WukongBot: A Brain-Spine Interface Proof of Concept Based on Non-Invasive Brain-Computer Interface for Spinal Cord Injury in a Monkey
1Sanya Research Institute of Hainan University, School of Biomedical Engineering, Hainan University, Sanya, China
2School of Information and Communication Engineering, Hainan University, Haikou, China
3Department of Neurosurgery, Beijing Tiantan Hospital, Capital Medical University, Beijing, China
4Department of Rehabilitation Medicine, Affiliated Haikou Hospital of Xiangya Medical College, Central South University, Haikou, China
5Rishena Medical Co., Ltd., Changzhou, China
aCo-first authors.
*Correspondence to: Fengyan Liang, E-mail: fyliang@hainanu.edu.cn; Qianqian Fan, E-mail: 15808943407@163.com
Received: September 26 2025; Revised: October 18 2025; Accepted: October 26 2025; Published Online: November 11 2025
Cite this paper:
Mo L, Chen L, Liao Y et al. WukongBot: A Brain-Spine Interface Proof of Concept Based on Non-Invasive Brain-Computer Interface for Spinal Cord Injury in a Monkey. BIO Integration 2025; 6: 1–3.
DOI: 10.15212/bioi-2025-0171. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Motor dysfunction constitutes a significant global public health burden. In China, greater than 20 million individuals have various forms of paralysis with stroke and spinal cord injury (SCI) being the leading causes. Epidemiologic data indicate that there are approximately 3.74 million existing SCI cases in China with approximately 90,000 new cases reported annually. Restoration of autonomous walking ability remains one of the most pressing rehabilitation goals for paralyzed patients.
In recent years epidural electrical stimulation (EES) has emerged as a promising intervention for functional recovery in SCI [1–3]. Existing studies have shown that EES can promote the recovery of voluntary motor control in SCI by facilitating motor neuron function and re-establishing functional connections between the brain and spinal cord, thereby restoring autonomous movement [4].
Concurrently, brain–spinal interfaces (BSIs) have established a direct information bridge between the brain and spinal cord. In 2023 the world’s first clinical-grade BSI system, which combined the invasive brain–computer interface (BCI) technology with EES, enabled an individual with an SCI to regain voluntary locomotion and enhance rehabilitation training outcomes. However, this invasive approach poses potential surgical risks, such as infection, and the neural decoding accuracy and real-time performance need further improvement [5].
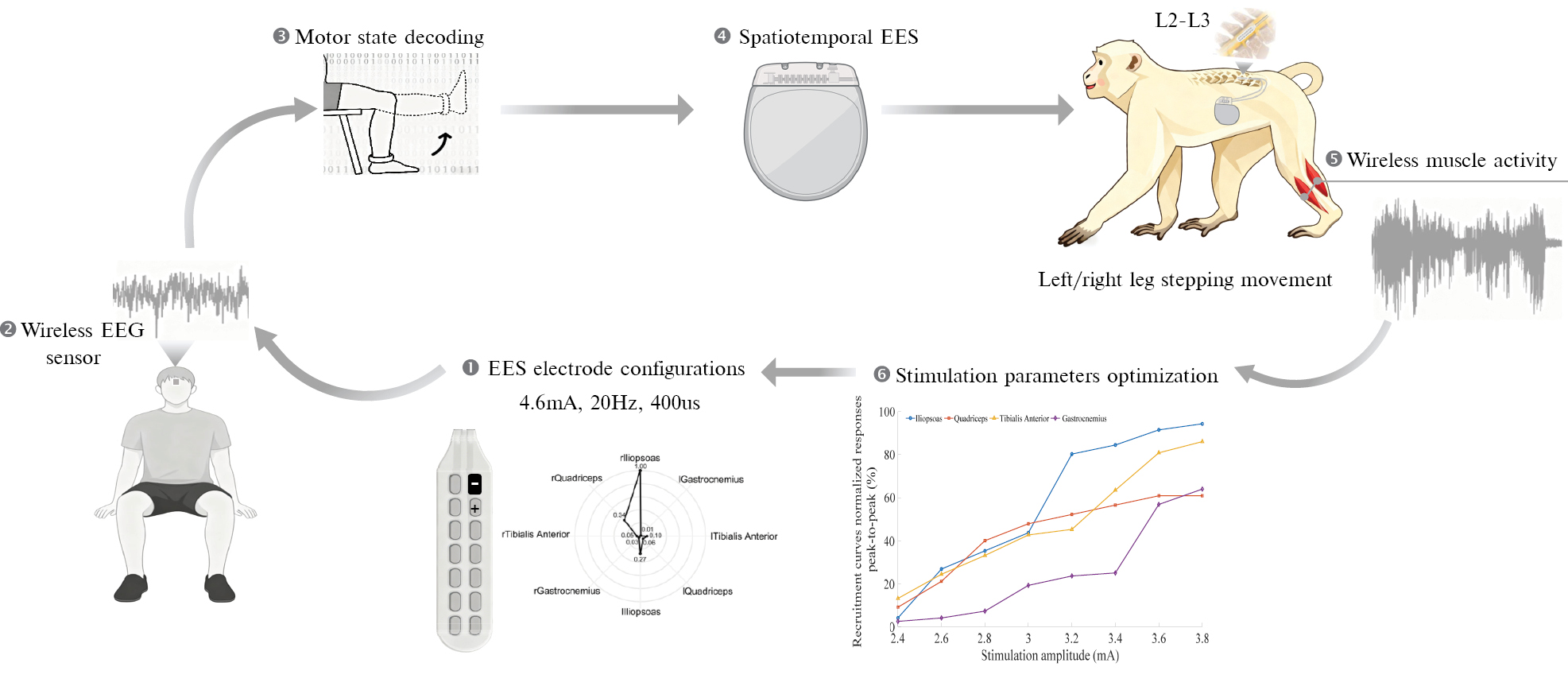
Nevertheless, current invasive approaches face limitations in terms of safety and clinical translatability. Therefore, we proposed a BSI design based on a single portable, non-invasive electroencephalography (EEG) sensor and conducted a preclinical validation and evaluation using a human–monkey experiment (Figure 1). An anesthetized macaque successfully exhibited left and right leg stepping-like movements controlled by human motion intentions. To the best of our knowledge, this is the first BSI proof-of-concept based on non-invasive BCI that controls EES with motion intentions to elicit locomotor activities.
| What is the cross-disciplinary question being addressed? |
| How can the integration of neuroscience, biomedical engineering, and machine learning facilitate the development of a brain-spine interface (BSI) based on a non-invasive brain-computer interface (BCI) to restore motor function in subjects with spinal cord injury (SCI)? |
| What is the main finding? |
| The novel BSI based on a non-invasive BCI enables the control of lower limb movements in a macaque using human motion intentions, achieving left and right leg stepping-like movement under anesthesia. |
Figure 1 Overflow of the BSI system. The anesthetized macaque successfully exhibited left and right leg stepping-like movements controlled by human intentions.
The proposed BSI system consisted of three components: (1) a single-channel flexible EEG sensor coupled with a self-developed online decoding module; (2) an electromyography (EMG) recording system; and (3) a spinal cord stimulation system (model CNS601B; Rishena Medical Co., Ltd., Changzhou, China) with a 16-channel electrode (model SCL305B-75; Rishena Medical Co., Ltd.).
A clinical team of neurosurgeons and veterinarians implanted the epidural spinal electrode at the T12–L2 level of a macaque (27 years old weighing 8.3 kg) and positioned the stimulator in the lateral dorsal region. The animal was in good health after 1 week of recovery. EES configurations were subsequently performed under anesthesia. EMG was used to identify and optimize the electrode configurations and parameter settings (amplitude, frequency, and pulse width) for single joint movements of the lower limbs. Wireless EMGs (Trigno; Delsys, Natick, MA, USA) were placed on the iliopsoas, quadriceps, tibialis anterior, and gastrocnemius of the lower limbs. After optimization, the parameters were set at 4.6 mA, 20 Hz, and 400 μs, respectively, enabling single joint movement of the hip and knee joints. Remarkably, the anesthetized macaque successfully exhibited bilateral stepping-like movements through spatiotemporally stimulation.
Electrooculography (EOG) signals were decoded to discriminate three control commands in the human subject experiment: left leg movement; right leg movement; idle and stop. This approach was chosen due to the higher signal-to-noise ratio and faster response time of EOG signals compared to motor imagery EEG. An application software was developed using PyQt5 with a modular architecture integrating data acquisition, preprocessing, feature extraction, prediction, transmission control protocol (TCP)-based wireless communication, and a graphical user interface (GUI), ensuring both efficient real-time processing and intuitive user interaction. The human subject was asked to look left, look right, and blink voluntarily. A 0.1–45 Hz band-pass filter was used in the signal preprocessing. The decoding algorithm was implemented using LightGBM with multimodal feature fusion. Each input sample consisted of the bandpass-filtered signals with 53 features derived from temporal (e.g., mean, variance, and Hjorth parameters), frequency (e.g., band power and spectral entropy), and non-linear domains (e.g., skewness, kurtosis, and waveform length). The LightGBM classifier had 31 leaves and a learning rate of 0.05, which was optimized for multiclass classification. The LightGBM classifier was trained with stratified a 10-fold cross-validation and achieved an average F1-score of 89.6% across 4 control commands.
Finally, in the online human–monkey BSI experiment, decoded outputs from the human subject were transmitted in real time to control the EES system implanted in the macaque, enabling spatiotemporally stimulation of lumbar spinal hotspots. The anesthetized macaque successfully exhibited left and right leg stepping-like movements controlled by human motion intentions (Supplementary Video 1). To the best of our knowledge, this is the first BSI proof-of-concept based on non-invasive BCI that controls EES with intentions to elicit locomotor activities. This approach avoids the surgical risks associated with invasive methods, while maintaining high decoding accuracy and real-time performance, thereby opening a new avenue for BSI technology. Future work will focus on further system optimization and the exploration of feasibility in clinical trials.
Several limitations of this study warrant consideration. First, experiments were performed in a controlled environment and the stability of decoding, long-term efficacy, and applicability to more complex daily motor tasks remain to be investigated. Second, the current amplitude of EES used to elicit the passive movements in this study was higher than the sensation threshold, which is the same as previously reported [2, 5]. Therefore, totally passive movement or passive walking by EES may not be feasible for SCI patients with persistent sensation. However, these patients can still benefit from long-term rehabilitation using EES. Third, due to high risks, this study is a proof of concept on an old macaque with weak muscle abilities and barely any locomotion. Stepping-like movements were successfully generated under anesthesia. Animals that are closer to the typical SCI age groups will be the subjects in a corollary study, then clinical trials will begin.
In conclusion, the current study demonstrated that a portable, non-invasive BCI-based online BSI system can successfully control a spinal cord stimulator to induce stepping-like movements in an anesthetized macaque with human motion intentions. These findings provide a novel technical pathway and translational potential for restoring locomotion in patients with motor paralysis.
Data availability statement
The data that support the findings of this study are available on reasonable request from the corresponding authors.
Ethics statement
All experimental procedures were approved by the Animal Welfare and Ethical Review Committee of Hainan University [Hainan, China] (Registration No.: HNUAUCC-202500449).
Author contributions
Conceptualization: L.M., L.C., and Y.L. Writing: L.M., and L.C. Statistical analysis: Y.R., B.L., Z.S., and W.X. Supervision: F.L., and Q.F. Review and editing: X.H., and M.Y.
Funding or acknowledgments
This research was supported by the Brain Science and Brain-like Intelligence Technology-National Science and Technology Major Projects (Grant Nos. 2025ZD0215202 and 2022ZD0208602), the National Natural Science Foundation of China (Nos. 32360196 and 32160204), the Key R&D Project of Hainan Province (Grant No. ZDYF2025SHFZ023), the Hainan Provincial Natural Science Foundation of China (No. 825QN566), the Hunan Provincial Natural Science Foundation of China (No. 2025JJ90272), the Joint Program on Health Science & Technology Innovation of Hainan Province (WSJK2025QN046), and the Hainan Province Clinical Medical Center (No. 0202067). Supported by the PhD Scientific Research anInnovation Foundation of The Education Department of Hainan ProvinceJoint Project of Sanya Yazhou Bay Science and Technology City (No. HSPHDSRF-2024-08-009).
Conflict of interest
The authors declare that there are no conflicts of interest.
Supplementary materials
Supplementary Video 1 available at https://bio-integration.org/wp-content/uploads/BSI-video.mp4.
References
- Wagner FB, Mignardot J-B, Le Goff-Mignardot CG, Demesmaeker R, Komi S, et al. Targeted neurotechnology restores walking in humans with spinal cord injury. Nature 2018;563(7729):65-71. [PMID: 30382197 DOI: 10.1038/s41586-018-0649-2]
- Rowald A, Komi S, Demesmaeker R, Baaklini E, Hernandez-Charpak SD, et al. Activity-dependent spinal cord neuromodulation rapidly restores trunk and leg motor functions after complete paralysis. Nat Med 2022;28(2):260-71. [PMID: 35132264 DOI: 10.1038/s41591-021-01663-5]
- Ren Y, Mo L, Lu J, Zhu P, Yin M, et al. Epidural electrical stimulation for functional recovery in incomplete spinal cord injury. Cyborg Bionic Syst 2025;6:0314. [PMID: 40697542 DOI: 10.34133/cbsystems.0314]
- Balaguer J-M, Prat-Ortega G, Ostrowski J, Borda L, Verma N, et al. Neural mechanisms underlying the recovery of voluntary control of motoneurons after paralysis with spinal cord stimulation. Neuron 2025;S0896-6273(25)00658-0. [PMID: 40975061 DOI: 10.1016/j.neuron.2025.08.023]
- Lorach H, Galvez A, Spagnolo V, Martel F, Karakas S, et al. Walking naturally after spinal cord injury using a brain-spine interface. Nature 2023;618(7963):126-33. [PMID: 37225984 DOI: 10.1038/s41586-023-06094-5]