A Strategy for Antigen Amplification via Nanozyme-Catalyzed Proximity Labeling
1Med-X Research Institute and School of Biomedical Engineering, Shanghai Jiao Tong University, Shanghai 200030, China
2Department of Hepatobiliary Surgery and Transplantation, Liver Cancer Institute, Key Laboratory of Carcinogenesis and Cancer Invasion of Ministry of Education, Zhongshan Hospital, Fudan University, Shanghai, China
*Corresponding author: Yinghui Men, Med-X Research Institute and School of Biomedical Engineering, Shanghai Jiao Tong University, Shanghai, 200030, China. E-mail: menyinghui@sjtu.edu.cn
Published Online: September 15 2025
Cite this paper:
Men Y, Shao K, Wu Y. A Strategy for Antigen Amplification via Nanozyme-Catalyzed Proximity Labeling. BIO Integration 2025; 1–2.
DOI: 10.15212/bioi-2025-1003. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Proximity labeling (PL) is a broad strategy that enables tagging of proteins within defined spatiotemporal proximity through enzyme-catalyzed reactive intermediates [1, 2]. Classically deployed as a discovery tool to chart protein interaction networks at nanometer precision, PL has evolved into a versatile toolbox including peroxidases, biotin ligases, and tyrosinase [3, 4]. These approaches have transformed proteomics by enabling sensitive, spatially resolved interrogation of protein neighborhoods. Despite its widespread use in proteomic studies, PL has not been systematically explored for therapeutic purposes.
In a recent study in Nature, Li et al. first translated PL from a proteomic discovery tool into a therapeutic strategy, by applying it to enhance clustering-induced receptor activation and amplify downstream signaling [5]. Clustering of cell surface receptors after antigen engagement, a central trigger of cellular signaling, controls activation of T cell receptors [6]. Cancer immunotherapies exploit this principle through monoclonal antibodies, bispecific engagers, and engineered T cells. However, a major limitation persists, because most tumor-associated antigens are displayed at heterogeneous and frequently insufficient densities on the cell surface, thus leading to a weak response [7].
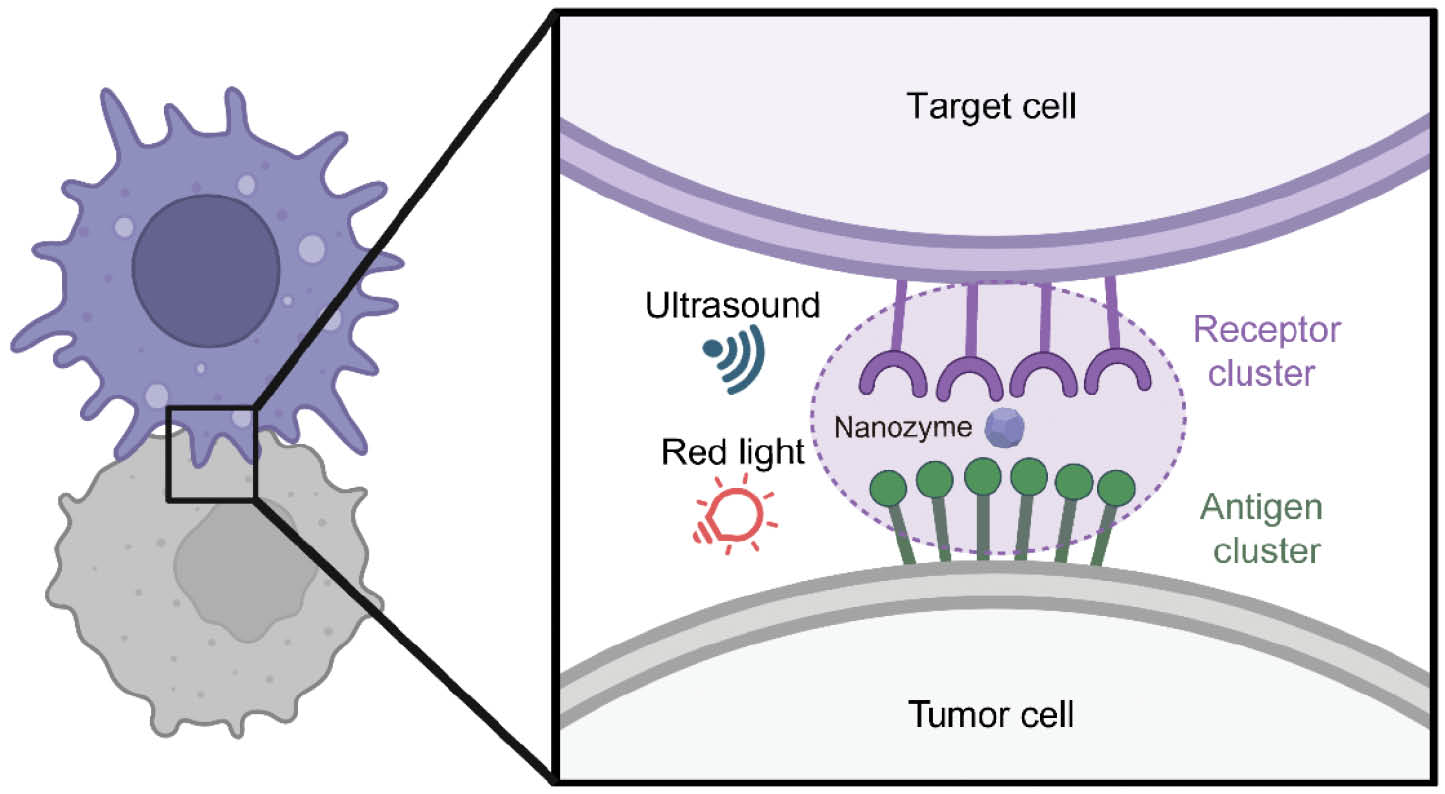
By creatively adapting proximity labeling for antigen engineering, the authors developed proximity antigen tagging of cytotoxic haptens (PATCH), a strategy that installs high-density synthetic antigen clusters directly on tumor cell surfaces under ultrasound or light control (Figure 1). PATCH overcomes several in vivo limitations of classical PL systems, including that peroxidases require highly toxic H2O2, biotin ligases require ATP, and blue-light photocatalysts have limited tissue penetration.
Figure 1 Schematic depicting proximity labeling-based antigen amplification.
In this study, Li et al. demonstrated that PATCH combines deep tissue penetration with high spatiotemporal specificity, thus enabling selective and efficient modulation of receptor clustering in vivo [5]. The authors synthesized a porphyrin-based porous coordination network (PCN) nanoparticle that functions as a nanozyme catalyst. In contrast to natural enzymes such as peroxidases or biotin ligases, which require exogenous gene expression and therefore involve long expression times and poor clinical feasibility, nanozymes can be directly applied without genetic manipulation and are more amenable to chemical modification [8, 9] The PCN nanozyme in this study was activated by red light and ultrasound, thereby producing free radicals and singlet oxygen that enabled efficient labeling of nearby proteins, such as bovine serum albumin. PATCH leverages red light and ultrasound as noninvasive stimuli to catalyze covalent, high-density amplification of synthetic antigens (FITC) in spatial proximity to target antigens. These synthetic antigen patches serve as potent scaffolds for FITC-binding bispecific T cell engagers, thus inducing robust T cell activation and tumor cell killing. In vivo, PATCH eradicated established tumors in mouse models, induced abscopal effects against untreated distal lesions, and generated durable immune memory protective against rechallenge.
Ultrasound, a widely used non-invasive modality with excellent tissue penetration, was first used in PL therapy to treat solid tumors. Unlike photocatalytic reactions, which are often limited by the shallow penetration of light into biological tissues, ultrasound can reach deep-seated tumors while causing minimal damage to surrounding healthy structures. This property not only facilitates treatment of deep-seated tumors but also enables precise spatiotemporal control over catalytic activity. Collectively, these attributes make ultrasound a promising modality to broaden the clinical applicability of PL, particularly for solid tumors that are otherwise inaccessible through conventional approaches.
Conceptually, PATCH reframes the approach to antigen targeting. Instead of engineering increasingly complex receptors to recognize sparse antigens, PATCH modifies the antigenic environment itself, by enhancing antigen density and distribution, and consequently drives receptor clustering. This inversion of the traditional paradigm marks a major step toward engineering the extracellular environment to control immune cell fate.
Despite these advances, several challenges must be addressed before clinical translation of PATCH. First, pharmacokinetics and scalability require validation in large animal models, particularly for systemic delivery and repeated dosing. Second, translational barriers, including regulatory considerations, scalable manufacturing of catalytic probes, and long-term biocompatibility, must be addressed to enable eventual clinical application.
PATCH highlights the therapeutic potential of proximity chemistry beyond proteomic discovery. In oncology, it could potentially be integrated with immune checkpoint inhibitors, adoptive T cell therapy, or antibody–drug conjugates to enhance treatment efficacy. In autoimmunity, selective amplification of tolerogenic ligands might recalibrate signaling thresholds. In addition, the PCN nanozyme provides a tunable platform that could be further engineered for enhanced catalytic efficiency, selective activation, and improved pharmacological properties, thereby expanding its utility across diverse biomedical applications.
In summary, the light/ultrasound-activated nanozyme–catalyzed PATCH extends the scope of PL from a proteomic discovery tool to a potential strategy for therapeutic antigen engineering. By amplifying receptor clustering with spatiotemporal precision, PATCH provides a framework for precise modulation of immune responses in vivo. Continued development of PL technologies and their integration into therapeutic settings may broaden the utility of chemistry and biology, and offer new opportunities for translational immunology and related biomedical fields.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
References
- Li J, Han S, Li H, Udeshi ND, Svinkina T, et al. Cell-surface proteomic profiling in the fly brain uncovers wiring regulators. Cell 2020;180(2):373-86.e15. [PMID: 31955847 DOI: 10.1016/j.cell.2019.12.029]
- Fazal FM, Han S, Parker KR, Kaewsapsak P, Xu J, et al. Atlas of subcellular RNA localization revealed by APEX-Seq. Cell 2019;178(2):473-90.e26. [PMID: 31230715 DOI: 10.1016/j.cell.2019.05.027]
- Maza JC, Bader DLV, Xiao L, Marmelstein AM, Brauer DD, et al. Enzymatic modification of N-terminal proline residues using phenol derivatives. J Am Chem Soc 2019;141(9):3885-92. [PMID: 30726077 DOI: 10.1021/jacs.8b10845]
- Qin W, Cho KF, Cavanagh PE, Ting AY. Deciphering molecular interactions by proximity labeling. Nature Methods. 2021;18(2):133-43. [PMID: 33432242 DOI: 10.1038/s41592-020-01010-5]
- Li S, Men Y, Wang Z, Wu Y, Sun H, et al. Amplifying antigen-induced cellular responses with proximity labelling. Nature. 2025. PMID: 40931066 DOI: 10.1038/s41586-025-09518-6.
- Su X, Ditlev JA, Hui E, Xing W, Banjade S, et al. Phase separation of signaling molecules promotes T cell receptor signal transduction. Science 2016;352(6285):595-9. [PMID: 27056844 DOI: 10.1126/science.aad9964]
- Velders MP, van Rhijn CM, Oskam E, Fleuren GJ, Warnaar SO, et al. The impact of antigen density and antibody affinity on antibody-dependent cellular cytotoxicity: relevance for immunotherapy of carcinomas. Br J Cancer 1998;78(4):478-83. [PMID: 9716030 DOI: 10.1038/bjc.1998.518]
- Li S, Li J, Du M, Deng G, Song Z, et al. Efficient gene silencing in intact plant cells using siRNA delivered by functional graphene oxide nanoparticles. Angew Chem Int Ed Engl 2022;61(40):e202210014. [PMID: 35921481 DOI: 10.1002/anie.202210014]
- Li S, Long Y, Deng G, Men Y, Lu F, et al. Manganese-based nanozyme enabled efficient mitigation of Huanglongbing-induced oxidative damage in Citrus. Environ Sci Nano 2025;12(1):701-15. [DOI: 10.1039/D4EN00519H]