Understanding Vascular Toxicity of Indigo Naturalis in Anti-Inflammatory Applications: Mendelian Randomization and Co-localization Study
1Institute of Chemical Biology, Shenzhen Bay Laboratory, Shenzhen 518132, China
2GoodMedX Tech Limited, Hong Kong SAR 999077, China
3College of Big Data and Internet, Shenzhen Technology University, Shenzhen 518118, China
4Medical School, Shenzhen University, Shenzhen 518060, China
*Correspondence to: Dr. Jie Liu, Institute of Chemical Biology, Shenzhen Bay Laboratory, Guangming District, Shenzhen 518132, China. E-mail: liujie@szbl.ac.cn
Received: August 28 2025; Revised: November 27 2025; Accepted: December 8 2025; Published Online: December 19 2025
Cite this paper:
Liu J, Liu C, Xiao H et al. Understanding Vascular Toxicity of Indigo Naturalis in Anti-Inflammatory Applications: Mendelian Randomization and Co-localization Study. BIO Integration 2025; 6: 1–11.
DOI: 10.15212/bioi-2025-0155. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Background: Indigo naturalis (IN), a traditional Chinese herbal medicine, shows promising potential for treating autoimmune inflammatory diseases. However, its clinical translation and application are being limited by concerns regarding vascular toxicity.
Methods: Mendelian randomization (MR) and colocalization analyses were conducted to systematically investigate the target-specific vascular risks associated with IN.
Results: Causal associations were identified between four targets of IN and multiple vascular disorders, including deep vein thrombosis (DVT) (OR = 1.001, 95% CI = 1.003-1.000, P = 0.008), among others. Notably, urate was identified as a mediator between IN and DVT, exhibiting a mild mediation effect (7.3%, P = 0.048).
Conclusion: For the first time, the causal effects of IN on vascular toxicity were demonstrated using MR. This study provides insights into the safety implications of IN for anti-inflammatory applications, which would facilitate evidence-based research on IN and its clinical translation.
Keywords
Indigo naturalis, Mendelian randomization, urate, vascular toxicity.
Introduction
Indigo naturalis (IN [Qing Dai in Chinese]) is a well-established anti-inflammatory herb used in traditional Chinese medicine (TCM) for the treatment of autoimmune inflammatory disorders, including psoriasis and ulcerative colitis (UC) [1]. As such, the clinical translation of IN has attracted considerable research efforts but is slowed by the poor understanding on IN-associated vascular toxicity [2–6]. The toxicity of IN is typically manifested in the form of portal vein thrombosis (PVT) [4] and pulmonary arterial hypertension (PAH) [7, 8] in multiple clinical trials and practices but the underlying mechanism has not been established. Therefore, a comprehensive understanding of the severe IN toxicity is warranted to facilitate a translation study involving IN.
Bis-indole alkaloid indigo (ING) and the ING isomer (indirubin [INB]) are the major organic components of IN (4% and 0.13%, respectively) and have been recognized as the active pharmaceutical ingredients (APIs) against psoriasis and UC by a TCM expert consensus [1]. ING and INB are potent agonists of the aryl hydrocarbon receptor (AhR) that participates in immunologic and metabolic regulation [1]. Moreover, INB is the antagonist for three types of kinases, i.e., fms-like tyrosine kinase 3 (FLT3) [9, 10], cyclin-dependent kinases (CDKs), and glycogen synthase kinase-3β (GSK-3β) [1, 11–13]. Therefore, we propose a hypothesis that IN-associated vascular toxicity is linked to the ING and INB targets, as reasoned below. Both alkaloids can elicit a pro-inflammatory effect by activating the AhR/CYP1A1 metabolic axis [8, 14] and subsequently promoting reactive oxygen species (ROS) production and NLRP3 activation [15, 16]. Notably, these adverse effects can be realized via the uptake of INB particles by macrophages within Peyer’s patches [15]. In addition, INB can cause inflammation due to potent cytotoxicity by inhibiting CDKs and GSK-3β [1, 11–13, 15, 17]. In fact, INB has been used in China clinically to kill leukemia cells since 2002 for the treatment of chronic myelocytic leukemia [18]. Vascular conditions are strongly associated with oxidative stress and inflammation [19, 20], which are linked to the biological effect of the three IN targets described above. Moreover, INB can disrupt some vascular repair functions by deactivating FLT3 because FLT3 mediates differentiation of hematopoietic progenitor cells [9] and development and maintenance of dendritic cells [10]. Taken together, these risk factors suggest that exposure to INB/ING would cause vascular toxicity by interacting with INB/ING targets, i.e., AhR, CDKs, GSK-3β, and FLT3. To date, the evidence supporting the hypothesis remains limited to one preclinical study involving the toxic effect of AhR activation by IN [8].
Mendelian randomization (MR) has emerged as a promising tool for drug safety prediction in pharmaceutical development as a prominent alternative to randomized controlled trials (RCTs) [21, 22]. The key advantages of MR include substantially reducing biases from reverse causation and residual confounding using genome-wide association study (GWAS) data [23, 24]. Briefly, MR enables a causal inference between drug exposure and toxicity risk by leveraging genetic variants associated with gene expression of drug targets (e.g., AhR, CDKs, GSK-3β, and FLT3) and phenotypic outcomes (e.g., vascular risks).
To the end, this work used MR to determine whether changes in gene expression of ING and INB targets (i.e., AhR, CDK1-20, GSK-3β, and FLT3) causally effect vascular risks. In this study it was hypothesized that there is a causal effect of AhR, CDK1-20, GSK-3β, and FLT3 expression on 30 vascular risks. AhR, FLT3, and CDK4/6/16 expression was shown to support a causal effect (P < 0.05) on multiple vascular risks using 2-sample MR and genetic co-localization analysis. Furthermore, MR indicated that urate mediates one vascular risk (i.e., deep venous thrombosis [DVT]) through AhR activation upon IN exposure. This novel work comprehensively provided MR evidence for causal effects of IN APIs (i.e., ING and INB) on vascular toxicity.
Methods
Study design and dataset description
As the primary analysis, MR was used to examine the following: the effects of ING/INB target gene expression on vascular risk. Upon identifying MR evidence of a causal effect, co-localization analysis was performed to confirm that exposure and outcomes are regulated by the same causal variants. The mediation effect was further investigated by screening metabolites, immune cells, and gut bacteria to identify potential biological mediators that explain the causal pathway between IN exposure and vascular outcomes. All GWAS summary statistics were derived from cohorts of European ancestry to minimize population stratification bias, as listed in Table S1, including 30 vascular conditions and 23 quantitative trait loci (eQTL) of gene expression (AhR, FLT3, GSK-3β, and CDK1-CDK20).
Selecting drug targets
Gene targets for ING and INB were collected from references [12, 13]. The eQTL datasets of the target genes (AhR, FLT3, GSK-3β, and CDK1-20) are listed in Table S1.
Two-sample MR and mediation MR analyses
The inverse variance weighted (IVW) MR method was selected as the main MR analysis method. Two-sample MR with an MR-based platform (P = 5 × 10−5) [25], where Mendelian Randomization Pleiotropy Residual Sum and Outlier (MR-PRESSO) was used. Multiple testing correction of the IVW results was carried out using the Benjamini-Hochberg method with a false discovery control level (α = 0.05) [26, 27]. The mediation effect was examined by screening metabolites, immune cells, and gut bacteria (P = 5 × 10−5) with GWAS summary datasets in the IEU GWAS database [28].
Sensitivity analysis
Several sensitivity analyses were performed to determine the presence of pleiotropy and heterogeneity. Pleiotropy analysis was mainly based on the MR-Egger intercept and heterogeneity tests using Cochran’s Q statistics. A significant MR-Egger intercept test (P < 0.05) was interpreted as evidence of directional pleiotropy.
Co-localization analysis
Co-localization analysis was performed using R package Coloc (version 3.2-1) [29]. Variants ± 100 kb of the reference variant were included. The 1000 Genomes v3 European ancestry dataset was used as the LD reference panel. Evidence for co-localization was defined as a posterior probability > 0.8.
Results
Effect of ING/INB targets on vascular risks
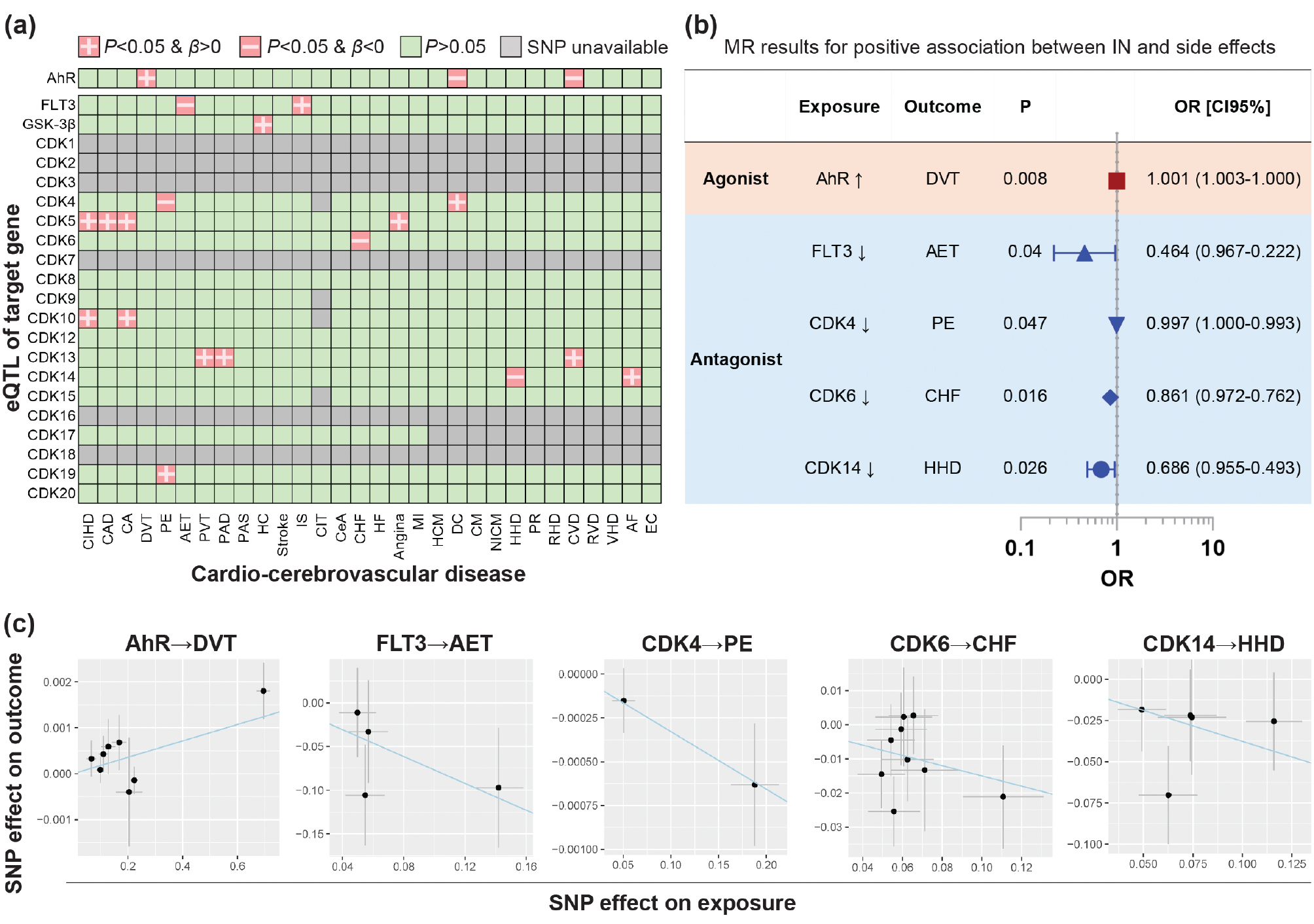
MR was performed with 30 vascular conditions as outcomes and 23 ING/INB targets (AhR, FLT3, GSK-3β, and CDK1-CDK20) as exposures at the P = 5 × 10−5 level (Figure 1A). This relatively lenient criterion (P = 5 × 10−5) was adopted for the purpose of increasing the number of available instrumental variables while ensuring validity, thereby enhancing the statistical power of the study. MR evidence of the causal effects of AhR, FLT3, and CDK4/6/14 expression on five vascular disorders was demonstrated after applying Benjamini-Hochberg correction (Tables 1 and S1–S6).
Figure 1 MR results at the P = 5 × 10−5 level. (A) Heatmap of MR results with eQTLs of ING/INB target genes (exposure) and 30 vascular risks (outcomes); (B) Summary of MR results indicative of a causal effect of ING/INB exposure on vascular toxicities; (C) Scatter plot of MR results. CIHD: chronic ischemic heart disease; CAD: coronary artery disease; CoA: coronary atherosclerosis; DVT: deep venous thrombosis; PE: pulmonary embolism; AET: arterial embolism and thrombosis; PVT: hemorrhoids and perianal venous thrombosis; PAD: peripheral artery disease; PAS: peripheral atherosclerosis; HC: high cholesterol; IS: ischemic stroke; CIT: carotid intima-media thickness; CeA: cerebral atherosclerosis; CHF: chronic heart failure; HF: heart failure; MI: myocardial infarction; HCM: hypertrophic cardiomyopathy; DC: dilated cardiomyopathy; CM: cardiomyopathy; NICM: non-ischemic cardiomyopathy; HHD: hypertensive heart disease; PR: polymyalgia rheumatic; RHD: rheumatic heart disease; CVD: cardiac valvular disease; RVD: rheumatic valve disease; VHD: valvular heart disease; AF: atrial fibrillation; EC: endocarditis.
Table 1 Results of MR Analyses with ING/INB as Exposures and Vascular Risks as Outcomes (P = 5 × 10−5).
| Exposure | Outcome | P Value Threshold | Number of SNPs | β | Causality Significance (P Value) |
|---|---|---|---|---|---|
| ING/INB → AhR | DVT | 5 × 10−5 | 8 | 0.0018 | 0.0075 |
| INB → FLT3 | AET | 5 × 10−5 | 4 | −0.7687 | 0.0405 |
| INB → CDK4 | PE | 5 × 10−5 | 2 | −0.0033 | 0.0472 |
| INB → CDK6 | CHF | 5 × 10−5 | 9 | −0.1500 | 0.0158 |
| INB → CDK14 | HHD | 5 × 10−5 | 5 | −0.3768 | 0.0255 |
DVT: deep venous thrombosis; AET: arterial embolism and thrombosis; PE: pulmonary embolism; CHF: chronic heart failure; HHD: hypertensive heart disease.
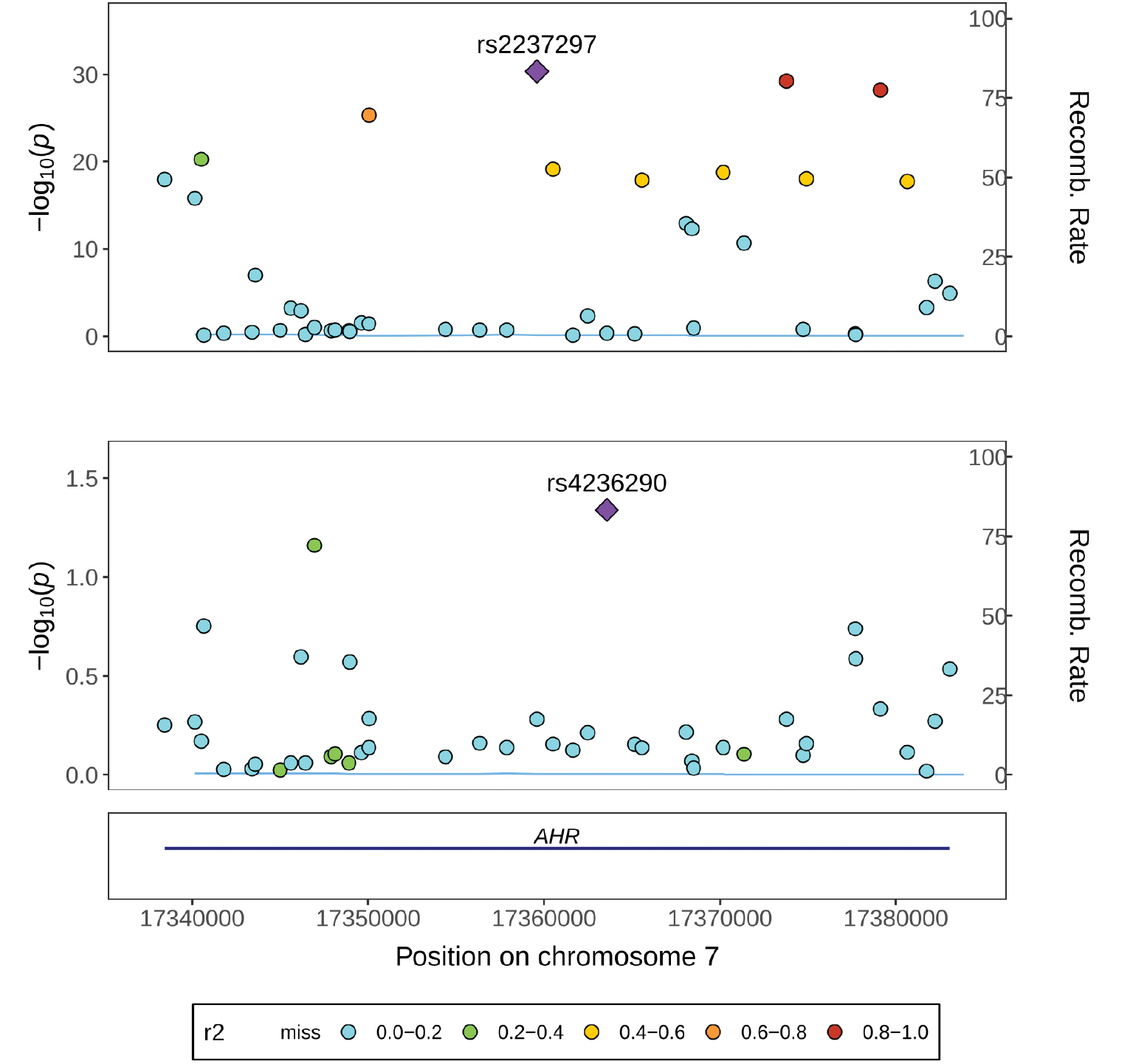
Specifically, MR demonstrated a positive effect of AhR expression on DVT (β = 0.001787) but a negative effect on dilated cardiomyopathy (DC) and cardiac valvular disease [CVD] (Figure 1A and Table 1). These data indicated that ING/INB potentially exert a pro-thrombotic effect as an AhR receptor agonist. Furthermore, co-localization analysis confirmed the AhR QTL and ING/INB exposure outcome (DVT) share the same causal variant (H4 = 0.91), as shown in Figure 2 and Table S7, which corroborates the putative causal effect of the exposure (ING/INB→AhR) on DVT.
Figure 2 Co-localization analysis. Regional plots for the AhR eQTL association with DVT within ± 100 kb of AhR (GRCh37/hg19 by Ensembl, Chr7: 17,338,246–17,385,776).
In addition, MR displayed a negative effect of FLT3 expression on arterial embolism and thrombosis [AET] (β = −0.7687) but a positive effect on ischemic stroke (IS). These data suggested that INB potentially leads to AET as an FLT3 receptor antagonist. Similarly, CDK4/6/14 expression was shown to be inversely associated with pulmonary embolism [PE] (β = −0.003284), congestive heart failure [CHF] (β = −0.15), and hypertensive heart disease [HHD] (β = −0.3768), respectively (Figure 1B and Table 1). These results indicated that ING/INB have the potential to cause PE, CHF, or HHD as a CDK receptor antagonist.
Sensitivity analysis was used to correct the issues of pleiotropy and heterogeneity. Cochran’s Q-test and funnel plot showed no evidence of heterogeneity and asymmetry (Table S8). The MR-Egger intercept showed weak evidence of pleiotropy at the directional level (Table S8). Potential horizontal pleiotropy was not detected using the MR-PRESSO global test (Table S8). In addition, the effect of each single nucleotide polymorphism (SNP) on the overall causal estimates was verified by leave-one-out analysis (Table S8), indicating that all SNPs were calculated to make the causal relationship significant.
Notably, no MR evidence demonstrated a causal effect of GSK-3β on 30 vascular disorders at a P = 5 × 10−5. Thus, GSK-3β was not included in the analysis.
Mediation effect
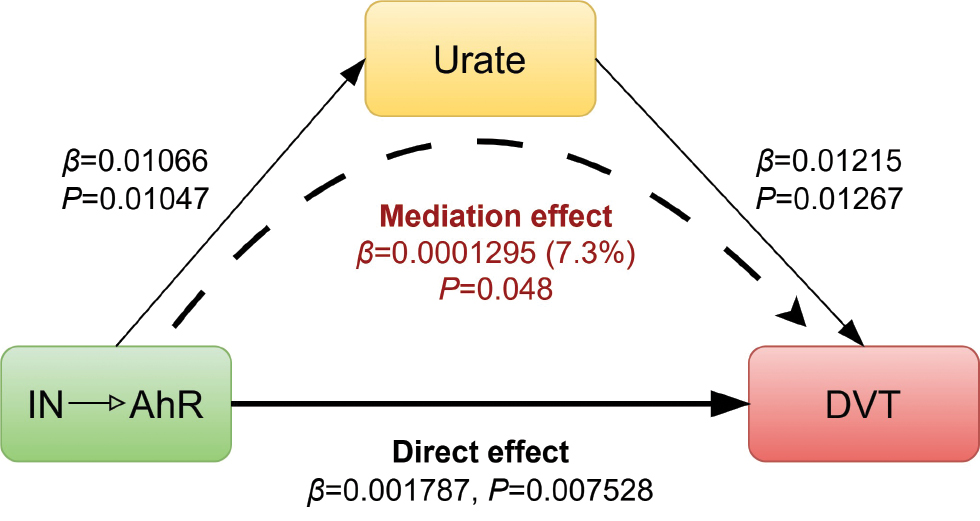
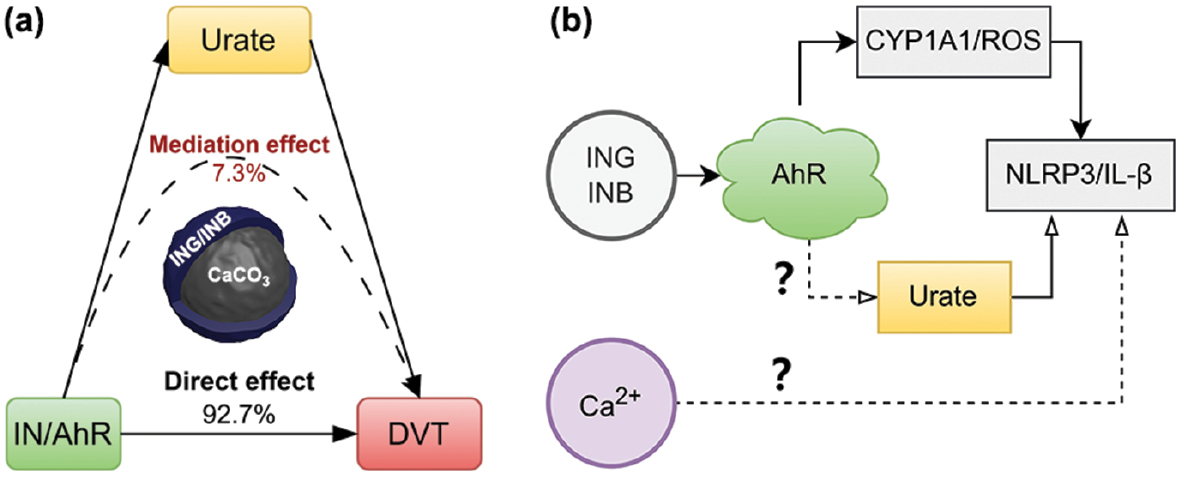
The mediation effect was studied by screening metabolites, immune cells, and gut bacteria using two-sample MR. Urate was identified as a potential mediator between AhR and DVT (Table S9). The indirect effect of urate was estimated to be 0.0001295 (95% CI = −0.0000008–0.0002598), accounting for 7.3% of the total effect (0.001787), as shown in Figure 3. In contrast, the direct effect (0.001657) comprises 92.7% of the total effect, suggesting that the influence of AhR activation via ING/INB (exposure) on DVT (outcome) is primarily driven by non-mediated pathways. This finding aligns with clinical studies. For example, elevated serum uric acid levels are associated with an increased risk of VTE recurrence [30]. Urate-lowering therapy mitigates the risks of hospitalized stroke and mortality in patients with gout [31]. No significant mediation effect was identified involving metabolites, immune cells, or gut bacteria in the current study.
Figure 3 Schematic diagram showing the mediation effect of urate (P = 5 × 10−5).
Discussion
This study determined the causal relationships between 2 IN APIs and 30 vascular conditions. The causal effects of IN exposure on DVT outcomes were identified and urate was shown to contribute to 7.3% of the total effect. In addition, IN exposure was casually associated with multiple vascular risks, i.e., embolism and thrombosis, including AET and PE, and heart diseases, including CHF and HHD, which are driven by the INB antagonistic effects on FLT3, CDK4, CDK6 and CDK14.
Notably, the IN toxicity has primarily been reported in Asian populations, while the datasets used in the current study were of European ancestry. Nevertheless, the findings of this MR study can be generalized to Asian and European populations, which can be supported by two perspectives. First, the four IN targets (AhR [32], FLT3 [9], GSK-3β [33], and CDKs [34]) are highly conserved across species and human populations. Second, multiple medications targeting these proteins are commonly used or currently undergoing clinical trials in Asian and European populations. For example, palbociclib, which targets CDKs, is a widely used anti-cancer drug across different ethnic groups. Tapinarof (targeting AhR) and midostaurin (targeting FLT3) are approved treatments for psoriasis and cancer in Chinese Taiwan, indicating that these targets are relevant across Asian and European populations. In addition, valproic acid (NCT01548066) has undergone clinical trial in South Korea, cilostazol (NCT06612593) is currently undergoing clinical trial, and tideglusib (NCT02858908) has undergone clinical trial in the United Kingdom. These trials indicated that GSK-3β is a common target for agents, including ING/INB, across diverse populations.
While IN has been shown through use in TCM to have an anti-inflammatory effect on autoimmune inflammatory disorders, the side effects on the vascular system have been typically observed in clinical trials and clinical practice [4, 7, 8]. This work has added MR evidence to these clinical observations. Accordingly, we present our interpretation of the MR evidence below with the latest advances in understanding the bioactivity, pharmacology, and toxicology of IN based on preclinical and clinical studies.
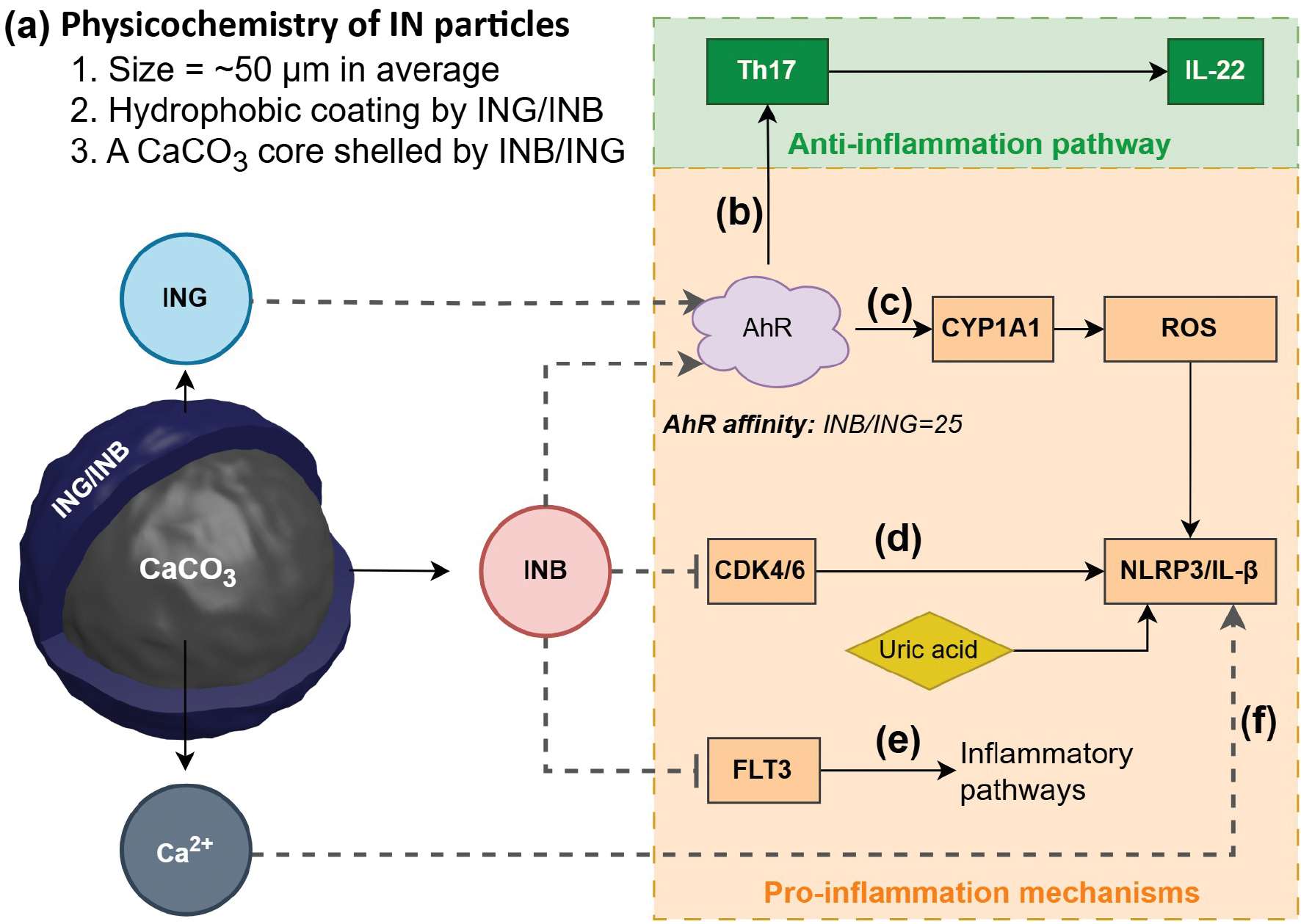
Pharmacologic duality of the AhR pathway
The IN vascular toxicity identified in this work can be explained based on the biphasic pharmacologic AhR properties. INB/ING can restore the Th17/Treg balance and consequently promote IL-22 secretion and intestinal mucosal repair with EC50 values of 0.2 and 5 nM, respectively [8, 12], which is the primary anti-inflammatory mechanism underlying IN [3, 35], as illustrated in Figure 4B. In contrast, this anti-inflammatory effect can shift to a pro-inflammatory response under specific pathologic conditions, and as a result vascular toxicities would be apparent. This transition involves the AhR/CYP1A1 metabolic axis [36], which can be overactivated by INB/ING with ROS overproduction, leading to the assembly of NLRP3 inflammasomes (Figure 4C). Moreover, NLRP3 inflammasomes can be promoted by INB/ING in other ways. For example, INB/ING can dampen the suppressive effect of AhR on NLRP3 inflammasome activity by depleting available AhR [37]. Similarly, excessive activation of AhR by INB/ING deplete endogenous AhR ligands via the AhR/CYP1A1 axis, which disrupts indole derivative homeostasis and consequently induces oxidative toxicity [38]. In fact, the presence of INB and ING in human urine at ~0.2 nM [12] underscores the significance of physiologic concentrations of INB and ING. In addition, urate was identified as a mediator between IN and DVT, exhibiting a mild mediation effect (7.3% of the total effect; P = 0.048). Indeed, elevated serum uric acid levels are associated with an increased risk of VTE recurrence [30]. Urate-lowering therapy has been shown to mitigate the risks of hospitalized patients with stroke and mortality in patients with gout [31]. This finding suggests the possible involvement of AhR in overexpression of xanthine oxidase and downregulation of uric acid transporters, which may contribute to the inflammatory processes underlying DVT. The finding implicates a novel proinflammatory mechanism underlying IN.
Figure 4 Physicochemistry and biology of IN. (A) Structural features, (B) anti-inflammatory, and (C-F) pro-inflammatory mechanisms underlying IN.
INB would contribute more significantly to IN toxicity than the INB isomer, ING. This is because the 25-fold greater AhR affinity of INB than ING amplifies pro-inflammatory signalling [8, 12], although the isomers share similar physicochemical properties (e.g., aqueous solubility = ~40 μg/mL; LogP = ~3.5) [39].
The target-specific vascular risks of IN should be analyzed within the context of the dose effect. Activation of AhR and subsequent induction of CYP1A1 by IN leads to the metabolic clearance of IN [40]. Therefore, with short-term IN dosing, the effects would be eliminated. However, with long-term IN treatment, sustained AhR activation and continuous induction of CYP1A1 occur, resulting in a prolonged accumulation of ROS. This persistent oxidative stress can contribute to various vascular disorders such as PAH [8]. Clinical observations suggest that this effect may need at least 6 months to manifest, as suggested by a reported case of PAH developing after IN treatment for 6 months [7].
Pro-inflammatory INB pleiotropy
INB is a potent inhibitor of FLT3 and CDKs [18]. INB causes inflammation through an inhibitory effect on FLT3 with an IC50 = 25 nM [13] and on CDKs with IC50 in the range of 50–100 nM [13] (Figure 4D, E). Indeed, FLT3 inhibition can trigger an immediate global change in gene expression, leading to significantly upregulated innate immune and inflammatory pathways [41–43]. Similarly, CDK4/6 inhibition can activate NLRP3 inflammasomes and consequently facilitate IL-1β secretion [44]. These reports indicated the pro-inflammatory INB pleiotropy.
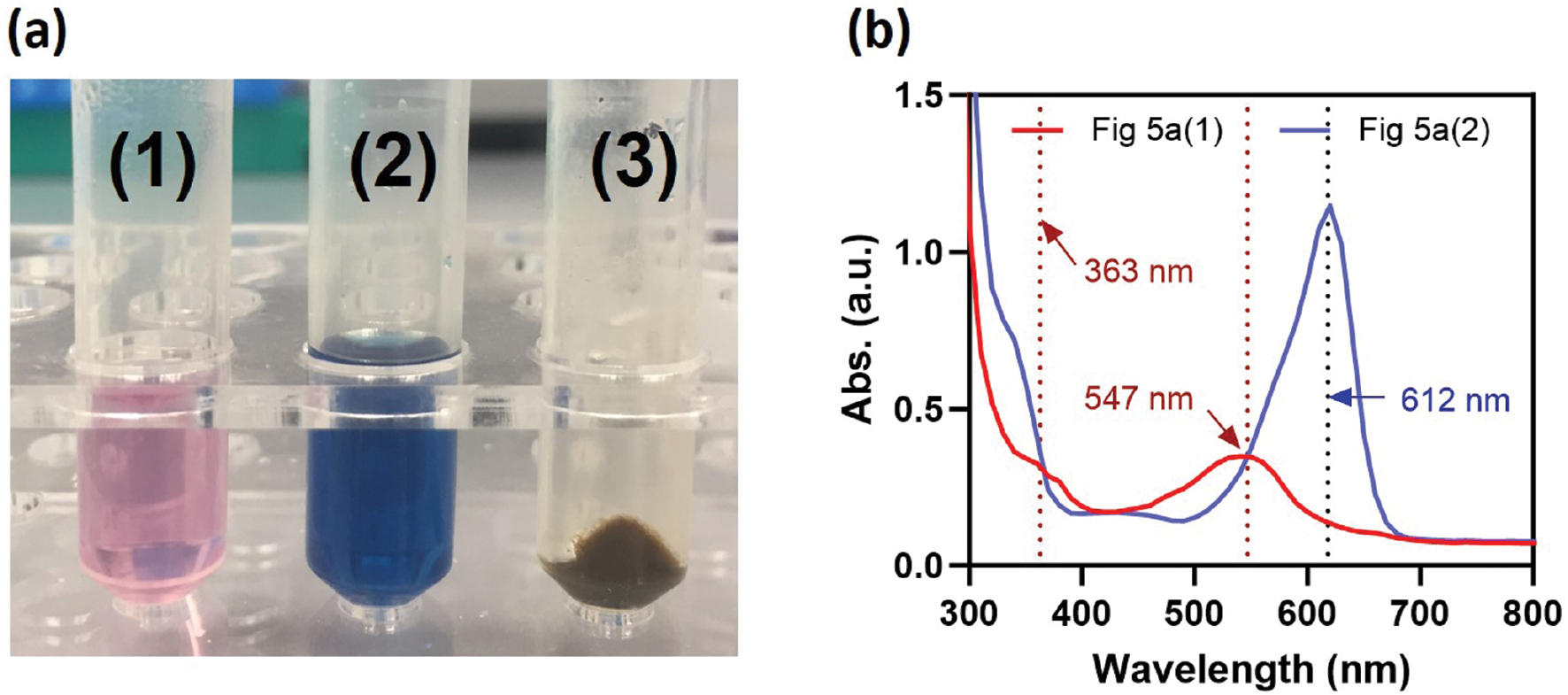
Interestingly, traditional folk practices of IN-based therapy involve washing IN with alcoholic drinks before oral administration to reduce the side effects in the digestive system. By analyzing IN extracts obtained through sequential washing with ethanol followed by dimethyl sulfoxide (DMSO), we found that INB was predominantly extracted by ethanol, while ING was mainly extracted by DMSO. The ethanol extract of IN appears pink with two characteristic absorption peaks at 363 and 547 nm (Figure 5), whereas the DMSO extract appears deep blue with a peak at 612 nm [45]. These data plausibly suggest that one toxic substance removed by alcoholic drinks would be INB. Both folk experience and laboratory analysis support the toxic potential of INB. Notably, the difference in solubility between INB and ING in ethanol suggests that INB and ING likely have distinct bioavailability profiles, which could influence toxicity, especially when factoring in food intake and the complex environment of the intestinal lumen, which warrants a detailed and thorough investigation.
Figure 5 Characterization of IN extracts. (A) Photograph of IN extracts obtained by (1) ethanol, followed by (2) DMSO, and (3) the remaining solid residue of IN after sequential washing with ethanol and DMSO; (B) UV-Vis spectra of the IN extracts shown in Figure 5A.
Pro-inflammatory particulate IN physicochemistry
IN is processed using CaO unlike common TCM herbs, then presented as microscale particles with an inorganic core of CaCO3 and a hydrophobic surface due to the coating of INB/ING [46], as illustrated in Figure 4A. This unique structure can potentiate INB/ING-mediated inflammation through the mechanisms postulated below. Released Ca2+ can activate NLRP3 via the calcium sensing receptor (CaSR) pathway [47]. Moreover, NLRP3 overload can lead to oxidative stress [48], which then collectively facilitates AhR/CYP1A1/ROS signaling. As a result, these events finally converge to hyperactivation of NLRP3 (Figure 4F).
In addition, the limited bioavailability of IN, which is caused by the hydrophobically coated surface from INB/ING, typically requires high doses of IN in clinical practice. This effect, in turn, increases the risk of off-target toxicity. In addition, the typical use of CaO in traditional herbal processing is of particular concern [49] and highlights the need to investigate toxic potentials of the interactions among Ca2+, herb chemicals, and biological systems.
Clinical implications for IN use
IN has been used by TCM for UC treatment [1, 35], while UC is associated with higher levels of cardiovascular risks [50, 51]. Clinicians should exercise caution because IN may worsen clinical outcomes by increasing cardiovascular risk when considering the vascular risks of IN use [52]. This effect is especially critical for corticosteroid-treated UC patients, who already face significantly higher risks of venous thromboembolism and ischemic heart disease [53]. Moreover, the chronic inflammatory nature of UC necessitates prolonged IN treatment, which raises the concern of cumulative ING/INB toxicity on the vascular system. Indeed, it has been reported that IN treatment may have an undesirable association with an increase in pulmonary arterial systolic pressure in UC patients [54]. In addition, IN is an anticancer agent used in TCM for the treatment of acute myeloid leukemia (AML) [18] but pro-inflammatory INB may potentially exacerbate AML because inflammatory signaling is typically involved in AML pathogenesis. In addition, the synergy of metabolites, such as uric acid in the IN pro-inflammatory effect, should be taken into consideration (Figure 4D). On the other hand, it is important to note that uric acid is known to have specific physiologic functions, including anti-inflammatory effects [55]. Given this complexity, the use of uric acid as a biomarker for a mediatory role in vascular risk warrants further study.
Genotyping of CYP1A1 genetic polymorphisms can aid in the personalized precision therapy of IN because the efficiency of ROS production via IN-induced AhR/CYP1A1 depends on CYP1A1 activity. In addition, IN treatment can be optimized using a metronomic therapy strategy, which involves administering low doses of IN on a continuous or frequent, regular schedule (such as daily or weekly) over an extended period since metronomic therapy is commonly used in cancer chemotherapy and is known to cause fewer and less severe side effects compared to standard chemotherapy.
Limitations of the study
This study had several limitations. First, a range of other active components of IN, such as alkaloids, nucleosides, amino acids, and inorganic elements [46], were not included, although ING and INB are the major active components of IN. For example, in addition to ING and INB, IN has many other indole alkaloids, such as isatin and tryptanthrine, which have been demonstrated to be anti-inflammatory [6]. Second, eQTLs may not capture post-transcriptional regulation and eQTL effects, which can vary with cell states/diseases. Integration of pQTLs (protein QTLs) and use of eQTLs from disease-relevant tissues would have improved the current study. In addition, MR findings suggest a potential causal relationship between genetic predisposition to an exposure and an outcome, yet do not demonstrate that therapeutic modifications of the exposure would significantly affect the outcome. Beyond, the current study was based on European ancestry, while IN is primarily used in Asian populations. This discrepancy may limit and compromise the understanding of IN toxicity as presented in the current study.
Conclusions
This study presents the first comprehensive investigation into the target-specific effect (AhR, CDK1-20, GSK-3β, and FLT3) of the herbal medicine, IN, on the vascular system. Utilizing MR analysis, significant causal associations of the IN exposure with multiple vascular conditions were identified, including DVT, PE, and AET, as well as related heart diseases, including CHF and HHD. Importantly, the findings herein identified urate as a key mediator in IN-induced DVT via AhR activation. These insights highlight critical safety considerations for developing IN-based anti-inflammatory therapeutics and guide biomarker monitoring in related clinical trials. Furthermore, this study proposed the potential role of pharmacologic duality in the AhR pathway, pleiotropy of INB, and physicochemistry of IN particles in the vascular toxicity of IN. Corollary studies will focus on mechanistic verification to advance the understanding of the vascular toxicity of IN.
In addition, the vascular risks of IN applications underscore a significant need to optimize traditional processing of IN to improve the safety profile, while maintaining therapeutic efficacy. To address the challenges, three key improvements on the traditional processing of IN are proposed as follows: (1) reducing the percentage of INB by chemical engineering; (2) removing Ca2+ through innovations in herb formulating technology; and (3) developing oral delivery systems of IN APIs to improve the therapeutic index.
Data availability statement
The data supporting this article are available in the manuscript or its ESI†.
Ethics statement
No direct interactions with human or animal subjects were involved. Therefore, ethical approval and informed consent were not required.
Author contributions
JL: Conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, resources, software, validation, visualization, writing – original draft, writing – review & editing. JCL: Methodology, software, writing – review & editing. HTX and ZPX: Writing – review & editing.
Acknowledgment
JL extends sincere gratitude to the Fenji (奋楫) Postdoctoral Program in Shenzhen Bay Lab of Guang Dong Province of China and the financial support from the IDEATION Programme of the Hong Kong Science & Technology Parks Corporation (Partner-23-159). The valuable suggestions of Dr. Ping Zheng on the methodology are sincerely appreciated.
Conflict of interest
The authors declare that there are no conflicts of interest.
Supplementary materials
Supplementary Material can be downloaded from https://bio-integration.org/wp-content/uploads/2025/12/bioi20250155_Supplemental.zip.
Graphical abstract
Highlights
- This study presented the first comprehensive Mendelian randomization evidence on the vascular toxicity of IN.
- Urate was identified as a mediator between IN (exposure) and DVT (outcome).
- It is proposed that the particulate characteristics of IN contributes to the toxicity and three key improvements in the traditional processing of IN are recommended.
In brief
For the first time, this study comprehensively presents Mendelian randomization evidence demonstrating the toxic effects of the active pharmaceutical ingredients in the herb indigo naturalis (IN). Specifically, it highlights a significant risk of deep vein thrombosis (DVT) mediated through the aryl hydrocarbon receptor (AhR). Moreover, urate was identified as a mediator between IN and DVT, exhibiting a mild mediation effect of 7.3%. These findings raise two important questions. First, does AhR upregulate xanthine oxidase or downregulate uric acid transporters, thereby contributing to the inflammatory processes underlying DVT? Second, does Ca2+ released from IN particles activate pro-inflammatory effects via calcium-sensing receptor?
References
- Xu Y, Lin C, Tan H, Bian Z. The double-edged sword effect of indigo naturalis. Food Chem Toxicol 2024;185:114476. [PMID: 38301993 DOI: 10.1016/j.fct.2024.114476]
- Sugimoto S, Naganuma M, Kiyohara H, Arai M, Ono K, et al. Clinical efficacy and safety of oral Qing-Dai in patients with ulcerative colitis: a single-center open-label prospective study. Digestion 2016;93(3):193-201. [PMID: 26959688 DOI: 10.1159/000444217]
- Naganuma M, Sugimoto S, Mitsuyama K, Kobayashi T, Yoshimura N, et al. Efficacy of indigo naturalis in a multicenter randomized controlled trial of patients with ulcerative colitis. Gastroenterology 2018;154(4):935-47. [PMID: 29174928 DOI: 10.1053/j.gastro.2017.11.024]
- Matsuno Y, Torisu T, Umeno J, Shibata H, Hirano A, et al. One-year clinical efficacy and safety of indigo naturalis for active ulcerative colitis: a real-world prospective study. Intest Res 2022;20(2):260-8. [PMID: 35508956 DOI: 10.5217/ir.2021.00124]
- Ben-Horin S, Kopylov U, Salomon N. Curcumin-QingDai combination as treatment for moderate-severe ulcerative colitis. Case Rep Gastroenterol 2022;16(3):563-8. [PMID: 36824698 DOI: 10.1159/000526646]
- Ozawa K, Mori D, Hatanaka A, Sawano T, Nakatani J, et al. Comparison of the anti-colitis activities of Qing Dai/Indigo Naturalis constituents in mice. J Pharmacol Sci 2020;142(4):148-56. [PMID: 32033881 DOI: 10.1016/j.jphs.2020.01.003]
- Naganuma M, Sugimoto S, Suzuki H, Matsuno Y, Araki T, et al. Adverse events in patients with ulcerative colitis treated with indigo naturalis: a Japanese nationwide survey. J Gastroenterol 2019;54(10):891-6. [PMID: 31102012 DOI: 10.1007/s00535-019-01591-9]
- Masaki T, Okazawa M, Asano R, Inagaki T, Ishibashi T, et al. Aryl hydrocarbon receptor is essential for the pathogenesis of pulmonary arterial hypertension. Proc Natl Acad Sci USA 2021;118(11):e2023899118. [PMID: 33836606 DOI: 10.1073/pnas.2023899118]
- Kazi JU, Rönnstrand L. FMS-like tyrosine kinase 3/FLT3: from basic science to clinical implications. Physiol Rev 2019;99(3):1433-66. [PMID: 31066629 DOI: 10.1152/physrev.00029.2018]
- Wilson KR, Villadangos JA, Mintern JD. Dendritic cell Flt3 – regulation, roles and repercussions for immunotherapy. Immunol Cell Biol 2021;99(9):962-71. [PMID: 34097779 DOI: 10.1111/imcb.12484]
- Hoessel R, Leclerc S, Endicott JA, Nobel MEM, Lawrie A, et al. Indirubin, the active constituent of a Chinese antileukaemia medicine, inhibits cyclin-dependent kinases. Nat Cell Biol 1999;1(1):60-7. [PMID: 10559866 DOI: 10.1038/9035]
- Adachi J, Mori Y, Matsui S, Takigami H, Fujino J, et al. Indirubin and indigo are potent aryl hydrocarbon receptor ligands present in human urine. J Biol Chemy 2001;276(34):31475-8. [PMID: 11425848 DOI: 10.1074/jbc.c100238200]
- Leclerc S, Garnier M, Hoessel R, Marko D, Bibb JA, et al. Indirubins inhibit glycogen synthase kinase-3 beta and CDK5/p25, two protein kinases involved in abnormal tau phosphorylation in Alzheimer’s disease. A property common to most cyclin-dependent kinase inhibitors? J Biol Chem 2001;276(1):251-60. [PMID: 11013232 DOI: 10.1074/jbc.M002466200]
- Sugihara K, Kitamura S, Yamada T, Okayama T, Ohta S, et al. Aryl hydrocarbon receptor-mediated induction of microsomal drug-metabolizing enzyme activity by indirubin and indigo. Biochem Biophys Res Commun 2004;318(2):571-8. [PMID: 15120638 DOI: 10.1016/j.bbrc.2004.04.066]
- Xu Y, Shi J, Mok HL, Lyu C, Chen J, et al. Macrophage-mediated transport of insoluble indirubin induces hepatic injury during intestinal inflammation. Adv Sci (Weinh) 2025;12(36):e02993. [PMID: 40546113 DOI: 10.1002/advs.202502993]
- Kwapiszewska G, Johansen AKZ, Gomez-Arroyo J, Voelkel NF. Role of the aryl hydrocarbon receptor/ARNT/cytochrome P450 system in pulmonary vascular diseases. Circ Res 2019;125(3):356-66. [PMID: 31242807 DOI: 10.1161/circresaha.119.315054]
- Wang H, Wang Z, Wei C, Wang J, Xu Y, et al. Anticancer potential of indirubins in medicinal chemistry: biological activity, structural modification, and structure-activity relationship. Eur J Med Chem 2021;223:113652. [PMID: 34161865 DOI: 10.1016/j.ejmech.2021.113652]
- Gaboriaud-Kolar N, Vougogiannopoulou K, Skaltsounis AL. Indirubin derivatives: a patent review (2010 – present). Expert Opin Ther Pat 2015;25(5):583-93. [PMID: 25887337 DOI: 10.1517/13543776.2015.1019865]
- Humphrey JD, Schwartz MA. Vascular mechanobiology: homeostasis, adaptation, and disease. Annu Rev Biomed Eng 2021;23(1):1-27. [PMID: 34255994 DOI: 10.1146/annurev-bioeng-092419-060810]
- Balta S. Endothelial dysfunction and inflammatory markers of vascular disease. Curr Vasc Pharmacol 2021;19(3):243-9. [PMID: 32316894 DOI: 10.2174/1570161118666200421142542]
- Bakker MK, van Straten T, Chong M, Paré G, Gill D, et al. Anti-epileptic drug target perturbation and intracranial aneurysm risk: mendelian randomization and colocalization study. Stroke 2023;54(1):208-16. [PMID: 36300369 DOI: 10.1161/strokeaha.122.040598]
- Yang Y, Chen B, Zheng C, Zeng H, Zhou J, et al. Association of glucose-lowering drug target and risk of gastrointestinal cancer: a mendelian randomization study. Cell Biosci 2024;14(1):36. [PMID: 38504335 DOI: 10.1186/s13578-024-01214-8]
- Iturrieta-Zuazo I, Walter S. Mendelian randomization: present and future of epidemiological studies in cardiology. Rev Esp Cardiol 2015;68(2):87-91. [PMID: 25449812 DOI: 10.1016/j.rec.2014.06.024]
- Deng Z, Long W, Duan H, Hui X, Tao T. Diabetes mellitus and pancreatic cancer: investigation of causal pathways through mendelian randomization analysis. BIOI 2023;4(4):160-69. [DOI: 10.15212/bioi-2023-0014]
- Hemani G, Zheng J, Elsworth B, Wade KH, Haberland V, et al. The MR-base platform supports systematic causal inference across the human phenome. ELife 2018;7:e34408. [PMID: 29846171 DOI: 10.7554/elife.34408]
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Stat Methodol 1995;57(1):289-300. [DOI: 10.1111/j.2517-6161.1995.tb02031.x]
- Hong S, Chen L, Zhou X, Huang Y, Tian Y, et al. Genetically predicted causal effects of gut microbiota on spinal pain: a two-sample Mendelian randomization analysis. Front Microbiol 2024;15:1357303. [PMID: 38591041 DOI: 10.3389/fmicb.2024.1357303]
- Kurilshikov A, Medina-Gomez C, Bacigalupe R, Radjabzadeh D, Wang J, et al. Large-scale association analyses identify host factors influencing human gut microbiome composition. Nat Genet 2021;53(2):156-65. [PMID: 33462485 DOI: 10.1038/s41588-020-00763-1]
- Giambartolomei C, Vukcevic D, Schadt EE, Franke L, Hingorani AD, et al. Bayesian test for colocalisation between pairs of genetic association studies using summary statistics. PLoS Genet 2014;10(5):e1004383. [PMID: 24830394 DOI: 10.1371/journal.pgen.1004383]
- Weng H, Li H, Zhang Z, Zhang Y, Xi L, et al. Association between uric acid and risk of venous thromboembolism in East Asian populations: a cohort and Mendelian randomization study. Lancet Reg Health West Pac 2023;39:100848. [PMID: 37565068 DOI: 10.1016/j.lanwpc.2023.100848]
- Yen F, Hsu C, Li H, Wei JC, Hwu C. Urate-lowering therapy may mitigate the risks of hospitalized stroke and mortality in patients with gout. PLoS One 2020;15(6):e0234909. [PMID: 32574194 DOI: 10.1371/journal.pone.0234909]
- Gasiewicz TA, Singh KP, Casado FL. Aryl hydrocarbon receptor and stem cells. Stem Cells Int 2017;2017:4602854. [PMID: 28163722 DOI: 10.1155/2017/4602854]
- del Ser T, Steinwachs KC, Gertz HJ, Andrés MV, Gómez-Carrillo B, et al. Treatment of Alzheimer’s disease with the GSK-3 inhibitor tideglusib: a pilot study. J Alzheimers Dis 2012;33(1):205-15. [PMID: 22936007 DOI: 10.3233/jad-2012-120805]
- Pluta AJ, Studniarek C, Murphy S, Norbury CJ. Cyclin-dependent kinases: masters of the eukaryotic universe. Wiley Interdiscip Rev RNA 2023;15(1):e1816. [PMID: 37718413 DOI: 10.1002/wrna.1816]
- Gu S, Xue Y, Liu X, Tang Y, Wang D, et al. Clinical, pharmacology and in vivo studies of QingDai (indigo naturalis) promotes mucosal healing and symptom improvement in ulcerative colitis by regulating the AHR-Th17/Treg pathway. J Inflamm 2024;21(1):44. [PMID: 39501249 DOI: 10.1186/s12950-024-00413-x]
- Yuan J, Sun X, Che S, Zhang L, Ruan Z, et al. AhR-mediated CYP1A1 and ROS overexpression are involved in hepatotoxicity of decabromodiphenyl ether (BDE-209). Toxicol Lett 2021;352:26-33. [PMID: 34571075 DOI: 10.1016/j.toxlet.2021.09.008]
- Huai W, Zhao R, Song H, Zhao J, Zhang L, et al. Aryl hydrocarbon receptor negatively regulates NLRP3 inflammasome activity by inhibiting NLRP3 transcription. Nat Commun 2014;5(1):4738. [PMID: 25141024 DOI: 10.1038/ncomms5738]
- Ye X, Li H, Anjum K, Zhong X, Miao S, et al. Dual role of indoles derived from intestinal microbiota on human health. Front Immunol 2022;13:903526. [PMID: 35784338 DOI: 10.3389/fimmu.2022.903526]
- Paul R, Blackburn RS, Bechtold T. Indigo and indigo colorants. In: Ullmann’s encyclopedia of industrial chemistry. Wiley-VCH Verlag GmbH & Co., KGaA; 2021. [DOI: 10.1002/14356007.a14_149.pub3]
- Schiering C, Wincent E, Metidji A, Iseppon A, Li Y, et al. Feedback control of AHR signalling regulates intestinal immunity. Nature 2017;542(7640):242-5. [DOI: 10.1038/nature21080]
- Melgar K, Walker MM, Jones LM, Bolanos LC, Hueneman K, et al. Overcoming adaptive therapy resistance in AML by targeting immune response pathways. Sci Transl Med 2019;11(508):eaaw8828. [PMID: 31484791 DOI: 10.1126/scitranslmed.aaw8828]
- Gebru MT, Atkinson JM, Young MM, Zhang L, Tang Z, et al. Glucocorticoids enhance the antileukemic activity of FLT3 inhibitors in FLT3-mutant acute myeloid leukemia. Blood 2020;136(9):1067-79. [PMID: 32396937 DOI: 10.1182/blood.2019003124]
- Gebru MT, Wang HG. Therapeutic targeting of FLT3 and associated drug resistance in acute myeloid leukemia. J Hematol Oncol 2020;13(1):155. [PMID: 33213500 DOI: 10.1186/s13045-020-00992-1]
- Weiss AM, Lopez MA 2nd, Rosenberger MG, Kim JY, Shen J, et al. Identification of CDK4/6 inhibitors as small molecule NLRP3 inflammasome activators that facilitate IL-1β secretion and T cell adjuvanticity. J Med Chem 2024;67(17):14974-85. [PMID: 39162654 DOI: 10.1021/acs.jmedchem.4c00516]
- Ju Z, Sun J, Liu Y. Molecular structures and spectral properties of natural indigo and indirubin: experimental and DFT studies. Molecules 2019;24(21):3831. [DOI: 10.3390/molecules24213831]
- Qi-Yue Y, Ting Z, Ya-Nan H, Sheng-Jie H, Xuan D, et al. From natural dye to herbal medicine: a systematic review of chemical constituents, pharmacological effects and clinical applications of indigo naturalis. Chin Med 2020;15(1):127. [PMID: 33317592 DOI: 10.1186/s13020-020-00406-x]
- Jäger E, Murthy S, Schmidt C, Hahn M, Strobel S, et al. Calcium-sensing receptor-mediated NLRP3 inflammasome response to calciprotein particles drives inflammation in rheumatoid arthritis. Nat Commun 2020;11(1):4243. [PMID: 32843625 DOI: 10.1038/s41467-020-17749-6]
- Peng T, Jou M. Oxidative stress caused by mitochondrial calcium overload. Ann N Y Acad Sci 2010;1201(1):183-8. [PMID: 20649555 DOI: 10.1111/j.1749-6632.2010.05634.x]
- Wu X, Wang S, Lu J, Jing Y, Li M, et al. Seeing the unseen of Chinese herbal medicine processing (Paozhi): advances in new perspectives. Chin Med 2018;13(1):4. [PMID: 29375653 DOI: 10.1186/s13020-018-0163-3]
- Wu H, Hu T, Hao H, Hill MA, Xu C, et al. Inflammatory bowel disease and cardiovascular diseases: a concise review. Eur Heart J Open 2021;2(1):oeab029. [PMID: 35919661 DOI: 10.1093/ehjopen/oeab029]
- Chen B, Collen LV, Mowat C, Isaacs KL, Singh S, et al. Inflammatory bowel disease and cardiovascular diseases. Am J Med 2022;135(12):1453-60. [PMID: 36058305 DOI: 10.1016/j.amjmed.2022.08.012]
- Singh S, Kullo IJ, Pardi DS, Loftus EV. Epidemiology, risk factors and management of cardiovascular diseases in IBD. Nat Rev Gastroenterol Hepatol 2014;12(1):26-35. [PMID: 25446727 DOI: 10.1038/nrgastro.2014.202]
- Sharma N, Tewatia P, Harvey PR, Kumar A. Controversies in venous thromboembolism risk assessment in inflammatory bowel disease: a narrative review. Diagnostics 2024;14(19):2112. [PMID: 39410515 DOI: 10.3390/diagnostics14192112]
- Orihara Y, Asakura M, Hida N, Kawai M, Sato T, et al. Effect of oral Qing-Dai medication on pulmonary arterial pressure levels in patients with ulcerative colitis. Circ J 2020;84(8):1339-45. [PMID: 32595175 DOI: 10.1253/circj.cj-19-1112]
- Wen S, Arakawa H, Tamai I. Uric acid in health and disease: from physiological functions to pathogenic mechanisms. Pharmacol Thers 2024;256:108615. [PMID: 38382882 DOI: 10.1016/j.pharmthera.2024.108615]