Armored and Cold Tumors: A Refractory Subset of Solid Cancer
1The Affiliated Cancer Hospital of Nanjing Medical University, Jiangsu Cancer Hospital, Jiangsu Institute of Cancer Research, Nanjing, Jiangsu, China
2The Fourth Clinical Medicine College, Nanjing Medical University, Nanjing, China
3The First Clinical Medicine College, Nanjing Medical University, Nanjing, China
4Department of Central Laboratory, Jintan Affiliated Hospital of Jiangsu University, Changzhou, China
5Department of Central Laboratory, Jintan Hospital, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Changzhou, China
6Department of Anatomical Pathology, Singapore General Hospital, Singapore
7Department of Physiology, School of Basic Medical Sciences, Nanjing Medical University, Nanjing, Jiangsu, China
8Department of General Surgery, The Affiliated Taizhou People’s Hospital of Nanjing Medical University, Taizhou, Jiangsu, China
9Department of Gynecology, Wuxi Maternity and Child Health Care Hospital, Affiliated Women’s Hospital of Jiangnan University, Wuxi, Jiangsu, China
10Department of Gynecology, Wuxi Maternal and Child Health Care Hospital, Wuxi Medical Center, Nanjing Medical University, Wuxi, Jiangsu, China
aRui Xu and Kai Yang contributed equally to this work.
*Correspondence to: Yichao Zhu, E-mail: zhuyichao@njmu.edu.cn; Yan Zhang, E-mail: fuyou2007@126.com; Jie Mei, E-mail: meijie199621@163.com
Received: July 9 2025; Revised: August 5 2025; Accepted: August 13 2025; Published Online: September 8 2025.
Cite this paper:
Xu R, Yang K, Cai Y et al. Armored and Cold Tumors: A Refractory Subset of Solid Cancer. BIO Integration 2025; 6: 1–6.
DOI: 10.15212/bioi-2025-0127. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Solid tumors are characterized by extensive extracellular matrix (ECM) remodeling prominently featuring massive collagen deposition. This dense collagen network does not act as inert scaffolding but actively orchestrates critical aspects of tumor progression and therapy resistance, thereby shaping the fate of cancer cells. Collagen influences cellular behavior through multiple mechanisms, including providing structural rigidity, modulating mechanotransduction signaling pathways, creating physical barriers to immune cell infiltration and drug penetration, and serving as a reservoir for signaling molecules. Here, we discuss recent findings regarding the critical roles of collagen in tumors and potential therapies for armored and cold tumors, a refractory subset demonstrating high collagen deposition and low immune infiltration.
Keywords
Armored and cold tumors, collagen, immune exclusion, tumor microenvironment.
Intratumoral collagen supports tumor progression
Collagen, the structural component in the extracellular matrix (ECM), provides structural integrity and support to various cell types in tumor tissues. Collagen also modulates cellular activities through signal transduction via interaction with cell surface receptors in specific cell types, including integrins, the discoidin domain receptors, osteoclast-associated receptor, glycoprotein VI, G6b-B, leukocyte-associated immunoglobulin-like receptors, and the mannose family receptor [1]. Intratumoral collagen, characterized by high density and stiffness, has diverse roles in malignant progression in most solid cancer types [1, 2], such as promoting tumor proliferation, metastasis, and resistance to therapeutic agents while hindering tumor progression.
Intratumoral collagen, primarily type I collagen, serves as a dual physical and biochemical barrier against anti-tumor immunity. It forms as a result of synergistic multifactorial effects. First, hyperactivation of tumor cells and recruited cancer-associated fibroblasts (CAFs) is the core mechanism driving aberrant collagen synthesis and deposition, wherein activated CAFs secrete excessive collagen and other ECM components under stimuli such as transforming growth factor-β (TGF-β) [3]. Second, imbalanced ECM remodeling exacerbates collagen accumulation. In this process, dysregulation of matrix metalloproteinases and their inhibitors impairs collagen degradation, thus promoting excessive cross-linking and thickening of deposited collagen fibers, and the formation of a dense, rigid fibrotic network [4]. Finally, this collagen-enriched microenvironment establishes immunosuppression through multiple mechanisms, including physical blockade and biochemical inhibition, primarily by (i) physically impeding contact between immune cells and tumor cells [5], (ii) restricting the cytotoxic functions of immune cells via mechanical stress [6], and (iii) releasing bioactive fragments that modulate immune cell activity [7]. Our previous research has systematically summarized the critical role of targeting collagen in sensitizing immunotherapy [8].
Definition of armored and cold tumors
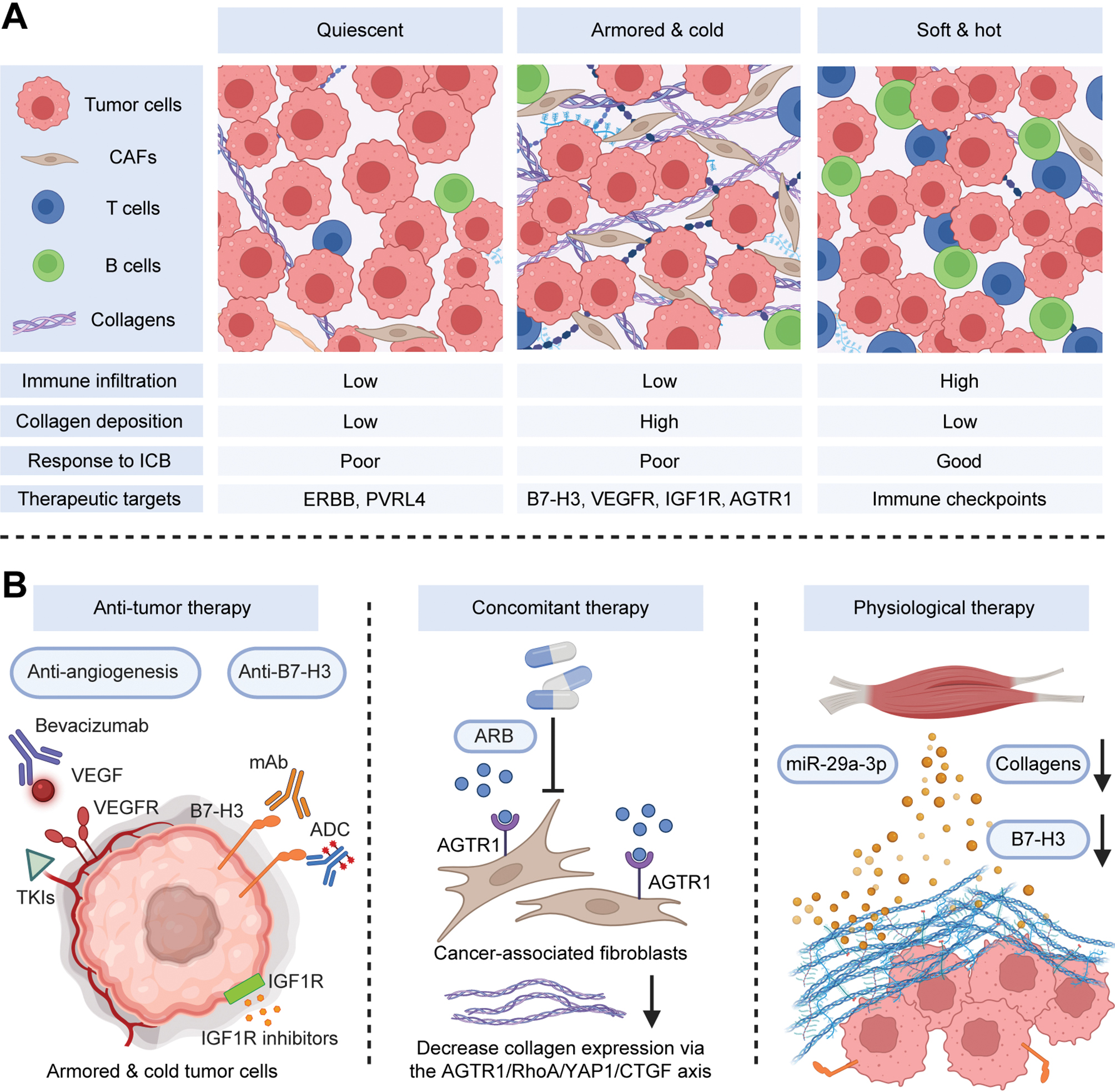
Despite the close association between the ECM and immune cells in the tumor microenvironment (TME), the contribution of the interaction between ECM and immune cells to patient stratification and immune checkpoint blockade (ICB) response prediction remains poorly understood. In our previous research [9], analysis of transcriptomic collagen activity and immune signatures in large-scale public cohorts led to the identification of three distinct immuno-collagenic subtypes predictive of ICB responses. Validation was performed using paraffin embedded cancer tissue microarrays obtained from the National Engineering Center for Biochip (Outdo Biotech, Shanghai), comprising 1,012 cases across 10 solid cancer types. The tumors were categorized into three subtypes: “soft and hot” (low collagen deposition and high immune infiltration), “armored and cold” (high collagen deposition and low immune infiltration), and “quiescent” (low collagen deposition and immune infiltration) (Figure 1A) [9]. In addition, Hamidi et al. have established a novel stratification incorporating ECM, immune infiltration, and tumor cell heterogeneity to predict clinical benefits from programmed death ligand 1 (PD-L1) blockade in urothelial carcinoma [10]. That study provided both an independent validation of, and an important complement to, our own research, thereby underscoring the critical importance of elucidating tumor cell heterogeneity in quiescent tumors [10]. In summary, although armored and cold tumors and quiescent tumors are “cold” tumors, as previously defined [11], armored and cold tumors demonstrate unique collagen-rich microenvironments that enable specific therapeutic strategies [12].
Figure 1 Schematic description of the molecular features of armored and cold tumors and corresponding treatments. (A) A framework was developed to stratify tumors according to collagen deposition and immune activity. Armored and cold tumors are accompanied by low immune infiltration and high collagen deposition, and show poor prognosis and resistance to ICB therapy. These tumors highly express B7-H3, angiogenesis markers, IGF1R, and AGTR1 (the target of ARBs). (B) Possible therapeutic strategies from three perspectives (anti-tumor therapy, concomitant therapy, and physiological therapy) are shown. Created with BioRender.com. Abbreviations: CAF, cancer associated fibroblast; TME, tumor microenvironment; ICB, immune checkpoint blockade; VEGF/VEGFA, vascular endothelial growth factor/A; VEGFR, VEGF receptor; AGTR1, angiotensin II receptor type 1; ARB, angiotensin receptor blocker; IGF1R, insulin like growth factor 1 receptor; mAb, monoclonal antibody; ADC, antibody–drug conjugate; TKIs, tyrosine kinase inhibitors; CAR T, chimeric antigen receptor T cell; NK, natural killer; RhoA, Ras homolog family member A; YAP1, Yes associated protein 1; CTGF, connective tissue growth factor.
Candidate therapies for armored and cold tumors
Anti-tumor therapy
To determine potential anti-tumor targets for armored and cold tumors, we conducted a pan-cancer analysis to assess the expression of various drug targets. Targets such as fibroblast growth factor receptor, platelet-derived growth factor receptor, immune inhibition-associated targets [such as B7 homolog 3 protein (B7-H3)], and angiogenesis-related targets [such as vascular endothelial growth factor A (VEGFA) and VEGF receptor (VEGFR)] exhibited high expression in armored and cold tumors [9]. B7-H3 has been associated with immuno-cold features and collagen accumulation in triple-negative breast cancer [13] and melanoma [14]. Interestingly, anti-B7-H3 therapy has been validated to be a feasible and robust candidate immunotherapy against advanced prostate cancer [15]. Therefore, anti-B7-H3 therapy might provide a valuable supplemental therapeutic strategy for armored and cold tumors. B7-H3-related clinical trials in solid cancer are summarized in Table 1. We believe that subgroup analyses based on the three distinct immuno-collagenic subtypes in these clinical trials are warranted to identify whether patients with armored and cold tumors might achieve enhanced benefits.
Table 1 B7-H3-Related Clinical Trials in Solid Cancers
| NCT Number | Cancer types | Interventions |
|---|---|---|
| NCT01918930 | Melanoma | MGA271 |
| NCT02381314 | Melanoma and non-small cell lung cancer | Enoblituzumab |
| NCT02628535 | Advanced solid tumors | MGD009 |
| NCT02982941 | Neuroblastoma, rhabdomyosarcoma, osteosarcoma, and others | Enoblituzumab |
| NCT03198052 | Lung cancer | CAR-T cell |
| NCT03406949 | Advanced solid tumors | MGD009 |
| NCT03729596 | Solid tumors | MGC018 |
| NCT04022213 | Desmoplastic small round cell tumors, and peritoneal cancer | Omburtamab I131 |
| NCT04077866 | Brain and nervous system tumors | CAR-T cell |
| NCT04129320 | Head and neck cancer | Enoblituzumab |
| NCT04145622 | Advanced solid tumors | DS7300 |
| NCT04185038 | Brain and nervous system tumors | CAR-T cell |
| NCT04385173 | Brain and nervous system tumors | CAR-T cell |
| NCT04432649 | Solid tumors | CAR-T cell |
| NCT04433221 | Sarcoma, osteoid sarcoma, and Ewing sarcoma | CAR-T cell |
| NCT04483778 | Solid tumors | CAR-T cell |
| NCT04544592 | B-cell acute lymphoblastic leukemia/non-Hodgkin lymphoma | CAR-T cell |
| NCT04630769 | Ovarian cancer | Enoblituzumab |
| NCT04634825 | Head and neck cancer | Enoblituzumab |
| NCT04637503 | Neuroblastoma | CAR-T cell |
| NCT04670068 | Epithelial ovarian cancer | CAR-T cell |
| NCT04691713 | Solid tumors | CAR-T cell |
| NCT04692948 | Acute myeloid leukemia | CAR-T cell |
| NCT04743661 | Brain and nervous system tumors | Omburtamab I131 |
| NCT04842812 | Solid tumors | CAR-T cell |
| NCT04864821 | Osteosarcoma, neuroblastoma, gastric cancer, and lung cancer | CAR-T cell |
| NCT04897321 | Solid tumors | CAR-T cell |
| NCT05063357 | Brain and nervous system tumors | Omburtamab I131 |
| NCT05064306 | Brain and nervous system tumors | Omburtamab I131 |
| NCT05143151 | Advanced pancreatic cancer | CAR-T cell |
| NCT05190185 | Malignant melanoma, lung cancer, and colorectal cancer | CAR-T cell |
| NCT05211557 | Ovarian cancer | CAR-T cell |
| NCT05241392 | Brain and nervous system tumors | CAR-T cell |
| NCT05276609 | Advanced solid tumors | HS-20093 |
| NCT05280470 | Extensive-stage small-cell lung cancer | DS7300 |
| NCT05293496 | Advanced solid tumors | MGC018 |
| NCT05323201 | Hepatocellular carcinoma | CAR-T cell |
| NCT05341492 | EGFR/B7H3-positive advanced lung cancer and breast cancer | CAR-T cell |
| NCT05366179 | Brain and nervous system tumors | CAR-T cell |
| NCT05405621 | Advanced solid tumors | BAT8009 |
| NCT05474378 | Brain and nervous system tumors | CAR-T cell |
| NCT05515185 | Advanced solid tumors | CAR-T cell |
| NCT05562024 | B7-H3-positive relapsed/refractory neuroblastoma | CAR-T cell |
| NCT05722171 | Relapsed/refractory acute myeloid leukemia | CAR-T cell |
| NCT05731219 | Relapsed/refractory acute myeloid leukemia | CAR-T cell |
| NCT05752877 | Brain and nervous system tumors | CAR-T cell |
| NCT05768880 | Brain and nervous system tumors | CAR-T cell |
| NCT05835687 | Brain and nervous system tumors | CAR-T cell |
| NCT05914116 | Advanced solid tumors | DB-1311 |
| NCT05991583 | Advanced malignant tumors | IBB0979 |
| NCT06018363 | Brain and nervous system tumors | CAR-T cell |
| NCT06052423 | Extensive-stage small-cell lung cancer | HS-20093 |
| NCT06112704 | Advanced solid tumors | HS-20093 |
| NCT06158139 | Pancreas cancer and relapse/resistant cancer | CAR-T cell |
| NCT06203210 | Small cell lung cancer | DS7300 |
| NCT06221553 | Brain and nervous system tumors | CAR-T cell |
| NCT06305299 | Ovarian cancer | CAR-T cell |
| NCT06347068 | Triple-negative breast cancer | CAR-T cell |
| NCT06362252 | Extensive-stage small-cell lung cancer | DS7300 |
| NCT06372236 | B7-H3-positive relapsed/advanced malignant solid tumors | CAR-T cell |
| NCT06422520 | Advanced solid tumors | BGB-C354 |
| NCT06482905 | Brain and nervous system tumors | CAR-T cell |
Furthermore, collagen deposition markedly influences tumor angiogenesis. Our prior systematic analysis across multiple cancer types has revealed a robust association between immuno-collagenic subtypes and angiogenic activity, and demonstrated that armored and cold tumors exhibit the highest angiogenic levels. Critically, collagen inhibition through diverse approaches effectively suppresses tumor angiogenesis in vivo [16]. In addition, armored and cold tumors have been found to display superior responsiveness to anti-angiogenic therapy in advanced lung adenocarcinoma cohorts [16]. Therefore, anti-angiogenic therapy is another promising strategy for targeting armored and cold tumors. Furthermore, insulin like growth factor 1 receptor (IGF1R) has emerged as an additional therapeutic target for armored and cold tumors. The IGF1R inhibitor picropodophyllin has been observed to enhance immunotherapy efficacy in preclinical models [17].
Concomitant therapy
Patients with cancer undergoing anti-tumor therapy are frequently prescribed multiple medications for pre-existing comorbidities or adverse effects from the anti-tumor therapy, and polypharmacy is common [18]. Considering the high prevalence of concomitant medications, we screened potential concomitant medications according to the established immuno-collagenic subtypes, and found that angiotensin II receptor 1 (AGTR1), the target of angiotensin receptor blockers (ARBs), has high expression in armored and cold tumors. ARBs inhibit type I collagen expression in CAFs via negatively regulating the RhoA/YAP axis, thus shaping an inflamed TME [19]. Therefore, the combination of ARBs with existing anti-tumor therapies might be effective for armored and cold tumors. Notably, a phase 2 clinical trial has reported that a combination of losartan followed by chemoradiotherapy has led to downstaging of locally advanced pancreatic ductal adenocarcinoma, a highly fibrotic solid tumor, and is associated with an R0 resection rate of 61% [20]. Therefore, more clinical trials of ARB combinations in settings not limited to pancreatic cancer, or to chemotherapy and radiotherapy, are urgently needed in armored and cold tumors.
Physiological therapy
Physiological therapies are aimed at enhancing and restoring the normal physiological functions of the human body. This spectrum of treatment includes primarily rehabilitation training and biofeedback therapy, among other therapies. Physical activity is widely understood to be healthful among both the scientific community and the public. Exercise has been found to increase T and natural killer (NK) cell infiltration, thereby controlling tumor growth [21]. However, how exercise promotes anti-tumor immunity and immunotherapy remains elusive. On the basis of the established immuno-collagenic subtypes, we have reported that exercise promotes muscle-derived extracellular vesicle-associated miR-29a-3p in ECM inhibition by directly targeting COL1A1, thereby enabling immune cell infiltration and immunotherapy. Clinically, miR-29a-3p correlates with decreased ECM and increased T cell infiltration in various cancer types [22]. In addition, B7-H3, a therapeutic target highly expressed in armored and cold tumors, is targeted by exercise-stimulated miR-29a-3p [23]. Although exercise exerts broad antitumor effects, based on the molecular features of the immuno collagenic subtypes and exercise induced molecular profiles, patients with armored and cold tumors are expected to achieve greater relative benefits than those with soft and hot or quiescent tumors—specifically in terms of extracellular matrix reduction, increased intratumoral T/NK cell infiltration, and potentiation of immunotherapy responses.
Shortcomings and future research directions
The studies described herein first defined the concept of armored and cold tumors in the scientific community and suggested possible therapeutic strategies from three perspectives: anti-tumor therapy, concomitant therapy, and physiological therapy (Figure 1B). However, further in-depth research should be conducted. First, in addition to concomitant medications, pre-existing comorbidities in patients with cancer warrant investigation. Attention should be paid to common pre-existing comorbidities, including type 2 diabetes, hypertension, abnormal lipid metabolism, and other diseases. Moreover, potential effects on the armored and cold transformation of the TME and the underlying molecular mechanisms should be investigated. Given the plasticity of the TME, non-invasive diagnostic technologies, such as deep learning with B-mode ultrasound, computed tomography (CT), magnetic resonance imaging (MRI), and visualization of the collagen in the TME, are urgently needed. Another promising research direction is using physical stimulation, such as ionizing radiation, light, electricity, magnetic fields, or ultrasound, to potentially decrease collagen production or induce collagen denaturation, thereby boosting anti-tumor immune responses [24]. Clinical trials guided by the characteristics of armored and cold tumors stand to benefit both cancer patients and oncologists. In addition, the development of novel biomaterials for collagen degradation is an important therapeutic method for further investigation.
Concluding remarks
Cancer is a complex systemic disease involving constant interactions among cancer cells, the ECM, and other cell types present in the TME. Collagen in the ECM plays an essential role during cancer progression, and consequently is a promising therapeutic target for cancer management. Although collagen has traditionally been considered a structural scaffold, it elicits a myriad of biophysical, biochemical, and cell biological alterations that extensively affect tumor metabolism, growth, and immunity. Leveraging the biological association between collagen deposition and immune activity in the TME has enabled the identification of armored and cold tumors, a refractory subset of solid cancer, as well as the proposal of potential therapeutic strategies. With advances and interdisciplinary integration in cell biology, oncology, material science, and nanotechnology, current translational priorities include (i) developing collagen-based imaging biomarkers to non-invasively stratify tumors and (ii) designing combination regimens that disrupt collagen barriers while enhancing immunotherapy penetration in armored and cold tumors. Such strategies have promise in accelerating clinical translation and improving patient quality of life.
Acknowledgments
We thank Dr. Yunlong Yang (Fudan University) for critical comments on this article.
Conflict of interest
Jie Mei is an Editorial Board Member of BIO Integration. He was not involved in the peer-review or handling of the manuscript. The other authors have no other competing interests to disclose.
References
- Su H, Karin M. Collagen architecture and signaling orchestrate cancer development. Trends Cancer 2023;9(9):764-73. [PMID: 37400314 DOI: 10.1016/j.trecan.2023.06.002]
- Sleeboom JJF, van Tienderen GS, Schenke-Layland K, van der Laan LJW, Khalil AA, et al. The extracellular matrix as hallmark of cancer and metastasis: from biomechanics to therapeutic targets. Sci Transl Med 2024;16(728):eadg3840. [PMID: 38170791 DOI: 10.1126/scitranslmed.adg3840]
- Caligiuri G, Tuveson DA. Activated fibroblasts in cancer: perspectives and challenges. Cancer Cell 2023;41(3):434-49. [PMID: 36917949 DOI: 10.1016/j.ccell.2023.02.015]
- Chitty JL, Cox TR. The extracellular matrix in cancer: from understanding to targeting. Trends Cancer 2025;S2405-8033(25)00126-8. [PMID: 40467384 DOI: 10.1016/j.trecan.2025.05.003]
- Salmon H, Franciszkiewicz K, Damotte D, Dieu-Nosjean M-C, Validire P, et al. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J Clin Invest 2012;122(3):899-910. [PMID: 22293174 DOI: 10.1172/JCI45817]
- O’Connor RS, Hao X, Shen K, Bashour K, Akimova T, et al. Substrate rigidity regulates human T cell activation and proliferation. J Immunol 2012;189(3):1330-9. [PMID: 22732590 DOI: 10.4049/jimmunol.1102757]
- Kessenbrock K, Plaks V, Werb Z. Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 2010;141(1):52-67. [PMID: 20371345 DOI: 10.1016/j.cell.2010.03.015]
- Wang Y, Zhang F, Qian Z, Jiang Y, Wu D, et al. Targeting collagen to optimize cancer immunotherapy. Exp Hematol Oncol 2025;14(1):101. [PMID: 40721833 DOI: 10.1186/s40164-025-00691-y]
- Mei J, Cai Y, Xu R, Li Q, Chu J, et al. Conserved immuno-collagenic subtypes predict response to immune checkpoint blockade. Cancer Commun (Lond) 2024;44(5):554-75. [PMID: 38507505 DOI: 10.1002/cac2.12538]
- Hamidi H, Senbabaoglu Y, Beig N, Roels J, Manuel C, et al. Molecular heterogeneity in urothelial carcinoma and determinants of clinical benefit to PD-L1 blockade. Cancer Cell 2024;42(12):2098-112.e4. [PMID: 39577421 DOI: 10.1016/j.ccell.2024.10.016]
- Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med 2018;24(5):541-50. [PMID: 29686425 DOI: 10.1038/s41591-018-0014-x]
- Baldari S, Di Modugno F, Nisticò P, Toietta G. Strategies for efficient targeting of tumor collagen for cancer therapy. Cancers (Basel) 2022;14(19):4706. [PMID: 36230627 DOI: 10.3390/cancers14194706]
- Mei J, Cai Y, Zhu H, Jiang Y, Fu Z, et al. High B7-H3 expression with low PD-L1 expression identifies armored-cold tumors in triple-negative breast cancer. NPJ Breast Cancer 2024;10(1):11. [PMID: 38280882 DOI: 10.1038/s41523-024-00618-6]
- Shen B, Mei J, Xu R, Cai Y, Wan M, et al. B7-H3 is associated with the armored-cold phenotype and predicts poor immune checkpoint blockade response in melanoma. Pathol Res Pract 2024;256:155267. [PMID: 38520953 DOI: 10.1016/j.prp.2024.155267]
- Shenderov E, De Marzo AM, Lotan TL, Wang H, Chan S, et al. Neoadjuvant enoblituzumab in localized prostate cancer: a single-arm, phase 2 trial. Nat Med 2023;29(4):888-97. [PMID: 37012549 DOI: 10.1038/s41591-023-02284-w]
- Mei J, Yang K, Zhang X, Luo Z, Tian M, et al. Intratumoral collagen deposition supports angiogenesis suggesting anti-angiogenic therapy in armored and cold tumors. Adv Sci (Weinh) 2025;12(10):e2409147. [PMID: 39823457 DOI: 10.1002/advs.202409147]
- Wan M, Mei J, Cai Y, Zhou J, Xue N, et al. Targeting IGF1R overcomes armored and cold tumor microenvironment and boosts immune checkpoint blockade in triple-negative breast cancer. Adv Sci (Weinh) 2025;e01341. [PMID: 40679093 DOI: 10.1002/advs.202501341]
- van Leeuwen RWF, Jansman FGA, van den Bemt PMLA, de Man F, Piran F, et al. Drug-drug interactions in patients treated for cancer: a prospective study on clinical interventions. Ann Oncol 2015;26(5):992-7. [PMID: 25628444 DOI: 10.1093/annonc/mdv029]
- Mei J, Chu J, Yang K, Luo Z, Yang J, et al. Angiotensin receptor blocker attacks armored and cold tumors and boosts immune checkpoint blockade. J Immunother Cancer 2024;12(9):e009327. [PMID: 39244215 DOI: 10.1136/jitc-2024-009327]
- Murphy JE, Wo JY, Ryan DP, Clark JW, Jiang W, et al. Total neoadjuvant therapy with FOLFIRINOX in combination with losartan followed by chemoradiotherapy for locally advanced pancreatic cancer: a phase 2 clinical trial. JAMA Oncol 2019;5(7):1020-7. [PMID: 31145418 DOI: 10.1001/jamaoncol.2019.0892]
- Pedersen L, Idorn M, Olofsson GH, Lauenborg B, Nookaew I, et al. Voluntary running suppresses tumor growth through epinephrine- and IL-6-dependent NK cell mobilization and redistribution. Cell Metab 2016;23(3):554-62. [PMID: 26895752 DOI: 10.1016/j.cmet.2016.01.011]
- Luo Z, Mei J, Wang X, Wang R, He Z, et al. Voluntary exercise sensitizes cancer immunotherapy via the collagen inhibition-orchestrated inflammatory tumor immune microenvironment. Cell Rep 2024;43(9):114697. [PMID: 39217611 DOI: 10.1016/j.celrep.2024.114697]
- Mei J, Luo Z, Cai Y, Wan R, Qian Z, et al. Altered atlas of exercise-responsive microRNAs revealing miR-29a-3p attacks armored and cold tumors and boosts anti-B7-H3 therapy. Research (Wash DC) 2025;8:0590. [PMID: 39845707 DOI: 10.34133/research.0590]
- Guo Z, Saw PE, Jon S. Non-invasive physical stimulation to modulate the tumor microenvironment: unveiling a new frontier in cancer therapy. BIOI 2024;5(1). [DOI: 10.15212/bioi-2024-0012]