Mechanoregulation of Cell Fate by Low-intensity Pulsed Ultrasound: Mechanisms and Advances in Regenerative Medicine
1Key Laboratory of Medical Imaging Precision Theranostics and Radiation Protection, College of Hunan Province, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
2Department of Medical Imaging, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
3Institute of Medical Imaging, Hengyang Medical School, University of South China, Hengyang, China
4Gulbali Institute, Charles Sturt University, Wagga Wagga, NSW 2678, Australia
5Australian Institute for Bioengineering and Nanotechnology, The University of Queensland, Brisbane, Queensland 4072, Australia
aThese authors contributed equally to this work and share the first authorship.
*Correspondence to: Meng Du, E-mail: dumeng_work@126.com; Li Li, E-mail: lili@csu.edu.au
Received: March 7 2025; Revised: May 1 2025; Accepted: May 15 2025; Published Online: July 30 2025
Cite this paper:
Lei L, Zhang Q, Du M et al. Mechanoregulation of Cell Fate by Low-intensity Pulsed Ultrasound: Mechanisms and Advances in Regenerative Medicine. BIO Integration 2025; 6: 1–19.
DOI: 10.15212/bioi-2025-0049. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
As a mechanical wave capable of transmitting thermal and mechanical energy, ultrasound has emerged as a pivotal tool in regenerative medicine due to its non-invasive nature. Low-intensity pulsed ultrasound (LIPUS), a mechanoregulatory technique independent of thermal effects, delivers controlled mechanical stimuli to activate endogenous mechanotransduction pathways, such as ion channels, transmembrane proteins, and cytoskeleton-mediated signaling cascades. These pathways regulate critical cellular processes, such as proliferation, differentiation, and apoptosis, positioning LIPUS as a promising modality for targeted modulation of cell fate. Preclinical and clinical studies have demonstrated the therapeutic efficacy of LIPUS across diverse applications, including bone repair, neural regeneration, and soft tissue rehabilitation. However, optimizing stimulation parameters and advancing clinical translation remain key challenges. This review summarizes the central role of LIPUS in promoting tissue regeneration through non-thermal regulation of cellular homeostasis and explores strategies to accelerate clinical adoption of LIPUS. By integrating mechanistic insights with translational perspectives, this review provides a roadmap for advancing LIPUS-driven regenerative medicine in the era of precision bioengineering.
Keywords
Biological effects, dental treatment, fracture healing, low-intensity pulsed ultrasound, neuromodulation, regenerative medicine, urologic condition.
Introduction
Ultrasound, defined as mechanical vibrations exceeding 20 kHz without ionizing radiation, serves dual diagnostic and therapeutic roles in medicine. Originally developed for imaging, the biological effects of ultrasound, which were first observed in 1927, have propelled the evolution of ultrasound into a versatile therapeutic tool [1, 2]. Therapeutic ultrasound modalities are categorized into thermal and non-thermal types. High-intensity focused ultrasound (HIFU), an early thermal application, induces coagulative necrosis and has gained clinical approval for tumor ablation and neurosurgical interventions [3]. In contrast, low-intensity ultrasound (LIU) predominantly exploits non-thermal effects, exemplified by an ability to trigger significant biological responses in neuromodulation with minimal temperature elevation (< 0.1°C), underscoring the pivotal role of mechanical forces in cellular regulation [4].
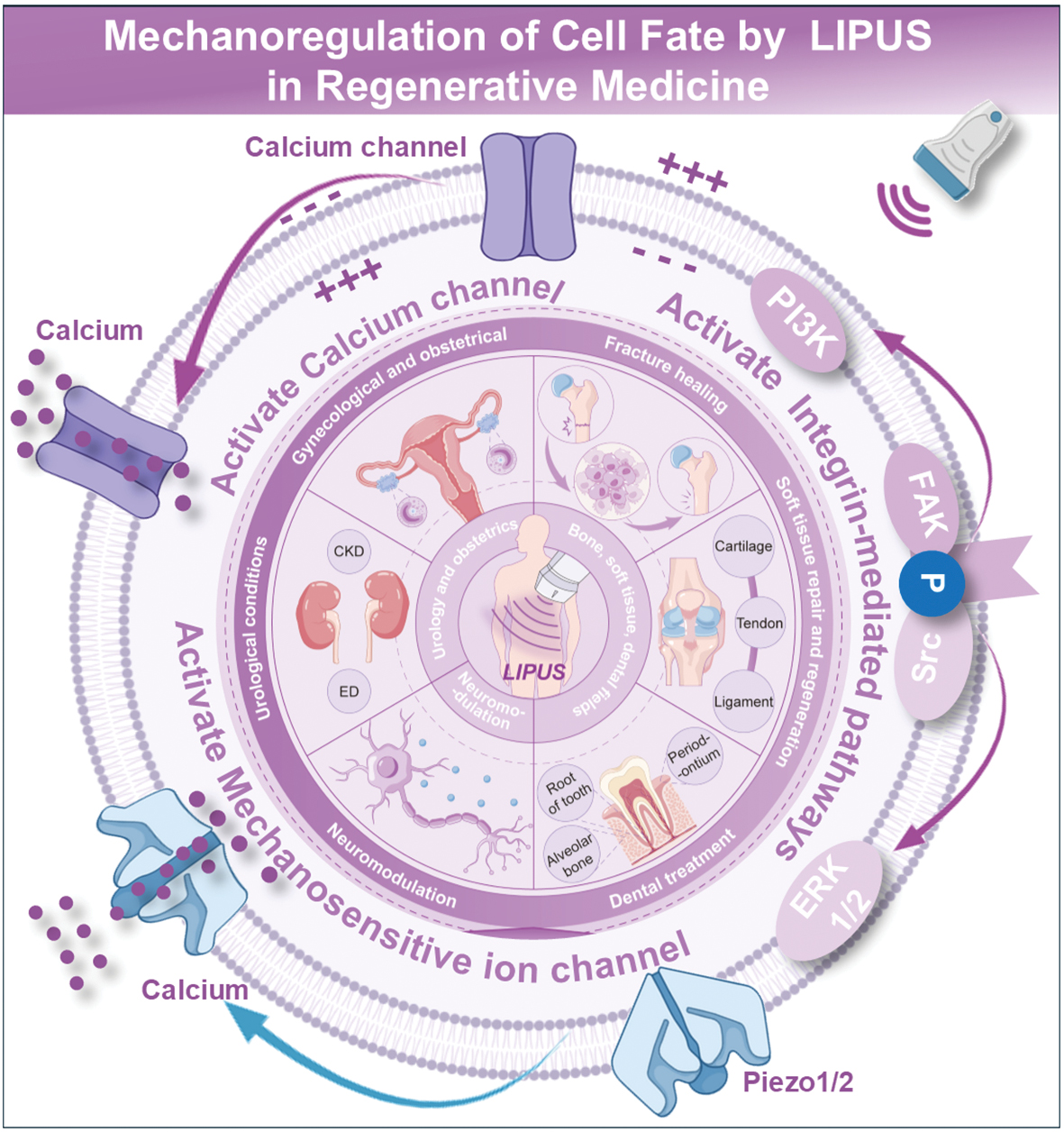
As a critical subset of LIU, low-intensity pulsed ultrasound (LIPUS) is characterized by low intensity (< 3 W/cm2), intermediate frequency (0.7–3 MHz), and pulsed-wave output. LIPUS generates mechanical signals through non-thermal mechanisms, including acoustic radiation force (steady momentum transfer in the direction of wave propagation) and cavitation effects, eliciting transient and sustained biological responses. Recent research has shown that mechanical stimuli promote cell differentiation during early embryogenesis and in vitro culture, which respond to morphogenetic movements, muscular activity, or substrate stiffness. Pathologic mechanical stimuli further disrupt effector cell proliferation and apoptosis, driving maladaptive remodeling and disease progression [5–7]. These mechanobiological principles now inform modern regenerative strategies, where the mechanical energy of ultrasound is harnessed to direct cell fate. Numerous studies have demonstrated its capacity to enhance cellular viability, stimulate proliferation, facilitate differentiation, and promote migration [8]. This mechanotransduction process occurs as LIPUS-generated mechanical waves activate ion channels or transmembrane proteins on the cell membrane, converting mechanical stimuli into biochemical signals that orchestrate cellular fate decisions. These signals subsequently regulate intracellular pathways, ultimately driving tissue repair and regeneration [9–11]. LIPUS activates intrinsic mechanotransduction networks, including ion channels and non-channel mediators. These pathways converge to regulate cellular processes critical for regeneration, including proliferation, differentiation, and apoptosis, which position LIPUS as a precision tool in regenerative medicine [11, 12].
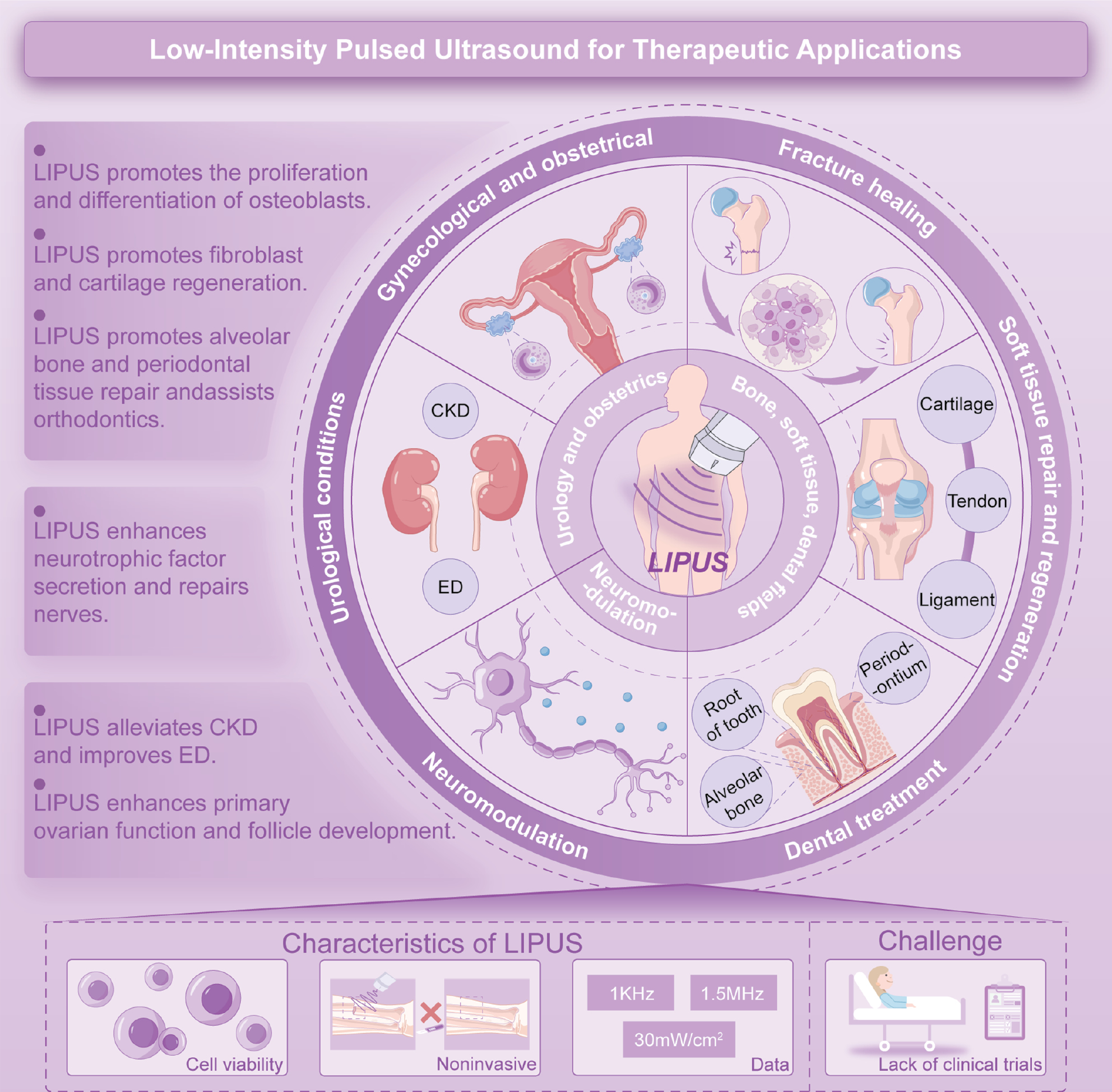
LIPUS applications span multiple clinical domains, offering a reliable and non-invasive treatment modality for fracture healing, soft tissue repair and regeneration, dental treatment, neuromodulation, urologic conditions, as well as obstetrics and gynecologic-related diseases [9, 10, 12–20]. The non-invasive nature of LIPUS addresses key limitations of traditional surgeries by reducing hospital length of stay, complications, and systemic burden on healthcare systems. Recent advances in regenerative medicine further expand the scope of LIPUS applications, enabling targeted interventions in neurologic repair. This review delineates the mechanoregulatory paradigms underpinning LIPUS therapy, exploring the dual modulation of ion channel-driven and cytoskeletal-mediated signaling cascades. In addition, we summarized translational advances across regenerative contexts, including skeletal, neural, renal, and reproductive systems, while highlighting unresolved challenges in parameter optimization and clinical validation (Figure 1). By bridging mechanistic insights with therapeutic innovation, this work aims to chart a roadmap for LIPUS-driven regenerative medicine in the era of precision bioengineering.
Figure 1 Schematic summary of LIPUS therapy in systemic treatments.
Mechanoregulation of cell fate: mechanisms underlying LIPUS for regenerative medicine
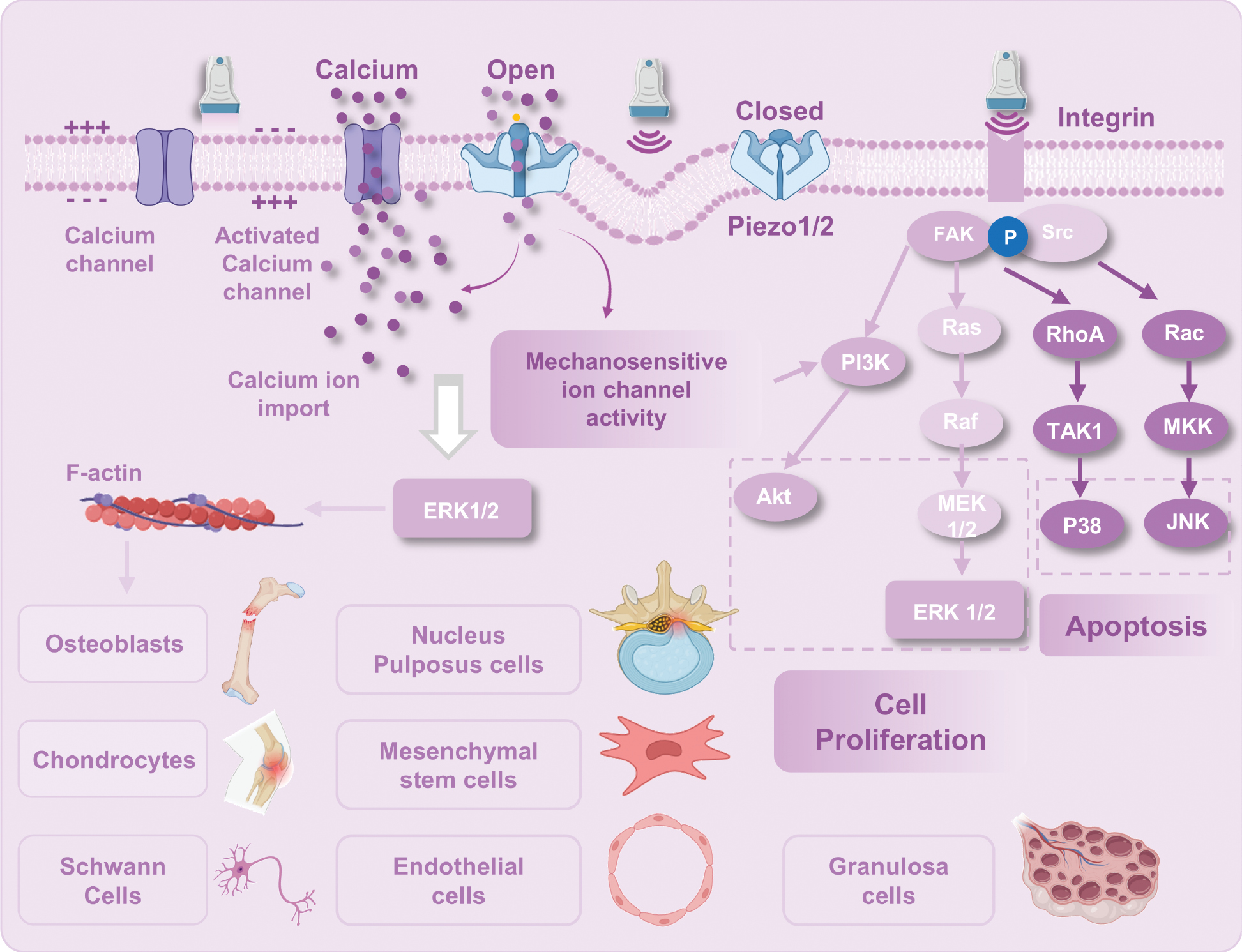
Ultrasound propagates through tissues as high-frequency pressure waves, generating mechanical forces (compression, tension, shear stress, and torque) that activate cellular mechanotransduction pathways. LIPUS, defined by non-thermal and pulsed parameters, exerts therapeutic effects primarily through mechanical interactions (Figure 2). A hallmark of ultrasound is the capacity to induce bioelectric signals via deformation of piezoelectric materials within tissues [21]. These bioelectric signals regulate critical processes, such as cell migration, proliferation, differentiation, neural conduction, muscle contraction, and tissue regeneration [22]. For example, bone collagen fibrils exhibit piezoelectric properties and mechanical stress induces bending and relaxation of the non-centrosymmetric conformation, which alters axial polarization to generate electrical signals [23, 24]. These signals activate voltage-gated calcium channels on cell membranes, modulating intra- and extra-cellular ion gradients [25].
Figure 2 Schematic diagram of LIPUS-mediated cellular fate modulation.
Calcium signaling in mechanotransduction
Calcium (Ca2+) signaling serves as a key initiating mechanism in mechanotransduction, coordinating diverse cellular functions [26]. Mechanical stimulation elevates cytosolic Ca2+ through extracellular influx (via voltage-gated channels) or release from intracellular stores [27, 28]. Ca2+ influx activates calmodulin-dependent pathways in osteoblasts, regulating transforming growth factor (TGF)-β1 levels and osteoblast behavior [23].
Mechanosensitive channels are multimeric membrane proteins responsive to mechanical forces that mediate ion flux (predominantly Ca2+) across cell membranes within milliseconds and convert mechanical stimuli into electrochemical signals. Mammalian Piezo1/2, which are non-selective cation channels with a Ca2+ preference, are activated by diverse mechanical cues, including membrane tension, shear stress, and stretching. Structural studies suggest that membrane tension shifts Piezo1 from a bent to an open conformation, enabling ion permeation [29, 30]. LIPUS-generated shear stress activates Mechanosensitive channels, such as TRP-4 in Caenorhabditis elegans sensory neurons and Piezo1 in primary mouse neurons, inducing Ca2+ influx and nuclear c-Fos expression to modulate neuronal activity [31, 32].
Integrin-mediated signaling and downstream pathways
Integrins are heterodimeric transmembrane glycoprotein receptors that serve dual roles in mechanotransduction [anchoring cells to the extracellular matrix (ECM) and converting mechanical stimuli into intracellular biochemical signals] [33]. This dual functionality positions integrins as central mediators of LIPUS-induced cell proliferation [34].
LIPUS generates cyclic mechanical strain through acoustic radiation force and microstreaming, directly acting on cell membranes or indirectly transmitting stress via ECM interactions [35]. These mechanical stimuli induce conformational changes in integrins, exposing intracellular domains to initiate downstream signaling. For example, during bone repair, LIPUS enhances integrin α5β1 binding to fibronectin and type I collagen by modulating mechanical strain gradients (1–10 με) in the callus region, thereby promoting osteoprogenitor cell migration and adhesion.
Activated integrins recruit focal adhesion adaptor proteins, triggering autophosphorylation of focal adhesion kinase (FAK) at Tyr397 and forming an FAK-Src complex [36]. This complex activates downstream signaling cascades. LIPUS significantly elevates phosphorylated FAK (p-FAK) levels in synovial cells, and FAK inhibition abolishes LIPUS-induced proliferation, underscoring FAK phosphorylation as a critical signaling node [37].
LIPUS-activated integrin-FAK signaling upregulates COX-2 expression, stimulating paracrine release of PGE2. PGE2 binds EP2/EP4 receptors on adjacent osteoblasts, activating the Wnt/β-catenin pathway to enhance bone formation. Integrin-mediated TGF-β1 receptor upregulation promotes aggrecan synthesis in intervertebral disc degeneration models and suppresses matrix metalloproteinase (MMP-3) activity, thereby preserving ECM integrity.
PI3K-Akt and MAPK pathways in proliferation and survival
The PI3K-Akt pathway, a key regulator of cell proliferation, is activated by LIPUS through multiple mechanisms [38]. Mechanical stimulation via integrins facilitates FAK/Src complex formation, which directly activates PI3K. Additionally, LIPUS enhances growth factor secretion (e.g., VEGF and TGF-β1), enabling ligand-receptor binding to RTK. RTK phosphorylation recruits PI3K via phosphotyrosine residues or Ras-dependent pathways. Activated PI3K converts phosphatidylinositol-4,5-bisphosphate (PIP2) to PIP3, which binds the pleckstrin homology (PH) domain of Akt, a serine/threonine kinase with three isoforms (Akt1, Akt2, and Akt3) expressed across tissues [39]. The PI3K/Akt axis promotes cell survival and proliferation by enhancing protein synthesis and inhibiting apoptosis. Akt phosphorylates and inactivates pro-apoptotic factors, while activating NF-κB to further bolster survival [40].
The Ras/ERK pathway, a branch of the MAPK cascade, governs cell proliferation, differentiation, and survival [41]. LIPUS stimulates MAPK signaling via integrin-mediated mechanotransduction. The MAPK family includes p38, ERK, and JNK. ERK1/2, which is ubiquitously expressed, regulates mitotic and post-mitotic functions, whereas JNK and p38 are stress-responsive kinases [42]. JNK phosphorylates c-Jun to enhance AP-1-mediated pro-apoptotic gene expression under stress conditions.
Wang et al. demonstrated dose-dependent effects of LIPUS on human adipose-derived stem cells (ADSCs). High-intensity LIPUS (210 mW/cm2) induced apoptosis via p38 MAPK activation, while low-intensity (70 mW/cm2) enhanced cell viability by suppressing p38 phosphorylation. p38 inhibitors reversed high-intensity LIPUS-induced apoptosis, confirming pathway specificity [43]. LIPUS activated ERK1/2 signaling in dental pulp stem cells (DPSCs), whereas periodontal ligament stem cells (PDLSCs) responded via JNK/p38 pathways. ERK1/2 or JNK/p38 inhibitors abolished LIPUS-induced proliferation in respective cell types, highlighting cell- and intensity-dependent MAPK regulation [44, 45].
LIPUS advances in regenerative medicine: regulating specific cells
LIPUS-mediated skeletal system repair
Promoting fracture healing
The global incidence of non-union/bone delayed healing has been reported to be between 5% and 10%, with an increasing trend [46]. Mechanical stimulation of bones is crucial for maintaining bone mass and structural stability. When bone is subjected to mechanical loading, the movement of fluid in the spaces around bone cells generates fluid shear stress, which stimulates osteoclasts and osteoblasts, enhancing anabolic bone remodeling activities to achieve appropriate bone resorption and subsequent new bone formation. As early as 1892, Wolff and colleagues proposed that LIPUS might induce bone formation by generating micro-mechanical stimulation in bone. Specifically, the differential absorption of LIPUS may create strain gradients in healing calluses, stimulating endosteal bone formation [47, 48]. Corradi and Cozzolino demonstrated that 500 mW/cm2 of ultrasound accelerated the healing of fresh radial fractures in rabbits compared to the control group. Similarly, Klug et al. reconfirmed acceleration of fracture healing with ultrasound in rabbits with tibial fractures [49]. Chang et al. showed that limbs treated with 500 mW/cm2 of ultrasound exhibited a 36% increase in new bone formation and an 80% increase in torsional stiffness compared to untreated limbs [50]. These studies collectively confirmed that LIPUS significantly stimulates and accelerates fresh fractures and in the treatment of aseptic and infected delayed unions and non-unions, improves healing rates 70%–93% [51].
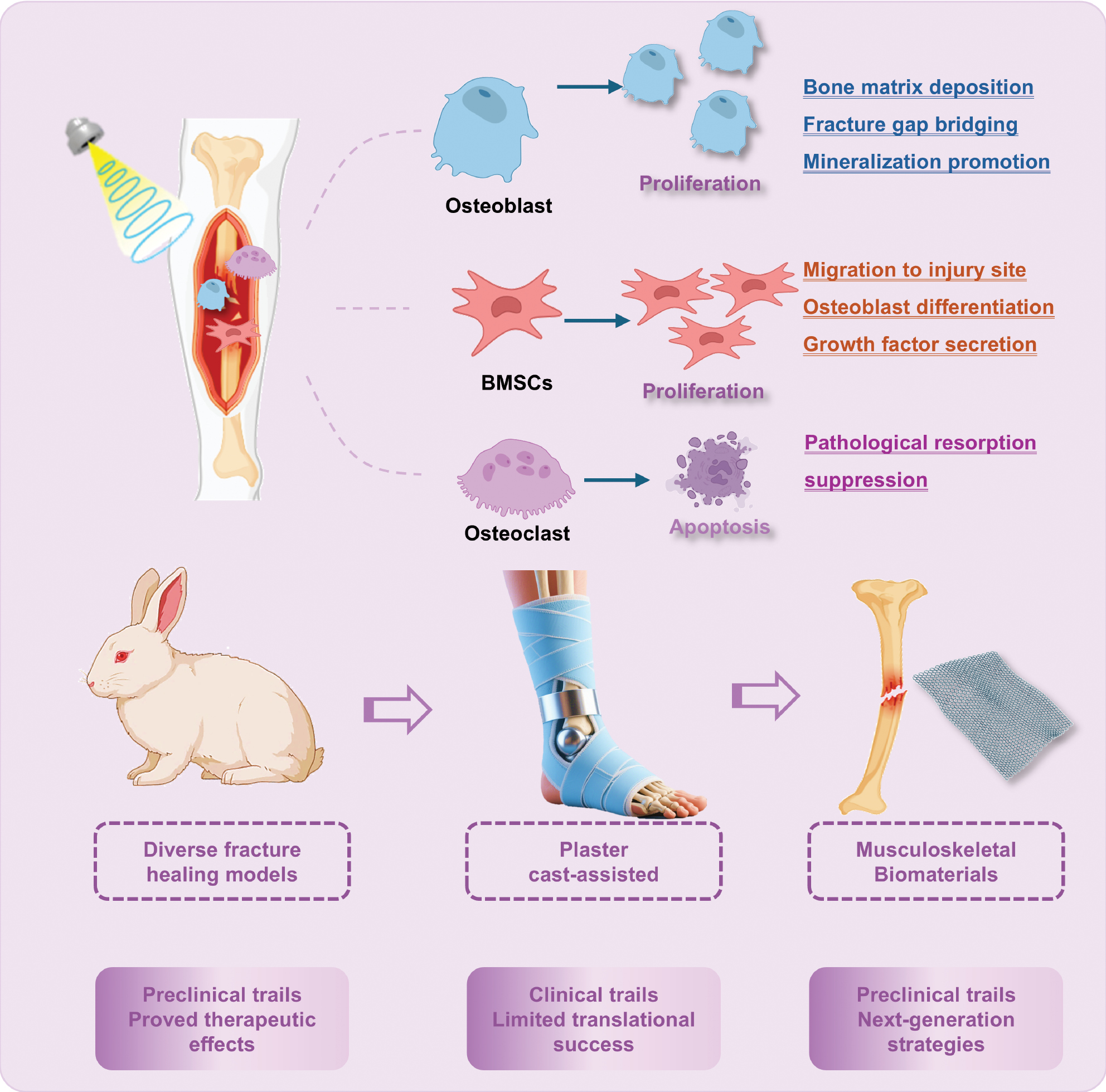
The exact mechanisms underlying the therapeutic effects of fracture healing have not been completely elucidated. Histologic evidence indicates that LIPUS modulates the activity of key cellular participants in bone repair, including osteoblasts, osteoclasts, and mesenchymal stem cells (MSCs) (Figure 3). Notably, these effects appear restricted to cells within soft tissues with a minimal impact observed on cells embedded in calcified bone matrices [51]. Osteoblasts accumulate at the injury site during fracture healing, actively synthesizing and secreting bone matrix to bridge the fracture gap and ultimately restoring both structural integrity and functional capacity of the bone.
Figure 3 Mechanisms and emerging trends of LIPUS in fracture repair.
Studies have suggested osteoblast proliferation and differentiation are mechanosensitive processes that are responsive to external mechanical, electrical, and magnetic stimuli [52]. These mechanotransduction pathways enable osteoblasts to adapt bone remodeling activities in accordance with mechanical loading conditions, thereby facilitating restoration of physiologic bone morphology and function [53, 54]. This mechanoregulatory framework provides a theoretical basis for LIPUS-mediated enhancement of osteoblast activity to accelerate fracture repair.
LIPUS exhibits pro-osteogenic effects by enhancing osteoblast proliferation, activation, and mineralization with mechanistic links to the upregulation of Sonic hedgehog (SHH) signaling. Experimental inhibition of SHH signaling significantly attenuates these effects, underscoring the critical role of SHH in mediating LIPUS therapeutic efficacy [55, 56]. Corollary studies revealed that LIPUS enhances calcium and phosphate uptake in pre-osteoblasts, facilitating mineralization processes [57]. Transcriptomic analyses performed by Tabuchi et al. identified LIPUS-induced upregulation of osteogenic regulators in murine pre-osteoblasts, including Cd200, Dmp1, Igf2, and Nr4a1, which are pivotal for osteoblast differentiation and skeletal development [58]. Notably, LIPUS stimulates COX-2 expression in osteoblasts, driving PGE2 synthesis [34, 59]. This bioactive lipid mediates the following two distinct reparative functions: recruitment of inflammatory cells through COX-2-dependent pathways, thereby triggering cytokine release to initiate tissue repair; and activation of the cAMP/PKA signaling cascade, which enhances osteoblast proliferation and promotes woven bone formation.
While preclinical data strongly support LIPUS as a bone anabolic modality, clinical outcomes remain heterogeneous. Randomized trials report conflicting evidence regarding LIPUS efficacy in long bone regeneration, especially in recalcitrant fractures with compromised healing potential [60] [Table 1]. Current clinical guidelines assign moderate-to-high confidence to the conclusion that LIPUS constitutes an inefficient allocation of healthcare resources for bone healing, driven by the lack of clinically significant benefits in fracture repair and disproportionate treatment costs [61]. This evidence-based assessment warrants a strategic shift toward prioritizing alternative interventions with higher mechanistic plausibility and demonstrated efficacy in accelerating osseous regeneration. Furthermore, research agendas should incorporate systematic investigations into the de-implementation of LIPUS in orthopedic practice, guided by frameworks for eliminating low-value care [62].
Table 1 Clinical Trials of LIPUS in Fracture Healing: Outcomes and Limitations
| Bone Fracture | LIPUS Protocol | Healing Results | Study Design (Population) | Year | Ref. |
|---|---|---|---|---|---|
| Closed or grade-I open tibial shaft fractures | 1.5 MHz, 200 μs burst, 1 kHz, 30 mW/cm2, 20 min/d for 20 weeks | Clinical healing: 86 ± 5.8 d (active) vs. 114 ± 10.4 d (placebo), P = 0.01. Overall healing: 96 ± 4.9 d (active) vs. 154 ± 13.7 d (placebo), P = 0.0001. | Prospective, randomized, double-blind, placebo-controlled multi-institutional trial (/) | 1994 | [150] |
| Dorsally angulated distal radial fractures | 1.5 MHz, 200 μs burst, 1 kHz, 30 mW/cm2, 20 min/d for 10 weeks | Time to union: 61 ± 3 d (active) vs. 98 ± 5 d (placebo), P < 0.0001 | Multicenter, prospective, randomized, double-blind, placebo-controlled trial (/) | 1997 | [151] |
| Fresh midshaft clavicle fractures, non-operatively treated | 1.5 MHz, 200 ms burst, 1 kHz, 30 mW/cm2, 20 min/d for 28 d | No significant difference in clinical healing time (P = 0.91) | Multicenter, double-blind, randomized, placebo-controlled trial [101] | 2008 | [61] |
| Delayed union of osteotomized fibula after high tibial osteotomy | 1.5 MHz, 200 μs burst, 1 kHz, 30 mW/cm2, 20 min/day at home | Osteoid thickness ↑47%, mineral apposition rate ↑27%, bone volume ↑33% (all P < 0.05). Direct bone formation observed vs controls | Prospective, double-blind, placebo-controlled, histomorphometric trial [13] | 2008 | [152] |
| Hallux valgus deformity treated with chevron osteotomy | 1.5 MHz, 200 ms burst, 1 kHz, 30 mW/cm2, 20 min/d | No significant differences in clinical or most radiographic parameters. Distal metatarsal articular angle stable in LIPUS group vs. relapse in placebo | Randomized, placebo-controlled, double-blind trial [44] | 2009 | [153] |
| Delayed unions of the tibial shaft | 1.5 MHz, 200 μs pulse, 1 kHz, 30 mW/cm2, 20 min/d for 16 weeks | BMD improvement 1.34× greater with LIPUS (P = 0.002). Bone gap area reduction favored LIPUS (P = 0.014) | Multicenter randomized sham-controlled trial [101] | 2010 | [154] |
| Posttraumatic tibial defects treated with callus distraction | 1.5 MHz, 200 μs impulse, 1 kHz, 30 mW/cm2, 20 min/d | Healing index: 33 d/cm (LIPUS) vs. 45 d/cm (control), 27% faster maturation. Fixator time shortened by 95 d in LIPUS group. | Prospective, randomized controlled trial [21] | 2014 | [155] |
| Standardized corticotomy in proximal tibial metaphysis | Daily 20 min treatment | No significant difference in time to frame removal. | Prospective, multicenter, double-blind, randomized controlled trial [62] | 2017 | [156] |
| Tibial shaft fractures managed with intramedullary nailing | Daily LIPUS | No significant difference in radiographic healing, functional recovery, or return to work. Mean cost higher for LIPUS. | Prospective, multicenter, randomized controlled trial (501) | 2017 | [157] |
| Mandibular prognathism treated with bilateral intraoral vertical ramus osteotomy | 1.5 MHz, 200 μs burst, 30 mW/cm2, 20 min/d for 3 weeks | Higher CT values in LIPUS group. Increased D1 grade area in LIPUS group on CT images. | Prospective cohort study [21] | 2019 | [158] |
Early investigations into LIPUS revealed dose-dependent limitations in bone healing efficacy. A seminal study by Warden et al. in 2001 demonstrated that conventional LIPUS parameters (30 mW/cm2 intensity) failed to mitigate spinal cord injury-induced bone loss, although alternative dosing regimens showed promise for intact bone preservation [63]. Subsequent analyses suggested that suboptimal acoustic intensity may critically undermine therapeutic outcomes. While the protocol using 30 mW/cm2 remains standard in clinical fracture management [64, 65], emerging preclinical evidence has identified 150 mW/cm2 as a superior intensity threshold, yielding enhanced bone mineral density, microarchitectural preservation, and biomechanical competence in both healthy and osteoporotic models [66]. Mechanistically, high-intensity LIPUS (150 mW/cm2) accelerates murine fracture healing through dual pathways: [1] potentiation of chondrocyte hypertrophy and callus chondrogenesis; and [2] stimulation of osteoblast recruitment and de novo bone formation, thereby optimizing endochondral ossification kinetics [67].
The therapeutic potential of LIPUS may be further amplified through integration with biomaterial scaffolds. Bioengineered matrices approved for bone tissue engineering could synergize with LIPUS-induced mechanotransduction, creating a regenerative niche to accelerate musculoskeletal repair [68]. However, material-specific parameter optimization represents a critical gap in current research. The acoustic responsiveness of biomaterials, which is influenced by composition, porosity, and viscoelastic properties, necessitates systematic characterization of intensity-frequency-duration matrices tailored to scaffold design. For example, hydrogel-based systems may require distinct intensity thresholds compared to ceramic or polymeric substrates to maximize energy coupling efficiency [69, 70].
Stimulating oral regeneration in orthodontics and periodontics
In addition to long bone applications, LIPUS has emerged as a novel therapeutic modality in dentistry, demonstrating promising therapeutic potential in orthodontics and periodontics despite the nascent integration into routine clinical practice [71]. Mechanistically, LIPUS suppresses pressure-induced alveolar bone resorption by downregulating Piezo1 expression while enhancing osteogenic capacity in mechanically stressed periodontal ligament cells [62]. Furthermore, LIPUS modulates the osteogenic potential of MSCs by promoting localized alveolar bone remodeling, increasing bone mineral density, mitigating vertical bone loss and gingival recession, and accelerating orthodontic tooth movement [63].
Preclinical studies have consistently indicated LIPUS efficacy in reducing orthodontically induced root resorption and enhancing osteoclast differentiation to optimize tooth displacement efficiency [54]. Clinical trials further validate these findings. El-Bialy et al. first demonstrated LIPUS potential in reducing root resorption and accelerating healing in a 2004 pilot trial [72] with subsequent studies confirming LIPUS effectiveness in human orthodontic root repair and introducing specialized low-power intraoral devices [73]. A single-blinded randomized controlled trial also established LIPUS utility in alleviating orthodontic separation pain, leveraging the safety, non-invasiveness, and cost-effectiveness of LIPUS [74, 75]. However, parameter optimization remains critical to maximize root resorption healing while minimizing potential pulpal side effects, which is a priority for future investigations.
LIPUS restores alveolar bone homeostasis by mitigating oxidative stress through PI3K-Akt/Nrf2 pathway regulation in periodontitis models [76]. The synergy of LIPUS with bone marrow-derived MSCs (BMSCs) enhances alveolar regeneration, which is partially attributed to improved BMSC homing to injury sites [77]. In addition to direct cellular effects, LIPUS amplifies the therapeutic potential of MSC-derived extracellular vehicles (EVs), which circumvent transplantation risks while delivering pro-regenerative signals. Zhang et al. demonstrated that LIPUS augments EV production and efficacy in counteracting experimental periodontitis-associated bone loss [78]. Despite robust preclinical evidence, clinical translation requires rigorous validation. Current research, while elucidating broad mechanistic frameworks, must address knowledge gaps in molecular cascades and parameter standardization to optimize LIPUS integration into precision dentistry.
Regulating cartilage degradation and soft tissue repair in osteoarthritis (OA)
LIPUS has shown promising potential in the management of OA by targeting the underlying mechanisms of cartilage degradation. OA is characterized by the progressive loss of cartilage, driven by inflammation and catabolic processes mediated by IL-1β and NF-κB. LIPUS has been shown to alleviate OA progression through multiple pathways, including a reduction in tissue degeneration, alleviation of pain, improved subchondral bone microstructure, and decreased sensory nerve distribution [79].
LIPUS has been shown to protect damaged cartilage in the early stage of OA by inhibiting the degradation of IL-β and stimulating the migration, proliferation, and differentiation of chondrocytes, especially in temporomandibular joint OA [80]. Furthermore, LIPUS enhances the inhibitory effect of synovial fluid on cartilage degradation, thereby worsening cartilage damage in the early stages of OA [81]. These findings provide valuable insights into the potential clinical application of LIPUS in the treatment of early symptomatic OA. In addition to the effects of LIPUS on cartilage, LIPUS has been shown to have a positive impact on tendon-bone healing [82]. Following LIPUS treatment at the patella-patellar tendon junction, significant increases in the histologic expression of VEGF were observed in chondrocytes and osteoblasts [83]. LIPUS also significantly increases the proportion of M2 macrophages and expression of anti-inflammatory mRNA, contributing to the therapeutic effects [84].
Sufficient vascular invasion has a critical role in the initiation of visceral ossification and soft tissue repair. LIPUS promotes tendon-bone junction healing by enhancing tissue perfusion and facilitating angiogenesis. Furthermore, MSCs are considered a promising autologous source for cartilage repair and numerous studies have demonstrated that LIPUS act as an effective stimulus to enhance MSC differentiation into chondrocytes in vivo [85, 86], thereby promoting tendon-bone injury healing. Compared to other external stimuli for tendon-bone healing, such as shockwaves, electrical stimulation, and combined magnetic fields, LIPUS is a safe, efficient, and cost-effective method. This feature makes the clinical application and translation of LIPUS more feasible in regenerative medicine [87].
Although numerous preclinical studies have demonstrated the therapeutic potential of LIPUS, the specific mechanisms underlying the effects of LIPUS in human musculoskeletal diseases, such as tendinopathy and OA, are incompletely understood [14]. Furthermore, translational studies have shown that the therapeutic benefits of LIPUS observed in animal models have bben difficult to replicate in patients [88]. Several clinical trials have failed to demonstrate the effectiveness of LIPUS in promoting ligament and muscle regeneration or alleviating symptoms in patients with tendinopathy and OA. For example, in clinical trials involving patellar tendinopathy and chronic lateral epicondylitis, the outcomes of the LIPUS treatment group were comparable to the control group (P > 0.05), indicating that the additional therapeutic effects of LIPUS in clinical applications are minimal [88, 89]. These discrepancies in translational studies may stem from physiologic differences between species, insufficient modeling of human pathophysiology in preclinical models, differences in clinical trial designs, and patient heterogeneity. Future research should focus on conducting large-scale, multi-center randomized controlled trials with standardized protocols to validate the efficacy of LIPUS across different disease stages and anatomic sites. Additionally, conducting mechanism studies using human-derived tissues or organoids may help bridge these translational gaps.
Delaying disc degeneration
Demonstrating robust pro-regenerative effects across diverse biological systems. Preclinical studies have highlighted the ability of LIPUS to stimulate fibroblast proliferation and enhance regeneration of cartilage, intervertebral discs, ligaments, and tendon sheaths [12]. Notably, LIPUS enables non-invasive healing of injured or degenerative intervertebral disc tissue by upregulating the transforming growth factor-beta 1 receptor in nucleus pulposus cells, thereby promoting cellular proliferation and ECM synthesis [44]. Mechanistic investigations have revealed that LIPUS treatment induces a 2.06-fold increase in tissue inhibitor of metalloproteinase-1 expression and a 2.3-fold elevation in monocyte chemoattractant protein-1 levels within nucleus pulposus cells, which facilitates macrophage recruitment and active participation in disc remodeling processes [90]. Taken together, these findings position LIPUS as a promising non-invasive therapeutic strategy for mitigating degenerative disc pathologies and alleviating chronic low back pain.
LIPUS-mediated neurologic repair
Accelerating peripheral nerve regeneration
LIPUS has shown significant therapeutic effects in neural modulation, especially in the peripheral nervous system [91]. Peripheral nerve injuries are common in clinical practice and often result in neuropathy, causing weakness, paralysis, sensory deprivation, neurogenic pain, and autonomic dysfunction. Repair of nerve injuries using neurotrophic factors has challenges, such as paradoxically inducing pain hypersensitivity through BDNF-mediated central sensitization and hyperexcitability of spinal WDR neurons [92]. LIPUS has demonstrated an essential role in peripheral nerve healing from multiple perspectives [18].
Mechanistically, LIPUS enhances neurotrophic factor secretion (e.g., BDNF and NGF), while suppressing inflammatory cascades in vitro, as demonstrated in models of peripheral neuropathy [93]. This effect is partially mediated through TrkB/Akt-CREB pathway activation, which upregulates neurotrophic expression and confers neuroprotection in rodent brains, suggesting therapeutic relevance for neurodegenerative disorders [35, 94]. LIPUS synergizes BMSC transplantation by augmenting BMSC survival and paracrine release of neurotrophic factors, yielding superior functional recovery compared to standalone cell therapy in preclinical models [95].
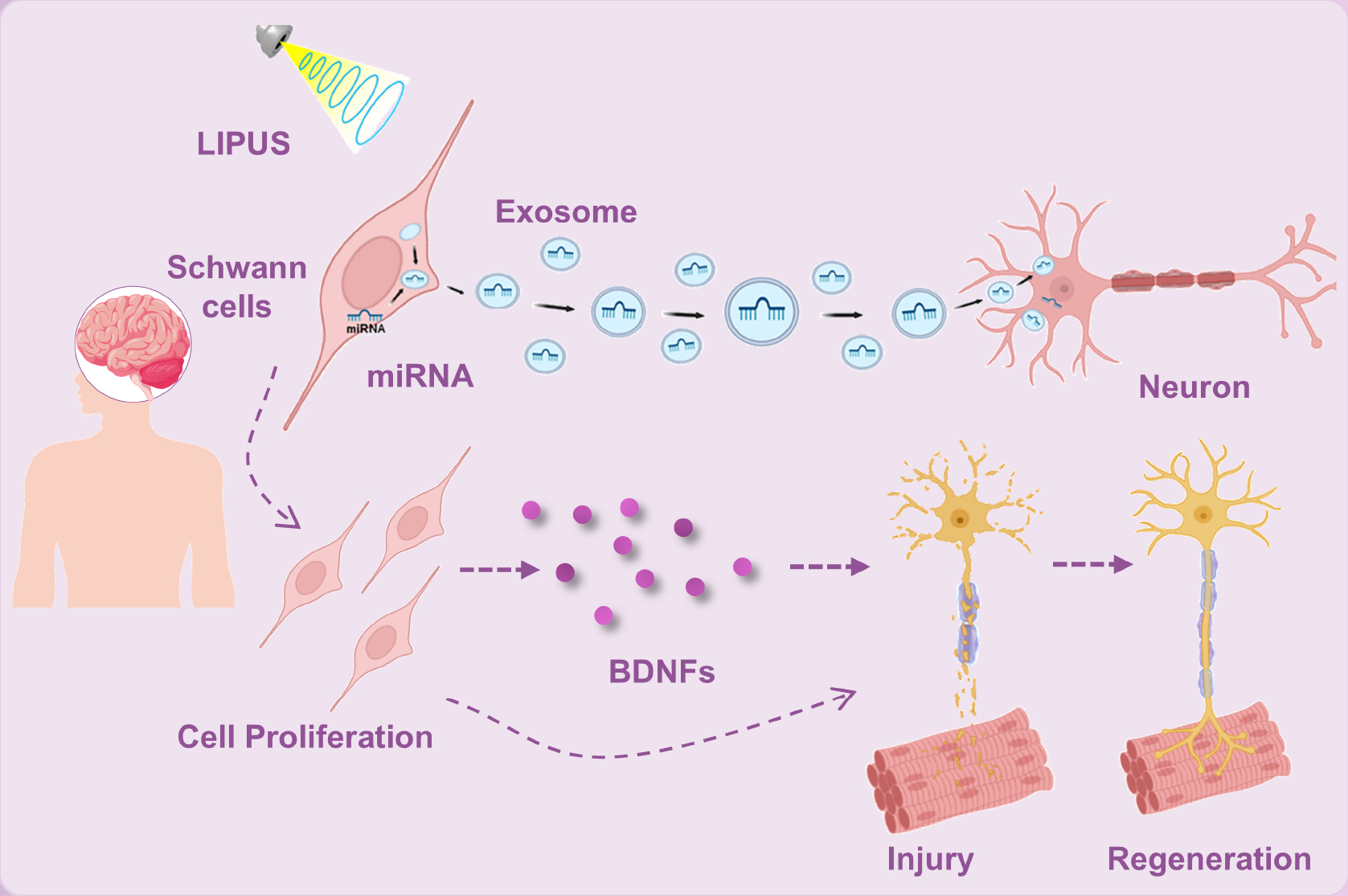
While a hypothesis suggests that LIPUS application directly enhances axonal regeneration, the majority of evidence supports the idea that LIPUS exerts therapeutic effects in peripheral nerve regeneration primarily through direct modulation of Schwann cell (SC) repair [19]. The mechanisms by which LIPUS positively influences nerve regeneration likely involve modulation of SCs, thereby facilitating the repair process. SCs are pivotal in peripheral nerve regeneration and have a critical role in the survival, regeneration, and reorganization of damaged axons.
It has been shown that LIPUS significantly impacts SC activity and proliferation. A seminal in vitro study published in 2009 demonstrated that LIPUS effectively promotes cell growth and expression of neurotrophic factor genes in SCs, highlighting the potential utility of LIPUS in peripheral nerve regeneration [96]. Subsequent work by Ren et al. validated LIPUS efficiency in enhancing SC viability and proliferation [97]. Furthermore, studies have indicated that LIPUS treatment may enhance nerve regeneration through the GSK-3β/β-catenin/cAMP signaling pathway.
Notably, previous research has shown that the absence of cyclin D1 in SC proliferation does not impede axonal regeneration or myelin regeneration in regenerating nerves [98]. These findings suggested that SC proliferation during Wallerian degeneration is not essential for the regeneration and functional recovery of damaged peripheral nerves. Consequently, further research is warranted to fully elucidate the molecular mechanisms underlying LIPUS promotion of nerve repair.
Recent investigations have explored additional avenues through which LIPUS may exert its therapeutic effects. For example, exosomes secreted by LIPUS-pretreated SCs have been shown to enhance axonal extension. Ye et al. reported that LIPUS stimulation regulates gene expression in pelvic neurons by modulating miRNA in exosomes derived from SCs, thereby activating the PI3K-Akt-FoxO signaling pathway to enhance nerve regeneration [99].
Innovative approaches to accelerating nerve healing involve enhancing SC function. A study demonstrated that co-culturing ADSCs with SCs combined with LIPUS stimulation synergistically enhances the myelination capacity of SCs [100]. This effect is attributed to the secretion of growth factors or cytokines by ADSCs, which promote the myelination process of SCs. However, this research remains largely confined to laboratory settings and awaits validation in clinical trials. To optimize the therapeutic effects of LIPUS, further research is necessary to explore the impact of prolonged application. Such studies will contribute to a deeper understanding of the underlying mechanisms and potential applications of LIPUS in clinical settings.
The repair efficiency of LIPUS in neural modulation is strongly influenced by the dose that is administered. While low-intensity ultrasound has been shown to promote nerve regeneration, higher intensities may hinder this process, as demonstrated in animal models [101]. Notably, there is considerable variability in the intensity parameters across studies with effective dosages for nerve regeneration ranging from 0.016 W/cm2 to 1 W/cm2 [102]. Additionally, the effects of LIPUS on SC proliferation and myelination depend on exposure time and duty cycle. Studies have indicated that repeated applications with higher duty cycles enhance SC proliferation with the most significant effects observed at a 50% duty cycle [103]. These findings suggest an incremental approach, starting with lower duty cycles and gradually increasing to the optimal level for maximal therapeutic benefit.
LIPUS exerts its neural regenerative effects by modulating neural cells and stem cell activity, regulating neurotrophic factors, pro-inflammatory cytokines, and activating critical signaling pathways [18]. While LIPUS has shown promising results in preclinical studies, LIPUS has not been widely implemented in clinical treatments for nerve injuries. The evidence underscores the potential of LIPUS as a non-invasive modality for neural repair. However, the variability in study protocols, such as differences in intensity, duration, and duty cycle, necessitates further investigation to establish standardized parameters for clinical use. Although progress is being made in understanding the molecular mechanisms underlying ultrasound-mediated neural modulation, a comprehensive mechanistic framework is still lacking. Addressing these gaps will be essential for translating LIPUS therapies into clinical practice, enabling a broader application in the treatment of peripheral nerve injuries (Figure 4).
Figure 4 LIPUS-mediated repair of peripheral nerve injury.
Modulating central nervous system function
The translational scope of LIPUS extends to central nervous system pathologies, with emerging evidence highlighting the multimodal therapeutic mechanisms. Transcranial LIPUS has demonstrated disease-modifying potential in Parkinson’s disease (PD) models. Specifically, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced mice exhibited improved motor performance in open-field and pole tests (P < 0.05) with elevated striatal antioxidant enzymes (T-SOD and GSH-PX; P < 0.03) post-LIPUS, while histologic analyses confirmed absence of tissue damage [104]. Parallel studies in 6-OHDA-lesioned rats revealed unilateral LIPUS stimulation upregulated glial cell line-derived neurotrophic factor (GDNF) by 1.98-fold in the substantia nigra pars compacta (SNpc) and attenuated lipidocalin-2-mediated neuroinflammation, which correlated with restored tyrosine hydroxylase density [105, 106].
By elevating endogenous BDNF levels and modulating neuroinflammatory pathways, LIPUS mitigates disease progression in Alzheimer’s disease (AD) and PD models, the effects of which were corroborated by improved cognitive outcomes in a randomized, double-blind trial of AD patients [105–108]. Notably, human trials in PD with cognitive impairment (PD-CI) further validate clinical translatability. Specifically, an 8-week LIPUS regimen (n = 28) significantly enhanced Mini-Mental State Examination (MMSE)/Montreal Cognitive Assessment (MoCA) scores (P < 0.05), reduced anxiety/depression indices (Beck Anxiety Inventory [BAI]/Beck Depression Inventory [BDI]), and upregulated serotonin, norepinephrine, and dopamine levels compared to sham controls, suggesting systemic neuromodulatory effects [6]. Pioneering human studies by Shindo et al. have safely applied transcranial LIPUS (0.5 MHz and 1.3 MPa spatial peak pressure) to modulate brain circuits, although long-term efficacy remains unvalidated [109].
Mechanistically, LIPUS synergizes with nanotechnology to overcome the blood-brain barrier (BBB). LIPUS-enhanced delivery of felodipine nanodrugs redirected endoplasmic reticulum stress toward antioxidative signaling in AD models, suppressed NLRP3 inflammasome activation, and rescued anxiety-like behaviors in 5xFAD mice [110]. Similarly, LIPUS-facilitated transport of 30-nm PEG-Q10 nanoparticles reduced amyloid plaques and formaldehyde toxicity in APP/PS1 mice with combined therapy outperforming standalone drug delivery [P < 0.05] [111]. These advances underscore the dual role of LIPUS as a standalone neuromodulator and a facilitator of precision drug delivery, positioning LIPUS as a cornerstone for next-generation neurotherapeutics.
LIPUS-mediated renal and erectile function recovery
Renal function recovery by targeting inflammation and fibrosis
The pathogenesis of chronic kidney disease (CKD) is largely influenced by renal inflammation and fibrosis. Therefore, inhibiting renal inflammation and fibrosis to protect kidney function is crucial [112]. LIPUS has been shown to have significant anti-inflammatory and repair properties, making LIPUS a promising treatment option for CKD [113]. Aibara et al. provided preliminary evidence indicating that daily LIPUS treatment has beneficial effects on renal fibrosis and inflammation in experimental models of hypertensive and diabetic nephropathy [114]. Moreover, it was found that LIPUS exposure directly inhibits the TGF-β1/Smad signaling pathway. These findings suggested that LIPUS may partly alleviate the progression of renal fibrosis by suppressing inflammatory cell infiltration. Corollary studies confirmed that LIPUS treatment effectively improves body weight, the albumin/globulin ratio, serum kidney function markers, renal pathologic changes, and renal fibrosis in CKD mice. LIPUS slows the progression of CKD by inhibiting epithelial-mesenchymal transition and age-related signaling [115]. Additionally, Ouyang et al. found that LIPUS application alleviates renal inflammation and fibrosis by inhibiting ferroptosis, thus protecting kidney function in CKD [116]. Furthermore, LIPUS may have a crucial role in modulating the TGF-β1/Smad and Nrf2/Keap1/HO-1 signaling pathways, thus inhibiting ferroptosis. The authors further confirmed that the therapeutic effects of LIPUS are related to modulation of the NF-kB and ferroptosis signaling pathways, with Fgf23, Fgg, and Cyp2c11 identified as potential LIPUS targets in improving renal fibrosis and dysfunction [117]. Recent studies have also shown that LIPUS effectively improves muscle mass loss, muscle weakness, protein expression related to muscle atrophy, and Akt inactivation in CKD-induced muscle atrophy [118]. However, further research is needed to fully understand the complex mechanisms and determine the potential clinical applications.
Chiang et al. demonstrated that LIPUS effectively reverses the renal and splenic inflammatory responses induced by ischemia/reperfusion injury in mice. This finding suggested that LIPUS, as a non-invasive therapeutic intervention, holds significant potential for treating renal injury or as an adjunct tool for acute kidney injury (AKI) management [119]. Macrophages have a critical role in the pathogenesis of AKI and LIPUS is being explored as an immunomodulatory intervention. Laboratory studies have shown that LIPUS effectively inhibits the expression of M1 macrophage-related genes, while significantly promoting the expression of M2 macrophage-related genes. This modulation is accompanied by suppression of apoptosis and a significant decrease in heat shock protein-70 levels, ultimately leading to the improvement of AKI caused by gemcitabine [120]. However, a comprehensive understanding of the long-term effects of LIPUS in AKI or chronic kidney disease (CKD) patients and optimization of LIPUS therapy is incomplete. Therefore, long-term follow-up studies are crucial to evaluate the efficacy of LIPUS post-AKI or CKD and gain insights into its long-term benefits.
Erectile function recovery
Erectile dysfunction (ED) is a common condition that negatively affects various physiologic and psychosocial aspects. It is estimated that the prevalence of ED exceeds 50% among men 40–70 years of age [121]. The extant literature relevant to the regenerative properties of LIPUS is promising and provides potential opportunities for many men to restore spontaneous erectile function [122]. The therapeutic effects of LIPUS in treating nerve damage and diabetes-induced ED have been explored. In 2015 Lei et al. reported that LIPUS therapy has encouraging results in improving erectile function and effectively reverses adrenal tissue lesions in diabetic rats [123]. Furthermore, damage to the corpus cavernosum endothelial cells has a crucial role in the development of diabetes-induced ED. Studies have shown that by activating mitochondrial autophagy, LIPUS effectively alleviates endothelial cell dysfunction in the corpus cavernosum caused by advanced glycation end-products [124].
Additionally, similar to other adjunct stem cell therapies utilizing LIPUS, combining MSC transplantation with LIPUS may become a synergistic therapy for diabetic ED. It has been reported that LIPUS enhances the therapeutic effects of MSCs in the treatment of diabetic ED by activating the Piezo-ERK-VEGF pathway in rats [125]. In 2019 a randomized, double-blind, placebo-controlled clinical trial conducted in a clinical setting first demonstrated the efficacy of LIPUS in treating mild-to-moderate ED induced by diabetes with no major adverse events [126]. The observed results can be attributed to the mechanical forces exerted by LIPUS and the ability of LIPUS to restore the pathologic changes in the corpus cavernosum. Chen et al. found that for younger, overall healthy patients with mild-to-moderate ED, LIPUS therapy performed 2 or 3 times a week showed similar therapeutic effects and safety [127].
Recent clinical studies suggest that LIPUS may also produce positive therapeutic effects in patients with severe ED, especially in the absence of other co-morbidities [128]. Moreover, the application of LIPUS stimulation may offer therapeutic benefits for ED induced by cavernous nerve injury [129]. Studies have shown that LIPUS effectively improves erectile function in rats with neurogenic ED, potentially by regulating YAP/TAZ-mediated mechanotransduction [130]. Further confirmation of these statements is pending. Research on the use of LIPUS in treating urinary system diseases is still in the early stages and additional multi-center randomized placebo-controlled studies are warranted before LIPUS can be adopted as a standard clinical treatment for ED. Dose studies with supportive measures will also be required at this stage.
Targeting apoptosis and stem cell activation in ovarian regeneration
Recent advances in bio-ultrasound research have propelled the clinical translation of ultrasound therapies in obstetrics and gynecology. While high-intensity ultrasound has established clinical utility in targeted gynecologic interventions, low-intensity modalities, such as LIPUS, are emerging as promising tools for ovarian dysfunction management. Preclinical studies highlight the potential in addressing primary ovarian insufficiency (POI), a condition associated with premature ovarian failure and systemic health risks. Mechanistically, LIPUS enhances follicular development by stimulating revascularization and follicular growth in animal models [131], while concurrently increasing sex hormone secretion, reducing follicular atresia, and suppressing granulosa cell apoptosis [132]. Tang et al. showed that LIPUS restores ovarian function in POI rats through histologic repair, normalizing estrous cycles, elevating follicle counts, and improving hormonal profiles [133].
A paradigm-shifting application involves combining LIPUS with MSC therapy (Table 2). MSCs are multipotent stromal cells isolated from diverse tissue sources, including bone marrow, adipose tissue, dental pulp, amniotic membrane, placenta, umbilical cord, and umbilical cord blood. These cells exhibit self-renewal capacity and multilineage differentiation potential [134]. Under specific conditions, MSCs can differentiate into diverse cell lineages of mesodermal origin, including osteocytes, chondrocytes, and adipocytes. MSCs have been extensively utilized in tissue engineering and are recognized as pivotal therapeutic agents in regenerative medicine [135, 136]. Pretreatment with LIPUS enhances MSC viability and directional migration, potentiating the therapeutic efficacy of MSCs in ovarian repair [137]. CXCR4-mediated chemotaxis may underlie the improved homing of LIPUS-primed MSCs to inflammatory ovarian microenvironments, where MSCs attenuate inflammation and inhibit germinal center apoptosis [138].
Table 2 Therapeutic Application of LIPUS in Conjunction with Stem Cells or Exosomes
| Diseases | Stem Cells | Parameter (LIPUS) | Therapeutic Effects | Ref. |
|---|---|---|---|---|
| Orthodontic tooth movement | hBMSCs | F = 1.5 MHz; I = 30 mW/cm2; DC = 20%; T = 20 min |
Enhanced BMSCs homing and promoted alveolar bone regeneration via LIPUS stimulation | [159] |
| Alveolar bone loss | BMSCs | Reduced alveolar bone resorption and accelerated tooth movement by modulating the cytoskeleton-Lamin A/C-YAP axis | [77] | |
| Periodontitis | Stem cells from apical papilla (SCAP) | F = 1.5 MHz; I = 90 mW/cm2; DC = 20%; T = 30 min |
Enhanced osteogenic differentiation and anti-inflammatory effects of SCAP-EVs via LIPUS stimulation | [78] |
| Spinal cord injury | BMSCs | I = 50 mW/cm2; T = 3 min |
Improved BMSCs viability, migration, and neurotrophic factor expression; promoted functional recovery | [95] |
| Diabetic erectile dysfunction | ADSCs | F = 1.7 MHz; I = 200 mW/cm2; DC = 20%; T = 5 min |
Enhanced ADSC-mediated angiogenesis and improved erectile function via the Piezo-ERK-VEGF axis | [125] |
| Premature ovarian insufficiency | hAD-MSCs | F = 0.25 MHz; I = 30 mW/cm2; DC = 20%; T = 30 min |
Enhanced migration and homing of hAD-MSCs to ovaries; improved ovarian function and reduced ovarian injury | [138] |
POI-associated complications extend beyond ovarian dysfunction because estrogen deficiency in POI often precipitates osteoporosis. LIPUS demonstrates dual therapeutic utility in patients with POI. Specifically, amplitude-modulated pulsed ultrasound (a variant of LIPUS) promotes bone formation and mitigates bone loss by optimizing acoustic energy distribution to osteocytes, thereby enhancing bone microstructure and strength [139]. In addition to direct ovarian repair, LIPUS may mitigate chemotherapy-induced gonadotoxicity. Emerging evidence suggests a cytoprotective role for LIPUS in reducing ovarian damage and preserving follicular reserves during chemotherapeutic regimens [137]. While preclinical data robustly support LIPUS efficacy, clinical validation remains imperative. Future research must prioritize mechanistic elucidation of LIPUS-induced cellular responses, tissue-specific parameter optimization, and translational frameworks to bridge the gap between experimental models and human applications.
Conclusions
LIPUS represents a promising non-invasive approach to regulate cellular behavior and tissue regeneration through mechanotransduction pathways with preclinical evidence underscoring the capacity of LIPUS to enhance osteogenesis, promote neural repair, and synergize with biomaterials. Notwithstanding the potential of LIPUS, clinical translation remains contentious, marked by inconsistent outcomes in fracture healing, musculoskeletal repair, and neural regeneration. As previously mentioned, although mechanistic studies highlight the ability of LIPUS to activate critical signaling cascades and modulate neurotrophic factors, clinical trials often fail to replicate these benefits. Key controversies arise from methodologic limitations, including unblinded designs, inadequate sham controls, and suboptimal parameter selection. Furthermore, physiologic disparities between preclinical models and humans complicate extrapolation, underscoring the need for translational frameworks that reconcile interspecies differences.
Furthermore, the mechanisms underlying the differential regulatory effects of ultrasound-induced mechanical forces across tissues require further elucidation. Substantial research has emphasized the critical need for system optimization of tissue-specific acoustic parameters, including intensity, frequency, pulse repetition frequency (PRF), and duty cycle [140]. Preclinical models demonstrate that higher intensities (e.g., 150 mW/cm2) achieve superior bone regeneration compared to conventional settings [66]. Notably, ultrasound parameters exhibit dual regulatory effects (stimulatory versus inhibitory) depending on the configurations. While elevated intensities enhance blood perfusion, excessive energy delivery risks BBB disruption [141]. This dichotomy may stem from threshold-dependent activation of cellular signaling pathways, in which lower frequencies (e.g., 350 kHz) preferentially activate TRPA1 channels, whereas higher frequencies (e.g., 43 MHz) stimulate Piezo1 channels [142]. Heterogeneous receptor distribution contributes to ultrasound-mediated tissue heterogeneity. Recent studies reveal that LIPUS activates the mechanosensitive receptor RET in colonic tissues via compressive stress waves (rather than shear strain or thermal effects), subsequently triggering the WNT/β-catenin pathway to promote cellular proliferation [11]. This mechanism parallels LIPUS-mediated bone repair involving Piezo1 activation or MAPK/ERK pathway induction, suggesting mechanotransduction (e.g., pressure-dependent protein conformational changes) as a conserved regulatory mechanism across tissues. Nevertheless, inter-tissue variability arises from receptor diversity and differential ultrasound energy absorption between compliant soft tissues (e.g., colon) and rigid osseous structures. Additionally, skull bone density variations pose challenges for LIPUS-mediated neuromodulation [143]. Tissue-specific parameter sensitivity is evident in cerebral blood flow (CBF) regulation, which responds strongly to intensity and stimulation duration but minimally to duty cycle adjustments [144]. Intriguingly, pulse repetition tuning may enable selective modulation of excitatory versus inhibitory neurons [145]. These findings collectively underscore the necessity for systematic exploration of the ultrasound parameter space to optimize therapeutic precision across biological systems.
Equally critical is the integration of LIPUS with adjuvant therapies to amplify the therapeutic impact. Coupling LIPUS with osteoconductive biomaterials or hydrogels could spatially guide cell differentiation and callus formation, while its synergy with stem cell therapies or growth factor delivery (e.g., BMP-2, VEGF) might enhance endogenous repair mechanisms [68, 146, 147]. Innovations, such as ultrasound-responsive nanocarriers for targeted drug release in neural or musculoskeletal niches, could further unlock combinatorial potential [148]. Clinically, bridging preclinical and human studies through advanced models, such as organ-on-chip systems or human-derived 3D tissue constructs, could elucidate species-specific mechanoresponses and refine parameter selection [149]. These efforts must be paralleled by translational research to dissect the molecular underpinnings of ultrasound-mediated cellular responses, ensuring parameter optimization aligns with human biology.
Data availability
All data generated and/or analyzed during this study are included in this published article.
Funding sources
This work was supported in part by the National Natural Science Foundation of China (grant number 82102054), the Clinical Research 4310 Program of the Affiliated Changsha Central Hospital of the University of South China (grant number 20214310NHYCG06), and the Health Research Project of Hunan Provincial Health Commission (grant number W20241010).
Declaration of competing interest
The authors declare no competing interests.
Graphical abstract
Highlights
- LIPUS uses non-thermal mechanical forces (radiation force, cavitation) for therapy.
- LIPUS activates cellular mechanotransduction via ion channels and cytoskeletal pathways.
- LIPUS promotes regeneration: cell proliferation, differentiation, and tissue repair.
- Clinical applications span skeletal, neural, renal, and reproductive systems.
- Review addresses LIPUS mechanisms, advances, and unresolved clinical challenges.
In brief
This review explores low-intensity pulsed ultrasound (LIPUS), an emerging non-invasive therapy leveraging mechanical forces for tissue regeneration. It details how LIPUS activates cellular mechanotransduction via ion channels and cytoskeletal pathways, driving proliferation and repair. We summarize translational advances across skeletal, neural, renal, and reproductive systems, while addressing key clinical challenges and future directions for precision regenerative medicine.
References
- Feng R, Sheng H, Lian Y. Advances in using ultrasound to regulate the nervous system. Neurol Sci 2024;45(7):2997-3006. [PMID: 38436788 DOI: 10.1007/s10072-024-07426-7]
- O’Reilly MA. Exploiting the mechanical effects of ultrasound for noninvasive therapy. Science 2024;385(6714):eadp7206. [PMID: 39265013 DOI: 10.1126/science.adp7206]
- Focused Ultrasound Foundation. 2023 State of the Field Report. Charlottesville, VA: Focused Ultrasound Foundation; 2023.
- Jiang X, Savchenko O, Li Y, Qi S, Yang T, et al. A review of low-intensity pulsed ultrasound for therapeutic applications. IEEE Trans Biomed Eng 2019;66(10):2704-18. [PMID: 30596564 DOI: 10.1109/TBME.2018.2889669]
- Hu X, Zhang Y. Developing biomaterials to mediate the spatial distribution of integrins. Biophys Rev 2021;2(4):041302. [PMID: 38504718 DOI: 10.1063/5.0055746]
- Chen J, Zhou Y, Liu S, Li C. Biomechanical signal communication in vascular smooth muscle cells. J Cell Commun Signal 2020;14(4):357-76. [PMID: 32780323 DOI: 10.1007/s12079-020-00576-1]
- Li L, Hanahan D. Hijacking the neuronal NMDAR signaling circuit to promote tumor growth and invasion. Cell 2013;153(1):86-100. [PMID: 23540692 DOI: 10.1016/j.cell.2013.02.051]
- Xia P, Shi Y, Wang X, Li X. Advances in the application of low-intensity pulsed ultrasound to mesenchymal stem cells. Stem Cell Res Ther 2022;13(1):214. [PMID: 35619156 DOI: 10.1186/s13287-022-02887-z]
- Guo X, Lv M, Lin J, Guo J, Lin J, et al. Latest progress of LIPUS in fracture healing: a mini-review. J Ultrasound Med 2024;43(4):643-55. [PMID: 38224522 DOI: 10.1002/jum.16403]
- Aimaijiang M, Liu Y, Zhang Z, Qin Q, Liu M, et al. LIPUS as a potential strategy for periodontitis treatment: a review of the mechanisms. Front Bioeng Biotechnol 2023;11:1018012. [PMID: 36911184 DOI: 10.3389/fbioe.2023.1018012]
- Zamfirov L, Nguyen NM, Fernández-Sánchez ME, Cambronera Ghiglione P, Teston E, et al. Acoustic-pressure-driven ultrasonic activation of the mechanosensitive receptor RET and of cell proliferation in colonic tissue. Nat Biomed Eng 2024. [PMID: 39706982 DOI: 10.1038/s41551-024-01300-9]
- Xin Z, Lin G, Lei H, Lue TF, Guo Y. Clinical applications of low-intensity pulsed ultrasound and its potential role in urology. Transl Androl Urol 2016;5(2):255-66. [PMID: 27141455 DOI: 10.21037/tau.2016.02.04]
- Palanisamy P, Alam M, Li S, Chow SKH, Zheng YP. Low-intensity pulsed ultrasound stimulation for bone fractures healing: a review. J Ultrasound Med 2022;41(3):547-63. [PMID: 33949710 DOI: 10.1002/jum.15738]
- Qin H, Du L, Luo Z, He Z, Wang Q, et al. The therapeutic effects of low-intensity pulsed ultrasound in musculoskeletal soft tissue injuries: focusing on the molecular mechanism. Front Bioeng Biotechnol 2022;10:1080430. [PMID: 36588943 DOI: 10.3389/fbioe.2022.1080430]
- Lai WC, Iglesias BC, Mark BJ, Wang D. Low-intensity pulsed ultrasound augments tendon, ligament, and bone-soft tissue healing in preclinical animal models: a systematic review. Arthroscopy 2021;37(7):2318-33e3. [PMID: 33621647 DOI: 10.1016/j.arthro.2021.02.019]
- Li F, Li Y, Zhu Y, Bao X, Wang L. Recent advances in basic studies of low-intensity pulsed ultrasound in periodontal tissue regeneration: a systematic review. Stem Cell Rev Rep 2024;20(8):2124-37. [DOI: 10.1007/s12015-024-10769-5]
- Zhong YX, Liao JC, Liu X, Tian H, Deng LR, et al. Low intensity focused ultrasound: a new prospect for the treatment of Parkinson’s disease. Ann Med 2023;55(2):2251145. [PMID: 37634059 DOI: 10.1080/07853890.2023.2251145]
- Liu X, Zou D, Hu Y, He Y, Lu J. Research progress of low-intensity pulsed ultrasound in the repair of peripheral nerve injury. Tissue Eng Part B Rev 2023;29(4):414-28. [PMID: 36785967 DOI: 10.1089/ten.TEB.2022.0194]
- Acheta J, Stephens SBZ, Belin S, Poitelon Y. Therapeutic low-intensity ultrasound for peripheral nerve regeneration – a Schwann cell perspective. Front Cell Neurosci 2021;15:812588. [PMID: 35069118 DOI: 10.3389/fncel.2021.812588]
- Ji X, Duan H, Wang S, Chang Y. Low-intensity pulsed ultrasound in obstetrics and gynecology: advances in clinical application and research progress. Front Endocrinol (Lausanne) 2023;14:1233187. [PMID: 37593351 DOI: 10.3389/fendo.2023.1233187]
- Cai K, Jiao Y, Quan Q, Hao Y, Liu J, et al. Improved activity of MC3T3-E1 cells by the exciting piezoelectric BaTiO3/TC4 using low-intensity pulsed ultrasound. Bioactive Mater 2021;6(11):4073-82. [PMID: 33997494 DOI: 10.1016/j.bioactmat.2021.04.016]
- Jin F, Li T, Wei Z, Xiong R, Qian L, et al. Biofeedback electrostimulation for bionic and long-lasting neural modulation. Nat Commun 2022;13(1):5302. [DOI: 10.1038/s41467-022-33089-z]
- Sun J, Xie W, Wu Y, Li Z, Li Y. Accelerated bone healing via electrical stimulation. Adv Sci (Weinh) 2024;e2404190. [PMID: 39115981 DOI: 10.1002/advs.202404190]
- Fukada E, Yasuda I. On the piezoelectric effect of bone. J Phys Soc Japan 1957;12:1158-62. [DOI: 10.1143/JPSJ.12.1158]
- Wright CS, Robling AG, Farach-Carson MC, Thompson WR. Skeletal functions of voltage sensitive calcium channels. Curr Osteoporos Rep 2021;19(2):206-21. [PMID: 33721180 DOI: 10.1007/s11914-020-00647-7]
- You J, Reilly GC, Zhen X, Yellowley CE, Chen Q, et al. Osteopontin gene regulation by oscillatory fluid flow via intracellular calcium mobilization and activation of mitogen-activated protein kinase in MC3T3-E1 osteoblasts. J Biol Chem 2001;276(16):13365-71. [PMID: 11278573 DOI: 10.1074/jbc.M009846200]
- Hung CT, Allen FD, Pollack SR, Brighton CT. Intracellular Ca2+ stores and extracellular Ca2+ are required in the real-time Ca2+ response of bone cells experiencing fluid flow. J Biomech 1996;29(11):1411-7. [PMID: 8894921 DOI: 10.1016/0021-9290(96)84536-2]
- Chen NX, Ryder KD, Pavalko FM, Turner CH, Burr DB, et al. Ca(2+) regulates fluid shear-induced cytoskeletal reorganization and gene expression in osteoblasts. Am J Physiol Cell Physiol 2000;278(5):C989-97. [PMID: 10794673 DOI: 10.1152/ajpcell.2000.278.5.C989]
- Xiao B. Mechanisms of mechanotransduction and physiological roles of PIEZO channels. Nat Rev Mol Cell Biol 2024;25(11):886-903. [PMID: 39251883 DOI: 10.1038/s41580-024-00773-5]
- Wu J, Lewis AH, Grandl J. Touch, tension, and transduction – the function and regulation of Piezo ion channels. Trends Biochem Sci 2017;42(1):57-71. [PMID: 27743844 DOI: 10.1016/j.tibs.2016.09.004]
- Ibsen S, Tong A, Schutt C, Esener S, Chalasani SH. Sonogenetics is a non-invasive approach to activating neurons in Caenorhabditis elegans. Nat Commun 2015;6:8264. [PMID: 26372413 DOI: 10.1038/ncomms9264]
- Qiu Z, Guo J, Kala S, Zhu J, Xian Q, et al. The mechanosensitive ion channel piezo1 significantly mediates in vitro ultrasonic stimulation of neurons. iScience 2019;21:448-57. [PMID: 31707258 DOI: 10.1016/j.isci.2019.10.037]
- Kanchanawong P, Calderwood DA. Organization, dynamics and mechanoregulation of integrin-mediated cell-ECM adhesions. Nat Rev Mol Cell Biol 2023;24(2):142-61. [PMID: 36168065 DOI: 10.1038/s41580-022-00531-5]
- Volarić D, Žauhar G, Chen J, Jerbić Radetić AT, Omrčen H, et al. The effect of low-intensity pulsed ultrasound on bone regeneration and the expression of osterix and cyclooxygenase-2 during critical-size bone defect repair. Int J Mol Sci 2024;25(7):3882. [PMID: 38612693 DOI: 10.3390/ijms25073882]
- Su WS, Wu CH, Chen SF, Yang FY. Transcranial ultrasound stimulation promotes brain-derived neurotrophic factor and reduces apoptosis in a mouse model of traumatic brain injury. Brain Stimul 2017;10(6):1032-41. [PMID: 28939348 DOI: 10.1016/j.brs.2017.09.003]
- Mitra SK, Schlaepfer DD. Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr Opin Cell Biol 2006;18(5):516-23. [PMID: 16919435 DOI: 10.1016/j.ceb.2006.08.011]
- Sato M, Nagata K, Kuroda S, Horiuchi S, Nakamura T, et al. Low-intensity pulsed ultrasound activates integrin-mediated mechanotransduction pathway in synovial cells. Ann Biomed Eng 2014;42(10):2156-63. [PMID: 25096496 DOI: 10.1007/s10439-014-1081-x]
- Lee HJ, Choi BH, Min BH, Park SR. Low-intensity ultrasound inhibits apoptosis and enhances viability of human mesenchymal stem cells in three-dimensional alginate culture during chondrogenic differentiation. Tissue Eng 2007;13(5):1049-57. [PMID: 17428192 DOI: 10.1089/ten.2006.0346]
- Zinda MJ, Johnson MA, Paul JD, Horn C, Konicek BW, et al. AKT-1, -2, and -3 are expressed in both normal and tumor tissues of the lung, breast, prostate, and colon. Clin Cancer Res 2001;7(8):2475-9. [PMID: 11489829]
- Datta SR, Dudek H, Tao X, Masters S, Fu H, et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 1997;91(2):231-41. [PMID: 9346240 DOI: 10.1016/s0092-8674(00)80405-5]
- Hashemi M, Mohandesi Khosroshahi E, Asadi S, Tanha M, Ghatei Mohseni F, et al. Emerging roles of non-coding RNAs in modulating the PI3K/Akt pathway in cancer. Non-coding RNA Res 2025;10:1-15. [PMID: 39296640 DOI: 10.1016/j.ncrna.2024.08.002]
- Johnson GL, Lapadat R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002;298(5600):1911-2. [PMID: 12471242 DOI: 10.1126/science.1072682]
- Wang Y, Jiang L, Xu T, Su Z, Guo X, et al. p38 MAPK signaling is a key mediator for low-intensity pulsed ultrasound (LIPUS) in cultured human omental adipose-derived mesenchymal stem cells. Am J Transl Res 2019;11(1):418-29. [PMID: 30787998]
- Gao Q, Walmsley AD, Cooper PR, Scheven BA. Ultrasound stimulation of different dental stem cell populations: role of mitogen-activated protein kinase signaling. J Endod 2016;42(3):425-31. [PMID: 26830427 DOI: 10.1016/j.joen.2015.12.019]
- Gao Q, Cooper PR, Walmsley AD, Scheven BA. Role of Piezo channels in ultrasound-stimulated dental stem cells. J Endod 2017;43(7):1130-6. [PMID: 28527849 DOI: 10.1016/j.joen.2017.02.022]
- Mills LA, Simpson AH. The relative incidence of fracture non-union in the Scottish population (5.17 million): a 5-year epidemiological study. BMJ Open 2013;3(2):e002276. [PMID: 23396560 DOI: 10.1136/bmjopen-2012-002276]
- Gross TS, Edwards JL, McLeod KJ, Rubin CT. Strain gradients correlate with sites of periosteal bone formation. J Bone Miner Res 1997;12(6):982-8. [PMID: 9169359 DOI: 10.1359/jbmr.1997.12.6.982]
- Harle J, Salih V, Knowles JC, Mayia F, Olsen I. Effects of therapeutic ultrasound on osteoblast gene expression. J Mater Sci Mater Med 2001;12(10-12):1001-4. [PMID: 15348355 DOI: 10.1023/a:1012825519114]
- Klug W, Franke WG, Knoch HG. Scintigraphic control of bone-fracture healing under ultrasonic stimulation: an animal experimental study. Eur J Nucl Med 1986;11(12):494-7. [PMID: 3089791 DOI: 10.1007/BF00252796]
- Chang WH, Sun JS, Chang SP, Lin JC. Study of thermal effects of ultrasound stimulation on fracture healing. Bioelectromagnetics 2002;23(4):256-63. [PMID: 11948604 DOI: 10.1002/bem.10009]
- Romano CL, Romano D, Logoluso N. Low-intensity pulsed ultrasound for the treatment of bone delayed union or nonunion: a review. Ultrasound Med Biol 2009;35(4):529-36. [PMID: 19097683 DOI: 10.1016/j.ultrasmedbio.2008.09.029]
- Zhang G, Li X, Wu L, Qin YX. Piezo1 channel activation in response to mechanobiological acoustic radiation force in osteoblastic cells. Bone Res 2021;9(1):16. [PMID: 33692342 DOI: 10.1038/s41413-020-00124-y]
- Young SR, Gerard-O’Riley R, Kim JB, Pavalko FM. Focal adhesion kinase is important for fluid shear stress-induced mechanotransduction in osteoblasts. J Bone Miner Res 2009;24(3):411-24. [PMID: 19016591 DOI: 10.1359/jbmr.081102]
- Tanaka E, Kuroda S, Horiuchi S, Tabata A, El-Bialy T. Low-intensity pulsed ultrasound in dentofacial tissue engineering. Ann Biomed Eng 2015;43(4):871-86. [PMID: 25672801 DOI: 10.1007/s10439-015-1274-y]
- Matsumoto K, Shimo T, Kurio N, Okui T, Ibaragi S, et al. Low-intensity pulsed ultrasound stimulation promotes osteoblast differentiation through hedgehog signaling. J Cell Biochem 2018;119(6):4352-60. [PMID: 28981158 DOI: 10.1002/jcb.26418]
- Zhou XY, Xu XM, Wu SY, Zhang ZC, Wang F, et al. Low-intensity pulsed ultrasound promotes spinal fusion and enhances migration and proliferation of MG63s through sonic hedgehog signaling pathway. Bone 2018;110:47-57. [PMID: 29414599 DOI: 10.1016/j.bone.2018.01.025]
- Tassinary JAF, Lunardelli A, Basso BS, Dias HB, Catarina AV, et al. Low-intensity pulsed ultrasound (LIPUS) stimulates mineralization of MC3T3-E1 cells through calcium and phosphate uptake. Ultrasonics 2018;84:290-5. [PMID: 29182945 DOI: 10.1016/j.ultras.2017.11.011]
- Tabuchi Y, Sugahara Y, Ikegame M, Suzuki N, Kitamura K, et al. Genes responsive to low-intensity pulsed ultrasound in MC3T3-E1 preosteoblast cells. Int J Mol Sci 2013;14(11):22721-40. [PMID: 24252911 DOI: 10.3390/ijms141122721]
- Veronick JA, Assanah F, Piscopo N, Kutes Y, Vyas V, et al. Mechanically loading cell/hydrogel constructs with low-intensity pulsed ultrasound for bone repair. Tissue Eng Part A 2018;24(3-4):254-63. [PMID: 28610471 DOI: 10.1089/ten.TEA.2016.0547]
- Puts R, Vico R, Beilfuss N, Shaka M, Padilla F, et al. Pulsed ultrasound for bone regeneration – outcomes and hurdles in the clinical application: a systematic review. Eur Cell Mater 2021;42:281-311. [PMID: 34647316 DOI: 10.22203/eCM.v042a20]
- Lubbert PH, van der Rijt RH, Hoorntje LE, van der Werken C. Low-intensity pulsed ultrasound (LIPUS) in fresh clavicle fractures: a multi-centre double blind randomised controlled trial. Injury 2008;39(12):1444-52. [PMID: 18656872 DOI: 10.1016/j.injury.2008.04.004]
- Prasad V, Ioannidis JP. Evidence-based de-implementation for contradicted, unproven, and aspiring healthcare practices. Implement Sci 2014;9:1. [PMID: 24398253 DOI: 10.1186/1748-5908-9-1]
- Warden SJ, Bennell KL, Matthews B, Brown DJ, McMeeken JM, et al. Efficacy of low-intensity pulsed ultrasound in the prevention of osteoporosis following spinal cord injury. Bone 2001;29(5):431-6. [PMID: 11704494 DOI: 10.1016/s8756-3282(01)00599-3]
- Watanabe Y, Arai Y, Takenaka N, Kobayashi M, Matsushita T. Three key factors affecting treatment results of low-intensity pulsed ultrasound for delayed unions and nonunions: instability, gap size, and atrophic nonunion. J Orthop Sci 2013;18(5):803-10. [PMID: 23775464 DOI: 10.1007/s00776-013-0415-0]
- Korstjens CM, Rutten S, Nolte PA, van Duin MA, Klein-Nulend J. Low-intensity pulsed ultrasound increases blood vessel size during fracture healing in patients with a delayed-union of the osteotomized fibula. Histol Histopathol 2018;33(7):737-46. [PMID: 29436706 DOI: 10.14670/HH-11-972]
- Sun S, Tang L, Zhao T, Kang Y, Sun L, et al. Longitudinal effects of low-intensity pulsed ultrasound on osteoporosis and osteoporotic bone defect in ovariectomized rats. Ultrasonics 2021;113:106360. [PMID: 33561635 DOI: 10.1016/j.ultras.2021.106360]
- Inoue S, Li C, Hatakeyama J, Jiang H, Kuroki H, et al. Higher-intensity ultrasound accelerates fracture healing via mechanosensitive ion channel Piezo1. Bone 2023;177:116916. [PMID: 37777037 DOI: 10.1016/j.bone.2023.116916]
- Jia W, Zhou Z, Zhan W. Musculoskeletal biomaterials: stimulated and synergized with low intensity pulsed ultrasound. J Funct Biomater 2023;14(10):504. [PMID: 37888169 DOI: 10.3390/jfb14100504]
- Li M, Hu X, Liu X, Zhao L, Zhao W, et al. 3D bioprinted piezoelectric hydrogel synergized with LIPUS to promote bone regeneration. Mater Today Bio 2025;31:101604. [DOI: 10.1016/j.mtbio.2025.101604]
- Chen K, Wang F, Sun X, Ge W, Zhang M, et al. 3D-printed zinc oxide nanoparticles modified barium titanate/hydroxyapatite ultrasound-responsive piezoelectric ceramic composite scaffold for treating infected bone defects. Bioact Mater 2025;45:479-95. [PMID: 39717367 DOI: 10.1016/j.bioactmat.2024.11.015]
- Vaddamanu SK, Alhamoudi FH, Vyas R, Gurumurthy V, Siurkel Y, et al. Attenuation of orthodontically induced inflammatory root resorption by using low-intensity pulsed ultrasound as a therapeutic modality – a systematic review. BMC Oral Health 2024;24(1):67. [PMID: 38200481 DOI: 10.1186/s12903-023-03741-1]
- El-Bialy T, El-Shamy I, Graber TM. Repair of orthodontically induced root resorption by ultrasound in humans. Am J Orthod Dentofacial Orthop 2004;126(2):186-93. [PMID: 15316473 DOI: 10.1016/j.ajodo.2004.02.010]
- Ang WT, Scurtescu C, Hoy W, El-Bialy T, Tsui YY, et al. Design and implementation of therapeutic ultrasound generating circuit for dental tissue formation and tooth-root healing. IEEE Trans Biomed Circuits Syst 2010;4(1):49-61. [PMID: 23853309 DOI: 10.1109/TBCAS.2009.2034635]
- Al-Hanbali LMS, Burhan AS, Hajeer MY, Nawaya FR. The effectiveness of low-level laser therapy and low-intensity pulsed ultrasound in reducing pain induced by orthodontic separation: a randomized controlled trial. BMC Oral Health 2024;24(1):166. [PMID: 38308275 DOI: 10.1186/s12903-024-03926-2]
- Badiee M, Tehranchi A, Behnia P, Khatibzadeh K. Efficacy of low-intensity pulsed ultrasound for orthodontic pain control: a randomized clinical trial. Front Dent 2021;18:38. [PMID: 35965719 DOI: 10.18502/fid.v18i38.7607]
- Ying S, Tan M, Feng G, Kuang Y, Chen D, et al. Erratum: Low-intensity pulsed ultrasound regulates alveolar bone homeostasis in experimental periodontitis by diminishing oxidative stress. Theranostics 2022;12(3):1337-40. [PMID: 32863960 DOI: 10.7150/thno.42508]
- Wang Y, Li J, Zhou J, Qiu Y, Song J. Low-intensity pulsed ultrasound enhances bone marrow-derived stem cells-based periodontal regenerative therapies. Ultrasonics 2022;121:106678. [PMID: 35051693 DOI: 10.1016/j.ultras.2021.106678]
- Zhang T, Chen Z, Zhu M, Jing X, Xu X, et al. Extracellular vesicles derived from human dental mesenchymal stem cells stimulated with low-intensity pulsed ultrasound alleviate inflammation-induced bone loss in a mouse model of periodontitis. Genes Dis 2023;10(4):1613-25. [PMID: 37397561 DOI: 10.1016/j.gendis.2022.06.009]
- Lee W, Georgas E, Komatsu DE, Qin YX. Daily low-intensity pulsed ultrasound stimulation mitigates joint degradation and pain in a post-traumatic osteoarthritis rat model. J Orthop Translat 2024;44:9-18. [PMID: 38161708 DOI: 10.1016/j.jot.2023.09.002]
- Tanaka E, Liu Y, Xia L, Ogasawara N, Sakamaki T, et al. Effectiveness of low-intensity pulsed ultrasound on osteoarthritis of the temporomandibular joint: a review. Ann Biomed Eng 2020;48(8):2158-70. [PMID: 32514932 DOI: 10.1007/s10439-020-02540-x]
- Kojima Y, Watanabe T. Low-intensity pulsed ultrasound irradiation attenuates collagen degradation of articular cartilage in early osteoarthritis-like model mice. J Exp Orthop 2023;10(1):106. [PMID: 37870591 DOI: 10.1186/s40634-023-00672-2]
- Ying ZM, Lin T, Yan SG. Low-intensity pulsed ultrasound therapy: a potential strategy to stimulate tendon-bone junction healing. J Zhejiang Univ Sci B 2012;13(12):955-63. [PMID: 23225850 DOI: 10.1631/jzus.B1200129]
- Lu H, Qin L, Cheung W, Lee K, Wong W, et al. Low-intensity pulsed ultrasound accelerated bone-tendon junction healing through regulation of vascular endothelial growth factor expression and cartilage formation. Ultrasound Med Biol 2008;34(8):1248-60. [PMID: 18378382 DOI: 10.1016/j.ultrasmedbio.2008.01.009]
- Xu Z, Li S, Wan L, Hu J, Lu H, et al. Role of low-intensity pulsed ultrasound in regulating macrophage polarization to accelerate tendon-bone interface repair. J Orthop Res 2023;41(5):919-29. [PMID: 36203341 DOI: 10.1002/jor.25454]
- Cui JH, Park SR, Park K, Choi BH, Min BH. Preconditioning of mesenchymal stem cells with low-intensity ultrasound for cartilage formation in vivo. Tissue Eng 2007;13(2):351-60. [PMID: 17518569 DOI: 10.1089/ten.2006.0080]
- Lee HJ, Choi BH, Min BH, Son YS, Park SR. Low-intensity ultrasound stimulation enhances chondrogenic differentiation in alginate culture of mesenchymal stem cells. Artif Organs 2006;30(9):707-15. [PMID: 16934100 DOI: 10.1111/j.1525-1594.2006.00288.x]
- Fu S, Lan Y, Wang G, Bao D, Qin B, et al. External stimulation: a potential therapeutic strategy for tendon-bone healing. Front Bioeng Biotechnol 2023;11:1150290. [PMID: 37064229 DOI: 10.3389/fbioe.2023.1150290]
- Warden SJ, Metcalf BR, Kiss ZS, Cook JL, Purdam CR, et al. Low-intensity pulsed ultrasound for chronic patellar tendinopathy: a randomized, double-blind, placebo-controlled trial. Rheumatology (Oxford) 2008;47(4):467-71. [PMID: 18270224 DOI: 10.1093/rheumatology/kem384]
- D’Vaz AP, Ostor AJ, Speed CA, Jenner JR, Bradley M, et al. Pulsed low-intensity ultrasound therapy for chronic lateral epicondylitis: a randomized controlled trial. Rheumatology (Oxford) 2006;45(5):566-70. [PMID: 16303817 DOI: 10.1093/rheumatology/kei210]
- Omi H, Mochida J, Iwashina T, Katsuno R, Hiyama A, et al. Low-intensity pulsed ultrasound stimulation enhances TIMP-1 in nucleus pulposus cells and MCP-1 in macrophages in the rat. J Orthop Res 2008;26(6):865-71. [PMID: 18240328 DOI: 10.1002/jor.20545]
- Jiang W, Wang Y, Tang J, Peng J, Wang Y, et al. Low-intensity pulsed ultrasound treatment improved the rate of autograft peripheral nerve regeneration in rat. Sci Rep 2016;6:22773. [PMID: 27102358 DOI: 10.1038/srep22773]
- Zhong Y, Huang C. BDNF/TrkB signaling pathway and WDR neurons: Core factors inducing central sensitization of neuropathic pain. Life Sci 2025;377:123796. [PMID: 40480621 DOI: 10.1016/j.lfs.2025.123796]
- Fontana F, Iacoponi F, Orlando F, Pratellesi T, Cafarelli A, et al. Low-intensity pulsed ultrasound increases neurotrophic factors secretion and suppresses inflammation in in vitro models of peripheral neuropathies. J Neural Eng 2023;20(2):026033. [PMID: 36930982 DOI: 10.1088/1741-2552/acc54e]
- Yang FY, Lu WW, Lin WT, Chang CW, Huang SL. Enhancement of neurotrophic factors in astrocyte for neuroprotective effects in brain disorders using low-intensity pulsed ultrasound stimulation. Brain Stimul 2015;8(3):465-73. [PMID: 25558041 DOI: 10.1016/j.brs.2014.11.017]
- Ning GZ, Song WY, Xu H, Zhu RS, Wu QL, et al. Bone marrow mesenchymal stem cells stimulated with low-intensity pulsed ultrasound: better choice of transplantation treatment for spinal cord injury: treatment for SCI by LIPUS-BMSCs transplantation. CNS Neurosci Ther 2019;25(4):496-508. [PMID: 30294904 DOI: 10.1111/cns.13071]
- Zhang H, Lin X, Wan H, Li JH, Li JM. Effect of low-intensity pulsed ultrasound on the expression of neurotrophin-3 and brain-derived neurotrophic factor in cultured Schwann cells. Microsurgery 2009;29(6):479-85. [PMID: 19308950 DOI: 10.1002/micr.20644]
- Ren C, Chen X, Du N, Geng S, Hu Y, et al. Low-intensity pulsed ultrasound promotes Schwann cell viability and proliferation via the GSK-3β/β-catenin signaling pathway. Int J Biol Sci 2018;14(5):497-507. [PMID: 29805301 DOI: 10.7150/ijbs.22409]
- Yang DP, Zhang DP, Mak KS, Bonder DE, Pomeroy SL, et al. Schwann cell proliferation during Wallerian degeneration is not necessary for regeneration and remyelination of the peripheral nerves: axon-dependent removal of newly generated Schwann cells by apoptosis. Mol Cell Neurosci 2008;38(1):80-8. [PMID: 18374600 DOI: 10.1016/j.mcn.2008.01.017]
- Ye K, Li Z, Yin Y, Zhou J, Li D, et al. LIPUS-SCs-Exo promotes peripheral nerve regeneration in cavernous nerve crush injury-induced ED rats via PI3K/Akt/FoxO signaling pathway. CNS Neurosci Ther 2023;29(11):3239-58. [PMID: 37157936 DOI: 10.1111/cns.14256]
- Yue Y, Yang X, Zhang L, Xiao X, Nabar NR, et al. Low-intensity pulsed ultrasound upregulates pro-myelination indicators of Schwann cells enhanced by co-culture with adipose-derived stem cells. Cell Prolif 2016;49(6):720-8. [PMID: 27625295 DOI: 10.1111/cpr.12298]
- Lowdon IM, Seaber AV, Urbaniak JR. An improved method of recording rat tracks for measurement of the sciatic functional index of de Medinaceli. J Neurosci Methods 1988;24(3):279-81. [PMID: 3419210 DOI: 10.1016/0165-0270(88)90173-2]
- Peng DY, Reed-Maldonado AB, Lin GT, Xia SJ, Lue TF. Low-intensity pulsed ultrasound for regenerating peripheral nerves: potential for penile nerve. Asian J Androl 2020;22(4):335-41. [PMID: 31535626 DOI: 10.4103/aja.aja_95_19]
- Huang B, Jiang Y, Zhang L, Yang B, Guo Y, et al. Low-intensity pulsed ultrasound promotes proliferation and myelinating genes expression of Schwann cells through NRG1/ErbB signaling pathway. Tissue Cell 2023;80:101985. [PMID: 36459840 DOI: 10.1016/j.tice.2022.101985]
- Zhou H, Niu L, Xia X, Lin Z, Liu X, et al. Wearable ultrasound improves motor function in an MPTP mouse model of Parkinson’s disease. IEEE Trans Biomed Eng 2019;66(11):3006-13. [PMID: 30794160 DOI: 10.1109/TBME.2019.2899631]
- Sung CY, Chiang PK, Tsai CW, Yang FY. Low-intensity pulsed ultrasound enhances neurotrophic factors and alleviates neuroinflammation in a rat model of Parkinson’s disease. Cereb Cortex 2021;32(1):176-85. [PMID: 34196669 DOI: 10.1093/cercor/bhab201]
- Song WS, Sung CY, Ke CH, Yang FY. Anti-inflammatory and neuroprotective effects of transcranial ultrasound stimulation on Parkinson’s disease. Ultrasound Med Biol 2022;48(2):265-74. [PMID: 34740497 DOI: 10.1016/j.ultrasmedbio.2021.10.001]
- Chen Y, Li Y, Du M, Yu J, Gao F, et al. Ultrasound neuromodulation: integrating medicine and engineering for neurological disease treatment. BIO Integr 2021;2:169-79. [DOI: 10.15212/bioi-2020-0026]
- Shimokawa H, Shindo T, Ishiki A, Tomita N, Ichijyo S, et al. A pilot study of whole-brain low-intensity pulsed ultrasound therapy for early stage of Alzheimer’s disease (LIPUS-AD): a randomized, double-blind, placebo-controlled trial. Tohoku J Exp Med 2022;258(3):167-75. [PMID: 36104179 DOI: 10.1620/tjem.2022.J078]
- Shindo T, Eguchi K, Monma Y, Kanai H, Yasuda S, et al. Optimal treatment conditions for low-intensity pulsed ultrasound therapy for Alzheimer’s disease: applications from mice to humans. J Med Ultrason (2001) 2024;51(3):419-27. [PMID: 38698287 DOI: 10.1007/s10396-024-01461-9]
- He X, Peng Y, Huang S, Xiao Z, Li G, et al. Blood brain barrier-crossing delivery of felodipine nanodrug ameliorates anxiety-like behavior and cognitive impairment in Alzheimer’s disease. Adv Sci (Weinh) 2024;11(34):e2401731. [PMID: 38981028 DOI: 10.1002/advs.202401731]
- Luo Q, Ye X, Xu J, Sun Z, Wang P, et al. Low-intensity pulsed ultrasound enhances delivery of 30 nm Q10 for improving mental and memory disorder in APP/PS1 mice. Drug Deliv Transl Res 2025. [PMID: 39994108 DOI: 10.1007/s13346-025-01814-y]
- Meng XM, Nikolic-Paterson DJ, Lan HY. Inflammatory processes in renal fibrosis. Nat Rev Nephrol 2014;10(9):493-503. [PMID: 24981817 DOI: 10.1038/nrneph.2014.114]
- Xu M, Wang L, Wu S, Dong Y, Chen X, et al. Review on experimental study and clinical application of low-intensity pulsed ultrasound in inflammation. Quant Imaging Med Surg 2021;11(1):443-62. [PMID: 33392043 DOI: 10.21037/qims-20-680]
- Aibara Y, Nakashima A, Kawano KI, Yusoff FM, Mizuki F, et al. Daily low-intensity pulsed ultrasound ameliorates renal fibrosis and inflammation in experimental hypertensive and diabetic nephropathy. Hypertension 2020;76(6):1906-14. [PMID: 33131306 DOI: 10.1161/HYPERTENSIONAHA.120.15237]
- Lin CY, Wang CC, Loh JZ, Chiang TC, Weng TI, et al. Therapeutic ultrasound halts progression of chronic kidney disease in vivo via the regulation of markers associated with renal epithelial-mesenchymal transition and senescence. Int J Mol Sci 2022;23(21):13387. [PMID: 36362179 DOI: 10.3390/ijms232113387]
- Ouyang ZQ, Shao LS, Wang WP, Ke TF, Chen D, et al. Low intensity pulsed ultrasound ameliorates Adriamycin-induced chronic renal injury by inhibiting ferroptosis. Redox Rep 2023;28(1):2251237. [PMID: 37652897 DOI: 10.1080/13510002.2023.2251237]
- Ouyang Z, Zhang G, Wang W, Shao L, Du X, et al. Transcriptome profile analysis revealed the potential mechanism of LIPUS treatment for Adriamycin-induced chronic kidney disease rat. Heliyon 2023;9(11):e21531. [PMID: 38027717 DOI: 10.1016/j.heliyon.2023.e21531]
- Lin CY, Weng TI, Loh JZ, Chan DC, Chiang CK, et al. Therapeutic ultrasound treatment for the prevention of chronic kidney disease-associated muscle wasting in mice. Artif Cells Nanomed Biotechnol 2023;51(1):268-75. [PMID: 37195194 DOI: 10.1080/21691401.2023.2213735]
- Chiang CK, Loh JZ, Yang TH, Huang KT, Wu CT, et al. Prevention of acute kidney injury by low intensity pulsed ultrasound via anti-inflammation and anti-apoptosis. Sci Rep 2020;10(1):14317. [DOI: 10.1038/s41598-020-71330-1]
- Gouda SAA, Aboulhoda BE, Abdelwahed OM, Abdallah H, Rashed L, et al. Low-intensity pulsed ultrasound (LIPUS) switched macrophage into M2 phenotype and mitigated necroptosis and increased HSP 70 in gentamicin-induced nephrotoxicity. Life Sci 2023;314:121338. [PMID: 36592788 DOI: 10.1016/j.lfs.2022.121338]
- Campbell JD, Burnett AL. Neuroprotective and nerve regenerative approaches for treatment of erectile dysfunction after cavernous nerve injury. Int J Mol Sci 2017;18(8):1794. [PMID: 28820434 DOI: 10.3390/ijms18081794]
- Chung E, Wang J. A state-of-art review of low intensity extracorporeal shock wave therapy and lithotripter machines for the treatment of erectile dysfunction. Expert Rev Med Devices 2017;14(12):929-34. [PMID: 29119841 DOI: 10.1080/17434440.2017.1403897]
- Lei H, Xin H, Guan R, Xu Y, Li H, et al. Low-intensity pulsed ultrasound improves erectile function in streptozotocin-induced type i diabetic rats. Urology 2015;86(6):1241.e11-8. [PMID: 26383610 DOI: 10.1016/j.urology.2015.07.026]
- Chen Y, Xiao M, Zhao L, Huang Y, Lin Y, et al. Low-intensity pulsed ultrasound counteracts advanced glycation end products-induced corpus cavernosal endothelial cell dysfunction via activating mitophagy. Int J Mol Sci 2022;23(23):14887. [PMID: 36499213 DOI: 10.3390/ijms232314887]
- Liu S, Jiang C, Hu J, Chen H, Han B, et al. Low-intensity pulsed ultrasound enhanced adipose-derived stem cell-mediated angiogenesis in the treatment of diabetic erectile dysfunction through the Piezo-ERK-VEGF axis. Stem Cells Int 2022;2022:6202842. [PMID: 35935181 DOI: 10.1155/2022/6202842]
- Cui W, Li H, Guan R, Li M, Yang B, et al. Efficacy and safety of novel low-intensity pulsed ultrasound (LIPUS) in treating mild to moderate erectile dysfunction: a multicenter, randomized, double-blind, sham-controlled clinical study. Transl Androl Urol 2019;8(4):307-19. [PMID: 31555554 DOI: 10.21037/tau.2019.07.03]
- Chen H, Li Z, Li X, Yang Y, Dai Y, et al. The efficacy and safety of thrice vs twice per week low-intensity pulsed ultrasound therapy for erectile dysfunction: a randomized clinical trial. J Sex Med 2022;19(10):1536-45. [PMID: 35999130 DOI: 10.1016/j.jsxm.2022.06.009]
- Gao QQ, Wang J, Li DS, Dai YT, Li ZR, et al. Efficacy and safety of low-intensity pulsed ultrasound (LIPUS) combined with tadalafil in the treatment of severe erectile dysfunction: a retrospective cohort study. Transl Androl Urol 2024;13(9):2045-54. [PMID: 39434752 DOI: 10.21037/tau-24-154]
- Chiang PK, Yang FY. A potential treatment of low intensity pulsed ultrasound on cavernous nerve injury for erectile dysfunction. Med Hypotheses 2019;122:19-21. [PMID: 30593410 DOI: 10.1016/j.mehy.2018.10.014]
- Liu Y, Pan XY, Zhang XX, Sun JL, Mao YH, et al. Role of mechanotransduction mediated by YAP/TAZ in the treatment of neurogenic erectile dysfunction with low-intensity pulsed ultrasound. Andrology 2023;11(7):1514-27. [PMID: 37042189 DOI: 10.1111/andr.13438]
- Abtahi NS, Eimani H, Vosough A, Shahverdi A, Fathi R, et al. Effect of therapeutic ultrasound on folliculogenesis, angiogenesis and apoptosis after heterotopic mouse ovarian transplantation. Ultrasound Med Biol 2014;40(7):1535-44. [PMID: 24785439 DOI: 10.1016/j.ultrasmedbio.2014.02.006]
- Xu H, Xia Y, Qin J, Xu J, Li C, et al. Effects of low intensity pulsed ultrasound on expression of B-cell lymphoma-2 and BCL2-associated X in premature ovarian failure mice induced by 4-vinylcyclohexene diepoxide. Reprod Biol Endocrinol 2021;19(1):113. [PMID: 34284777 DOI: 10.1186/s12958-021-00799-w]
- Tang H, Liu Y, Fan Y, Li C. Therapeutic effects of low-intensity pulsed ultrasound on premature ovarian insufficiency. Ultrasound Med Biol 2021;47(8):2377-87. [PMID: 34088530 DOI: 10.1016/j.ultrasmedbio.2021.04.022]
- Nurkovic J, Dolicanin Z, Mustafic F, Mujanovic R, Memic M, et al. Mesenchymal stem cells in regenerative rehabilitation. J Phys Ther Sci 2016;28(6):1943-8. [PMID: 27390452 DOI: 10.1589/jpts.28.1943]
- Lou S, Duan Y, Nie H, Cui X, Du J, et al. Mesenchymal stem cells: biological characteristics and application in disease therapy. Biochimie 2021;185:9-21. [PMID: 33711361 DOI: 10.1016/j.biochi.2021.03.003]
- Hosseini S, Taghiyar L, Safari F, Baghaban Eslaminejad M. Regenerative medicine applications of mesenchymal stem cells. Adv Exp Med Biol 2018;1089:115-41. [PMID: 29767289 DOI: 10.1007/5584_2018_213]
- Zhou Y, Zhu F, Zhou Y, Li X, Zhao S, et al. The value of low-intensity pulsed ultrasound in reducing ovarian injury caused by chemotherapy in mice. Reprod Biol Endocrinol 2024;22(1):51. [PMID: 38671458 DOI: 10.1186/s12958-024-01216-8]
- Ling L, Hou J, Wang Y, Shu H, Huang Y. Effects of low-intensity pulsed ultrasound on the migration and homing of human amnion-derived mesenchymal stem cells to ovaries in rats with premature ovarian insufficiency. Cell Transplant 2022;31:9636897221129171. [PMID: 36282038 DOI: 10.1177/09636897221129171]
- Zhou X, Sun S, Chen Y, Liu C, Li D, et al. Pulsed frequency modulated ultrasound promotes therapeutic effects of osteoporosis induced by ovarian failure in mice. Ultrasonics 2023;132:106973. [PMID: 36893552 DOI: 10.1016/j.ultras.2023.106973]
- Li F, Gallego J, Tirko NN, Greaser J, Bashe D, et al. Low-intensity pulsed ultrasound stimulation (LIPUS) modulates microglial activation following intracortical microelectrode implantation. Nat Commun 2024;15(1):5512. [PMID: 38951525 DOI: 10.1038/s41467-024-49709-9]
- Mesiwala AH, Farrell L, Wenzel HJ, Silbergeld DL, Crum LA, et al. High-intensity focused ultrasound selectively disrupts the blood-brain barrier in vivo. Ultrasound Med Biol 2002;28(3):389-400. [PMID: 11978420 DOI: 10.1016/s0301-5629(01)00521-x]
- Oh SJ, Lee JM, Kim HB, Lee J, Han S, et al. Ultrasonic neuromodulation via astrocytic TRPA1. Curr Biol 2019;29(20):3386-401.e8. [PMID: 31588000 DOI: 10.1016/j.cub.2019.08.021]
- Robertson JL, Cox BT, Jaros J, Treeby BE. Accurate simulation of transcranial ultrasound propagation for ultrasonic neuromodulation and stimulation. J Acoust Soc Am 2017;141(3):1726. [PMID: 28372121 DOI: 10.1121/1.4976339]
- Yuan Y, Wang Z, Liu M, Shoham S. Cortical hemodynamic responses induced by low-intensity transcranial ultrasound stimulation of mouse cortex. Neuroimage 2020;211:116597. [PMID: 32018004 DOI: 10.1016/j.neuroimage.2020.116597]
- Blackmore DG, Razansky D, Götz J. Ultrasound as a versatile tool for short- and long-term improvement and monitoring of brain function. Neuron 2023;111(8):1174-90. [PMID: 36917978 DOI: 10.1016/j.neuron.2023.02.018]
- Chen Y, Yang H, Wang Z, Zhu R, Cheng L, et al. Low-intensity pulsed ultrasound promotes mesenchymal stem cell transplantation-based articular cartilage regeneration via inhibiting the TNF signaling pathway. Stem Cell Res Ther 2023;14(1):93. [PMID: 37069673 DOI: 10.1186/s13287-023-03296-6]
- Yan S, Wang D, Zhang L, Gan T, Yao H, et al. LIPUS-S/B@NPs regulates the release of SDF-1 and BMP-2 to promote stem cell recruitment-osteogenesis for periodontal bone regeneration. Front Bioeng Biotechnol 2023;11:1226426. [PMID: 37469445 DOI: 10.3389/fbioe.2023.1226426]
- Rabut C, Yoo S, Hurt RC, Jin Z, Li H, et al. Ultrasound technologies for imaging and modulating neural activity. Neuron 2020;108(1):93-110. [PMID: 33058769 DOI: 10.1016/j.neuron.2020.09.003]
- Monteduro AG, Rizzato S, Caragnano G, Trapani A, Giannelli G, et al. Organs-on-chips technologies – a guide from disease models to opportunities for drug development. Biosen Bioelectron 2023;231:115271. [PMID: 37060819 DOI: 10.1016/j.bios.2023.115271]
- Heckman JD, Ryaby JP, McCabe J, Frey JJ, Kilcoyne RF. Acceleration of tibial fracture-healing by non-invasive, low-intensity pulsed ultrasound. J Bone Joint Surg Am 1994;76(1):26-34. [PMID: 8288661 DOI: 10.2106/00004623-199401000-00004]
- Kristiansen TK, Ryaby JP, McCabe J, Frey JJ, Roe LR. Accelerated healing of distal radial fractures with the use of specific, low-intensity ultrasound. A multicenter, prospective, randomized, double-blind, placebo-controlled study. J Bone Joint Surg Am 1997;79(7):961-73. [PMID: 9234872 DOI: 10.2106/00004623-199707000-00002]
- Rutten S, Nolte PA, Korstjens CM, van Duin MA, Klein-Nulend J. Low-intensity pulsed ultrasound increases bone volume, osteoid thickness and mineral apposition rate in the area of fracture healing in patients with a delayed union of the osteotomized fibula. Bone 2008;43(2):348-54. [PMID: 18538648 DOI: 10.1016/j.bone.2008.04.010]
- Zacherl M, Gruber G, Radl R, Rehak PH, Windhager R. No midterm benefit from low intensity pulsed ultrasound after chevron osteotomy for hallux valgus. Ultrasound Med Biol 2009;35(8):1290-7. [PMID: 19540659 DOI: 10.1016/j.ultrasmedbio.2009.03.008]
- Schofer MD, Block JE, Aigner J, Schmelz A. Improved healing response in delayed unions of the tibia with low-intensity pulsed ultrasound: results of a randomized sham-controlled trial. BMC Musculoskelet Disord 2010;11:229. [PMID: 20932272 DOI: 10.1186/1471-2474-11-229]
- Salem KH, Schmelz A. Low-intensity pulsed ultrasound shortens the treatment time in tibial distraction osteogenesis. Int Orthop 2014;38(7):1477-82. [PMID: 24390009 DOI: 10.1007/s00264-013-2254-1]
- Simpson AH, Keenan G, Nayagam S, Atkins RM, Marsh D, et al. Low-intensity pulsed ultrasound does not influence bone healing by distraction osteogenesis: a multicentre double-blind randomised control trial. Bone Joint J 2017;99-b(4):494-502. [PMID: 28385939 DOI: 10.1302/0301-620X.99B4.BJJ-2016-0559.R1]
- Tarride JE, Hopkins RB, Blackhouse G, Burke N, Bhandari M, et al. Low-intensity pulsed ultrasound for treatment of tibial fractures: an economic evaluation of the TRUST study. Bone Joint J 2017;99-b(11):1526-32. [PMID: 29092994 DOI: 10.1302/0301-620X.99B11.BJJ-2017-0737]
- Arimoto S, Hasegawa T, Takeda D, Tateishi C, Akashi M, et al. Effect of low-intensity pulsed ultrasound after intraoral vertical ramus osteotomy. Oral Surg Oral Med Oral Pathol Oral Radiol 2019;128(6):581-9. [PMID: 31227453 DOI: 10.1016/j.oooo.2019.03.009]
- Wu T, Zheng F, Tang HY, Li HZ, Cui XY, et al. Low-intensity pulsed ultrasound reduces alveolar bone resorption during orthodontic treatment via Lamin A/C-yes-associated protein axis in stem cells. World J Stem Cells 2024;16(3):267-86. [PMID: 38577236 DOI: 10.4252/wjsc.v16.i3.267]