Low-intensity Pulsed Ultrasound Enhances NK Cell Adoptive Therapy by Modulating the Wnt/β-catenin Signaling Pathway
1Key Laboratory of Medical Imaging Precision Theranostics and Radiation Protection, The Affiliated Changsha Central Hospital, University of South China, College of Hunan Province, Changsha, Hunan, China
2Institute of Medical Imaging, Hengyang Medical School, University of South China, Hengyang, Hunan, China
3The Seventh Affiliated Hospital, Hunan Veterans Administration Hospital, Hengyang Medical School, University of South China, Changsha, Hunan, China
4Department of Biliary-Pancreatic Surgery, Renji Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China
5Department of Biology, Science Faculty, Selcuk University, Campus, Konya, Turkey
*Correspondence to: Gökhan Zengin, Department of Biology, Science Faculty, Selcuk University, Campus, Konya, Turkey. E-mail: gokhanzengin@selcuk.edu.tr; Meng Du, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, Hunan, 410004, China. E-mail: dumeng_work@126.com
Received: February 15 2025; Revised: April 30 2025; Accepted: May 17 2025; Published Online: July 15 2025
Cite this paper:
Feng R, Gu A, Zengin G et al. Low-intensity Pulsed Ultrasound Enhances NK Cell Adoptive Therapy by Modulating the Wnt/β-catenin Signaling Pathway. BIO Integration 2025; 6: 1–8.
DOI: 10.15212/bioi-2025-0036. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Natural killer cell adoptive therapy is a novel immunotherapy strategy for the treatment of multiple refractory tumors, including ovarian cancer. However, natural killer cells’ anti-tumor effectiveness is limited by their viability and cytotoxicity. Our preliminary study suggested that ultrasound irradiation improves the therapeutic effects of natural killer cells on ovarian cancer through an unclear mechanism. The Wnt/β-catenin signaling pathway is a complex protein network system associated with the maturation and function of natural killer cells. Therefore, herein, to reveal the precise mechanism underlying the effects of ultrasound on natural killer cells, we measured the expression of Wnt/β-catenin signaling pathway proteins before and after ultrasound irradiation. The expression of therapeutic factors secreted by natural killer cells increased after ultrasound irradiation. Therefore, this study provides an additional strategy for optimizing the therapeutic efficiency of natural killer cells.
Keywords
Adoptive cell therapy, natural killer cells, ultrasound, Wnt/β-catenin signaling pathway.
Introduction
Ovarian cancer is a gynecological cancer with a high mortality rate [1]. Immunotherapy based on the immune recognition and immunological modulation mechanisms of cancer cells has garnered substantial attention because of its optimal therapeutic efficacy. Natural killer (NK) cells are intrinsic immune cells critical in killing malignant pathogens and solid tumor cells without major histocompatibility complex class I restriction and prior sensitization [2, 3]. Use of immunotherapy based on NK cells has increased and has become a major research direction in immunotherapy. Activated NK cells secrete various cytokines and enhance anti-tumor effects through adaptive immune responses [4]. The concentration of secreted cytokines is key in NK cells’ killing of tumor cells via antibody-dependent cell-mediated cytotoxicity or inflammatory action [5]. In recent years, remarkable progress has been made in adoptive therapy based on NK cells. However, challenges in anti-tumor immunotherapy persist; for example, serial immunosuppressive factors in the tumor microenvironment inhibit the anti-tumor efficacy of NK cells, and consequently limit their anti-tumor efficacy in vitro and in vivo [6–8]. The number and killing activity of NK cells are significantly associated with their therapeutic effects on cancer treatment [9]. Consequently, increasing NK cell numbers and cytotoxicity has emerged as a major strategy for promoting NK cell-based immunotherapy.
Mechanical stimulation, such as electric, magnetic, or laser stimulation [10, 11], triggers cellular differentiation or alters cellular metabolic function [12]. However, low-energy laser irradiation promotes the proliferation of both normal cells and tumor cells [13]. Among various physical stimulations, low-intensity pulsed ultrasound (LIPUS) has shown promising application potential in cell proliferation, migration, and differentiation, because it is non-invasive, cost-effective, and easily performed [14, 15]. LIPUS is a form of ultrasound with a much lower energy intensity (<3 W/cm2) than traditional therapeutic ultrasound [16]. However, the influence of LIPUS on the tumor-killing function of NK cells remains unknown. Two studies have shown that after treatment with high-intensity focused ultrasound, the number of NK cells, as well as the expression of granzyme B and perforin, increases in treated areas [17, 18]. Another study has indicated that the expression of perforin and granzyme B in NK cells significantly increases after activation of the Wnt/β-catenin signaling pathway, through inhibition of glycogen synthase kinase-3β (GSK3β) [19]. In addition, Tang et al. have shown that LIPUS activates the Wnt/β-catenin signaling pathway in osteoblasts [20]. Moreover, both LIPUS alone and the activated Wnt/β-catenin signaling pathway have been found to inhibit the NF-kB pathway [21, 22]. On this basis, we hypothesized that LIPUS might enhance the cytotoxicity of NK cells by activating the Wnt/β-catenin signaling pathway.
A series of clinical studies have been conducted on NK cell therapy (including NCT05528341, NCT05776355, and NCT05213195). This study was aimed at providing a safe, non-invasive, economical, and simple adjuvant enhancement strategy for NK cell relay therapy that can easily be translated and applied clinically. We preliminarily explored the mechanism underlying the effects of LIPUS on NK cell cytotoxicity and the Wnt/β-catenin signaling pathway. In a co-incubation system comprising NK cells and SK-OV-3 cells, we detected secreted amounts of granzyme A, granzyme B, and human perforin, as well as the expression levels of Wnt/β-catenin signaling pathway-associated proteins. Finally, to test the effect of LIPUS on the anti-tumor activity of NK cells, we examined various treatments in tumor-bearing mice, and assessed the anti-tumor activity of NK cells on the basis of changes in tumor volume and perforin levels (Figure 1).
Figure 1 Schematic diagram of low-intensity pulsed ultrasound-enhanced NK cell anti-tumor therapy. Low-intensity pulsed ultrasound irradiation of NK cells activates the Wnt/β-catenin signaling pathway and subsequently promotes the expression and secretion of the cytokines granzyme A, granzyme B, and perforin by NK cells. These factors enhance the anti-tumor ability of NK cells, thereby enabling effective killing of SK-OV-3 tumor cells and inhibiting the growth of ovarian cancer.
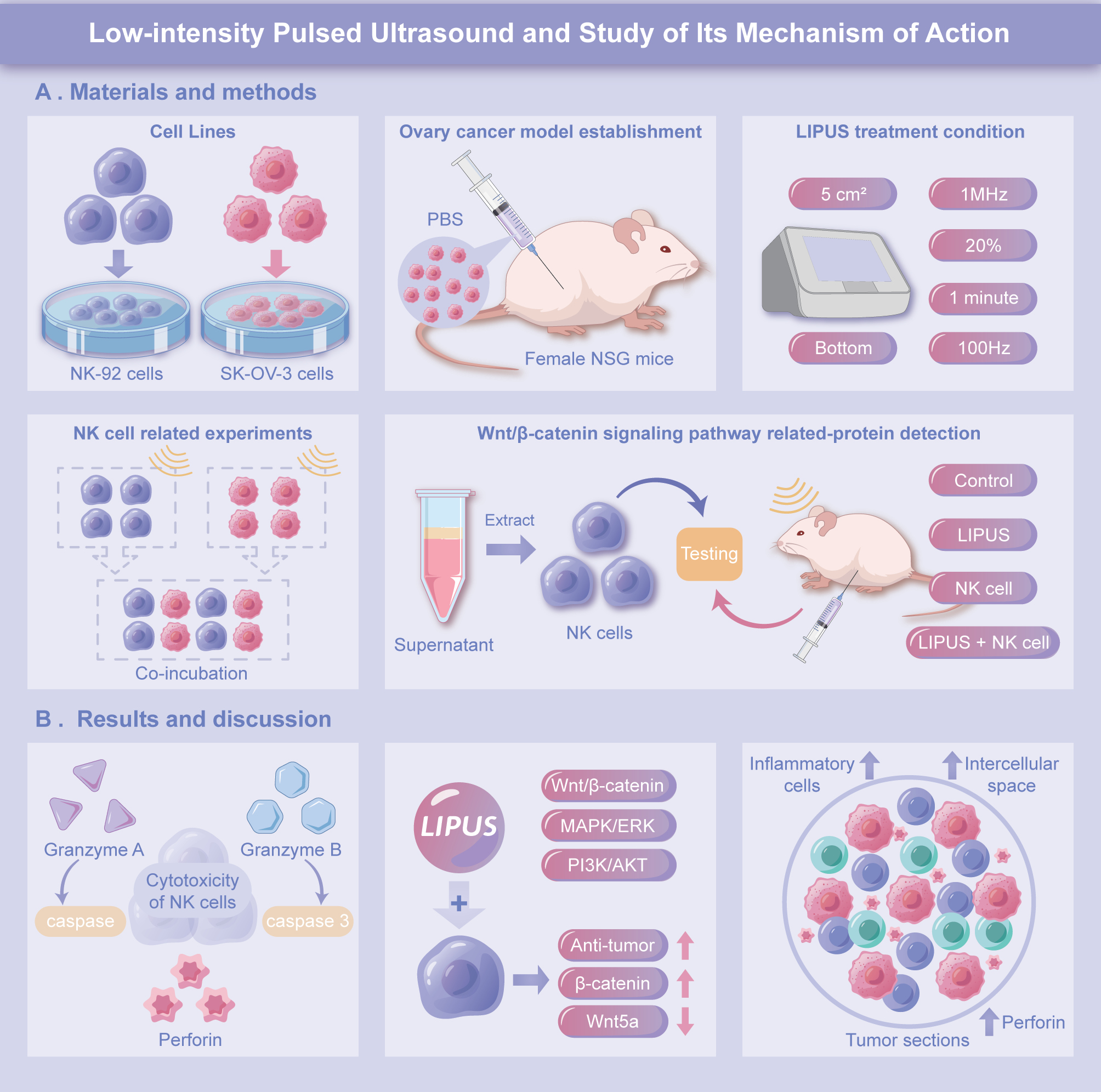
Materials and methods
Cell lines
SK-OV-3 human ovarian cancer cells and NK-92 cells were obtained from ATCC. The SK-OV-3 cells were cultured in RPMI-1640 medium with 2 mM L-glutamine, 10% FBS, and 1% penicillin/streptomycin, whereas the NK-92 cells were cultured in NK-92 medium containing MEMα, inositol, folic acid, 0.1 mM β-mercaptoethanol, 12.5% horse serum, 12.5% FBS, and 1% penicillin/streptomycin (Thermo Fisher Scientific, Co., America).
Ovarian cancer model establishment
All animal experiments complied with the ARRIVE guidelines and were performed in accordance with the U.K. Animals (Scientific Procedures) Act of 1986 and associated guidelines in EU Directive 2010/63/EU for animal experiments. All animal experiments were approved by the University of South China Animal Experiment Ethics Review. NOD-SCID IL2rγ null (NSG) female mice were obtained from the Guangdong Medical Laboratory Animal Center. To establish a tumor-bearing mouse model, we resuspended 6 × 106 SK-OV-3 cells in 0.2 mL PBS and subcutaneously injected them into the right anterolateral thigh skin in mice. After the tumor volumes reached 50–100 mm3, tumor-bearing mice were used in experiments.
LIPUS treatment condition
We examined the cells and animals with a Sonovitro (Shengxiang Hi-Tech, Co., Ltd., China) ultrasonic transfection apparatus. The following ultrasound equipment parameters were applied in all experiments: focused ultrasonic probe located at the bottom of the orifice plate; ultrasonic head area of 5 cm2; acoustic frequency of 1 MHz; duty cycle of 20%; pulsed repetition frequency of 100 Hz; and irradiation time of 1 min. All controls were subjected to the same conditions, except that the LIPUS apparatus was turned off.
NK cell activity, and granzyme A, granzyme B, and human perforin release assays
NK-92 cells were seeded into 96-well plates (1 × 104 cells per well) and incubated overnight. Subsequently, the cells were irradiated with LIPUS at an acoustic power of 0.2 W/cm2, 0.4 W/cm2, or 0.6 W/cm2. Three replicate groups were analyzed for each group. After 6 and 12 hours of incubation following irradiation, the cells were collected. A CCK-8 kit (Beyotime Biotechnology Co., Shanghai, China) was used to detect NK cell activity. SK-OV-3 human ovarian cancer cells were seeded into 96-well plates and incubated overnight in 100 μL RPMI-1640 medium with 2 mM L-glutamine, 10% FBS, and 1% penicillin/streptomycin. First, the NK cells were irradiated with LIPUS at an acoustic power of 0.2 W/cm2, 0.4 W/cm2, or 0.6 W/cm2. Three replicate groups were analyzed for each group. After LIPUS irradiation, the NK cells were co-incubated with SK-OV-3 cells in a 2:1 ratio in 96-well plates. After 6 or 12 hours of incubation, supernatants were collected for granzyme A, granzyme B, and human perforin detection with ELISA kits (DAKEWE Co., China), according to the manufacturer’s protocol.
Detection of Wnt/β-catenin signaling pathway proteins
The LIPUS irradiation procedure and the co-incubation of NK cells and SK-OV-3 cells were as previously described. Supernatants collected from the co-culture system were centrifuged (at 178 g) for extraction of the NK cells. Total cellular proteins were extracted with RIPA lysis reagent (Cell Signaling Technology, Co., America) and used for detection of Wnt5a and β-catenin expression via western blotting. Quantitative expression levels were analyzed in BIO-RAD Image Lab Software.
Anti-tumor activity of NK cells combined with LIPUS against SK-OV-3 ovarian xenografts
Ovarian cancer xenografts were established with SK-OV-3 cells. After the tumor volume reached 100 cm3, 20 tumor-bearing mice were randomly divided into four groups (control, LIPUS irradiation alone, NK cell treatment alone, and NK cells combined with LIPUS irradiation treatment), with similar tumor volumes in each group. For NK cell treatment, 1.0 × 107 NK cells per mouse were administered intraperitoneally on days 0, 3, 6, and 9. LIPUS irradiation was performed on days 1, 4, 7, and 10, with an acoustic power of 0.6 W/cm2. Tumor volume was measured every 3 days with the following formula: volume = ((tumor length) × (tumor width)2)/2. Twelve days after treatment, or after the tumor volume exceeded 2,000 mm3, tumor-bearing mice were euthanized through anesthesia, and tumor tissues and sera were collected and used for perforin detection with ELISA. A portion of tumor tissue was subjected to H&E staining.
Statistical analysis
One-way ANOVA was used to analyze all quantitative results in this study in GraphPad Prism 8 (GraphPad Software Inc.). Expression levels of Wnt5a and β-catenin were quantified in grayscale in ImageJ software. Quantitative data are presented as mean ± SD.
Results and discussion
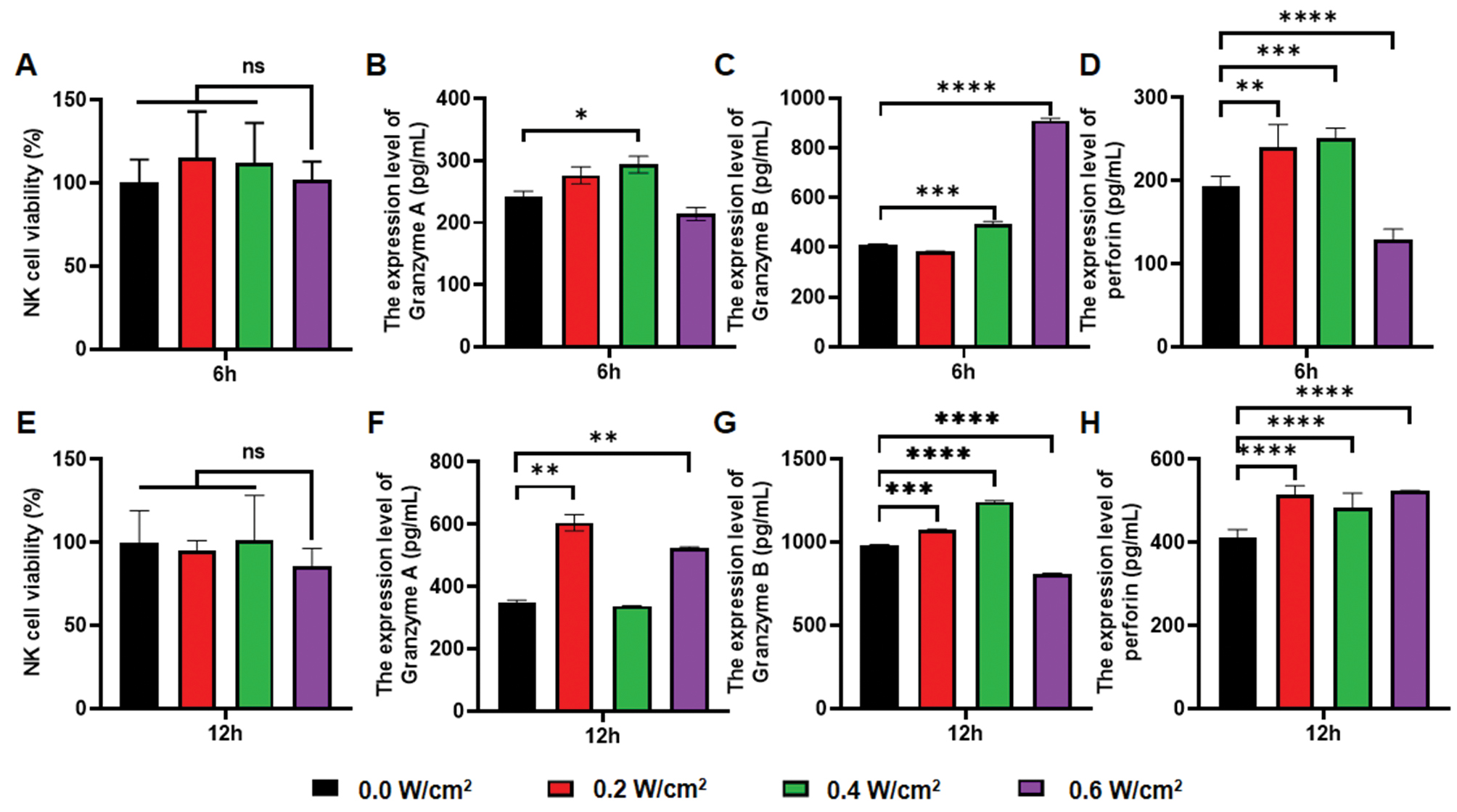
Granzyme A, a factor with trypsin-like effects, induces apoptosis primarily by activating the independent caspase pathway [23]. Unlike granzyme A, granzyme B is a product derived from aspartic acid, which activates the caspase 3 and 7 pathways, and thereby causes DNA breakage [24]. The cytotoxic substances secreted by NK cells include perforin and granzyme. Perforin is a glycoprotein that promotes granzyme entry into the cytoplasm and causes cell apoptosis [25, 26]. Moreover, Cichocki has demonstrated that perforin expression is associated with Wnt/β-catenin signaling pathway activity [19]. Herein, we determined the proliferation rates of NK cells, as well as the concentrations of granzyme A, granzyme B, and human perforin, in a coculture system comprising irradiated NK cells and SK-OV-3 cells. No significant differences were observed in NK cell viability at 6 h and 12 h after irradiation with LIPUS intensities of 0.2 W/cm2, 0.4 W/cm2, or 0.6 W/cm2 (Figure 2A, E, n=3, P>0.05), thus suggesting that LIPUS did not affect the NK cell proliferation rate. We subsequently subjected NK cells to 0.4 W/cm2 LIPUS irradiation, after co-culture with SK-OV-3 cells for 6 hours. These cells showed higher expression of granzyme A (20.6% higher; n=3, P<0.05), granzyme B (20.2% higher; n=3, P<0.001), and perforin (30.6% higher; n=3, P<0.001) than observed in cells with no LIPUS irradiation (Figure 2B–D). After 12 hours of co-incubation, the group treated with a sonication intensity of 0.2 W/cm2 showed a uniform increase in expression, comprising a 74.13% increase in granzyme A (n=3, P<0.01), 9.76% increase in granzyme B (n=3, P<0.001), and 24.78% increase in perforin (n=3, P<0.0001). In contrast, there was an inconsistent trend in the expression levels of each cytokine in the group treated with 0.6 W/cm2 ultrasound intensity (Figure 2F–H). On the basis of these findings, in conjunction with previous studies, we hypothesized that the increased cytokine expression might be associated with activation of the Wnt/β-catenin signaling pathway, and that the degree of activation of this pathway might correlate with ultrasound intensity.
Figure 2 Viability of NK cells and secretion levels of functional proteins by NK cells after LIPUS irradiation at varying acoustic power. A, E. Viability of NK cells 6 h (A) and 12 h (E) after exposure to various LIPUS irradiation intensities. B, F. Secretion levels of granzyme A in SK-OV-3 and NK cell co-culture system supernatants 6 h (B) and 12 h (F) after exposure to various LIPUS irradiation intensities. C, G. Secretion levels of granzyme B in SK-OV-3 and NK cell co-culture system supernatants 6 h (C) and 12 h (G) after exposure to various intensities of LIPUS irradiation. D, H. Secretion levels of perforin in SK-OV-3 and NK cell co-culture system supernatants 6 h (D) and 12 h (H) after exposure to various intensities of LIPUS irradiation. All experiments involved sequential irradiation with ultrasound intensities of 0.0 W/cm2, 0.2 W/cm2, 0.4 W/cm2, and 0.6 W/cm2 (1 MHz, pulse repetition frequency 100 Hz, duty cycle 20%, time 1 min). The results were statistically analyzed with one-way ANOVA. *P<0.05, **P<0.01, ***P<0.001, and ****P<0.0001 were considered statistically significant (n=3 per group).
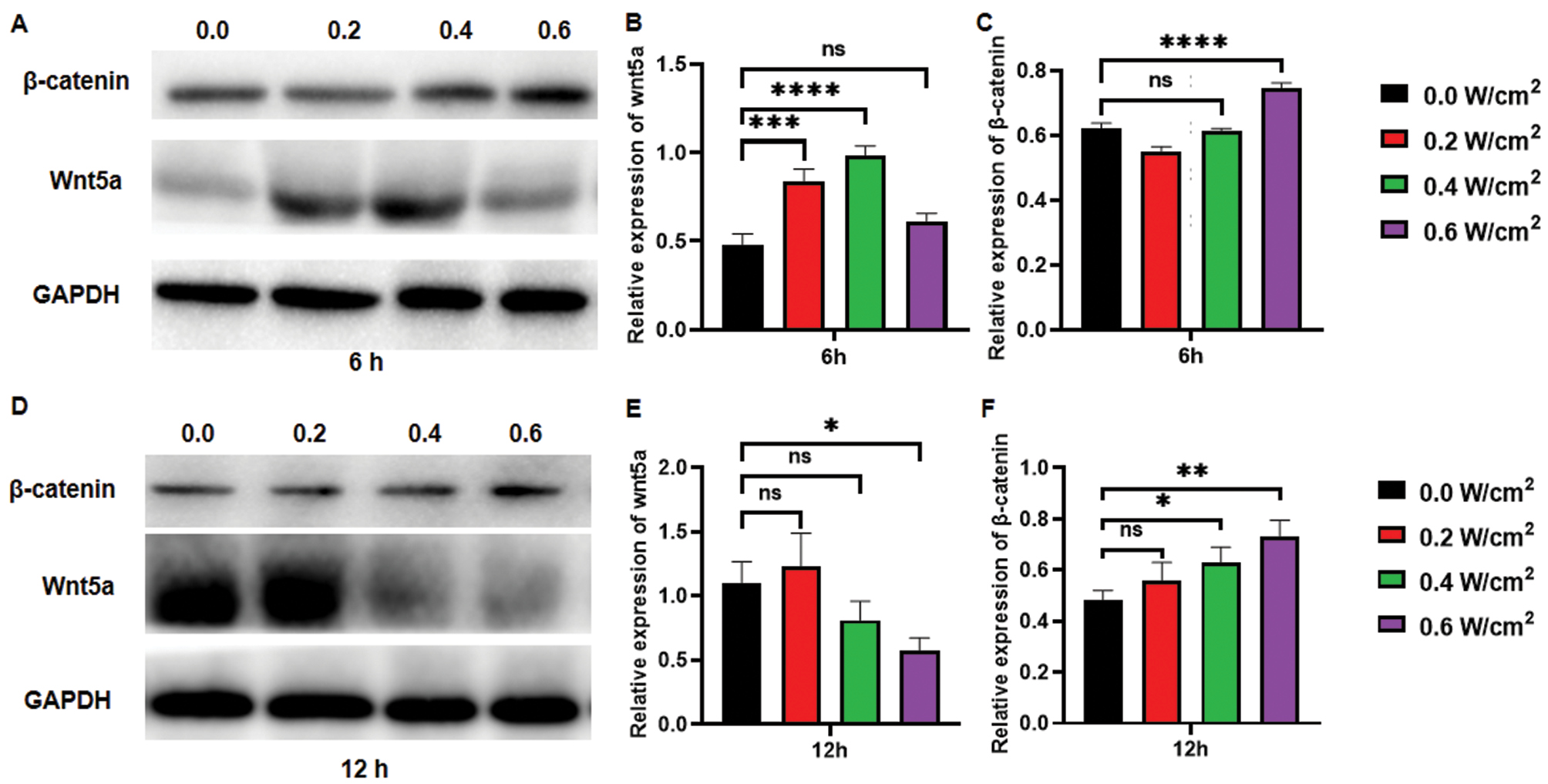
Consequently, we investigated whether the underlying mechanism might be associated with the Wnt/β-catenin signaling pathway. Tang et al. have demonstrated that LIPUS activates the Wnt/β-catenin signaling pathway in bone tissue and prevents bone loss [20]; therefore, LIPUS stimulation is associated with Wnt/β-catenin signaling pathway activity. In addition, ultrasound upregulates MAPK/ERK and PI3K/AKT pathway-related proteins, which in turn contribute to NK cell proliferation and cytokine secretion [27]. Our results indicated an increase in Wnt5a protein expression by 1.1-fold (n=3, P<0.001) and 1.4-fold (n=3, P<0.0001) 6 h after irradiation with 0.2 W/cm2 and 0.4 W/cm2 ultrasound, respectively, whereas no significant change in β-catenin protein expression was observed. However, with an ultrasound intensity of 0.6 W/cm2, Wnt5a protein did not significantly increase (n=3, P>0.05), whereas β-catenin protein increased by 20.97% (n=3, P<0.0001) (Figure 3A–C). At 12 h after 0.4 W/cm2 and 0.6 W/cm2 ultrasound irradiation, Wnt5a protein content significantly decreased, whereas β-catenin protein expression increased by 24.39% (n=3, P<0.05) and 46.34% (n=3, P<0.01), respectively, thus suggesting that Wnt5a protein might be associated with the levels of activated β-catenin protein (Figure 3D–F). On the basis of the above results, we hypothesized that LIPUS might first activate the Wnt/β-catenin signaling pathway and Wnt5a protein expression; subsequently promote the expression of the downstream protein β-catenin; and finally activate the downstream expression of secretion of virulence factors, such as granzyme and perforin.
Figure 3 Expression of Wnt5a and β-catenin in NK cells after LIPUS irradiation. A. Western blot results of Wnt5a and β-catenin in NK cells 6 hours after LIPUS irradiation at various intensities. B, C. Quantitative grayscale analysis of western blot bands of Wnt5a (B) and β-catenin (C) in NK cells 6 hours after LIPUS irradiation at various intensities. D. Western blot results of Wnt5a and β-catenin in NK cells 12 hours after LIPUS irradiation at various intensities. E, F. Quantitative grayscale analysis of western blot bands of Wnt5a (E) and β-catenin (F) in NK cells 6 hours after LIPUS irradiation at various intensities. All experiments involved sequential irradiation with ultrasound intensities of 0.0 W/cm2, 0.2 W/cm2, 0.4 W/cm2, and 0.6 W/cm2 (1 MHz, pulse repetition frequency 100 Hz, duty cycle 20%, time 1 min). Expression levels of Wnt5a and β-catenin were quantified in grayscale in ImageJ. The results were statistically analyzed with one-way ANOVA. *P<0.05, **P<0.01, ***P<0.001, and ****P<0.0001 were considered statistically significant (n=3 per group).
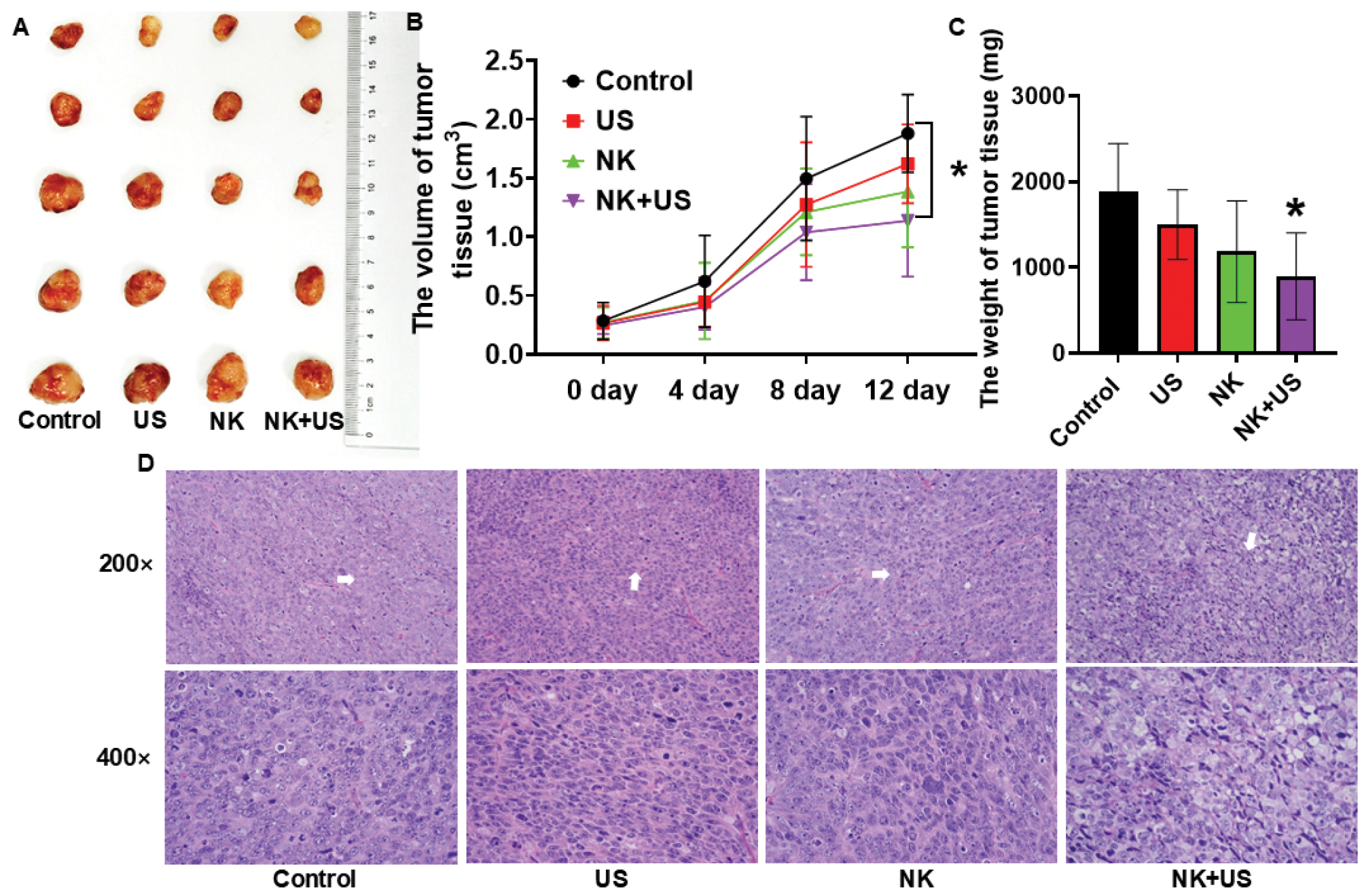
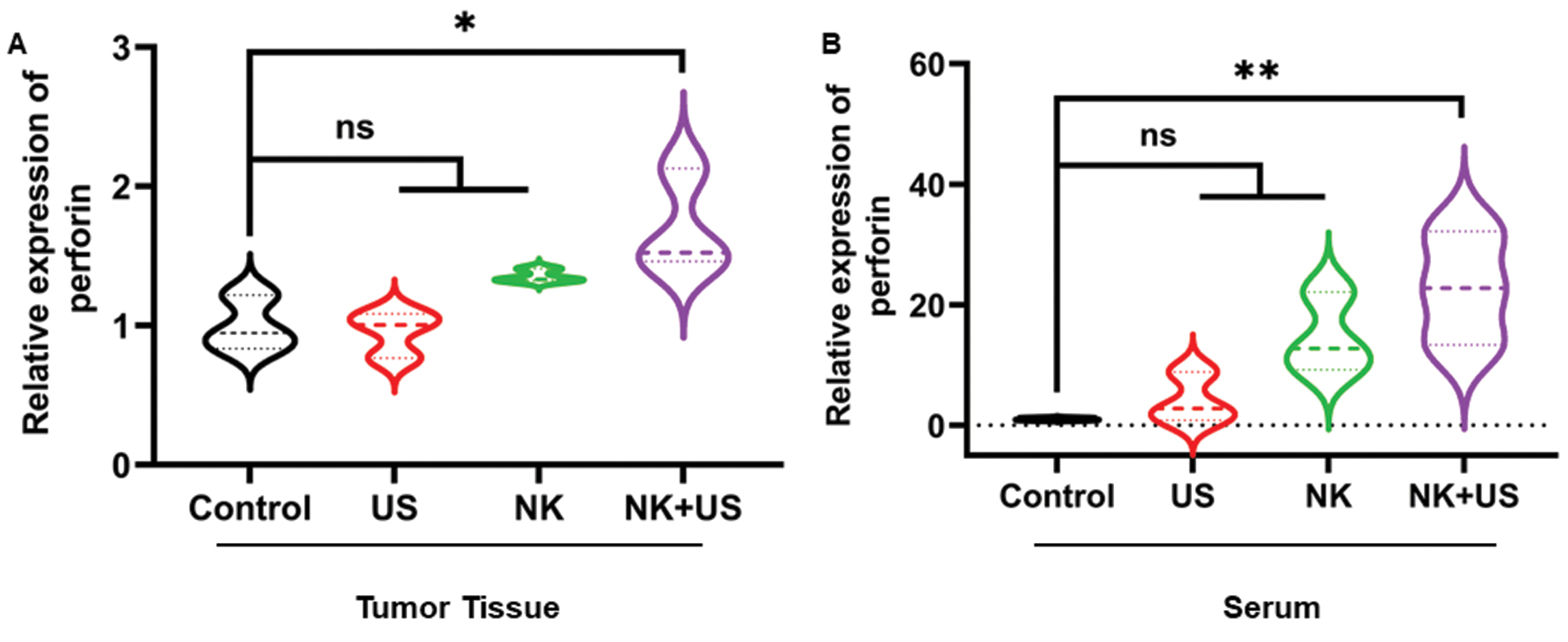
Furthermore, we verified that NK cells subjected to LIPUS irradiation achieved greater anti-tumor efficacy than NK cells without LIPUS irradiation or LIPUS irradiation alone. On day 12 after treatment, the tumor volume and weight in the NK cell combined with LIPUS treatment group were significantly lower than those in the Control group (tumor volume decreased by 39.52%, tumor weight decreased by 52.75%, n=5, P<0.05). (Figure 4A–C). According to tumor tissue histology, in the group with NK cells combined with LIPUS treatment, the intercellular space in the tumor tissue showed widening and contained abundant inflammatory cells. Simultaneously, the presence of vacuole-like structures indicated cancer cell necrosis and deformation (Figure 4D). Compared with the Control group, the NK cell combined with ultrasound treatment group showed a 70.41% increase in perforin secretion in tumor tissue (n=3, P<0.05) and a 21-fold increase in serum (n=3, P<0.01) (Figure 5).
Figure 4 Anti-tumor activity of NK cells against ovarian cancer xenografts (scale bar: 50 μm). A. Photographs of tumor tissue in vitro after various treatments. B. Volume change curves of tumors under various treatments. C. Tumor tissue weights in the indicated groups. D. Histological slices of tumor tissues after various treatments. These results were statistically analyzed with one-way ANOVA. *P<0.05 was considered statistically significant (n=5 per group).
Figure 5 Secretion of perforin in vivo after various treatments. A. Secretion of perforin in tumor tissue after various treatments. B. Secretion of perforin in serum after various treatments. The results were statistically analyzed with one-way ANOVA. *P<0.05 was considered statistically significant (n=3 per group).
Herein, LIPUS was found to enhance NK cells’ anti-tumor efficacy by activating the Wnt/β-catenin signaling pathway. However, some prior studies have concluded that LIPUS significantly activates the Wnt/β-catenin signaling pathway, whereas others have reported that LIPUS suppresses the Wnt/β-catenin signaling pathway [28]. These discrepant findings might be associated with the applied ultrasound parameters. According to previous research, 0.5 W/cm2 LIPUS enhances iPSC–NCSC proliferation ability, whereas 1.5 W/cm2 LIPUS has opposite effects [29]. In addition, we observed inconsistency in the results obtained with various ultrasound intensities and sampling times; therefore, additional subgroups and longer observation times must be examined in subsequent studies to further optimize this treatment regimen. In addition, although LIPUS is safe and noninvasive, and has been demonstrated by several recent clinical studies to provide advantages of safety and noninvasiveness in short-term treatment [30–32], an in-depth examination of the effects on normal tissues as well as the long-term effects on the disease is lacking. Therefore, the mid- and long-term effects of LIPUS on normal tissues and disease sites must be explored to enable application of this technique in late clinical stages.
In conclusion, this study provides a precise, controllable, safe, and effective strategy for enhancing NK therapy through LIPUS. The combination of LIPUS was found to increase the cytotoxicity and anti-tumor efficiency of NK cells in ovarian cancer. Future research is needed to clarify the detailed relationship between changes in the Wnt/β-catenin signaling pathway in NK cells and various ultrasound parameters, to further refine the mechanism.
Data availability statement
All data analyzed during this study are included in this published article. Additional data are available from the corresponding author on reasonable request.
Ethics statement
All animal experiments complied with the ARRIVE guidelines and were performed in accordance with the U.K. Animals (Scientific Procedures) Act of 1986 and associated guidelines in EU Directive 2010/63/EU for animal experiments. All animal experiments were approved by the University of South China Animal Experiment Ethics Review.
Author contributions
Renjie Feng, Conceptualization, Writing—original draft, Writing—review & editing. Meng Du: Conceptualization, Supervision, Writing—review & editing, Funding acquisition. Ao Gu: Investigation, Methods. Gökhan Zengin: Conceptualization, Supervision, Writing—review & editing.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82272028, 81971621, and 82102087), the Natural Science Foundation of Hunan Province (2022JJ30039 and 2022JJ40392), and the Natural Science Foundation of Guangdong Province (2021A1515011177).
Conflict of interest
The authors declare that there are no conflicts of interest.
References
- Konstantinopoulos PA, Matulonis UA. Clinical and translational advances in ovarian cancer therapy. Nat Cancer 2023;4(9):1239-57. [PMID: 37653142 DOI: 10.1038/s43018-023-00617-9]
- Shimasaki N, Jain A, Campana D. NK cells for cancer immunotherapy. Nat Rev Drug Discov 2020;19(3):200-18. [PMID: 31907401 DOI: 10.1038/s41573-019-0052-1]
- Vivier E, Rebuffet L, Narni-Mancinelli E, Cornen S, Igarashi RY, Fantin VR. Natural killer cell therapies. Nature 2024;626(8000):727-36. [PMID: 38383621 DOI: 10.1038/s41586-023-06945-1]
- Lin C, Horwitz ME, Rein LAM. Leveraging natural killer cell innate immunity against hematologic malignancies: from stem cell transplant to adoptive transfer and beyond. Int J Mol Sci 2022;24(1):204. [PMID: 36613644 DOI: 10.3390/ijms24010204]
- Shin MH, Kim J, Lim SA, Kim J, Kim SJ, et al. NK cell-based immunotherapies in cancer. Immune Netw 2020;20(2):e14. [PMID: 32395366 DOI: 10.4110/in.2020.20.e14]
- Zhu H, Blum RH, Bjordahl R, Gaidarova S, Rogers P, et al. Pluripotent stem cell-derived NK cells with high-affinity noncleavable CD16a mediate improved antitumor activity. Blood 2020;135(6):399-410. [PMID: 31856277 DOI: 10.1182/blood.2019000621]
- Li Y, Hermanson DL, Moriarity BS, Kaufman DS. Human iPSC-derived natural killer cells engineered with chimeric antigen receptors enhance anti-tumor activity. Cell Stem Cell 2018;23(2):181-92.e5. [PMID: 30082067 DOI: 10.1016/j.stem.2018.06.002]
- Du N, Guo F, Wang Y, Cui J. NK cell therapy: a rising star in cancer treatment. Cancers (Basel) 2021;13(16):4129. [PMID: 34439285 DOI: 10.3390/cancers13164129]
- Jiang D, Zhang J, Mao Z, Shi J, Ma P. Driving natural killer cell-based cancer immunotherapy for cancer treatment: An arduous journey to promising ground. Biomed Pharmacother. 2023;165:115004.
- Rix A, Lederle W, Theek B, Lammers T, Moonen C, et al. Advanced ultrasound technologies for diagnosis and therapy. J Nucl Med 2018;59(5):740-6. [PMID: 29496981 DOI: 10.2967/jnumed.117.200030]
- O’Brien WD Jr. Ultrasound-biophysics mechanisms. Prog Biophys Mol Biol 2007;93(1-3):212-55. [PMID: 16934858 DOI: 10.1016/j.pbiomolbio.2006.07.010]
- Al Musawi MS, Jaafar MS, Al-Gailani B, Ahmed NM, Suhaimi FM, et al. Effects of low-level laser irradiation on human blood lymphocytes in vitro. Lasers Med Sci 2017;32(2):405-11. [PMID: 28044209 DOI: 10.1007/s10103-016-2134-1]
- Bamps M, Dok R, Nuyts S. Low-level laser therapy stimulates proliferation in head and neck squamous cell carcinoma cells. Front Oncol 2018;8:343. [PMID: 30211121 DOI: 10.3389/fonc.2018.00343]
- Huang P. An integrated approach to ultrasound imaging in medicine and biology. BIO Integration 2020;1(3):105-9. [DOI: 10.15212/bioi-2020-0036]
- Phan TN, Fan CH, Yeh CK. Application of ultrasound to enhancing stem cells associated therapies. Stem Cell Rev Rep 2023;19(6):1709-25. [PMID: 37119453 DOI: 10.1007/s12015-023-10546-w]
- Liang W, Liang B, Yan K, Zhang G, Zhuo J, et al. Low-intensity pulsed ultrasound: a physical stimulus with immunomodulatory and anti-inflammatory potential. Ann Biomed Eng 2024;52(8):1955-81. [PMID: 38683473 DOI: 10.1007/s10439-024-03523-y]
- Lu P, Zhu X-Q, Xu Z-L, Zhou Q, Zhang J, et al. Increased infiltration of activated tumor-infiltrating lymphocytes after high intensity focused ultrasound ablation of human breast cancer. Surgery 2009;145(3):286-93. [PMID: 19231581 DOI: 10.1016/j.surg.2008.10.010]
- Abe S, Nagata H, Crosby EJ, Inoue Y, Kaneko K, et al. Combination of ultrasound-based mechanical disruption of tumor with immune checkpoint blockade modifies tumor microenvironment and augments systemic antitumor immunity. J Immunother Cancer 2022;10(1):e003717. [PMID: 35039461 DOI: 10.1136/jitc-2021-003717]
- Cichocki F, Valamehr B, Bjordahl R, Zhang B, Rezner B, et al. GSK3 inhibition drives maturation of NK cells and enhances their antitumor activity. Cancer Res 2017;77(20):5664-75. [PMID: 28790065 DOI: 10.1158/0008-5472.CAN-17-0799]
- Tang L, An S, Zhang Z, Fan X, Guo J, et al. MSTN is a key mediator for low-intensity pulsed ultrasound preventing bone loss in hindlimb-suspended rats. Bone 2021;143:115610. [PMID: 32829040 DOI: 10.1016/j.bone.2020.115610]
- Liu S, Zhou M, Li J, Hu B, Jiang D, et al. LIPUS inhibited the expression of inflammatory factors and promoted the osteogenic differentiation capacity of hPDLCs by inhibiting the NF-κB signaling pathway. J Periodontal Res 2020;55(1):125-40. [PMID: 31541455 DOI: 10.1111/jre.12696]
- Ma B, Hottiger MO. Crosstalk between Wnt/β-catenin and NF-κB signaling pathway during inflammation. Front Immunol 2016;7:378. [PMID: 27713747 DOI: 10.3389/fimmu.2016.00378]
- Zhou Z, He H, Wang K, Shi X, Wang Y, et al. Granzyme A from cytotoxic lymphocytes cleaves GSDMB to trigger pyroptosis in target cells. Science 2020;368(6494):eaaz7548. [PMID: 32299851 DOI: 10.1126/science.aaz7548]
- Rasi V, Hameed OA, Matthey P, Bera S, Grandgenett DP, et al. Improved purification of human granzyme A/B and granulysin using a mammalian expression system. Front Immunol 2022;13:830290. [PMID: 35300343 DOI: 10.3389/fimmu.2022.830290]
- Guan X, Guo H, Guo Y, Han Q, Li Z, et al. Perforin 1 in cancer: mechanisms, therapy, and outlook. Biomolecules 2024;14(8):910. [PMID: 39199299 DOI: 10.3390/biom14080910]
- Laskarin G, Babarovic E, Kifer N, Bulimbasic S, Sestan M, et al. Involvement of M1-activated macrophages and perforin/granulysin expressing lymphocytes in IgA vasculitis nephritis. Int J Mol Sci 2024;25(4):2253. [PMID: 38396930 DOI: 10.3390/ijms25042253]
- Lee IC, Wu HJ, Liu HL. Dual-frequency ultrasound induces neural stem/progenitor cell differentiation and growth factor utilization by enhancing stable cavitation. ACS Chem Neurosci 2019;10(3):1452-61. [PMID: 30608667 DOI: 10.1021/acschemneuro.8b00483]
- Liao B, Guan M, Tan Q, Wang G, Zhang R, et al. Low-intensity pulsed ultrasound inhibits fibroblast-like synoviocyte proliferation and reduces synovial fibrosis by regulating Wnt/β-catenin signaling. J Orthop Translat 2021;30:41-50. [PMID: 34611513 DOI: 10.1016/j.jot.2021.08.002]
- Lv Y, Zhao P, Chen G, Sha Y, Yang L. Effects of low-intensity pulsed ultrasound on cell viability, proliferation and neural differentiation of induced pluripotent stem cells-derived neural crest stem cells. Biotechnol Lett 2013;35(12):2201-12. [PMID: 24078117 DOI: 10.1007/s10529-013-1313-4]
- Jia L, Li D, Wei X, Chen J, Zuo D, et al. Efficacy and safety of focused low-intensity pulsed ultrasound versus pulsed shortwave diathermy on knee osteoarthritis: a randomized comparative trial. Sci Rep 2022;12(1):12792. [PMID: 35896688 DOI: 10.1038/s41598-022-17291-z]
- Mohamad Yusoff F, Kajikawa M, Yamaji T, Kishimoto S, Maruhashi T, et al. Low-intensity pulsed ultrasound improves symptoms in patients with Buerger disease: a double-blinded, randomized, and placebo-controlled study. Sci Rep 2024;14(1):13704. [PMID: 38871832 DOI: 10.1038/s41598-024-64118-0]
- Li W, Li X, Kong Z, Chen B, Zhou H, et al. Efficacy of low-intensity pulsed ultrasound in the treatment of COVID-19 pneumonia. Ultraschall Med 2023;44(6):e274-e83. [PMID: 37467781 DOI: 10.1055/a-2133-0835]