Recent Developments in Diabetes Management: Exploring Receptors, Pathways, and Compounds
Pallavi Pandey1, Vikash Jakhmola1,*
, Supriyo Saha1
and Anand Gaurav2
1Department of Pharmaceutical Chemistry, Uttaranchal Institute of Pharmaceutical Science, Uttaranchal University, Prem Nagar, Dehradun, Uttarakhand 248007, India
2Department of Pharmaceutical Sciences, School of Health Sciences and Technology, UPES, Dehradun 248007, Uttarakhand, India
*Correspondence to: Vikash Jakhmola, E-mail: jakhmola.1979@gmail.com
Received: November 18 2024; Revised: January 11 2025; Accepted: April 3 2025; Published Online: May 7 2025
Cite this paper:
Pandey P, Jakhmola V, Saha S et al. Recent Developments in Diabetes Management: Exploring Receptors, Pathways, and Compounds. BIO Integration 2025; 6: 1–28.
DOI: 10.15212/bioi-2024-0137. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Diabetes mellitus (DM) is a widespread metabolic disorder with profound effects on vital organs. Often referred to as a “systemic” or “multi-organ” disorder due to the impact on key organs, such as the kidneys, pancreas, eyes, and heart, DM is characterized by elevated blood glucose levels due to insufficient insulin production. DM consists of three subtypes (type 1 DM, type 2 DM, and gestational DM). Approximately 537 million adults are living with DM, a number predicted to increase to 643 million by 2030, according to the International Diabetes Federation (IDF) data. With the rising prevalence of DM, the Pan American Health Organization and World Health Organization estimate an age-standardized mortality rate of 20.9 deaths per 100,000 people. Therefore, it is important to study the prevention and cure of DM and determine the underlying mechanism and regulation. Numerous receptors and pathways help to regulate DM. This article delves into the intricate regulatory mechanisms underlying DM involving receptors and pathways, such as KATP (sulfonylurea), 5′AMP-activated protein kinase enzyme (biguanides), PPAR gamma, alpha-glucosidase, Glucose transporters (GLUT4), dipeptide peptidase-4 (DPP-4), and sodium-glucose co-transporters type 2 (SGLT2). The article highlights recent advances from 2020–2024 in synthesizing compounds targeting specific receptors for diabetes management. In so doing, insights into newly synthesized compounds, marketed drugs, phytoconstituents, and drugs currently in clinical trials will be provided. The role of each receptor in maintaining glucose homeostasis is reviewed in detail and various compounds with potential as effective antidiabetic agents are evaluated. This comprehensive review presents the pharmacologic mechanisms underlying these receptors and an in-depth analysis of promising new drug candidates.
Keywords
biguanides, DM, DPP-4, GLUT4, PPARγ, SGLT2, sulfonylureas.
Introduction
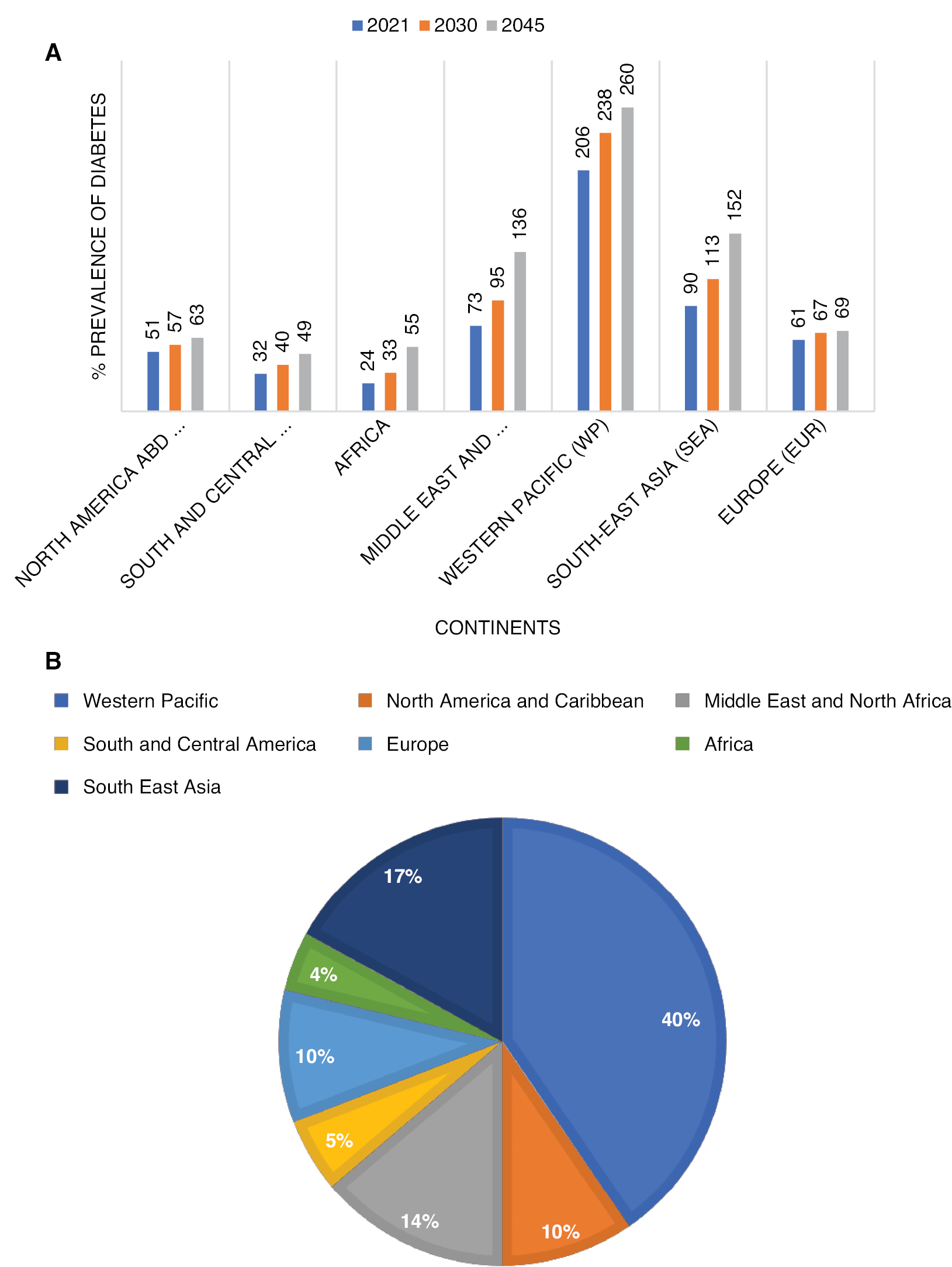
Diabetes mellitus (DM) is a widespread metabolic disorder in which glucose levels become too high, a condition known as hyperglycemia. DM develops when the pancreas fails to generate enough insulin or when cells develop insulin resistance [1, 2]. The word “mellitus” is a Latin word that resembles sweet, which means that the blood glucose concentration in the body is increased. According to data from the American Diabetes Association, a fasting blood glucose level < 116 mg/dL is considered normal. A fasting blood glucose level ≥ 126 mg/dL or a glucose level > 200 mg/dL in a randomly collected specimen is considered elevated [3]. If an elevated glucose level persists, chronic diabetes-related problems that impact numerous organs may occur, such as retinopathy, nephropathy, peripheral neuropathy, peripheral arterial disorders, and cerebrovascular disease [1]. The International Diabetes Federation (IDF) states estimated that 527 million adults between 20 and 79 years of age were living with DM in 2021. Moreover, DM is projected to affect 643 million people or 1 in 9 adults by 2030 and the number will increase to 784 million or 1 in 8 adults by 2045. Nearly 6.7 million fatalities were attributed to DM in 2021 [4, 5]. The IDF also published data on DM according to geographic region and worldwide (Figure 1A). In addition, the IDF has provided data on the rise in DM across continents, which is detailed in Figure 1B [5].
Figure 1 A) Predicted prevalence of diabetes in different continents by IDF. B) Percentage of increase in diabetes cases.
Diabetes is of three types based on the causes (type 1 DM, type 2 DM, and gestational DM). Type 1 DM, which is associated with autoimmune disease, primarily affects pancreatic cells, leading to a reduction or impairment in insulin synthesis [6]. There were 530,000 new cases of type 1 DM across all age groups, with 201,000 of these cases occurring in individuals < 20 years of age, according to IDF data from 2022. The IDF organized this data segmented by age group and incidence of DM [5]. Metabolic irregularities, such as hypertension, obesity, and dyslipidemia, are associated with type 2 DM and are risk factors for cardiovascular disease. Type 2 DM, also referred to as non-insulin-dependent DM, is characterized by pancreatic beta cell dysfunction, which impairs insulin use [7].
Gestational diabetes mellitus (GDM) is the third type of DM. GDM is characterized by any level of glucose intolerance detected during pregnancy. GDM complicates approximately 200,000 pregnancies annually (approximately 7% of all pregnancies). A fasting blood glucose level > 126 mg/dL or an average blood glucose concentration > 200 mg/dL meets the diagnostic criteria for GDM. When hyperglycemia of this magnitude is identified, the assessment of GDM should follow both one- and two-step methodologies. The one-step strategy involves administering an oral glucose tolerance test (OGTT) without initially evaluating plasma or serum glucose levels. In contrast, the two-step approach begins with an initial screening, during which plasma or serum glucose levels are measured 1 h after a 50-g oral glucose load. Subsequently, an OGTT is performed on the subgroup of women who exceed the glucose threshold value [8].
Another important factor that affects the prevalence of DM is lifestyle. Some food groups and dietary compounds, such as monounsaturated fatty acids, fruits, vegetables, dietary fibers, fish, magnesium, and nuts, have been shown to potentially offer protection against DM, possibly by improving insulin sensitivity and exerting anti-inflammatory effects. Conversely, consuming red and processed meats, as well as saturated fats, may increase the risk of developing type 2 DM [9]. In addition to lifestyle and gender, there are several other risk factors for DM, such as a lack of exercise, age, cigarette smoking, obesity, dyslipidemia, and a family history of DM [10].
This review focuses on recent advances (from 2020–2024) in synthesis of compounds targeting specific receptors for DM management. Furthermore, insights into newly synthesized compounds, marketed drugs, phytoconstituents, and drugs currently in clinical trials are provided.
Sulfonylureas
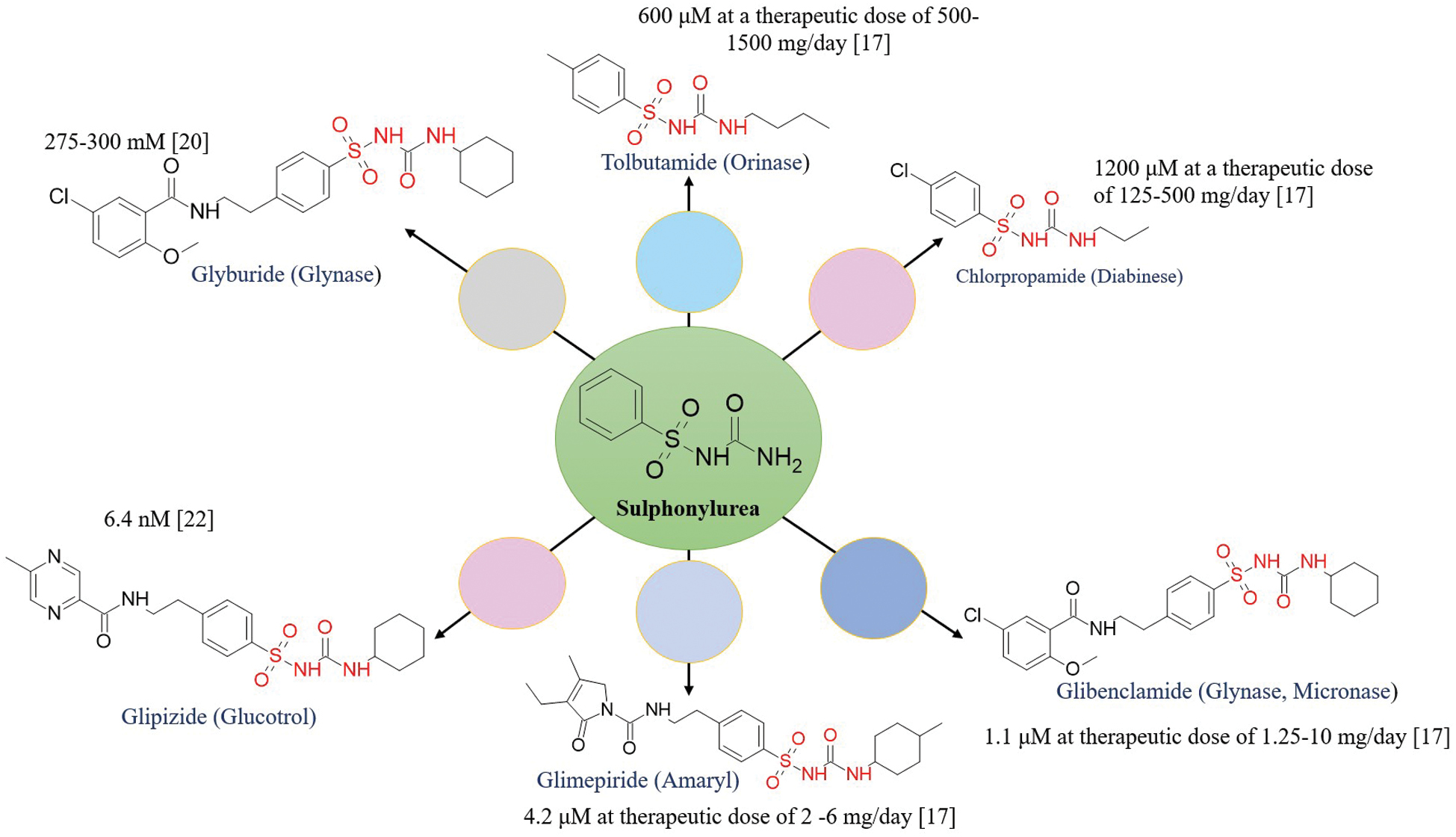
Sulfonylureas are a well-established class of antidiabetic drugs that are recommended for mono- or combination-therapy in management of DM. Sulfonylureas facilitate release of insulin from the beta-pancreatic cells, the details of which are provided below. However, recent studies have highlighted additional antidiabetic effects beyond β-cell stimulation. Sulfonylureas have been shown to inhibit peroxisome proliferator-activated receptor-γ (PPARγ) phosphorylation at Ser273 in primary human white adipocytes, leading to a favorable antidiabetic expression profile. This effect is characterized by upregulation of insulin-sensitizing adipokines and downregulation of adipokines associated with insulin resistance [11]. A detailed overview of available sulfonylurea drugs, along with the IC50 values, is presented in Figure 2, further illustrating the potency and pharmacologic profiles.
Figure 2 Marketed available sulfonylurea derivatives and IC50s.
Physiology of insulin release and role of sulfonylurea inhibitors
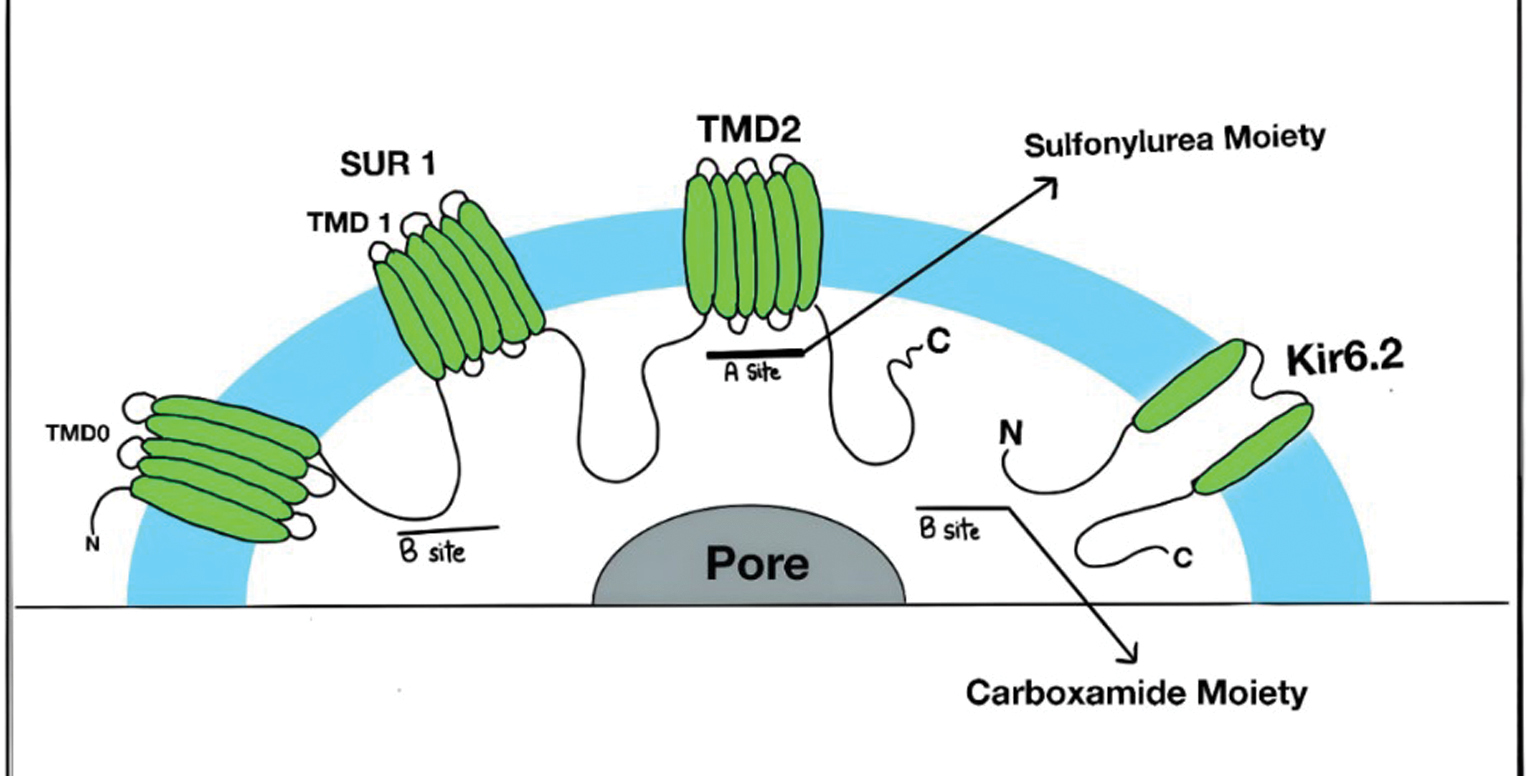
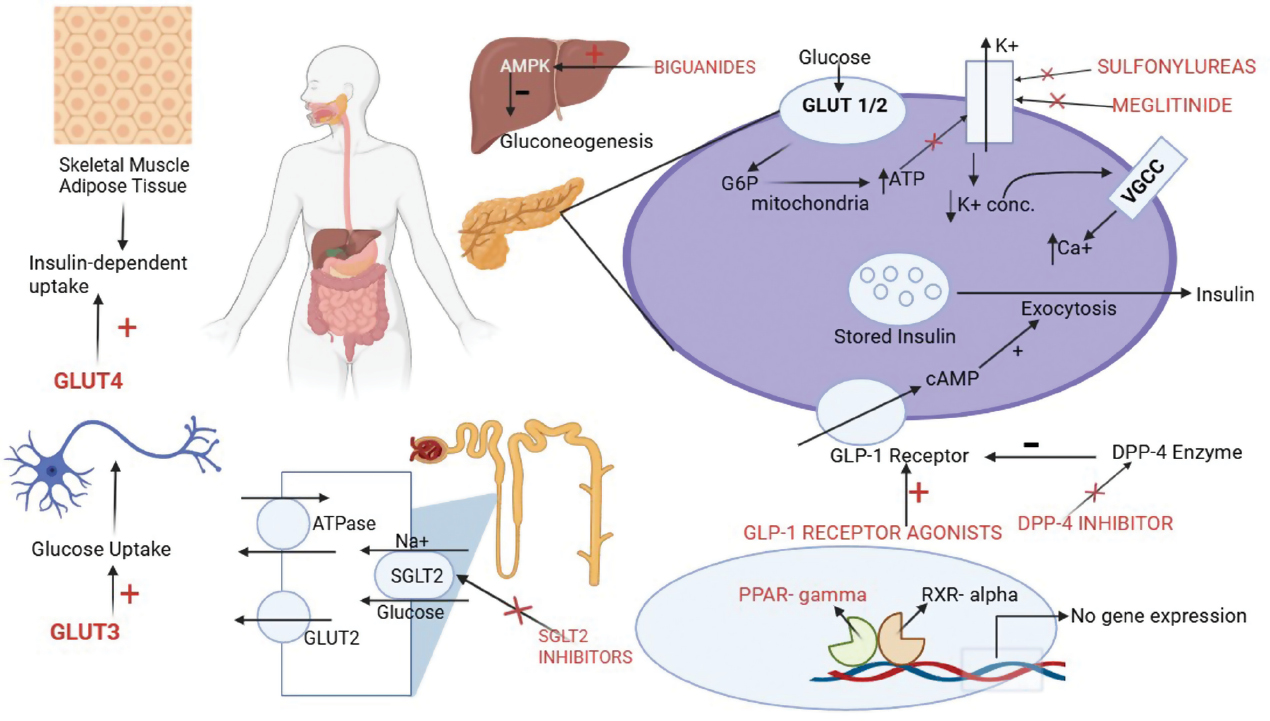
Pancreatic beta cells regulate insulin secretion through ATP-sensitive potassium (KATP) channels. KATP channels consist of Kir6.2 and SUR1 subunits, which respond to fluctuations in the cytosolic adenosine triphosphate (ATP): adenosine diphosphate (ADP) ratio. At low plasma glucose levels, ATP production decreases, keeping KATP channels open and allowing the efflux of potassium ions. This mechanism prevents calcium influx and suppresses insulin secretion. When the plasma glucose level rises, glucose metabolism increases the ATP:ADP ratio, leading to KATP channel closure. The resulting depolarization activates calcium channels, causing calcium-mediated exocytosis of insulin granules. The detailed molecular make-up of the KATP channel complex is shown in Figure 3. Mutations in KATP channel subunits, encoded by KCNJ11 and ABCC8 genes, significantly impact insulin secretion and glucose homeostasis. Loss-of-function mutations result in persistent hyper-insulinemic hypoglycemia due to excessive insulin release, while gain-of-function mutations reduce ATP sensitivity, leading to hyperglycemia and conditions, such as permanent neonatal diabetes mellitus and transient neonatal diabetes mellitus [12].
Figure 3 Molecular make-up of the KATP channel complex (transmembrane domain [TMD]) and binding site of sulfonylurea [12]. SUR: sulfonylurea receptor, TMD: transmembrane domain.
Sulfonylurea inhibitors are widely used in type 2 DM, target the SUR1 subunit of the KATP channel, and mimics ATP action to force channel closure. This process induces β-cell depolarization, calcium influx, and insulin secretion. First-generation sulfonylureas, such as tolbutamide, selectively bind to a single site on SUR1, while second-generation sulfonylureas, including glimepiride and glibenclamide, interact with multiple binding sites, which enhances potency. Additionally, meglitinides, which are structurally related to sulfonylureas, allosterically bind to regulate KATP channel activity with a rapid onset and short duration of action, making meglitinides effective for postprandial glucose control [13, 14].
Recent advances in sulfonylurea and combination therapy
Recent research highlights promising advances in sulfonylurea therapy, particularly when combined with other potent moieties. While traditional sulfonylureas, like gliclazide, effectively stimulate insulin release from pancreatic beta cells, traditional sulfonylureas carry the risk of hypoglycemia, especially at higher doses. To address these concerns, the study that focused on combining two potent moieties is detailed in Table 1 [15].
Table 1 Recently Synthesized Sulfonylureas with Combined Moieties
| S.No | Structure | Combination | Result | References |
|---|---|---|---|---|
| 1 | 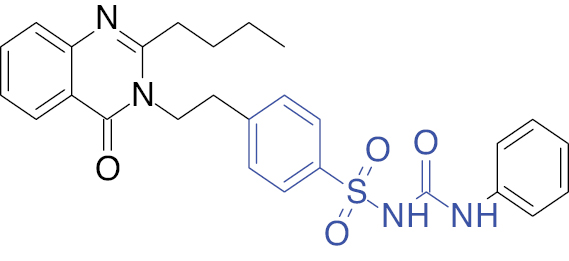 |
Quinazoline
|
Percentage of reduced blood glucose level in rats, 78.2% | [16] |
| 2 | 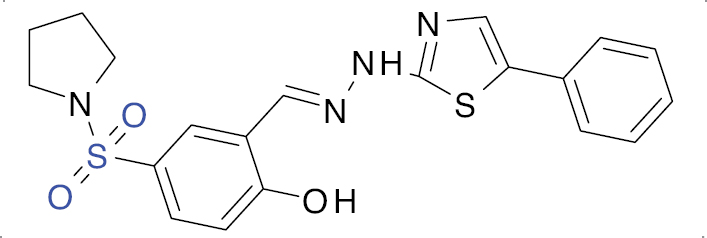 |
Thiazole
|
IC50 value, 3.30 μM (α-amylase) and 3.02 μM (α-glucosidase) | [17] |
| 3 | 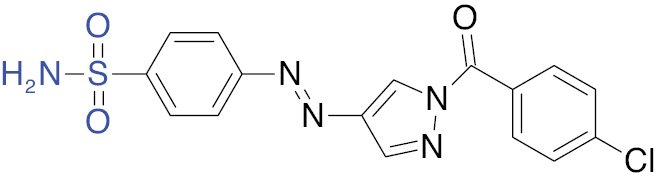 |
Pyrazole
|
IC50 value, 1.13 μM (α-glucosidase) | [18] |
| 4 | 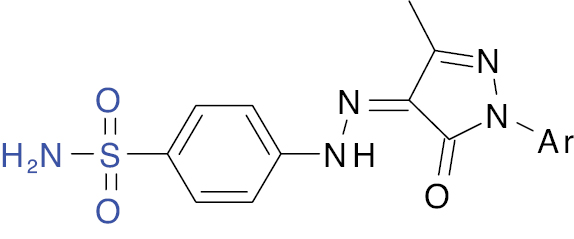 Ar: NO2C6H4 |
Pyrazole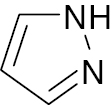 |
IC50 value, 25.53 μM | [19] |
| 5 | 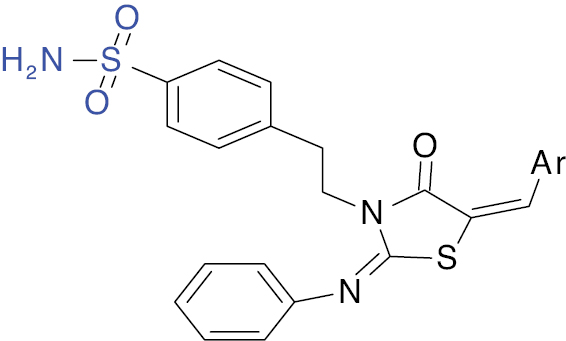 |
Thiazolidin-4-one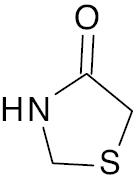 |
IC50 value, 29.51 μg/ml (α-amylase). | [20] |
| 6 | 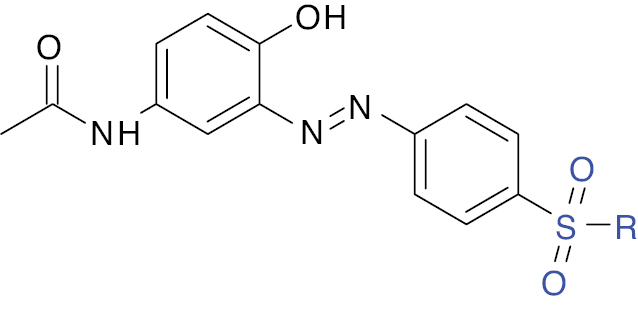 |
Paracetamol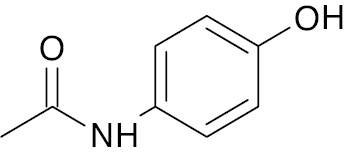 |
IC50 value, 1.55 μM (α-amylase) and 1.39 μM (α-glucosidase) | [21] |
Biguanides
Biguanides, a class of antidiabetic drugs with metformin as the primary agent, have been the cornerstone of type 2 DM management for decades due to efficacy, safety, and cost-effectiveness. Metformin features a guanidine core modified with methyl groups at the N1 and N2 positions. This structure contributes to the hydrophilic property, necessitating organic cation transporter 1 transporters for cellular uptake. Metformin exhibits slow absorption (peak plasma concentration, 1–3 h for immediate-release formulations), negligible plasma protein binding, and primary elimination through renal clearance [16, 22].
Recent studies (2023–2024) highlight the multifaceted mechanism underlying metformin beyond glycemic control, positioning metformin as a versatile agent with therapeutic potential in metabolic, cardiovascular, and inflammatory disorders. Metformin is known to inhibit mitochondrial complex I, which reduces hepatic gluconeogenesis by suppressing glycerol-3-phosphate shuttle activity. Metformin also modulates gut microbiota and lowers tumor necrosis factor-α, interleukin-6, and C-reactive protein levels, thereby mitigating chronic inflammation in obesity-driven diabetes. It has also been shown that metformin provides benefits beyond glycemic control by positively impacting macrovascular and microvascular outcomes. Metformin improves cardiovascular health by reducing insulin resistance, endothelial dysfunction, and oxidative stress, while inhibiting atherosclerotic plaque formation. Clinical evidence indicates a 30% lower risk of myocardial infarction in patients with type 2 DM using metformin compared to sulfonylureas [23, 24]. Additionally, metformin has a role in nephropathy by attenuating renal fibrosis through AMP-activated protein kinase-mediated suppression of transforming growth factor-β1 signaling. Clinical studies have shown that metformin slows the decline of eGFR by 1.2 mL/min/year in patients with chronic kidney disease stages 2–3. Metformin enhances mitochondrial function in peripheral nerves, which reduces oxidative damage and a corresponding 22% reduction in progression of neuropathy over 5 years [25, 26].
Furthermore, metformin has been linked to a reduced cancer risk by downregulating the mTOR pathway and insulin/IGF-1 signaling. Clinical evidence suggests a 31% lower risk of obesity-related cancers (e.g., breast and colorectal cancer) in patients using metformin [27].
Peroxisome proliferator activating receptor gamma
PPARγ is a nuclear receptor that regulates the expression of genes involved in glucose and lipid metabolism, adipogenesis, and inflammation. PPARγ has a pivotal role in enhancing insulin sensitivity by improving glucose uptake in peripheral tissues, particularly in adipocytes and muscle cells. Recent studies have increasingly focused on pan-PPAR agonists, compounds that simultaneously activate all three isoforms of peroxisome proliferator-activated receptors [PPARs] (PPAR-α, PPAR-β/δ, and PPAR-γ). These agonists hold significant potential in regulating pathways that contribute to antidiabetic effects. One of the key benefits is improving insulin sensitivity through activation of PPAR-γ is enhanced glucose uptake and utilization in peripheral tissues. Additionally, pan-PPAR agonists regulate lipid metabolism by activating PPAR-α, promoting fatty acid oxidation, and reducing triglyceride levels in the liver and plasma. These effects help alleviate hepatic steatosis and improve overall insulin action. Pan-PPAR agonists also have a critical role in reducing inflammation by modulating inflammatory responses through PPAR-γ and PPAR-β/δ. By decreasing the production of pro-inflammatory cytokines, pan-PPAR agonists create a more favorable inflammatory environment, which is often disrupted in conditions, like insulin resistance. Furthermore, activation of PPAR-β/δ enhances glucose metabolism by promoting the switch from glucose to fatty acid oxidation in skeletal muscle, thereby improving the overall metabolic profile. Another important mechanism is the regulation of hormonal signaling. Pan-PPAR agonists influence hormones involved in glucose homeostasis, such as adiponectin, which not only enhances insulin sensitivity but also exhibits anti-inflammatory properties [28, 29]. These multifaceted actions of pan-PPAR agonists make pan-PPAR agonists promising candidates for the development of novel therapies targeting metabolic disorders, including type 2 DM and associated complications. Chiglitazar (Bilessglu®) became the first pan-PPAR receptor agonist approved in China in October 2021 for the treatment of type 2 DM. This milestone highlights the growing interest in pan-PPAR agonists as a novel therapeutic approach for metabolic disorders [30]. In addition to chiglitazar, other compounds, such as Bezafibrate and E17241, also target the pan-PPAR receptor, further emphasizing the potential of this drug class in regulating metabolic pathways and improving outcomes in patients with type 2 DM [31].
Chemistry
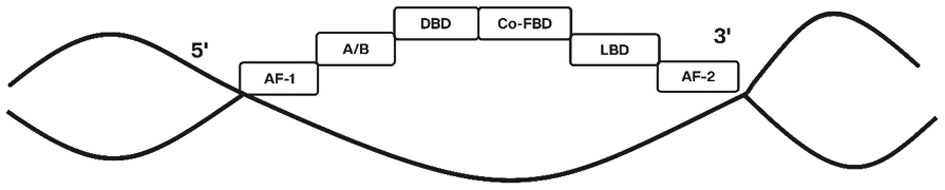
The nuclear receptor, PPARγ, is comprised of five domains (labeled A-E) that span from the N-terminus to the C-terminus. The N-terminal region includes the A and B domains, which feature the intrinsically disordered activated function (AF) 1 domain. This AF1 domain is involved in ligand-independent co-regulatory binding but lacks a major binding site due to the absence of a conserved amino acid sequence [32].
The C domain of PPARγ serves as the DNA-binding domain and is the most conserved region. The C domain of PPARγ contains two zinc-binding sites crucial for function, enabling the receptor to bind to specific DNA sequences and regulate gene transcription. The D domain acts as a hinge, providing structural flexibility and linking the DNA-binding domain to the ligand-binding domain, facilitating conformational changes upon ligand binding [33].
The E domain is responsible for ligand binding, accommodating both endogenous ligands, like fatty acids and synthetic drugs. The E domain also contains the AF2 region, which is essential for ligand-dependent activation of the receptor. Notably, the DNA-binding domain structure is quite similar to the ligand-binding domain, highlighting a conserved mechanism across these regions [34].
The F domain, located at the C-terminus, is less well-characterized but is believed to have a role in the interaction between co-regulators and other proteins. The overall structure of PPARγ enables PPARγ to effectively regulate gene expression involved in metabolism and the pharmacologic role (structure demonstrated in Figure 4) [24, 35].
Figure 4 Different binding domains of PPARγ. AF: activation function, DBD: DNA binding domain, C0-FBD: Co-activator binding, LBD: ligand-binding domain.
PPARγ consists of three subunits (a polar head, a linker, and a hydrophobic tail). The acidic head is a thiazolidinedione moiety, the linker is a benzyl group, and the hydrophilic tail is a pyridine in pioglitazone and rosiglitazone.
Mechanism of action
PPARγ is a nuclear receptor present in various cells, including adipose tissues, the colon, and macrophages. PPARγ is located in the nuclear membrane and the drugs within the class bind to PPARγ, activating PPARγ and causing PPARγ to dimerize with the retinoic X receptor (RXR). The dimer then interacts with DNA, influencing gene transcription and leading to protein synthesis. As a result, PPARγ agonists enhance protein synthesis and release, which increases insulin sensitivity (detailed in Table 2) [36].
Table 2 Recently Synthesized PPARγ and Its Combination Moiety
| S.No | Structure | Combination | Result | References |
|---|---|---|---|---|
| 1 | 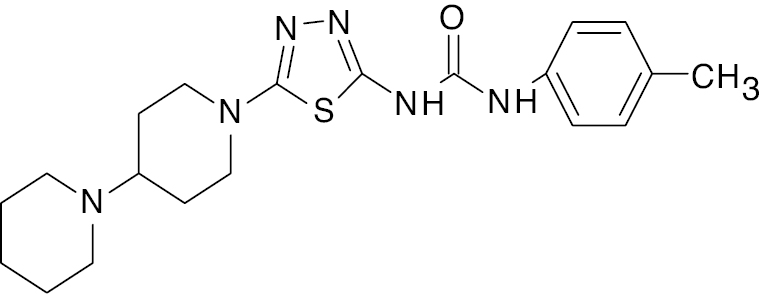 |
1, 3, 4-thiadiazole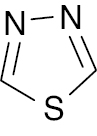 |
89.7% increase in blood glucose levels | [46] |
| 2 | 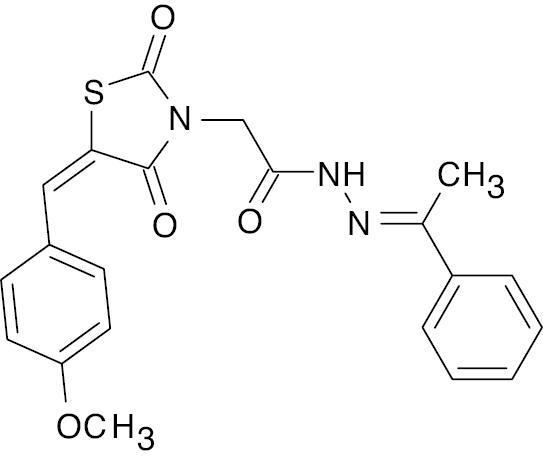 |
Thiazolidine- 2,4-dione |
Percentage change in fasting blood glucose level, 54.4±4.8 | [47] |
| 3 | 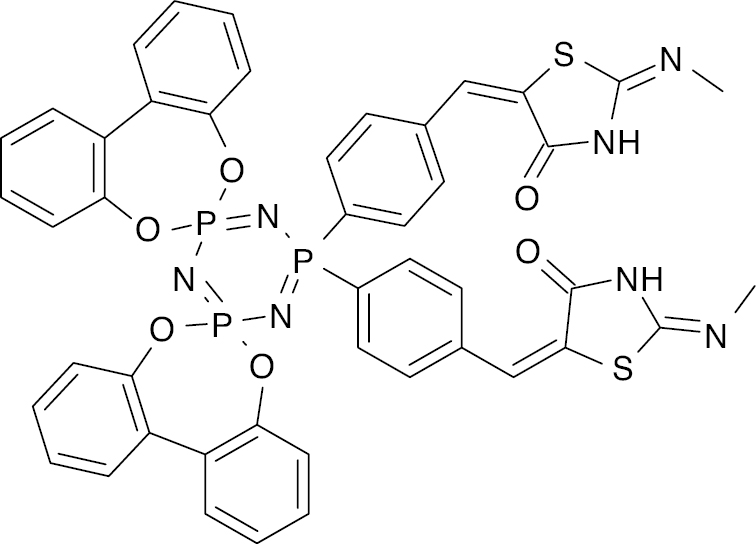 |
Phosphazene |
Higher glucose uptake compared to pioglitazone | [48] |
| 4 | 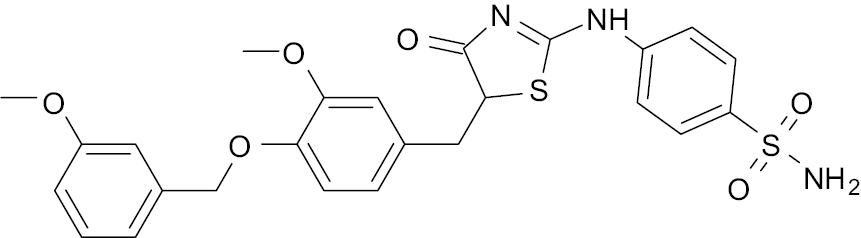 |
Benzenesulfonamide |
PPAR activation increased by 31.7% and 10-fold increase in blood insulin level and C-peptide level by 48.4%. | [49] |
| 5 | 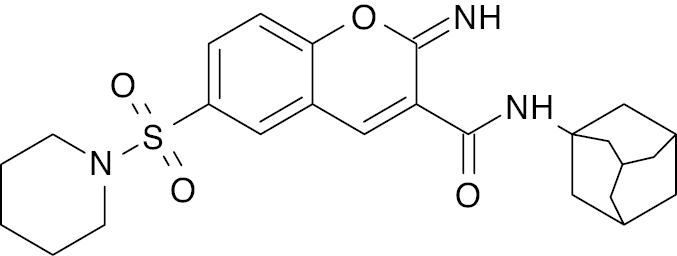 |
2H- chromene |
IC50 value, 4.653 μg/mL | [50] |
| 6 | 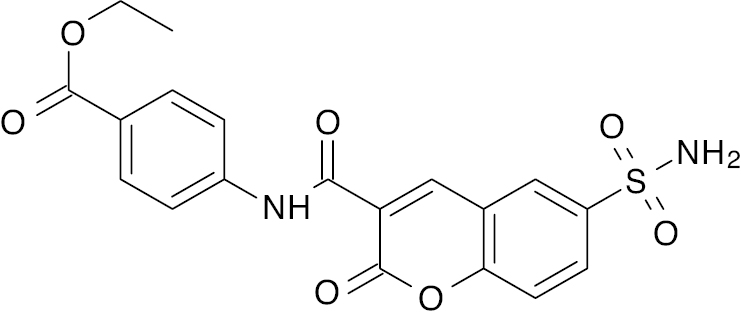 |
2H- Chromene-6-sulfonamide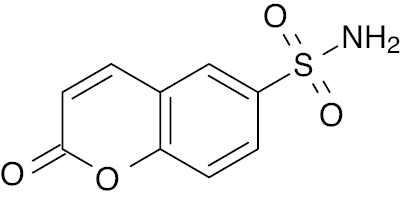 |
IC50 value, 3.706 μg/mL | [51] |
| 7 | 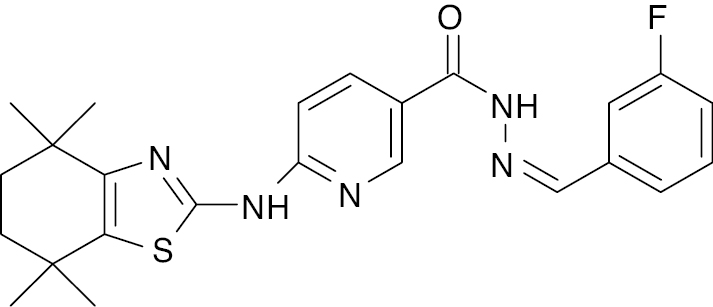 |
Thiazole- pyridine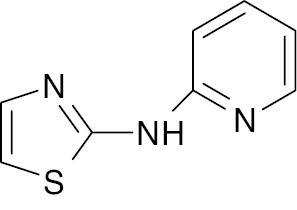 |
Reduction in blood glucose level from 226.8 ± 5.10 to 115.5 ± 3.09 mg/dL | [52] |
| 8 | 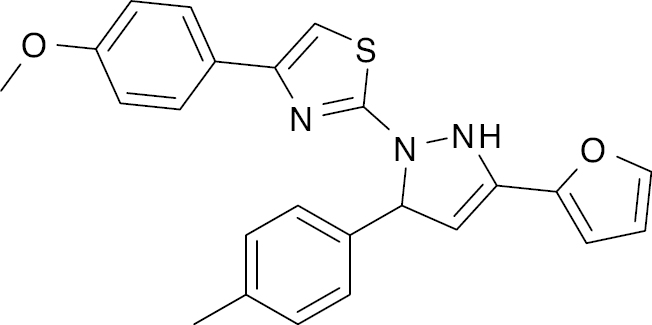 |
Thiazole |
PPARγ activation increase, 72%-79% | [53] |
| 9 | 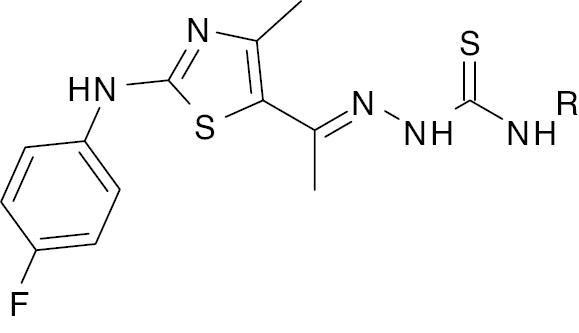 |
Thiazole-Thiosemicarbazones |
Thiazole- thiosemicarbazones derivative IC50 values, 0.938 ± 0.023 and 0.947 ± 0.024 ng/mL | [54]. |
| 10 | 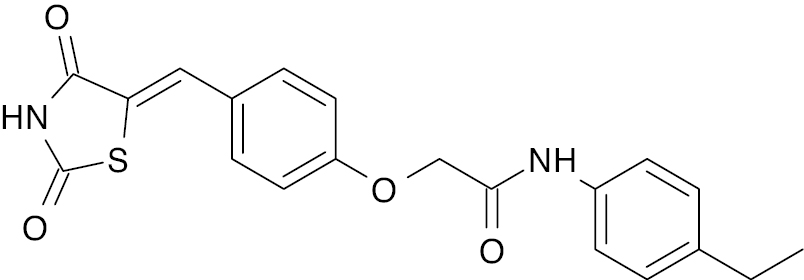 |
Thiazolidinediones |
Reduction in blood glucose level by 108.5 ± 2.171 mg/dL | [55] |
| 11 | 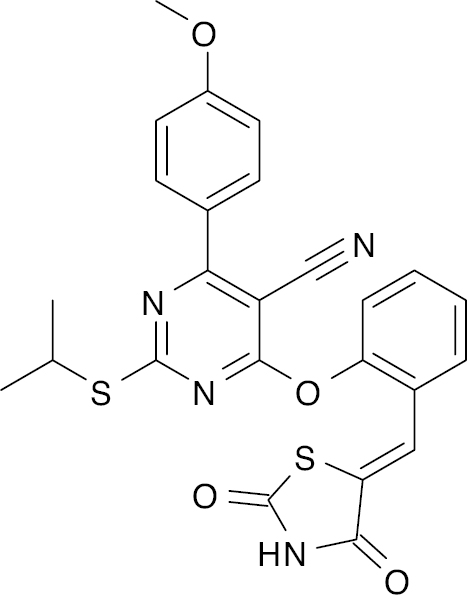 |
Pyrimidine |
Lower blood glucose level to 145.2 mg/dl | [56] |
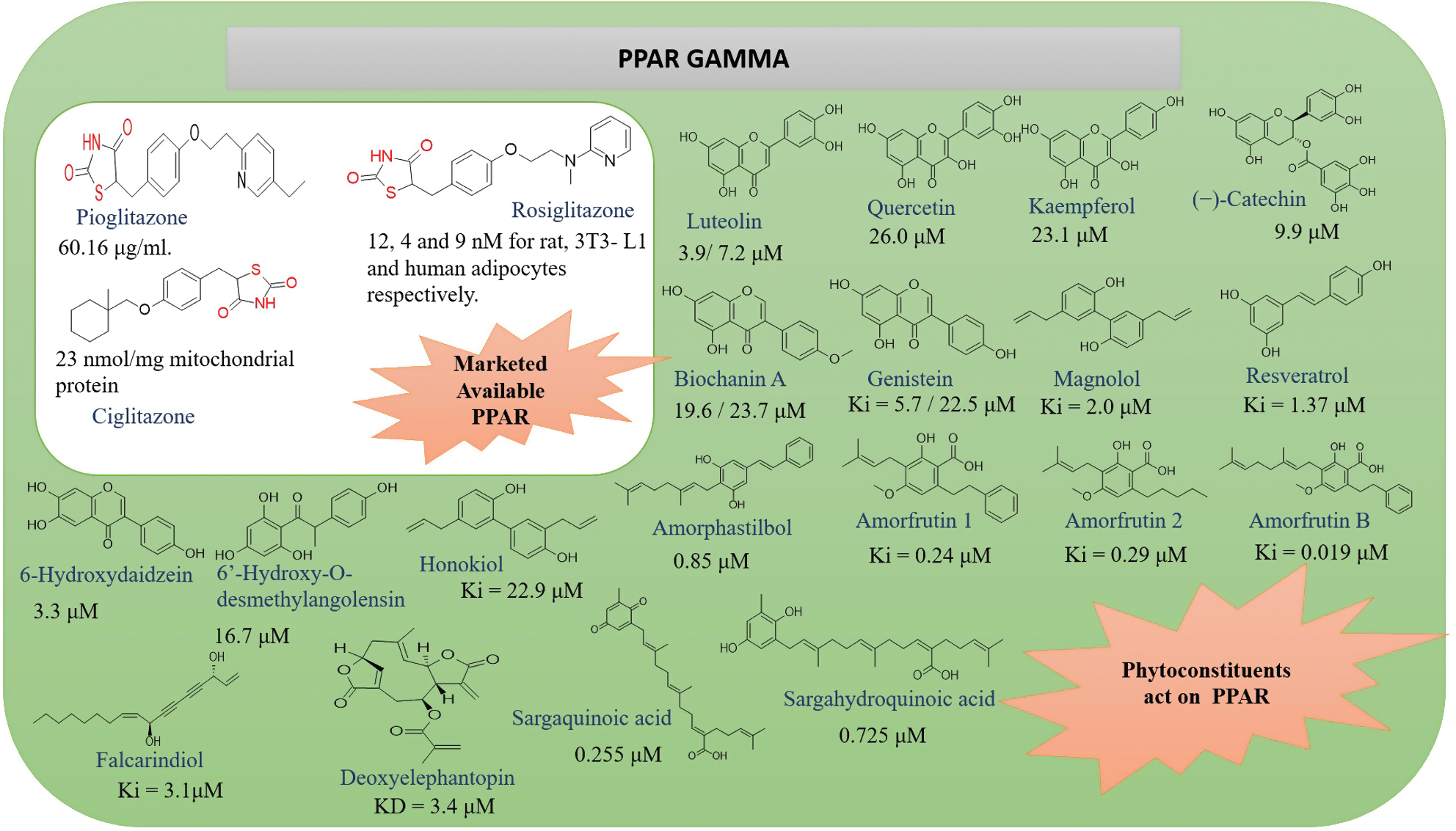
The phytoconstituents listed in Figure 5 have been identified as exhibiting anti-diabetic properties by acting on the PPARγ receptor [37–43].
Figure 5 Marketed available drugs, phytoconstituents, and IC50s.
Recent advances in PPARγ and combination therapy
Current research includes selective PPARγ modulation, such as INT131, which provides insulin-sensitizing effects without the adverse cardiovascular impact of traditional PPARγ agonists. A 2024 study reported that INT131-based combinations reduce HbA1c by 1.2% and LDL by 18% in patients with diabetics dyslipidemia. Additionally, novel dual PPARα/γ agonists (e.g., tesaglitazar and saroglitazar) demonstrate synergistic improvements in glycemic control, lipid metabolism, and insulin resistance. A 2024 meta-analysis of 20 randomized controlled trials (6058 patients) concluded that combining PPAR agonists with metformin reduces the fasting glucose level by 22.07 mg/dL (P < 0.001), the HbA1c by 0.53% (P < 0.001), and the HOMA-IR by 1.26 units (P = 0.006) with no significant increase in adverse events compared to metformin alone. Saroglitazar, a newer dual agonist, has also been shown to improve lipid profiles and reduce cardiovascular risks. Additionally, combination therapy (illustrated in Table 2) shows promise in amplifying glycemic control and addressing multiple pathways of metabolic dysfunction. This multi-faceted approach aims to improve long-term outcomes by leveraging the role of PPARγ in lipid and glucose metabolism [44, 45].
Alpha-glucosidase inhibitor
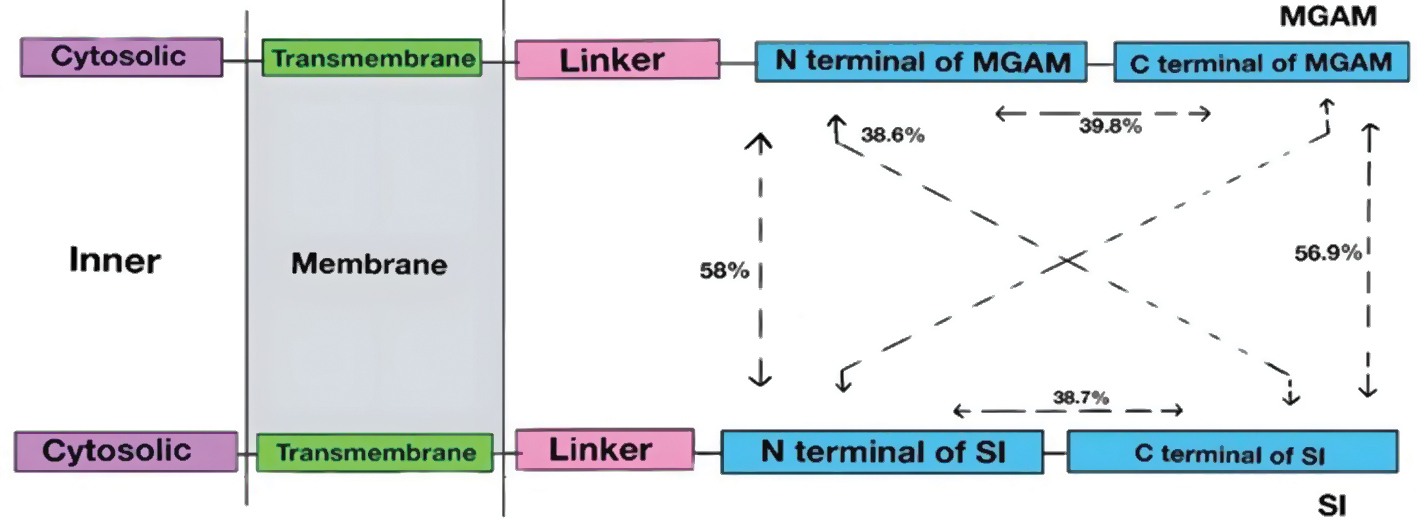
Alpha-glucosidase is an enzyme that belongs to the family of glycosyl hydrolases. There are two distinct forms of alpha-glucosidase in the gut (maltase-glucoamylase [MGAM] and sucrase-isomerase [SI]). Each domain of these enzymes catalyzes the hydrolysis of maltose at the alpha 1-,4-glycosidic bond (Figure 6) [57].
Figure 6 Human alpha-glucosidase and location of maltose-glucoamylase (MGAM) and sucrase-isomaltose (SI) situated within the intestinal brush membrane [58].
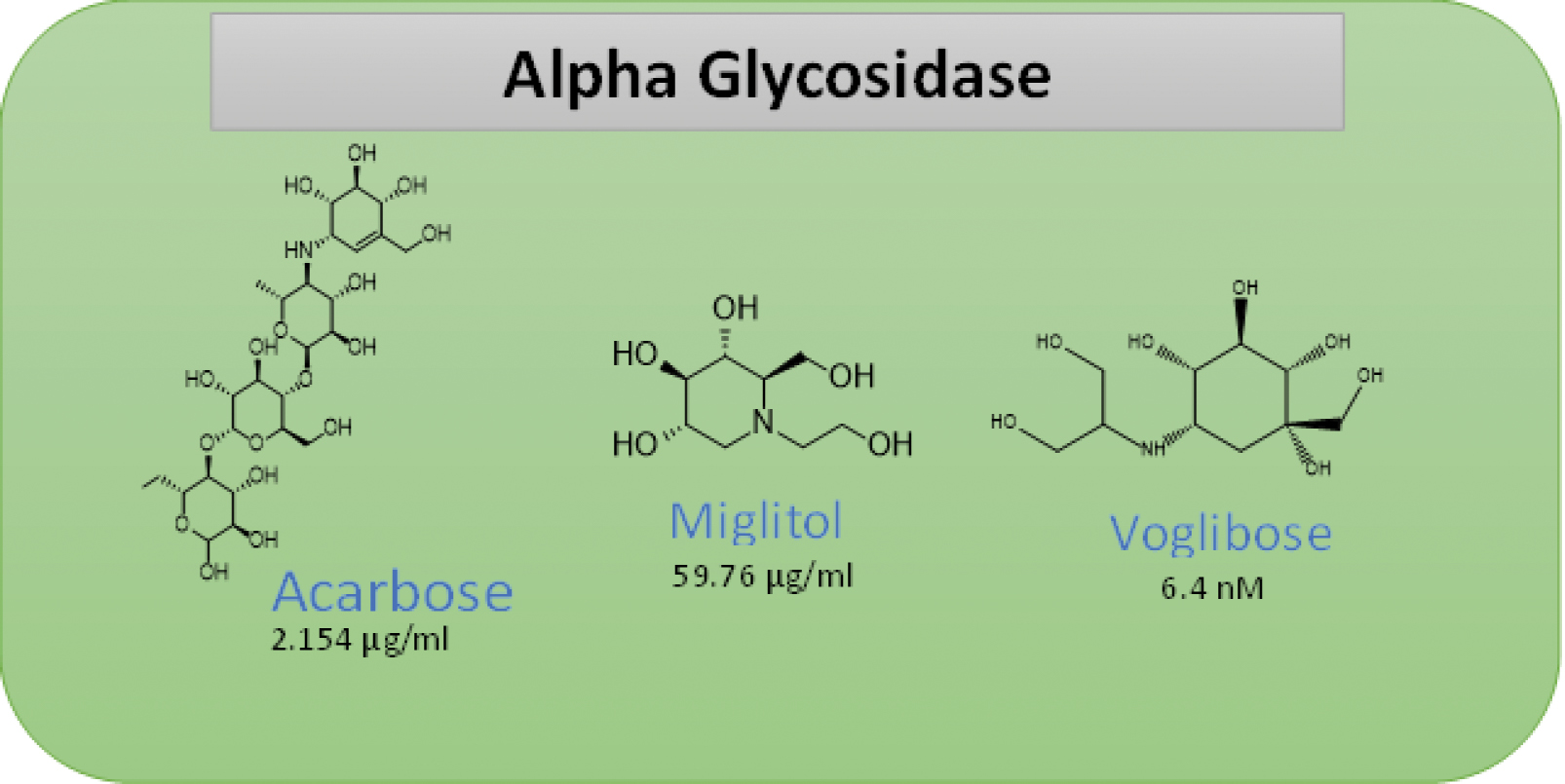
Alpha-glucosidase cleaves alpha-related D-glucose residues from the non-reducing end of polysaccharides and disaccharides, which hydrolyzes alpha-related D-glucose residents into glucose. Therefore, inhibiting alpha-glucosidase is one of the simplest strategies for controlling the postprandial blood glucose level. Acarbose and miglitol are prominent alpha-glucosidase inhibitors. Acarbose is a bacterial oligosaccharide that is an analog to a glucosidase substrate (acarbose and voglibose are amino sugars). In contrast, miglitol belongs to a structurally distinct class of compounds with a cyclohexane moiety (azasugar; Figure 7) [59].
Figure 7 Marketed available alpha glucoside drugs and IC50s.
Mechanism of action
Alpha-glucosidase inhibitors function by preventing the intestinal enzyme brush boundary alpha-glucosidase from functioning. Alpha-glucosidase usually cleaves large glucose molecules into smaller fragments for absorption. By inhibiting alpha-glucosidase, alpha-glucosidase inhibitors prevent the breakdown and subsequent absorption of glucose, inflicting glucose to remain inside the intestinal lumen. This effect leads to gastrointestinal side effects, such as bloating and gastric upset [7, 60].
Recent advances in alpha-glycosidase and combination therapy
Recent studies show that combining alpha-glucosidase inhibitors with antidiabetic medications and other moieties (Table 3) enhance glycemic control significantly by lowering HbA1c, fasting plasma glucose, and postprandial glucose levels, while reducing the side effects [61–63].
Table 3 Recently Synthesized Alpha Glycoside and the Combination Moiety
| S.No | Structure | Combination | Result | References |
|---|---|---|---|---|
| 1 | 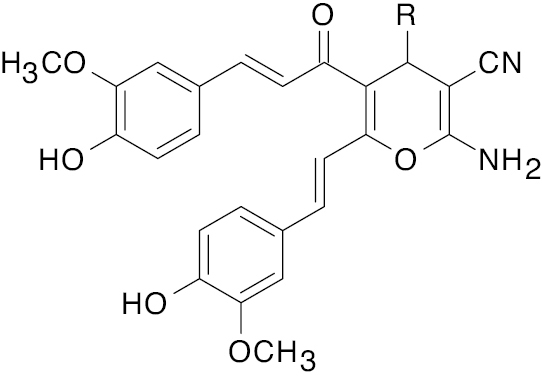 R= bis-functionalized |
Curcumin derivative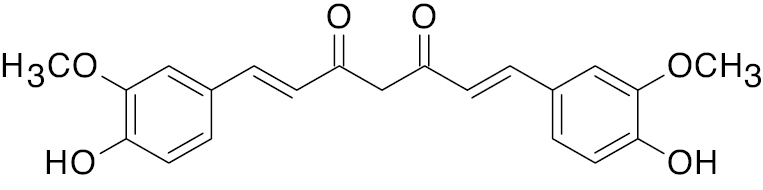 |
IC50, 3.01 μg | [64] |
| 2 | 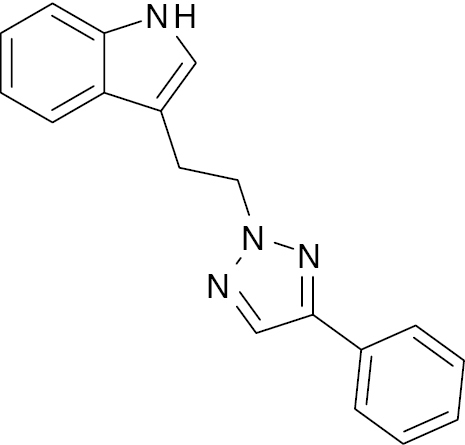 |
Triazole clubbed indole derivative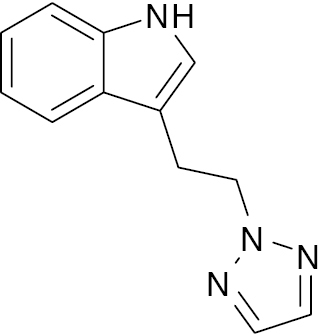 |
IC50, 10.1 μM | [65] |
| 3 | 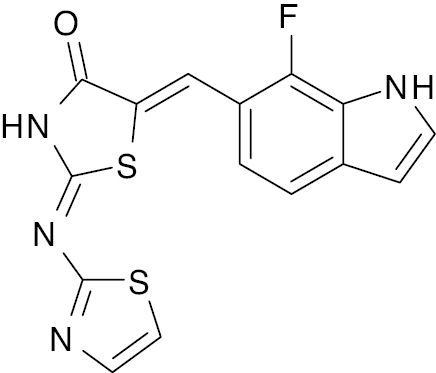 |
Thiazolidine |
IC50 values, alpha-amylase (1.5 μM) and alpha glycosidase (2.40 μM) | [66] |
| 4 | 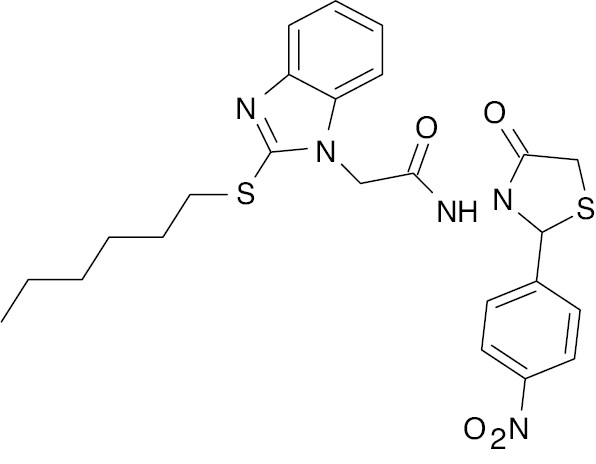 |
Mercaptobenzimidazole |
IC50, 5.22 μM | [67] |
| 5 | 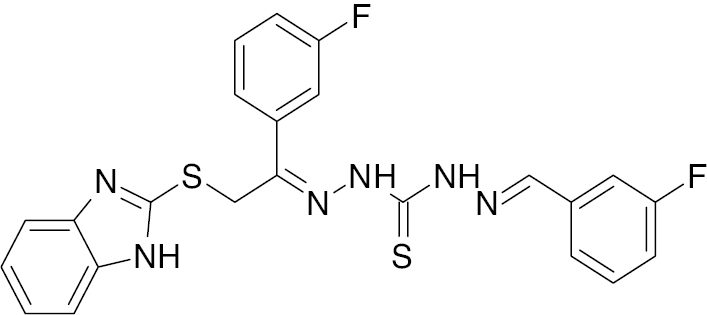 |
Benzimidazole with thiosemicarbazone |
IC50 value, alpha glycosidase (1.30 μM) and alpha-amylase (1.20 μM) | [68] |
| 6 | 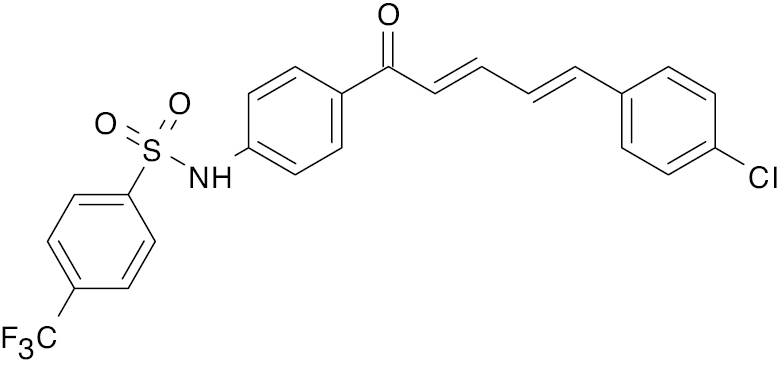 |
Diarylpentadienone |
IC50, 5.69 μM (alpha glycosidase) | [69] |
| 7 | 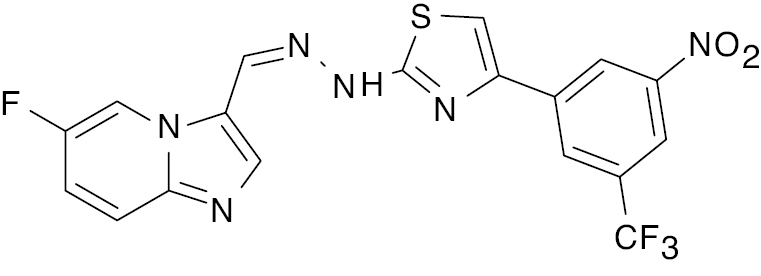 |
Imidazopyridine |
IC50, 5.57 μM (alpha glycosidase) | [70] |
| 8 | 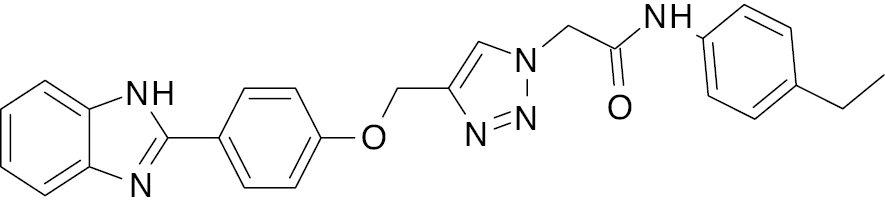 |
Benzimidazole with phenoxy acetamide |
50.0% inhibition, 108 μM; IC50, 25.2 μM (alpha glycosidase) | [71] |
| 9 | 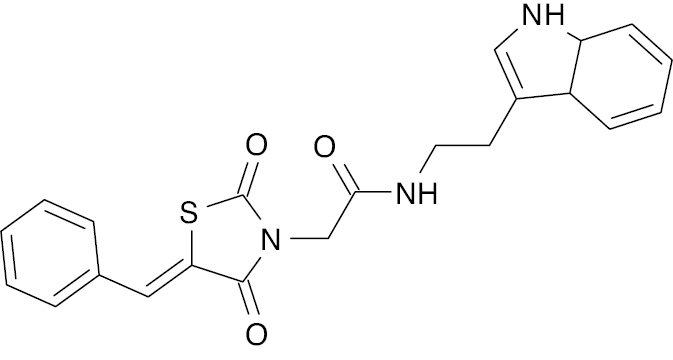 |
Indole derivative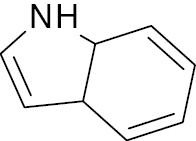 |
IC50, 2.35 μM (alpha glycosidase) | [72] |
| 10 | 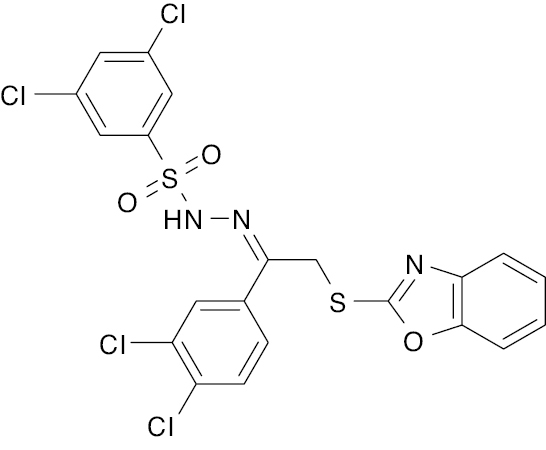 |
Benzoxazole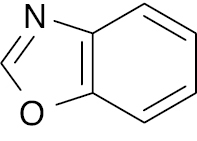 |
IC50 value, alpha glycosidase (1.20 μM) and alpha-amylase (1.10 μM) | [73] |
| 11 | 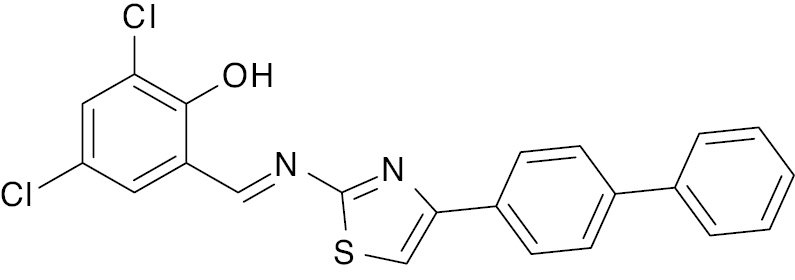 |
Thiazole |
IC50 value, alpha glycosidase (0.5 μM) and alpha-amylase (0.6 μM) | [74] |
| 12 | 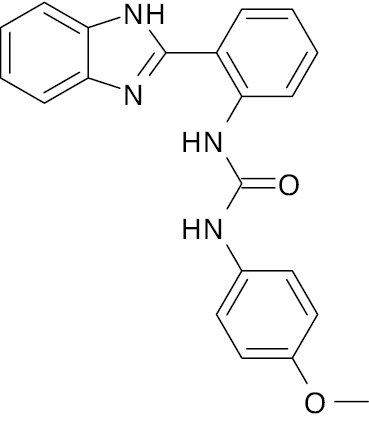 |
Benzimidazole urea derivative |
IC50 value, alpha glycosidase (17.47 μM) and alpha-amylase (18.65 μM) | [75] |
| 13 | 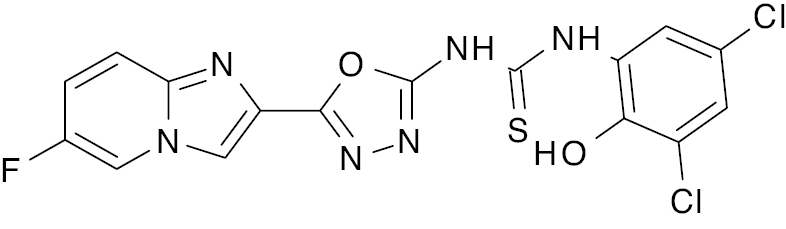 |
Imidazopyridine |
IC50 value, alpha glycosidase (0.90 μM) and alpha-amylase (1.10 μM) | [76] |
| 14 |  |
Benzimidazole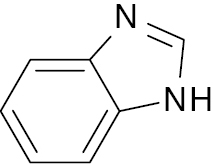 |
IC50 value, alpha glycosidase (2.70 μM) and alpha-amylase (1.30 μM) | [77] |
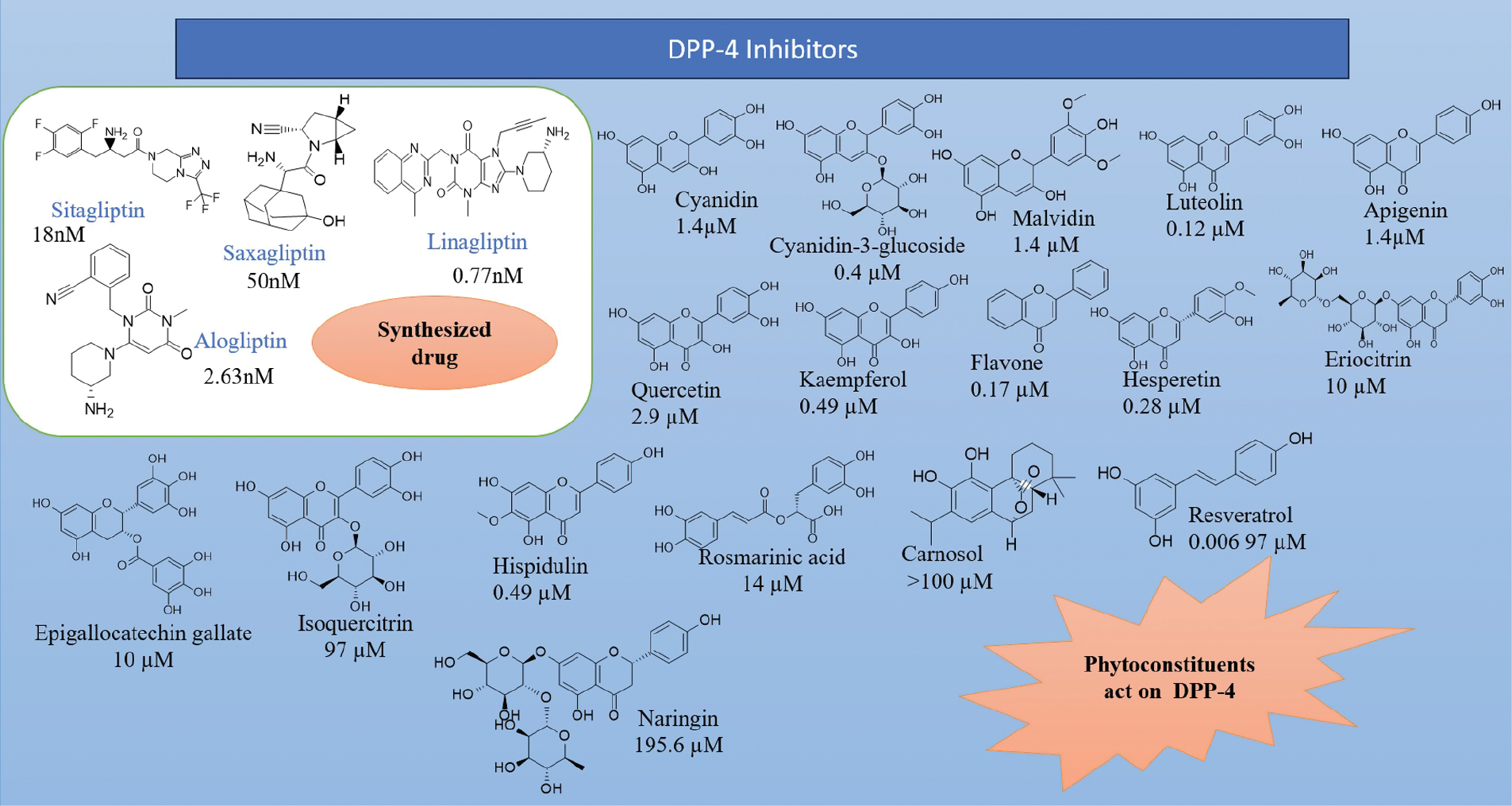
Dipeptidyl peptidase-4 (DPP-4) inhibitors
DPP-4 inhibitors are utilized in the management of type 2 DM. Recent studies have shown that DPP-4 inhibitors have positive renal and cardiovascular effects. Tablets, like sitagliptin, saxagliptin, and linagliptin, are regularly used in blended remedies with different antidiabetic medications, together with metformin or sodium-glucose co-transporters type 2 (SGLT2) inhibitors, for additional glucose control [78].
Mechanism of action
DPP-4, also known as adenosine deaminase complexing protein 2 (ADCP-2) or CD26, is a glycoprotein (110 kDa) expressed on the surface of various cells [79, 80]. Under normal physiologic conditions, food intake stimulates the production of glucagon-like peptide-1 (GLP-1), an incretin hormone that activates the GLP-1 receptor, a Gs protein-coupled receptor. This activation triggers adenyl cyclase, converting ATP into cAMP, which in turn stimulates insulin release. However, GLP-1 is rapidly degraded by the enzyme, DPP-4, which is on the vascular endothelium. To counteract this effect, DPP-4 inhibitors are administered to inhibit DPP-4, thereby prolonging GLP-1 activity and enhancing insulin secretion [81].
It has been observed that the phytoconstituents, cyanidin, cyanidin-3-glucoside, malvidin, luteolin, apigenin, quercetin, kaempferol, flavone, hesperetin, naringenin, eriocitrin, genistein, resveratrol, epigallocatechin gallate, gallic acid, caffeic acid, isoquercitrin, eriodictyol, hispidulin, rosmarinic acid, carnosol, and naringin, help reduce blood glucose levels by acting on the DPP-4 receptor (Figure 8) [82–86].
Figure 8 Marketed available drugs, phytoconstituents, and IC50s.
Recent advances in DPP-4 and combination therapy
Recent advancements in DPP-4 inhibitors focus on enhanced combination treatments to achieve better glycemic control, while reducing side effects. DPP-4 inhibitors, which are known for extending GLP-1 activity to enhance insulin secretion and reduce glucagon levels, are often combined with other agents, such as metformin, SGLT-2 inhibitors, and other therapeutic moieties, as shown in Table 4; drugs under clinical trials are listed in Table 5. A 2024 pilot study confirmed that the combination of DPP-4 inhibitors with SGLT-2 inhibitors significantly decrease HbA1c levels while improving physical function in patients with type 2 DM. This dual approach is particularly beneficial for patients at risk for cardiovascular disease. Additionally, recent meta-analyses indicated that combining DPP-4 inhibitors with metformin yields superior glycemic control compared to monotherapy because the combination enhances GLP-1 concentrations more effectively than DPP-4 inhibitor monotherapy [87]. A 2024 consensus paper further supports the use of DPP-4 inhibitors with metformin and/or SGLT-2 inhibitors due to the complementary mechanisms of action [88]. Furthermore, several DPP-4 inhibitors, such as dutogliptin and gosogliptin, are currently under clinical trials (Table 5), and have demonstrated promising potency (IC50 values) and therapeutic efficacy. These emerging agents are expected to expand the existing options for combination therapy in the management of type 2 DM. Notably, combination therapies with SGLT-2 inhibitors also support cardiovascular and renal health, which are critical for managing DM-related complications. However, current guidelines suggest considering individual patient profiles to optimize DPP-4 therapy choices because of the efficacy in reducing mortality is limited [89].
Table 4 Recent Synthesized DPP-4 and the Combination Moiety
| S.No | Structure | Combination | Result | References |
|---|---|---|---|---|
| 1 | 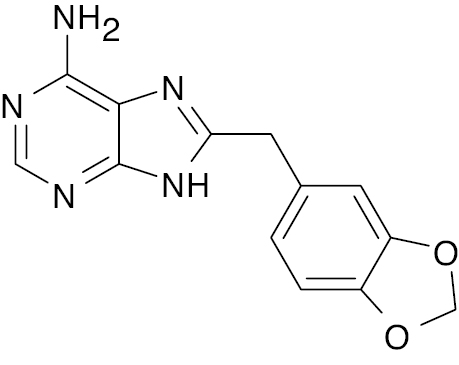 |
Purine |
Reduce fasting hyperglycemia value by 24%. | [90] |
| 2 | 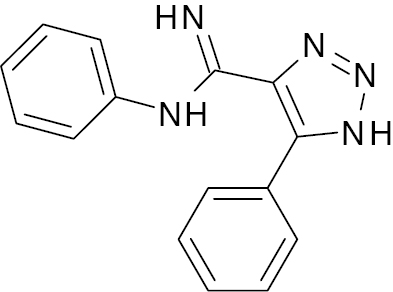 |
Triazole |
Inhibitory activity of DDP-4 with IC50 of 14.75nM | [91] |
| 3 | 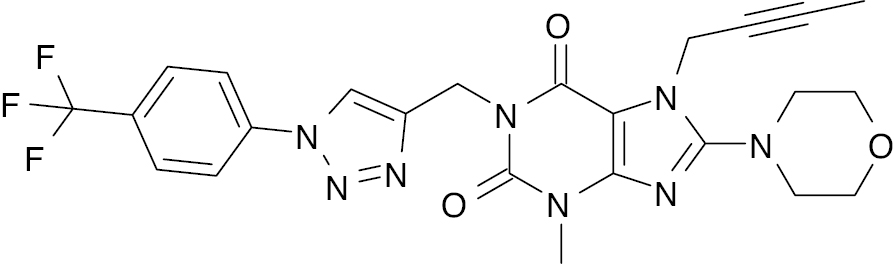 |
Xanthine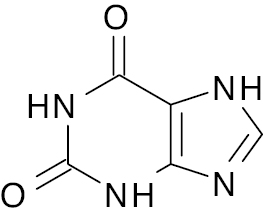 |
Percentage inhibition at 100 nM (78.53%) and IC50 (16.34 nM) | [92] |
| 4 | 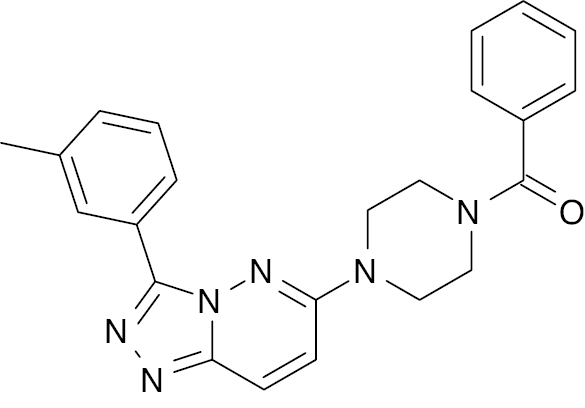 |
Piperazine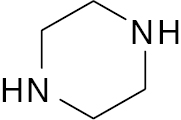 |
IC50, 0.75 nM | [93] |
| 5 | 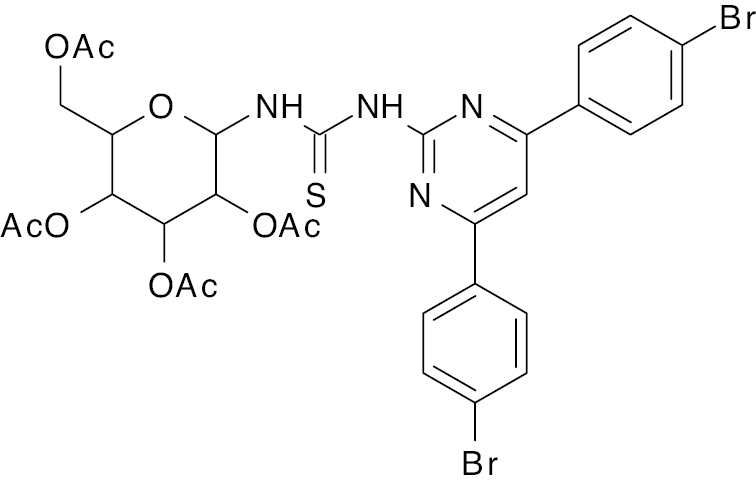 |
Thiourea |
IC50, 2.53 nM | [94] |
| 6 | 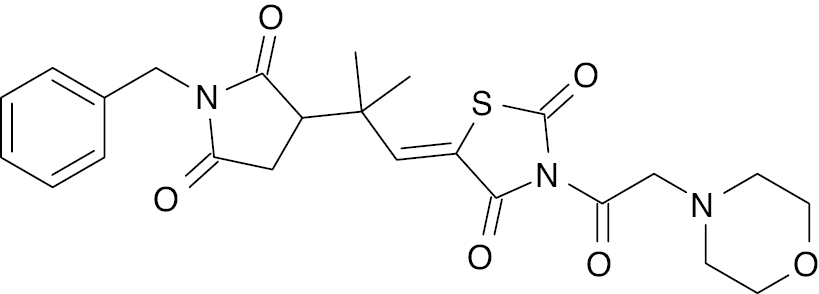 |
Succinimide Thiazolidine 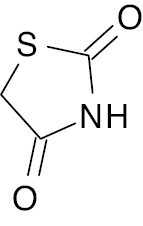 |
IC50, 4.22 nM | [95] |
Table 5 Drugs under Clinical Trials that act on the DPP-4 Receptor
| Clinical trial | |||
|---|---|---|---|
| Vildagliptin [113] | 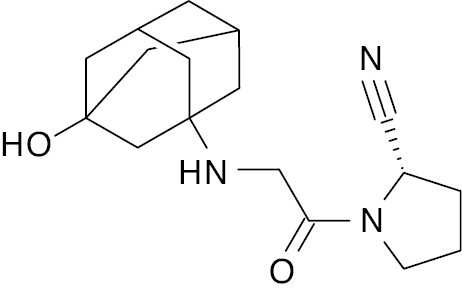 |
Approved in Europe | 110.95 μg/mL [96] or 62 nM [97] |
| MK-3102 (Omarigliptin) [113] | 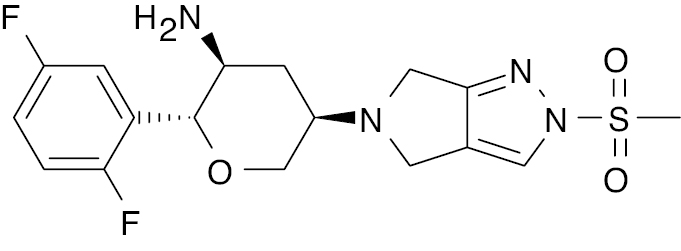 |
Approved in Japan | 1.6 nM [98] |
| Gemigliptin [113] | 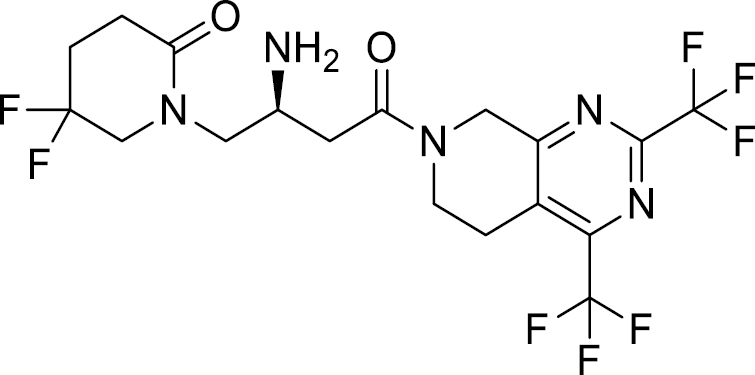 |
Approved in Korea | 6.31 nM [99] |
| SYR-472 (Trelagliptin) [113] | 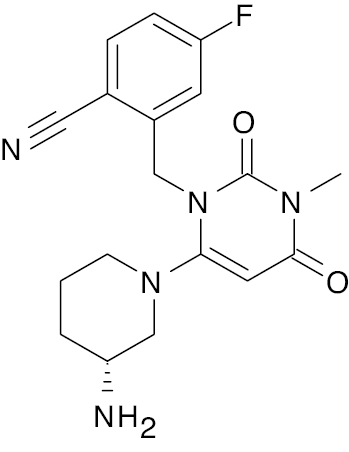 |
Approved in Japan | 4 nM [100] |
| Anagliptin [113] | 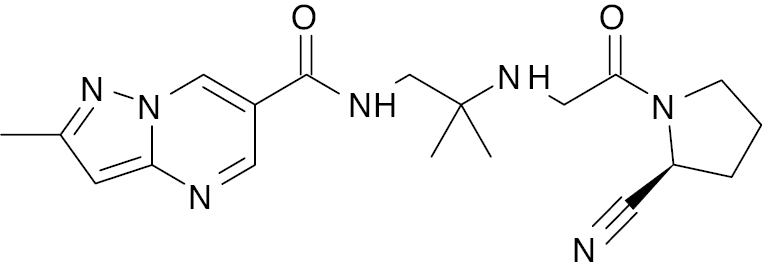 |
Approved in Japan | 3.8 nM [101] |
| Teneligliptin [113] | 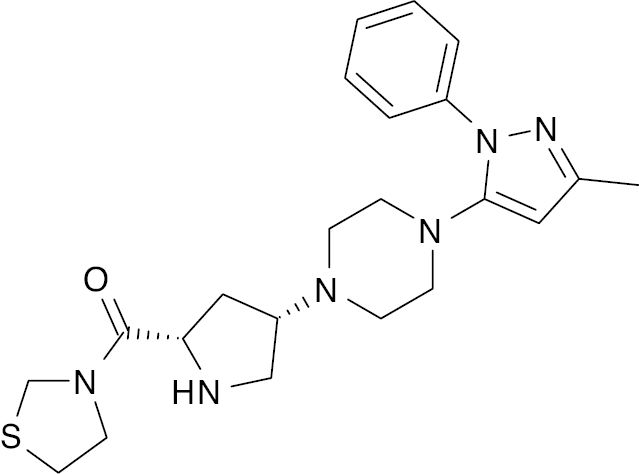 |
Approved in Japan and Korea | 1 nM [102] |
| Dutogliptin [113] | 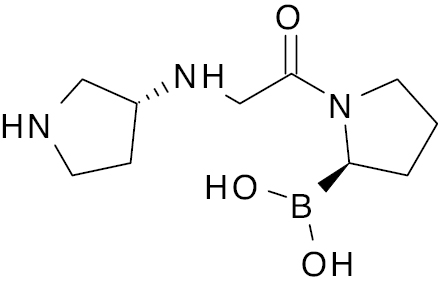 |
Phase 3 | 25 nM [103] |
| Gosogliptin [113] | 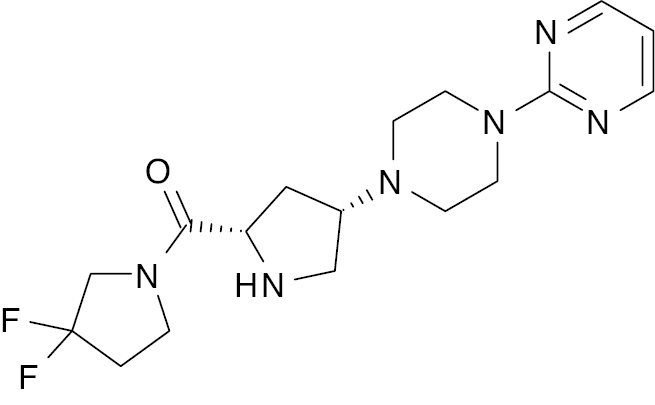 |
Phase 3 | 0.013 nM [103] |
| DA-1229 (Evogliptin) [113] | 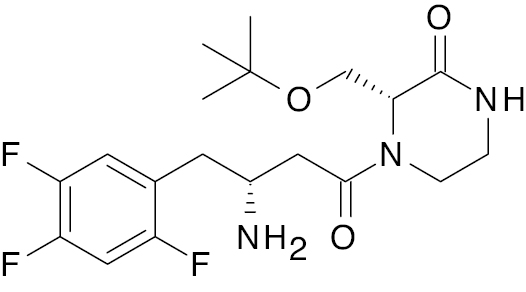 |
Phase 3 | 7.5 nM [104] |
GLUT4
Glucose transporters (GLUT4) are transmembrane proteins that transport across the cell membrane. GLUT4 provide glucose entry into the skeletal and adipose tissue. This process is achieved through insulin-induced GLUT4 translocation, which increases the rate of glucose flux into a cell. The GLUT-4 receptor consists of 12 transmembrane domains with unique N- and C-terminal sequences that have a crucial role in insulin signaling and membrane trafficking. The primary function of GLUT-4 is to facilitate glucose transport across cell membranes by stimulating glucose uptake into muscle and adipose cells in response to insulin [105]. It has been observed that Capparis moon, Sophora alopecuroides, and Gundelia tournefortii exhibit antidiabetic effects through the GLUT-4 pathway [106].
Recent advances in GLUT4 and combination therapy
Recent research focuses on ways to enhance GLUT4 function or expression to improve insulin sensitivity in diabetic patients. Combination therapies that include agents, such as metformin, thiazolidinediones, or GLP-1 agonists (Table 6), have shown promise in boosting GLUT4 activity and improving glycemic control [107].
Table 6 Recently Synthesized GLUT4 and the Combination Moiety
| S.No | Structure | Combination | Result | References |
|---|---|---|---|---|
| 1 | 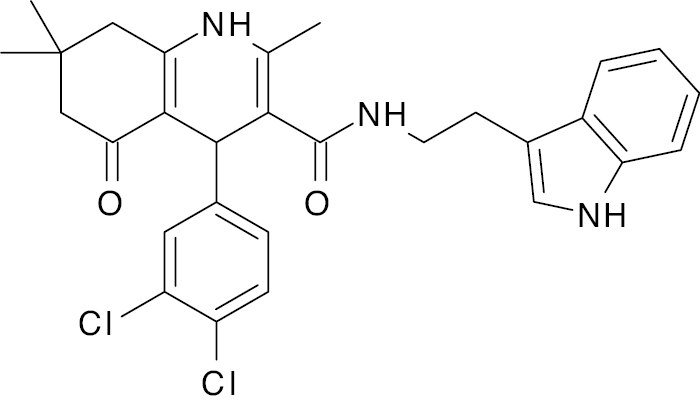 |
Indole |
21.6 % reduction in blood glucose level | [108] |
| 2 | 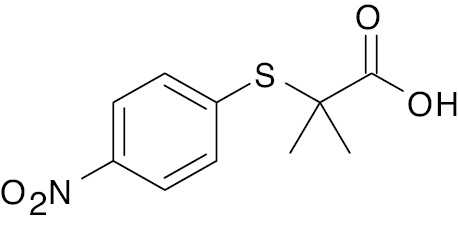 |
Phenoxy isobutyric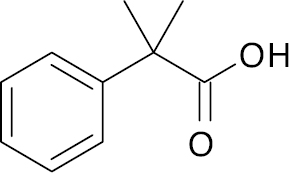 |
-50.1% maximal percentage of glycemic-lowering effect | [109] |
Sodium-glucose co-transporter type 2 inhibitors (SGLT2is)
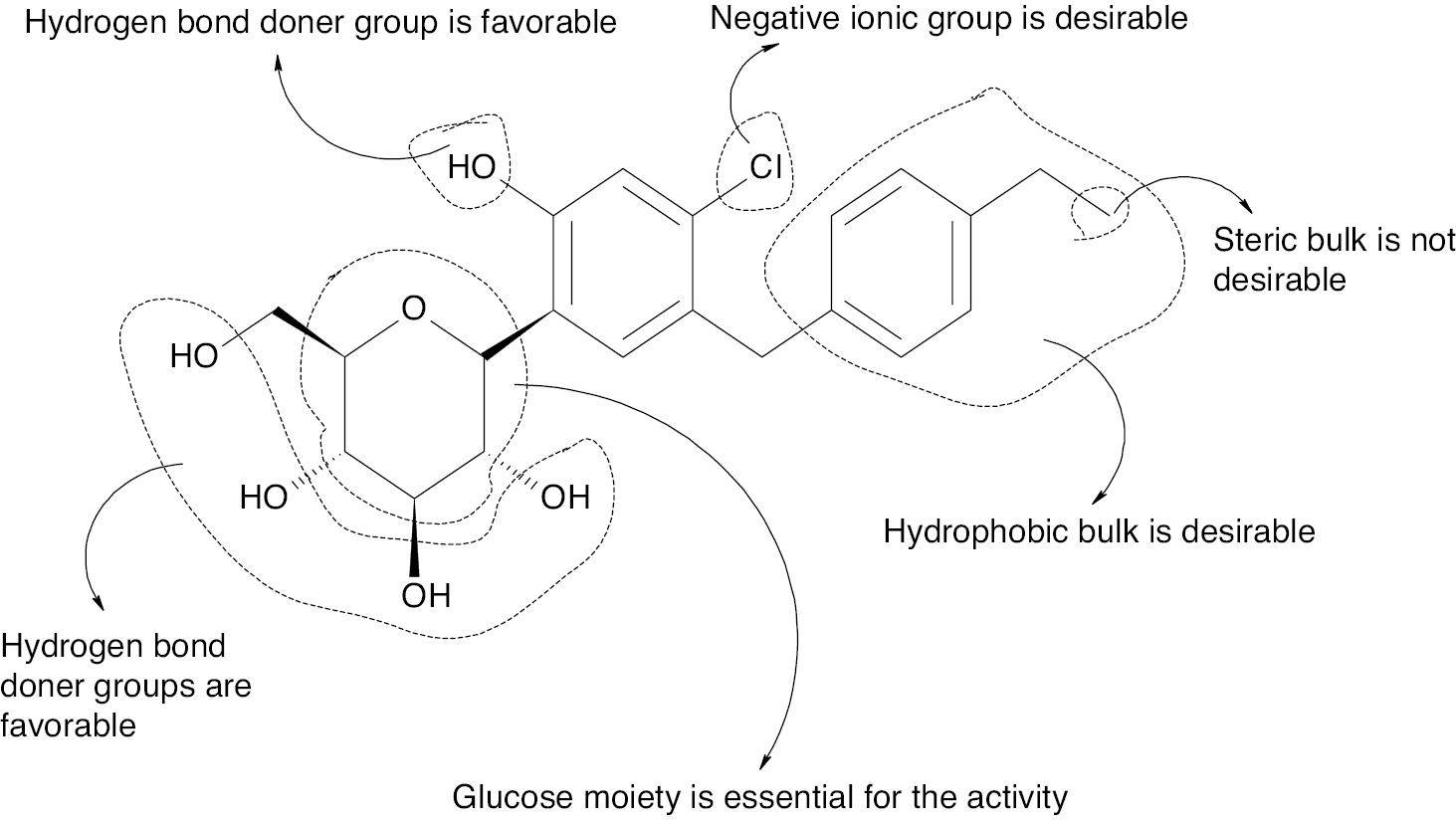
Sodium-glucose co-transporters (SGLTs) have a crucial role in glucose reabsorption in the kidneys and glucose absorption in the intestines. SGLT1 and SGLT2 belong to the SLC5A gene family, with SGLT2 (SLC5A2) predominantly expressed in the renal cortex. SGLT2 consists of approximately 670 amino acids arranged into 14 transmembrane helices, 10 of which are responsible for glucose and sodium binding [110]. The inhibitory activity of SGLT2 inhibitors is influenced by hydrogen substitution on the glucose moiety. The presence of a bulky benzene ring negatively affects activity due to steric hindrance, whereas a hydrogen bond donor group at the para position of the benzene ring is attached to the sugar moiety enhances activity. Additionally, hydrogen bond donor groups at various positions on the sugar moiety are essential for improving SGLT2 inhibitory action, as illustrated in Figure 9.
Figure 9 Key features identified through a 3D-QSAR study for the design of novel molecules as SGLT2 inhibitors.
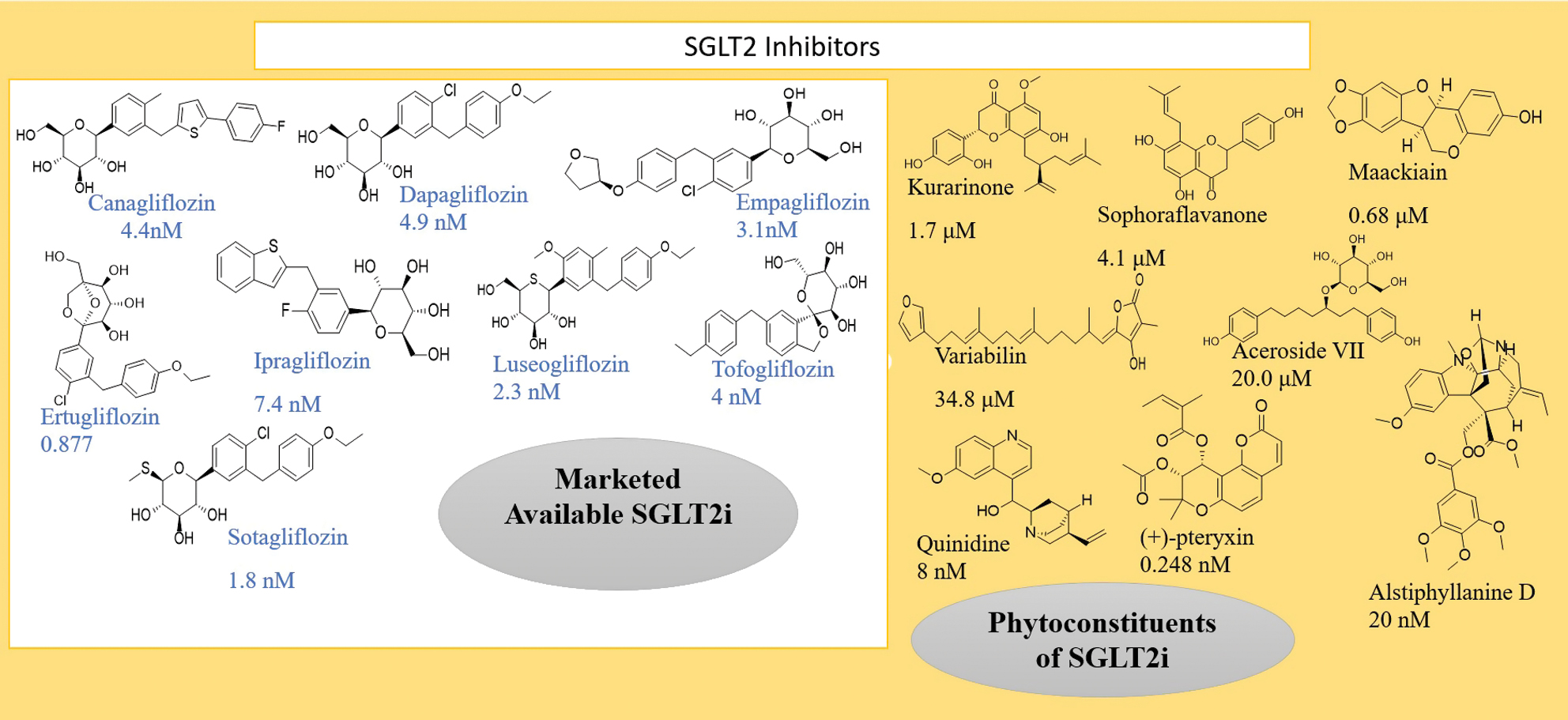
SGLT2 inhibitors share a common oxane-3,4,5-triol moiety with different substitutions at the 4th carbon of the oxane ring. For example, canagliflozin contains fluorophenyl, thiophene, and methyl phenyl substitutions, while dapagliflozin, empagliflozin, and ipragliflozin feature ethoxyphenyl, oxyphenyl, and benzothiophene substitutions. Other drugs in this category include ertugliflozin and luseogliflozin, as shown in Figure 10. Despite the benefits in reducing glucose reabsorption, SGLT2 inhibitors have side effects. SGLT2 inhibitors may cause changes in the genital area by increasing moisture levels, which when combined with higher glucose concentrations in genital secretions, creates an environment conducive to yeast growth. This effect increases the risk of genital tract infections, particularly fungal infections, such as yeast infections [111, 112].
Figure 10 Marketed available drugs, phytoconstituents, and IC50s.
Mechanism of action
SGLT2 inhibitors function by promoting glycosuria, thereby effectively lowering blood glucose levels. These inhibitors target the SGLT2 protein in the renal proximal tubules, which is responsible for reabsorbing approximately 90% of filtered glucose. By blocking SGLT2, these drugs reduce renal glucose reabsorption, leading to increased glucose excretion through urine and a subsequent decrease in blood glucose levels [113]. Beyond the glucose-lowering effects, SGLT2 inhibitors also provide therapeutic benefits in managing DM-related complications, such as nephropathy and retinopathy [114].
Additionally, some phytoconstituents have been shown to act on the SGLT2 receptor and contribute to reducing blood glucose levels. These phytoconstituents include kurarinone, sophoraflavanone, maackiain, variabilin, formononetin, pterocarpin, aceroside VII, 10-methoxy-N(1)-methylburnamine, 17-O-veratrate, alstiphyllanine D, (+)-pteryxin, and quinidine (Figure 11) [115–118].
Recent advances in SGLT2is and combination therapy
Recent advances in SGLUT2 inhibitors for management of DM emphasize that combination therapies, especially with drugs, like GLP-1 receptor agonists and metformin.
SGLT2 inhibitor and GLP-1 receptor agonist combination therapy has become a viable treatment strategy because these medications work through complementary mechanisms. SGLT2 inhibitors reduce blood glucose and improve cardiovascular outcomes, while GLP-1 agonists enhance insulin secretion and promote weight loss. Recent studies have shown that this dual therapy may reduce major cardiovascular events, heart failure hospitalizations, and overall mortality in diabetic patients with cardiovascular disease. Other studies have shown that combining SGLT2is with other potent moieties enhances effectiveness (Tables 7 and 8 show drugs under clinical trials) [119, 120].
Table 7 Recenlyt Synthesized SGLT2i and the Combination Moiety
| S.No | Structure | Combination | Result | References |
|---|---|---|---|---|
| 1 | A.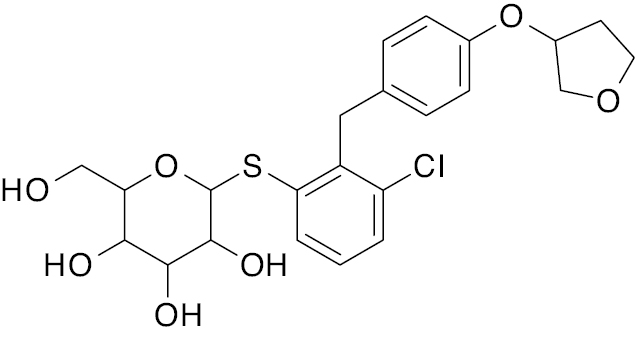 B. 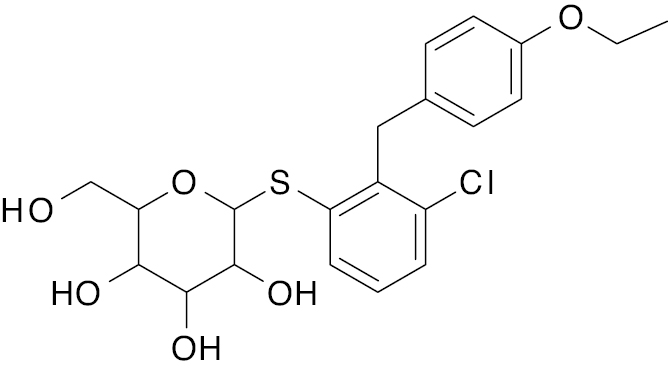 C. 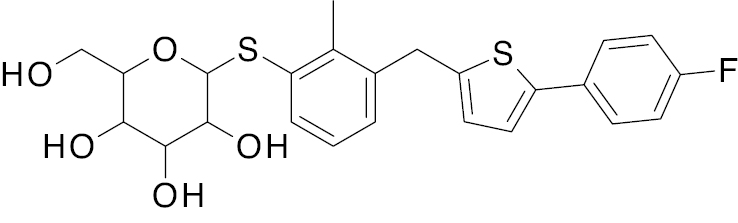 |
Empagliflozin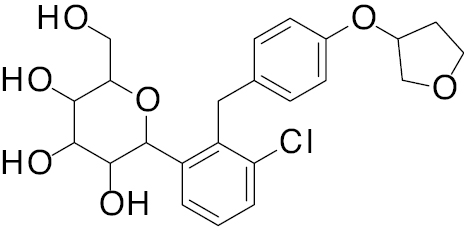 Dapagliflozin 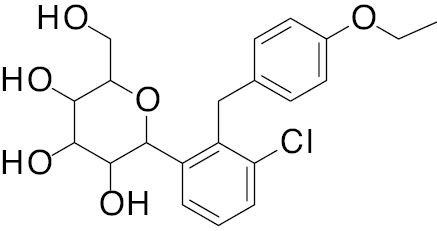 Canagliflozin 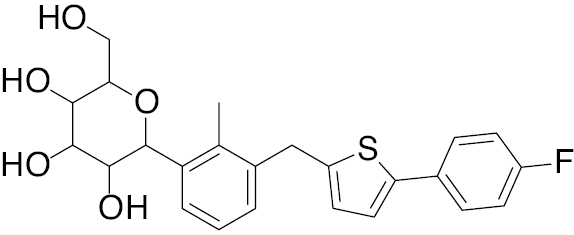 |
A. IC50, 2.0 nM B. IC50, 2.6 nM C. IC50, 2.5 nM |
[121] |
| 2 | 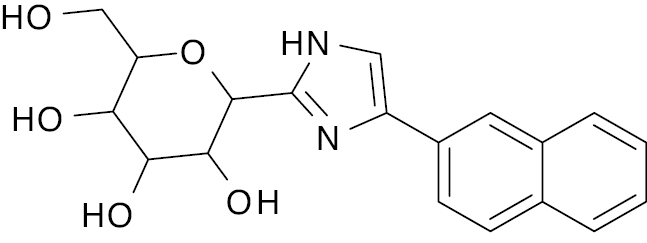 |
D-glucopyranosyl |
IC50, 3.50 μM | [122] |
| 3 | 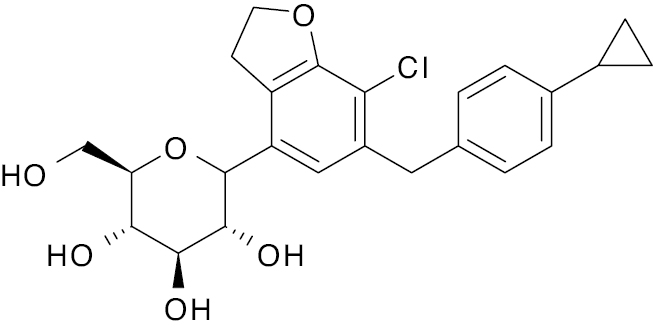 |
Glycosidase |
IC50, 0.460 μM | [123] |
| 4 | 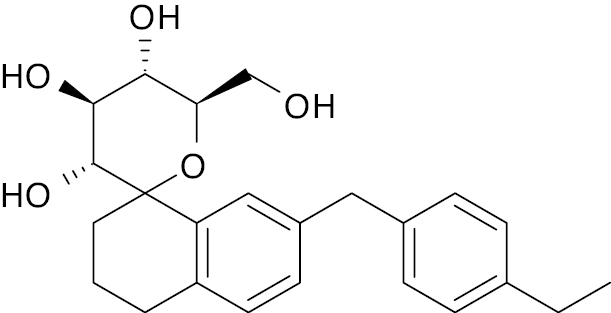 |
Glycosidase |
IC50, 436 nM | [124] |
Table 8 Synthetics, Phytoconstituents, and Drugs under Clinical Trials that Act on SGLT2
| Clinical trial | |||
|---|---|---|---|
| Pioglitazone [125] | 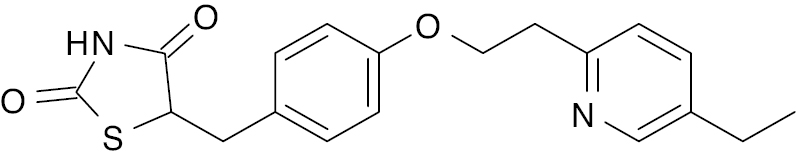 |
Phase 4 Usually administered in combination with metformin, glimepiride, and alogliptin [126] |
60.16 μg/mL |
| Tofogliflozin (Developed by Chugai Pharmaceutica and approved in Japan) [127, 128] | 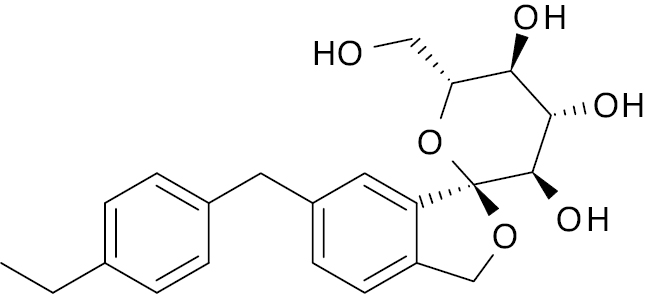 |
Phase 3 | 4.5 nM |
| Remogliflozin Etabonate [129] | 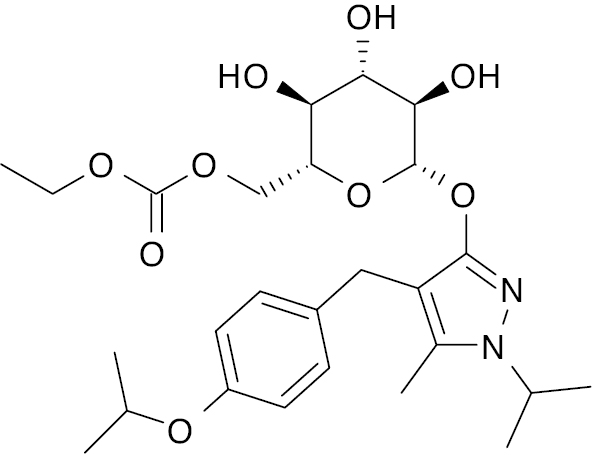 |
Phase IIb | 43 and 39 μM |
G protein-coupled receptor (GPCR)
GPCRs have a critical role in regulating insulin secretion and maintaining pancreatic beta cell function, making GPCRs essential in the management of type 2 DM. For example, the GLP-1 receptor helps stimulate insulin release from pancreatic beta cells and drugs that target this receptor have become a key treatment option for T2D [130].
Mechanism of action
GLP-1 is activated by the beta cell surface, leading to conformational changes in the receptor that are unable to interact with the intracellular G proteins. GPCR enhances insulin exocytosis through this pathway, the process by which insulin is released from beta cells into the bloodstream, thereby lowering blood glucose levels after meals. Additionally, GPCR kinases (GRKs) and beta cell arrest help regulate the activity of these receptors by promoting receptor internalization and recycling. This regulation is essential to prevent beta-cell stress and overactivation, which can contribute to cell damage over time. Apart from aiding insulin release, GPCR signaling also supports beta-cell health by activating pathways that prevent apoptosis and promote cell growth. This effect helps sustain an adequate beta-cell mass for insulin production, which is critical for long-term glucose control [77].
Recent advances in GPCR and combination therapy
New therapeutic strategies involving GPCR agonists, especially incretin-based therapies, such as GLP-1 and GIP receptor agonists, have shown promising results in managing glucose levels and improving metabolic health by supporting β-cell functionality. Emerging dual therapies that act on multiple GPCR targets simultaneously, such as combining GLP-1 with glucagon or GIP, have demonstrated enhanced efficacy in glucose control and cardiovascular health. These combination approaches show potential, not only in glycemic management, but also in reducing risks of complications linked to cardiovascular and kidney health, addressing broader aspects of metabolic disease. GPCR-targeted therapies are thus gaining traction as a foundation for DM treatment strategies that offer metabolic benefits beyond traditional therapies (Table 9) [131, 132].
Table 9 Recently Synthesized GPCR and the Combination Moiety
| S.No | Structure | Combination | Result | References |
|---|---|---|---|---|
| 1 | 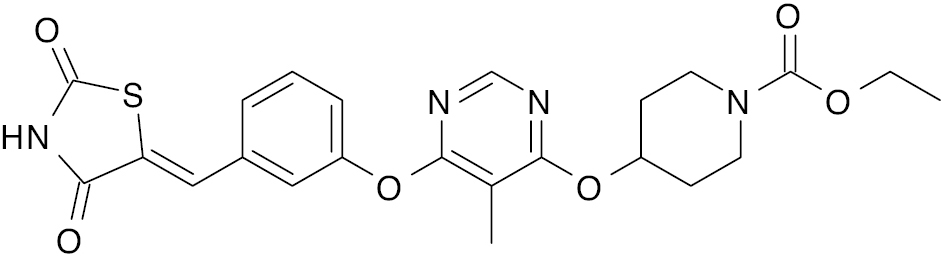 |
Thiazolidine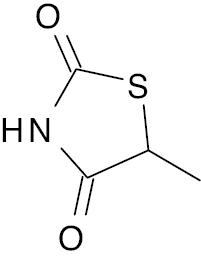 |
EC50 (43nM) and Emax (160%) | [133] |
| 2 | 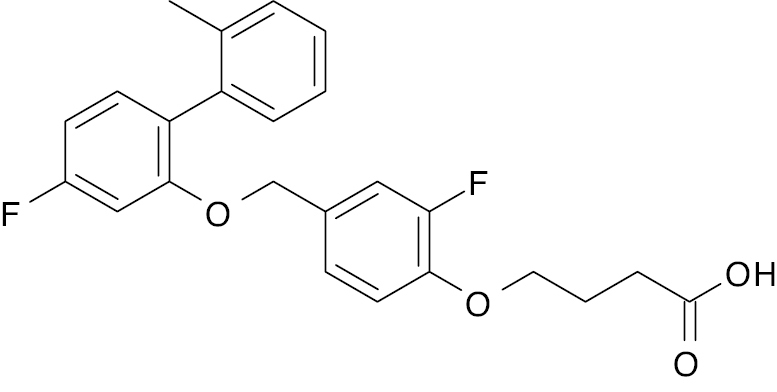 |
TUG-891 derivative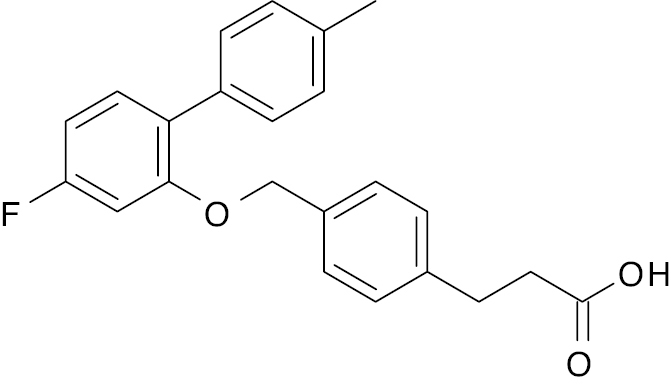 |
EC50, 37.5 nM | [134] |
| 3 | 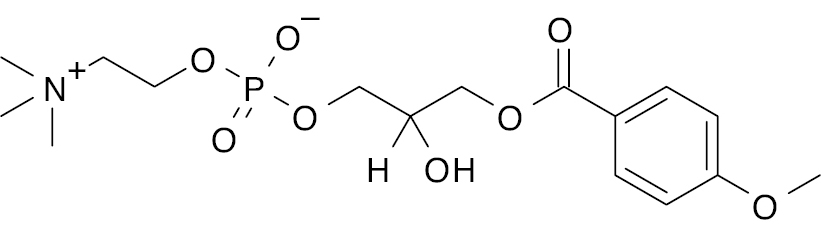 |
Lysophosphatidylcholine with anisic acid derivative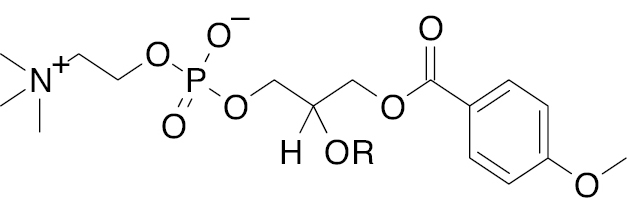 |
Acts as an antagonist of GPR40, GPR55, and GPR119, and increases insulin secretion up to X-fold |
[135] |
| 4 | CPL207280 (Novel GPR40/free fatty acid receptor)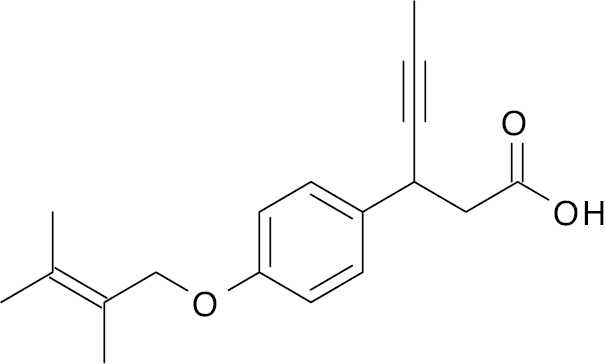 |
– | EC50 (270 nM) and stimulates insulin secretion 3.9-fold | [136] |
| 5 | 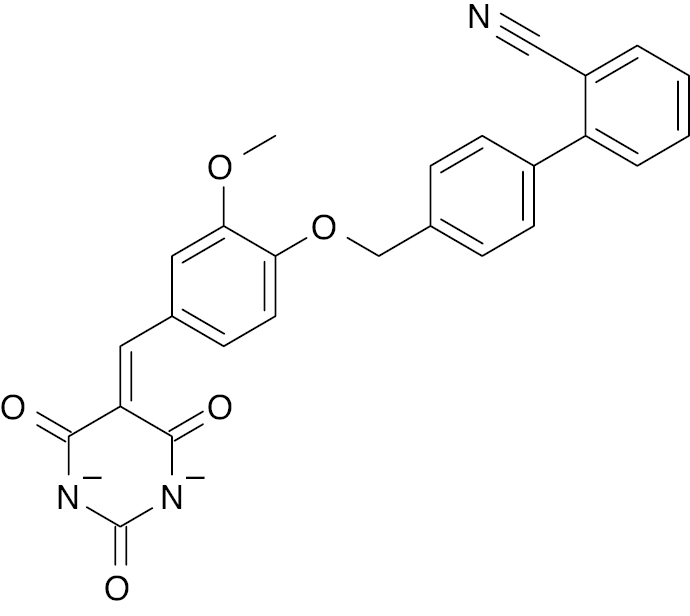 |
Barbiturates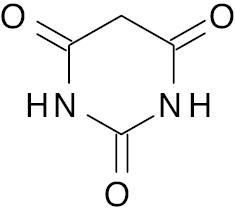 |
Increases GPR40 expression by 7-fold | [137] |
| 6 | 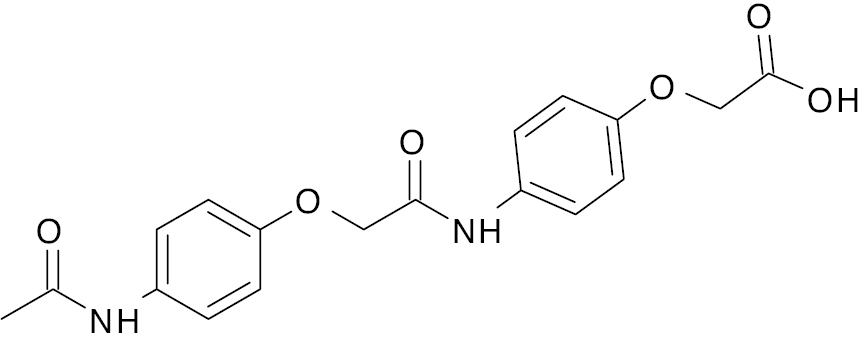 |
Phenoxyacetic derivative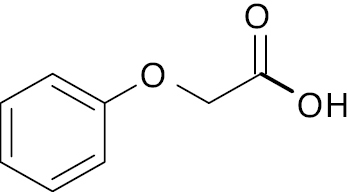 |
Increases GLUT4 expression 7-fold | [138] |
| 7 | 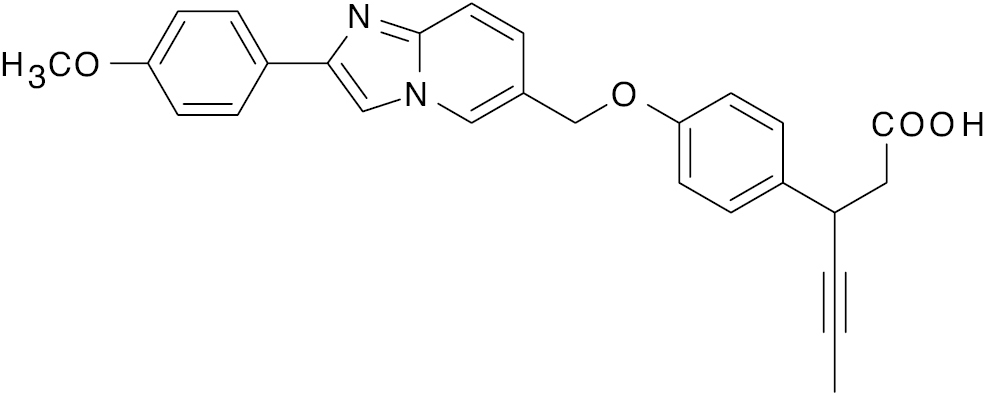 |
Imidazo- pyridine |
EC50, 47 nM | [139] |
Discussion
Recent evidence and ongoing research across various classes of antidiabetic agents, including sulfonylureas, biguanides, PPARγ agonists, and DPP-4 inhibitors, underscore the complexity of achieving optimal glycemic control while mitigating cardiovascular and renal risks. Synthesizing recent progress and identifying future research priorities is crucial for developing more effective and personalized therapeutic strategies. Sulfonylureas, while effective in stimulating insulin release, may not offer significant cardiovascular benefits and are associated with an increased risk of hypoglycemia, highlighting the need for caution and careful patient selection. Biguanides, particularly metformin, have demonstrated a cardioprotective effect by reducing the risk of myocardial infarction by 30% compared to sulfonylureas and offering renal benefits. However, the efficacy of biguanides may vary among different patient populations, emphasizing the importance of personalized treatment approaches. PPARγ agonists, including pan-PPAR agonists, have emerged as promising therapeutic targets due to the ability to improve insulin sensitivity, regulate lipid metabolism, and modulate inflammatory responses. However, challenges remain in minimizing adverse effects, such as weight gain and fluid retention. Selective PPARγ modulators (SPPARMs) and combination therapies with other antidiabetic agents offer potential solutions to enhance efficacy while reducing cardiovascular risks. DPP-4 inhibitors enhance incretin levels, leading to improved insulin secretion and glucose homeostasis. Studies have suggested that combining DPP-4 inhibitors with SGLT2 inhibitors yields better glycemic control and cardiovascular protection. Emerging DPP-4 inhibitors under clinical trials may expand therapeutic options, but careful patient profiling is necessary due to limitations in mortality reduction. Despite these advances, significant gaps persist. The cardiovascular benefits of SGLT2 inhibitors vary based on heart failure status and metformin cardioprotection differs between obese and lean patients. Long-term safety concerns, such as hypoglycemia with sulfonylureas and vitamin B12 deficiency with metformin, warrant careful monitoring.
In addition to the well-established receptors involved in management of DM, several other receptors have emerged as potential targets for regulating blood glucose levels. One such target is the neurokinin 2 receptor (NK2R). NK2R agonists have shown promise as potential treatments for type 2 DM and obesity. These agonists reduce appetite, enhance energy expenditure, and improve insulin sensitivity. Studies utilizing hyperinsulinemic-euglycemic clamp techniques have demonstrated that NK2R activation acutely enhances insulin sensitivity [141]. Another promising target is TGR5, a bile acid receptor belonging to the G-protein-coupled receptor (GPCR) superfamily. TGR5 has a crucial role in regulating blood glucose levels through mechanisms, such as stimulating insulin secretion from pancreatic beta cells and promoting energy expenditure. Activation of TGR5 has demonstrated beneficial effects on glucose metabolism, making TGR5 a compelling target for the development of novel treatments for type 2 DM. By improving insulin secretion and enhancing energy homeostasis, TGR5 activation offers a promising strategy for managing type 2 diabetes [142].
In conclusion, the landscape of DM management is shifting towards a more holistic approach that prioritizes prevention of complications with glycemic control. While current therapies offer various benefits, ongoing research is essential to optimize treatment strategies and develop new agents that provide comprehensive protection against cardiovascular and renal complications. Personalized therapy, informed by large-scale trials comparing outcomes across drug classes, will be crucial for improving patient outcomes and reducing the burden of type 2 DM and associated complications.
Limitation
The receptors mentioned herein have limitations that need to be considered while targeting the specific receptor, such as sulfonylureas. While effective in stimulating insulin secretion, sulfonylureas carry a significant risk of hypoglycemia, weight gain, and beta-cell exhaustion with limited long-term cardiovascular benefits. Biguanides, like metformin, can cause gastrointestinal discomfort and are contraindicated in patients with renal impairment due to the risk of lactic acidosis, with limited efficacy in severe insulin deficiency. PPARγ agonists improve insulin sensitivity but are associated with weight gain, fluid retention, bone fractures, and potential cardiovascular risks. Alpha-glucosidase inhibitors, such as acarbose, are limited by frequent gastrointestinal side effects and modest efficacy in reducing HbA1c. DPP-4 inhibitors provide modest glucose-lowering effects and carry rare risks of pancreatitis and joint pain without significant weight loss benefits. GLUT4 dysfunction remains challenging because no direct activators exist, limiting the therapeutic potential. SGLT2 inhibitors effectively lower blood glucose but increase the risk of genital and urinary infections, dehydration, and rare cases of euglycemic diabetic ketoacidosis with reduced efficacy in advanced renal impairment. G-protein-coupled receptors, including TGR5, are promising but face challenges, such as off-target effects, limited clinical data, and potential unintended side effects due to the widespread presence of GPCRs in the body.
Conclusion and future scope
The current study underscores the complexity of blood glucose management in DM, highlighting the crucial roles of various pathways, from sulfonylureas to GPCR inhibitors. Given the chronic nature and increasing prevalence of DM, it is imperative to develop therapies that effectively target specific receptors associated with glucose management. We have studied different pathways and receptors that help maintain the blood glucose level and compiled all the recently synthesized derivatives from 2020–2024, which act on each receptor and show the improved potency of the medication. Future research should focus on combination therapy to enhance receptor specificity and improve target interactions. Furthermore, computational studies and pharmacophore modeling should be prioritized to design more selective and potent inhibitors. These approaches can help identify key molecular features required for receptor binding, optimizing drug-target interactions, and minimizing off-target effects. By integrating advanced computational techniques, such as molecular docking, QSAR, and AI-driven drug discovery, researchers can develop more precise and efficient therapeutic agents for diabetes management.
Acknowledgments
This work was funded under a seed money grant from Uttaranchal University (UU/DRI/SM/2024-25/017).
Consent for publication
None.
Statement on conflicts of interest
No conflicts of interest.
Graphical abstract
Highlights
- Diabetes mellitus is a systemic metabolic disorder impacting key organs, such as the heart, kidney, and eyes.
- KATP, AMPK, GLUT4, PPARγ, DPP-4, and SGLT2 are central targets in glucose regulation.
- Sulfonylurea, biguanides, and alpha-glucosidase inhibitors show promise in combination therapy.
- Novel targets such as NK2R and TGR5 are emerging in diabetes research.
- Challenges include hypoglycemia risk, renal limitation, and cardiovascular side effects.
In brief
Recent advancement in diabetes therapy focuses on targeting multiple receptors and pathways to improve glucose regulation. Key mechanisms include sulfonylurea, biguanides, PPARγ agonists, DPP-4 inhibitors, and SGLT2 inhibitors. Novel compounds and combination therapies show promise in enhancing insulin sensitivity and glycemic control. Despite these strides, challenges like hypoglycemia risk, cardiovascular safety, and personalized treatment remain central to optimizing diabetes management.
References
- Matoori S. Diabetes and its complications. ACS Pharmacol Transl Sci 2022;5(8):513-5. [DOI: 10.1021/acsptsci.2c00122]
- Pal D, Kumar S, Saha S. Antihyperglycemic activity of phenyl and ortho-hydroxy phenyl linked imidazolyl triazolo hydroxamic acid derivatives. Int J Pharm Pharm Sci 2017;9(12):247-51. [DOI: 10.22159/ijpps.2017v9i12.22086]
- Pories WJ, Albrecht RJ. Etiology of type II diabetes mellitus: role of the foregut. World J Surg 2001;25(4):527-31. [PMID: 11344408 DOI: 10.1007/s002680020348]
- Mishra V, Nayak P, Sharma M, Albutti A, Alwashmi AS, et al. Emerging treatment strategies for diabetes mellitus and associated complications: an update. Pharmaceutics 2021;13(10):1568. [PMID: 34683861 DOI: 10.3390/pharmaceutics13101568]
- IDF Diabetes Atlas, 10th edition 2022. Available from: https://diabetesatlas.org/ [accessed on 31 July 2023].
- Katsarou A, Gudbjörnsdottir S, Rawshani A, Dabelea D, Bonifacio E, et al. Type 1 diabetes mellitus. Nat Rev Dis Primers 2017;3:17016. [PMID: 28358037 DOI: 10.1038/nrdp.2017.16]
- Padhi S, Nayak AK, Behera A. Type II diabetes mellitus: a review on recent drug based therapeutics. Biomed Pharmacother 2020;131:110708. [PMID: 32927252 DOI: 10.1016/j.biopha.2020.110708]
- American Diabetes Association. Gestational diabetes mellitus. Diabetes Care 2004;27:S88-90. [PMID: 14693936 DOI: 10.2337/diacare.27.2007.S88]
- Rahati S, Shahraki M, Arjomand G, Shahraki T. Food pattern, lifestyle and diabetes mellitus. Int J High Risk Behav Addict 2014;3(1):e8725. [PMID: 24971303 DOI: 10.5812/ijhrba.8725]
- Saha S, Pal D. Role of histone deacetylase inhibitors on viral replication: a review. Biointerface Res Appl Chem 2023;13(6):589. [DOI: 10.33263/BRIAC136.589]
- Haas B, Hass MDS, Voltz A, Vogel M, Walther J, et al. Sulfonylureas exert antidiabetic action on adipocytes by inhibition of PPARγ serine 273 phosphorylation. Mol Metab 2024;85:101956. [PMID: 38735390 DOI: 10.1016/j.molmet.2024.101956]
- Lang V, Light PE. The molecular mechanisms and pharmacotherapy of ATP-sensitive potassium channel gene mutations underlying neonatal diabetes. Pharmacogenomics Pers Med 2010;3:145-61. [PMID: 23226049 DOI: 10.2147/PGPM.S6969]
- Muller AA. Managing pediatric ingestions of the new antidiabetic agents. J Emerg Nurs 2004;30(2):183-4. [PMID: 15039680 DOI: 10.1016/j.jen.2003.12.011]
- Reynaldo F, Fayed R. Religious fasting, diabetes management, and medication dosage adjustments. Medsurg Nursing 2022;31(1):4498. [DOI: 10.62116/msj.2022.31.1.44]
- Sahin I, Bakiner O, Demir T, Sari R, Atmaca A. Current position of gliclazide and sulfonylureas in the contemporary treatment paradigm for type 2 diabetes: a scoping review. Diabetes Ther 2024;15(8):1687-716. [PMID: 38935188 DOI: 10.1007/s13300-024-01612-8]
- El-Zahabi MA, Bamanie FH, Ghareeb S, Alshaeri HK, Alasmari MM, et al. Design, synthesis, molecular modeling and anti-hyperglycemic evaluation of quinazoline-sulfonylurea hybrids as peroxisome proliferator-activated receptor gamma (PPARγ) and sulfonylurea receptor (SUR) agonists. Int J Mol Sci 2022;23(17):9605. [PMID: 36077003 DOI: 10.3390/ijms23179605]
- Khamees Thabet H, Ammar YA, Imran M, Hamdy Helal M, Ibrahim Alaqel S, et al. Unveiling anti-diabetic potential of new thiazole-sulfonamide derivatives: design, synthesis, in vitro bio-evaluation targeting DPP-4, α-glucosidase, and α-amylase with in-silico ADMET and docking simulation. Bioorg Chem 2024;151:107671. [PMID: 39067419 DOI: 10.1016/j.bioorg.2024.107671]
- Ahmed A, Zaib S, Bhat MA, Saeed A, Altaf MZ, et al. Acyl pyrazole sulfonamides as new antidiabetic agents: synthesis, glucosidase inhibition studies, and molecular docking analysis. Front Chem 2024;12:1380523. [PMID: 38694406 DOI: 10.3389/fchem.2024.1380523]
- Ayoup MS, Khaled N, Abdel-Hamid H, Ghareeb DA, Nasr SA, et al. Novel sulfonamide derivatives as multitarget antidiabetic agents: design, synthesis, and biological evaluation. RSC Adv 2024;14(11):7664-75. [PMID: 38440282 DOI: 10.1039/D4RA01060D]
- Ranade SD, Alegaon SG, Khatib NA, Gharge S, Kavalapure RS, et al. Design, synthesis, molecular dynamic simulation, DFT analysis, computational pharmacology and decoding the antidiabetic molecular mechanism of sulphonamide-thiazolidin-4-one hybrids. J Mol Struct 2024;1311:138359. [DOI: 10.1016/j.molstruc.2024.138359]
- Khamees Thabet H, Ragab A, Imran M, Helal MH, Ibrahim Alaqel S, et al. Discovery of new anti-diabetic potential agents based on paracetamol incorporating sulfa-drugs: design, synthesis, α-amylase and α-glucosidase inhibitors with docking simulation. Eur J Med Chem 2024;275:116589. [PMID: 38878516 DOI: 10.1016/j.ejmech.2024.116589]
- Campbell RK, White Jr JR, Saulie BA. Metformin: a new oral biguanide. Clin Ther 1996;18(3):360-71. [PMID: 8829014 DOI: 10.1016/S0149-2918(96)80017-8]
- Bailey CJ. Metformin: effects on micro and macrovascular complications in type 2 diabetes. Cardiovasc Drugs Ther 2008;22:215-24. [PMID: 18288595 DOI: 10.1007/s10557-008-6092-0]
- Di Magno L, Di Pastena F, Bordone R, Coni S, Canettieri G. The mechanism of action of biguanides: new answers to a complex question. Cancers 2022;14(13):3220. [PMID: 35804992 DOI: 10.3390/cancers14133220]
- Yi H, Huang C, Shi Y, Cao Q, Chen J, et al. Metformin attenuates renal fibrosis in a mouse model of adenine-induced renal injury through inhibiting TGF-β1 signaling pathways. Front Cell Dev Biol 2021;9:603802. [PMID: 33614642 DOI: 10.3389/fcell.2021.603802]
- Demaré S, Kothari A, Calcutt NA, Fernyhough P. Metformin as a potential therapeutic for neurological disease: mobilizing AMPK to repair the nervous system. Expert Rev Neurother 2021;21(1):45-63. [PMID: 33161784 DOI: 10.1080/14737175.2021.1847645]
- Corleto KA, Strandmo JL, Giles ED. Metformin and breast cancer: current findings and future perspectives from preclinical and clinical studies. Pharmaceuticals 2024;17(3):396. [PMID: 38543182 DOI: 10.3390/ph17030396]
- Li W, Wang Y, Liu C, Yu Y, Xu L, et al. Evaluation of the regulatory effect of the pan-PPAR agonist chiglitazar on the dawn phenomenon. Diabetes Ther 2025;16(4):731-48. [DOI: 10.1007/s13300-025-01708-9]
- Barb D, Kalavalapalli S, Leiva EG, Bril F, Huot-Marchand P, et al. Pan-PPAR agonist lanifibranor improves insulin resistance and hepatic steatosis in patients with T2D and MASLD. J Hepatol 2025. [PMID: 39824443 DOI: 10.1016/j.jhep.2024.12.045]
- Deeks ED. Chiglitazar: first approval. Drugs 2022;82(1):87-92. [PMID: 34846697 DOI: 10.1007/s40265-021-01648-1]
- Sheng R, Li Y, Wu Y, Liu C, Wang W, et al. A pan-PPAR agonist E17241 ameliorates hyperglycemia and diabetic dyslipidemia in KKAy mice via up-regulating ABCA1 in islet, liver, and white adipose tissue. Biomed Pharmacother 2024;172:116220. [PMID: 38308968 DOI: 10.1016/j.biopha.2024.116220]
- Iltz JL, Baker DE, Setter SM, Keith Campbell R. Exenatide: an incretin mimetic for the treatment of type 2 diabetes mellitus. Clin Ther 2006;28(5):652-65. [PMID: 16861088 DOI: 10.1016/j.clinthera.2006.05.006]
- Inoue Y, Emoto M, Inoue H, Kaku K, Kaneko T. Characterization of the binding sites for [3H]glibenclamide in rat liver membranes. Eur J Pharmacol 1995;284(1-2):77-82. [PMID: 8549639 DOI: 10.1016/0014-2999(95)00368-u]
- Lee CR, Pieper JA, Frye RF, Hinderliter AL, Blaisdell JA, et al. Tolbutamide, flurbiprofen, and losartan as probes of CYP2C9 activity in humans. J Clin Pharmacol 2003;43(1):84-91. [PMID: 12520632 DOI: 10.1177/0091270002239710]
- Qaoud MT, Almasri I, Önkol T. Peroxisome proliferator-activated receptors as superior targets for treating diabetic disease, design strategies – review article. Turk J Pharm Sci 2022;19(3):353-70. [PMID: 35775494 DOI: 10.4274/tjps.galenos.2021.70105]
- Bosenberg LH, Van Zyl DG. The mechanism of action of oral antidiabetic drugs: a review of recent literature. J Endocrinol Metab Diabetes South Afr 2008;13(3):80-8. [DOI: 10.1080/22201009.2008.10872177]
- Sangeetha R. Luteolin in the management of type 2 diabetes mellitus. Curr Res Nutr Food Sci J 2019;7(2):393-8. [DOI: 10.12944/CRNFSJ.7.2.09]
- Mueller M, Jungbauer A. Culinary plants, herbs and spices – a rich source of PPARγ ligands. Food Chem 2009;117(4):660-7. [DOI: 10.1016/j.foodchem.2009.04.063]
- Mueller M, Lukas B, Novak J, Simoncini T, Genazzani AR, et al. Oregano: a source for peroxisome proliferator-activated receptor γ antagonists. J Agric Food Chem 2008;56(24):11621-30. [PMID: 19053389 DOI: 10.1021/jf802298w]
- Ansari P, Choudhury ST, Seidel V, Rahman AB, Aziz MA, et al. Therapeutic potential of quercetin in the management of type-2 diabetes mellitus. Life 2022;12(8):1146. [PMID: 36013325 DOI: 10.3390/life12081146]
- Shi GJ, Li Y, Cao QH, Wu HX, Tang XY, et al. In vitro and in vivo evidence that quercetin protects against diabetes and its complications: a systematic review of the literature. Biomed Pharmacother 2019;109:1085-99. [PMID: 30551359 DOI: 10.1016/j.biopha.2018.10.130]
- Fang XK, Gao J, Zhu DN. Kaempferol and quercetin isolated from Euonymus alatus improve glucose uptake of 3T3-L1 cells without adipogenesis activity. Life Sci 2008;82(11-12):615-22. [PMID: 18262572 DOI: 10.1016/j.lfs.2007.12.021]
- Alnuaimi S, Reljic T, Abdulla FS, Memon H, Al-Ali S, et al. PPAR agonists as add-on treatment with metformin in management of type 2 diabetes: a systematic review and meta-analysis. Sci Rep 2024;14(1):8809. [PMID: 38627464 DOI: 10.1038/s41598-024-59390-z]
- Skoczyńska A, Ołdakowska M, Dobosz A, Adamiec R, Gritskevich S, et al. PPARs in clinical experimental medicine after 35 years of Worldwide Scientific Investigations and medical experiments. Biomolecules 2024;14(7):786. [PMID: 39062500 DOI: 10.3390/biom14070786]
- Vaishnav Y, Dewangan D, Verma S, Mishra A, Thakur AS, et al. PPAR gamma targeted molecular docking and synthesis of some new amide and urea substituted 1, 3, 4-thiadiazole derivative as antidiabetic compound. J Heterocycl Chem 2020;57(5):2213-24. [DOI: 10.1002/jhet.3941]
- Najmi A, Alam MS, Thangavel N, Taha MME, Meraya AM, et al. Synthesis, molecular docking, and in vivo antidiabetic evaluation of new benzylidene-2, 4-thiazolidinediones as partial PPAR-γ agonists. Sci Rep 2023;13(1):19869. [PMID: 37963936 DOI: 10.1038/s41598-023-47157-x]
- Al Neyadi SS, Adem A, Amir N, Abdou IM. Targeting PPARγ receptor using new phosphazene derivative containing thiazolidinedione: design, synthesis, and glucose uptake. Open J Med Chem 2020;10(2):35-45. [DOI: 10.4236/ojmc.2020.102003]
- Ali IH, Hassan RM, El Kerdawy AM, Abo-Elfadl MT, Abdallah HMI, et al. Novel thiazolidin-4-one benzenesulfonamide hybrids as PPARγ agonists: design, synthesis and in vivo anti-diabetic evaluation. Eur J Med Chem 2024;269:116279. [PMID: 38460271 DOI: 10.1016/j.ejmech.2024.116279]
- Thabet HK, Abusaif MS, Imran M, Helal MH, Alaqel SI, et al. Discovery of novel 6-(piperidin-1-ylsulfonyl)-2H-chromenes targeting α-glucosidase, α-amylase, and PPAR-γ: Design, synthesis, virtual screening, and anti-diabetic activity for type 2 diabetes mellitus. Comput Biol Chem 2024;111:108097. [PMID: 38772048 DOI: 10.1016/j.compbiolchem.2024.108097]
- Thabet HK, Ragab A, Imran M, Hamdy Helal M, Ibrahim Alaqel S, et al. Innovation of 6-sulfonamide-2 H-chromene derivatives as antidiabetic agents targeting α-amylase, α-glycosidase, and PPAR-γ inhibitors with in silico molecular docking simulation. RSC Adv 2024;14(22):15691-705. [PMID: 38746843 DOI: 10.1039/D4RA02143F]
- Patchipala SB, Pasupuleti VR, Audipudi AV, Bollikolla HB. Synthesis, in-vivo anti-diabetic & anticancer activities and molecular modelling studies of tetrahydrobenzo[d]thiazole tethered nicotinohydrazide derivatives. Arab J Chem 2022;15(2):103546. [DOI: 10.1016/j.arabjc.2021.103546]
- Fadaly WAA, Elshewy A, Nemr MTM, Abdou K, Sayed AM, et al. Discovery of novel thiazole derivatives containing pyrazole scaffold as PPAR-γ agonists, α-glucosidase, α-amylase and COX-2 inhibitors; design, synthesis and in silico study. Bioorg Chem 2024;152:107760. [DOI: 10.1016/j.bioorg.2024.107760]
- Fayed EA, Thabet A, Abd El-Gilil SM, Elsanhory HHM, Ammar YA. Fluorinated thiazole–thiosemicarbazones hybrids as potential PPAR-γ agonist and α-amylase, α-glucosidase antagonists: design, synthesis, in silico ADMET and docking studies and hypoglycemic evaluation. J Mol Struct 2024;1301:137374 [DOI: 10.1016/j.molstruc.2023.137374]
- Gharge S, Alegaon SG, Ranade SD, Khatib NA, Kavalapure RS, et al. Design, synthesis of new 2, 4-thiazolidinediones: in-silico, in-vivo anti-diabetic and anti-inflammatory evaluation. Eur J Med Chem Rep 2024;11:100151. [DOI: 10.1016/j.ejmcr.2024.100151]
- Amin S, Sheikh KA, Iqubal A, Ahmed Khan M, Shaquiquzzaman M, et al. Synthesis, in-silico studies and biological evaluation of pyrimidine based thiazolidinedione derivatives as potential anti-diabetic agent. Bioorg Chem 2023;134:106449. [PMID: 36889200 DOI: 10.1016/j.bioorg.2023.106449]
- Safithri M, Sari YP. Inhibition of α-glucosidase activity by ethanolic extract of Melia azedarach L. leaves. IOP Conf Ser Earth Environ Sci 2016;31(1):012025. [DOI: 10.1088/1755-1315/31/1/012025]
- Nakamura S, Takahira K, Tanabe G, Muraoka O, Nakanishi I. Homology modeling of human alpha-glucosidase catalytic domains and SAR study of salacinol derivatives. Open J Med Chem 2012;2(3):50-60. [DOI: 10.4236/ojmc.2012.23007]
- Mauldina MG, Sauriasari R, Elya B. α-Glucosidase inhibitory activity from ethyl acetate extract of Antidesma bunius (L.) Spreng stem bark containing triterpenoids. Pharmacogn Mag 2017;13(52):590-4. [PMID: 29200718 DOI: 10.4103/pm.pm_25_17]
- Dabhi AS, Bhatt NR, Shah MJ. Voglibose: an alpha glucosidase inhibitor. J Clin Diagn Res 2013;7(12):3023-7. [PMID: 24551718 DOI: 10.7860/JCDR/2013/6373.3838]
- Xie X, Wu C, Hao Y, Wang T, Yang Y, et al. Benefits and risks of drug combination therapy for diabetes mellitus and its complications: a comprehensive review. Front Endocrinol 2023;14:1301093. [DOI: 10.3389/fendo.2023.1301093]
- Broome DT, Kodali A, Phillips D, Makin V, Mendlovic D, et al. Combined dipeptidyl peptidase 4 inhibitor and α-glucosidase inhibitor treatment in postprandial hypoglycemia. Clin Diabetes 2022;40(1):116-9. [PMID: 35221483 DOI: 10.2337/cd21-0042]
- Usman B, Sharma N, Satija S, Mehta M, Vyas M, et al. Recent developments in alpha-glucosidase inhibitors for management of type-2 diabetes: an update. Curr Pharm Des 2019;25(23):2510-25. [PMID: 31333110 DOI: 10.2174/1381612825666190717104547]
- Tavaf Z, Dangolani SK, Yousefi R, Panahi F, Shahsavani MB, et al. Synthesis of new curcumin derivatives as influential antidiabetic α-glucosidase and α-amylase inhibitors with anti-oxidant activity. Carbohydr Res 2020;494:108069. [PMID: 32563890 DOI: 10.1016/j.carres.2020.108069]
- Ritu, Sharma P, Gupta GD, Asati V. Design, synthesis and antidiabetic study of triazole clubbed indole derivatives as α-glucosidase inhibitors. Bioorg Chem 2023;139:106750. [PMID: 37499530 DOI: 10.1016/j.bioorg.2023.106750]
- Khan S, Iqbal S, Rahim F, Shah M, Hussain R, et al. New biologically hybrid pharmacophore thiazolidinone-based indole derivatives: synthesis, in vitro alpha-amylase and alpha-glucosidase along with molecular docking investigations. Molecules 2022;27(19):6564. [PMID: 36235098 DOI: 10.3390/molecules27196564]
- Khan SA, Ali M, Latif A, Ahmad M, Khan A, et al. Mercaptobenzimidazole-based 1,3-thaizolidin-4-ones as antidiabetic agents: synthesis, in vitro α-glucosidase inhibition activity, and molecular docking studies. ACS Omega 2022;7(32):28041-51. [PMID: 35990459 DOI: 10.1021/acsomega.2c01969]
- Ullah H, Khan S, Rahim F, Taha M, Iqbal R, et al. Benzimidazole bearing thiosemicarbazone derivatives act as potent α-amylase and α-glucosidase inhibitors; synthesis, bioactivity screening and molecular docking study. Molecules 2022;27(20):6921. [PMID: 36296520 DOI: 10.3390/molecules27206921]
- Abdullah MA, Lee YR, Mastuki SN, Leong SW, Wan Ibrahim WN, et al. Development of diarylpentadienone analogues as alpha-glucosidase inhibitor: synthesis, in vitro biological and in vivo toxicity evaluations, and molecular docking analysis. Bioorg Chem 2020;104:104277. [PMID: 32971414 DOI: 10.1016/j.bioorg.2020.104277]
- Hussain R, Rehman W, Khan S, Maalik A, Hefnawy M, et al. Imidazopyridine-based thiazole derivatives as potential antidiabetic agents: synthesis, in vitro bioactivity, and in silico molecular modeling approach. Pharmaceuticals 2023;16(9):1288. [PMID: 37765096 DOI: 10.3390/ph16091288]
- Shayegan N, Iraji A, Bakhshi N, Moazzam A, Faramarzi MA, et al. Design, synthesis, and in silico studies of benzimidazole bearing phenoxyacetamide derivatives as α-glucosidase and α-amylase inhibitors. J Mol Struct 2022;1268:133650. [DOI: 10.1016/j.molstruc.2022.133650]
- Hu C, Liang B, Sun J, Li J, Xiong Z, et al. Synthesis and biological evaluation of indole derivatives containing thiazolidine-2, 4-dione as α-glucosidase inhibitors with antidiabetic activity. Eur J Med Chem 2024;264:115957. [PMID: 38029465 DOI: 10.1016/j.ejmech.2023.115957]
- Khan S, Rahim F, Rehman W, Nawaz M, Taha M, et al. New benzoxazole-based sulphonamide hybrids analogs as potent inhibitors of α-amylase and α-glucosidase: Synthesis and in vitro evaluation along with in silico study. Arab J Chem 2022;15(12):104341. [DOI: 10.1016/j.arabjc.2022.104341]
- Khan I, Rehman W, Rahim F, Hussain R, Khan S, et al. Synthesis, in vitro biological analysis and molecular docking studies of new thiadiazole-based thiourea derivatives as dual inhibitors of a-amylase and a-glucosidase. Arab J Chem 2023;16(9):105078. [DOI: 10.1016/j.arabjc.2023.105078]
- Aroua LM, Alosaimi AH, Alminderej FM, Messaoudi S, Mohammed HA, et al. Synthesis, molecular docking, and bioactivity study of novel hybrid benzimidazole urea derivatives: a promising α-amylase and α-glucosidase inhibitor candidate with antioxidant activity. Pharmaceutics 2023;15(2):457. [PMID: 36839780 DOI: 10.3390/pharmaceutics15020457]
- Hussain R, Rehman W, Rahim F, Khan S, Taha M, et al. Discovery of imidazopyridine derived oxadiazole-based thiourea derivatives as potential anti-diabetic agents: synthesis, in vitro antioxidant screening and in silico molecular modeling approaches. J Mol Struct 2023;1293:136185. [DOI: 10.1016/j.molstruc.2023.136185]
- Hussain R, Iqbal S, Shah M, Rehman W, Khan S, et al. Synthesis of novel benzimidazole-based thiazole derivatives as multipotent inhibitors of α-amylase and α-glucosidase: in vitro evaluation along with molecular docking study. Molecules 2022;27(19):6457. [PMID: 36234994 DOI: 10.3390/molecules27196457]
- Jiménez-Martí E, Hurtado-Genovés G, Aguilar-Ballester M, Martínez-Hervás S, González-Navarro H. Novel therapies for cardiometabolic disease: recent findings in studies with hormone peptide-derived g protein coupled receptor agonists. Nutrients 2022;14(18):3775. [PMID: 36145148 DOI: 10.3390/nu14183775]
- Kameoka J, Tanaka T, Nojima Y, Schlossman SF, Morimoto C. Direct association of adenosine deaminase with a T cell activation antigen, CD26. Science 1993;261(5120):466-9. [PMID: 8101391 DOI: 10.1126/science.8101391]
- Röhrborn D, Wronkowitz N, Eckel J. DPP4 in diabetes. Front Immunol 2015;6:386. [PMID: 26284071 DOI: 10.3389/fimmu.2015.00386]
- Green BD, Flatt PR, Bailey CJ. Dipeptidyl peptidase IV (DPP IV) inhibitors: a newly emerging drug class for the treatment of type 2 diabetes. Diab Vasc Dis Res 2006;3(3):159-65. [PMID: 17160910 DOI: 10.3132/dvdr.2006.024]
- Rashmi R, Bojan Magesh S, Mohanram Ramkumar K, Suryanarayanan S, Venkata SubbaRao M. Antioxidant potential of naringenin helps to protect liver tissue from streptozotocin-induced damage. Rep Biochem Mol Biol 2018;7(1):76-84. [PMID: 30324121]
- Liu F, Wang TTY, Tang Q, Xue C, Li RW, et al. Malvidin 3-glucoside modulated gut microbial dysbiosis and global metabolome disrupted in a murine colitis model induced by dextran sulfate sodium. Mol Nutr Food Res 2019;63(21):e1900455. [PMID: 31444937 DOI: 10.1002/mnfr.201900455]
- Nabavi SF, Braidy N, Gortzi O, Sobarzo-Sanchez E, Daglia M, et al. Luteolin as an anti-inflammatory and neuroprotective agent: a brief review. Brain Res Bull 2015;119:1-11. [PMID: 26361743 DOI: 10.1016/j.brainresbull.2015.09.002]
- Andres S, Pevny S, Ziegenhagen R, Bakhiya N, Schäfer B, et al. Safety aspects of the use of quercetin as a dietary supplement. Mol Nutr Food Res 2018;62(1). [PMID: 29127724 DOI: 10.1002/mnfr.201700447]
- Magnani C, Isaac VL, Correa MA, Salgado HR. Caffeic acid: a review of its potential use in medications and cosmetics. Anal Methods 2014;6(10):3203-10. [DOI: 10.1039/C3ay41807C]
- Saini K, Sharma S, Khan Y. DPP-4 inhibitors for treating T2DM – hype or hope? An analysis based on the current literature. Front Mol Biosci 2023;10:1130625. [PMID: 37287751 DOI: 10.3389/fmolb.2023.1130625]
- Kalra S, Bhattacharya S, Dhingra A, Das S, Kapoor N, et al. Expert consensus on dipeptidyl peptidase-4 inhibitor-based therapies in the modern era of type 2 diabetes mellitus management in India. Cureus 2024;16(6):e61766. [PMID: 38975525 DOI: 10.7759/cureus.61766]
- Lyu YS, Oh S, Kim JH, Kim SY, Jeong MH. Comparison of SGLT2 inhibitors with DPP-4 inhibitors combined with metformin in patients with acute myocardial infarction and diabetes mellitus. Cardiovasc Diabetol 2023;22(1):185. [PMID: 37481509 DOI: 10.1186/s12933-023-01914-4]
- Kazeem IM, Awenegieme NO. Apple and banana displayed better inhibition of dipeptidyl peptidase IV activity than vildagliptin. In: Proceedings of the Annual Meeting of the Japanese Pharmacological Society, WCP2018 (18th World Congress of Basic and Clinical Pharmacology). Japanese Pharmacol Soc 2018;PO1-5-8. [DOI: 10.1254/jpssuppl.WCP2018.0_PO1-5-8]
- Bindu B, Vijayalakshmi S, Manikandan A. Synthesis and discovery of triazolo-pyridazine-6-yl-substituted piperazines as effective anti-diabetic drugs; evaluated over dipeptidyl peptidase-4 inhibition mechanism and insulinotropic activities. Eur J Med Chem 2020;187:111912. [DOI: 10.1016/j.ejmech.2019.111912]
- Ngoc Toan V, Son Hai D, Thi Kim Van H, Minh Tri N, Ngoc Toan D, et al. Design, synthesis, inhibitory activity, and molecular simulations study for d-glucose-conjugated thioureas containing pyrimidine ring as multitarget inhibitors against α-amylase, α-glucosidase, DDP-4, and PTP1B in type 2 diabetes mellitus. RSC Med Chem 2024;15(10):3395-417. [PMID: 39185455 DOI: 10.1039/d4md00334a]
- Huneif MA, Mahnashi MH, Jan MS, Shah M, Almedhesh SA, et al. New succinimide–thiazolidinedione hybrids as multitarget antidiabetic agents: design, synthesis, bioevaluation, and molecular modelling studies. Molecules 2023;28(3):1207. [PMID: 36770873 DOI: 10.3390/molecules28031207]
- Meurot C, Martin C, Sudre L, Breton J, Bougault C, et al. Liraglutide, a glucagon-like peptide 1 receptor agonist, exerts analgesic, anti-inflammatory and anti-degradative actions in osteoarthritis. Sci Rep 2022;12(1):1567. [PMID: 35091584 DOI: 10.1038/s41598-022-05323-7]
- Wang T, Wang J, Hu X, Huang XJ, Chen GX. Current understanding of glucose transporter 4 expression and functional mechanisms. World J Biol Chem 2020;11(3):76-98. [PMID: 33274014 DOI: 10.4331/wjbc.v11.i3.76]
- Chong K, Chang JK, Chuang LM. Recent advances in the treatment of type 2 diabetes mellitus using new drug therapies. Kaohsiung J Med Sci 2024;40(3):212-20. [PMID: 38183334 DOI: 10.1002/kjm2.12800]
- Oh H, Nguyen HD, Yoon IM, Ahn BR, Kim MS. Antidiabetic effect of gemigliptin: a systematic review and meta-analysis of randomized controlled trials with Bayesian inference through a quality management system. Sci Rep 2021;11(1):20938. [PMID: 34686738 DOI: 10.1038/s41598-021-00418-z]
- Kasamatsu H, Chino T, Oyama N, Nakaya T, Hasegawa M. Bullous pemphigoid receiving a novel long-acting dipeptidyl-peptidase-4 (DPP-4) inhibitor omarigliptin in a patient with type 2 diabetes: a case report. J Cutan Immunol Allergy 2021;4(4):89-91. [DOI: 10.1002/cia2.12162]
- Patel BD, Ghate MD. Recent approaches to medicinal chemistry and therapeutic potential of dipeptidyl peptidase-4 (DPP-4) inhibitors. Eur J Med Chem 2014;74:574-605. [PMID: 24531198 DOI: 10.1016/j.ejmech.2013.12.038]
- Madar DJ, Kopecka H, Pireh D, Yong H, Pei Z, et al. Discovery of 2-[4-{{2-(2S, 5R)-2-cyano-5-ethynyl-1-pyrrolidinyl]-2-oxoethyl]amino]-4-methyl-1-piperidinyl]-4-pyridinecarboxylic acid (ABT-279): a very potent, selective, effective, and well-tolerated inhibitor of dipeptidyl peptidase-IV, useful for the treatment of diabetes. J Med Chem 2006;49(21):6416-20. [PMID: 17034148 DOI: 10.1016/B978-0-12-409547-2.12443-6]
- Kuthati Y, Rao VN, Busa P, Wong CS. Teneligliptin exerts antinociceptive effects in rat model of partial sciatic nerve transection induced neuropathic pain. Antioxidants 2021;10(9):1438. [PMID: 34573072 DOI: 10.3390/antiox10091438]
- Hall A, Neelamkavil S, Jones R. CNS, pain, metabolic syndrome, cardiovascular, tissue, fibrosis and urinary incontinence. 2017. Available from: https://www.alkemlabs.com/pdf/adverse/Evogliptin.pdf
- Singla RK, Kumar R, Khan S, Mohit Kumari K, Garg A. Natural products: potential source of DPP-IV inhibitors. Curr Protein Pept Sci 2019;20(12):1218-25. [PMID: 31057098 DOI: 10.2174/1389203720666190502154129]
- Chayah M, Luque-González A, Gómez-Pérez V, Salagre D, Al-Shdaifat A, et al. Synthesis and anti-diabetic activity of an 8-purine derivative as a novel DPP-4 inhibitor in obese diabetic Zücker rats. Drug Des Devel Ther 2024;18:1133-41. [PMID: 38618281 DOI: 10.2147/DDDT.S450917]
- Dastjerdi HF, Naderi N, Nematpour M, Rezaee E, Mahboubi-Rabbani M, et al. Design, synthesis and anti-diabetic activity of novel 1, 2, 3-triazole-5-carboximidamide derivatives as dipeptidyl peptidase-4 inhibitors. J Mol Struct 2020;1221:128745. [DOI: 10.1016/j.molstruc.2020.128745]
- Narsimha S, Battula KS, Ravinder M, Reddy YN, Nagavelli VR. Design, synthesis and biological evaluation of novel 1,2,3-triazole-based xanthine derivatives as DPP-4 inhibitors. J Chem Sci 2020;132:1-9. [DOI: 10.1007/s12039-020-1760-0]
- Katiyar S, Ahmad S, Kumar A, Ansari A, Bisen AC, et al. Design, synthesis, and biological evaluation of 1,4-dihydropyridine-indole as a potential antidiabetic agent via GLUT4 translocation stimulation. J Med Chem 2024;67(14):11957-74. [PMID: 39013034 DOI: 10.1021/acs.jmedchem.4c00570]
- Colin-Lozano B, Torres-Gomez H, Hidalgo-Figueroa S, Chávez-Silva F, Estrada-Soto S, et al. Synthesis, in vitro, in vivo and in silico antidiabetic bioassays of 4-nitro (thio) phenoxyisobutyric acids acting as unexpected PPARγ modulators: an in combo study. Pharmaceuticals 2022;15(1):102. [PMID: 35056159 DOI: 10.3390/ph15010102]
- Wright EM. SGLT2 inhibitors: physiology and pharmacology. Kidney360. 2021;2(12):2027-37. [PMID: 35419546 DOI: 10.34067/KID.0002772021]
- Uitrakul S, Aksonnam K, Srivichai P, Wicheannarat S, Incomenoy S. The incidence and risk factors of urinary tract infection in patients with type 2 diabetes mellitus using SGLT2 inhibitors: a real-world observational study. Medicines 2022;9(12):59. [PMID: 36547992 DOI: 10.3390/medicines9120059]
- Bhattacharya S, Asati V, Mishra M, Das R, Kashaw V, et al. Integrated computational approach on sodium-glucose co-transporter 2 (SGLT2) Inhibitors for the development of novel antidiabetic agents. J Mol Struct 2021;1227:129511. [DOI: 10.1016/j.molstruc.2020.129511]
- Wakisaka M, Nagao T. Sodium glucose cotransporter 2 in mesangial cells and retinal pericytes and its implications for diabetic nephropathy and retinopathy. Glycobiology 2017;27(8):691-5. [PMID: 28535208 DOI: 10.1093/glycob/cwx047]
- Tupas GD, Otero MC, Ebhohimen IE, Egbuna C, Aslam M. Antidiabetic lead compounds and targets for drug development. Phytochem Lead Compounds N Drug Discov 2020;127-41. [DOI: 10.1016/B978-0-12-817890-4.00008-1]
- Ritzel R, Roussel R, Bolli GB, Vinet L, Brulle-Wohlhueter C, et al. Patient-level meta-analysis of the EDITION 1, 2 and 3 studies: glycaemic control and hypoglycaemia with new insulin glargine 300 U/ml versus glargine 100 U/ml in people with type 2 diabetes. Diabetes Obes Metab 2015;17(9):859-67. [PMID: 25929311 DOI: 10.1111/dom.12485]
- Holden SE, Jenkins-Jones S, Morgan CL, Schernthaner G, Currie CJ. Glucose-lowering with exogenous insulin monotherapy in type 2 diabetes: dose association with all-cause mortality, cardiovascular events and cancer. Diabetes Obes Metab 2015;17(4):350-62. [PMID: 25399739 DOI: 10.1111/dom.12412]
- Mashraqi MM, Chaturvedi N, Alam Q, Alshamrani S, Bahnass MM, et al. Biocomputational prediction approach targeting FimH by natural SGLT2 inhibitors: a possible way to overcome the uropathogenic effect of SGLT2 inhibitor drugs. Molecules 2021;26(3):582. [PMID: 33499241 DOI: 10.3390/molecules26030582]
- Dias TR, Alves MG, Casal S, Oliveira PF, Silva BM. Promising potential of dietary (poly)phenolic compounds in the prevention and treatment of diabetes mellitus. Curr Med Chem 2017;24(4):334-54. [PMID: 27593957 DOI: 10.2174/0929867323666160905150419]
- Li C, Luo J, Jiang M, Wang K. The efficacy and safety of the combination therapy with GLP-1 receptor agonists and SGLT-2 inhibitors in type 2 diabetes mellitus: a systematic review and meta-analysis. Front Pharmacol 2022;13:838277. [PMID: 35185588 DOI: 10.3389/fphar.2022.838277]
- Pan HC, Chen JY, Chen HY, Yeh FY, Huang TT, et al. Sodium-glucose cotransport protein 2 inhibitors in patients with type 2 diabetes and acute kidney disease. JAMA Netw Open 2024;7(1):e2350050. [PMID: 38170522 DOI: 10.1001/jamanetworkopen.2023.50050]
- Feng GJ, Guo YF, Tang Y, Li M, Jia Y, et al. Design, synthesis, and biological evaluation of thioglucoside analogues of gliflozin as potent new gliflozin drugs. J Med Chem 2023;66(17):12536-43. [PMID: 37608596 DOI: 10.1021/acs.jmedchem.3c01138]
- Sipos Á, Szennyes E, Hajnal NÉ, Kun S, Szabó KE, et al. Dual-target compounds against type 2 diabetes mellitus: proof of concept for sodium dependent glucose transporter (SGLT) and glycogen phosphorylase (GP) inhibitors. Pharmaceuticals 2021;14(4):364. [PMID: 33920838 DOI: 10.3390/ph14040364]
- Kong YK, Song KS, Jung ME, Kang M, Kim HJ, et al. Discovery of GCC5694A: A potent and selective sodium glucose co-transporter 2 inhibitor for the treatment of type 2 diabetes. Bioorg Med Chem Lett 2022;56:128466. [PMID: 34813882 DOI: 10.1016/j.bmcl.2021.128466]
- Wang S, Chen K, Guo F, Zhu W, Liu C, et al. C–H Glycosylation of native carboxylic acids: discovery of antidiabetic SGLT-2 inhibitors. ACS Cent Sci 2023;9(6):1129-39. [DOI: 10.1021/acscentsci.3c00201]
- Powell D, Zambrowicz B, Morrow L, Beysen C, Hompesch M, et al. OR22-5 Sotagliflozin decreases postprandial glucose and insulin by delaying intestinal glucose absorption. J Endocr Soc 2019;3(suppl 1):OR22-5. [DOI: 10.1210/js.2019-OR22-5]
- Clinical Trial report. Antidiabetic Drugs for Steatotic Liver Disease. Available from: https://clinicaltrials.gov/ct2/show/NCT03646292?term=drugs&cond=diabetic+sglt2&draw=2&rank=9 [Last accessed on 15 Aug 2023].
- Pioglitazon. Available from: https://www.drugbank.ca/drugs/DB01132 [Last accessed on 15 Aug 2023].
- Rosenstock J, Vico M, Wei L, Salsali A, List JF. Effects of dapagliflozin, an SGLT2 inhibitor, on HbA(1c), body weight, and hypoglycemia risk in patients with type 2 diabetes inadequately controlled on pioglitazone monotherapy. Diabetes Care 2012;35(7):1473-8. [PMID: 22446170 DOI: 10.2337/dc11-1693]
- Dobbins RL, O’Connor-Semmes R, Kapur A, Kapitza C, Golor G, et al. Remogliflozin etabonate, a selective inhibitor of the sodium-dependent transporter 2 reduces serum glucose in type 2 diabetes mellitus patients. Diabetes Obes Metab 2012;14(1):15-22. [PMID: 21733056 DOI: 10.1111/j.1463-1326.2011.01462.x]
- Varney MJ, Benovic JL. The role of G protein-coupled receptors and receptor kinases in pancreatic β-cell function and diabetes. Pharmacol Rev 2024;76(2):267-99. [PMID: 38351071 DOI: 10.1124/pharmrev.123.001015]
- Pocai A. G protein-coupled receptors and obesity. Front Endocrinol 2023;14:1301017. [PMID: 38161982 DOI: 10.3389/fendo.2023.1301017]
- Zheng Z, Zong Y, Ma Y, Tian Y, Pang Y, et al. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal Transduct Target Ther 2024;9(1):234. [PMID: 39289339 DOI: 10.1038/s41392-024-01931-z]
- Pola S, Shah SR, Pingali H, Zaware P, Thube B, et al. Discovery of a potent G-protein-coupled receptor 119 agonist for the treatment of type 2 diabetes. Bioorg Med Chem 2021;35:116071. [PMID: 33611013 DOI: 10.1016/j.bmc.2021.116071]
- Wang X, Li X, Wei S, Wang M, Xu Y, et al. Discovery of novel and selective G-protein coupled receptor 120 (GPR120) Agonists for the treatment of type 2 diabetes mellitus. Molecules 2022;27(24):9018. [PMID: 36558150 DOI: 10.3390/molecules27249018]
- Drzazga A, Okulus M, Rychlicka M, Biegała Ł, Gliszczyńska A, et al. Lysophosphatidylcholine containing anisic acid is able to stimulate insulin secretion targeting G protein coupled receptors. Nutrients 2020;12(4):1173. [PMID: 32331428 DOI: 10.3390/nu12041173]
- Bazydlo-Guzenda K, Buda P, Matloka M, Mach M, Stelmach F, et al. CPL207280, a novel G protein–coupled receptor 40/free fatty acid receptor 1–specific agonist, shows a favorable safety profile and exerts antidiabetic effects in type 2 diabetic animals. Mol Pharmacol 2021;100(4):335-47. [PMID: 34349026 DOI: 10.1124/molpharm.121.000260]
- Juárez-Cruz S, Estrada-Soto S, Colín-Lozano B, Marquina-Rodríguez H, Delgado-Aguilar T, et al. Design and synthesis of barbiturates and hydantoins with multitarget antidiabetic effect. J Mex Chem Soc 2024;68(4):609-23. [DOI: 10.29356/jmcs.v68i4.2284]
- Hidalgo-Figueroa S, Rodríguez-Luévano A, Almanza-Pérez JC, Giacoman-Martínez A, Ortiz-Andrade R, et al. Synthesis, molecular docking, dynamic simulation and pharmacological characterization of potent multifunctional agent (dual GPR40-PPARγ agonist) for the treatment of experimental type 2 diabetes. Eur J Pharmacol 2021;907:174244. [PMID: 34116041 DOI: 10.1016/j.ejphar.2021.174244]
- Ye Z, Liu C, Zou F, Cai Y, Chen B, et al. Discovery of novel potent GPR40 agonists containing imidazo[1,2-a]pyridine core as antidiabetic agents. Bioorg Med Chem 2020;28(13):115574. [DOI: 10.1016/j.bmc.2020.115574]
- Bhatt DL, Szarek M, Pitt B, Cannon CP, Leiter LA, et al. Sotagliflozin in patients with diabetes and chronic kidney disease. N Engl J Med 2021;384(2):129-39. [PMID: 33200891 DOI: 10.1056/NEJMoa2030186]
- Aronne LJ, Sattar N, Horn DB, Bays HE, Wharton S, et al. Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: the SURMOUNT-4 randomized clinical trial. JAMA 2024;331(1):38-48. [PMID: 38078870 DOI: 10.1001/jama.2023.24945]
- Ueki K, Tanizawa Y, Nakamura J, Yamada Y, Inagaki N, et al. Long-term safety and efficacy of alogliptin, a DPP-4 inhibitor, in patients with type 2 diabetes: a 3-year prospective, controlled, observational study (J-BRAND Registry). BMJ Open Diabetes Res Care 2021;9(1):e001787. [PMID: 33441417 DOI: 10.1136/bmjdrc-2020-001787]
- Sass F, Ma T, Ekberg JH, Kirigiti M, Ureña MG, Dollet L, et al. NK2R control of energy expenditure and feeding to treat metabolic diseases. Nature 2024;635:1-4. [PMID: 39537932 DOI: 10.1038/s41586-024-08207-0]
- Wang XX, Edelstein MH, Gafter U, Qiu L, Luo Y, et al. G protein-coupled bile acid receptor TGR5 activation inhibits kidney disease in obesity and diabetes. J Am Soc Nephrol 2016;27(5):1362-78. [PMID: 26424786 DOI: 10.1681/ASN.2014121271]