Asymmetric Cell Division as a Mechanistic Contributor to Cancer with a Focus on Glioblastoma
1Leon M Goldstein School, Brooklyn, NY, USA
2Advanced Gynecology and Surgery, Maimonides Medical Center, Brooklyn, NY, USA
3Cancer Research Institute, Jinan University, Guangzhou, China
4Tianjin Yinuo Biomedical Co., Ltd, Tianjin, China
aThese authors contribute equally.
*Correspondence to: Genia Bekker, Advanced Gynecology and Surgery, Maimonides Medical Center, Brooklyn, NY, USA. E-mail: Bekkermd@hotmail.com; Hengrui Liu, Tianjin Yinuo Biomedical Co., Ltd, Tianjin, China. E-mail: lh@yinuobiomedical.cn; Co-author: Rebecca Golin, E-mail: rebeccagolin10@gmail.com
Received: April 26 2025; Revised: May 29 2025; Accepted: June 16 2025; Published Online: July 4 2025.
Cite this paper:
Golin R, Bekker G, Liu H. Asymmetric Cell Division as a Mechanistic Contributor to Cancer with a Focus on Glioblastoma. BIO Integration 2025; 6: 1–8.
DOI: 10.15212/bioi-2025-0076. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Asymmetric cell division (ACD) has a crucial role in normal cellular differentiation and tissue homeostasis. The mechanisms underlying ACD are highly intricate and involve coordinated molecular and cellular processes. Dysregulation of ACD has been implicated in various human cancers by contributing to malignant tumor initiation, progression, metastasis, and treatment resistance. Although numerous studies have explored the relationship between ACD and cancer, many questions remain unanswered. This literature review aims to evaluate the potential biological significance of ACD in cancer with a focus on the diagnostic and prognostic relevance to glioblastoma. A comprehensive PubMed search was conducted from 2008 to the present using keywords, such as “asymmetric cell division”, “cancer”, “glioblastoma”, and “tumorigenesis”. The selected articles were analyzed to assess ACD-related data and the clinical correlations. Special emphasis was placed on glioblastomas, an aggressive brain tumor with limited improvement in patient survival over recent years. The review underscores the crucial role of ACD in normal tissue homeostasis and ACD dysregulation in cancer initiation, progression, therapeutic resistance, and metastatic potential. Understanding how ACD contributes to cancer heterogeneity may provide insights into innovative strategies for tumor detection, monitoring, and treatment. Future research into the molecular mechanisms governing ACD could facilitate the development of novel glioblastoma therapies aimed at restoring or modulating ACD processes to improve patient outcomes.
Keywords
Asymmetric cell division, cancer, glioblastoma, tumorigenesis.
Introduction
Cancer is characterized by uncontrolled cell growth, invasion of surrounding tissues, and in many cases, metastasis to distant organs [1]. For these reasons, cancer remains one of the most significant challenges in modern medicine. Among the diverse array of cancers, glioblastoma (GBM) stands out as the most aggressive and lethal primary brain tumor in adults [2]. GBM is marked by rapid proliferation, extensive infiltration into surrounding brain tissue, resistance to conventional therapies, and poor patient prognosis and is classified as a World Health Organization (WHO) grade IV glioma [3, 4].
Asymmetric cell division (ACD), a conserved mechanism for generating cellular diversity, has recently gained attention for its role in GBM [5]. Dysregulated ACD may contribute to germline stem cell persistence [6], tumor growth [7], therapeutic resistance [5, 8], and recurrence [9, 10]. ACD research has provided insights into tumor growth and treatment resistance and highlighted potential therapeutic targets [5]. This paper aims to close gaps in understanding the relationship between ACD and tumorigenesis by focusing on abnormal ACD mechanisms that contribute to malignancies.
A literature review was performed using PubMed publications from 2010 to the present. The aim of the review was to provide insights into potential therapeutic strategies for GBM by analyzing regulatory control elements.
Narrative literature review approach
A narrative literature search was performed in PubMed to identify studies exploring ACD mechanisms in GBM. The search was limited to English-language articles published between January 2010 and April 2025 using the keywords, “glioblastoma” AND “asymmetric cell division”. The initial query returned 12 records. Titles and abstracts were screened for relevance to GBM-specific ACD pathways and regulatory proteins. Studies lacking direct mechanistic data or focused solely on non-glioma models were excluded. Five key articles that provided insights into molecular regulators (e.g., TRIM3 and BMP4-SMAD signaling) and their effects on GBM stem-like cell division were selected for detailed discussion after screening. A formal PRISMA flow diagram was not included given the narrative review format. Instead, the selection process described above to ensure transparency while allowing focused, mechanistic synthesis of the most pertinent GBM-ACD literature.
Discovery of ACD
Edwin Conklin (1863–1952), an American biologist, was the first to observe that during the development of ascidian embryos, a distinct yellow-colored cytoplasm is unevenly distributed during cell division, leading to different cell fates in the daughter cells [11]. This discovery provided the first demonstration of ACD. This historical observation underscores the fundamental importance of cytoplasmic determinants in cell fate decisions, a concept that has since been recognized as a cornerstone in developmental biology [12].
Despite Conklin’s observations over a century ago, the mechanistic understanding of ACD has progressed slowly. While the detailed mechanisms of ACD are beyond the scope of this paper, the focus will be on the relevance of ACD to cancer initiation, tumor growth, progression, and potential therapeutic strategies. Understanding the relationship between ACD and cancer is crucial because dysregulation has been implicated in cancer stem cell (CSC) maintenance and therapeutic resistance, especially in aggressive cancers like GBM.
The mechanism underlying ACD
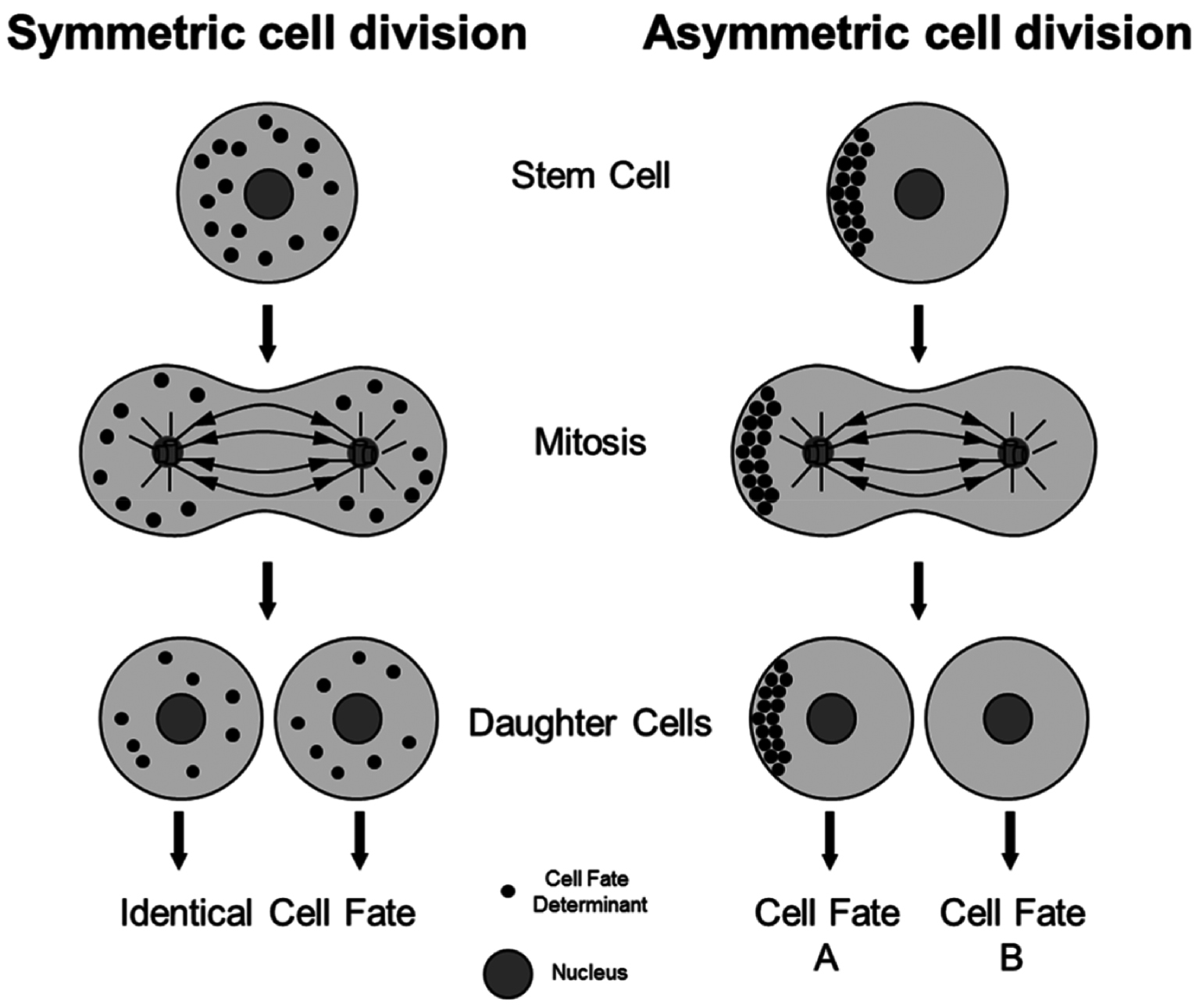
A key principle of ACD is the establishment of distinct sibling cell fates by mechanisms linked to mitosis (Figure 1). Figure 1 illustrates the fundamental differences between symmetric cell division (SCD) and ACD. SCD results in two daughter cells with identical fates that is achieved through the equal distribution of cell fate determinants, as shown in panel (a). In contrast, panel (b) depicts ACD, in which daughter cells inherit different amounts or compositions of cell fate determinants, resulting in distinct cell fates [13].
Figure 1 Symmetric versus asymmetric cell division. During a symmetric cell division. Figure was reproduced from reference [43] with permission from Frontiers (Copyright 2014).
Asymmetric fate can be established through exposure to cell-extrinsic signaling cues. Alternatively, asymmetric inheritance of cell-intrinsic cell fate determinants, such as proteins or RNA, can induce asymmetric cell fate decisions [14]. ACD, a process through which a single cell divides into two distinct daughter cells, is critical for maintaining healthy stem cell populations and tissue integrity. ACD is particularly important within stem cell populations [15]. Stem cells are specialized cells capable of dividing and differentiating into various cell types, which contributes to tissue renewal and repair. Stem cells generate diverse cell types, such as blood, bone, and muscle cells, while simultaneously self-renewing to maintain the stem cell population. Importantly, stem cells have a crucial role in cancer treatment, especially in hematologic malignancies like blood cancers, as well as in regenerative medicine [16].
The discovery of diagnostic and therapeutic strategies depends on unlocking the mechanisms underlying ACD together with its cancer-related functions. Cell differentiation, tissue homeostasis, and cancer development are intimately linked to the proper regulation of ACD. Dysregulation of this process can lead to tumorigenesis and therapeutic resistance, underscoring the importance of ongoing research in this field.
Relevance of ACD and cancer
ACD and tumorigenesis
Cancer is a complex, multifaceted disease characterized by uncontrolled cell growth and differentiation [17]. Cancer arises through the stepwise acquisition of genetic mutations that provide a survival advantage to affected cells. The cell cycle has been one of the most studied mechanisms in cancers [18, 19]. Many proteins well known for roles in cell cycle control and DNA repair, such as CDK2 [20] and RAD51 [21], as well as factors like CNIH4 [22] and AIMP1 [23] that influence signaling and trafficking, have more recently been implicated in the regulation of ACD in cancer. Dysregulation of ACD in GBM stem-like cells is driven by loss of the spindle-orientation regulator, TRIM3. BMP4-mediated shifts in fate determinant localization and altered distribution of receptors, such as EGFR and p75NTR, contribute directly to tumor initiation, heterogeneity, and treatment resistance [24]. ACD is critical for maintaining tissue homeostasis and generating cellular diversity in normal tissues [25]. Stem cells dynamically switch between SCD and ACD under physiologic conditions to preserve homeostasis [26]. This process requires highly coordinated intra- and extra-cellular signaling events. When dysregulated, however, ACD increases the risk of malignant transformation [27].
ACD in stem and progenitor cells
Stem and progenitor cells are defined by a self-renewal capacity and ability to produce differentiated progeny (Figure 2). A delicate balance between these processes is essential for developmental cellular diversity and adult tissue maintenance [28]. Dysregulation of ACD disrupts this balance, potentially driving tumorigenesis by producing aggressive, fast-dividing daughter cell clones that are often resistant to treatment.
Figure 2 Dynamics of asymmetric and symmetric divisions of stem cells.
Emerging evidence supports the notion that ACD functions as a tumor-suppressive mechanism. Disruption of ACD in Drosophila neuroblasts has been associated with abnormal proliferation and genomic instability, suggesting a potential role in early carcinogenesis [29]. Studies have indicated that oncogenic mutations can hijack ACD pathways, disrupting mechanisms responsible for cell proliferation, cycle progression, and genomic integrity.
For example, Mukherjee et al. demonstrated that CSCs, which drive tumor initiation and propagation, often rely on symmetric divisions. Loss of ACD may thus result in tissue dysregulation and contribute to the development and growth of tumors. Aberrant ACD also promotes stem-like cell accumulation and limits differentiation, which reinforces cancer stemness [30, 31].
ACD and cancer treatment resistance
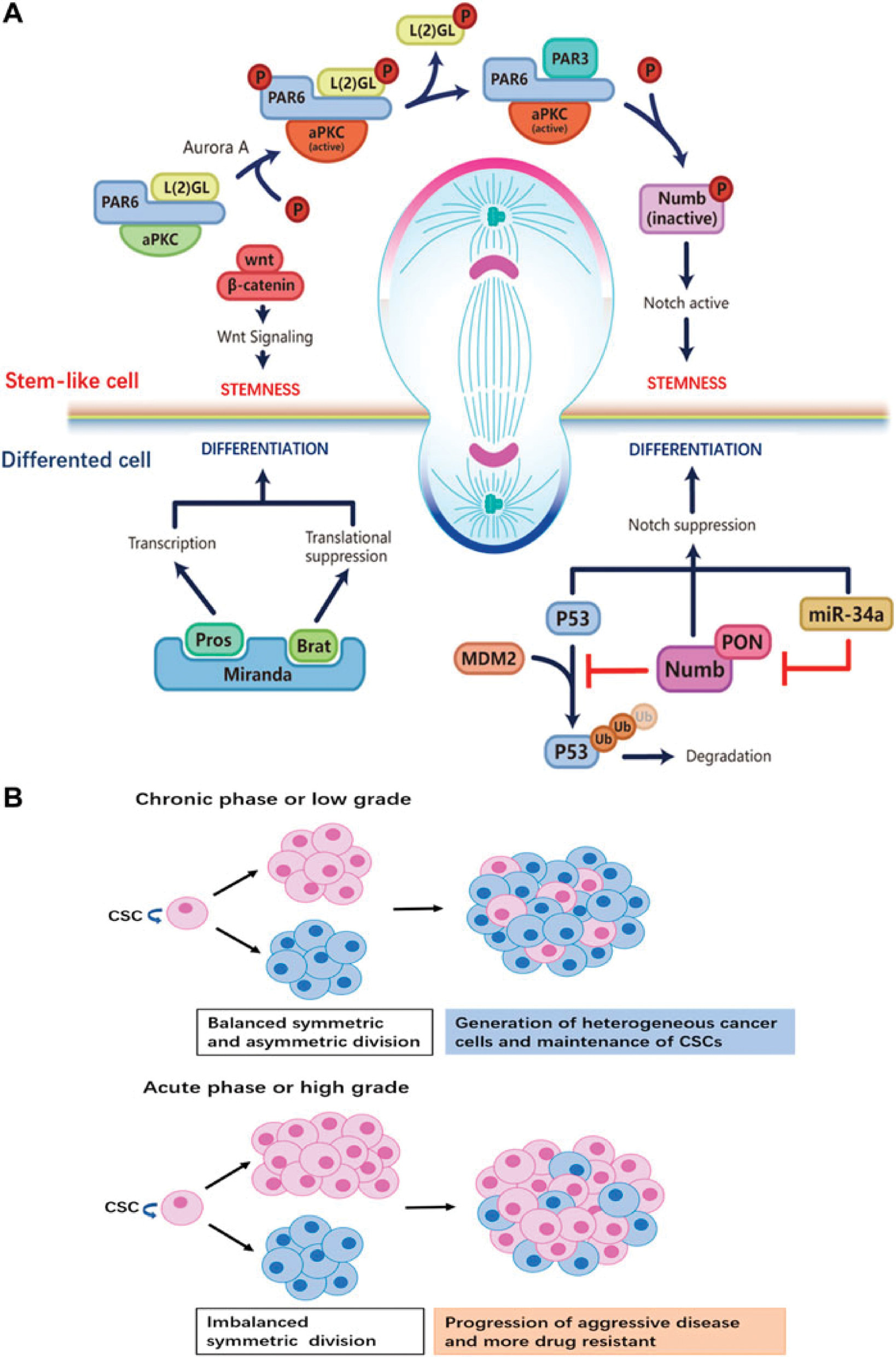
Dysregulation of ACD not only skews the balance between stem-like and differentiated progeny but also endows one daughter cell with molecular features that drive treatment resistance (Figure 1A,B). The PAR3–aPKC complex becomes phosphorylated and localizes to the apical cortex under normal ACD, which then activates Wnt/β-catenin signaling and maintains stemness. The basal cortex concentrates Numb (inactivated by aPKC phosphorylation) and Miranda, which recruit differentiation factors (Prospero and Brat) to suppress stem-cell programs via transcriptional and translational repression. Aberrant aPKC activation or loss of cortical polarity proteins in cancer leads to missegregation of these determinants; both daughters may inherit stemness signals or conversely may not, which disrupts tissue homeostasis.
Mislocalized PAR6/aPKC enhances Notch activity through Numb inactivation in GBM CSCs, promoting symmetric self-renewal and expanding the resistant pool. Simultaneously, disruption of the p53-MDM2 axis, often through upregulation of the p53-inhibitors PON or miR-34a, impairs apoptosis in the basal daughter cell, further biasing survival. Previous studies directly demonstrated that ACD produces one daughter enriched for EGFR and p75NTR, which exhibits heightened resilience to temozolomide and radiation, while the sibling remains sensitive [27].
Moreover, key repair and cell-cycle regulators, such as RAD51 [21] and CDK2 [20], are preferentially retained in the resistant daughter following spindle-orientation defects driven by TRIM3 loss, equipping the resistant daughter with superior DNA-damage response capacity and proliferative drive. Thus, ACD dysregulation creates a self-reinforcing loop of resistance; polarizing signals go awry, fate determinants mispartition, survival pathways dominate, and resistant clones expand.
Restoring ACD fidelity, whether by correcting cortical polarity through TRIM3 restoration, modulating aPKC activity to re-establish proper PAR3/Numb localization, or leveraging BMP4 to bias divisions toward differentiation, could rebalance cell-fate outcomes. Tracking asymmetric distribution of EGFR/p75NTR or nuclear p53 status at the single-cell resolution may serve as early biomarkers of emerging resistance. By targeting the molecular circuitry of ACD, we may not only curb tumor heterogeneity but also sensitize GBM and other solid tumors to existing therapies (Figure 3).
Figure 3 Regulatory mechanisms of ACD in CSCs and the impact on tumor progression. Figure was reproduced from reference [27] with permission from Frontiers (Copyright 2022).
Previous studies discovered that ACD generates progeny with increased growth factor receptor expression, such as EGFR [32, 33] and p75NTR [34], conferring greater therapeutic resistance in glioblastoma CSCs. Similarly, resistance to radiation therapy (RT) in head and neck cancers has been linked to accelerated stem cell division and impaired ACD, underscoring the relevance of ACD dysregulation in cancer treatment resistance [27].
ACD in GBM
Despite these promising mechanistic insights, translating “ACD therapy” into the clinic faces two major challenges. First, BMP4 is a large cytokine with poor blood–brain barrier (BBB) permeability [35]. Strategies under investigation to enhance CNS delivery include convection-enhanced delivery, nanoparticle carriers engineered for receptor-mediated transcytosis, and focused ultrasound to transiently open the BBB. Second, traditional cell lines fail to capture the full heterogeneity of GBM. Patient-derived organoids and orthotopic xenografts now serve as more faithful preclinical platforms to evaluate ACD-modulating interventions. These models allow assessment of intra-tumoral drug penetration, off-target effects, and efficacy across diverse GBM genetic backgrounds, bridging in vitro findings to in vivo proof-of-concept.
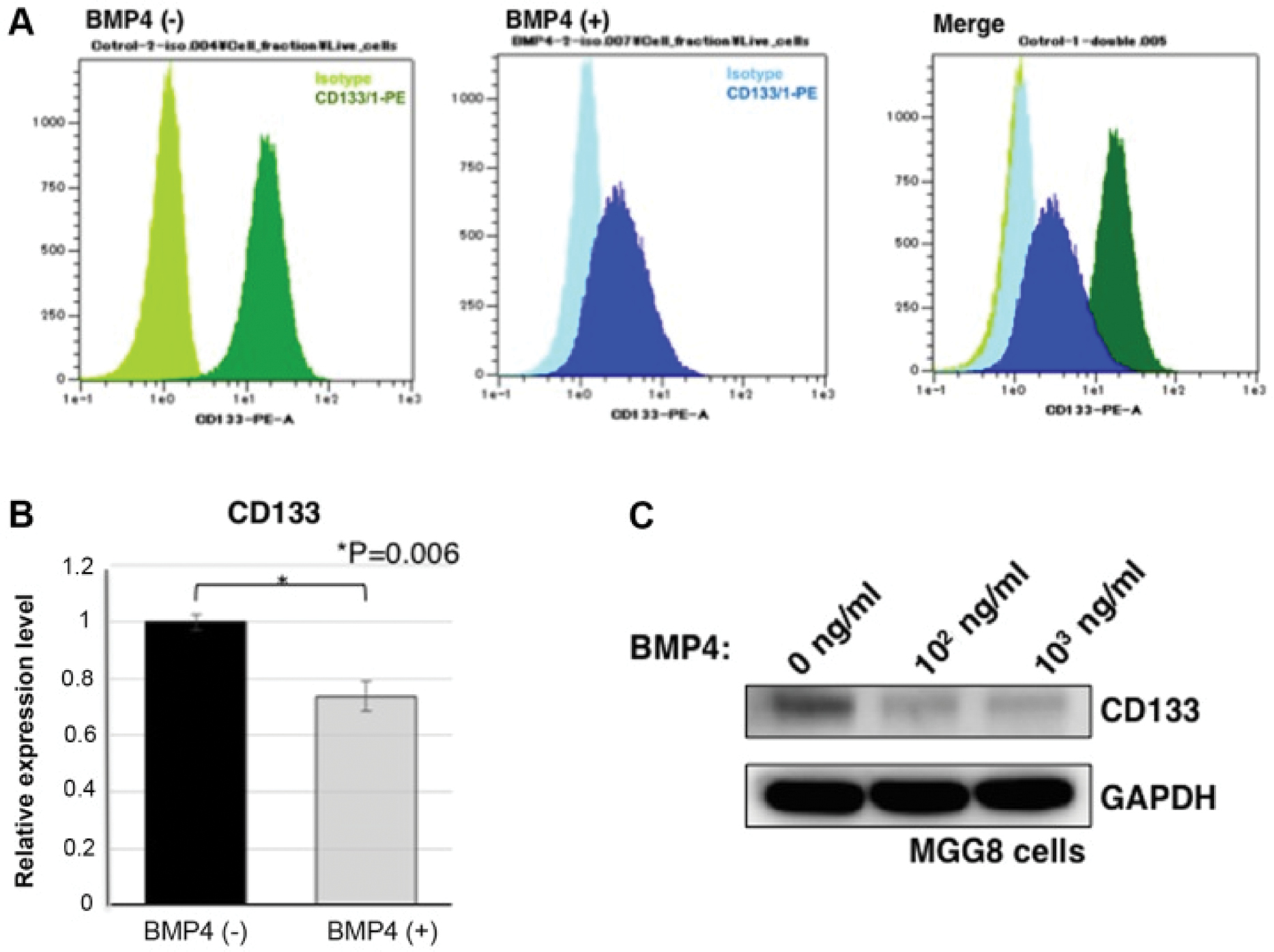
Importantly, TRIM3, a brain-enriched E3 ubiquitin ligase, has emerged as a GBM-specific regulator of ACD. Whereas TRIM3 expression is markedly reduced in > 80% of primary GBM specimens, the TRIM3 levels remain comparatively stable in most non–CNS tumors [36, 37]. Loss of TRIM3 disrupts mitotic spindle orientation in glioma stem-like cells, skewing divisions toward symmetric self-renewal and driving tumor expansion. Restoration of TRIM3 expression in GBM models re-establishes proper ACD, diminishes stemness marker expression, and suppresses tumor growth. This tumor-selective vulnerability underscores the therapeutic promise of targeting TRIM3-mediated ACD pathways in GBM (Figure 4).
Figure 4 BMP4-induced ACD and CD133 downregulation in glioblastoma stem-like cells. Figure was reproduced from reference [35] with permission from Spandidos Publications (Copyright 2020). BMP4 exposure markedly reduces the CD133 level in MGG8 cells. (A) Flow cytometry of CD133-PE staining in MGG8 cells treated with 100 ng/mL of BMP4 for 3 days versus untreated controls; light green and light blue trace the IgG1-PE isotype background, while green and blue depict specific CD133-PE signals. (B) qPCR quantification of CD133 transcripts following the same BMP4 regimen, shown as the mean ± SEM of three independent experiments (*P = 0.006). (C) Western blot analysis of CD133 protein in MGG8 cells after 3 days of BMP4 treatment at 100 ng/mL and 1000 ng/mL, with GAPDH serving as a loading control. BMP4, bone morphogenetic protein 4; PE, phycoerythrin.
Prospective and future directions
Molecular mechanisms of ACD
Future research should focus on elucidating the intricate molecular signaling pathways that regulate ACD, including the roles of polarity proteins, spindle orientation, and cell-fate determinants.
ACD in CSCs
Investigating the relationship between ACD and CSCs could provide insights into tumor initiation, progression, and resistance to therapies. Identifying molecular signatures of ACD dysregulation in CSCs may lead to novel diagnostic markers.
Therapeutic targeting of ACD
Developing therapeutic strategies to modulate ACD, such as restoring ACD in CSCs, holds promise for cancer treatment [38, 39]. For example, targeting regulators like TRIM3 or using BMP4-based therapies could be explored further.
ACD dynamics across cancer types
Comparative studies across different cancer types can uncover cancer-specific and universal aspects of ACD dysregulation, potentially leading to broad-spectrum or tailored therapeutic approaches.
Clinical translation
Translating findings from preclinical models to clinical applications requires systematic investigation, including the development of biomarkers for ACD activity and patient stratification for personalized therapies [40].
Single-cell multi-omics profiling of ACD heterogeneity and AI-driven data augmentation
Single-cell RNA sequencing (scRNA-seq) has become a cornerstone of cancer research [41], enabling high-resolution profiling of tumor heterogeneity and the elucidation of mechanisms like ACD in GBM stem-like cells. Leveraging paired scRNA-seq and single-cell ATAC-seq will enable concurrent mapping of transcriptomic states and chromatin accessibility in individual GBM stem-like cells undergoing SCD versus ACD. This integrative approach can identify gene regulatory networks and epigenetic modulators that govern division fate, reveal subpopulations of GSCs with distinct ACD profiles, and uncover correlations between ACD heterogeneity and microenvironmental cues [27]. Emerging spatial multi-omics platforms further allow these analyses to be performed within intact tumor architecture, enhancing translational relevance in patient-derived organoid and orthotopic xenograft models.
In addition to these mechanistic and translational priorities, emerging AI-driven strategies, such as generative adversarial networks (GANs), hold great promise for overcoming current data limitations in GBM ACD research. GANs can be trained on scarce RNA-seq datasets to produce realistic synthetic expression profiles, effectively generating intermediate phenotypic states that are rarely captured in patient samples and bypassing the ethical and privacy constraints of human data [42]. By integrating these synthetic profiles with genuine single-cell ATAC-seq and proteomic datasets, it becomes possible to train multi-modal GAN architectures that reveal the regulatory networks driving SCD versus ACD in GBM stem cells. By embedding established biological priors into the GAN training process and subsequently validating the generated samples in patient-derived organoid or orthotopic xenograft models, one can ensure both relevance and reproducibility. Incorporating GAN-based augmentation into our future GBM ACD studies thus offers a scalable, standardized framework for biomarker discovery, mechanistic hypothesis testing, and ultimately, the development of predictive signatures of treatment response.
Summary
ACD is fundamental to maintaining tissue homeostasis and cellular diversity [13]. Dysregulation of this process is implicated in GBM initiation, progression, and treatment resistance. A deeper understanding of ACD molecular underpinnings may facilitate the development of innovative diagnostic tools and therapeutic interventions, potentially improving outcomes in GBM. By prioritizing these research areas proposed in this paper, scientists can further uncover the critical roles of ACD in cancer biology and develop innovative strategies to combat malignancies more effectively.
Data availability statement
Not applicable.
Ethical statement
No direct interactions with human or animal subjects were involved. Therefore, ethical approval and informed consent were not required.
Author contributions
Genia Bekker and Rebecca Golin wrote the draft of the paper. Hengrui Liu edited the paper and supervised the project.
Funding
No funding or sponsorship was received for this study.
Acknowledgments
We thank Weifen Chen, Zongxiong Liu, Yaqi Yang, and Bryan Liu for their support.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
About the invited author Genia Bekker, MD, FACOG, FACS: Dr. Genia Bekker is the Fellow of the American College of Obstetricians and Gynecologists and the Fellow of the American College of Surgeons. Dr. Bekker provides comprehensive gynecological care to women in the Sheepshead Bay area of Brooklyn, New York. A board-certified OB/GYN, she is dedicated to delivering high-quality care to women at all stages of life with compassion and professionalism. With nearly 20 years of experience in gynecology, Dr. Bekker has authored multiple scientific publications and has presented her work at numerous professional meetings worldwide. She is a Fellow of both the American College of Obstetricians and Gynecologists and the American College of Surgeons. Her clinical expertise includes minimally invasive procedures, such as endometrial ablation and TVT™ for female urinary incontinence. Dr. Bekker remains highly active in both clinical and surgical practice, offering a full spectrum of women’s health services. Since founding Advanced Gynecology & Surgery in 2006 alongside Dr. Faina Akselrod, MD, FACOG, FACS, she has been committed to providing comprehensive obstetric and gynecologic care to women throughout her community.
References
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011;144(5):646-74. [PMID: 21376230 DOI: 10.1016/j.cell.2011.02.013]
- Chen R, Smith-Cohn M, Cohen AL, Colman H. Glioma subclassifications and their clinical significance. Neurotherapeutics 2017;14(2):284-97. [PMID: 28281173 DOI: 10.1007/s13311-017-0519-x]
- Wang LM, Englander ZK, Miller ML, Bruce JN. Malignant glioma. Adv Exp Med Biol 2023;1405:1-30. [PMID: 37452933 DOI: 10.1007/978-3-031-23705-8_1]
- Liu H, Hamaia SW, Dobson L, Weng J, Hernández FL, Beaudoin CA, et al. The voltage-gated sodium channel β3 subunit modulates C6 glioma cell motility independently of channel activity. Biochim Biophys Acta Mol Basis Dis 2025;1871(16):167844. [PMID: 40245999 DOI: 10.1016/j.bbadis.2025.167844]
- Hitomi M, Chumakova AP, Silver DJ, Knudsen AM, Pontius WD, Murphy S, et al. Asymmetric cell division promotes therapeutic resistance in glioblastoma stem cells. JCI Insight 2021;6(3):e130510. [PMID: 33351787 DOI: 10.1172/jci.insight.130510]
- Deng W, Lin H. Spectrosomes and fusomes anchor mitotic spindles during asymmetric germ cell divisions and facilitate the formation of a polarized microtubule array for oocyte specification in Drosophila. Dev Biol 1997;189(1):79-94. [PMID: 9281339 DOI: 10.1006/dbio.1997.8669]
- Daynac M, Petritsch CK. Regulation of asymmetric cell division in mammalian neural stem and cancer precursor cells. Results Probl Cell Differ 2017;61:375-99. [PMID: 28409314 DOI: 10.1007/978-3-319-53150-2_17]
- Samanta P, Bhowmik A, Biswas S, Sarkar R, Ghosh R, Pakhira S, et al. Therapeutic effectiveness of anticancer agents targeting different signaling molecules involved in asymmetric division of cancer stem cell. Stem Cell Rev Rep 2023;19(5):1283-306. [PMID: 36952080 DOI: 10.1007/s12015-023-10523-3]
- Wei M, Nurjanah U, Li J, Luo X, Hosea R, Li Y, et al. YY2-DRP1 axis regulates mitochondrial fission and determines cancer stem cell asymmetric division. Adv Sci 2023;10(23):e2207349. [PMID: 37300334 DOI: 10.1002/advs.202207349]
- Tamori S, Matsuda C, Kasai T, Ohno S, Sasaki K, Akimoto K. Asymmetric cell division of ALDH1-positive cancer stem cells generates glycolytic metabolically diverse cell populations. Sci Rep 2025;15(1):13932. [PMID: 40263471 DOI: 10.1038/s41598-025-97985-2]
- Gonzalez C. Centrosomes in asymmetric cell division. Curr Opin Struct Biol 2021;66:178-82. [PMID: 33279730 DOI: 10.1016/j.sbi.2020.10.023]
- Chen C, Yamashita YM. Centrosome-centric view of asymmetric stem cell division. Open Biol. 2021;11(1):200314. [PMID: 33435817 DOI: 10.1098/rsob.200314]
- Sunchu B, Cabernard C. Principles and mechanisms of asymmetric cell division. Development 2020;147(13):dev167650. [PMID: 32601056 DOI: 10.1242/dev.167650]
- Loeffler D, Wehling A, Schneiter F, Zhang Y, Müller-Bötticher N, Hoppe PS, et al. Asymmetric lysosome inheritance predicts activation of haematopoietic stem cells. Nature 2019;573(7774):426-9. [PMID: 31485073 DOI: 10.1038/s41586-019-1531-6]
- Venkei ZG, Yamashita YM. Emerging mechanisms of asymmetric stem cell division. J Cell Biol 2018;217(11):3785-95. [PMID: 30232100 DOI: 10.1083/jcb.201807037]
- Kochendoerfer AM, Modafferi F, Dunleavy EM. Centromere function in asymmetric cell division in Drosophila female and male germline stem cells. Open Biol 2021;11(11):210107. [PMID: 34727723 DOI: 10.1098/rsob.210107]
- Sonkin D, Thomas A, Teicher BA. Cancer treatments: past, present, and future. Cancer Genet 2024;286-287:18-24. [PMID: 38909530 DOI 10.1016/j.cancergen.2024.06.002]
- Li R, Xiao C, Liu H, Huang Y, Dilger JP, Lin J. Effects of local anesthetics on breast cancer cell viability and migration. BMC Cancer 2018;18(1):666. [PMID: 29914426 DOI: 10.1186/s12885-018-4576-2]
- Liu H, Dilger JP, Lin J. The role of transient receptor potential melastatin 7 (TRPM7) in cell viability: a potential target to suppress breast cancer cell cycle. Cancers 2020;12(1):131. [PMID: 31947967 DOI: 10.3390/cancers12010131]
- Liu H, Weng J. A comprehensive bioinformatic analysis of cyclin-dependent kinase 2 (CDK2) in glioma. Gene 2022;822:146325. [PMID: 35183683 DOI: 10.1016/j.gene.2022.146325]
- Liu H, Weng J. A pan-cancer bioinformatic analysis of RAD51 regarding the values for diagnosis, prognosis, and therapeutic prediction. Front Oncol 2022;12:858756. [PMID: 35359409 DOI: 10.3389/fonc.2022.858756]
- Liu H, Li Y. Potential roles of cornichon family AMPA receptor auxiliary protein 4 (CNIH4) in head and neck squamous cell carcinoma. Cancer Biomark 2022;35(4):439-50. [PMID: 36404537 DOI: 10.3233/CBM-220143]
- Li Y, Liu H. Clinical powers of aminoacyl tRNA synthetase complex interacting multifunctional protein 1 (AIMP1) for head-neck squamous cell carcinoma. Cancer Biomark 2022;34(3):359-74. [PMID: 35068446 DOI: 10.3233/CBM-210340]
- Buss JH, Begnini KR, Lenz G. The contribution of asymmetric cell division to phenotypic heterogeneity in cancer. J Cell Sci 2024;137(5):jcs261400. [PMID: 38334041 DOI: 10.1242/jcs.261400]
- Morrison SJ, Kimble J. Asymmetric and symmetric stem-cell divisions in development and cancer. Nature 2006;441(7097):1068-74. [PMID: 16810241 DOI: 10.1038/nature04956]
- Yan K, Wang M, Qiu Z, Xu M. A cell model about symmetric and asymmetric stem cell division. J Theor Biol 2023;560:111380. [PMID: 36509138 DOI: 10.1016/j.jtbi.2022.111380]
- Li Z, Zhang YY, Zhang H, Yang J, Chen Y, Lu H. Asymmetric cell division and tumor heterogeneity. Front Cell Dev Biol 2022;10:938685. [PMID: 35859890 DOI: 10.3389/fcell.2022.938685]
- Chartier NT, Hyenne V, Labbé JC. Mechanisms of asymmetric cell division: from model organisms to tumorigenesis. Med Sci (Paris) 2010;26(3):251-7. [PMID: 20346274 DOI: 10.1051/medsci/2010263251]
- Zhang S, Wu S, Yao R, Wei X, Ohlstein B, Guo Z. Eclosion muscles secrete ecdysteroids to initiate asymmetric intestinal stem cell division in Drosophila. Dev Cell 2024;59(1):125-40.e112. [PMID: 38096823 DOI: 10.1016/j.devcel.2023.11.016]
- Mukherjee S, Kong J, Brat DJ. Cancer stem cell division: when the rules of asymmetry are broken. Stem Cells Dev 2015;24(4):405-16. [PMID: 25382732 DOI: 10.1089/scd.2014.0442]
- Mukherjee S, Brat DJ. Molecular programs underlying asymmetric stem cell division and their disruption in malignancy. Results Probl Cell Differ 2017;61:401-21. [PMID: 28409315 DOI: 10.1007/978-3-319-53150-2_18]
- Ferron SR, Pozo N, Laguna A, Aranda S, Porlan E, Moreno M, et al. Regulated segregation of kinase Dyrk1A during asymmetric neural stem cell division is critical for EGFR-mediated biased signaling. Cell Stem Cell 2010;7(3):367-79. [PMID: 20804972 DOI: 10.1016/j.stem.2010.06.021]
- Wang YX, Feige P, Brun CE, Hekmatnejad B, Dumont NA, Renaud JM, et al. EGFR-Aurka signaling rescues polarity and regeneration defects in dystrophin-deficient muscle stem cells by increasing asymmetric divisions. Cell Stem Cell 2019;24(3):419-32.e416. [PMID: 30713094 DOI: 10.1016/j.stem.2019.01.002]
- Chan JR, Jolicoeur C, Yamauchi J, Elliott J, Fawcett JP, Ng BK, et al. The polarity protein Par-3 directly interacts with p75NTR to regulate myelination. Science 2006;314(5800):832-6.
- Koguchi M, Nakahara Y, Ito H, Wakamiya T, Yoshioka F, Ogata A, et al. BMP4 induces asymmetric cell division in human glioma stem-like cells. Oncol Lett 2020;19(2):1247-54. [PMID: 31966054 DOI: 10.3892/ol.2019.11231]
- Chen G, Kong J, Tucker-Burden C, Anand M, Rong Y, Rahman F, et al. Human Brat ortholog TRIM3 is a tumor suppressor that regulates asymmetric cell division in glioblastoma. Cancer Res 2014;74(16):4536-48. [PMID: 24947043 DOI: 10.1158/0008-5472.CAN-13-3703]
- Mukherjee S, Tucker-Burden C, Zhang C, Moberg K, Read R, Hadjipanayis C, et al. Drosophila Brat and human ortholog TRIM3 maintain stem cell equilibrium and suppress brain tumorigenesis by attenuating notch nuclear transport. Cancer Res 2016;76(8):2443-52. [PMID: 26893479 DOI: 10.1158/0008-5472.CAN-15-2299]
- Chen HF, Wu KJ. LncRNAs and asymmetric cell division: the epigenetic mechanisms. Biomed J 2024;48(2):100774. [PMID: 39059582 DOI: 10.1016/j.bj.2024.100774]
- Ma S, Wu J, Liu Z, He R, Wang Y, Liu L, et al. Quantitative characterization of cell physiological state based on dynamical cell mechanics for drug efficacy indication. J Pharm Anal 2023;13(4):388-402. [PMID: 37181289 DOI: 10.1016/j.jpha.2023.03.002]
- Goetz LH, Schork NJ. Personalized medicine: motivation, challenges, and progress. Fertil Steril 2018;109(6):952-63. [PMID: 29935653 DOI: 10.1016/j.fertnstert.2018.05.006]
- Liu H, Dong A, Rasteh AM, Wang P, Weng J. Identification of the novel exhausted T cell CD8 + markers in breast cancer. Sci Rep 2024;14(1):19142. [PMID: 39160211 DOI: 10.1038/s41598-024-70184-1]
- Ai X, Smith MC, Feltus FA. Generative adversarial networks applied to gene expression analysis: an interdisciplinary perspective. Comput Syst Oncol. 2023;3(3):e1050. [DOI: 10.1002/cso2.1050]
- Berika M, Elgayyar ME, El-Hashash AH. Asymmetric cell division of stem cells in the lung and other systems. Front Cell Dev Biol 2014;2:33. [PMID: 25364740 DOI: 10.3389/fcell.2014.00033]