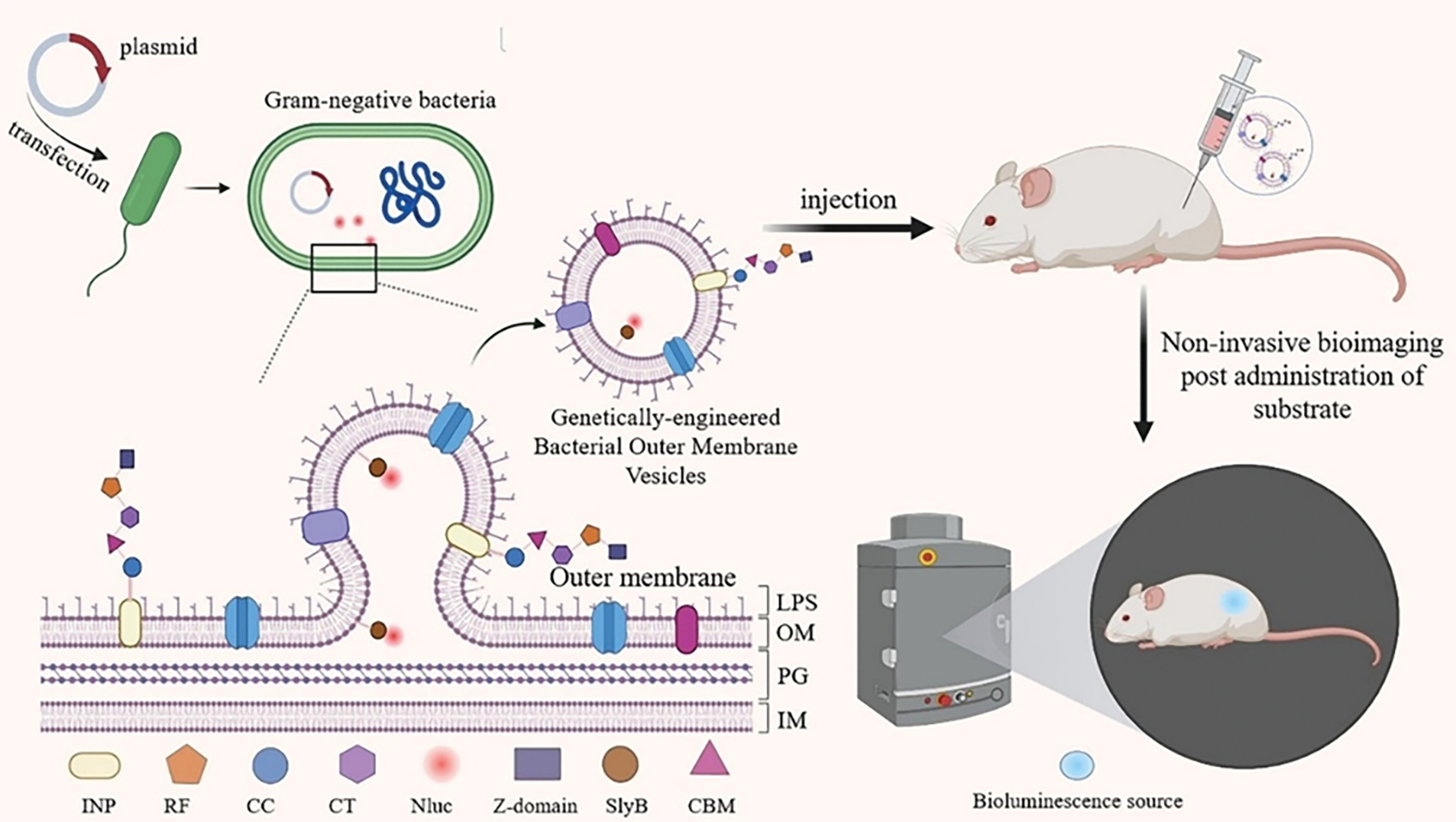
In 2019 the team was able to detect the most intense bioluminescence shortly after substrate injection by isolating OMVs containing NanoLuc and Z structural domains from genetically engineered E. coli and injecting the OMVs subcutaneously into mice under mild substrate reaction conditions (Figure 4). The bacterial OMVs had low cytotoxicity and high biocompatibility. In addition, the bacterial OMVs used in this study could be further functionalized with dockerin-tagged foreign proteins by adding dockerin-tagged foreign proteins to further functionalize the OMVs with relevant antibodies to recognize biomarkers on cancer cells for bioluminescence-based tumor-targeted imaging. The unique properties of engineered multifunctional OMVs can be used for molecular imaging [143].
Figure 4 Engineered OMVs enable targeted tumor bioluminescence imaging. Gram-negative bacteria were transfected with plasmids encoding nanoluciferase (Nluc) and a targeting moiety. SlyB lipoprotein transported intracellularly expressed Nluc into OMVs. An INP-based scaffold incorporating CC, CT, and RF adhesin domains display surface Z-domains for antibody conjugation. Subcutaneously injected engineered OMVs generated strong bioluminescent signals post-substrate administration, demonstrating low cytotoxicity, high biocompatibility, and sensitive tumor-targeting capability. Figure 4 was created in BioRender. (2025) https://BioRender.com/6tkqqcb.
Harnessing CMNVs in photoacoustic (PA) imaging
PA imaging is a non-ionizing, non-invasive mode that combines optical and ultrasonic imaging modalities [144]. When a pulsed laser irradiates tissue, light energy is transformed into heat energy by endogenous or exogenous contrast agents, resulting in an instantaneous increase in local tissue temperature, followed by thermoelastic expansion and the generation of sound waves. The acoustic waves are captured by an ultrasound transducer and data processing based on the arrival time, which transforms the acoustic waves into PA images [145]. PA imaging provides a high spatial resolution and a significant contrast compared to optical and ultrasound imaging [146]. PA imaging has been widely used for breast, skin, blood vessel, and adipose tissue imaging. Although endogenous contrast agents, such as hemoglobin, can produce PA imaging signals, PA imaging does not provide further tissue information in the absence of exogenous contrast agents. Therefore, it is critical to produce exogenous contrast agents. Some small organic molecules, such as cyano dyes, are limited in practical application because of rapid degradation in water, poor photostability, a short in vivo half-life, a tendency to bind to serum in the blood, and rapid clearance after intravenous injection [147]. One way to increase the PA signal intensity of this type of contrast agent is to increase the accumulation of the probe in the tissue of interest. The use of cell membrane-derived vesicles for the transport of such contrast agents can achieve this goal. For example, ICG-loaded PLGA camouflaged on A549 cell membranes encapsulating perfluorocarbons can be actively targeted to tumor tissues for homologous targeting [148]. The biocarrier can also be genetically modified in the original cells of the vesicle or directly modify the vesicle with ligands so that the biocarrier can spontaneously encapsulate the endogenous contrast agent and possess active targeting properties during the formation process.
Melanin, as a natural light absorber with high absorption capacity in the visible and near-infrared windows, is ideal for enhancing contrast in PA imaging [149, 150]. In addition, melanin has a high photothermal conversion efficiency, making melanin ideal for use in photothermal therapy, in which light is used to selectively irradiate melanin-containing tissues and induce localized heating that kills tumor cells. The ability of melanin to act as a contrast agent and a therapeutic agent potentially makes melanin clinically integrable.
Gujrati et al. thus developed a molecular approach to encapsulate melanin in cell membrane-derived nanocarriers. E. coli expressing a tyrosinase transgene was transformed into a low endotoxin strain (OMVΔmsbB) by inactivating the msb gene [13]. The melanin produced by this strain passively binds to the cytoplasm and cell membrane, resulting in the formation of bacterial OMVs encapsulating the biopolymer-melanin (OMVMel). Experimental results have shown that OMVMel produces a strong MSOT signal both mimicking and in vivo. Exposure to a pulsed near-infrared light source produces local heating that inhibits tumor growth in vivo. suggesting that melanin-coated OMVs is a promising therapeutic agent for PA imaging.
The use of cell membrane-derived vesicles of mammalian origin is more biologically safe compared to the uncertainty of immunogenicity of bacterial exocysts. Ding et al. used folate-modified erythrocyte membranes as nanocarriers and graphene QDs as a nano-enzymatic and responsive molecule with peroxidase activity {[2,2-azidobis (3-ethylbenzothiazolidine-6-sulfonic acid)] (ABTS)}. Exocystic vesicles from EVs isolated from tumors were used as nanocarriers of the photosensitizer, Chlorin e6 (Ce6), for effective targeted PA imaging of malignant tumors. In addition, EVs-loaded Ce6 showed a stronger PA signal in imaging compared to free or liposome-loaded Ce6 [151].
Harnessing CMNVs in MRI
MRI, as a non-invasive clinical modality, provides exceptional spatial resolution and soft tissue contrast. The image contrast primarily depends on tissue magnetic properties, including proton density and T1/T2 relaxation times of water protons. However, insufficient magnetic heterogeneity between tumors and normal tissues often compromises MRI contrast. To address this limitation, paramagnetic metal ions and paramagnetic metal ion chelates serve as contrast agents that modulate tissue relaxation times to enhance imaging contrast [152]. Gadolinium-based contrast agents and superparamagnetic iron oxide nanoparticles (SPIONs) represent widely used MRI contrast materials. SPIONs exhibit super-paramagnetism, high saturation magnetization, favorable biocompatibility, and low toxicity, functioning as negative MRI contrast agents with significant biomedical utility [153–155]. Nevertheless, inadequate targeting remains a major challenge for SPIONs. Specifically, sub-micromolar concentrations in target tissues often fall below MRI detection thresholds. EV-derived carriers offer a promising solution by providing enhanced targeting specificity and biosafety [156].
In 2014 Hu et al. first demonstrated MRI detection of EVs loaded with super-paramagnetic iron oxide nanoparticles (SPIONs), establishing applicability in neurologic disease theranostics [157]. Wang et al. subsequently conjugated stem cell-derived EVs with SPIONs, achieving targeted brain MRI in rats through significant T2 relaxation time shortening and enhanced tissue contrast [158]. Further advancing this approach, astrocyte-derived EVs carrying ultra-small SPIONs (USPIOs) were efficiently delivered to the brain via nasal administration with significantly elevated ΔR2 values detectable on day 1 and USPIOs particles observed in 85.7% of astrocytes, validating utility as blood-brain barrier (BBB)-traversing carriers for brain disorders [159].
EVs lacking innate targeting specificity, such as those derived from macrophages, have attracted considerable interest due to low immunogenicity, scalable production, and inherent BBB penetration capability. Surface engineering strategies, including covalent modification and ligand-receptor interactions, enable active targeting for precision delivery [160, 161]. Engineering EVs with arginine (R)-glycine (G)-glutamic acid (E) [RGE] peptides, which are specific ligands for neuropilin-1 overexpressed in gliomas, yielded an RGE-Exo-SPION/Cur system in which MRI revealed substantially higher contrast-to-noise ratios (CNRs) and sharper tumor boundaries compared to controls [162–164]. Genetic engineering approaches further enhanced vesicle-based MRI performance. Galisova et al. used CRISPR/Cas9 to generate HEK293 cells stably expressing the SARS-CoV-2 spike receptor-binding domain (RBD), secreting RBD-retaining EVs (EVsRBD). Loading SPIONs into EVsRBD produced a 40-fold greater CNR enhancement in ACE2-positive tumors than free SPIONs within 4 h post-IV injection, enabling non-invasive monitoring of RBD-ACE2 binding dynamics for tracking viral variants and evaluating therapeutic sensitivity [165].
Optimizing contrast agent size constitutes a critical design parameter for EV-based MRI probes. Studies have indicated that USPIOs fail to generate detectable MR signals, even at maximum feasible concentrations (5 mgFe/mL), which limits utility for in vivo imaging [166]. Conversely, clinically approved large-particle agents, like ferucarbotran (Resovist®), demonstrate superior performance, including higher SNRs than USPIOs [167], efficient EV loading without compromising structural integrity, and distinct signal attenuation in T2-weighted imaging. Intramuscular injection experiments revealed concentrated Resovist®-labeled EV signals at target sites (muscle tissue), whereas USPIO-labeled counterparts diffused into subcutaneous fat, confirming that SPIONs > 50 nm enhance tissue retention and local targeting precision [168]. The established safety profile of Resovist®, biocompatibility, and good manufacturing practices (GMP)-compliant manufacturing synergize with EV carriers to accelerate clinical translation of this integrated platform.
Discussion
Nanoprobes offer superior stability, imaging quality, and specificity when compared to traditional probes. CMNVs are a key platform for advanced imaging, supporting nanoprobe design, and loading. CMNV biocompatibility and low immunogenicity enhance targeting and address key limitations in high-quality imaging. However, clinical translation requires overcoming critical challenges, such as heterogeneity control, scalable purification, and safety and immunogenicity balance for long-term safety.
Verifying long-term safety is essential due to parental cell endotoxicity for OMV-based probes or vaccines. The MenPF1 OMV vaccine from Neisseria meningitidis B H44/76 is safe and tolerable in healthy adults [169]. A phase I trial showed increased serum IgG but poor memory B cell response and antibody persistence [170], suggesting engineered OMVs offer long-term safety potential for immunotherapy, although novel adjuvants or nanocarriers are needed to boost antigen presentation. Progress has also been achieved for EV long-term safety. While prolonged EV circulation may be unnecessary, preclinical studies have shown rapid intravenous clearance in minutes [171]. Routine plasma transfusions transfer trillions of allogeneic EVs without adverse events, supporting biocompatibility [172]. However, CMNVs face heterogeneity and standardization challenges. Size and composition vary significantly with parent cell growth stage, culture conditions, and isolation methods (e.g., ultracentrifugation), causing batch-to-batch differences in potency and immunogenicity. Thus, standardized assays for purity, antigen integrity, unified production, isolation, characterization guidelines, and technologies, like microfluidic chips for automated modification, are urgently needed to ensure study reproducibility [79].
CMNVs should prioritize low immunogenicity and ease of sourcing for nanoprobe design in the future. As excellent natural carriers, genetically engineering source cells to intrinsically carry reporter molecules or therapeutics could enable theranostic integration.
Data availability statement
All data generated and/or analyzed during this study are included in this published article.
Author contributions
Lingling Lei: Investigation, Formal analysis, Writing – Original Draft. Meng Du: Conceptualization, Methodology, Supervision, Writing – Review & Editing. Jianjian Zhang: Conceptualization, Resources, Supervision, Writing – Review & Editing. Yuguang Mu: Validation, Supervision, Writing – Review & Editing.
Funding/Acknowledgment
This work was supported in part by the National Natural Science Foundation of China (grant number 82102054), the Clinical Research 4310 Program of the Affiliated Changsha Central Hospital of the University of South China (grant number 20214310NHYCG06), and the Health Research Project of Hunan Provincial Health Commission (grant number W20241010).
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Graphical abstract
Highlights
- Cell membrane vesicles overcome rapid immune clearance of nanoprobes.
- Inherited biofunctionalization enables precise targeting and enhanced imaging.
- Unified platform for fluorescence/bioluminescence/MRI/photoacoustic imaging applications.
- Scalability and stability challenges limit clinical translation.
In brief
CMNVs are biomimetic nanoplatforms overcoming rapid clearance and toxicity of traditional nanoprobes. Leveraging inherited membrane proteins or minimally disruptive modifications, CMNVs enhance targeting, prolong circulation, and improve imaging precision across fluorescence, bioluminescence, photoacoustic imaging, and MRI. Challenges in scalable production, stability, and standardization require resolution for clinical translation.
References
- Deng H, Li LX, Ju J, Mo X, Ge G, et al. Multifunctional nanoprobes for macrophage imaging. Biomaterials 2022;290:121824. [PMID: 36209580 DOI: 10.1016/j.biomaterials.2022.121824]
- Li S, Wei J, Yao Q, Song X, Xie J, et al. Emerging ultrasmall luminescent nanoprobes for in vivo bioimaging. Chem Soc Rev 2023;52(5):1672-96. [PMID: 36779305 DOI: 10.1039/d2cs00497f]
- Xiao W, Gao H. The impact of protein corona on the behavior and targeting capability of nanoparticle-based delivery system. Int J Pharm 2018;552(1-2):328-39. [PMID: 30308270 DOI: 10.1016/j.ijpharm.2018.10.011]
- Song Y, Feng D, Shi W, Li X, Ma H. Parallel comparative studies on the toxic effects of unmodified CdTe quantum dots, gold nanoparticles, and carbon nanodots on live cells as well as green gram sprouts. Talanta 2013;116:237-44. [PMID: 24148399 DOI: 10.1016/j.talanta.2013.05.022]
- Llop J, Lammers T. Nanoparticles for cancer diagnosis, radionuclide therapy and theranostics. ACS Nano 2021;15(11):16974-81. [PMID: 34748314 DOI: 10.1021/acsnano.1c09139]
- Zhou Y, Dai Z. New strategies in the design of nanomedicines to oppose uptake by the mononuclear phagocyte system and enhance cancer therapeutic efficacy. Chem Asian J 2018;13(22):3333-40. [PMID: 29441706 DOI: 10.1002/asia.201800149]
- Hu T, Huang Y, Liu J, Shen C, Wu F, et al. Biomimetic cell-derived nanoparticles: emerging platforms for cancer immunotherapy. Pharmaceutics 2023;15(7):1821. [PMID: 37514008 DOI: 10.3390/pharmaceutics15071821]
- Hong G, Robinson JT, Zhang Y, Diao S, Antaris AL, et al. In vivo fluorescence imaging with Ag2S quantum dots in the second near-infrared region. Angew Chem Int Ed Engl 2012;51(39):9818-21. [PMID: 22951900 DOI: 10.1002/anie.201206059]
- Choi HS, Liu W, Misra P, Tanaka E, Zimmer JP, et al. Renal clearance of quantum dots. Nat Biotechnol 2007;25(10):1165-70. [PMID: 17891134 DOI: 10.1038/nbt1340]
- Fitzpatrick JAJ, Andreko SK, Ernst LA, Waggoner AS, Ballou B, et al. Long-term persistence and spectral blue shifting of quantum dots in vivo. Nano Lett 2009;9(7):2736-41. [PMID: 19518087 DOI: 10.1021/nl901534q]
- Antaris AL, Chen H, Cheng K, Sun Y, Hong G, et al. A small-molecule dye for NIR-II imaging. Nat Mater 2016;15(2):235-42. [PMID: 26595119 DOI: 10.1038/nmat4476]
- Luo W, Dai Y, Chen Z, Yue X, Andrade-Powell KC, et al. Spatial and temporal tracking of cardiac exosomes in mouse using a nano-luciferase-CD63 fusion protein. Commun Biol 2020;3(1):114. [PMID: 32157172 DOI: 10.1038/s42003-020-0830-7]
- Gujrati V, Prakash J, Malekzadeh-Najafabadi J, Stiel A, Klemm U, et al. Bioengineered bacterial vesicles as biological nano-heaters for optoacoustic imaging. Nat Commun 2019;10(1):1114. [PMID: 30846699 DOI: 10.1038/s41467-019-09034-y]
- Zhu D, Li Y, Zhang Z, Xue Z, Hua Z, et al. Recent advances of nanotechnology-based tumor vessel-targeting strategies. J Nanobiotechnology 2021;19(1):435. [PMID: 34930293 DOI: 10.1186/s12951-021-01190-y]
- Lv Y, Liu M, Zhang Y, Wang X, Zhang F, et al. Cancer cell membrane-biomimetic nanoprobes with two-photon excitation and near-infrared emission for intravital tumor fluorescence imaging. ACS Nano 2018;12(2):1350-8. [PMID: 29338190 DOI: 10.1021/acsnano.7b07716]
- Legrand N, Huntington ND, Nagasawa M, Bakker AQ, Schotte R, et al. Functional CD47/signal regulatory protein alpha (SIRP(alpha)) interaction is required for optimal human T- and natural killer- (NK) cell homeostasis in vivo. Proc Natl Acad Sci U S A 2011;108(32):13224-9. [PMID: 21788504 DOI: 10.1073/pnas.1101398108]
- Pan S, Zhang Y, Huang M, Deng Z, Zhang A, et al. Urinary exosomes-based engineered nanovectors for homologously targeted chemo-chemodynamic prostate cancer therapy via abrogating EGFR/AKT/NF-kB/IkB signaling. Biomaterials 2021;275:120946. [PMID: 34119884 DOI: 10.1016/j.biomaterials.2021.120946]
- Reuveni T, Motiei M, Romman Z, Popovtzer A, Popovtzer R. Targeted gold nanoparticles enable molecular CT imaging of cancer: an in vivo study. Int J Nanomedicine 2011;6:2859-64. [PMID: 22131831 DOI: 10.2147/IJN.S25446]
- Khademi S, Sarkar S, Shakeri-Zadeh A, Attaran N, Kharrazi S, et al. Targeted gold nanoparticles enable molecular CT imaging of head and neck cancer: An in vivo study. Int J Biochem Cell Biol 2019;114:105554. [PMID: 31276787 DOI: 10.1016/j.biocel.2019.06.002]
- Ma L, Liu T-W, Wallig MA, Dobrucki IT, Dobrucki LW, et al. Efficient targeting of adipose tissue macrophages in obesity with polysaccharide nanocarriers. ACS Nano 2016;10(7):6952-62. [PMID: 27281538 DOI: 10.1021/acsnano.6b02878]
- Suk JS, Xu Q, Kim N, Hanes J, Ensign LM. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv Drug Deliv Rev 2016;99(Pt A):28-51. [PMID: 26456916 DOI: 10.1016/j.addr.2015.09.012]
- Zhang P, Sun F, Liu S, Jiang S. Anti-PEG antibodies in the clinic: Current issues and beyond PEGylation. J Control Release 2016;244(Pt B):184-93. [PMID: 27369864 DOI: 10.1016/j.jconrel.2016.06.040]
- Yang Q, Lai SK. Anti-PEG immunity: emergence, characteristics, and unaddressed questions. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2015;7(5):655-77. [PMID: 25707913 DOI: 10.1002/wnan.1339]
- Jarrett BR, Frendo M, Vogan J, Louie AY. Size-controlled synthesis of dextran sulfate coated iron oxide nanoparticles for magnetic resonance imaging. Nanotechnology 2007;18(3):035603. [PMID: 19636126 DOI: 10.1088/0957-4484/18/3/035603]
- Iacob AT, Lupascu FG, Apotrosoaei M, Vasincu IM, Tauser RG, et al. Recent biomedical approaches for chitosan based materials as drug delivery nanocarriers. Pharmaceutics 2021;13(4):587. [PMID: 33924046 DOI: 10.3390/pharmaceutics13040587]
- Yu M, Zheng J. Clearance pathways and tumor targeting of imaging nanoparticles. ACS Nano 2015;9(7):6655-74. [PMID: 26149184 DOI: 10.1021/acsnano.5b01320]
- Yan J, Wu Q, Zhao Z, Wu J, Ye H, et al. Light-assisted hierarchical intratumoral penetration and programmed antitumor therapy based on tumor microenvironment (TME)-amendatory and self-adaptive polymeric nanoclusters. Biomaterials 2020;255:120166. [PMID: 32544718 DOI: 10.1016/j.biomaterials.2020.120166]
- Li H-J, Du J-Z, Liu J, Du X-J, Shen S, et al. Smart superstructures with ultrahigh pH-sensitivity for targeting acidic tumor microenvironment: instantaneous size switching and improved tumor penetration. ACS Nano 2016;10(7):6753-61. [PMID: 27244096 DOI: 10.1021/acsnano.6b02326]
- Liu X, Liu Z, Dong K, Wu S, Sang Y, et al. Tumor-activatable ultrasmall nanozyme generator for enhanced penetration and deep catalytic therapy. Biomaterials 2020;258:120263. [PMID: 32798740 DOI: 10.1016/j.biomaterials.2020.120263]
- Liu Y, Lu Y, Zhu X, Li C, Yan M, et al. Tumor microenvironment-responsive prodrug nanoplatform via co-self-assembly of photothermal agent and IDO inhibitor for enhanced tumor penetration and cancer immunotherapy. Biomaterials 2020;242:119933. [PMID: 32151859 DOI: 10.1016/j.biomaterials.2020.119933]
- Han M, Huang-Fu M-Y, Guo W-W, Guo N-N, Chen J, et al. MMP-2-Sensitive HA end-conjugated poly(amidoamine) dendrimers via click reaction to enhance drug penetration into solid tumor. ACS Appl Mater Interfaces 2017;9(49):42459-70. [PMID: 29143522 DOI: 10.1021/acsami.7b10098]
- Huang J, Xian S, Liu Y, Chen X, Pu K, et al. A renally clearable activatable polymeric nanoprobe for early detection of hepatic ischemia-reperfusion injury. Adv Mater 2022;34(24):e2201357. [PMID: 35436014 DOI: 10.1002/adma.202201357]
- Ruan B, Yu M, Zhou Y, Xu W, Liu Y, et al. Size-transformable superoxide-triggered nanoreporters for crosstalk-free dual fluorescence/chemiluminescence imaging and urinalysis in living mice. Angew Chem Int Ed Engl 2023;62(31):e202305812. [PMID: 37258940 DOI: 10.1002/anie.202305812]
- De Jong WH, Hagens WI, Krystek P, Burger MC, Sips AJAM, et al. Particle size-dependent organ distribution of gold nanoparticles after intravenous administration. Biomaterials 2008;29(12):1912-9. [PMID: 18242692 DOI: 10.1016/j.biomaterials.2007.12.037]
- Yang Y, Yang T, Chen F, Zhang C, Yin B, et al. Degradable magnetic nanoplatform with hydroxide ions triggered photoacoustic, MR imaging, and photothermal conversion for precise cancer theranostic. Nano Lett 2022;22(8):3228-35. [PMID: 35380847 DOI: 10.1021/acs.nanolett.1c04804]
- Hwang J, Jeong Y, Park JM, Lee KH, Hong JW, et al. Biomimetics: forecasting the future of science, engineering, and medicine. Int J Nanomedicine 2015;10:5701-13. [PMID: 26388692 DOI: 10.2147/IJN.S83642]
- Kroll AV, Fang RH, Zhang L. Biointerfacing and applications of cell membrane-coated nanoparticles. Bioconjug Chem 2017;28(1):23-32. [PMID: 27798829 DOI: 10.1021/acs.bioconjchem.6b00569]
- Peng P, Hu J. [Erythrocytes-camouflaged nanoparticles: a promising delivery system for drugs and vaccines]. Sheng Wu Gong Cheng Xue Bao 2023;39(1):159-76. [PMID: 36738208 DOI: 10.13345/j.cjb.220166]
- Pasto A, Giordano F, Evangelopoulos M, Amadori A, Tasciotti E. Cell membrane protein functionalization of nanoparticles as a new tumor-targeting strategy. Clin Transl Med 2019;8(1):8. [PMID: 30877412 DOI: 10.1186/s40169-019-0224-y]
- Rao L, Bu L-L, Xu J-H, Cai B, Yu G-T, et al. Red blood cell membrane as a biomimetic nanocoating for prolonged circulation time and reduced accelerated blood clearance. Small 2015;11(46):6225-36. [PMID: 26488923 DOI: 10.1002/smll.201502388]
- Wei X, Gao J, Fang RH, Luk BT, Kroll AV, et al. Nanoparticles camouflaged in platelet membrane coating as an antibody decoy for the treatment of immune thrombocytopenia. Biomaterials 2016;111:116-23. [PMID: 27728811 DOI: 10.1016/j.biomaterials.2016.10.003]
- Xu C-H, Ye P-J, Zhou Y-C, He D-X, Wei H, et al. Cell membrane-camouflaged nanoparticles as drug carriers for cancer therapy. Acta Biomater 2020;105:1-14. [PMID: 32001369 DOI: 10.1016/j.actbio.2020.01.036]
- Choi MJ, Lee YK, Choi KC, Lee DH, Jeong HY, et al. Tumor-targeted erythrocyte membrane nanoparticles for theranostics of triple-negative breast cancer. Pharmaceutics 2023;15(2):350. [PMID: 36839675 DOI: 10.3390/pharmaceutics15020350]
- Li B, Wang F, Gui L, He Q, Yao Y, et al. The potential of biomimetic nanoparticles for tumor-targeted drug delivery. Nanomedicine (Lond) 2018;13(16):2099-118. [PMID: 30226404 DOI: 10.2217/nnm-2018-0017]
- Chen Y, Zhao G, Wang S, He Y, Han S, et al. Platelet-membrane-camouflaged bismuth sulfide nanorods for synergistic radio-photothermal therapy against cancer. Biomater Sci 2019;7(8):3450-9. [PMID: 31268067 DOI: 10.1039/c9bm00599d]
- Liu Y, Luo J, Chen X, Liu W, Chen T. Cell membrane coating technology: a promising strategy for biomedical applications. Nanomicro Lett 2019;11(1):100. [PMID: 34138027 DOI: 10.1007/s40820-019-0330-9]
- Gao C, Wu Z, Lin Z, Lin X, He Q. Polymeric capsule-cushioned leukocyte cell membrane vesicles as a biomimetic delivery platform. Nanoscale 2016;8(6):3548-54. [PMID: 26804725 DOI: 10.1039/c5nr08407e]
- Chen C, Song M, Du Y, Yu Y, Li C, et al. Tumor-associated-macrophage-membrane-coated nanoparticles for improved photodynamic immunotherapy. Nano Lett 2021;21(13):5522-31. [PMID: 34133181 DOI: 10.1021/acs.nanolett.1c00818]
- Zhen X, Cheng P, Pu K. Recent advances in cell membrane-camouflaged nanoparticles for cancer phototherapy. Small 2019;15(1):e1804105. [PMID: 30457701 DOI: 10.1002/smll.201804105]
- Leibacher J, Henschler R. Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells. Stem Cell Res Ther 2016;7:7. [PMID: 26753925 DOI: 10.1186/s13287-015-0271-2]
- Timaner M, Letko-Khait N, Kotsofruk R, Benguigui M, Beyar-Katz O, et al. Therapy-educated mesenchymal stem cells enrich for tumor-initiating cells. Cancer Res 2018;78(5):1253-65. [PMID: 29301792 DOI: 10.1158/0008-5472.CAN-17-1547]
- Rao L, Bu L-L, Cai B, Xu J-H, Li A, et al. Cancer cell membrane-coated upconversion nanoprobes for highly specific tumor imaging. Adv Mater 2016;28(18):3460-6. [PMID: 26970518 DOI: 10.1002/adma.201506086]
- Chen Z, Zhao P, Luo Z, Zheng M, Tian H, et al. Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dual-modal imaging and photothermal therapy. ACS Nano 2016;10(11):10049-57. [PMID: 27934074 DOI: 10.1021/acsnano.6b04695]
- Fang RH, Hu C-M, Chen KNH, Luk BT, Carpenter CW, et al. Lipid-insertion enables targeting functionalization of erythrocyte membrane-cloaked nanoparticles. Nanoscale 2013;5(19):8884-8. [PMID: 23907698 DOI: 10.1039/c3nr03064d]
- Dehaini D, Wei X, Fang RH, Masson S, Angsantikul P, et al. Erythrocyte-platelet hybrid membrane coating for enhanced nanoparticle functionalization. Adv Mater 2017;29(16):1606209. [PMID: 28199033 DOI: 10.1002/adma.201606209]
- Chugh V, Vijaya Krishna K, Pandit A. Cell membrane-coated mimics: a methodological approach for fabrication, characterization for therapeutic applications, and challenges for clinical translation. ACS Nano 2021;15(11):17080-123. [PMID: 34699181 DOI: 10.1021/acsnano.1c03800]
- Zeng Y, Li S, Zhang S, Wang L, Yuan H, et al. Cell membrane coated-nanoparticles for cancer immunotherapy. Acta Pharm Sin B 2022;12(8):3233-54. [PMID: 35967284 DOI: 10.1016/j.apsb.2022.02.023]
- Gangadaran P, Hong CM, Ahn B-C. An update on in vivo imaging of extracellular vesicles as drug delivery vehicles. Front Pharmacol 2018;9:169. [PMID: 29541030 DOI: 10.3389/fphar.2018.00169]
- Jia S, Zocco D, Samuels ML, Chou MF, Chammas R, et al. Emerging technologies in extracellular vesicle-based molecular diagnostics. Expert Rev Mol Diagn 2014;14(3):307-21. [PMID: 24575799 DOI: 10.1586/14737159.2014.893828]
- Vlassov AV, Magdaleno S, Setterquist R, Conrad R. Exosomes: current knowledge of their composition, biological functions, and diagnostic and therapeutic potentials. Biochim Biophys Acta 2012;1820(7):940-8. [PMID: 22503788 DOI: 10.1016/j.bbagen.2012.03.017]
- Nolte-’t Hoen E, Cremer T, Gallo RC, Margolis LB. Extracellular vesicles and viruses: are they close relatives? Proc Natl Acad Sci U S A 2016;113(33):9155-61. [PMID: 27432966 DOI: 10.1073/pnas.1605146113]
- Escrevente C, Keller S, Altevogt P, Costa J. Interaction and uptake of exosomes by ovarian cancer cells. BMC Cancer 2011;11:108. [PMID: 21439085 DOI: 10.1186/1471-2407-11-108]
- Kalimuthu S, Gangadaran P, Li XJ, Oh JM, Lee HW, et al. In Vivo therapeutic potential of mesenchymal stem cell-derived extracellular vesicles with optical imaging reporter in tumor mice model. Sci Rep 2016;6:30418. [PMID: 27452924 DOI: 10.1038/srep30418]
- Gangadaran P, Rajendran RL, Lee HW, Kalimuthu S, Hong CM, et al. Extracellular vesicles from mesenchymal stem cells activates VEGF receptors and accelerates recovery of hindlimb ischemia. J Control Release 2017;264:112-26. [PMID: 28837823 DOI: 10.1016/j.jconrel.2017.08.022]
- Bunggulawa EJ, Wang W, Yin T, Wang N, Durkan C, et al. Recent advancements in the use of exosomes as drug delivery systems. J Nanobiotechnology 2018;16(1):81. [PMID: 30326899 DOI: 10.1186/s12951-018-0403-9]
- Li X, Tsibouklis J, Weng T, Zhang B, Yin G, et al. Nano carriers for drug transport across the blood-brain barrier. J Drug Target 2017;25(1):17-28. [PMID: 27126681 DOI: 10.1080/1061186X.2016.1184272]
- Kim H, Jang H, Cho H, Choi J, Hwang KY, et al. Recent advances in exosome-based drug delivery for cancer therapy. Cancers (Basel) 2021;13(17):4435. [PMID: 34503245 DOI: 10.3390/cancers13174435]
- Kim H, Kim EH, Kwak G, Chi S-G, Kim SH, et al. Exosomes: cell-derived nanoplatforms for the delivery of cancer therapeutics. Int J Mol Sci 2020;22(1):14. [PMID: 33374978 DOI: 10.3390/ijms22010014]
- Hoshino A, Costa-Silva B, Shen T-L, Rodrigues G, Hashimoto A, et al. Tumour exosome integrins determine organotropic metastasis. Nature 2015;527(7578):329-35. [PMID: 26524530 DOI: 10.1038/nature15756]
- Frontiers Editorial Office. Retraction: targeting and therapy of glioblastoma in a mouse model using exosomes derived from natural killer cells. Front Immunol 2019;10:1770. [PMID: 31379887 DOI: 10.3389/fimmu.2019.01770]
- Qian R, Jing B, Jiang D, Gai Y, Zhu Z, et al. Multi-antitumor therapy and synchronous imaging monitoring based on exosome. Eur J Nucl Med Mol Imaging 2022;49(8):2668-81. [PMID: 35091755 DOI: 10.1007/s00259-022-05696-x]
- Kimiz-Gebologlu I, Oncel SS. Exosomes: large-scale production, isolation, drug loading efficiency, and biodistribution and uptake. J Control Release 2022;347:533-43. [PMID: 35597405 DOI: 10.1016/j.jconrel.2022.05.027]
- Ming-Kun C, Zi-Xian C, Mao-Ping C, Hong C, Zhuang-Fei C, et al. Engineered extracellular vesicles: a new approach for targeted therapy of tumors and overcoming drug resistance. Cancer Commun (Lond) 2024;44(2):205-25. [PMID: 38155418 DOI: 10.1002/cac2.12518]
- Yang D, Zhang W, Zhang H, Zhang F, Chen L, et al. Progress, opportunity, and perspective on exosome isolation – efforts for efficient exosome-based theranostics. Theranostics 2020;10(8):3684-707. [PMID: 32206116 DOI: 10.7150/thno.41580]
- Shen J, Ma Z, Xu J, Xue T, Lv X, et al. Exosome isolation and detection: from microfluidic chips to nanoplasmonic biosensor. ACS Appl Mater Interfaces 2024;16(18):22776-93. [PMID: 38676635 DOI: 10.1021/acsami.3c19396]
- Jeong S, Park J, Pathania D, Castro CM, Weissleder R, et al. Integrated magneto-electrochemical sensor for exosome analysis. ACS Nano 2016;10(2):1802-9. [PMID: 26808216 DOI: 10.1021/acsnano.5b07584]
- Li Y, Ma X, Yue Y, Zhang K, Cheng K, et al. Rapid surface display of mRNA antigens by bacteria-derived outer membrane vesicles for a personalized tumor vaccine. Adv Mater 2022;34(20):e2109984. [PMID: 35315546 DOI: 10.1002/adma.202109984]
- Li M, Zhou H, Yang C, Wu Y, Zhou X, et al. Bacterial outer membrane vesicles as a platform for biomedical applications: an update. J Control Release 2020;323:253-68. [PMID: 32333919 DOI: 10.1016/j.jconrel.2020.04.031]
- Xie J, Li Q, Haesebrouck F, Van Hoecke L, Vandenbroucke RE. The tremendous biomedical potential of bacterial extracellular vesicles. Trends Biotechnol 2022;40(10):1173-94. [PMID: 35581020 DOI: 10.1016/j.tibtech.2022.03.005]
- Kaparakis-Liaskos M, Ferrero RL. Immune modulation by bacterial outer membrane vesicles. Nat Rev Immunol 2015;15(6):375-87. [PMID: 25976515 DOI: 10.1038/nri3837]
- Li M, Li S, Zhou H, Tang X, Wu Y, et al. Chemotaxis-driven delivery of nano-pathogenoids for complete eradication of tumors post-phototherapy. Nat Commun 2020;11(1):1126. [PMID: 32111847 DOI: 10.1038/s41467-020-14963-0]
- Chen Q, Rozovsky S, Chen W. Engineering multi-functional bacterial outer membrane vesicles as modular nanodevices for biosensing and bioimaging. Chem Commun (Camb) 2017;53(54):7569-72. [PMID: 28636010 DOI: 10.1039/c7cc04246a]
- Kim OY, Park HT, Dinh NTH, Choi SJ, Lee J, et al. Bacterial outer membrane vesicles suppress tumor by interferon-γ-mediated antitumor response. Nat Commun 2017;8(1):626. [PMID: 28931823 DOI: 10.1038/s41467-017-00729-8]
- Xiang S, Khan A, Yao Q, Wang D. Recent advances in bacterial outer membrane vesicles: Effects on the immune system, mechanisms and their usage for tumor treatment. J Pharm Anal 2024;14(12):101049. [PMID: 39840399 DOI: 10.1016/j.jpha.2024.101049]
- Wang X, Quinn PJ. Lipopolysaccharide: biosynthetic pathway and structure modification. Prog Lipid Res 2010;49(2):97-107. [PMID: 19815028 DOI: 10.1016/j.plipres.2009.06.002]
- Collins SM, Brown AC. Bacterial outer membrane vesicles as antibiotic delivery vehicles. Front Immunol 2021;12:733064. [PMID: 34616401 DOI: 10.3389/fimmu.2021.733064]
- Needham BD, Carroll SM, Giles DK, Georgiou G, Whiteley M, et al. Modulating the innate immune response by combinatorial engineering of endotoxin. Proc Natl Acad Sci U S A 2013;110(4):1464-9. [PMID: 23297218 DOI: 10.1073/pnas.1218080110]
- Zhu Z, Antenucci F, Villumsen KR, Bojesen AM. Bacterial outer membrane vesicles as a versatile tool in vaccine research and the fight against antimicrobial resistance. mBio 2021;12(4):e0170721. [PMID: 34372691 DOI: 10.1128/mBio.01707-21]
- Orench-Rivera N, Kuehn MJ. Environmentally controlled bacterial vesicle-mediated export. Cell Microbiol 2016;18(11):1525-36. [PMID: 27673272 DOI: 10.1111/cmi.12676]
- Prados-Rosales R, Weinrick BC, Piqué DG, Jacobs WR Jr, Casadevall A, et al. Role for Mycobacterium tuberculosis membrane vesicles in iron acquisition. J Bacteriol 2014;196(6):1250-6. [PMID: 24415729 DOI: 10.1128/JB.01090-13]
- Keenan JI, Allardyce RA. Iron influences the expression of Helicobacter pylori outer membrane vesicle-associated virulence factors. Eur J Gastroenterol Hepatol 2000;12(12):1267-73. [PMID: 11192314 DOI: 10.1097/00042737-200012120-00002]
- Toyofuku M, Zhou S, Sawada I, Takaya N, Uchiyama H, et al. Membrane vesicle formation is associated with pyocin production under denitrifying conditions in Pseudomonas aeruginosa PAO1. Environ Microbiol 2014;16(9):2927-38. [PMID: 24112564 DOI: 10.1111/1462-2920.12260]
- Zariri A, Beskers J, van de Waterbeemd B, Hamstra HJ, Bindels THE, et al. Meningococcal outer membrane vesicle composition-dependent activation of the innate immune response. Infect Immun 2016;84(10):3024-33. [PMID: 27481244 DOI: 10.1128/IAI.00635-16]
- Macdonald IA, Kuehn MJ. Stress-induced outer membrane vesicle production by Pseudomonas aeruginosa. J Bacteriol 2013;195(13):2971-81. [PMID: 23625841 DOI: 10.1128/JB.02267-12]
- van de Waterbeemd B, Zomer G, van den Ijssel J, van Keulen L, Eppink MH, et al. Cysteine depletion causes oxidative stress and triggers outer membrane vesicle release by Neisseria meningitidis; implications for vaccine development. PLoS One 2013;8(1):e54314. [PMID: 23372704 DOI: 10.1371/journal.pone.0054314]
- Bernadac A, Gavioli M, Lazzaroni JC, Raina S, Lloubès R. Escherichia coli tol-pal mutants form outer membrane vesicles. J Bacteriol 1998;180(18):4872-8. [PMID: 9733690 DOI: 10.1128/JB.180.18.4872-4878.1998]
- Szczepaniak J, Press C, Kleanthous C. The multifarious roles of Tol-Pal in Gram-negative bacteria. FEMS Microbiol Rev 2020;44(4):490-506. [PMID: 32472934 DOI: 10.1093/femsre/fuaa018]
- Luzio JP, Pryor PR, Bright NA. Lysosomes: fusion and function. Nat Rev Mol Cell Biol 2007;8(8):622-32. [PMID: 17637737 DOI: 10.1038/nrm2217]
- Gujrati V, Lee M, Ko Y-J, Lee S, Kim D, et al. Bioengineered yeast-derived vacuoles with enhanced tissue-penetrating ability for targeted cancer therapy. Proc Natl Acad Sci U S A 2016;113(3):710-5. [PMID: 26715758 DOI: 10.1073/pnas.1509371113]
- Armstrong J. Yeast vacuoles: more than a model lysosome. Trends Cell Biol 2010;20(10):580-5. [PMID: 20685121 DOI: 10.1016/j.tcb.2010.06.010]
- Nguyen N-H, Nguyen N-T, Kim YH, Min J. Yeast-derived vacuoles as a novel carrier with enhanced hCMEC/D3 cell monolayer penetration. Biotechnol J 2023;18(2):e2200393. [PMID: 36321515 DOI: 10.1002/biot.202200393]
- Wickner W. Yeast vacuoles and membrane fusion pathways. EMBO J 2002;21(6):1241-7. [PMID: 11889030 DOI: 10.1093/emboj/21.6.1241]
- Zieger M, Mayer A. Yeast vacuoles fragment in an asymmetrical two-phase process with distinct protein requirements. Mol Biol Cell 2012;23(17):3438-49. [PMID: 22787281 DOI: 10.1091/mbc.E12-05-0347]
- Choi W, Shin W-R, Kim Y-H, Min J. Inducing a proinflammatory response with bioengineered yeast vacuoles with TLR2-binding peptides (VacT2BP) as a drug carrier for daunorubicin delivery. ACS Appl Mater Interfaces 2023;15(35):41258-70. [PMID: 37615983 DOI: 10.1021/acsami.3c06669]
- Nevoigt E. Progress in metabolic engineering of Saccharomyces cerevisiae. Microbiol Mol Biol Rev 2008;72(3):379-412. [PMID: 18772282 DOI: 10.1128/MMBR.00025-07]
- Liang Y, Duan L, Lu J, Xia J. Engineering exosomes for targeted drug delivery. Theranostics 2021;11(7):3183-95. [PMID: 33537081 DOI: 10.7150/thno.52570]
- Boudna M, Campos AD, Vychytilova-Faltejskova P, Machackova T, Slaby O, et al. Strategies for labelling of exogenous and endogenous extracellular vesicles and their application for in vitro and in vivo functional studies. Cell Commun Signal 2024;22(1):171. [PMID: 38461237 DOI: 10.1186/s12964-024-01548-3]
- Vader P, Mol EA, Pasterkamp G, Schiffelers RM. Extracellular vesicles for drug delivery. Adv Drug Deliv Rev 2016;106(Pt A):148-56. [PMID: 26928656 DOI: 10.1016/j.addr.2016.02.006]
- Anas A, Okuda T, Kawashima N, Nakayama K, Itoh T, et al. Clathrin-mediated endocytosis of quantum dot-peptide conjugates in living cells. ACS Nano 2009;3(8):2419-29. [PMID: 19653641 DOI: 10.1021/nn900663r]
- Wang Z, Mo H, He Z, Chen A, Cheng P. Extracellular vesicles as an emerging drug delivery system for cancer treatment: Current strategies and recent advances. Biomed Pharmacother 2022;153:113480. [PMID: 36076581 DOI: 10.1016/j.biopha.2022.113480]
- Jiang A, Nie W, Xie H-Y. In vivo imaging for the visualization of extracellular vesicle-based tumor therapy. ChemistryOpen 2022;11(9):e202200124. [PMID: 36101512 DOI: 10.1002/open.202200124]
- Jing B, Qian R, Jiang D, Gai Y, Liu Z, et al. Extracellular vesicles-based pre-targeting strategy enables multi-modal imaging of orthotopic colon cancer and image-guided surgery. J Nanobiotechnology 2021;19(1):151. [PMID: 34022897 DOI: 10.1186/s12951-021-00888-3]
- Sun J-X, Xu J-Z, An Y, Ma S-Y, Liu C-Q, et al. Future in precise surgery: fluorescence-guided surgery using EVs derived fluorescence contrast agent. J Control Release 2023;353:832-41. [PMID: 36496053 DOI: 10.1016/j.jconrel.2022.12.013]
- Han Z, Liu S, Pei Y, Ding Z, Li Y, et al. Highly efficient magnetic labelling allows MRI tracking of the homing of stem cell-derived extracellular vesicles following systemic delivery. J Extracell Vesicles 2021;10(3):e12054. [PMID: 33489014 DOI: 10.1002/jev2.12054]
- Yang Y, Zhang F. Molecular fluorophores for in vivo bioimaging in the second near-infrared window. Eur J Nucl Med Mol Imaging 2022;49(9):3226-46. [PMID: 35088125 DOI: 10.1007/s00259-022-05688-x]
- Duan Y, Liu B. Recent advances of optical imaging in the second near-infrared window. Adv Mater 2018;30(47):e1802394. [PMID: 30182451 DOI: 10.1002/adma.201802394]
- Su Y, Yu B, Wang S, Cong H, Shen Y. NIR-II bioimaging of small organic molecule. Biomaterials 2021;271:120717. [PMID: 33610960 DOI: 10.1016/j.biomaterials.2021.120717]
- Hong G, Diao S, Antaris AL, Dai H. Carbon nanomaterials for biological imaging and nanomedicinal therapy. Chem Rev 2015;115(19):10816-906. [PMID: 25997028 DOI: 10.1021/acs.chemrev.5b00008]
- Shiraki T, Miyauchi Y, Matsuda K, Nakashima N. Carbon nanotube photoluminescence modulation by local chemical and supramolecular chemical functionalization. Acc Chem Res 2020;53(9):1846-59. [PMID: 32791829 DOI: 10.1021/acs.accounts.0c00294]
- Bruns OT, Bischof TS, Harris DK, Franke D, Shi Y, et al. Next-generation in vivo optical imaging with short-wave infrared quantum dots. Nat Biomed Eng 2017;1:0056. [PMID: 29119058 DOI: 10.1038/s41551-017-0056]
- Li X, Zha M, Li Y, Ni J-S, Min T, et al. Sub-10 nm aggregation-induced emission quantum dots assembled by microfluidics for enhanced tumor targeting and reduced retention in the liver. Angew Chem Int Ed Engl 2020;59(49):21899-903. [PMID: 32841464 DOI: 10.1002/anie.202008564]
- Wang R, Zhou L, Wang W, Li X, Zhang F. In vivo gastrointestinal drug-release monitoring through second near-infrared window fluorescent bioimaging with orally delivered microcarriers. Nat Commun 2017;8:14702. [PMID: 28281530 DOI: 10.1038/ncomms14702]
- He S, Chen S, Li D, Wu Y, Zhang X, et al. High affinity to skeleton rare earth doped nanoparticles for near-infrared II imaging. Nano Lett 2019;19(5):2985-92. [PMID: 30983358 DOI: 10.1021/acs.nanolett.9b00140]
- Hemmateenejad B, Shamsipur M, Khosousi T, Shanehsaz M, Firuzi O. Antioxidant activity assay based on the inhibition of oxidation and photobleaching of L-cysteine-capped CdTe quantum dots. Analyst 2012;137(17):4029-36. [PMID: 22814421 DOI: 10.1039/c2an35588d]
- Shamsipur M, Rajabi HR. Pure zinc sulfide quantum dot as highly selective luminescent probe for determination of hazardous cyanide ion. Mater Sci Eng C Mater Biol Appl 2014;36:139-45. [PMID: 24433896 DOI: 10.1016/j.msec.2013.12.001]
- Xu Z, Huang H, Xiong X, Wei X, Guo X, et al. A near-infrared light-responsive extracellular vesicle as a “Trojan horse” for tumor deep penetration and imaging-guided therapy. Biomaterials 2021;269:120647. [PMID: 33450584 DOI: 10.1016/j.biomaterials.2020.120647]
- Chen L-L, Zhao L, Wang Z-G, Liu S-L, Pang D-W. Near-infrared-II quantum dots for in vivo imaging and cancer therapy. Small 2022;18(8):e2104567. [PMID: 34837314 DOI: 10.1002/smll.202104567]
- Liu A-A, Cui R, Zong X, Jia J, Hu Y, et al. Live-cell synthesis of biocompatible quantum dots. Nat Protoc 2025;20(7):1884-914. [PMID: 40097832 DOI: 10.1038/s41596-024-01133-5]
- Zhou J, Yang Y, Zhang CY. Toward biocompatible semiconductor quantum dots: from biosynthesis and bioconjugation to biomedical application. Chem Rev 2015;115(21):11669-717. [PMID: 26446443 DOI: 10.1021/acs.chemrev.5b00049]
- Liu A-A, Sun E-Z, Wang Z-G, Liu S-L, Pang D-W. Artificially regulated synthesis of nanocrystals in live cells. Natl Sci Rev 2021;9(6):nwab162. [PMID: 35874310 DOI: 10.1093/nsr/nwab162]
- Hong Z-Y, Liu H-M, Yu Z-L, Chen G, Wu M. Nanometer-sized and near-infrared-II fluorescent extracellular vesicles for noninvasive high-contrast imaging of tumors with high EGFR expression. ACS Appl Nano Mater 2023;6(13):11481-90. [DOI: 10.1021/acsanm.3c01527]
- Li S, Ruan Z, Zhang H, Xu H. Recent achievements of bioluminescence imaging based on firefly luciferin-luciferase system. Eur J Med Chem 2021;211:113111. [PMID: 33360804 DOI: 10.1016/j.ejmech.2020.113111]
- England CG, Ehlerding EB, Cai W. NanoLuc: a small luciferase is brightening up the field of bioluminescence. Bioconjug Chem 2016;27(5):1175-87. [PMID: 27045664 DOI: 10.1021/acs.bioconjchem.6b00112]
- Coralli C, Cemazar M, Kanthou C, Tozer GM, Dachs GU. Limitations of the reporter green fluorescent protein under simulated tumor conditions. Cancer Res 2001;61(12):4784-90. [PMID: 11406553]
- Contag CH, Bachmann MH. Advances in in vivo bioluminescence imaging of gene expression. Annu Rev Biomed Eng 2002;4:235-60. [PMID: 12117758 DOI: 10.1146/annurev.bioeng.4.111901.093336]
- Welsh DK, Noguchi T. Cellular bioluminescence imaging. Cold Spring Harb Protoc 2012;2012(8):pdb.top070607. [PMID: 22854570 DOI: 10.1101/pdb.top070607]
- Harris DA, Patel SH, Gucek M, Hendrix A, Westbroek W, et al. Exosomes released from breast cancer carcinomas stimulate cell movement. PLoS One 2015;10(3):e0117495. [PMID: 25798887 DOI: 10.1371/journal.pone.0117495]
- Setti M, Osti D, Richichi C, Ortensi B, Del Bene M, et al. Extracellular vesicle-mediated transfer of CLIC1 protein is a novel mechanism for the regulation of glioblastoma growth. Oncotarget 2015;6(31):31413-27. [PMID: 26429879 DOI: 10.18632/oncotarget.5105]
- Rodríguez M, Silva J, Herrera A, Herrera M, Peña C, et al. Exosomes enriched in stemness/metastatic-related mRNAS promote oncogenic potential in breast cancer. Oncotarget 2015;6(38):40575-87. [PMID: 26528758 DOI: 10.18632/oncotarget.5818]
- Hood JL, San RS, Wickline SA. Exosomes released by melanoma cells prepare sentinel lymph nodes for tumor metastasis. Cancer Res 2011;71(11):3792-801. [PMID: 21478294 DOI: 10.1158/0008-5472.CAN-10-4455]
- Gangadaran P, Li XJ, Lee HW, Oh JM, Kalimuthu S, et al. A new bioluminescent reporter system to study the biodistribution of systematically injected tumor-derived bioluminescent extracellular vesicles in mice. Oncotarget 2017;8(66):109894-914. [PMID: 29299117 DOI: 10.18632/oncotarget.22493]
- Hikita T, Miyata M, Watanabe R, Oneyama C. Sensitive and rapid quantification of exosomes by fusing luciferase to exosome marker proteins. Sci Rep 2018;8(1):14035. [PMID: 30232365 DOI: 10.1038/s41598-018-32535-7]
- Huang Y, Beringhs AO, Chen Q, Song D, Chen W, et al. Genetically engineered bacterial outer membrane vesicles with expressed nanoluciferase reporter for in vivo bioluminescence kinetic modeling through noninvasive imaging. ACS Appl Bio Mater 2019;2(12):5608-15. [PMID: 35021555 DOI: 10.1021/acsabm.9b00690]
- Zeng Y, Dou T, Ma L, Ma J. Biomedical photoacoustic imaging for molecular detection and disease diagnosis: “always-on” and “turn-on” probes. Adv Sci (Weinh) 2022;9(25):e2202384. [PMID: 35773244 DOI: 10.1002/advs.202202384]
- Jiang Y, Pu K. Advanced photoacoustic imaging applications of near-infrared absorbing organic nanoparticles. Small 2017;13(30). [PMID: 28597608 DOI: 10.1002/smll.201700710]
- Fu Q, Zhu R, Song J, Yang H, Chen X. Photoacoustic imaging: contrast agents and their biomedical applications. Adv Mater 2019;31(6):e1805875. [PMID: 30556205 DOI: 10.1002/adma.201805875]
- Chaudhary Z, Khan GM, Abeer MM, Pujara N, Wan-Chi Tse B, et al. Efficient photoacoustic imaging using indocyanine green (ICG) loaded functionalized mesoporous silica nanoparticles. Biomater Sci 2019;7(12):5002-15. [PMID: 31617526 DOI: 10.1039/c9bm00822e]
- Li S, Jiang W, Yuan Y, Sui M, Yang Y, et al. Delicately designed cancer cell membrane-camouflaged nanoparticles for targeted 19F MR/PA/FL imaging-guided photothermal therapy. ACS Appl Mater Interfaces 2020;12(51):57290-301. [PMID: 33231083 DOI: 10.1021/acsami.0c13865]
- Stritzker J, Kirscher L, Scadeng M, Deliolanis NC, Morscher S, et al. Vaccinia virus-mediated melanin production allows MR and optoacoustic deep tissue imaging and laser-induced thermotherapy of cancer. Proc Natl Acad Sci U S A 2013;110(9):3316-20. [PMID: 23401518 DOI: 10.1073/pnas.1216916110]
- Fan Q, Cheng K, Hu X, Ma X, Zhang R, et al. Transferring biomarker into molecular probe: melanin nanoparticle as a naturally active platform for multimodality imaging. J Am Chem Soc 2014;136(43):15185-94. [PMID: 25292385 DOI: 10.1021/ja505412p]
- Jang Y, Kim H, Yoon S, Lee H, Hwang J, et al. Exosome-based photoacoustic imaging guided photodynamic and immunotherapy for the treatment of pancreatic cancer. J Control Release 2021;330:293-304. [PMID: 33359580 DOI: 10.1016/j.jconrel.2020.12.039]
- Lu Z-R, Laney V, Li Y. Targeted contrast agents for magnetic resonance molecular imaging of cancer. Acc Chem Res 2022;55(19):2833-47. [PMID: 36121350 DOI: 10.1021/acs.accounts.2c00346]
- Sanvicens N, Marco MP. Multifunctional nanoparticles–properties and prospects for their use in human medicine. Trends Biotechnol 2008;26(8):425-33. [PMID: 18514941 DOI: 10.1016/j.tibtech.2008.04.005]
- Hohnholt MC, Geppert M, Dringen R. Treatment with iron oxide nanoparticles induces ferritin synthesis but not oxidative stress in oligodendroglial cells. Acta Biomater 2011;7(11):3946-54. [PMID: 21763792 DOI: 10.1016/j.actbio.2011.06.052]
- Du C, Liu X, Hu H, Li H, Yu L, et al. Dual-targeting and excretable ultrasmall SPIONs for T1-weighted positive MR imaging of intracranial glioblastoma cells by targeting the lipoprotein receptor-related protein. J Mater Chem B 2020;8(11):2296-306. [PMID: 32100784 DOI: 10.1039/c9tb02391g]
- Rayamajhi S, Marasini R, Nguyen TDT, Plattner BL, Biller D, et al. Strategic reconstruction of macrophage-derived extracellular vesicles as a magnetic resonance imaging contrast agent. Biomater Sci 2020;8(10):2887-904. [PMID: 32300771 DOI: 10.1039/d0bm00128g]
- Hu L, Wickline SA, Hood JL. Magnetic resonance imaging of melanoma exosomes in lymph nodes. Magn Reson Med 2015;74(1):266-71. [PMID: 25052384 DOI: 10.1002/mrm.25376]
- Wang J, Zhu X, Li C, Cai L, Pei W, et al. Efficient EVs extraction through the conjugation of superparamagnetic iron oxide nanoparticles for the targeted delivery in rat brain. Mater Today Chem 2022;23.
- Kutchy NA, Ma R, Liu Y, Buch S, Hu G. Extracellular vesicle-mediated delivery of ultrasmall superparamagnetic iron oxide nanoparticles to mice brain. Front Pharmacol 2022;13:819516. [PMID: 35462907 DOI: 10.3389/fphar.2022.819516]
- Wang J, Li W, Lu Z, Zhang L, Hu Y, et al. The use of RGD-engineered exosomes for enhanced targeting ability and synergistic therapy toward angiogenesis. Nanoscale 2017;9(40):15598-605. [PMID: 28990632 DOI: 10.1039/c7nr04425a]
- Tian T, Zhang H-X, He C-P, Fan S, Zhu Y-L, et al. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials 2018;150:137-49. [PMID: 29040874 DOI: 10.1016/j.biomaterials.2017.10.012]
- Jia G, Han Y, An Y, Ding Y, He C, et al. NRP-1 targeted and cargo-loaded exosomes facilitate simultaneous imaging and therapy of glioma in vitro and in vivo. Biomaterials 2018;178:302-16. [PMID: 29982104 DOI: 10.1016/j.biomaterials.2018.06.029]
- Chen L, Zhang G, Shi Y, Qiu R, Khan AA. Neuropilin-1 (NRP-1) and magnetic nanoparticles, a potential combination for diagnosis and therapy of gliomas. Curr Pharm Des 2015;21(37):5434-49. [PMID: 26377659 DOI: 10.2174/1381612821666150917092658]
- Chen L, Miao W, Tang X, Zhang H, Wang S, et al. Inhibitory effect of neuropilin-1 monoclonal antibody (NRP-1 MAb) on glioma tumor in mice. J Biomed Nanotechnol 2013;9(4):551-8. [PMID: 23621013 DOI: 10.1166/jbn.2013.1623]
- Galisova A, Zahradnik J, Allouche-Arnon H, Morandi MI, Abou Karam P, et al. Genetically engineered MRI-trackable extracellular vesicles as SARS-CoV-2 mimetics for mapping ACE2 binding in vivo. ACS Nano 2022;16(8):12276-89. [PMID: 35921522 DOI: 10.1021/acsnano.2c03119]
- Lüdtke-Buzug K, Haegele J, Biederer S, Sattel TF, Erbe M, et al. Comparison of commercial iron oxide-based MRI contrast agents with synthesized high-performance MPI tracers. Biomed Tech (Berl) 2013;58(6):527-33. [PMID: 23787462 DOI: 10.1515/bmt-2012-0059]
- Bulte JWM. Superparamagnetic iron oxides as MPI tracers: a primer and review of early applications. Adv Drug Deliv Rev 2019;138:293-301. [PMID: 30552918 DOI: 10.1016/j.addr.2018.12.007]
- Capuzzo AM, Piccolantonio G, Negri A, Bontempi P, Lacavalla MA, et al. Comparison between USPIOs and SPIOs for multimodal imaging of extracellular vesicles extracted from adipose tissue-derived adult stem cells. Int J Mol Sci 2024;25(17):9701. [PMID: 39273647 DOI: 10.3390/ijms25179701]
- Awanye AM, Chang C-M, Wheeler JX, Chan H, Marsay L, et al. Immunogenicity profiling of protein antigens from capsular group B Neisseria meningitidis. Sci Rep 2019;9(1):6843. [PMID: 31048732 DOI: 10.1038/s41598-019-43139-0]
- Rollier CS, Dold C, Marsay L, Linder A, Green CA, et al. Human B cell responses to dominant and subdominant antigens induced by a meningococcal outer membrane vesicle vaccine in a phase I trial. mSphere 2022;7(1):e0067421. [PMID: 35080470 DOI: 10.1128/msphere.00674-21]
- Witwer KW, Wolfram J. Extracellular vesicles versus synthetic nanoparticles for drug delivery. Nat Rev Mater 2021;6(2):103-6. [PMID: 36117545 DOI: 10.1038/s41578-020-00277-6]
- Xia Y, Zhang J, Liu G, Wolfram J. Immunogenicity of extracellular vesicles. Adv Mater 2024;36(33):e2403199. [PMID: 38932653 DOI: 10.1002/adma.202403199]
- Liu H, Zhang H, Han Y, Hu Y, Geng Z, et al. Bacterial extracellular vesicles-based therapeutic strategies for bone and soft tissue tumors therapy. Theranostics 2022;12(15):6576-94. [PMID: 36185613 DOI: 10.7150/thno.78034]