Novel CAR-T Cells Specifically Targeting Nectin4 Exhibit Effective Anti-Tumor Efficacy in Bladder Cancer Cell Lines
1School of Biotechnology and State Key Laboratory of Bioreactor Engineering, East China University of Science and Technology, Shanghai 200237, P.R. China
2Shanghai Yihao Biological Technology Co., Ltd., Shanghai 200231, P.R. China
3Shanghai Key Laboratory of New Drug Design, School of Pharmacy, East China University of Science and Technology, Shanghai 200237, P.R. China
*Correspondence to: Xingyuan Ma, School of Biotechnology and State Key Laboratory of Bioreactor Engineering, East China University of Science and Technology, 130 Meilong Road, Shanghai 200237, P.R. China. E-mail: maxy@ecust.edu.cn; Xiaoli Tian, Shanghai Yihao Biological Technology Co., Ltd., E-mail: ceo@shmlrs.cn; Wenyun Zheng, Shanghai Key Laboratory of New Drug Design, School of Pharmacy, East China University of Science and Technology, E-mail: zwy@ecust.edu.cn
Received: February 25 2025; Revised: March 18 2025; Accepted: April 3 2025; Published Online: April 28 2025.
Cite this paper:
Niu J, Liu G, Zhang S et al. Novel CAR-T Cells Specifically Targeting Nectin4 Exhibit Effective Anti-Tumor Efficacy in Bladder Cancer Cell Lines. BIO Integration 2025; 6: 1–11.
DOI: 10.15212/bioi-2025-0041. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Background: Bladder cancer (BLCA) is one of the most common malignancies and the second most frequent urogenital tract tumor. Cell and gene therapy, which offer many advantages in treating BLCA, are urgently needed. However, there is an important limitation of the currently reported single chain antibody used as a chimeric antigen receptor (CAR) targeting domain. Specifically, CAR aggregation leads to CAR-T depletion, which may originate from the linker peptide and folding stability between the variable domains (VH and VL) of the single chain antibody of the CAR. Humanized, small size, strong affinity single domain antibodies (variable domain of heavy chain of heavy-chain antibody [VHH]) derived from camelids are promising alternatives.
Methods: Second-generation Nectin4-targeted VHH-CAR-T cells were constructed and the specific killing efficacy was determined against BLCA cells in vitro. VHH Nectin4-CAR lentivirus was transduced into human T cells and CAR-T cell phenotypes were analyzed by flow cytometry. Cell killing efficacy was assessed using Nectin4-positive BLCA cells (SW780 and RT4) and Nectin4-negative U87-MG cells as controls using the xCELLigence Real Time Cell Analysis system. Cytokine secretion expression (IFN-γ and IL-2) were measured by an enzyme-linked immunosorbent assay.
Results: VHHNectin4-CAR lentivirus treatment increased the proportion of CD4+ T and memory T cells. VHHNectin4-CAR-T cells had increased specific killing ability compared to the control using VH-VL-based CAR-T cells, specifically recognized Nectin4+ BLCA cells, secreted cytokines, and mediated cell apoptosis. Furthermore, VHHNectin4-CAR-T had no effect on Nectin4– U87-MG cell growth.
Conclusions: VHHNectin4-CAR-T cells were established with potent killing ability that specifically recognized Nectin4+ BLCA cells in vitro.
Statement of Significance: Second-generation targeted Nectin4 VHH-CAR-T cells were established. VHHNectin4-CAR-T cells with strong killing ability specifically recognized Nectin4+ BLCA cells in vitro.
Keywords
Bladder cancer, CAR-T, CGT, Nectin4, VHH.
Introduction
Bladder cancer (BLCA) is the most common urinary tract malignancy and one of the most prevalent cancers worldwide [1]. Although conventional therapies, such as tumor resection, chemotherapy, and radiation, have been proven to be effective, conventional therapies have many limitations, including drug resistance, cancer recurrence, and severe adverse effects [2–4]. Standard therapy for non-muscle invasive bladder cancer, including carcinoma in situ, involves intravesical instillation of bacillus Calmette Guerin, which triggers a local immune reaction that ultimately promotes elimination of BLCA cells [5]. However, BCG treatment of NMIBC has been shown to be ineffective in approximately 40% of patients [6]. Therefore, new therapeutic approaches for the treatment of BLCA are urgently needed.
In recent years new cancer treatments, such as immunotherapy (cell and gene therapy), have emerged. Chimeric antigen receptor (CAR) T-cell therapy is one of the most promising immunotherapeutic approaches. Indeed, six CAR T therapies have been approved by the Food and Drug Administration (FDA) for hematologic malignancies [7, 8]. However, CAR immunotherapy for solid tumors lags significantly behind liquid tumors due to CAR-T cell manufacturing, inefficient CAR-T cell trafficking and infiltration into tumor sites, therapy-associated toxicity [9], and antigen escape [10]. Ciltacabtagene autoleucel was the first variable domain of heavy chain of heavy-chain antibody (VHH)-based CAR-T product approved by the FDA, suggesting that nanobody-based CAR-T products could be an effective means to treat solid tumors [11, 12].
Previous studies have shown that Nectin4 has a pivotal role in the occurrence, invasion, and metastasis of BLCA and is one of the crucial targets for diagnosis and treatment [13]. The Nectin4-specific nanobody was screened from a phage display library constructed as described in our previous study [14]. Then, Nectin4 nanobody was utilized to construct a second-generation recombinant lentiviral CAR plasmid. Subsequently, lentivirus was transferred to cells to obtain Nectin4 targeted VHH-CAR-T (VHHNectin4-CAR-T) cells. VHHNectin4-CAR-T exhibited excellent cytotoxicity and targeting at the cellular level, and specifically killed tumor cells, and released corresponding cytokines, indicating the potential of VHHNectin4-CAR-T for BLCA immunotherapy.
Materials and methods
Cell lines and culture conditions
HEK293T, RT4, SW780, 5637, U87-MG, HGC-27, MDA-MB-231, SW620, ES-2, SKOV-3, and HOS cells were obtained from the Chinese Academy of Cell Bank Sciences (Shanghai, China). The HEK293T cell line was cultured in Dulbecco’s modified DMEM medium (BI, Göttingen, Germany). HGC-27, MB-231, and 5637 cells were maintained in RPMI-1640 medium (Biosera, Cholet, France). SW780 and SW620 were maintained in L-15 medium (Gibco, Grand Island, NY, USA). RT4, ES-2, and SKOV-3 cells were maintained in McCoy’s 5A medium (Gibco). HOS and U87-MG cells were maintained in MEM medium (Gibco). All cell lines were supplemented with 10% heat-inactivated fetal bovine serum (Gibco) and 1% penicillin-streptomycin (Biosera) and cultured in an incubator at 37°C with 5% CO2. The humanized camelids single-domain phage library displays synthesized single-domain antibodies based on camelid VHH frames constructed by our laboratory [14].
CAR construction
Humanized VHHs for anti-Nectin4 VHH-CAR and anti-Nectin4 scFv-CAR were screened from a phage display library constructed, as described in our previous study [14]. scFv derived from hNec4.11 (CN 112088167 A) was incorporated into basic CARs composed of CD8α hinges, CD28 transmembrane domains, and CD28 and CD3ζ signaling domains. The two full-length DNA sequences, which had undergone human codon optimization, were synthesized by Sangon Biotech (Shanghai, China) and subsequently inserted into the lentiviral CAR expression vector (pCDH-CMV-EF1-GFP) via two specific restriction enzyme sites.
Production of lentivirus and T cell transduction
Nectin4 CAR lentivirus vector (pCDH-CMV-EF1-VHH-CAR-GFP and pCDH-CMV-EF1-scFv-CAR-GFP) with package plasmids (PLP1, PLP2, and PLP-VSVG) transfected HEK293T cells using PEI transfection reagent (Polysciences, Warrington, PA, USA) at a ratio of 23.1:16.5:16.5:9.9. The virus-containing supernatants were collected 72 h later, concentrated, and stored at −80 °C.
Fresh blood was obtained from healthy donors with consent and approval of the Ethics Committee. Human peripheral blood mononuclear cells (PBMCs) were isolated over a density gradient using a Ficoll kit (GE, Boston, USA). Next, T cells were enriched with ActSep® CD3/CD28 separation & activation magnetic beads (T&L Biotechnology, Beijing, China) to activate cells for 48 h prior to lentiviral infection. Then, activated T cells were transduced by lentiviral particles at a multiplicity of infection = 20. The cells were continuously cultured for 7 days and the cells were collected for in vitro experiments. Untransduced T cells (NC-T) and transfected GFP lentivirus (Mock-T) were prepared as a negative control and scFv-CAR-T was prepared as a positive control. All T cells were derived from different healthy donors.
All experiments were conducted in accordance with the guidelines and regulations of the Research Ethics Committees of East China University of Science and Technology (Shanghai, China), as approved by the mentioned committees (REC No. 20181223).
Flow cytometry and antibodies
mAbs against Nectin4 antibody (R&D, Minneapolis, MN, USA) and FITC-rabbit anti-goat IgG (H+L) secondary antibody (ABclonal Biotechnology Co., Ltd, Wuhan, China) were used to detect Nectin4 protein expression on cancer cells by flow cytometry. APC-Cy7-CD3, APC-CD4, PE-CD8, brilliant violet 421-CD197 (CCR7), FITC-CD45RA, PE-CD223 (LAG-3), brilliant violet 421-CD366 (Tim-3), and APC-CD279 (PD-1) (Biolegend, San Diego, CA, USA), HA-Tag (C29F4) rabbit mAb (PE conjugate; CST, Boston, Danvers, USA) were used for CAR expression. The cells were rinsed twice with PBS, dyed, and incubated with mAb for 20 min at 4 °C in the dark. The cells were collected for analysis on a flow cytometer (NovoCyte D3000; ACEA, Shanghai, China) and the data were analyzed using NovaExpress software (Shanghai, China).
Cytotoxicity assays
Nectin4-positive BLCA cells (SW780 and RT4) and Nectin4-negative U87-MG cells were cultured at a density of 2 × 104 cells/well in 96-well E-plates (ACEA) at 37 °C in a 5% CO2 incubator overnight, then NC-T cells, mock-T cells, scFv-CAR-T, and VHH-CAR-T cells were co-cultured with target cells at different effector-to-target (E:T) ratios (0.5:1 and 1:1). The viability of target cells was detected using the Real-Time Cell Analysis system [RTCA] (ACEA).
Analysis of cytokine secretion
Tumor cells were co-cultured with NC-T cells, mock-T cells, scFv-CAR-T cells, and VHH-CAR-T cells in 96-well plates for 24 h without exogenous cytokines. Then, the supernatants were collected and cytokines (IFN-γ and TNF-α) were measured using ELISA kits (eBioscience, San Diego, CA, USA) according to the manufacturer’s instructions.
Statistical analysis
Statistical analysis and graph generation were performed using GraphPad Prism software (v.8.0; Boston, MA, USA). All data are expressed as the mean ± SD. All experiments were repeated at least 3 times. Student’s t-test was used to compare the two groups. One- or two-way ANOVA was used for multi-group comparisons to determine statistically significant differences between the samples. A P value < 0.05 was significant and the significance level is shown in figures as *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001.
Results
Establishment of Nectin4 targeting CAR molecules
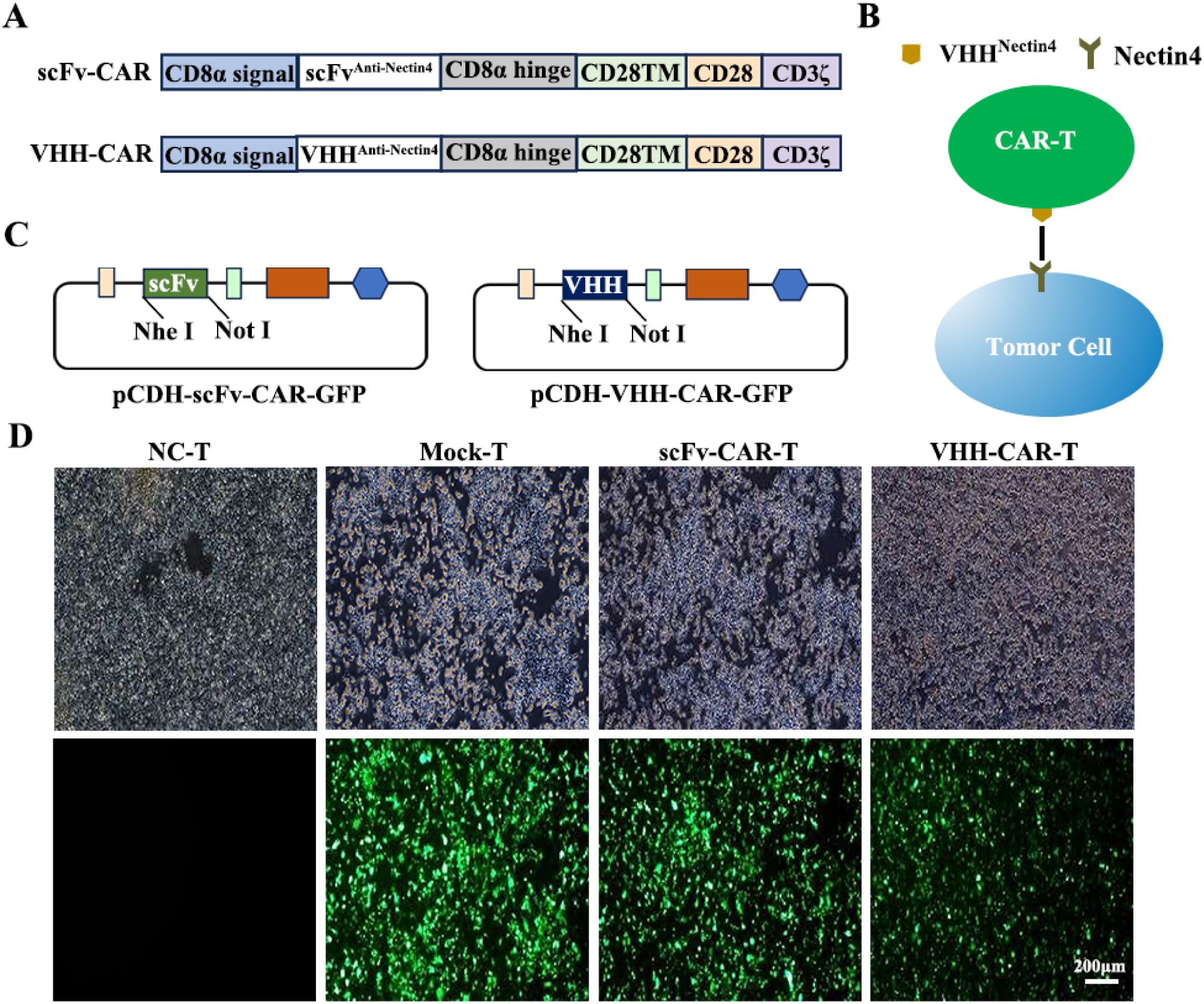
Nectin4 targeting CAR was constructed and anti-Nectin4 scFv/nanoantibody was used to bind antigen outside of the cell. The extracellular antigen binding domain was connected to the intra-cellular domain via a CD8α hinge and a CD28 transmembrane region. The intracellular domain consisted of a CD28 co-stimulatory region and a CD3ζ T-cell activation region. The CARs in the present study were second generation [CAR2G] (Figure 1A and B). scFv-CAR and VHH-CAR DNA sequence with or without an HA tag were synthesized and inserted into the lentiviral vector (pCDH-CMV-EF1α-GFP; Figure S4, Figure 1C). The single-chain variable fragment (scFv)-CAR and VHH-CAR sizes were 1478 and 1148 bp, respectively (Figure S1). The fluorescence pattern and lentivirus titer were determined (Table S1) to verify whether the recombinant lentiviral vector plasmid was successfully transfected and the corresponding lentiviral particles were released. The fluorescence pattern revealed positive CAR expression in HEK-293T cells (Figure 1D), indicating that the recombinant lentiviral vector plasmid was successfully transfected and the corresponding lentiviral particles were released.
Figure 1 Establishment and expression of the Nectin4 targeting CAR molecules. (A) Schematic diagram of CARs, including the CD8 signal peptide (SP), scFv/VHH, CD8α hinge, CD28TM, CD28, and CD3ζ domain. (B) Diagram of a lentiviral vector encoding CARs. (C) Schematic diagram of pCDH-VHH-CAR-GFP and pCDH-scFv-CAR-GFP. (D) GFP expression was observed by fluorescence microscope in HEK-293T cells transfected with NC, mock, scFv-CAR, and VHH-CARs after 72 h. Bar = 200 μm.
Preparation and evaluation of CAR-T cells
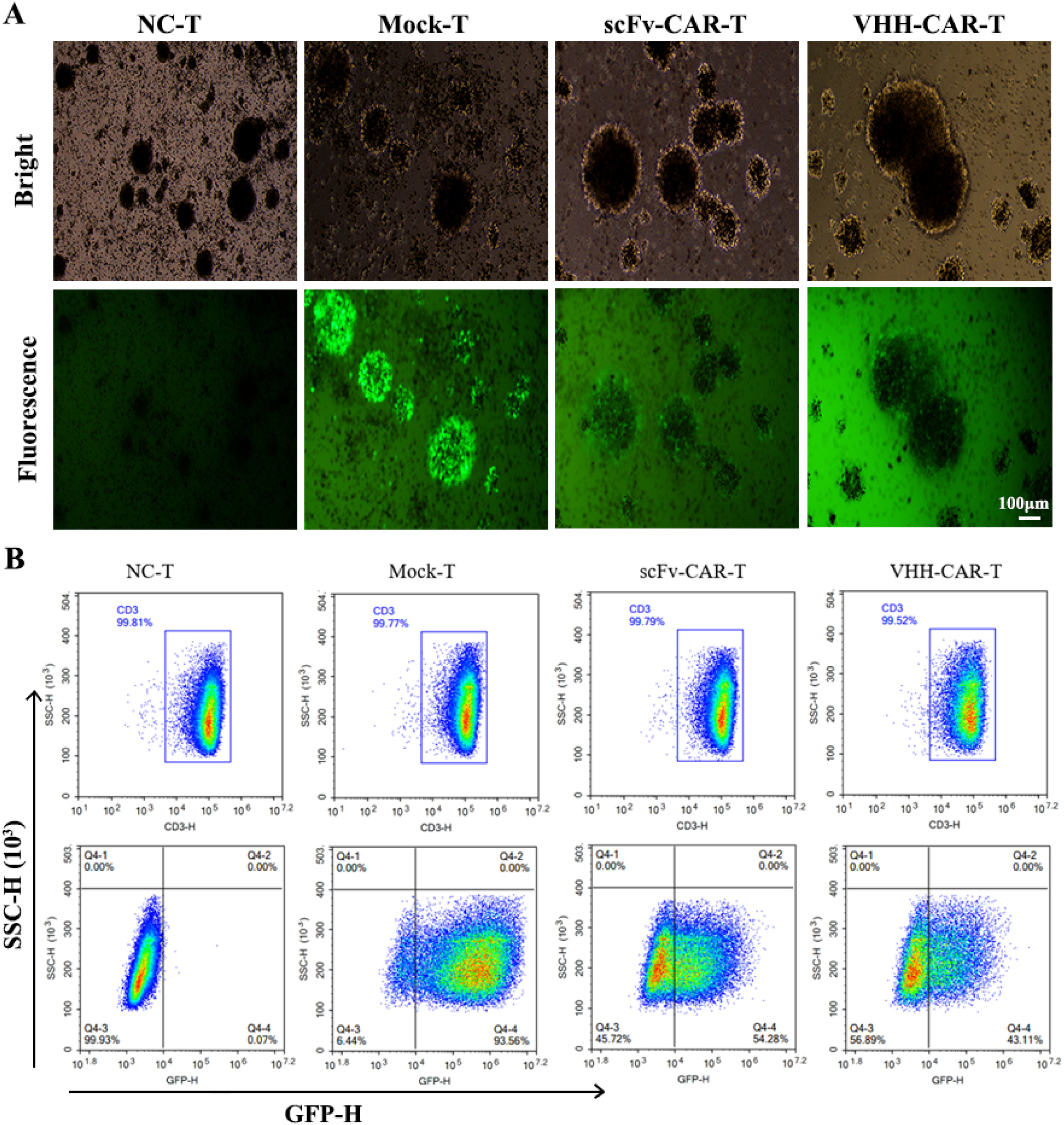
Mock, scFv-CAR, and VHHNectin4-CAR lentiviruses successfully infected T cells to determine whether the prepared CAR was expressed in T cells. As shown in Figure 2A, green fluorescence (GFP) was observed in the lentivirus-infected T cells after 48 h as expected, except in the NC-T cells, suggesting that the three lentiviruses were successfully transfected into T cells. The percentage of CD3+ T cell (Figure 2B) was detected by flow cytometry. The purity of CD3+ T cells was > 99%, indicating that the T cells were specifically proliferating in large numbers. Next, flow cytometry (FCM) showed that the GFP-positive rates in mock-T, scFv-CAR-T, and VHHNectin4-CAR-T cells were 93.56%, 54.28%, and 43.11%, respectively (Figure 2B), which indicated that scFv/VHH affects GFP expression. In addition, the HA rates in VHHNectin4-CAR (HA tag)-T cells were also detected using flow cytometry and the results indicated that the percentages of GFP and HA-positive cells in VHHNectin4-CAR (HA tag) T cells were comparable, suggesting that GFP+ cells effectively represent CAR+ cells (Figure S4).
Figure 2 Generation of Nectin4 CAR-T cells. (A) The fluorescence images of NC-T, mock-T, scFv-CAR-T, and VHH-CAR-T cells transfected by lentiviral vectors after 48 h. (B) The positive rate of GFP in the T cells detected by FCM. Bar = 100 μm.
CAR affects T-cell phenotype
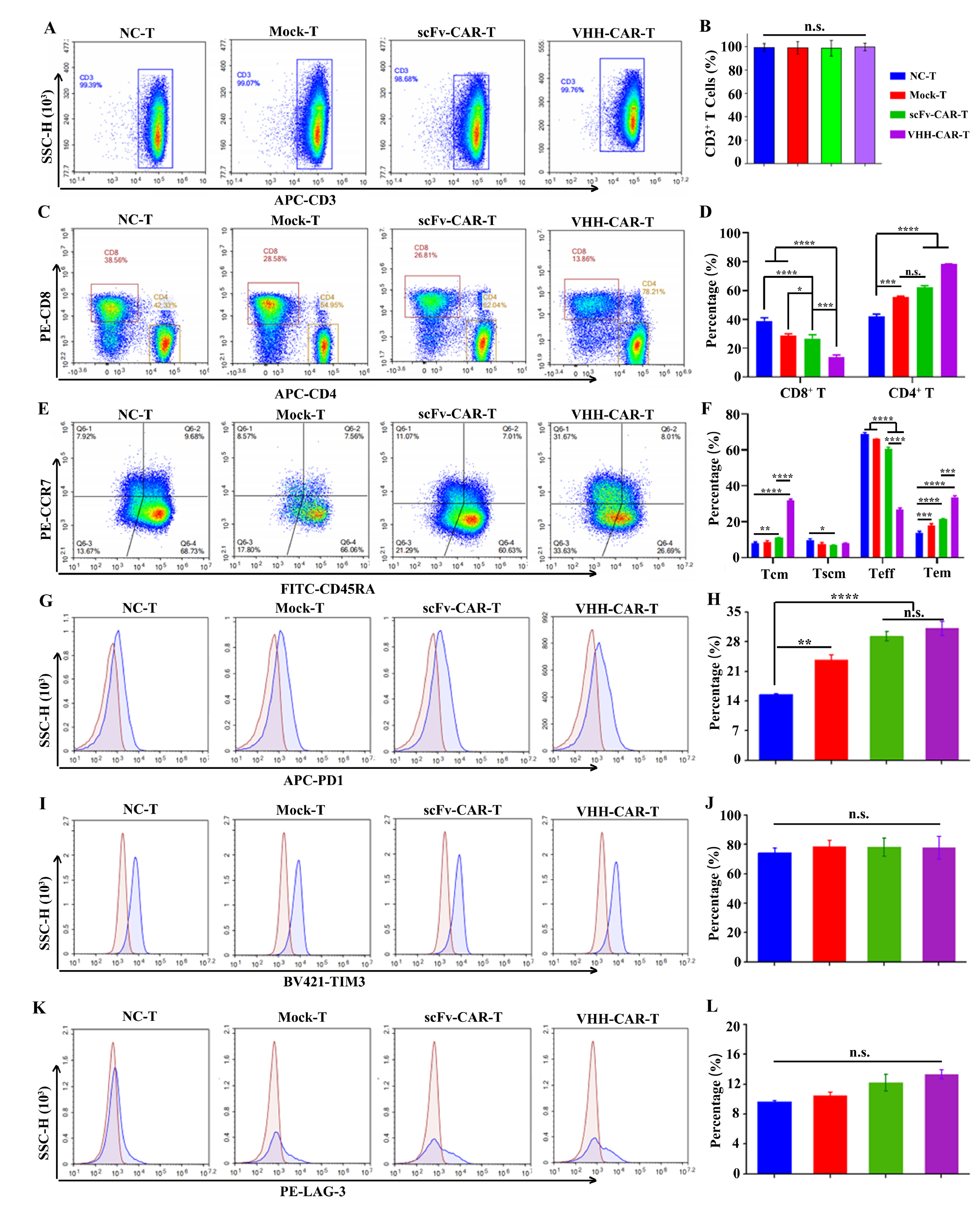
Different T cell typing is significant for CAR-T cell immunotherapy and can significantly influence the effect of immunotherapy [15, 16]. Cell surface marker expression of CAR-T (CD3, CD4, CD8, CD45RA, and CCR7) was detected by FCM to explore the effect of CAR on T cell subtypes. The proportion of CD4+ cells in mock-T, scFv-CAR-T, and VHHNectin4-CAR-T cells was increased and the percentage of CD8+ cells was significantly decreased compared to NC-T cells (Figure 3A–D), indicating that CD4+ and CD8+ T cell immunophenotyping changed under the influence of lentivirus transfection. Compared to mock-T cells, the proportion of CD8+ significantly decreased and the proportion of CD4+ significantly increased in scFv-CAR-T and VHHNectin4-CAR-T cells, suggesting that the recombinant CAR lentivirus had a greater impact on CD4+ and CD8+ T cell immunophenotyping. The proportion of CD8+ T cells decreased and the proportion of CD4+ cells increased significantly in the VHHNectin4-CAR-T group compared to scFv-CAR-T.
Figure 3 Cell phenotype of VHH-CAR-T cell. The ratios of CD3+ (A-B), CD4+/CD8+ (C-D), and CD45RA+/CCR7+ (E-F) in CAR-T and NC-T cells were detected by flow cytometry. PD1 (G-H), TIM3 (I-J), and LAG3 (K-L) expression in CAR-T and NC-T cells was detected by flow cytometry. All data are shown as the mean ± SD (n = 3) and analyzed by Student’s t-test. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001; n.s., not significant.
T cells can be divided into four subsets based on the surface expression of CD45RA and CCR7 [17] during T cell differentiation. Teff cells can proliferate and differentiate after stimulation by an antigen. Contact with host cells and activation of lysosomal enzymes in target cells cause lysis and death of host cells [18]. Therefore, flow cytometry was used to detect immunophenotyping on the surface of T cells. The proportion of Teff cells based on VHHNectin4-CAR-T cell typing was significantly reduced compared to scFv-CAR-T cells and the total number of CD4+ T cells and remaining memory T cells were significantly increased (Figure 3E-F). A previous study showed that CD4+ T cells are essential for formation of cytolytic CD8+ T cells against tumors [19]. The higher proportion of CD4+ T cells in VHHNectin4-CAR-T cells suggested that there might be significant differences in the anti-tumor mechanisms and functions between VHHNectin4-CAR-T and scFv-CAR-T, warranting further investigation.
VHH Nectin4-CAR affects immune checkpoint expression
Emerging research suggests that T-cell activation and associated immune responses are influenced by immune checkpoints [20, 21]. Checkpoint-related protein expression on the surface of T cells (PD-1, TIM-3, and LAG-3) was detected by FCM to determine the effect of CAR on T cell immune checkpoints. PD-1 protein expression on the surface of lentivirus-transfected mock-T, scFv-CAR-T, and VHHNectin4-CAR-T was increased compared to NC-T (Figure 3G-H), suggesting that PD-1 expression on the surface of T cells was affected by VHHNectin4-CAR lentivirus. However, there was no significant differences in the levels of TIM-3 (Figure 3I-J) and LAG-3 expression (Figure 3K-L). These results indicate that the level of PD-1 expression on the surface of T cells was affected by scFv-CAR and VHHNectin4-CAR lentivirus.
VHHNectin4 CAR-T cells display targeted toxicity to BLCA cells
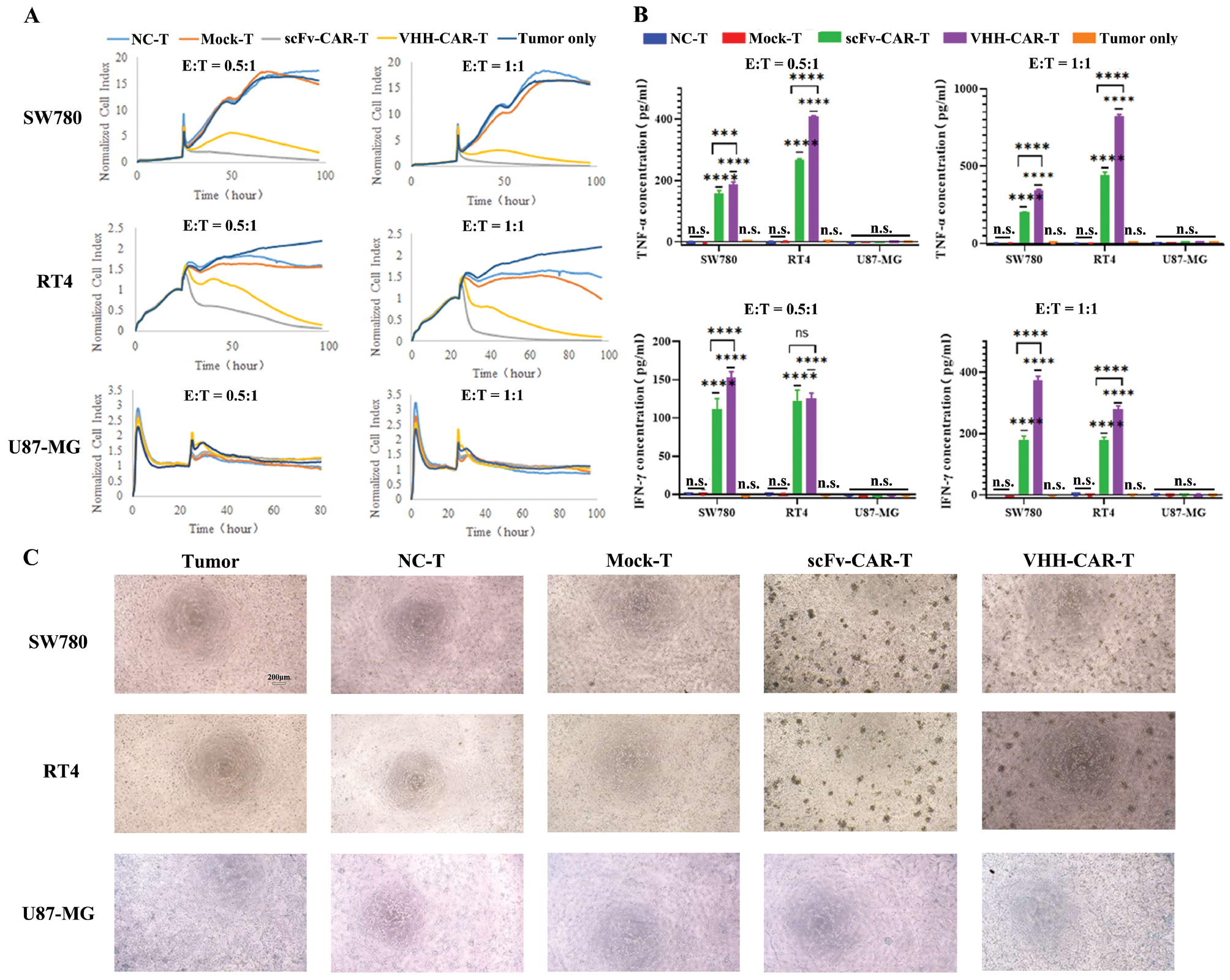
RT4 and SW780 cells were co-cultured with Nectin4 CAR-T cells at different E:T Ratio to determine the presence of targeted toxicity of Nectin4 CAR-T to Nectin4+ tumor cells (Figure S2). The cell index was detected using the RTCA system, which showed that the VHH Nectin4-CAR-T and scFv-CAR-T cell index decreased significantly in RT4 and SW780 cells (Figure 4A). Similarly, the release of cytokines (TNF-α and IFN-γ) was significantly increased in SW780 and RT4 cells treated with VHH Nectin4-CAR-T. In addition, cytokines (TNF-α and IFN-γ) released by the VHH Nectin4-CAR-T cell group was significantly higher than the scFv-CAR-T cell group (Figure 4B). Notably, there was no significant difference in the Nectin4– cell line (U87-MG) and VHH Nectin4-CAR-T and scFv-CAR-T group cell index and cytokine release, indicating that the VHH Nectin4-CAR-T and scFv-CAR-T cells were targeted and unresponsive to Nectin4– cells. The aggregation of T cells to the site of tumor cells signifies the killing ability of T cells [22]. The effects of different T cells on tumor cells were directly observed under a microscope and the results showed that there were obvious T cell clusters in the scFv-CAR-T and VHH-CAR-T groups (Figure 4C). These findings indicate that VHH Nectin4-CAR-T may exhibit strong killing activity against Nectin4+ cancer cells by releasing related cytokines.
Figure 4 Evaluation of the killing ability of VHH-CAR-T cells in vitro. (A) The cytotoxic effect of VHH-CAR-T cells on tumor cells detected using the RTCA system. RT4, SW780, and U87-MG cells were co-cultured with different CAR-T cells for 24 h at different E:T ratios. The cell killing assay was detected using the RTCA system. (B) TNF-α and IFN-γ expression was detected by ELISA analysis. (C) The morphology of different T cells co-cultured with cancer cell lines (SW780, RT4, and U87-MG). All data are shown as the mean ± SD (n = 3) and analyzed using Student’s t-test. ***P < 0.001 and ****P < 0.0001; n.s., not significant.
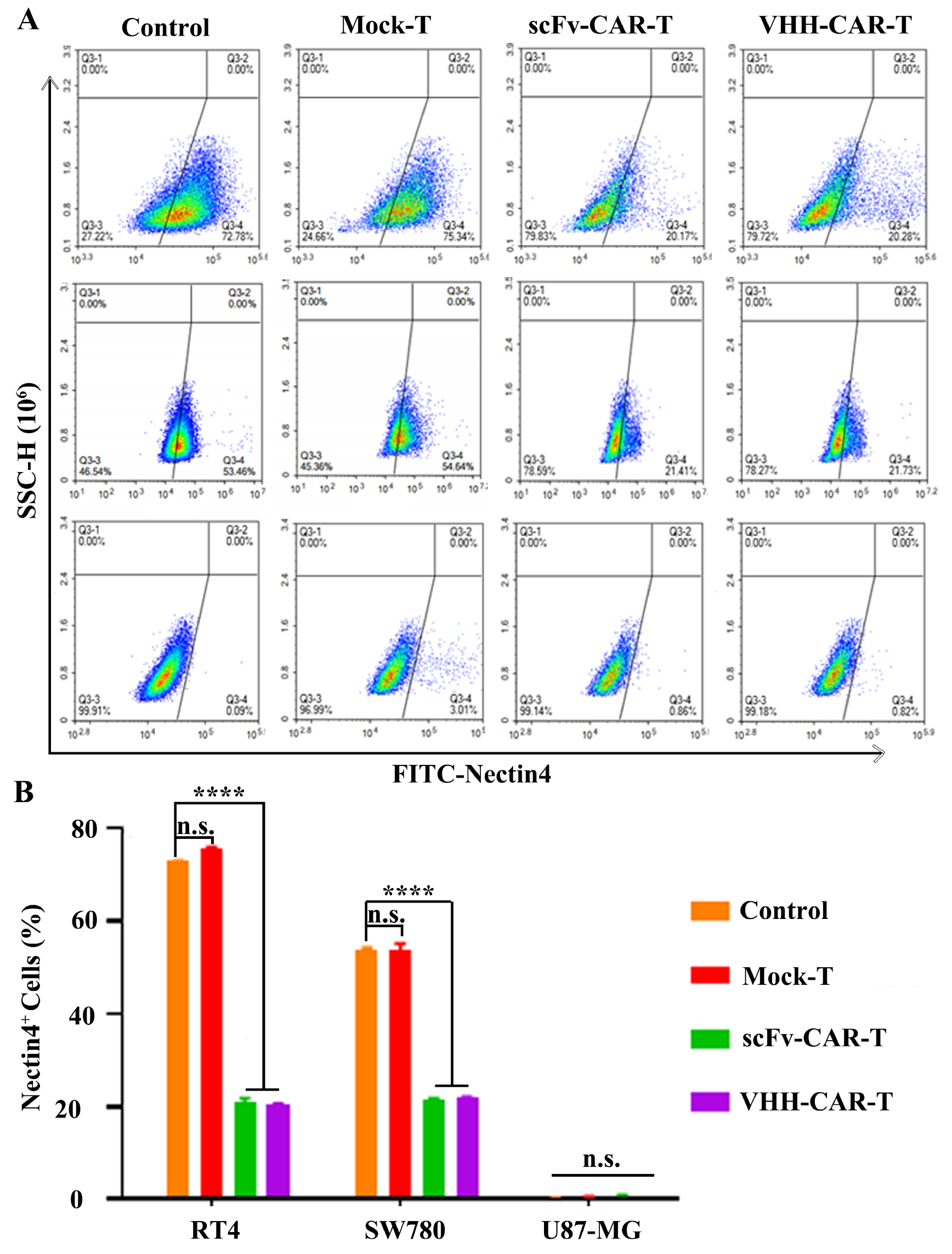
VHH Nectin4-CAR-T reduced Nectin4 expression on the surface of cancer cells
Surface expression of Nectin4 in SW780, RT4, and U87-MG cell lines was examined by flow cytometry after co-incubation with different T cells (Figure 5, Figure S5) to determine the changes in surface Nectin4 expression after co-culturing target cells with different T cells. Nectin4 expression on the surface of SW780 and RT4 cells decreased significantly compared to control and mock-T groups after co-culture with scFv-CAR-T and VHHNectin4-CAR-T cells for 5 h (Figure 5A and B), while there was no significant difference in the Nectin4-negative cell line (U87-MG). The experimental results demonstrated that VHHNectin4-CAR-T and scFv-CAR-T target Nectin4+ cells. Furthermore, the surface antigen of cancer cells was shown to decrease gradually, which may be a mechanism underlying immune escape of cancer cells.
Figure 5 Nectin4 expression on tumor cells surface was detected by FCM. (A) Nectin4 expression on the surface of cancer cells treated with Mock-T, scFv-CAR-T, and VHH-CAR-T cells was detected by FCM. (B) Quantitative analysis of (A). All data are shown as the mean ± SD (n = 3) and analyzed using Student’s t-test. ****P < 0.0001; n.s., not significant.
Discussion
There are currently >1000 CAR-T therapies undergoing clinical trials with the majority targeting relapsed/refractory hematologic malignancies. More and more studies involving solid tumors focus on finding tumor-associated antigens but only a few clinical trials have shown satisfactory results due to severe side effects and toxicity [23]. Li et al. reported that the fourth-generation Nectin4-7.19 CAR-T cells exhibit greater proliferation, migration, and cytotoxicity than second-generation Nectin4 CAR-T cells, in which Nectin4-7.19 CAR-T consist of a scFv derived from an antibody against human Nectin4 [24]. VHHs have also been used as the antigen-recognition domains of CAR-T cells due to the small size, high stability, and ability to recognize hidden antigen epitopes. Safarzadeh Kozani et al. developed a VHH-based CD19-redirected CAR-T cells capable of mediating robust anti-tumor effects [25]. Herein second-generation Nectin4 CAR-T cells are described, in which VHHNectin4 CAR-T consisted of single-domain antibodies (VHHs), which were shown to possess potent proliferation, and cytotoxicity and significant anti-tumor effects in vitro.
Recent reports have revealed the correlation between variations in the function of T cell subpopulations and the efficacy of CAR-T cell immunotherapy [26]. TSCM, expressing a CD45RA+ CCR7+ T population, possesses higher effectiveness and persistence against tumors than TCM [27]. Both CD8+ and CD4+ T subsets exhibit synergistic anti-tumor CAR-T activities because CD4+ cells are conducive to developing CD8+ memory functions [28]. The expression of CAR in the CD4+ T subset was markedly increased, whereas the CD8+ subset significantly decreased. The proportion of Tem subpopulation of VHH-CAR-T was higher than NC-T cells but the VHHNectin4-CAR-T Teff subpopulation decreased compared to NC-T and scFv-CAR-T, which may be related to the weak killing ability of VHHNectin4-CAR-T. Studies have shown that increased memory T-cell (Tem) phenotypic markers may delay CAR T-cell exhaustion [29]. Interestingly, the increased VHHNectin4 CAR-T Tem subpopulation may delay CAR T-cell exhaustion, thereby improving CAR T-cell persistence.
T cells rapidly expand to reach the appropriate number relative to the tumor load after being transported to the tumor and encountering homologous antigens [30, 31]. Immune checkpoints can lead to T cell exhaustion and suppression of immune function [32]. For example, PD-1 inhibits T cell activation after binding to PD-L1 on the tumor [33] and TIM-3 and LAG-3 regulate T cell depletion [34]. TIM-3 and LAG-3 assist tumor cells to inhibit the activity of immune cells and act as a tumor immune checkpoint. The present study showed that there was no significant difference in LAG-3 and TIM-3 expression among the four types of T cells but PD-1 expression in the three types of T cells infected with virus was significantly increased. PD-1 expression was significantly increased in VHHNectin4-CAR-T cells compared to scFv-CAR-T, which could lead to a greater likelihood of VHHNectin4-CAR-T cell failure, which was liin agreement with the results of other CAR-T studies [35].
Enfortumab vedotin (EV) is an antibody-drug conjugate targeting Nectin4 and approved for treatment-refractory metastatic urothelial cancer. Despite the unprecedented success of CAR-T cell therapy in hematologic malignancies, CAR-T cell therapy has not had similar results in solid tumors. Success in this area is hampered by several factors, including the lack of desirable tumor antigens, poor transport of T cells to the tumor, and a hostile tumor microenvironment [36]. In the current study, Nectin4 targeted CAR-T (VHHNectin4-CAR-T) cells was established as a therapeutic target in the clinic setting based on the safety and efficacy of Nectin4 and capability and security was confirmed in vitro. Nanoantibodies (VHH), which are the smallest antigen-binding fragments reported to date, were used instead of traditional single-chain antibodies (scFv). Nanoantibodies have been utilized in CAR-T therapy as the extracellular antigen-binding structural domain of CAR-T. Nanoantibodies have the advantages of small molecular weight, easy expression, strong penetration, good stability, low immunogenicity, and homology with humans [37, 38], demonstrating excellent therapeutic efficacy.
Conclusion
Construction of second-generation VHHNectin4-CAR-T cells was demonstrated in the current study that target Nectin4, which is abnormally expressed on BLCA cell surface but not on normal cell surfaces. Specifically, VHHNectin4-CAR lentivirus treatment increased the proportion of CD4+ T and memory T cells. In vitro experiments demonstrated that VHHNectin4-CAR-T cells with strong specific killing ability can specifically recognize Nectin4+ bladder cancer cells, secrete cytokines, and kill cancer cells. Fortunately, VHHNectin4-CAR-T had no effect on the growth of U87-MG cells due to the lack of Nectin4 expression on the surface of U87-MG cells. Taken together, the data herein will hopefully support the potential of VHHNectin4-CAR-T as a targeted drug for the treatment of BLCA. Moreover, VHH Nectin4-CAR-T cells must be further investigated in preclinical xenograft models of Nectin4+ BLCA before advancing into clinical trials.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Key Research and Development Project of China (2018YFA0902800), the National Natural Science Foundation (32471003), the Open Funding Project of the State Key Laboratory of Bioreactor Engineering, and the Science and Technology Commission of Shanghai Municipality (Grant Nos. 19ZR1454700, 202H1020600, and 220H1210700).
Contributions
XYM, XLT, and WYZ participated in the research design and guidance. JPN and SZ performed the experiments, analyzed the data, and wrote the manuscript. CNX, GDL, and ZWX assisted with the mouse experiments and modified the manuscript. All authors have read the manuscript and approved the content.
Availability of data and materials
All datasets generated and analyzed during this study are included in the published article. Additional data are available from the corresponding author upon reasonable request.
Ethics declarations
Ethics approval and consent to participate
Fresh blood was obtained from healthy donors with their consent after approval by the Ethics Committee.
Consent for publication
Not applicable
Animal Research Statement
Not applicable
Conflict of Interest Statement
The authors declare no competing interests. Guodi Liu is an employee of Shanghai Yihao Biological Technology Co., Ltd.
Supplementary materials
Supplementary Material can be downloaded from https://bio-integration.org/wp-content/uploads/2025/04/bioi20250041_Supplemental.pdf.
Graphical abstract
Highlights
- Engineered novel Nectin4 targeted VHH-CAR-T cells.
- VHHNectin4-CAR-T cells could specifically recognize Nectin4+ BLCA cells, exhibit strong specific killing ability, secrete cytokines, and mediate cell apoptosis.
- VHHNectin4-CAR-T had no effect on the growth of Nectin4− U87-MG cells.
In brief
A novel Nectin4-targeted VHH-CAR-T cell was established. It effectively killed Nectin4+ bladder cancer cells in vitro, with enhanced specific killing and immune-related effects.
References
- Dobruch J, Oszczudłowski M. Bladder cancer: current challenges and future directions. Medicina 2021;57:749. [PMID: 34440955 DOI: 10.3390/medicina57080749]
- Wei Q-Y, Xu Y-M, Lau ATY. Recent progress of nanocarrier-based therapy for solid malignancies. Cancers 2020;12:2783. [PMID: 32998391 DOI: 10.3390/cancers12102783]
- Compérat E, Amin MB, Cathomas R, Choudhury A, De Santis M, et al. Current best practice for bladder cancer: a narrative review of diagnostics and treatments. Lancet 2022;400:1712-21. [PMID: 36174585 DOI: 10.1016/S0140-6736(22)01188-6]
- Shan S, Niu J, Yin R, Shi J, Zhang L, et al. Peroxidase from foxtail millet bran exerts anti-colorectal cancer activity via targeting cell-surface GRP78 to inactivate STAT3 pathway. Acta Pharm Sin B 2022;12:1254-70. [PMID: 35530132 DOI: 10.1016/j.apsb.2021.10.004]
- Seidl C. Targets for therapy of bladder cancer. Semin Nucl Med 2020;50:162-70. [PMID: 32172801 DOI: 10.1053/j.semnuclmed.2020.02.006]
- Weinberg MS, Zafar A, Magdamo C, Chung SY, Chou WH, et al. Association of BCG vaccine treatment with death and dementia in patients with non-muscle-invasive bladder cancer. JAMA Netw Open 2023;6:e2314336. [PMID: 37204792 DOI: 10.1001/jamanetworkopen.2023.14336]
- Ellis GI, Sheppard NC, Riley JL. Genetic engineering of T cells for immunotherapy. Nat Rev Genet 2021;22:427-47. [PMID: 33603158 DOI: 10.1038/s41576-021-00329-9]
- Kumar AR, Devan AR, Nair B, Vinod BS, Nath LR. Harnessing the immune system against cancer: current immunotherapy approaches and therapeutic targets. Mol Biol Rep 2021;48:8075-95. [PMID: 34671902 DOI: 10.1007/s11033-021-06752-9]
- Abbasi S, Totmaj MA, Abbasi M, Hajazimian S, Goleij P, et al. Chimeric antigen receptor T (CAR-T) cells: novel cell therapy for hematological malignancies. Cancer Med 2023;12:7844-58. [PMID: 36583504 DOI: 10.1002/cam4.5551]
- Zhang X, Zhu L, Zhang H, Chen S, Xiao Y. CAR-T cell therapy in hematological malignancies: current opportunities and challenges. Front Immunol 2022;13:927153. [PMID: 35757715 DOI: 10.3389/fimmu.2022.927153]
- Bagchi S, Yuan R, Engleman EG. Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Annu Rev Pathol 2021;16:223-49. [PMID: 3197221 DOI: 10.1146/annurev-pathol-042020-042741]
- Safarzadeh Kozani P, Naseri A, Mirarefin SMJ, Salem F, Nikbakht M, et al. Nanobody-based CAR-T cells for cancer immunotherapy. Biomark Res 2022;10:24. [PMID: 35468841 DOI: 10.1186/s40364-022-00371-7]
- Ghali F, Vakar-Lopez F, Roudier MP, Garcia J, Arora S, et al. Metastatic bladder cancer expression and subcellular localization of nectin-4 and trop-2 in variant histology: a rapid autopsy study. Clin Genitourin Cancer 2023;21:669-78. [PMID: 37344281 DOI: 10.1016/j.clgc.2023.05.014]
- Nie J, Ma X, Hu F, Miao H, Feng X, et al. Designing and constructing a phage display synthesized single domain antibodies library based on camel VHHs frame for screening and identifying humanized TNF-α-specific nanobody. Biomed Pharmacother 2021;137:111328. [PMID: 33571835 DOI: 10.1016/j.biopha.2021.111328]
- Golubovskaya V, Wu L. Different subsets of T cells, memory, effector functions, and CAR-T immunotherapy. Cancers (Basel) 2016;8:36. [PMID: 26999211 DOI: 10.3390/cancers8030036]
- Sethi GS, Gracias DT, Gupta RK, Carr D, Miki H, et al. Anti-CD3 inhibits circulatory and tissue-resident memory CD4 T cells that drive asthma exacerbations in mice. Allergy 2023;78:2168-80. [PMID: 36951658 DOI: 10.1111/all.15722]
- Kunert A, Basak EA, Hurkmans DP, Balcioglu HE, Klaver Y, et al. CD45RA+CCR7– CD8 T cells lacking co-stimulatory receptors demonstrate enhanced frequency in peripheral blood of NSCLC patients responding to nivolumab. J Immunother Cancer 2019;7:149. [PMID: 31176366 DOI: 10.1186/s40425-019-0608-y]
- Zhang Y, Li W, Zhai J, Jin Y, Zhang L, et al. Phenotypic and functional characterizations of CD8+ T cell populations in malignant pleural effusion. Exp Cell Res 2022;417:113212. [PMID: 35588796 DOI: 10.1016/j.yexcr.2022.113212]
- Zander R, Schauder D, Xin G, Nguyen C, Wu X, et al. CD4+ T cell help is required for the formation of a cytolytic CD8+ T cell subset that protects against chronic infection and cancer. Immunity 2019;51:1028-42.e1024. [PMID: 31810883 DOI: 10.1016/j.immuni.2019.10.009]
- Bod L, Kye YC, Shi J, Torlai Triglia E, Schnell A, et al. B-cell-specific checkpoint molecules that regulate anti-tumour immunity. Nature 2023;619:348-56. [PMID: 37344597 DOI: 10.1038/s41586-023-06231-0]
- Datar I, Sanmamed MF, Wang J, Henick BS, Choi J, et al. Expression analysis and significance of PD-1, LAG-3, and TIM-3 in human non-small cell lung cancer using spatially resolved and multiparametric single-cell analysis. Clin Cancer Res 2019;25:4663-73. [PMID: 31053602 DOI: 10.1158/1078-0432.Ccr-18-4142]
- Notarbartolo S, Abrignani S. Human T lymphocytes at tumor sites. Semin Immunopathol 2022;44:883-901. [PMID: 36385379 DOI: 10.1007/s00281-022-00970-4]
- Hartmann J, Schüßler-Lenz M, Bondanza A, Buchholz CJ. Clinical development of CAR T cells-challenges and opportunities in translating innovative treatment concepts. EMBO Mol Med 2017;9:1183-97. [PMID: 28765140 DOI: 10.15252/emmm.201607485]
- Li F, Zhao S, Wei C, Hu Y, Xu T, et al. Development of Nectin4/FAP-targeted CAR-T cells secreting IL-7, CCL19, and IL-12 for malignant solid tumors. Front Immunol 2022;13:958082. [PMID: 36479116 DOI: 10.3389/fimmu.2022.958082]
- Safarzadeh Kozani P, Safarzadeh Kozani P, Rahbarizadeh F. Humanization of the antigen-recognition domain does not impinge on the antigen-binding, cytokine secretion, and antitumor reactivity of humanized nanobody-based CD19-redirected CAR-T cells. J Transl Med 2024;22:679. [PMID: 39054481 DOI: 10.1186/s12967-024-05461-8]
- Klebanoff CA, Scott CD, Leonardi AJ, Yamamoto TN, Cruz AC, et al. Memory T cell-driven differentiation of naive cells impairs adoptive immunotherapy. J Clin Invest 2016;126:318-34. [PMID: 26657860 DOI: 10.1172/jci81217]
- Vahidi Y, Faghih Z, Talei AR, Doroudchi M, Ghaderi A. Memory CD4+ T cell subsets in tumor draining lymph nodes of breast cancer patients: a focus on T stem cell memory cells. Cell Oncol (Dordrecht) 2018;41:1-11. [PMID: 28994018 DOI: 10.1007/s13402-017-0352-6]
- Ahrends T, Busselaar J, Severson TM, Babala N, de Vries E, et al. CD4+ T cell help creates memory CD8+ T cells with innate and help-independent recall capacities. Nat Commun 2019;10:5531. [PMID: 31797935 DOI: 10.1038/s41467-019-13438-1]
- Pietrobon V, Todd LA, Goswami A, Stefanson O, Yang Z, et al. Improving CAR T-cell persistence. Int J Mol Sci 2021;22:10828. [PMID: 4639168 DOI: 10.3390/ijms221910828]
- Adachi K, Kano Y, Nagai T, Okuyama N, Sakoda Y, et al. IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor. Nat Biotechnol 2018;36:346-51. [PMID: 29505028 DOI: 10.1038/nbt.4086]
- Iida Y, Yoshikawa R, Murata A, Kotani H, Kazuki Y, et al. Local injection of CCL19-expressing mesenchymal stem cells augments the therapeutic efficacy of anti-PD-L1 antibody by promoting infiltration of immune cells. J Immunother Cancer 2020;8:e000582. [PMID: 32675195 DOI: 10.1136/jitc-2020-000582]
- Cao T, Zhang W, Wang Q, Wang C, Ma W, et al. Cancer SLC6A6-mediated taurine uptake transactivates immune checkpoint genes and induces exhaustion in CD8+ T cells. Cell 2024;187:2288-304.e27. [PMID: 38565142 DOI: 10.1016/j.cell.2024.03.011]
- Ngiow SF, Young A, Jacquelot N, Yamazaki T, Enot D, et al. A threshold level of intratumor CD8+ T-cell PD1 expression dictates therapeutic response to anti-PD1. Cancer Res 2015;75:3800-11. [PMID: 26208901 DOI: 10.1158/0008-5472.Can-15-1082]
- Kozłowski M, Borzyszkowska D, Cymbaluk-Płoska A. The role of TIM-3 and LAG-3 in the microenvironment and immunotherapy of ovarian cancer. Biomedicines 2022;10:2826. [PMID: 36359346 DOI: 10.3390/biomedicines10112826]
- Pfeifer R, Al Rawashdeh W, Brauner J, Martinez-Osuna M, Lock D, et al. Targeting stage-specific embryonic antigen 4 (SSEA-4) in triple negative breast cancer by CAR T cells results in unexpected on target/off tumor toxicities in mice. Int J Mol Sci 2023;24:9184. [PMID: 37298141 DOI: 10.3390/ijms24119184]
- Singh AK, McGuirk JP. CAR T cells: continuation in a revolution of immunotherapy. Lancet Oncol 2020;21:e168-78. [PMID: 32135120 DOI: 10.1016/s1470-2045(19)30823-x]
- Bao C, Gao Q, Li LL, Han L, Zhang B, et al. The application of nanobody in CAR-T therapy. Biomolecules 2021;11:238. [PMID: 33567640 DOI: 10.3390/biom11020238]
- Berdeja JG, Madduri D, Usmani SZ, Jakubowiak A, Agha M, et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. Lancet 2021;398:314-24. [PMID: 34175021 DOI: 10.1016/s0140-6736(21)00933-8]