Vitcylation of Lysine: The Novel Mechanism of Vitamin C in Tumor Treatment
1State Key Laboratory of Systems Medicine for Cancer, Shanghai Cancer Institute, Renji Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China
2Shanghai Key Laboratory for Cancer Systems Regulation and Clinical Translation, Shanghai Jiading District Central Hospital, Shanghai, China
3Department of Biliary-Pancreatic Surgery, Renji Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China
4School of Food and Biological Engineering, Xuzhou University of Technology, Xuzhou, China
5Department of Pharmacy, The First Affiliated Hospital of Guangxi Medical University, Nanning, China
6Department of Biology, Science Faculty, Selcuk University, Campus, Konya, Turkey
aThese authors contributed equally to this work.
*Correspondence to: Jin-Wei Zhou, PhD, Xuzhou University of Technology, Xuzhou, Jiangsu, China. E-mail: zhoujw86@126.com; Chen Li, PhD, Guangxi Medical University, Nanning, Guangxi, China. E-mail: chen.li.scholar@outlook.com; Gokhan Zengin, PhD, Selcuk Universitesi, Konya, Konya 42130, Turkey. E-mail: gokhanzengin@selcuk.edu.tr; Meng-Yao Li, PhD, Shanghai Jiao Tong University, 1630 Dongfang Road, Shanghai, China Shanghai 200240, China. E-mail: limy@sioc.ac.cn
Received: March 17 2025; Revised: April 15 2025; Accepted: April 29 2025; Published Online: May 21 2025
Cite this paper:
Li J, Gu A, Tang N et al. Vitcylation of Lysine: The Novel Mechanism of Vitamin C in Tumor Treatment. BIO Integration 2025; 6: 1–6.
DOI: 10.15212/bioi-2025-0057. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Vitamin C (vitC), an essential nutrient for human health, has critical roles in numerous biological processes. VitC’s potential in disease prevention and treatment is a focus of ongoing research. Recent clinical studies have highlighted its promising effects in combating various tumor diseases, thus spurring further exploration of its anti-tumor mechanisms. Historically, the known anti-tumor mechanisms of vitC have included redox regulation and epigenetic modifications. He et al. have now uncovered a novel pathway in which covalent modification directly targets immune signaling proteins (Cell, 2025, 188, 1858–1877. DOI: 10.1016/j.cell.2025.01.043). These findings, particularly lysine vitcylation, substantially change the understanding of vitC’s mechanistic landscape. This Commentary comprehensively analyzes He’s study elucidating the key mechanisms underlying vitC’s anti-tumor efficacy. Furthermore, it thoroughly examines the newly proposed mechanism and discusses potential implications for clinical applications as well as future challenges.
Keywords
Cancer therapy, Immune activation, Lysine vitcylation, Vitamin C.
This Commentary is based on the article “Lysine vitcylation is a vitamin C-derived protein modification that enhances STAT1-mediated immune response” (Cell, 2025, 188, 1858–1877. DOI: 10.1016/j.cell.2025.01.043) [1].
Introduction
Vitamin C (vitC) plays critical roles in numerous biological processes. Its potential therapeutic applications encompass metabolism, age-related diseases, and other associated biological processes [2, 3]. A substantial body of basic and clinical research underscores the major role of vitamin C in tumor therapy [4, 5]. However, its therapeutic potential, particularly at supraphysiological doses, has been extensively debated for decades. Initial research demonstrated the selective cytotoxicity of vitC in cancer cells via pro-oxidant hydrogen peroxide formation. Subsequent studies suggested its involvement in epigenetic regulation through ten-eleven translocation (TET) enzyme activation and destabilization of hypoxia-inducible factor (HIF) [6, 7]. Although vitC has shown promising preclinical effects in kirsten ratsarcoma viral oncogene homolog-mutant colorectal cancer and hematologic malignancies, its clinical translation has been impeded by inconsistent efficacy and unresolved mechanistic questions—particularly regarding how vitC influences immune regulation beyond these established pathways [8].
Recent advances have shown that vitC has dual roles in tumor biology: at low doses, it acts as an antioxidant, whereas at pharmacologic concentrations (0.1–20 mM), it has pro-oxidant and immunomodulatory effects. High-dose vitC enhances immunotherapy responses in murine models, but the molecular basis underlying this effect is unknown. Similarly, the mechanisms underlying the paradoxical effects of vitC on immune cells, namely the suppression of T cell inflammation in autoimmune settings and the promotion of dendritic cell development, are poorly understood [9, 10].
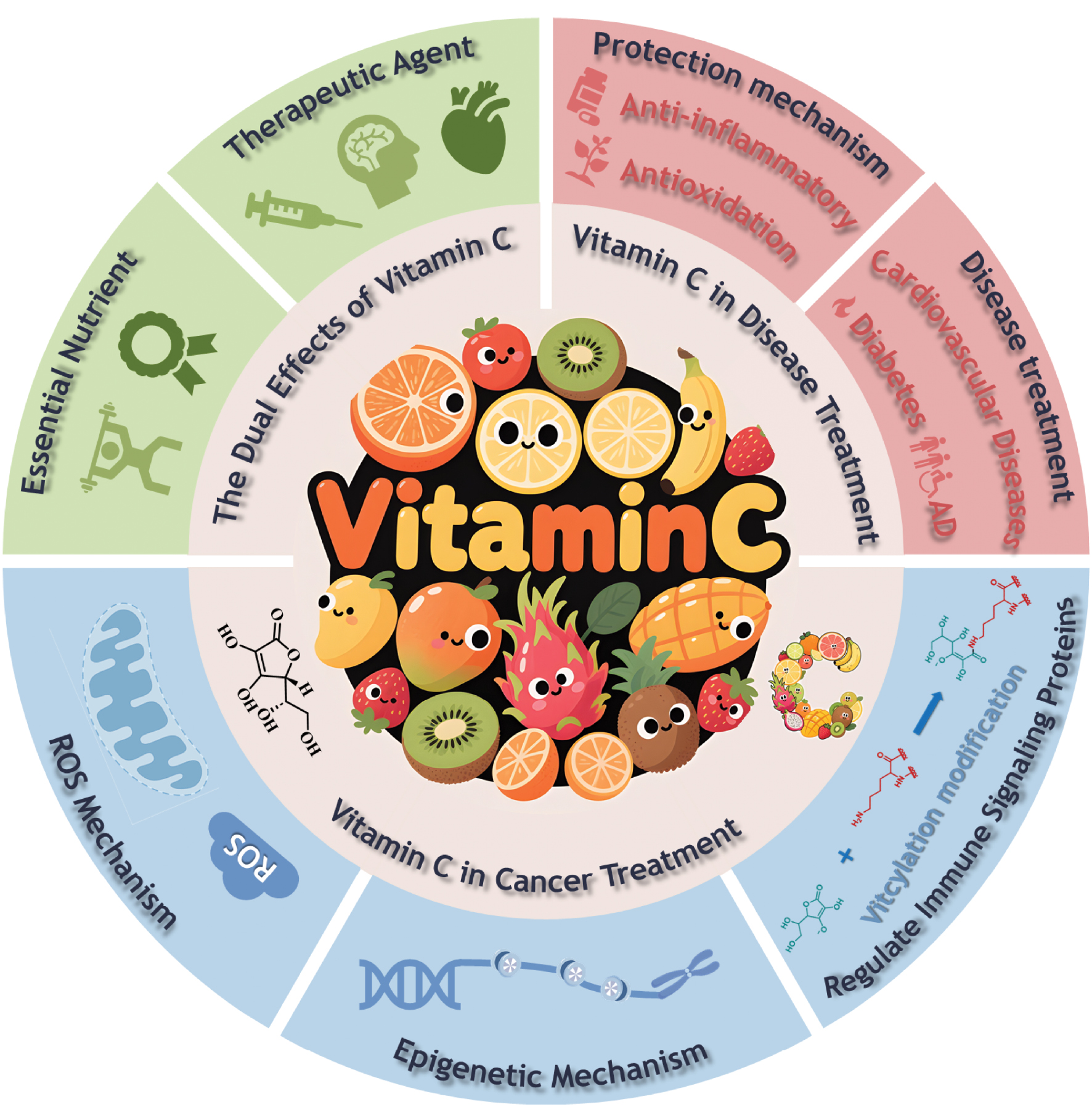
He et al.’s seminal study advances understanding of vitC’s mechanism by identifying lysine vitcylation—an a novel post-translational modification in which vitC directly modifies lysine residues via its lactone moiety [1]. Through a series of biochemical and functional studies, the authors have demonstrated that the vitcylation of STAT1 at lysine-298 (K298) maintains phosphorylation at tyrosine-701 (Y701) by inhibiting dephosphorylation by T cell protein-tyrosine phosphatase (TCPTP). This modification enhances interferon signaling, bolsters MHC-I-mediated antigen presentation, and facilitates CD8+ T cell-dependent tumor suppression. By linking vitC’s chemical reactivity to immune activation, this work resolves enduring controversies regarding vitC’s anti-cancer mechanisms and may pave the way to novel therapeutic approaches (Figure 1).
Figure 1 Benefits of vitamin C in human health.
Existing knowledge and gaps
Despite these well-characterized roles, the therapeutic application of high-dose vitamin C in oncology remains contentious. Preclinical models have indicated selective cytotoxicity of vitC via reactive oxygen species-dependent mechanisms in cancer cells, in which pharmacologic concentrations (1–20 mM) lead to production of extracellular hydrogen peroxide, thus taking advantage of the defective antioxidant machinery of malignant cells [11]. Pro-oxidant activity is particularly promising in KRAS-mutant colorectal cancer models; however, clinical trials have shown mixed results, with responses confined to certain genetic backgrounds, such as TET2-mutant acute myeloid leukemia. The discrepancy between preclinical and clinical results highlights the mechanistic complexity that remains to be resolved, particularly regarding the interactions between vitC and immune pathways.
Emerging evidence suggests that vitC may exert immunomodulatory effects through poorly defined mechanisms. For example, vitC increases dendritic cell maturation by facilitating TET2-mediated demethylation of immune gene promoters and subsequently enhancing antigen presentation. In addition, vitC polarizes naïve T cells into anti-tumor effector phenotypes, while suppressing pro-inflammatory responses in an autoimmune context, thereby indicating context-dependent modulation of the immune response. However, these effects have largely been associated with epigenetic or redox mechanisms, and whether vitC directly affects the post-translational signaling hubs required for immune activation remains unknown [12, 13].
Signal transducer and activator of transcription 1 (STAT1) is the master regulator of interferon signaling. Activation of STAT1 via tyrosine-701 (Y701) phosphorylation induces MHC-I expression and antigen presentation, thereby linking innate and adaptive immunity. STAT1 activity is tightly controlled by TCPTP, which dephosphorylates STAT1 at Y701, thereby avoiding STAT1 hyperactivation and autoimmune pathology. STAT1 dysregulation has been associated with chronic inflammatory diseases and cancer immune evasion. However, no previous studies have reported a direct association of vitC with either STAT1 post-translational modification or preventing dephosphorylation. This knowledge gap underscores a larger, more important point: although vitC has pleiotropic effects, its ability to directly regulate immune signaling proteins via covalent modifications, independently of ROS or epigenetic mechanisms, was previously unexplored [14].
He et al. [1] have directly addressed these gaps by uncovering lysine vitcylation as a novel modification driven by vitC. This discovery challenges the currently held paradigm regarding vitC’s indirect immune effects.
Pioneering discoveries: highlights from He et al. [1]
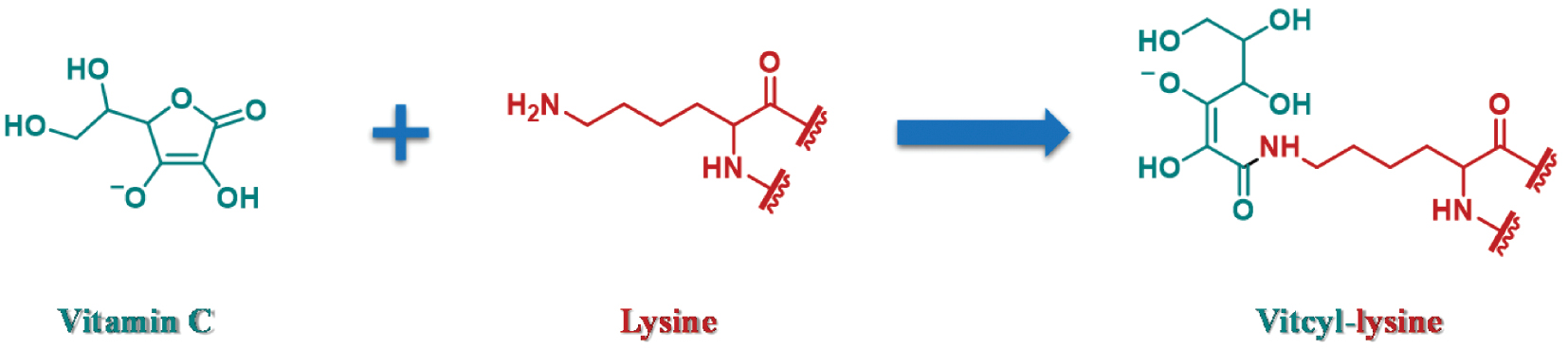
The study by He et al. [1] fundamentally alters the understanding of the mechanistic landscape of vitC, by identifying lysine vitcylation—a covalent modification in which the lactone moiety of ascorbate is directly conjugated to lysine residues through a nucleophilic substitution reaction (Figure 2). Through a variety of techniques, including mass spectrometry, isotopic labeling, and biochemical assays, the authors established that vitcylation occurs in a manner dependent on both pH and dose, and mitochondrial proteins exhibit enhanced modification levels, because of the alkaline microenvironment. This modification markedly differs from ascorbylation, in which a non-enzymatic adduct is formed by oxidized vitC derivatives, and it displays strict lysine specificity, as demonstrated through peptide substitution experiments in which arginine or cysteine residues were not modified.
Figure 2 Vitcylation of lysine.
The key finding of this study was that STAT1 is vitcylated at lysine-298 (K298), a conserved residue in the STAT1 coiled-coil domain. Structural modeling indicated that the steric hindrance created by vitcylation destabilizes the interaction between STAT1 and its phosphatase TCPTP, thus preventing STAT1 dephosphorylation. This mechanism was confirmed through a mutational analysis in which STAT1-K298R knock-in cells were unable to maintain the phosphorylation or activation of interferon-stimulated genes after treatment with vitamin C, whereas these phenotypes were rescued by wild-type STAT1. Vitcylation had no effect on the interaction between STAT1 and its kinase Janus kinase 1, thus confirming that the effect of vitcylation was selective in the regulation of phosphatase and was not due to upstream activation.
In preclinical models, vitC-induced STAT1 vitcylation was demonstrated to confer potent anti-tumor immunity. In syngeneic mouse tumor models (E0771 and PP breast cancer), administration of high-dose vitC (4 g/kg/day intraperitoneally) increased infiltration of CD8+ T cells and activation of dendritic cells within tumors, whereas these effects were not observed in STAT1-null or K298R-mutant tumors. Investigation of the underlying mechanism revealed that vitcylated STAT1 augments MHC-I antigen presentation through the upregulation of Tap1 and B2m, thereby enhancing recognition by cytotoxic T lymphocytes. These results were further validated in human cancer cell lines (Cal-51, MCF7), in which vitC treatment increased HLA class I expression and tumor sensitivity to PD-1 checkpoint blockade in co-culture assays.
Importantly, the effects of vitC on STAT1 vitcylation and immune activation were not subject to regulation by ROS and TET enzymes. Although vitC can produce ROS at supraphysiological doses (> 2 mM), both STAT1 vitcylation and immune activation were found to be independent of oxidative stress in rescue experiments with TET2 or HIF1α knockout, thus indicating that STAT1 vitcylation is not merely a consequence of vitC’s redox or epigenetic effects but instead is a primary mechanism underlying the immunomodulatory activity of vitC.
Mechanistic implications
He et al.’s [1] lysine vitcylation findings provide a novel understanding of vitC’s action, by uncovering a third axis independent of the canonical redox or epigenetic effects. The authors demonstrated that vitcylation directly affects STAT1 and consequently boosts immune signaling—without a need for TET2-mediated DNA demethylation or ROS production—thereby answering previously unresolved questions in vitC biology. This conclusion was further supported by experiments indicating that TET2 and HIF1α knockout cells still showed STAT1 phosphorylation and MHC-I upregulation after vitC treatment, thus definitively indicating that vitcylation is independent of these pathways. This independence suggests that vitcylation is a key mechanism underlying vitC’s immunomodulatory effects, particularly in scenarios in which epigenetic or oxidative stress mechanisms are inactive or saturated.
Structurally, vitcylation recapitulates the functional effects of STAT1 gain-of-function mutations seen in chronic mucocutaneous candidiasis. The STAT1-K298N mutation, for example, destabilizes TCPTP binding by altering the electrostatic interactions with E281/E284 in the antiparallel dimer conformation, thus resulting in hyperphosphorylation and autoimmune pathology. Molecular dynamics simulations by He et al. [1] have indicated that vitcylated K298 also destabilizes these salt bridges, thereby mimicking the K298N phenotype in a transient, pharmacologically tunable manner. Consequently, vitcylation might potentially be used to achieve controlled immune potentiation without the chronic hyperactivation observed in genetic disorders.
The pH dependence of vitcylation suggests additional compartmentalized regulatory possibilities. Mitochondria contain an alkaline matrix (pH ~8.0) and were found to have greater protein vitcylation than the cytosol (pH ~7.2) in He’s study. These results are consistent with previous findings indicating that mitochondrial proteins are often lysine-modified (e.g., through acetylation or succinylation) because of their distinct pH and redox environment [15]. The mitochondrial vitcylation preference suggests interesting possibilities for metabolic-immune communication in cancers dependent on oxidative phosphorylation or fatty acid oxidation—processes enriched in mitochondrial enzymes.
Finally, the proteomic data hint at potential effects extending far beyond STAT1. In murine cells, more than 1,400 vitaminylated proteins were identified, including metabolic enzymes (e.g., GAPDH and IDH1) and DNA repair factors (e.g., PARP1). The functional importance of these modifications remains to be determined, but the findings suggest the existence of a widespread regulatory network in which vitC controls diverse cellular processes through covalent lysine modifications. This finding might broaden the therapeutic potential of vitC to metabolic and aging-related diseases, in which protein dysregulation is a key feature [16].
Lysine residues subjected to post-translational modifications have essential functions in multiple biological processes. VitC plays a critical role in protein modification, particularly in the reversible post-translational modification of lysine residues. VitC also regulates histone methylation status by modulating the activity of histone demethylases, thereby controlling gene expression. For instance, vitC specifically removes H3K9me2 marks in embryonic stem cells through Kdm3a/b-mediated demethylation, and consequently influences gene expression patterns. Moreover, vitC has been implicated in DNA modification processes. In green algae, vitC serves as a co-substrate for the CMD1 enzyme, which catalyzes the conversion of 5-methylcytosine to a novel DNA modification. This modification is essential for photosynthesis and metabolic regulation [17]. Lysine propionylation is a prevalent post-translational modification in plants, and vitC may modulate photosynthesis and metabolic pathways by regulating this modification. Additionally, lysine methylation and acetylation are critical post-translational modifications, and vitC may influence nervous system development and disease progression by affecting dynamic changes in these modifications.
Clinical translation: opportunities and challenges
The results from He et al. [1] provide a strong rationale for the use of high-dose vitC as an immune adjuvant in cancer, by linking vitcylation to STAT1-mediated antigen presentation. The findings provide a potential mechanism underlying the clinical observations of enhanced checkpoint inhibitor efficacy by vitC in preclinical models. Magrì et al. have shown that vitC works in concert with anti-PD1 therapy in murine melanoma, thereby increasing tumor-infiltrating CD8+ T cells 2.5-fold [10]. He et al. [1] add to this model by demonstrating that STAT1 vitcylation is required for synergy, because the STAT1-K298R mutation abrogates both MHC-I upregulation and T cell activation. VitC might therefore be used in combination with immunotherapies focused on PD-1/PD-L1 or CTLA-4 in tumors in which STAT1 signaling is intact but antigen presentation is deficient [18].
Considerable pharmacokinetic challenges remain in achieving therapeutic vitcylation in humans. Murine models with intraperitoneal doses of 4 g/kg/day achieved plasma concentrations of ~20 mM. In contrast, human trials with intravenous administration of 100 g/day have shown peak plasma levels of only 2–5 mM [19]. These studies illustrate the need for better delivery methods, such as nanoparticle encapsulation or intratumoral injection, to increase tumor-selective accumulation and limit systemic exposure. Interestingly, He et al. [1] found higher vitC uptake in tumor tissue than in the spleen or plasma, owing to the Warburg effect-induced expression of sodium-dependent vitC transporters (SVCT2) on malignant cells. Exploiting this metabolic weakness might help enhance specificity.
Safety considerations add an additional layer of complexity to clinical translation. Although high-dose vitC is typically well tolerated, case reports have indicated a risk of oxalate nephropathy in patients who have renal insufficiency and hemolysis, and are deficient in glucose-6-phosphate dehydrogenase (G6PD). He et al. [1] suggest that the pH dependence of vitcylation might result in fewer off-target effects in normal tissues (which have a neutral pH) than tumors (which have an acidic microenvironment); however, this hypothesis must be tested in human trials. Moreover, the large-scale proteomic reach of vitcylation—extending to more than 1,400 identified modified proteins—is cause for concern regarding unintended effects, such as the dysregulation of metabolic enzymes or DNA repair machinery.
Patient stratification is a key challenge remaining to be addressed. Although He et al. [1] used STAT1-proficient models, clinical responses to vitC are expected to be heterogeneous. For instance, tumors containing STAT1 mutations or epigenetically silenced interferon pathways might be resistant to treatment, whereas those with intact JAK-STAT signaling might respond. Biomarkers such as STAT1 phosphorylation status, MHC-I expression, or TAP1/B2M transcript levels might aid in the selection of patients for this approach, as suggested in previous work combining vitC and demethylating agents. In addition, examining the potential of combining vitcylation and other existing therapies, such as PARP inhibitors, that target vitcylated DNA repair proteins, should prove interesting.
Conclusion
He et al.’s [1] findings regarding lysine vitcylation re-establish vitC as a direct modulator of protein function, thus linking its roles as an essential nutrient and therapeutic agent. The study clarifies how ascorbate covalently modifies STAT1 and consequently increases interferon signaling and anti-tumor immunity. These findings resolve longstanding ambiguities concerning the mechanisms underlying high-dose vitC’s action, and indicate their independence from ROS generation or TET2-mediated epigenetics. This paradigmatic shift parallels earlier advances in the understanding of ACE2’s dual role in viral entry and metabolic regulation, wherein a single molecule presides over diverse biological processes through distinct structural interactions.
Vitcylation provides a tunable approach to enhance immune checkpoint therapy while circumventing the chronic hyperactivation associated with STAT1 gain-of-function mutations. However, to realize this potential, pharmacokinetic limitations and safety challenges must be overcome, particularly the risk of off-target vitcylation in normal tissues. Future investigations should encompass combination therapy strategies integrated with chemotherapy or radiotherapy, as well as the delivery of vitC combined with various drugs via nano-drug-carrying particles within a novel delivery system.
In addition, future research should focus on biomarker-driven clinical trials and mechanistic investigation of vitC’s proteomic landscape. Harnessing the metabolic specificity and pH-dependent activity of vitC should enable its potential to be achieved as a safe, cost-effective adjuvant in oncology and other fields. The findings might herald a new era in which the intersection of nutrient biology and precision medicine is used to address human diseases. These findings should provide valuable insights for future studies, particularly in exploring the role of vitC in modulating other signaling pathways beyond the STAT1 pathway in the molecular mechanism through which vitcylation enhances anti-tumor immunity. Specifically, future studies should investigate whether other members of the STAT family are regulated by vitcylation. Given that prior studies have indicated that STAT3 activation may contribute to immunosuppression and thereby increase tumor risk, studying vitcylation’s regulation of other signaling pathways, including STAT3, is a promising and noteworthy research direction.
Declaration of competing interests
The authors declare that they have no conflicts of interest.
Author contributions
Jiatong Li and Meng-Yao Li conceived this work; Jiatong Li wrote the manuscript; Ao Gu and Nannan Tang reviewed the manuscript; Jin-Wei Zhou, Chen Li, Gokhan Zengin, and Meng-Yao Li revised the manuscript.
References
- He X, Wang Q, Cheng X, Wang W, Li Y, et al. Lysine vitcylation is a vitamin C-derived protein modification that enhances STAT1-mediated immune response. Cell 2025;188(7):1858-77 [PMID: 40023152 DOI: 10.1016/j.cell.2025.01.043]
- Wang Y, Dong L, Zhao M, Yin S, Da P, et al. Causal relationship between circulating vitamin C and 25-hydroxyvitamin D concentrations and common mental disorders – a Mendelian randomization study. Food Sci Human Well 2024;13(4):1877-85. [DOI: 10.26599/FSHW.2022.9250156]
- Hamdi E, Hidouri S, Muniz-Gonzalez A-B, Kechnebbou M, Amara S. Harnessing silica nanoparticles grafted with ascorbic acid to alleviate oxidative stress and impaired brain activity in rats. Nano TransMed 2025;4:100074. [DOI: 10.1016/j.ntm.2025.100074]
- Wang J, Liao ZX. Research progress of microrobots in tumor drug delivery. Food Med Homol 2024;1(2):9420025. [DOI: 10.26599/FMH.2024.9420025]
- Li MY, Gu A, Li J, Tang N, Matin M, et al. Exploring food and medicine homology: potential implications for cancer treatment innovations. Acta Materia Medica 2025;4(2):200-6. [DOI: 10.15212/amm-2025-0003]
- Agathocleous M, Meacham CE, Burgess RJ, Piskounova E, Zhao Z, et al. Ascorbate regulates haematopoietic stem cell function and leukaemogenesis. Nature 2017;549(7673):476-81. [DOI: 10.1038/nature23876]
- Cimmino L, Dolgalev I, Wang Y, Yoshimi A, Martin GH, et al. Restoration of TET2 function blocks aberrant self-renewal and leukemia progression. Cell 2017;170(6):1079-95. [PMID: 28823558 DOI: 10.1016/j.cell.2017.07.032]
- Yun J, Mullarky E, Lu C, Bosch KN, Kavalier A, et al. Vitamin C selectively kills KRAS and BRAF mutant colorectal cancer cells by targeting GAPDH. Science 2015;350(6266):1391-6. [PMID: 26541605 DOI: 10.1126/science.aaa5004]
- Ngo B, Van Riper JM, Cantley LC, Yun J. Targeting cancer vulnerabilities with high-dose vitamin C. Nat Rev Cancer 2019;19(5):271-82. [PMID: 30967651 DOI: 10.1038/s41568-019-0135-7]
- Magrì A, Germano G, Lorenzato A, Lamba S, Chilà R, et al. High-dose vitamin C enhances cancer immunotherapy. Sci Transl Med 2020;12(532):eaay8707. [PMID: 32102933 DOI: 10.1126/scitranslmed.aay8707]
- Chen Q, Espey MG, Krishna MC, Mitchell JB, Corpe CP, et al. Pharmacologic ascorbic acid concentrations selectively kill cancer cells: action as a pro-drug to deliver hydrogen peroxide to tissues. Proc Natl Acad Sci 2005;102:13604-609. [PMID: 16157892 DOI: 10.1073/pnas.0506390102]
- Alyatkin S, Sigurdsson H, Askitopoulos A, Töpfer JD, Lagoudakis PG. Quantum fluids of light in all-optical scatterer lattices. Nat Commun 2021;12(1):5571. [PMID: 34552069 DOI: 10.1038/s41467-021-25845-4]
- Huang M, Gong G, Deng Y, Long X, Long W, et al. Crosstalk between cancer cells and the nervous system. Med Adv 2023;1(3):173-89. [DOI: 10.1002/med4.27]
- Stark GR, Darnell JE Jr. The JAK-STAT pathway at twenty. Immunity 2012;36(4):503-14. [PMID: 22520844 DOI: 10.1016/j.immuni.2012.03.013]
- Weinert BT, Schölz C, Wagner SA, Iesmantavicius V, Su D, et al. Lysine succinylation is a frequently occurring modification in prokaryotes and eukaryotes and extensively overlaps with acetylation. Cell Rep 2013;4(4):842-51. [PMID: 23954790 DOI: 10.1016/j.celrep.2013.07.024]
- López-Otín C, Galluzzi L, Freije JMP, Madeo F, Kroemer G. Metabolic control of longevity. Cell 2016;166(4):802-21. [PMID: 27518560 DOI: 10.1016/j.cell.2016.07.031]
- Xue JH, Chen GD, Hao F, Chen H, Fang Z, et al. A vitamin-C-derived DNA modification catalysed by an algal TET homologue. Nature 2019;569(7757):581-5. [DOI: 10.1038/s41586-019-1160-0]
- Topalian SL, Drake CG, Pardoll DM. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell 2015;27(4):450-61. [PMID: 25858804 DOI: 10.1016/j.ccell.2015.03.001]
- Monti DA, Mitchell E, Bazzan AJ, Littman S, Zabrecky G, et al. Phase I evaluation of intravenous ascorbic acid in combination with gemcitabine and erlotinib in patients with metastatic pancreatic cancer. PLoS One 2012;7(1):e29794. [PMID: 22272248 DOI: 10.1371/journal.pone.0029794]