Role of Photofunctionalization in Orthopedic Customized Implants
1Sports Medicine Service, Peking University Fourth School of Clinical Medicine, No. 31, Xinjiekou East Street, Beijing 100035, China
2Sports Medicine Service, Beijing Jishuitan Hospital, No. 31, Xinjiekou East Street, Beijing 100035, China
3Thayer School of Engineering, Dartmouth College, Hanover, NH 03755, USA
4Department of Ophthalmology and Visual Sciences and Wisconsin Institute for Discovery, University of Wisconsin-Madison, Madison, Wisconsin 53706, USA
5Shanghai Jiao Tong University School of Medicine, Shanghai Ninth People’s Hospital, 639 Zhizaoju Road, Huangpu District, Shanghai, China
*Correspondence to: Chuan Yin, Shanghai Jiao Tong University School of Medicine, Shanghai Ninth People’s Hospital, 639 Zhizaoju Road, Huangpu District, Shanghai, China, Tel: +86-010-18811770509. E-mail: yinchuan123@pku.org.cn
Received: May 14 2025; Revised: August 10 2025; Accepted: August 21 2025; Published Online: September 8 2025
Cite this paper:
Liu Y, Li P, Li X et al. Role of Photofunctionalization in Orthopedic Customized Implants. BIO Integration 2025; 1–6.
DOI: 10.15212/bioi-2025-0094. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Ultraviolet (UV) photofunctionalization is a promising surface activation strategy for orthopedic implants that offers a safe, coating-free, and intraoperatively applicable approach to enhance bone implant integration. This commentary evaluates the translational potential of UV photofuntionalization and outlines key mechanisms and applications. By removing hydrocarbons and increasing hydrophilicity and protein adsorption, UV treatment transforms titanium into a biologically active interface that accelerates osteoblast attachment, proliferation, and differentiation. Evidence from in vitro and in vivo models, including diabetic and osteoporotic conditions, demonstrates consistently improved osseointegration, while early clinical data indicate reduced healing time and enhanced implant stability, especially in spinal fusion and large bone defect reconstruction. Moreover, UV photofunctionalization synergizes with advanced implant technologies, such as 3D-printed porous scaffolds, ion-doped alloys, and polymer–metal composites, enhancing biological performance without altering geometry or introducing chemical agents. Despite challenges in standardization and workflow integration, the intraoperative applicability and favorable safety profile of UV photofunctionalization support readiness for clinical adoption. Given the demand for effective strategies in complex or biologically compromised bone reconstructions, UV photofunctionalization represents a novel, additive-free, deployable method that significantly improves osseointegration across implant materials, even under high-risk conditions, ultimately enhancing outcomes and reducing implant failure.
Keywords
Bone defects, osseointegration, photofunctionalization, surface modification.
Introduction
Achieving stable and predictable bone implant integration remains a key goal in orthopaedic surgery, particularly for large bone defects or patients with compromised bone quality. Conditions, such as osteoporosis and diabetes, impair osseointegration, prolong healing, and increase implant failure risk. The advent of 3D-printed, patient-specific titanium implants has improved anatomic fit in complex reconstructions but the biological inertness of titanium alloys remains a challenge, especially in compromised environments. Conventional strategies, such as hydroxyapatite coatings or bioactive ceramics, offer early fixation advantages but increase fabrication complexity, cost, and potential biocompatibility concerns due to exogenous materials [1]. These limitations highlight the demand for a biologically effective, additive-free surface treatment.
Ultraviolet (UV) photofunctionalization has emerged as a promising solution for osteointegration. This technique removes surface contaminants and restores the bioactivity of titanium by enhancing hydrophilicity and protein affinity, thereby promoting osteogenic cell adhesion. Notably, UV alters only surface physicochemistry without affecting macrostructure or requiring coatings, thus enabling intraoperative use with minimal workflow disruption [2]. Among existing techniques, UV photofunctionalization is the only clinically feasible method for real-time surface bioactivation. A growing body of preclinical and early clinical evidence supports the efficacy of UV photfunctionalization, demonstrating improved cell attachment, faster bone formation, and enhanced stability, even under impaired healing. Clinically, UV-treated implants have reached functional loading nearly twice as fast as untreated controls. Although much current data derives from dental and preclinical models, outcomes suggest strong translational potential in orthopaedics, particularly for revision cases and segmental reconstructions requiring accelerated integration.
Titanium and its alloys are widely used in orthopaedics due to exceptional mechanical properties and biocompatibility. Among metallic biomaterials, titanium stands out for its high strength-to-weight ratio, low density, and corrosion resistance, making titanium ideal for long-term implantation. These materials also form a passive oxide layer that further enhances resistance to corrosion and ion release. In addition to orthopaedics, the physical and chemical properties have led to applications in aerospace and marine industries. However, despite these advantages, titanium remains biologically inert, necessitating surface modifications, such as UV photofunctionalization, to improve early adhesion and integration.
This commentary highlights the translational potential of UV photofunctionalization in orthopaedics. We outline the biological mechanisms through which UV enhances implant activity, review in vitro, in vivo, and clinical evidence, and discuss challenges, such as standardization and workflow integration. We also explore how UV complements emerging strategies, including porous 3D-printed scaffolds, ion-doped surfaces, and hybrid composites, to improve outcomes. A forward-looking perspective is provided, emphasizing how this light-based, additive-free technique may redefine the bone–implant interface in orthopedic practice.
Mechanistic insights: how UV photofunctionalization enhances implant surfaces
Biological aging of titanium and the rationale for UV photofunctionalization
Titanium surfaces undergo a phenomenon over time known as “biological aging,” which significantly reduces osteoconductivity. This process involves the accumulation of hydrocarbon contaminants from the environment, leading to increased hydrophobicity and impaired protein adsorption [3]. Studies have shown that freshly prepared titanium surfaces support nearly 90% osseointegration [4], while 4-week aged surfaces demonstrate only approximately 60% integration. The decline in biological performance is also evident at the cellular level; aged titanium surfaces exhibit a 50% reduction in osteoblast attachment and proliferation compared to newly prepared ones [5]. Furthermore, the protein adsorption capacity of titanium decreases with aging, compromising initial cell–material interactions critical for osseointegration.
UV photofunctionalization effectively reverses the aging process. UV photofunctionalization restores hydrophilicity and enhances the surface ability to attract osteogenic proteins and cells through photodegradation of surface hydrocarbons. UV-treated titanium implants demonstrated significantly improved bone–implant contact and bone volume in a critical preclinical study using a gap healing model compared to untreated controls, even in regions with poor native bone contact. These findings provide a mechanistic rationale for UV treatment as a restorative surface strategy to counteract the time-dependent decline in bioactivity inherent to titanium materials.
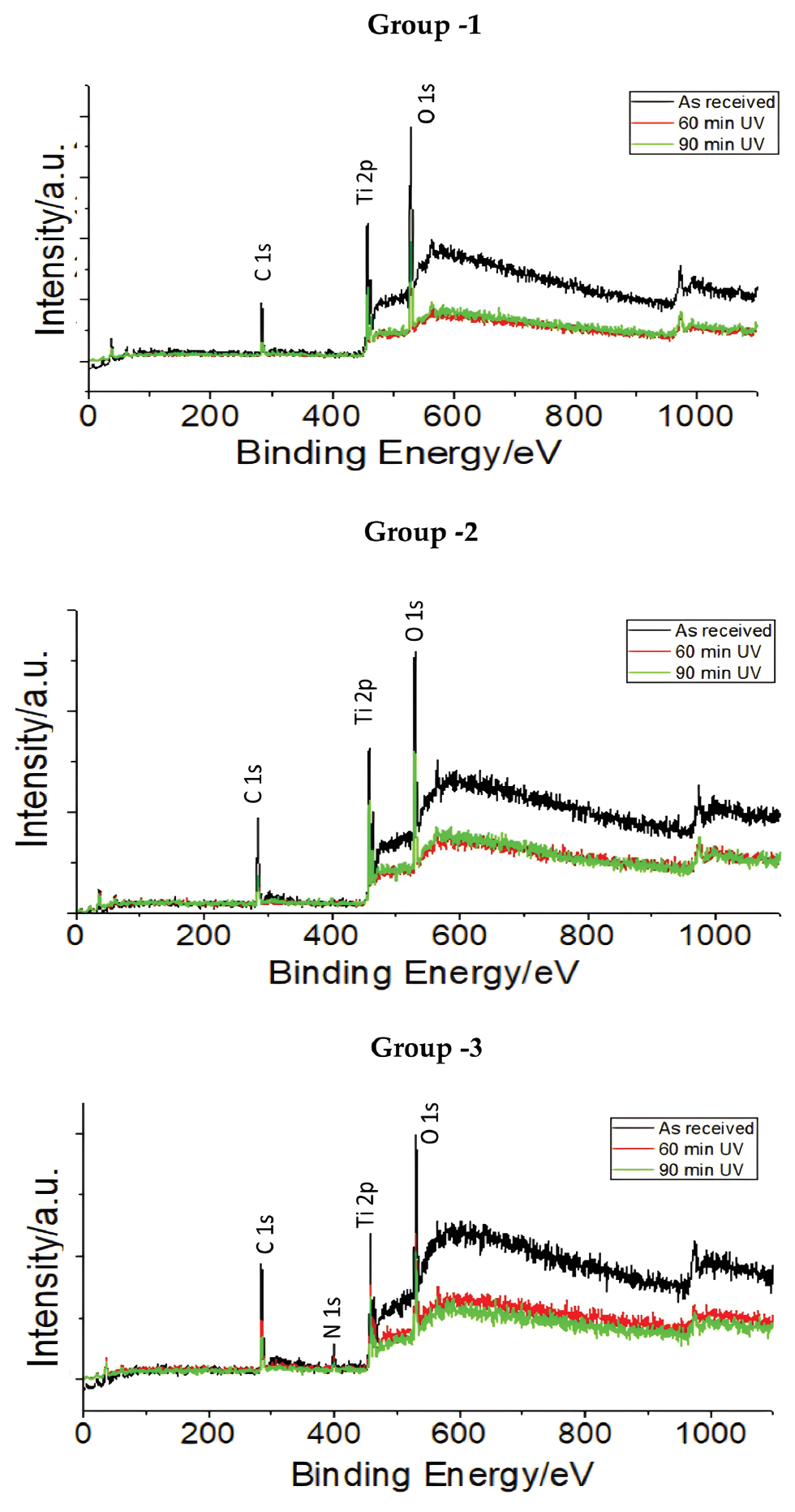
UV photofunctionalization induces a cascade of physicochemical and biological modifications that collectively improve the osteoconductivity of titanium and other implant materials. One of the primary effects of UV photofunctionalization is removal of surface-bound hydrocarbons, a process often referred to as reversing “biological aging.” Titanium exposed to ambient air accumulates a hydrophobic organic layer that inhibits cellular interactions. UVC irradiation (~254 nm) activates the photocatalytic properties of the titanium dioxide surface, generating reactive oxygen species (ROS) that break carbon–carbon and carbon–oxygen bonds. This process leads to the oxidative degradation and desorption of hydrocarbons [6], restoring the native titanium oxide layer and significantly increasing surface energy and wettability. This surface activation process is further evidenced by spectroscopic analysis, which demonstrates a decrease in surface carbon contamination and a shift from Ti4+ to Ti3+ states, reflecting photocatalytic-driven chemical changes following UV exposure (Figure 1). Following decontamination, the surface becomes super-hydrophilic, with water contact angles approaching zero. This result enhances absorption of biological fluids and promotes uniform adsorption of osteogenic proteins, such as fibronectin, vitronectin, albumin, and growth factors [7]. These proteins are derived from endogenous sources, eliminating the need for external coatings. The resulting protein layer creates a bioactive interface conducive to stable cell attachment and matrix development.
Figure 1 Spectroscopic confirmation of UV-induced photocatalytic surface activation on titanium implants. X-ray photoelectron spectroscopy (XPS) demonstrates the transformation of Ti4+ to Ti3+ and a marked reduction in hydrocarbon contamination after UV treatment, supporting the mechanism of photocatalysis-enhanced surface reactivity. Reproduced from ref [23]. Available under a CC-BY license. Copyright 2023 Kheur et al.
UV exposure also modulates the electrostatic properties of the titanium surface. Formation of Ti3+ sites and oxygen vacancies shifts the surface to a more negatively charged and hydroxylated state, enhancing interactions with positively charged protein domains and allowing divalent cations, like calcium, to bridge protein-surface and cell-surface interfaces [8]. These changes support osteoblast attachment, cytoskeletal organization, and the formation of mature focal adhesions, as demonstrated in in vitro studies that compared UV-treated and untreated surfaces. Once adherent, osteoblasts show increased metabolic activity, proliferation, and viability, which are benefits linked to reduced oxidative stress and enhanced mitochondrial function. These early responses drive osteogenic differentiation with upregulation of markers, like alkaline phosphatase (ALP), osteocalcin, and accelerated mineral matrix deposition. UV-treated surfaces initiate apatite formation within 24 h in simulated body fluid (SBF), far sooner than untreated controls, underscoring the superior mineralization potential.
Together, these effects promote rapid and intimate bone–implant contact, a process often termed “super-osseointegration.” UV photofunctionalization enhances surface cleanliness, hydrophilicity, protein adsorption, and electrostatics, turning bioinert titanium into a highly osteoinductive platform. Importantly, this transformation is achieved without altering implant geometry or applying chemical coatings, thus preserving mechanical integrity and simplifying clinical use. UV-treated surfaces perform as well as or better than complex biomaterial coatings in multiple comparative studies, even under systemic challenges, like diabetes or osteoporosis [9]. This combination of efficacy, safety, and practicality strongly supports the translational potential of UV photofunctionalization in orthopaedic surgery.
Translational challenges
Despite a strong preclinical foundation, UV photofunctionalization has yet to be adopted as a routine orthopaedic tool. A key limitation of UV photofunctionalization is the lack of standardized protocols. Current studies vary widely in UV wavelength, intensity, and exposure time (ranging from 12–20 min) making comparisons difficult and hindering regulatory alignment [2]. Establishing consensus on optimal treatment parameters, including dosage and lamp specifications, is essential. Even brief exposures have shown biological benefits, suggesting that practical protocols are achievable [2]. Another concern is the transient nature of surface activation. The super-hydrophilic and electrostatically charged surface generated by UV treatment begins to degrade upon re-exposure to ambient air, a reversion of the “biological aging” process. While some studies suggest that the effect can last several days-to-weeks [10], the most reliable approach remains intraoperative application, ensuring peak bioactivity at implantation. These challenges are primarily logistical, not scientific. With growing experimental evidence and the emergence of more user-friendly devices, the field is increasingly positioned to implement UV photofunctionalization, especially in cases requiring accelerated osseointegration, such as large bone defects or revision surgeries in compromised bone. Adoption in orthopaedics also lags behind dental applications, where UV has been widely shown to improve implant stability and bone contact. Barriers include the complex geometry and size of orthopaedic implants, which complicate uniform surface illumination and a traditional reliance on coatings or pharmacologic enhancers. While portable UV systems suitable for surgical use are becoming available, clinical uptake remains limited. Importantly, UV photofunctionalization introduces no foreign materials and avoids the risks of coatings or bioactive additives, offering a cleaner and potentially safer alternative. Regulatory clarity and orthopaedic-specific clinical trials will be key to broader adoption.
Recent advances in light-based surface activation have introduced vacuum ultraviolet (VUV) systems that enable complete photofunctionalization within 1 min. This high-energy light (~172 nm) accelerates hydrocarbon decomposition, achieving surface superhydrophilicity and protein adsorption faster than conventional UV treatment [11]. In addition to titanium, VUV also enhances soft tissue responses on zirconia and promotes oral fibroblast attachment, indicating broader material applicability [12]. These findings mark a critical step toward routine intraoperative use because rapid treatment times align better with surgical workflows and reduce contamination risk. In summary, the primary challenges in implementing UV photofunctionalization are logistical rather than scientific, given that this technique reliably amplifies implant bioactivity without requiring any coatings or structural modifications. As standardized protocols emerge and surgical familiarity grows, this additive-free, light-based method could seamlessly become a routine intraoperative tool, like the use of antibiotic bone cement or irrigation, and offering significant biological benefits with minimal procedural burden.
Synergy with advanced implant technologies
UV photofunctionalization is most impactful when used in conjunction with emerging implant technologies. Rather than functioning solely as a surface modification, UV photofunctionalization amplifies the bioactivity of modern implant designs without altering the geometry, chemistry, or manufacturing complexity. This compatibility enables UV photofunctionalization to complement strategies, such as additive manufacturing, ion-doped surfaces, and hybrid materials, addressing both mechanical and biological demands in challenging orthopaedic reconstructions [13].
Additive manufacturing facilitates the creation of patient-specific titanium implants with tailored porosity for large bone defects. While mechanically robust, 3D-printed titanium remains biologically inert post-fabrication. UV treatment enhances hydrophilicity across both external and internal lattice structures without compromising form or strength and improving cell adhesion and tissue ingrowth, even within deep pore networks. For example, Yin et al. demonstrated that an omnidirectional UV delivery system significantly improved osseointegration and bone fill in porous Ti implants relative to untreated controls [14]. Ion-doped materials incorporating elements, like copper, calcium, or strontium, promote osteogenesis and angiogenesis. UV photofunctionalization enhances these effects by modulating surface physicochemistry, optimizing ion release kinetics and protein adsorption. UV-treated copper-doped titanium shows superior vascular and osteogenic marker expression compared to either method alone. Similarly, combining calcium ion adsorption with UV leads to increased mineral deposition and early matrix formation, while UV alone improves protein adsorption, potentially reducing the need for high dopant concentrations. UV activation also expands the utility of polymers, such as PEEK, which suffers from poor bioactivity despite bone-mimicking mechanical properties [15]. UV irradiation increases PEEK wettability and protein affinity without altering mechanical integrity, leading to improved early-stage cell behavior. Comparable benefits have been observed in ceramics, like zirconia and metal–polymer composites, highlighting the adaptability of UV across diverse biomaterials.
Furthermore, UV photofunctionalization can enhance drug-loaded or growth factor–coated implants by improving surface affinity and biological signaling. This feature makes UV-treated surfaces promising platforms for future smart biomaterials, such as stimuli-responsive scaffolds or gene-activated constructs. By creating a clean, hydrophilic interface, UV treatment promotes molecular interactions and cellular recruitment essential for regenerative applications.
In summary, UV photofunctionalization serves as a versatile biological amplifier across implant platforms, ranging from 3D-printed scaffolds and ion-doped alloys-to-polymer composites. By instantly maximizing surface osteoconductivity without altering implant geometry or introducing exogenous materials, UV photofunctionalization ensures that diverse implants engage host tissue at peak bioactivity. Early studies combining UV with such advanced implant systems consistently report accelerated bone regeneration, faster healing, and fewer complications [16] compared to untreated controls. The unique intraoperative deployability and compatibility with patient-specific implants underscore the promise of UV photofunctionalization as a transformative adjunct in regenerative orthopaedics.
Clinical outlook and future directions
Although UV photofunctionalization is well-established in dental applications, growing evidence from in vitro, in vivo, and early clinical studies supports relevance in orthopaedics. UV-treated titanium consistently promotes osteoblast adhesion, proliferation, and differentiation at the cellular level. Increased alkaline phosphatase (ALP) activity and calcium deposition have been observed as early as 7–14 days after seeding. These effects are not surface-dependent and have been demonstrated on SLA-treated, sandblasted, and 3D-printed titanium, underscoring broad applicability [14].
Animal studies reinforce this finding. As summarized in Table 1 of the original review, models, such as rat femoral defects and rabbit tibial metaphysis, show that UV treatment significantly improves bone–implant contact (BIC), new bone formation, and mechanical strength (e.g., push-in force) within 4–6 weeks post-implantation. Hirota et al. further demonstrated that photofunctionalized titanium screws implanted in rat segmental femoral defects exhibited enhanced mechanical stability and faster bone bridging, even in large non-critical size defects [17]. In a related study, Park et al. engineered bone-implant interfaces using UV-treated titanium microfibers, which promoted early-stage osteointegration and cell alignment in vivo [18]. These results affirm UV photofunctionalization as a potent surface modification strategy in diverse preclinical platforms. In one study UV-treated Ti6Al4V implants yielded ~40% higher BIC and nearly doubled mechanical fixation compared to controls. These benefits persist even in osteoporotic or diabetic conditions, where UV activation improves trabecular architecture and peri-implant density and is likely due to enhanced local protein binding and pro-angiogenic effects [19]. These findings are beginning to translate clinically.
Table 1 In Vitro and In Vivo Studies Demonstrating the UV Photofunctionalization Effects on Osseointegration
| Year | Author | Model/Design | Implant/Material Type | UV Conditions | Key Findings |
|---|---|---|---|---|---|
| 2009 | Aita et al. [24] | In vitro + rat femur implant model | Commercially pure titanium | UV-C, 15 min | UV enhances osseointegration and cell adhesion on titanium surfaces. |
| 2014 | Hirota et al. [25] | In vitro + rat trabecular bone | Porous titanium | UV-C, 15 min | UV treatment improves bone volume and bone architecture in porous titanium. |
| 2016 | Hirota et al. [22] | In vitro + rat femoral defect model | Porous Ti implant | UV-C, 15 min | UV-treated implants increase trabecular infill and early bone formation. |
| 2017 | Kim et al. [9] | In vitro + rat | SLA-treated titanium discs | UV-C + Ozone, 10 min | UV + ozone synergistically promote mineralization and ALP activity. |
| 2024 | Wang et al. [26] | In vitro + rabbit femur defect | Cu-doped porous Ti (3D-printed) | UV-C, 15 min | UV + Cu-doped porous titanium shows enhanced hydrophilicity and integration. |
In addition to standard models, UV photofunctionalization has shown promising results under compromised healing conditions that mimic high-risk patient populations. In a rat model of osteoporosis, UV-treated titanium implants significantly improved mechanical anchorage and bone implant contact compared to untreated controls, demonstrating restored osseointegration despite systemic bone loss. Similarly, in a type 2 diabetes rat model, photofunctionalization mitigated the impaired healing response typically associated with hyperglycemia, leading to enhanced peri-implant bone volume and trabecular structure [20]. These findings underscore the robustness of UV surface activation in biologically challenging environments and strengthen its clinical relevance for elderly and metabolically compromised patients. As shown in Table 2, initial orthopaedic reports demonstrate early implant stability and rapid bone bridging with UV-treated implants. In spinal fusion, titanium cages exhibited early radiographic fusion, even in compromised patients. Preclinical studies on UV-treated titanium mesh also support its osteogenic efficacy. In a series of animal models, Hirota et al. reported enhanced bone volume and integration around photofunctionalized meshes used for guided bone regeneration and maxillofacial augmentation. For large defect reconstruction, porous UV-activated meshes achieved complete bony union in shorter-than-expected timelines, with intraoperative evidence of vascular ingrowth and favorable implant handling [21]. These clinical observations align with preclinical studies showing UV-treated surfaces upregulate VEGF and other angiogenic factors, suggesting added benefits beyond osseointegration. Additional clinical studies further support the translational maturity of UV photofunctionalization. In a 7-year prospective study, Hirota et al. demonstrated durable implant success and minimal complications with UV-treated surfaces, underscoring the long-term reliability [13]. A case-control study comparing regular and complex cases revealed enhanced early stability in UV-treated implants, even under challenging anatomical conditions [22]. Moreover, UV photofunctionalization was associated with reduced early implant failure rates, reinforcing its value in clinical risk mitigation.
Table 2 Clinical Evidence Supporting UV Photofunctionalization in Implant Applicationss
| Year | Author | Study Type | Implant/Material Type | UV Parameters | Orthopaedic Relevance/Conclusion |
|---|---|---|---|---|---|
| 2016 | Hirota et al. [22] | Controlled trial (25 vs. 24 patients) | Porous titanium implant | UV-C, 15 min | UV improved early bone formation and osseointegration across cases. |
| 2019 | Tominaga et al. [27] | Prospective controlled (lumbar fusion) | Spinal fusion titanium rod | UV-C, 15 min | First orthopaedic-focused UV application; showed modest benefit. |
| 2020 | Hirota et al. [13] | Single-center prospective (70 implants) | Titanium dental implants | UV-C, 15 min | UV effective even in cancer-related or complex bone conditions. |
| 2021 | Choi et al. [28] | Randomized clinical trial (low-density bone) | Titanium dental implant | UV-C, 15–20 min | UV improves early stability in compromised bone environments. |
Importantly, these outcomes are achieved without coatings, grafts, or specialized manufacturing. UV treatment is a residue-free, light-based method that can be applied intraoperatively, reactivating aged or processed implants at the point of care. This gives UV treatment logistical and regulatory advantages, especially for use in osteoporotic fracture repair, revision arthroplasty, or personalized 3D-printed implants. Compatibility with porous scaffolds, ion-integrated surfaces, and bioresponsive platforms makes UV treatment particularly well-suited for high-demand clinical scenarios. With continued evidence accumulation, UV photofunctionalization is well-positioned to become a standard intraoperative adjunct. As shown in Tables 1 and 2, the ability of UV photofunctionalization to accelerate cellular responses, promote earlier fixation, and function across diverse geometries offers a compelling rationale for broader use. Future prospective trials will be critical for defining indications and optimizing protocols.
In conclusion, UV photofunctionalization provides a novel, scientifically robust, and clinically adaptable strategy to boost the bioactivity of orthopaedic implant surfaces across a wide range of materials and designs. UV photofunctionalization uniquely achieves real-time surface activation without any chemical coatings or structural modifications and consistently enhances cellular responses and osseointegration, even in patient-specific 3D-printed scaffolds. These advantages, coupled with proven success in high-risk scenarios (including osteoporosis, diabetes, severe bone loss, and large bone defects), position UV photofunctionalization as a strong candidate for routine surgical use. By significantly improving implant integration and success rates in these challenging contexts, this additive-free approach promises to reduce revision surgeries, accelerate healing, and ultimately improve patient outcomes. As healthcare systems increasingly prioritize treatments that are both cost-effective and biologically efficacious, UV photofunctionalization represents a forward-looking, scalable innovation that addresses both clinical and societal needs.
Conflict of interest statement
The authors declare that they have no conflicts of interest related to this work.
References
- Kaneko S, Yamamoto Y, Wada K, Kumagai G, Harada Y, et al. Ultraviolet irradiation improves the hydrophilicity and osteo-conduction of hydroxyapatite. J Orthop Surg Res 2020;15(1):425. [PMID: 32948233 DOI: 10.1186/s13018-020-01949-3]
- Ogawa T. Ultraviolet photofunctionalization of titanium implants. Int J Oral Maxillofac Implants 2014;29(1):e95-102. [PMID: 24451893 DOI: 10.11607/jomi.te47]
- Kido D, Komatsu K, Suzumura T, Matsuura T, Cheng J, et al. Influence of surface contaminants and hydrocarbon pellicle on the results of wettability measurements of titanium. Int J Mol Sci 2023;24(19):14688. [PMID: 37834133 DOI: 10.3390/ijms241914688]
- Att W, Hori N, Takeuchi M, Ouyang J, Yang Y, et al. Time-dependent degradation of titanium osteoconductivity: an implication of biological aging of implant materials. Biomaterials 2009;30(29):5352-63. [PMID: 19595450 DOI: 10.1016/j.biomaterials.2009.06.040]
- Hori N, Att W, Ueno T, Sato N, Yamada M, et al. Age-dependent degradation of the protein adsorption capacity of titanium. J Dent Res 2009;88(7):663-7. [PMID: 19641155 DOI: 10.1177/0022034509339567]
- Hirota M, Ikeda T, Sugita Y, Ishijima M, Hirota S, et al. Impaired osteoblastic behavior and function on saliva-contaminated titanium and its restoration by UV treatment. Mater Sci Eng C Mater Biol Appl 2019;100:165-77. [PMID: 30948050 DOI: 10.1016/j.msec.2019.03.008]
- Cheng K, Hong Y, Yu M, Lin J, Weng W, et al. Modulation of protein behavior through light responses of TiO2 nanodots films. Sci Rep 2015;5:13354. [PMID: 26306638 DOI: 10.1038/srep13354]
- Raines AL, Berger MB, Schwartz Z, Boyan BD. Osteoblasts grown on microroughened titanium surfaces regulate angiogenic growth factor production through specific integrin receptors. Acta Biomater 2019;97:578-86. [PMID: 31349056 DOI: 10.1016/j.actbio.2019.07.036]
- Kim HS, Lee JI, Yang SS, Kim BS, Kim BC, et al. The effect of alendronate soaking and ultraviolet treatment on bone-implant interface. Clin Oral Implants Res 2017;28(9):1164-72. [PMID: 27458172 DOI: 10.1111/clr.12933]
- Lee JB, Jo YH, Choi JY, Seol YJ, Lee YM, et al. The effect of ultraviolet photofunctionalization on a titanium dental implant with machined surface: an in vitro and in vivo study. Materials (Basel). 2019;12(13):2078. [PMID: 31261627 DOI: 10.3390/ma12132078]
- Suzumura T, Matsuura T, Komatsu K, Ogawa T. A novel high-energy vacuum ultraviolet light photofunctionalization approach for decomposing organic molecules around titanium. Int J Mol Sci 2023;24(3):1978. [PMID: 36768297 DOI: 10.3390/ijms24031978]
- Komatsu K, Matsuura T, Suzumura T, Shibata R, Chen PC, et al. Vacuum ultraviolet (VUV)-induced physicochemical engineering of titanium: enhanced fibroblast activity, redox system, and glycosaminoglycan binding for soft tissue integration. ACS Appl Bio Mater 2025;8(5):4166-85. [PMID: 40249645 DOI: 10.1021/acsabm.5c00283]
- Hirota M, Ozawa T, Iwai T, Mitsudo K, Ogawa T. UV-mediated photofunctionalization of dental implant: a seven-year results of a prospective study. J Clin Med 2020;9(9):2733. [PMID: 32847061 DOI: 10.3390/jcm9092733]
- Yin C, Zhang T, Wei Q, Cai H, Cheng Y, et al. Surface treatment of 3D printed porous Ti6Al4V implants by ultraviolet photofunctionalization for improved osseointegration. Bioact Mater 2022;7:26-38. [PMID: 34466715 DOI: 10.1016/j.bioactmat.2021.05.043]
- Roy M, Kubacki J, Psiuk B, Mrozek-Wilczkiewicz A, Malarz K, et al. Photofunctionalization effect and biological ageing of PEEK, TiO2 and ZrO2 abutments material. Mater Sci Eng C Mater Biol Appl 2021;121:111823. [PMID: 33579466 DOI: 10.1016/j.msec.2020.111823]
- Choi SH, Shin J, Cha JK, Kwon JS, Cha JY, et al. Evaluation of success rate and biomechanical stability of ultraviolet-photofunctionalized miniscrews with short lengths. Am J Orthod Dentofacial Orthop 2021;159(2):158-66. [PMID: 33243706 DOI: 10.1016/j.ajodo.2019.12.018]
- Hirota M, Tanaka M, Ishijima M, Iwasaki C, Park W, Ogawa T. Effect of Photofunctionalization on Ti6Al4V Screw Stability Placed in Segmental Bone Defects in Rat Femurs. J Oral Maxillofac Surg 2016;74(4):861.e1-16. [PMID: 26704430 DOI: 10.1016/j.joms.2015.11.016]
- Park W, Ishijima M, Hirota M, Soltanzadeh P, Ogawa T. Engineering bone-implant integration with photofunctionalized titanium microfibers. J Biomater Appl 2016;30(8):1242-1250. [PMID: 26656313 DOI: 10.1016/10.1177/0885328215620034]
- Kemuriyama S, Aita H, Maida T, Kawamura N, Nezu T, et al. Effect of photofunctionalization on titanium bone-implant integration in ovariectomized rats. Dent Mater J 2023;42(1):11-8. [PMID: 36123044 DOI: 10.4012/dmj.2022-081]
- Sugita Y, Honda Y, Kato I, Kubo K, Maeda H, et al. Role of photofunctionalization in mitigating impaired osseointegration associated with type 2 diabetes in rats. Int J Oral Maxillofac Implants 2014;29(6):1293-300. [PMID: 25397793 DOI: 10.11607/jomi.3480]
- Hirota M, Ikeda T, Tabuchi M, Ozawa T, Tohnai I, et al. Effects of ultraviolet photofunctionalization on bone augmentation and integration capabilities of titanium mesh and implants. Int J Oral Maxillofac Implants 2017;32(1):52-62. [PMID: 28095515 DOI: 10.11607/jomi.4891]
- Hirota M, Ozawa T, Iwai T, Ogawa T, Tohnai I. Implant stability development of photofunctionalized implants placed in regular and complex cases: a case-control study. Int J Oral Maxillofac Implants 2016;31(3):676-86. [PMID: 27183088 DOI: 10.11607/jomi.4115]
- Kheur S, Kheur M, Madiwal V, Sandhu R, Lakha T. et al. In-Vitro evaluation of photofunctionalized implant surfaces in a high-glucose microenvironment simulating diabetics. J Funct Biomater 2023;14(3):130. [PMID: 36976054 DOI: 10.3390/jfb14030130]
- Aita H, Hori N, Takeuchi M, Suzuki T, Yamada M, et al. The effect of ultraviolet functionalization of titanium on integration with bone. Biomaterials 2009;30(6):1015-25. [PMID: 19042016 DOI: 10.1016/j.biomaterials.2008.11.004]
- Hirota M, Ikeda T, Tabuchi M, Iwai T, Tohnai I, et al. Effect of ultraviolet-mediated photofunctionalization for bone formation around medical titanium mesh. J Oral Maxillofac Surg 2014;72(9):1691-702. [PMID: 25109583 DOI: 10.1016/j.joms.2014.05.012]
- Wang J, Jing Z, Yin C, Wang Z, Zeng S, et al. Coatless modification of 3D-printed Ti6Al4V implants through tailored Cu ion implantation combined with UV photofunctionalization to enhance cell attachment, osteogenesis and angiogenesis. Colloids Surf B Biointerfaces 2024;238:113891. [PMID: 38615392 DOI: 10.1016/j.colsurfb.2024.113891]
- Tominaga H, Matsuyama K, Morimoto Y, Yamamoto T, Komiya S, et al. The effect of ultraviolet photofunctionalization of titanium instrumentation in lumbar fusion: a non-randomized controlled trial. BMC Musculoskelet Disord 2019;20(1):292. [PMID: 31208382 DOI: 10.1186/s12891-019-2672-3]
- Choi B, Lee YC, Oh KC, Lee JH. Effects of photofunctionalization on early osseointegration of titanium dental implants in the maxillary posterior region: a randomized double-blinded clinical trial. Int J Implant Dent 2021;7(1):37. [PMID: 33969450 DOI: 10.1186/s40729-021-00318-x]