Wearable Bioelectronics: Toward Future Personal Health Monitoring
1National & Local Joint Engineering Research Center of Technical Fiber Composites for Safety and Healthy, School of Textile & Clothing, Nantong University, No. 9 Seyuan Road, Nantong 226019, Jiangsu, China
2Department of Mechanical Engineering, Texas A&M University, College Station, TX 77843, USA
3The First Clinical Medicine College, Nanjing Medical University, Nanjing 211166, China
*Correspondence to: Yao Wang, E-mail: a2573300264@163.com; Jie Mei, E-mail: meijie1996@njmu.edu.cn; Xinyang He, E-mail: hexinyang@ntu.edu.cn
Received: December 10 2025; Revised: December 23 2025; Accepted: January 11 2026; Published Online: February 5 2026
Cite this paper:
Li B, Wang Y, Mei J et al. Wearable Bioelectronics: Toward Future Personal Health Monitoring. BIO Integration 2026; 7: 1–10.
DOI: 10.15212/bioi-2025-0219. Available at: https://bio-integration.org/
Download citation
© 2026 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Soft and sustainable wearable bioelectronics are emerging as key platforms for personal health monitoring and digital healthcare. These systems can continuously acquire electrical, mechanical, and biochemical signals during daily life by integrating skin-like soft materials, hydrogel-based interfaces, and environmentally conscious device architectures with wireless communication and data analytics and in some cases, deliver closed-loop interventions. Recent advances have enabled multimodal skin-interfaced patches for chronic disease management, soft cardiovascular and neural interfaces for long-term regulation, and intelligent hydrogel dressings for wound monitoring and therapy. At the same time, concepts of biodegradability, self-healing, and reduced electronic waste are being incorporated to align device lifetimes with therapeutic needs. In this Perspective, these key material and system-level strategies are summarized, representative applications in chronic diseases, cardiovascular and neural regulation, and wound care are highlighted, and the remaining challenges in long-term biocompatibility and stability, data governance and regulation, and the integration of artificial intelligence and miniaturized architectures for future soft bioelectronic systems are discussed.
Keywords
Bioelectronics; digital healthcare; multimodal physiological sensing.
Introduction
Bioelectronic technologies are increasingly moving from hospital equipment to skin-interfaced systems that can follow individuals through everyday life. Wearable bioelectronics, built on recent advances in soft materials, microelectronics, and wireless communication, are shifting health monitoring from occasional, clinic-based measurements to continuous and non-invasive tracking of physiologic and biochemical signals in daily life [1–4]. Such skin-interfaced electronics open a path toward earlier detection of disease exacerbations and more personalized management of chronic cardiovascular, metabolic, and inflammatory conditions by resolving fluctuations in heart rhythm, respiration, motion, or sweat chemistry, and when appropriate, coupling these readouts to electrical, thermal, or pharmacologic interventions. It is worth noting that this vision depends not only on precise sensing but also on long-term comfort, reliability and seamless integration into real-world care pathways, themes that motivated the present work [5–7]. Lab-on-skin platforms have become more capable and are increasingly regarded as key building blocks of precision medicine and digital health infrastructures. However, most commercial wearables still rely on rigid chips, brittle interconnects, aggressive adhesives, and disposable batteries, which can cause motion artefacts, skin irritation, and electronic waste, and typically provide data streams that are rich in quantity but limited in clinical specificity [8–12]. Soft, sustainable, and intelligent skin-interfaced bioelectronics have therefore attracted growing interest as a route to more comfortable, environmentally responsible, and clinically meaningful personal health monitoring platforms [13–20].
In the past decade a wide variety of soft wearable systems that address different elements of this challenge have been reported. Ultrathin “electronic skin” patches, hydrogel-based electrodes, and other organic mixed ionic-electronic conductors reduce mechanical and chemical mismatch at the biotic-abiotic interface, improving signal fidelity during long-term wear [21–26]. Transient and eco-friendly designs that incorporate biodegradable substrates, bioresorbable conductors, and self-powered modules aim to match device lifetime to therapeutic need, while minimizing the burden of device retrieval and end-of-life waste. At the same time, multimodal sensing arrays linked to edge computing and cloud-based analytics are beginning to transform dense, longitudinal bio-signals into digital biomarkers that can trigger timely alerts or closed-loop interventions [27–30]. In this Perspective, there is a focus on these three converging directions, including soft skin-like interfaces, sustainable device architectures, and intelligent data handling, and how the integration across chronic disease management, cardiovascular and neural regulation, and smart wound care could turn current prototypes into practical wearable bioelectronic systems for truly personalized, digitally connected healthcare is discussed. Terminology is used as follows: wearable bioelectronics refers to body-worn systems that interface with skin or tissue for continuous sensing and when relevant, therapeutic actuation; and skin-interfaced bioelectronics denotes the subset designed for conformal contact with skin or wound beds. OMIECs denotes organic mixed ionic-electronic conductors and SaMD denotes software as a medical device.
Many existing reviews discuss soft bio-interfaces, sustainability or transient electronics, and artificial intelligence (AI) or digital-health analytics as separate topics. This Perspective treats such medical devices as a coupled design space and emphasizes how hydrogel and OMIEC interfaces, sustainability constraints, and SaMD-level data governance jointly shape translation from prototypes-to-deployable systems.
Key technologies and advances
This section summarizes the enabling technologies that underpin the three threads outlined in the Introduction: soft skin-like interfaces; sustainable device architectures; and intelligent data handling. First, there is a focus on materials and bio-interfaces that reduce biotic-abiotic mismatch. Then, how sustainability-oriented architectures and system integration are shaping practical wearable platforms are highlighted. The evolution of bioelectronics from rigid, lab-based instruments-to-personal health monitoring platforms has been driven by concurrent advances in soft materials, microfabrication, and energy systems. Wearable bioelectronics now represent the leading edge of this transition, moving from simple activity trackers-to-“lab-on-skin” systems capable of acquiring complex physiologic and biochemical signals in real time. A central theme in this development is the need to reconcile the profound mechanical, electrical, and chemical mismatch between conventional silicon-based electronics and soft, curvilinear, dynamic tissues. This challenge has prompted a shift from rigid chips and thick encapsulants-to-ultrathin, stretchable architectures that approximate the mechanics of skin, allowing conformal adhesion, reduced motion artifacts, and improved comfort during prolonged wear [31–34].
At the materials level, elastomeric substrates, such as PDMS, still provide the basic mechanical framework but the functional core of many contemporary devices is built from intrinsically conductive and ion-permeable components. Hydrogels exemplify such skin-interfacing electrodes because the high water content and tissue-like modulus support efficient ionic conduction and low interfacial impedance, while the softness accommodates repeated deformation without causing irritation [8, 35–37]. The emergence of OMIECs further bridge the gap between the biologic ionic milieu and the electronic domain of sensors and circuits, enabling highly efficient signal transduction in electrophysiology, chemical sensing, and neuromodulation. In parallel, nanomaterials, such as metallic nanoparticles, silver nanowires, carbon nanotubes, graphene, and MXenes, are dispersed within soft matrices to form percolating networks that combine high conductivity with stretchability. The large specific surface area also provides abundant sites for biorecognition, which is crucial for detecting biomarkers at low concentrations [38–41].
Within this broader materials toolbox, hydrogel-based bioelectronics have emerged as a particularly versatile platform. Pristine hydrogels behave as soft electrolytes in which dissolved ions carry charge. Ionic conductivity depends on ion type, concentration, and polymer-network morphology, and is sufficient for low-frequency impedance sensing or as an ionic bridge between metal contacts and tissue. For applications that demand lower impedance and higher current density, including low-noise electrophysiology, high-charge-density stimulation, and dense flexible interconnects, composite hydrogels incorporating PEDOT:PSS, metallic nanowires, carbon nanotubes, graphene, or MXenes provide mixed ionic–electronic conduction over a broad range of moduli [42, 43]. The key design problem is to increase the density of electronic pathways without sacrificing softness, transparency, or long-term stability. In practice, this issue has led to hierarchical filler networks, strong filler–polymer interactions, and tough double-network or organohydrogel architectures that decouple modulus from fracture toughness and maintain stable conductivity under large strains [44, 45].
These principles translate naturally into two archetypal device classes (skin-interfaced patches and hydrogel-based wound dressings). In the former device, hydrogel electrodes and interconnects offer lower contact impedance and fewer motion artifacts than dry or gelled Ag/AgCl counterparts, supporting long-term ECG, EMG, and EEG recordings as well as strain and motion sensing during daily activity [46]. In the wound setting, hydrogels already provide a moist healing environment. Hydrogels become intelligent dressings that can detect infection, report wound status, and deliver therapy in a more controlled fashion when engineered to host embedded pH, temperature, oxygen or glucose sensors, microheaters, and drug reservoirs. Zwitterionic, catechol-functional, and supramolecular formulations further ensure strong, reversible adhesion on wet tissue, while resisting protein fouling and bacterial colonization, which is essential for maintaining signal fidelity and reducing secondary damage during dressing changes [47–49]. Recent work has extended these concepts to multimodal, closed-loop systems in which spatially patterned hydrogel domains simultaneously monitor mechanical, thermal, and biochemical signals and drive actuators, such as heaters or electrical stimulators, under guidance from machine-learning models.
In recent years these advances in soft interfaces have increasingly been viewed through a sustainability lens. Conventional wearable and implantable devices rely heavily on non-degradable plastics, persistent metals, and disposable batteries, raising concerns about electronic waste, long-term biocompatibility, and the environmental footprint of large-scale deployment. As a result, there is growing interest in transient and eco-friendly bioelectronic systems that align device lifetime with therapeutic need, then undergo controlled degradation into benign products. At the structural level, natural polymers, such as cellulose, chitosan, alginate, gelatin, hyaluronic acid, and sericin, together with synthetic degradable matrices, such as poly(lactic acid) [PLA], poly(ε-caprolactone) [PCL], and poly(lactic-co-glycolic acid) [PLGA], serve as hydrogel scaffolds and substrates derived from renewable resources and capable of enzymatic or hydrolytic breakdown [50–52]. Degradable conductors, including bioabsorbable metals (Mg, Zn, Fe, and Mo) and tailored conductive polymers or carbon-based composites, are incorporated to maintain electrical performance during the intended service period, then gradually lose conductivity as the conductors are resorbed [53–55].
Sustainability during use depends not only on end-of-life degradation but also on prolonging functional lifetime and minimizing biofouling. Self-healing hydrogels based on dynamic supramolecular interactions or reversible covalent chemistries can restore mechanical integrity and conductive pathways after damage, reducing device replacement rates. Antimicrobial functions, provided either intrinsically by zwitterionic or cationic polymers or extrinsically by silver nanoparticles, ZnO, MXenes, or photothermal agents, suppress bacterial colonization at skin and wound interfaces and reduce the need for aggressive topical antibiotics [56–58]. It is worth noting that these self-healing and antibacterial features are particularly powerful when integrated into smart wound dressings, in which continuous sensing of pH, temperature, and bacterial burden can trigger local drug release or electrical stimulation, while the material autonomously reseals after mechanical disruption.
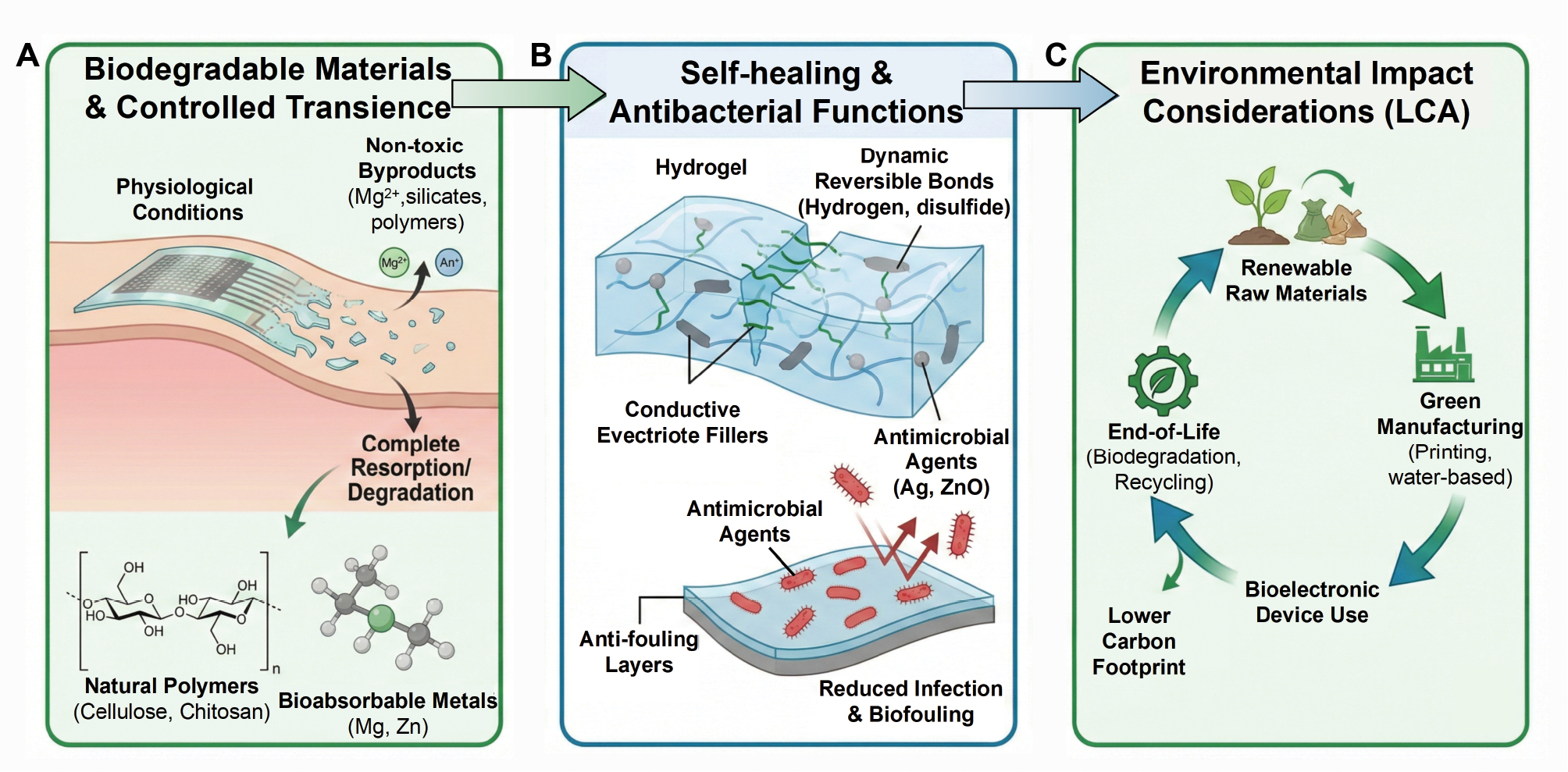
These developments point toward a new generation of soft, sustainable bioelectronic platforms (Figure 1). Conductivity, mechanical compliance, adhesion, degradation time, and antimicrobial activity are treated as coupled variables in this design space rather than isolated targets (Figure 1A). Life cycle considerations are increasingly being considered with traditional performance metrics, such as signal-to-noise ratio or charge injection capacity, from raw material procurement and solvent usage to end-of-life dissolution (Figure 1B) [59–61]. Consequently, the same set of material principles supports a continuum of devices ranging from simple skin electrodes-to-complex, AI-assisted wound dressings and transient implants (Figure 1C). The following sections build on this foundation to examine how these platforms can be deployed in chronic disease management, cardiovascular and neural regulation, and digital wound care.
Figure 1 Sustainable and eco-friendly bioelectronic systems. (A) Biodegradable materials and controlled transience using natural polymers and bioabsorbable metals that resorb into non-toxic by-products. (B) Self-healing and antibacterial hydrogel systems incorporating dynamic reversible bonds, conductive fillers, and antimicrobial/anti-fouling components. (C) Environmental impact considerations and life-cycle assessment, from renewable raw materials and green manufacturing-to-device use and end-of-life degradation or recycling.
Applications for soft and sustainable bioelectronics
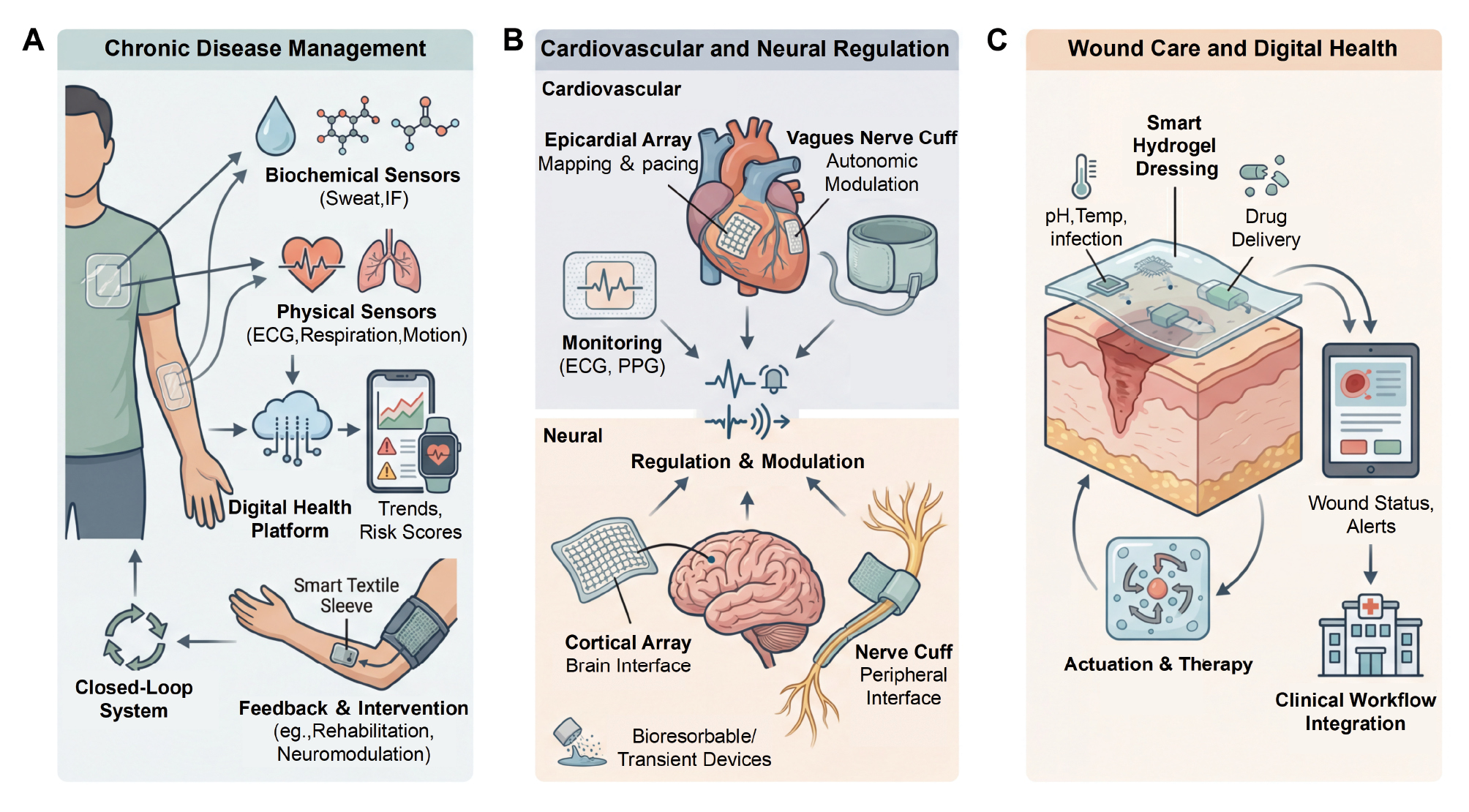
The discussion now translates the enabling materials and system concepts introduced above into application-level needs and clinical or home-care workflows. To keep the narrative concrete, representative use cases are grouped into three domains in which continuous sensing and, feedback and intervention when appropriate, can change outcomes, as follows: chronic disease management; cardiovascular and neural regulation; and wound care within broader digital health infrastructures (Figure 2) [62–64]. In each case, continuous sensing is coupled to different degrees with feedback and intervention, shifting care from episodic monitoring toward more proactive and personalized management.
Figure 2 Applications of soft and sustainable bioelectronics in digital health. (A) Chronic disease management with multimodal skin-interfaced sensors, digital health platforms, and closed-loop intervention. (B) Cardiovascular and neural regulation using soft patches, epicardial arrays, nerve cuffs, and transient implants. (C) Wound care and digital health integration via smart hydrogel dressings that combine sensing, drug delivery, and connected clinical workflows.
For chronic diseases, such as diabetes, heart failure, and chronic obstructive pulmonary disease, important physiologic changes can unfold over hours or days, making short clinic visits a poor sampling strategy (Figure 2A). Skin-interfaced electronics address this mismatch by collecting data continuously during daily life, provided the devices remain comfortable under sweat, movement, and routine handling [62]. Ultra-flexible patches that combine soft elastomer substrates, stretchable interconnects, and on-board processing can record heart rate, respiration, temperature, and motion, while biochemical sensors and microfluidic sampling layers extend monitoring to glucose, lactate, cortisol, and cytokines in sweat, interstitial fluid, and tears [63, 65, 66]. These heterogeneous streams are aggregated on digital health platforms that transform raw time series into trends, alerts, and risk scores. In practice, adherence depends as much on feedback, reminders, and visualization strategies as on sensor precision [45]. When such monitoring is connected to local actuation-electrical stimulation modules, smart textile sleeves or closed-loop drug delivery signals related to joint loading, muscle activity, or metabolic status can be used to guide rehabilitation, neuromodulation, or therapy titration in real time [67–69].
Cardiovascular and neural regulation represent another major application space (Figure 2B). Soft chest and neck patches that integrate stretchable electrodes, strain sensors, and photoplethysmography components enable long-term recording of electrocardiograms, pulse waveforms, and subtle mechanical signatures of cardiac performance in cardiovascular medicine [70]. These platforms support arrhythmia detection, surrogate blood-pressure estimation, and heart-failure monitoring with mechanical compliance and stable skin contact emerging as key requirements for reliable data [71]. In addition to monitoring, flexible epicardial arrays and low-modulus vagus-nerve cuffs made from stretchable conductors and compliant substrates are being explored for direct regulation of cardiac and autonomic function [72, 73]. Here, transient or bioresorbable device concepts are particularly attractive because devices designed to operate for a finite therapeutic period then dissolve can reduce the need for surgical removal and align well with sustainable materials strategies. Neural interfaces impose even stricter constraints because brain and peripheral nerve tissues are highly sensitive to chronic mechanical irritation. Ultrathin polymer substrates, soft encapsulation, and hydrogel coatings are therefore favored for cortical surface arrays, injectable mesh electronics, and peripheral nerve cuffs, which underpin neuromodulation approaches for epilepsy, Parkinson’s disease, chronic pain, and organ control, and provide continuous neural data for personalized computational models of disease dynamics [74].
Wound care offers a third, highly illustrative setting in which hydrogel-based materials and sustainability concerns intersect (Figure 2C). Hydrogels are already widely used as dressings for acute and chronic wounds. Embedding pH, temperature, oxygen, and impedance sensors with heaters, electrical stimulators, and drug reservoirs convert these familiar matrices into smart dressings without disrupting existing clinical workflows [42]. A notable example is the wireless smart bandage that integrates hydrogel electrodes, flexible sensors, and a remotely powered circuit to monitor wound status and deliver electrical cues in a simple feedback loop, accelerating closure and reducing infection in animal models [75]. Building on the wireless smart bandage, conductive hydrogel dressings combined with machine-learning models can classify wound state and suggest or adjust therapy patterns, effectively linking local bioelectronic feedback to wider digital adherence frameworks [43]. Once connected to smartphones and cloud platforms, data from smart dressings, cardiovascular patches, and metabolic sensors are visualized together, enabling dynamic risk assessment for patients who often live with multiple chronic conditions. At the same time, the environmental footprint of frequent dressing changes has prompted strategies that separate reusable electronic modules from single-use hydrogel interfaces or employ bioresorbable conductors and substrates wherever possible, reinforcing the role of wound-care bioelectronics as elements of a resource-aware digital health ecosystem [76, 77].
Several common design principles persist across chronic disease management, cardiovascular and neural regulation, and wound care. These design principles include soft and conformal interfaces, multimodal sensing that covers electrical, mechanical, and biochemical signals, and device life cycles that are acceptable for patients and the environment.
Table 1 brings these aspects together for different application domains and representative device platforms. Table 1 shows that the dominant signals and clinical roles differ from case-to-case but the underlying material choices and architectural strategies are closely related. Taken as a whole, these applications suggest that soft and sustainable bioelectronic platforms can help to move healthcare from occasional and reactive interventions toward more continuous, proactive, and personalized management.
Table 1 Representative Application Domains for Soft and Sustainable Bioelectronics and the Main Characteristics
| Application Domain | Representative Device Platform (Examples, Refs.) | Dominant Signals or Functions | Main Practical Advantages | Refs. |
|---|---|---|---|---|
| Chronic disease management | Skin-interfaced multimodal patches for vital signs and biochemical sensing | Heart rate, activity, temperature, glucose, and metabolites | Long-term wearability; earlier detection of loss of control | [4, 99] |
| Cardiovascular regulation | Soft ECG/PPG chest or neck patches; flexible epicardial arrays | ECG, PPG, pulse waveforms, and local activation maps | Ambulatory rhythm monitoring; support for pacing and mapping | [70, 72] |
| Neural regulation | Flexible cortical and peripheral nerve interfaces | Local field potentials, spikes, and stimulation currents | Circuit-level neuromodulation with improved tissue compatibility | [100] |
| Wound care | Wireless smart hydrogel dressings with integrated sensors and stimulators | pH, temperature, oxygenation, impedance; local stimulation | Early infection detection; guided local therapy; fewer dressing changes | [51, 75] |
| Digital health integration | Cloud-linked multi-sensor systems combining wearables and dressings | Aggregated physiologic and wound-related data streams | Telemedicine, risk stratification, and longitudinal analytics | [101] |
Abbreviations: ECG: electrocardiogram; PPG: photoplethysmography; ROS: reactive oxygen species; pH: acidity.
Challenges and future directions
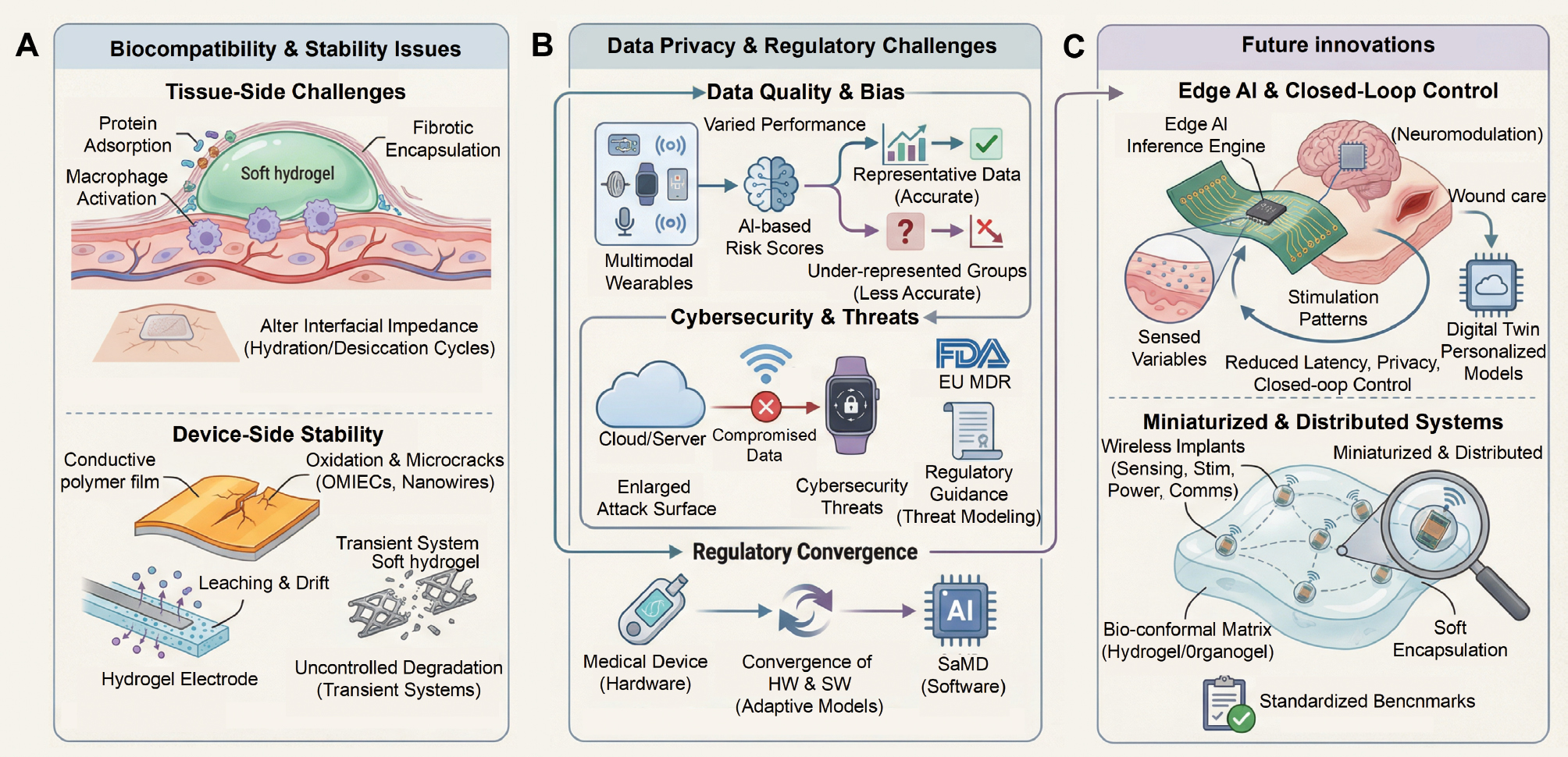
The preceding sections have shown how soft, hydrogel-based, and sustainable bioelectronic platforms can bridge mechanical and chemical gaps at the biotic–abiotic interface and enable applications in chronic disease management, cardiovascular and neural regulation, and smart wound care. Herein these three challenges were used as a simple organizing spine to clarify what currently limits translation and what near-term opportunities could best directly accelerate real-world deployment. Translating these concepts into robust, widely deployed technologies requires confronting three tightly coupled challenges [long-term biocompatibility and stability, data governance and regulation, and the integration of AI and miniaturized architecture; Figure 3] [74].
Figure 3 Challenges and future directions in soft bioelectronics. (A) Biocompatibility and stability issues at the tissue and device sides. (B) Data quality, bias, cybersecurity, and regulatory convergence for multimodal wearables. (C) Future innovations based on edge AI, closed-loop control, and miniaturized, distributed bioelectronic systems.
From a materials and device perspective, soft hydrogels, organogels, and bioresorbable substrates alleviate the modulus mismatch that has historically driven foreign-body responses around rigid implants but do not eliminate biological reactivity. Even mechanically compliant systems must contend with protein adsorption, macrophage activation, fibrotic encapsulation, and microbial colonization over months-to-years, particularly in neural and cardiovascular settings [78]. On the device side, soft conductors, such as OMIECs, metallic nanowire networks, and MXene-based composites, operate in electrolytes or humid environments that promote oxidation, delamination, and microcrack formation under strain [79–81]. Leaching of dopants, swelling-induced microstructural changes, and hydrolysis of crosslinks can cause gradual drift in conductivity and charge-injection capacity in hydrogel electrodes and interconnects. These issues are amplified in transient systems, in which hydrolysable substrates and bioresorbable metals are intentionally designed to degrade; controlling degradation kinetics is essential to avoid premature loss of function or residual fragments persisting longer than intended [82, 83]. Increased biodegradability or programmed transience can trade off against electrical performance and long-term stability. Degradable substrates and encapsulants often increase water and ion permeability, which can accelerate swelling, dopant migration, and corrosion, leading to rising impedance and signal drift. Conversely, strategies that preserve electrical performance (e.g., stronger encapsulation or denser conductive networks) can slow degradation and may leave residues that persist longer than desired. In practice, reliability, stability, durability, and longevity are distinct but intertwined. A device may be mechanically robust yet electrically unstable or stable under benchtop testing but unreliable under the stochastic loads of daily life (Figure 3A) [84]. User comfort and multi-day wearability often set the practical limit for real-world performance. Breathability, sweat and hygiene, skin irritation, and pressure points from stiff or bulky components can reduce adherence during extended use and should be considered with electrical metrics. Therefore, long-term implants need combinations of mechanically matched hydrogels or elastomers, robust encapsulation, graded interfaces, and self-diagnostic circuits that detect drift before catastrophic failure, whereas disposable patches and dressings primarily demand predictable performance over days-to-weeks and benign degradation products.
On the information side, dense longitudinal data streams from multimodal wearables and smart dressings raise questions about data quality, bias, privacy, and cybersecurity (Figure 3B). Performance of clinical and research wearables can vary with skin tone, body habitus, co-morbidities, and contextual factors, so AI-based risk scores or wound-healing classifiers may systematically underperform in under-represented groups if datasets and validation strategies are not carefully designed [85, 86]. Persistent connectivity enlarges the attack surface, and compromised devices can leak sensitive health information, or in extreme cases, be manipulated to disrupt therapy. Regulators have started to respond. For example, the US FDA now treats cybersecurity as a core quality-system consideration and has issued guidance on threat modelling, secure updates, and vulnerability disclosure, while similar expectations are emerging under the EU MDR [87, 88]. At the same time, regulatory pathways are complicated by the convergence of hardware and software. Biosensors embedded in wearables or dressings are regulated as medical devices, whereas AI algorithms that analyze outputs may fall under the category of SaMD with separate requirements for clinical evidence and lifecycle monitoring [89]. Existing frameworks are largely oriented toward “locked” algorithms that do not change after approval and offer limited guidance for adaptive or continuously learning models, even though such models are natural fits for long-term bioelectronic monitoring [90, 91]. Key regulatory questions include how updates are bounded and documented, what update thresholds trigger re-validation, and how each deployed model version is traced to its training data and evidence base for adaptive or continuously learning models. Continuous adaptation also complicates post-market surveillance because algorithm drift can be confounded with sensor drift and population shift. This complication motivates predefined monitoring metrics, clear rollback mechanisms, and secure update pipelines for model deployment. Harmonizing device- and data-centric regulations, clarifying responsibilities among manufacturers, healthcare institutions, and cloud providers, and defining acceptable levels of transparency and explainability for safety-critical decisions remain open tasks.
Looking ahead, several technological trajectories may help address these cross-cutting challenges (Figure 3B). One technological trajectory is the migration of AI from remote servers to the edge of the network, where energy-efficient inference engines embedded in flexible electronics or companion modules can perform on-device feature extraction, anomaly detection, and closed-loop control, thereby reducing latency, bandwidth requirements, and privacy risks [92]. Such edge-AI architectures are particularly attractive for neuromodulation and wound care, in which local feedback between sensed variables and stimulation patterns must operate on timescales that are difficult to achieve through cloud-mediated loops. Local feedback aligns naturally with digital-twin concepts in which personalized models are continuously updated, while only summary parameters are transmitted to remote server. A second direction is the miniaturization and distribution of bioelectronic modules. Next-generation wireless implants aim to integrate sensing, stimulation, energy harvesting, and communication into millimeter-scale packages that function as networks rather than single devices. Advances in hydrogel, organogel, and hybrid gel encapsulation enable these modules to be embedded within soft, tissue-conformal matrices that provide mechanical buffering and in some cases, bioresorbable scaffolds [93–96]. Ultimately, progress will depend not only on new materials and circuits but also on shared evaluation practices. Emerging perspectives in bioelectronic medicine argue for common quantitative benchmarks for reliability, stability, and longevity across device classes with standardized reporting of dataset composition and algorithmic performance. Coupling such benchmarks as life-cycle assessments for materials and environmental impact would help ensure that future soft bioelectronic platforms are not only intelligent and minimally invasive but also safe, equitable, and sustainable.
A closely related extension is oncology, in which soft, skin-conformal platforms can couple longitudinal biomarker sensing with localized actuation to support outpatient or home-based care. Wearable biosensing platforms offer new prospects for tumor management by integrating flexible sensors and stimulators for continuous monitoring and precisely triggered therapy. For example, microneedle-based patches can non-invasively detect tumor markers and use ultrasound or electrical stimulation to trigger drug release, thereby enhancing immune responses. Such devices allow for long-term home-based monitoring, reduce hospital visits, and provide a feasible approach to personalized cancer therapy [97]. Among the devices, the wearable flexible ultrasound microneedle patch (wf-UMP) utilizes ultrasound stimulation and microneedle-delivered piezoelectric nanoparticles for tumor treatment, while enabling real-time monitoring of drug diffusion, reactive oxygen species (ROS) generation, and immune modulation processes [98].
Conclusion
Soft and sustainable bioelectronics are reshaping how the human body is monitored, understood, and treated across timescales from minutes-to-years by enabling continuous acquisition of electrical, mechanical, and biochemical signals during daily life rather than only in brief clinic visits. By tailoring skin-interfaced platforms, hydrogel-based interfaces and environmentally conscious device architectures to approximate the mechanics, hydration, and chemistry of living tissues, similar material and design principles can be deployed across chronic disease management, cardiovascular and neural regulation, and intelligent wound care embedded within broader digital health infrastructures. The central challenge now is to translate these prototypes into robust, manufacturable systems that are acceptable to patients and the environment, relying on materials that combine long-term biocompatibility with programmable degradation, devices that integrate multimodal sensing with closed-loop actuation and on-device processing, and data infrastructures that protect privacy, while still enabling population-level learning. If these technical and regulatory hurdles can be addressed in a coordinated way, soft and sustainable bioelectronics are well-positioned to support a shift from episodic, reactive care toward more continuous, proactive and personalized health management, operating as an unobtrusive yet functionally rich layer within future digital healthcare ecosystems.
Acknowledgement
Drs. He and Mei thank the China Association for Science and Technology’s Young Talent Support Program for Doctoral Students for support. Drs. He and Mei also thank Yao Wang for his contributions and support to this work.
Conflict of interest
Jie Mei is an Editorial Board Member of BIO Integration. He was not involved in the peer-review or handling of the manuscript. The other authors have no other competing interests to disclose.
References
- Jeong JW, Yeo WH, Akhtar A, Norton JJ, Kwack YJ, et al. Materials and optimized designs for human-machine interfaces via epidermal electronics. Adv Mater 2013;25(47):6839-46. [PMID: 24327417 DOI: 10.1002/adma.201301921]
- Ray T, Choi J, Reeder J, Lee SP, Aranyosi AJ, et al. Soft, skin-interfaced wearable systems for sports science and analytics. Curr Opin Biomed Eng 2019;9:47-56. [DOI: 10.1016/J.COBME.2019.01.003]
- Wang Y, Yang Y, Liu M. Electrophoretic deposition of halloysite nanotubes/PVA composite coatings for corrosion protection of metals. Appl Mater Today 2022;29:101657. [DOI: 10.1016/j.apmt.2022.101657]
- Gao W, Emaminejad S, Nyein HYY, Challa S, Chen K, et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016;529(7587):509-14. [PMID: 26819044 DOI: 10.1038/nature16521]
- Stuart T, Hanna J, Gutruf P. Wearable devices for continuous monitoring of biosignals: challenges and opportunities. APL Bioeng 2022;6(2):021502. [PMID: 35464617 DOI: 10.1063/5.0086935]
- Huang G, Chen X, Liao C. AI-driven wearable bioelectronics in digital healthcare. Biosensors (Basel) 2025;15(7):410. [PMID: 40710060 DOI: 10.3390/bios15070410]
- Xiang L, Wang Y, Xia F, Liu F, He D, et al. An epidermal electronic system for physiological information acquisition, processing, and storage with an integrated flash memory array. Sci Adv 2022;8(33):eabp8075. [PMID: 35977018 DOI: 10.1126/sciadv.abp8075]
- Ershad F, Patel S, Yu C. Wearable bioelectronics fabricated in situ on skins. NPJ Flex Electron 2023;7(1):32. [PMID: 38665149 DOI: 10.1038/s41528-023-00265-0]
- Zang J, An Q, Li B, Zhang Z, Gao L, et al. A novel wearable device integrating ECG and PCG for cardiac health monitoring. Microsyst Nanoeng 2025;11(1):7. [PMID: 39814701 DOI: 10.1038/s41378-024-00858-3]
- Sun X, Wu B, Chiang HC, Deng H, Zhang X, et al. Tumour DDR1 promotes collagen fibre alignment to instigate immune exclusion. Nature 2021;599(7886):673-8. [PMID: 34732895 DOI: 10.1038/s41586-021-04057-2]
- Wang L, Feng L, Sun Z, He X, Wang R, et al. Flexible, self-cleaning, and high-performance ceramic nanofiber-based moist-electric generator enabled by interfacial engineering. Sci China Technol Sci 2022;65(2):450-7. [DOI: 10.1007/s11431-021-1969-y]
- Wang L, He X, Hao Y, Zheng M, Wang R, et al. Rational design of stretchable and highly aligned organic/inorganic hybrid nanofiber films for multidirectional strain sensors and solar-driven thermoelectrics. Sci China Mater 2023;66(2):707-15. [DOI: 10.1007/s40843-022-2160-8]
- Liu G, Lv Z, Batool S, Li MZ, Zhao P, et al. Biocompatible material-based flexible biosensors: from materials design to wearable/implantable devices and integrated sensing systems. Small 2023;19(27):e2207879. [PMID: 37009995 DOI: 10.1002/smll.202207879]
- Wang Y, Haick H, Guo S, Wang C, Lee S, et al. Skin bioelectronics towards long-term, continuous health monitoring. Chem Soc Rev 2022;51(9):3759-93. [DOI: 10.1039/D2CS00207H]
- Lyu Q, Gong S, Yin J, Dyson JM, Cheng W. Soft wearable healthcare materials and devices. Adv Healthc Mater 2021;10(17):e2100577. [PMID: 34019737 DOI: 10.1002/adhm.202100577]
- He X, Shi XL, Wu X, Li C, Liu WD, et al. Three-dimensional flexible thermoelectric fabrics for smart wearables. Nat Commun 2025;16(1):2523. [PMID: 40082483 DOI: 10.1038/s41467-025-57889-1]
- He X, Cai J, Liu M, Ni X, Liu W, et al. Multifunctional, wearable, and wireless sensing system via thermoelectric fabrics. Engineering 2024;35:158-67. [DOI: 10.1016/j.eng.2023.05.026]
- He X, Li B, Cai J, Zhang H, Li C, et al. A waterproof, environment-friendly, multifunctional, and stretchable thermoelectric fabric for continuous self-powered personal health signal collection at high humidity. Susmat 2023;3(5):709-20. [DOI: 10.1002/sus2.155]
- He X, Li C, Zhu S, Cai J, Yang G, et al. Layer-by-layer self-assembly of durable, breathable and enhanced performance thermoelectric fabrics for collaborative monitoring of human signal. Chem Eng J 2024;490:151470. [DOI: 10.1016/j.cej.2024.151470]
- He X, Liu M, Cai J, Li Z, Teng Z, et al. Waste cotton-derived fiber-based thermoelectric aerogel for wearable and self-powered temperature-compression strain dual-parameter sensing. Engineering 2024;39:235-43. [DOI: 10.1016/j.eng.2024.01.015]
- Liu Y, Pharr M, Salvatore GA. Lab-on-skin: a review of flexible and stretchable electronics for wearable health monitoring. ACS Nano 2017;11(10):9614-35. [PMID: 28901746 DOI: 10.1021/acsnano.7b04898]
- Hua J, Su M, Sun X, Li J, Sun Y, et al. Hydrogel-based bioelectronics and their applications in health monitoring. Biosensors (Basel) 2023;13(7):696. [PMID: 37504095 DOI: 10.3390/bios13070696]
- Kim DH, Lu N, Ma R, Kim YS, Kim RH, et al. Epidermal electronics. Science 2011;333(6044):838-43. [PMID: 21836009 DOI: 10.1126/science.1206157]
- He X, Zhang X, Zhang H, Li C, Luo Q, et al. Facile fabrication of stretchable and multifunctional thermoelectric composite fabrics with strain-enhanced self-powered sensing performance. Compos Commun 2022;35:101275. [DOI: 10.1016/j.coco.2022.101275]
- He X, Gu J, Hao Y, Zheng M, Wang L, et al. Continuous manufacture of stretchable and integratable thermoelectric nanofiber yarn for human body energy harvesting and self-powered motion detection. Chem Eng J 2022;450:137937. [DOI: 10.1016/j.cej.2022.137937]
- Hao Y, Yan Q, Liu H, He X, Zhang P, et al. A stretchable, breathable, and self-adhesive electronic skin with multimodal sensing capabilities for human-centered healthcare. Adv Funct Mater 2023;33(44):2303881. [DOI: 10.1002/adfm.202303881]
- Hosseini ES, Dervin S, Ganguly P, Dahiya R. Biodegradable materials for sustainable health monitoring devices. ACS Appl Bio Mater 2021;4(1):163-94. [PMID: 33842859 DOI: 10.1021/acsabm.0c01139]
- Song JW, Ryu H, Bai W, Xie Z, Vázquez-Guardado A, et al. Bioresorbable, wireless, and battery-free system for electrotherapy and impedance sensing at wound sites. Sci Adv 2023;9(8):eade4687. [PMID: 36812305 DOI: 10.1126/sciadv.ade4687]
- Gong S, Lu Y, Yin J, Levin A, Cheng W. Materials-driven soft wearable bioelectronics for connected healthcare. Chem Rev 2024;124(2):455-553. [PMID: 38174868 DOI: 10.1021/acs.chemrev.3c00502]
- Matsumura G, Honda S, Kikuchi T, Mizuno Y, Hara H, et al. Real-time personal healthcare data analysis using edge computing for multimodal wearable sensors. Device 2025;3(2):100597. [DOI: 10.1016/j.device.2024.100597]
- Kim S, Kang J, Lee I, Jang J, Park CB, et al. An intrinsically stretchable multi-biochemical sensor for sweat analysis using photo-patternable ecoflex. NPJ Flex Electron 2023;7(1):33. [DOI: 10.1038/s41528-023-00268-x]
- Cheng P, Dai S, Liu Y, Li Y, Hayashi H, et al. An intrinsically stretchable power-source system for bioelectronics. Device 2024;2(1):100216. [DOI: 10.1016/j.device.2023.100216]
- Gong T, Li ZN, Liang H, Li Y, Tang X, et al. High-sensitivity wearable sensor based on a MXene nanochannel self-adhesive hydrogel. ACS Appl Mater Interfaces 2023;15(15):19349-61. [PMID: 37036936 DOI: 10.1021/acsami.3c01748]
- Kwon Y, Kim J, Kim H, Kang TW, Lee J, et al. Printed nanomaterials for all-in-one integrated flexible wearables and bioelectronics. ACS Appl Mater Interfaces 2024;16(49):68016-26. [PMID: 39586587 DOI: 10.1021/acsami.4c17939]
- Wu J, Gu M, Travaglini L, Lauto A, Ta D, et al. Organic mixed ionic–electronic conductors based on tunable and functional poly(3,4-ethylenedioxythiophene) copolymers. ACS Appl Mater Interfaces 2024;16(22):28969-79. [PMID: 38778796 DOI: 10.1021/acsami.4c03229]
- Mei J, Cai Y, Xu R, Li Q, Chu J, et al. Conserved immuno-collagenic subtypes predict response to immune checkpoint blockade. Cancer Commun (Lond) 2024;44(5):554-75. [PMID: 38507505 DOI: 10.1002/cac2.12538]
- He X, Wu C, Zhang T, Chen Z, Wang L, et al. Ultrahigh thermoelectric properties of PEDOT:PSS films by dedoping and π-π overlapping with 4-(1,3-dimethyl-2,3-dihydro-1H-benzoimidazol-2-yl)phenyl)dimethylamine (N-DMBI). Adv Funct Mater 2025;35(43):2506872. [DOI: 10.1002/adfm.202506872]
- Yang H, Li J, Xiao X, Wang J, Li Y, et al. Topographic design in wearable MXene sensors with in-sensor machine learning for full-body avatar reconstruction. Nat Commun 2022;13(1):5311. [PMID: 36085341 DOI: 10.1038/s41467-022-33021-5]
- Zhu Y, Chen B, Liu Y, Tan T, Gao B, et al. Recent advances in conductive hydrogels for electronic skin and healthcare monitoring. Biosensors (Basel) 2025;15(7):463. [PMID: 40710112 DOI: 10.3390/bios15070463]
- Yuk H, Lu B, Zhao X. Hydrogel bioelectronics. Chem Soc Rev 2019;48(6):1642-67. [DOI: 10.1039/C8CS00595H]
- Mo F, Lin Y, Liu Y, Zhou P, Yang J, et al. Advances in ionic conductive hydrogels for skin sensor applications. Mater Sci Eng R Rep 2025;165:100989. [DOI: 10.1016/j.mser.2025.100989]
- Niu Y, Zhao Z, Yang L, Lv D, Sun R, et al. Towards intelligent wound care: hydrogel-based wearable monitoring and therapeutic platforms. Polymers 2025;17(13):1881. [DOI: 10.3390/polym17131881]
- She Y, Liu H, Yuan H, Li Y, Liu X, et al. Artificial intelligence-assisted conductive hydrogel dressings for refractory wounds monitoring. Nanomicro Lett 2025;17(1):319. [PMID: 40603806 DOI: 10.1007/s40820-025-01834-w]
- Zhu Y, Zhang J, Song J, Yang J, Du Z, et al. A multifunctional pro-healing zwitterionic hydrogel for simultaneous optical monitoring of pH and glucose in diabetic wound treatment. Adv Funct Mater 2020;30(6):1905493. [DOI: 10.1002/adfm.201905493]
- Deng D, Liang L, Su K, Gu H, Wang X, et al. Smart hydrogel dressing for machine learning-enabled visual monitoring and promote diabetic wound healing. Nano Today 2025;60:102559. [DOI: 10.1016/j.nantod.2024.102559]
- Wu S, Lu J, Gao H, Yang Y, He N, et al. A multifunctional wearable patch based on hydrogel for strain sensing and epidermal sweat-analyzing. Chem Eng J 2025;521:166828. [DOI: 10.1016/j.cej.2025.166828]
- Yang X, Chai L, Huang Z, Zhu B, Liu H, et al. Smart photonic crystal hydrogels for visual glucose monitoring in diabetic wound healing. J Nanobiotechnology 2024;22(1):618. [PMID: 39395993 DOI: 10.1186/s12951-024-02905-7]
- Wang P, Wang G, Sun G, Bao C, Li Y, et al. A flexible-integrated multimodal hydrogel-based sensing patch. Nanomicro Lett 2025;17(1):156. [PMID: 39982550 DOI: 10.1007/s40820-025-01656-w]
- Iriya R, Braswell B, Mo M, Zhang F, Haydel SE, et al. Deep learning-based culture-free bacteria detection in urine using large-volume microscopy. Biosensors (Basel) 2024;14(2):89. [PMID: 38392008 DOI: 10.3390/bios14020089]
- Choi Y, Koo J, Rogers JA. Inorganic materials for transient electronics in biomedical applications. MRS Bull 2020;45(2):103-12. [DOI: 10.1557/mrs.2020.25]
- Wang C, Sani ES, Gao W. Wearable bioelectronics for chronic wound management. Adv Funct Mater 2022;32(17):2111022. [PMID: 36186921 DOI: 10.1002/adfm.202111022]
- Tan MJ, Owh C, Chee PL, Kyaw AKK, Kai D, et al. Biodegradable electronics: cornerstone for sustainable electronics and transient applications. J Mater Chem C 2016;4(24):5531-58. [DOI: 10.1039/C6TC00678G]
- Song H, Wang H, Gan T, Shi S, Zhou X, et al. Gelatin biogel–liquid metal composite transient circuits for recyclable flexible electronics. Adv Mater Technol 2024;9(14):2301483. [DOI: 10.1002/admt.202301483]
- Quan L, Xin Y, Wu X, Ao Q. Mechanism of self-healing hydrogels and application in tissue engineering. Polymers (Basel) 2022;14(11):2184. [PMID: 35683857 DOI: 10.3390/polym14112184]
- Shi W, Li H, Xu C, Wu G, Chen J, et al. Ag@polydopamine-functionalized borate ester-linked chitosan hydrogel integrates monitoring with wound healing for epidermal sensor. NPJ Flex Electron 2024;8(1):82. [DOI: 10.1038/s41528-024-00366-4]
- Hu XL, Shang Y, Yan KC, Sedgwick AC, Gan HQ, et al. Low-dimensional nanomaterials for antibacterial applications. J Mater Chem B 2021;9(17):3640-61. [DOI: 10.1039/D1TB00033K]
- Li L, Ji X, Chen K. Conductive, self-healing, and antibacterial Ag/MXene-PVA hydrogel as wearable skin-like sensors. J Biomater Appl 2023;37(7):1169-81. [PMID: 36189748 DOI: 10.1177/08853282221131137]
- Jing X, Xu C, Su W, Ding Q, Ye B, et al. Photosensitive and conductive hydrogel induced innerved bone regeneration for infected bone defect repair. Adv Healthc Mater 2023;12(3):e2201349. [PMID: 36325633 DOI: 10.1002/adhm.202201349]
- Zheng Z, Li X, Jiang G, Cheng W, Zhao D, et al. Cellulosic flexible electronic materials: recent advances in structural design, functionalization, and smart applications. Macromol Rapid Commun 2025:e00557. [PMID: 40920433 DOI: 10.1002/marc.202500557]
- Dulal M, Afroj S, Ahn J, Cho Y, Carr C, et al. Toward sustainable wearable electronic textiles. ACS Nano 2022;16(12):19755-88. [PMID: 36449447 DOI: 10.1021/acsnano.2c07723]
- Rizzarelli P, La Carta S, Rapisarda M, Valenti G. Analytical methods in resorbable polymer development and degradation tracking. In:Materials for Biomedical Engineering. Elsevier; 2019. pp. 351-408. [DOI: 10.1016/B978-0-12-818415-8.00013-9]
- Zhu Y, Li J, Kim J, Li S, Zhao Y, et al. Skin-interfaced electronics: a promising and intelligent paradigm for personalized healthcare. Biomaterials 2023;296:122075. [PMID: 36931103 DOI: 10.1016/j.biomaterials.2023.122075]
- Tu J, Flynn CD, Yeom J, Wu Z, Kelley SO, et al. Wearable biomolecular sensing nanotechnologies in chronic disease management. Nat Nanotechnol 2025;20(10):1388-404. [PMID: 41094264 DOI: 10.1038/s41565-025-02010-2]
- Lu T, Lin Q, Yu B, Hu J. A systematic review of strategies in digital technologies for motivating adherence to chronic illness self-care. NPJ Health Syst 2025;2(1):13. [DOI: 10.1038/s44401-025-00017-4]
- Zhao C, Park J, Root SE, Bao Z. Skin-inspired soft bioelectronic materials, devices and systems. Nat Rev Bioeng 2024;2(8):671-90. [DOI: 10.1038/s44222-024-00194-1]
- Cui Y, Chen Q, Bao W, Yao H, Huang Z, et al. Dual-catalytic polymerization of high-performance PEDOT thermoelectric fabrics for self-powered sensing. Chem Eng J 2025;523:168819. [DOI: 10.1016/j.cej.2025.168819]
- Zhang S, Tan R, Meng S, Zhang K, Hu J. Wearable bioelectronics for home-based monitoring and treatment of muscle atrophy. Adv Sci 2025;12(33):e02831. [PMID: 40650709 DOI: 10.1002/advs.202502831]
- Wang HP, Bi ZY, Zhou Y, Zhou YX, Wang ZG, et al. Real-time and wearable functional electrical stimulation system for volitional hand motor function control using the electromyography bridge method. Neural Regen Res 2017;12(1):133-42. [PMID: 28250759 DOI: 10.4103/1673-5374.197139]
- Yang Q, Hu Z, Rogers JA. Functional hydrogel interface materials for advanced bioelectronic devices. Acc Mater Res 2021;2(11):1010-23. [DOI: 10.1021/accountsmr.1c00142]
- Chen S, Qi J, Fan S, Qiao Z, Yeo JC, et al. Flexible wearable sensors for cardiovascular health monitoring. Adv Healthc Mater 2021;10(17):e2100116. [PMID: 33960133 DOI: 10.1002/adhm.202100116]
- Sarna NS, Desai SH, Kaufman BG, Curry NM, Hanna AM, et al. Enhanced and sustained T cell activation in response to fluid shear stress. iScience 2024;27(6):109999. [PMID: 38883838 DOI: 10.1016/j.isci.2024.109999]
- Xu L, Gutbrod SR, Bonifas AP, Su Y, Sulkin MS, et al. 3D multifunctional integumentary membranes for spatiotemporal cardiac measurements and stimulation across the entire epicardium. Nat Commun 2014;5(1):3329. [PMID: 24569383 DOI: 10.1038/ncomms4329]
- Terutsuki D, Yoroizuka H, Osawa Si, Ogihara Y, Abe H, et al. Totally organic hydrogel-based self-closing cuff electrode for vagus nerve stimulation. Adv Healthc Mater 2022;11(23):e2201627. [PMID: 36148587 DOI: 10.1002/adhm.202201627]
- Campbell A, Wu C. Chronically implanted intracranial electrodes: tissue reaction and electrical changes. Micromachines (Basel) 2018;9(9):430. [PMID: 30424363 DOI: 10.3390/mi9090430]
- Jiang Y, Trotsyuk AA, Niu S, Henn D, Chen K, et al. Wireless, closed-loop, smart bandage with integrated sensors and stimulators for advanced wound care and accelerated healing. Nat Biotechnol 2023;41(5):652-62. [PMID: 36424488 DOI: 10.1038/s41587-022-01528-3]
- Smith L, Simpson G, Holt S, Dambha-Miller H. Digital applications to support self-management of multimorbidity: a scoping review. Int J Med Inform 2025;202:105988. [PMID: 40424867 DOI: 10.1016/j.ijmedinf.2025.105988]
- Gagnon MP, Ouellet S, Attisso E, Supper W, Amil S, et al. Wearable devices for supporting chronic disease self-management: scoping review. Interact J Med Res 2024;13(1):e55925. [PMID: 39652850 DOI: 10.2196/55925]
- Wu SZ, Al-Eryani G, Roden DL, Junankar S, Harvey K, et al. A single-cell and spatially resolved atlas of human breast cancers. Nat Genet 2021;53(9):1334-47. [PMID: 34493872 DOI: 10.1038/s41588-021-00911-1]
- Jiao C, Liu J, Yan S, Xu Z, Hou Z, et al. Hydrogel-based soft bioelectronic interfaces and their applications. J Mater Chem C 2025;13(6):2620-45. [DOI: 10.1039/D4TC04150J]
- Lee MY, Lee ES, Ko NY, Kim HJ, Kim DH, et al. Emerging roles of hydrogels, organogels, and their hybrids in soft bioelectronics and bioplatforms. NPJ Biosens 2025;2(1):35. [DOI: 10.1038/s44328-025-00055-w]
- Zhou M, Wu J, Huang L, Miao M, Cui Y, et al. Smart textiles: sustainable, self-powered, portable, and durable wearable textiles for human health monitoring. Chem Eng J 2025;525:170097. [DOI: 10.1016/j.cej.2025.170097]
- Mariello M. Reliability and stability of bioelectronic medicine: a critical and pedagogical perspective. Bioelectron Med 2025;11(1):16. [PMID: 40646664 DOI: 10.1186/s42234-025-00179-4]
- Chen B, Zhu Y, Yu R, Feng Y, Han Z, et al. Recent progress of biomaterial-based hydrogels for wearable and implantable bioelectronics. Gels 2025;11(6):442. [PMID: 40558741 DOI: 10.3390/gels11060442]
- Zhao Z, Fu H, Tang R, Zhang B, Chen Y, et al. Failure mechanisms in flexible electronics. Int J Smart Nano Mater 2023;14(4):510-65. [DOI: 10.1080/19475411.2023.2261775]
- Huhn S, Axt M, Gunga HC, Maggioni MA, Munga S, et al. The impact of wearable technologies in health research: scoping review. JMIR Mhealth Uhealth 2022;10(1):e34384. [PMID: 35076409 DOI: 10.2196/34384]
- Obermeyer Z, Powers B, Vogeli C, Mullainathan S. Dissecting racial bias in an algorithm used to manage the health of populations. Science 2019;366(6464):447-53. [PMID: 31649194 DOI: 10.1126/science.aax2342]
- Pawnikar V, Patel M. Biosensors in wearable medical devices: regulatory framework and compliance across US, EU, and Indian markets. Ann Pharm Fr 2025;83(4):637-48. [PMID: 40020872 DOI: 10.1016/j.pharma.2025.02.007]
- Mihan A, Pandey A, Van Spall HG. Mitigating the risk of artificial intelligence bias in cardiovascular care. Lancet Digit Health 2024;6(10):e749-54. [PMID: 39214762 DOI: 10.1016/S2589-7500(24)00155-9]
- Reddy S. Global harmonization of artificial intelligence-enabled software as a medical device regulation: addressing challenges and unifying standards. Mayo Clin Proc Digit Health 2025;3(1):100191. [PMID: 40207007 DOI: 10.1016/j.mcpdig.2024.100191]
- Benjamens S, Dhunnoo P, Meskó B. The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database. NPJ Digit Med 2020;3(1):118. [PMID: 32984550 DOI: 10.1038/s41746-020-00324-0]
- Vokinger KN, Feuerriegel S, Kesselheim AS. Continual learning in medical devices: FDA’s action plan and beyond. Lancet Digit Health 2021;3(6):e337-8. [PMID: 33933404 DOI: 10.1016/S2589-7500(21)00076-5]
- Shin U, Ding C, Zhu B, Vyza Y, Trouillet A, et al. NeuralTree: a 256-channel 0.227-μJ/class versatile neural activity classification and closed-loop neuromodulation SoC. IEEE J Solid-State Circuits 2022;57(11):3243-57. [PMID: 36744006 DOI: 10.1109/jssc.2022.3204508]
- Seo D, Neely RM, Shen K, Singhal U, Alon E, et al. Wireless recording in the peripheral nervous system with ultrasonic neural dust. Neuron 2016;91(3):529-39. [PMID: 27497221 DOI: 10.1016/j.neuron.2016.06.034]
- Park S, Yuk H, Zhao R, Yim YS, Woldeghebriel EW, et al. Adaptive and multifunctional hydrogel hybrid probes for long-term sensing and modulation of neural activity. Nat Commun 2021;12(1):3435. [PMID: 34103511 DOI: 10.1038/s41467-021-23802-9]
- Wang X, Sun X, Gan D, Soubrier M, Chiang HY, et al. Bioadhesive and conductive hydrogel-integrated brain-machine interfaces for conformal and immune-evasive contact with brain tissue. Matter 2022;5(4):1204-23. [DOI: 10.1016/j.matt.2022.01.012]
- Sun J, Wu X, Zhang X, Huang W, Zhong X, et al. Radiomic model associated with tumor microenvironment predicts immunotherapy response and prognosis in patients with locoregionally advanced nasopharyngeal carcinoma. Research (Wash D C) 2025;8:0749. [PMID: 40556946 DOI: 10.34133/research.0749]
- Zhang Z, Zhu Z, Zhou P, Zou Y, Yang J, et al. Soft bioelectronics for therapeutics. ACS Nano 2023;17(18):17634-67. [PMID: 37677154 DOI: 10.1021/acsnano.3c02513]
- Xue H, Jin J, Huang X, Tan Z, Zeng Y, et al. Wearable flexible ultrasound microneedle patch for cancer immunotherapy. Nat Commun 2025;16(1):2650. [PMID: 40102412 DOI: 10.1038/s41467-025-58075-z]
- Kim J, Campbell AS, de Ávila BEF, Wang J. Wearable biosensors for healthcare monitoring. Nat Biotechnol 2019;37(4):389-406. [PMID: 30804534 DOI: 10.1038/s41587-019-0045-y]
- Song E, Li J, Won SM, Bai W, Rogers JA. Materials for flexible bioelectronic systems as chronic neural interfaces. Nat Mater 2020;19(6):590-603. [PMID: 32461684 DOI: 10.1038/s41563-020-0679-7]
- Zhu P, Peng H, Rwei AY. Flexible, wearable biosensors for digital health. Med Nov Technol Devices 2022;14:100118. [DOI: 10.1016/j.medntd.2022.100118]