Phytochemicals as a Promising Approach to Rheumatoid Arthritis: Current Perspectives
1School of Pharmaceutical Sciences, Faculty of Pharmacy, IFTM University, Moradabad, Uttar Pradesh 244102, India
*Correspondence to: Vidhan Chand Bala, School of Pharmaceutical Sciences, Faculty of Pharmacy, IFTM University, Moradabad, Uttar Pradesh 244102, India, Mobile No.: +917500773087. E-mail: vidhanchandbala07@gmail.com
Received: July 29 2025; Revised: November 9 2025; Accepted: December 14 2025; Published Online: February 13 2026
Cite this paper:
Bala VC, Gupta AK. Phytochemicals as a Promising Approach to Rheumatoid Arthritis: Current Perspectives. BIO Integration 2026; 7: 1–26.
DOI: 10.15212/bioi-2025-0142. Available at: https://bio-integration.org/
Download citation
© 2026 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
In recent years, phytochemicals and medicinal plants have increasingly been used to treat autoimmune diseases, such as rheumatoid arthritis (RA). RA, a systemic inflammatory disease, is a chronic condition that affects primarily the joints, which are lined by synovial membranes, and leads to pain, diminished mobility, and joint deterioration. Oxidative stress, synovial hyperplasia, immune cell infiltration, and the production of pro-inflammatory cytokines, such as TNF-α and IL-6, are key factors in RA development. Herbal medicine is an effective alternative to conventional treatments, such as biologics, nonsteroidal anti-inflammatory drugs, and disease-modifying antirheumatic drugs, which are commonly used but can lose effectiveness or cause adverse effects. Phytotherapy therefore provides a promising complementary approach. This review provides an in-depth analysis of the pathophysiology of RA, therapeutic targets, drug resistance, and current therapeutic boundaries, with a focus on the roles of phytochemicals such as lignans, flavonoids, alkaloids, terpenoids, and phenolic compounds. By targeting key pro-inflammatory cytokines such as TNF-α and IL-6, and reformed molecular pathways such as those involving NF-κB, MAPK, and Nrf2/HO-1, these phytochemicals have potent anti-inflammatory and anti-rheumatoid arthritis properties.
Keywords
Anti-inflammatory; autoimmune disease; medicinal plants; phytochemicals; rheumatoid arthritis.
Introduction
Rheumatoid arthritis (RA) is a condition that causes joint inflammation, and leads to bone and cartilage damage. Approximately 0.5–1% of people worldwide are affected, particularly women between 40 and 60 years of age [1, 2]. Reactive oxygen species (ROS), matrix metalloproteinases (MMPs), pro-inflammatory cytokines (including tumor necrosis factor (TNF)-α, interleukin [IL]-1β, IL-6, and IL-17), and other mediators are overproduced as a result of immune dysregulation, which prolongs joint injury [3, 4]. Current treatments, including disease-modifying antirheumatic drugs (DMARDs) such as methotrexate, biologics, corticosteroids, and nonsteroidal anti-inflammatory drugs (NSAIDs), can halt disease progression but frequently have substantial adverse effects (hepatotoxicity and infection risk) and poor long-term efficacy [5, 6]. Many patients turn to alternative therapies because traditional medications cannot completely regulate cytokine cascades or oxidative stress [7, 8].
Natural chemicals originating from plants have garnered increasing attention as potential new treatments for RA. Alkaloids, flavonoids, terpenoids, phenolic compounds, saponins, tannins, lignans, and other chemical groups are among the many bioactive phytochemicals found in medicinal plants, which have long been used to treat inflammatory diseases [9–12]. In preclinical settings, many of these substances show strong antioxidant and anti-inflammatory properties. For example, triterpenoid saponins inhibit the nuclear factor-kappa B (NF-κB) pathway and consequently decrease inflammatory responses, whereas flavonoids target IL-6 and TNF-α, key cytokines involved in RA pathogenesis. Numerous exemplary phytochemicals have been examined in autoimmune disease studies [13]. Interestingly, phytochemicals frequently work by controlling molecular pathways that jointly decrease pro-inflammatory mediators and increase anti-inflammatory cytokines [14].
Cytokine production, NF-κB/mitogen-activated protein kinase (MAPK) signaling, oxidative stress, and inflammatory enzymes are generally targets of RA management [15]. Whereas phytochemicals may exert their effects on overlapping pathways with possibly fewer negative effects, conventional DMARDs target immunosuppression. For example, many phytochemicals suppress downstream cytokine release through the obstructive NLRP3 inflammasome pathway, cyclooxygenase-2 (COX-2) expression, or NF-κB activation [16, 17]. Additionally, they support antioxidant defenses by upregulating enzymes including superoxide dismutase (SOD) in the response to ROS by inflammatory synovium [18–20].
Preclinical research evaluating phytochemicals in RA models has markedly increased in the past decade. This research has involved both in vivo animal models (such as collagen-induced arthritis [CIA] and adjuvant-induced arthritis [AIA]) and in vitro experiments (such as lipopolysaccharide [LPS]-stimulated synoviocytes). To highlight mechanistic understanding, this review focuses exclusively on preclinical evidence. We explore the sources and effects of phytochemicals by categorizing them into classes, such as flavonoids, alkaloids, terpenoids, phenolic compounds, saponins, tannins, and lignans. Molecular markers such as NF-κB, MAPK, COX-2, cytokines, and oxidative stress pathways are highlighted. The figures depict key signaling pathways influenced by phytochemicals, whereas the tables summarize plant sources and experimental methods. In the Methods, we define the literature search approach. The potential of phytochemicals as therapeutic mediators for inflammatory illnesses is discussed in the Conclusion, including suggestions for future lines of investigation. This systematic review explains the routes through which phytochemicals decrease inflammation, according to preclinical research. In summary, the study illuminates the ability of phytochemicals to address inflammation and oxidative stress in the framework of RA. Future research should focus on well-designed clinical trials to further explore their therapeutic potential in managing inflammatory diseases.
Literature search
This narrative review was based on a comprehensive literature search conducted in the PubMed, Scopus, and Google Scholar databases, to identify preclinical studies exploring the anti-rheumatoid arthritis potential of phytochemicals and plant-derived compounds. Search terms included rheumatoid arthritis; phytochemicals; plant extract; anti-inflammatory; in vitro; in vivo; and specific phytochemical classes, such as flavonoids, alkaloids, terpenoids, phenolic compounds, saponins, tannins, and lignans. Both in vitro models (e.g., fibroblast-like synoviocytes [FLSs] and RAW 264.7 macrophages) and in vivo models (e.g., CIA and AIA) were included when they provided meaningful mechanistic or pharmacological evidence. Clinical and computational studies were excluded except when they contributed helpful perspectives. The collected data included phytochemical identity, plant source, experimental model, molecular targets, and biological effects, such as cytokine modulation, oxidative stress, and histopathological improvements. Overall, this narrative review provides an overview of preclinical evidence highlighting mechanistic patterns, without indicating systemic evaluation or quantifiable comparison.
Pathophysiology of RA, molecular targets for RA treatment, and the most common pathway
Pathophysiology of RA
RA progression can generally be divided into three interconnected stages: (1) the initiation or preclinical stage, characterized by the initiation of autoreactive T and B cells, and the generation of autoantibodies such as rheumatoid factor (RF) and anti-citrullinated protein antibodies (ACPAs); (2) the acute or non-specific inflammatory stage, involving synovial inflammation, immune cell infiltration, and cytokine overproduction (e.g., TNF-α, IL-1β, and IL-6); and (3) the chronic destructive or inflammatory stage, including persistent synovitis indications such as pannus formation, cartilage degradation, and bone erosion [21, 22]. These successive overlapping phases characterize the transition from autoimmune initiation to irreversible joint damage [23].
Initiation or preclinical stage
Autoantibody production is associated with serious symptoms, such as joint damage and elevated risk of death [24], probably because of the production of anti-citrullinated protein antibodies (ACPAs), which form immune complexes with citrullinated antigens. After these complexes bind RFs, complement is activated [25–28]. The ability to identify autoimmune reactions to citrullinated self-proteins has been a major development in recent years. The extent and strength of the association between shared epitopes and ACPA-positive and ACPA-negative epitopes varies among patients with RA. Because several non-HLA genes associated with RA susceptibility show substantial similarity across different genomes, researchers have speculated that ACPA-positive and ACPA-negative RA might be two genetically different forms of RA [29, 30]. Arginine has been shown to be converted into citrulline by peptidylarginine deiminases (PADs) when specific environmental factors change. Citrullinated proteins, after being presented to T cells by antigen-presenting cells via specific major histocompatibility complexes (MHCs), also produce ACPAs and trigger autoimmune reactions to citrullinated self-antigens in patients with RA [32]. Another non-MHC genetic risk factor for RA is peptidyl arginine deiminase type 4 (PADI4). However, in Asian patients with RA, bone injury has been associated with the PADI4 risk allele regardless of ACPA positivity [33]. Activated immune responses in tissues are unknown; however, ACPAs bind citrullinated residues on several proteins, such as histones, vimentin, fibronectin, fibrinogen, type II collagen, and alphaenolase [34]. Pre-RA, or circulating ACPAs, have been identified as many as 10 years before diagnosis [35]. Serum cytokine concentrations rise in tandem with the diversity and concentration of ACPA epitopes over time. ACPA and RF concentrations fall with good treatment, although patients scarcely ever become ACPA negative. In contrast, patients may seroconvert to RF negativity as a result of more frequent and dramatic RF decreases [35]. Autoantibodies directed against other post-translational protein modifications may also form, but anti-carbamylated protein and acetylated protein autoantibodies have also been found in patients with RA [36].
Acute or non-specific inflammatory stage
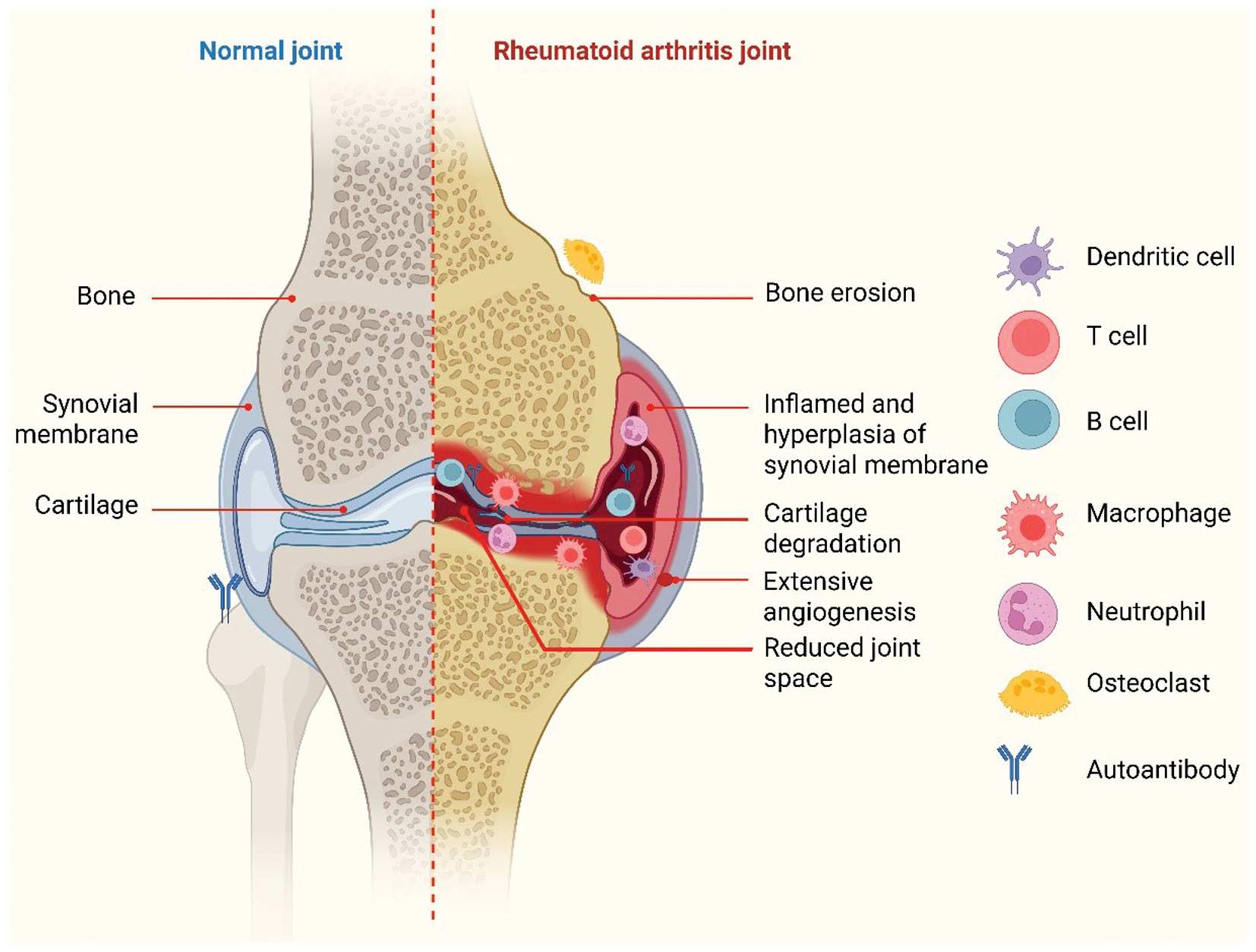
In RA, synovial inflammation elicited by immunological activation results in joint swelling. Leukocytes entering the synovial compartment are a characteristic of the observed edema [29]. The cellular makeup of RA synovitis is characterized by buildup of innate immune cells (such as dendritic cells, monocytes, mast cells, and innate lymphoid cells) and adaptive immune cells (such as T-helper (Th)1 and Th17 cells, B cells, plasmablasts, and plasma cells). Dendritic cells are stimulated by specific environmental or genetic variables and subsequently stimulate innate immunity [37]. After their recruitment and activation, dendritic cells cause T cells to release proinflammatory and bone-destroying cytokines, such as IL-1β, IL-6, and TNF-α, as well as MMPs, and to activate B cells, macrophages, synoviocytes, chondrocytes, and osteoclasts [38]. Therefore, the combination of innate and adaptive immune pathways facilitates tissue damage and remodeling in the nearby bone marrow and synovium [39]. Angiogenesis, which is necessary to provide nutrition and oxygen to enlarged joints, can lead to ongoing inflammation in RA and attract white blood cells to inflamed joints. Angiogenesis is triggered by proangiogenic factors [40]. In the synovium intima, FLSs develop a distinct invasive phenotype that encourages extracellular matrix invasion and aggravates joint damage [41] (Figure 1).
Figure 1 Normal joints and joints with rheumatoid arthritis. In RA, joint swelling reflects immune-stimulated synovial inflammation. T cells, dendritic cells, B cells, macrophages, and osteoclasts are examples of the innate and adaptive immune cells that accumulate in RA sinusitis. Angiogenesis, muscle atrophy, and synovial thickening are caused by immune response components that are pro-inflammatory and bone-destructive, thus resulting in loss of bone or cartilage (figure created with BioRender.com).
Increasing evidence highlights the critical role of the gut-joint axis in rheumatoid arthritis pathogenesis. Dysbiosis, characterized by diminished microbial diversity and enrichment in pro-inflammatory taxa such as Prevotella copri, has been associated with autoimmune activation and loss of immune tolerance. The gut microbiota influences systemic inflammation through modulation of intestinal permeability, LPS translocation, and cytokine production [42]. Many bioactive phytochemicals (such as polyphenols, flavonoids, and alkaloids) exert a portion of their anti-inflammatory effects via the gut microbiota, by enhancing beneficial genera (e.g., Lactobacillus and Bifidobacterium) and increasing the production of short-chain fatty acids, such as butyrate and propionate. These microbial metabolites regulate regulatory T (Treg) cell differentiation and inhibit Th17-driven cytokine cascades (IL-6, IL-17, and TNF-α), thereby restoring immune homeostasis. This gut microbial metabolism of phytochemicals provides a crucial mechanistic link between dietary or plant-derived compounds and their systemic anti-arthritic effects, and emphasizes the therapeutic relevance of targeting gut dysbiosis alongside joint inflammation in RA [43].
Chronic destructive or inflammatory stage
The nature of immune cells in the development and progression of RA has been examined in recent research. Local interstitial cells and their role in RA pathophysiology have also received increasing attention in recent years. The structural foundation of organs and tissues is made up of stromal cells [44]. These cells are believed to identify infections, initiate immunological responses, and perform immune tasks. Typical stromal cells include fibroblasts found in the gut, skin, gums, and synovium. Innate immune receptors, particularly Toll-like receptors (TLRs), are expressed by these cells [45, 46]. These stromal cells additionally release cytokines and chemokines, and use MHC II receptors to present and express antigens. Consequently, the innate immune system includes these stromal cells [34].
FLSs’ typical physiological activity in non-pathological synovial tissue includes forming the synovium’s lining layer, secreting synovial fluid, lubricating joint proteins, and supplying plasma protein for the surrounding cartilage and joint cavity [47]. Additionally, by generating matrix components such as collagen, FLSs contribute to the ongoing remodeling of the synovium and preserve synovium homeostasis. FLSs markedly change from benign mesenchymal cells to aggressive and destructive tumor-like cells when RA inflammation is present [48]. Decreased susceptibility to apoptosis; overexpression of adhesion molecules; and aberrant production of cytokines, chemokines, and MMPs are among the unique characteristics of these altered RA FLSs, which are important in RA development and progression [48].
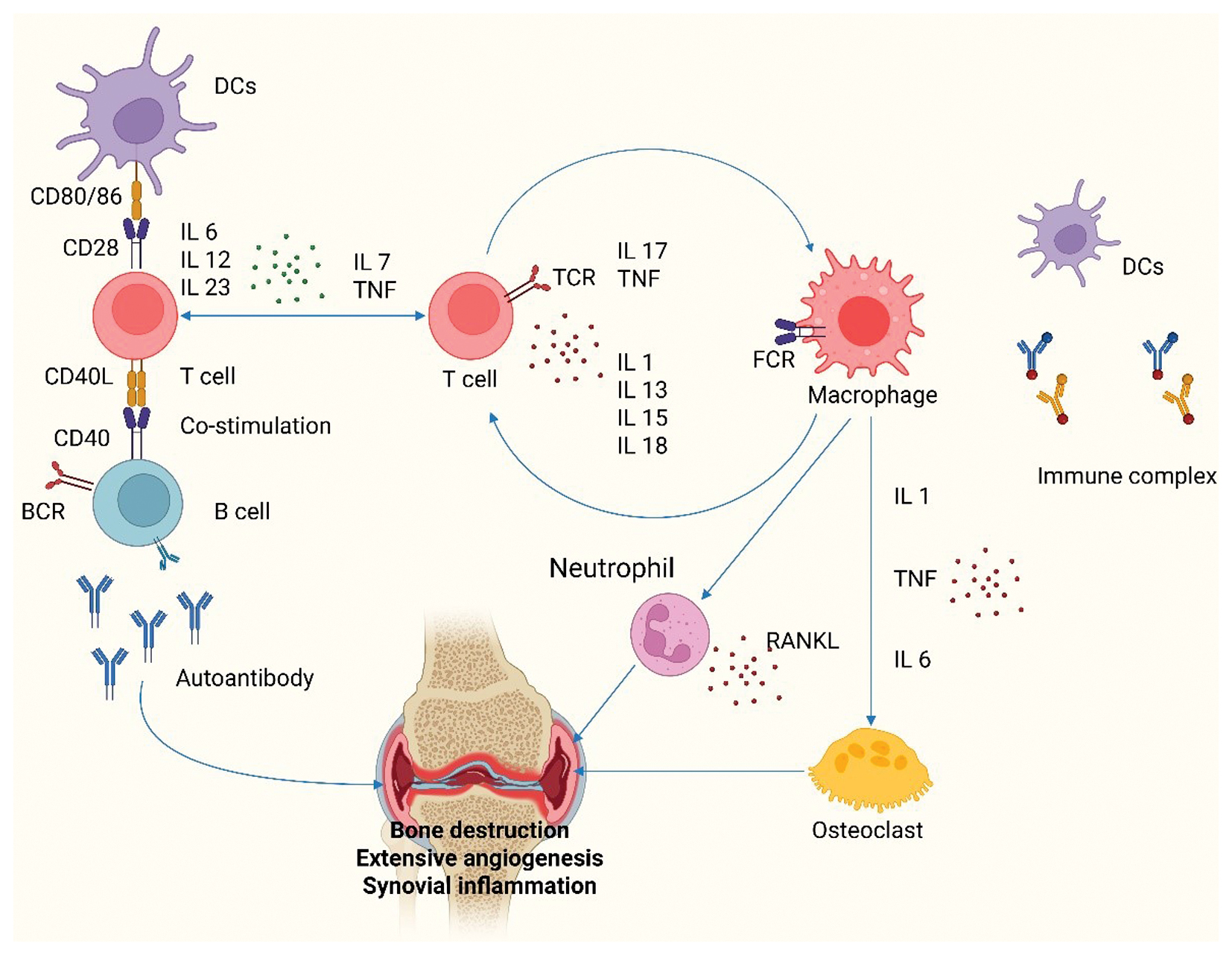
The inflammatory environment of the synovial compartment is controlled by a complex network of cytokines and chemokines; several clinical interventions have indicated that granulocyte-monocyte colony-stimulating factor, IL-6, and TNF-α are crucial to the process shown in Figure 2 [49]. By stimulating endothelial cells, drawing immune cells to the synovial compartment, stimulating fibroblasts, and accumulation of activated T and B cells, cytokines and chemokines increase inflammation. Th cells differentiate into Th1, Th2, Th17, and Treg cells, according to the cytokine microenvironment, whereas activated B cells, with the aid of antigen-presenting cells and Th cells, differentiate into plasma cells, which produce and secrete various immunoglobulins. Th1 cytokine secretion increases Th17 infiltration and IL-17 synthesis in synovial tissues during RA [50]. A subpopulation of CD4+ T cells called follicular helper T (Tfh) cells facilitate germinal center responses by supplying the signals required for the development of long-lived antigen-secreting plasma cells and high-affinity antibody synthesis [51]. Uncontrolled proliferation of Tfh cells has been noted in several systemic autoimmune diseases; moreover, in patients with RA, the frequency of circulating Tfh-like cells, their subtypes, and synovially infiltrating Th cells correlate with the disease process [52]. Monocytes and macrophages use receptor activator of nuclear factor κB ligand (RANKL) to trigger the generation of osteoclasts, whereas fibroblasts interact directly with the RANK receptor on dendritic cells, macrophages, and preosteoclasts [53]. At the so-called “bare area,” where the periosteal synovial membrane, bone, and cartilage meet, bony erosions occur [54]. Cytokines activate genes encoding systems that can worsen inflammation and cellular and tissue damage by binding homologous receptors and initiating a variety of intracellular signal transduction events [55].
Figure 2 Cellular and cytokine interactions in the pathogenesis of rheumatoid arthritis (figure created with BioRender.com).
Molecular targets for RA treatment and the most common pathway
Numerous molecular targets and signaling pathways are involved in the intricate pathophysiology of RA, which have been extensively investigated to create efficacious treatment plans. Pro-inflammatory cytokines, immune cells, and intracellular signaling molecules are important molecular targets for RA treatment [56].
TNF-α, a pro-inflammatory cytokine essential to the pathophysiology of RA, is among the best-known molecular targets in RA treatment. Infliximab, adalimumab, and etanercept are examples of TNF-α inhibitors that have demonstrated notable effectiveness in lowering inflammation and delaying the course of disease [57]. IL-1 and IL-6 are other notable cytokine targets. Tocilizumab and other IL-6 receptor inhibitors have shown promise in treating RA symptoms and avoiding joint degeneration [58]. Despite being less frequently used than TNF-α and IL-6 inhibitors, IL-1 receptor antagonists, including anakinra, have demonstrated potential in RA treatment [59].
Immune cell-specific molecules have become notable therapeutic targets in RA, in addition to cytokine targets. Drugs such as rituximab, a monoclonal antibody that depletes CD20-positive B cells, can be used to target B cells, which are essential for autoantibody generation and T cell activation [60]. T cell-targeted treatments, such as abatacept, which binds CD80/CD86 on antigen-presenting cells and subsequently suppresses T cell co-stimulation, have been shown to be effective in RA [61]. A new method of treating RA uses Janus kinase (JAK) inhibitors, such as tofacitinib and baricitinib, which target intracellular signaling pathways implicated in cytokine generation and immune cell activation [44].
The inflammatory cascade involving TNF-α and its downstream signaling molecules is the most frequently identified route associated with the pathophysiology of RA and targeted for therapy [29]. The NF-κB route, the MAPK system, and the JAK-STAT pathway are among the intracellular signaling pathways activated by TNF-α. Pro-inflammatory mediators, MMPs, and other substances that cause synovial inflammation and joint degradation are produced through these pathways [62]. When the TNF-α pathway interacts with other cytokines, such as IL-6 and IL-1, it forms a network of inflammatory signals. TNF-α inhibitors, which target the TNF-α pathway, are highly successful in controlling RA symptoms and delaying the course of the illness in many individuals [63].
The JAK-STAT signaling system, which is involved in the transmission of signals from many cytokines, such as IL-6, interferon-gamma, and several interleukins, is another important mechanism in RA pathophysiology [64]. JAK inhibitors targeting this route have demonstrated encouraging outcomes in RA treatment, particularly in patients who do not respond well to biologic therapies or traditional DMARDs. The JAK-STAT pathway is a favorable target for RA therapy because it is essential for immune cell activation, differentiation, and cytokine generation [64].
Because B cells aid in the generation of autoantibodies and the activation of T cells, the B cell receptor signaling pathway plays a crucial role in the pathophysiology of RA [65]. Rituximab and other B-cell depletion treatments targeting this pathway have demonstrated effectiveness in controlling RA symptoms and averting joint deterioration. Additionally, as demonstrated by abatacept’s ability to effectively suppress T cell activation, the T cell receptor signaling pathway and co-stimulatory pathways are crucial targets in RA treatment [66].
The molecular and immunological mechanisms outlined above, including TNF-α, IL-6, JAK/STAT, MAPK, and NF-κB signaling, directly represent the therapeutic nodes modulated by many phytochemicals. Therefore, the subsequent sections examine how specific classes of plant-derived compounds, such as flavonoids, alkaloids, terpenoids, and phenolic compounds, target these same cytokines and intracellular signaling cascades [67]. By mapping phytochemical action onto these molecular pathways, this review provides a cohesive understanding of how natural products can modulate the immune dysfunction characteristic of RA [68].
Current pharmacological treatments and limitations
Decreasing inflammation, controlling pain, and delaying the disease course are the main goals of current RA treatment approaches [69]. The mainstay RA treatment consists of DMARDs, including methotrexate, the most frequently administered conventional DMARD [70]. Methotrexate can have adverse effects such as fatigue, nausea, and liver damage, although it works well for many patients. Biologic DMARDs that target certain inflammatory pathways have shown potential in situations in which traditional DMARDs are insufficient. These consist of inhibitors of JAK, IL, and TNF [71]. However, because of the formation of anti-drug antibodies, biologic DMARDs can lose their effectiveness over time and are associated with elevated infection risk [72]. Although glucocorticoids, such as prednisone, are frequently used to treat short-term symptoms, they can cause serious long-term adverse effects, such as weight gain, osteoporosis, and elevated risk of cardiovascular disease [73]. Although they decrease pain, NSAIDs might not change the course of a disease and, when used for extended periods, can increase cardiovascular disease risk or cause gastrointestinal problems [74]. A substantial fraction of patients with RA do not experience remission or low disease activity with current medications, although numerous therapy alternatives are available. Furthermore, many patients’ access to biologic DMARDs is restricted by their high cost, particularly in settings with low resources [75, 76]. Because RA is heterogeneous, each patient’s response to treatment can substantially differ; therefore, a trial-and-error method is necessary to determine the best course of action. In addition to being time-consuming, this procedure can cause patients to experience needless adverse effects [77]. Concerns regarding possible undiscovered dangers associated with extended use have also been raised, given that the long-term safety profiles of more recent biologic and targeted synthetic DMARDs remain unclear [78]. The burden of managing RA is increased for patients and healthcare systems by the requirement for regular monitoring and laboratory testing to evaluate medication effectiveness and identify any toxicities. Identifying biomarkers to forecast therapy response and individualize treatment plans, as well as creating more targeted medicines with better safety profiles, are becoming increasingly important. Although rheumatoid arthritis treatment has substantially advanced in the past few decades, more accessible, safe, and effective therapeutic choices remain necessary to enhance outcomes for all patients with RA [79].
Herbal remedies in the treatment of rheumatoid arthritis
In preclinical or clinical trials, several medicinal plants have been experimentally confirmed to aid in RA treatment. Pharmacological investigations directly or indirectly assessing the most frequently described medicinal plants used to treat RA are included in this discussion. Table 1 shows a list of herbal medicines with anti-RA properties.
Table 1 Herbal Remedies for Anti-Rheumatoid Arthritis
| Scientific Name | Part Used | Active Constituents | Method (Model) | Outcome | References |
|---|---|---|---|---|---|
| Aframomum meleguetta | Seeds | Essential oils | CFA-induced arthritis in rats | Antioxidant; inhibits NO and COX; decreases paw edema and inflammation | [180, 181] |
| Ajuga iva | Aerial parts | Flavonoids, Iridoids | CIA rats | ↓ IL-1β, IL-6, TNF-α; cartilage protection | [182] |
| Akebia quinata | Stem | Triterpenoids | AIA rats | ↓ TNF-α; anti-inflammatory | [183] |
| Allium cepa | Bulb | Quercetin | LPS macrophages; AIA rats | ↓ NO, PGE2; suppresses NF-κB and cytokines | [184, 185] |
| Allium sativum | Bulb | Allicin, ajoene | RAW264.7; rats with CFA-induced arthritis | ↓ COX-2, iNOS, TNF-α, IL-1β, RF | [186] |
| Aloe barbadensis | Leaf gel | Anthraquinones | LPS RAW264.7; rats with CFA-induced arthritis | ↓ COX-2, iNOS, PGE2; ↓ paw swelling | [187, 188] |
| Alpinia officinarum | Rhizomes | Flavonoids | LPS RAW264.7; rats with CFA-induced arthritis | ↓ NO, IL-1β; antioxidant | [189] |
| Ammodaucus leucotrichus | Seeds | Phenolics | CIA and rats with CFA-induced arthritis | ↓ MDA, MPO; ↑ GSH; cartilage protection | [190] |
| Anacyclus pyrethrum | Roots | Alkylamides | Rats with CFA-induced arthritis | ↓ Cytokines; antioxidant | [191, 192] |
| Argania spinosa | Kernel oil | Tocopherols | LPS RAW264.7; mice with CFA-induced arthritis | ↓ NO, TNF-α; macrophage modulation | [193] |
| Artemisia iwayomogi | Leaves | Flavonoids | LPS macrophages; AIA rats | ↓ TNF-α, IL-6; antioxidant | [194] |
| Asphodelus tenuifolius | Tuber | Phenolics | Rats with CFA-induced arthritis | ↓ COX-2, iNOS, TNF-α, IL-1β, RF | [195, 196] |
| Bambusa arundinacea | Seeds | Phenolics | RA rats | ↓ Paw swelling, TNF-α, RF | [197] |
| Beta vulgaris | Root | Betalains | AIA rats | ↓ TNF-α; improves antioxidant enzymes | [198] |
| Boerhavia diffusa | Roots | Punarnavine | LPS cells; LPS mice | ↓ TNF-α, IL-1β, IL-6 | [199] |
| Brassica oleracea | Seeds | Glucosinolates | Rats with CFA-induced arthritis | ↓ IL-1β, PGE2 | [200] |
| Brassica rapa | Roots | Phenolics | LPS macrophages; rats with CFA-induced arthritis | ↓ NF-κB, TNF-α, IL-6, NO; ↑ antioxidant | [201] |
| Camellia sinensis | Leaves | EGCG | B-cell assay; CIA rodents | ↓ BAFF; anti-inflammatory | [202] |
| Calotropis procera | Latex | Cardiac glycosides | Rats with CFA-induced arthritis | ↓ TNF-α, IL-6, PGE2; ↓ joint damage | [203] |
| Capparis spinosa | Fruit/leaves | Flavonoids | LPS RAW264.7; rats with CFA-induced arthritis | ↓ NO, TNF-α; inhibits COX-2, NF-κB | [204] |
| Capsicum annuum | Fruits | Capsaicin | LPS macrophages; rats with CFA-induced arthritis | ↓ IL-6, TNF-α; inhibits COX-2 | [205] |
| Carum carvi | Seeds | Carvone | Rats with CFA-induced arthritis | ↓ Swelling, RF, IL-6; antioxidant | [192] |
| Cassia absus | Whole plant | Anthraquinones | AIA rats | ↓ TNF-α, IL-1β; ↓ leukocytes | [24] |
| Caulophyllum robustum | Root | Alkaloids | LPS RAW264.7; CIA mice | ↓ IL-1, IL-6, TNF-α; inhibits NF-κB | [206] |
| Ceratonia siliqua | Pods/seeds | Tannins | Rats with CFA-induced arthritis | ↓ MDA; ↑ SOD, GSH; joint protection | [26] |
| Cinnamomum camphora | Leaves | Camphor | LPS RAW264.7; rats with CFA-induced arthritis | ↓ TNF-α, NO; ↓ COX-2 | [207] |
| Cinnamomum cassia | Bark | Cinnamaldehyde | LPS macrophages; rats with CFA-induced arthritis | ↓ Cytokines; inhibits NF-κB | [208] |
| Citrus aurantium | Leaves | Flavonoids | Rats with CFA-induced arthritis | ↓ CRP, cytokines; ↓ inflammation | [209] |
| Citrus limon | Peel/juice | Limonene, flavonoids | LPS macrophages; AIA rats | ↓ TNF-α, IL-6; ↑ SOD, GSH | [210] |
| Coriandrum sativum | Leaves/seeds | Linalool | LPS macrophages; rats with CFA-induced arthritis | ↓ Cytokines; antioxidant | [211, 212] |
| Crocus sativus | Stigma | Crocin, safranal | LPS macrophages; rats with CFA-induced arthritis | ↓ NF-κB, COX-2, TNF-α | [213] |
| Cucumis sativus | Fruit | Cucurbitacins | RAW264.7; rats with CFA-induced arthritis | ↓ IL-1β, TNF-α; antioxidant | [214] |
| Cucurbita maxima | Seeds | Sterols, fatty acids | Rats with CFA-induced arthritis | ↓ TNF-α, IL-1β; cartilage protection | [215] |
| Curculigo rhizoma | Rhizomes | Curculigoside A | CIA/AA rats | ↓ TNF-α, IL-6; inhibits JAK/STAT, NF-κB | [216] |
| Curcuma longa | Rhizomes | Curcumin | LPS macrophages; CIA rats | ↓ NF-κB, COX-2, TNF-α; antioxidant | [217, 218] |
| Elettaria cardamomum | Seeds | 1,8-Cineole | LPS RAW264.7; rats with CFA-induced arthritis | ↓ NO, TNF-α; ↓ oxidative damage | [219] |
| Equisetum arvense | Whole plant | Silicic acid | LPS macrophages; rats with CFA-induced arthritis | ↓ iNOS, COX-2; antioxidant | [220] |
| Eucalyptus globulus | Leaves | 1,8-Cineole | LPS macrophages; rats with CFA-induced arthritis | ↓ IL-1β, TNF-α; antioxidant | [221] |
| Eugenia caryophyllata | Flower buds | Eugenol | LPS RAW264.7 | ↓ NF-κB, COX-2, TNF-α | [222] |
| Ficus carica | Fruit/leaf | Ficin, polyphenols | Rats with CFA-induced arthritis | ↓ Oxidative stress; improved histology | [223] |
| Foeniculum vulgare | Seeds | Anethole | LPS macrophages; rats with CFA-induced arthritis | ↓ TNF-α, IL-6; antioxidant | [224] |
| Glycine max | Seeds | Genistein | RAW264.7; CIA mice | ↓ TNF-α; inhibits T-cell activation | [225] |
| Glycine tabacina | Roots | Isoflavonoids | CIA mice | ↓ TNF-α, IL-6; antioxidant | [226] |
| Glycyrrhiza glabra | Roots | Glycyrrhizin | LPS macrophages; rats with CFA-induced arthritis | ↓ TNF-α, IL-1β; ↑ antioxidants | [227] |
| Harpagophytum procumbens | Roots | Harpagoside | Human synovial cells; AIA rats | ↓ COX-2, NO, MMPs; ↓ cartilage damage | [228] |
| Helianthus annuus | Seeds (oil) | Linoleic acid | LPS macrophages; rats with CFA-induced arthritis | ↓ IL-1β, TNF-α; ↑ antioxidant enzymes | [229] |
| Illicium verum | Fruit | Anethole | LPS RAW264.7; rats with CFA-induced arthritis | ↓ NO, PGE2; ↓ COX-2 | [230] |
| Ipomoea batatas | Tuber | Anthocyanins | RAW264.7; CIA mice | ↓ NF-κB, IL-1β, TNF-α | [231] |
| Juglans regia | Leaves | Polyphenols | LPS macrophages; AIA rats | ↓ TNF-α, IL-6; ↑ antioxidant activity | [232–235] |
| Lavandula angustifolia | Flowers | Linalool, linalyl acetate | LPS RAW264.7; rats with CFA-induced arthritis | ↓ TNF-α, NO; antioxidant | [236] |
| Lepidium sativum | Seeds | Flavonoids | Rats with CFA-induced arthritis | ↓ Oxidative stress, cytokines | [236] |
| Linum usitatissimum | Seeds | Lignans, ALA | TNF-α synoviocytes; CIA mice | ↓ MMPs; protects cartilage | [237, 238] |
| Lippia citriodora | Leaves | Verbascoside | LPS RAW264.7; AIA mice | ↓ TNF-α, NF-κB; antioxidant | [54] |
| Marrubium vulgare | Leaves | Marrubiin | Rats with CFA-induced arthritis | ↓ TNF-α; antioxidant | [239, 240] |
| Nardostachys jatamansi | Rhizomes | Jatamansone | RAW264.7; rats with CFA-induced arthritis | ↓ TNF-α, COX-2; ↓ bone erosion | [241] |
| Nerium oleander | Leaves | Cardiac glycosides | LPS macrophages; rats with CFA-induced arthritis | ↓ NO, TNF-α; antioxidant | [57] |
| Nyctanthes arbor-tristis | Leaves, bark | Iridoids | Cell cultures; RA models | ↓ inflammatory mediators | [242, 243] |
| Ocimum basilicum | Leaves | Eugenol, linalool | LPS RAW264.7; mice with CFA-induced arthritis | ↓ TNF-α, IL-6; antioxidant | [244] |
| Olea europaea | Leaves, oil | Oleuropein | LPS macrophages; rats with CFA-induced arthritis | ↓ COX-2, iNOS; ↓ TNF-α | [245] |
| Opuntia monacantha | Pulp/peel | Betalains | LPS RAW264.7; rats with CFA-induced arthritis | ↓ IL-6, TNF-α; antioxidant | [246] |
| Panax ginseng | Roots | Ginsenosides | RAFLS, T cells; rats with CFA-induced arthritis | ↓ NF-κB, TNF-α; immunomodulatory | [247, 248] |
| Peganum harmala | Seeds | β-Carbolines | LPS macrophages; rats with CFA-induced arthritis | ↓ TNF-α, IL-1β; COX-2 inhibition | [249] |
| Phoenix dactylifera | Seeds/pulp | Polyphenols | LPS RAW264.7; rats with CFA-induced arthritis | ↓ TNF-α, IL-6; inhibits NF-κB | [250] |
| Piper longum | Fruit | Piperine | RAW264.7; AIA rats | ↓ NF-κB, IL-1β, TNF-α; ↓ COX-2 | [251] |
| Plumbago zeylanica | Roots | Plumbagin | LPS RAW264.7; rats with CFA-induced arthritis | ↓ TNF-α, IL-6; inhibits NF-κB, JAK/STAT | [252] |
| Punica granatum | Peel/juice | Punicalagin | RAFLS; CIA rats | ↓ MMPs, NF-κB, COX-2 | [253] |
| Ricinus communis | Seeds (oil) | Ricinoleic acid | Rats with CFA-induced arthritis | ↓ Inflammatory mediators | [254] |
| Rosmarinus officinalis | Leaves | Rosmarinic acid | LPS macrophages; rats with CFA-induced arthritis | ↓ TNF-α, IL-1β; antioxidant | [255] |
| Sesamum indicum | Seeds, oil | Sesamin | LPS macrophages; CIA rats | ↓ TNF-α, IL-6; ↓ COX-2 | [256] |
| Silybum marianum | Whole plant | Silymarin | AIA rats | ↓ Leukotrienes; antioxidant | [257] |
| Tinospora cordifolia | Stem | Alkaloids, terpenoids | LPS RAW264.7; CIA rats | ↓ IL-6, TNF-α, NO; ↓ JAK/STAT | [258] |
| Tripterygium wilfordii | Roots | Triptolide | Macrophages; CIA rats | ↓ NF-κB, MAPK; ↓ cytokines, bone erosion | [259] |
| Trigonella foenum-graecum | Seeds | Saponins, flavonoids | Rats with CFA-induced arthritis | ↓ IL-6, TNF-α; cartilage protection | [260, 261] |
| Urtica pilulifera | Leaves | Flavonoids | Rats with CFA-induced arthritis | ↓ TNF-α, IL-1β; antioxidant | [262] |
| Withania somnifera | Roots | Withanolides | Rats with CFA-induced arthritis | ↓ TNF-α, IL-1β; ↑ antioxidant enzymes | [263] |
| Zingiber officinale Rosc. | Rhizomes | Gingerols, shogaols | RAFLS, CIA rats | ↓ NF-κB, COX-2, TNF-α, IL-1β | [264] |
| Ziziphus nummularia | Leaves, stem bark | Polyphenols | LPS RAW264.7; rats with CFA-induced arthritis | ↓ NF-κB, NO, PGE2, TNF-α | [265] |
Bioactive phytochemical classes and their effects
The pathological mechanisms in RA comprise long-term cytokine release (TNF-α and IL-6); NF-κB and MAPK activation; oxidative stress; and proliferation of FLSs. Against this molecular backdrop, phytochemicals exert their therapeutic effects. Each class of phytochemicals discussed herein acts on one or more of these immunological targets, thereby mirroring or complementing the pharmacological action of biologics and DMARDs.
Flavonoids
Flavonoids, polyphenolic pigments found in a wide variety of fruits, vegetables, and herbs, including tea, ginger, parsley, and onions, have known antioxidant and anti-inflammatory qualities. In RA models, many flavonoids have demonstrated anti-arthritic properties, primarily by inhibiting pro-inflammatory cytokines [80]. One such promising flavonol is galangin, which is present in propolis, honey, and Alpinia galanga. In mice with CIA, galangin has been shown to decrease joint inflammation and suppress the growth of synoviocytes that resemble RA fibroblasts [81]. In synoviocytes, galangin mechanistically decreases TNF-α, IL-1β, IL-18, COX-2, and inducible nitric oxide synthase (iNOS) levels by downregulating NF-κB and NLRP3 signaling. This flavonoid prevents oxidative damage to synoviocytes by inhibiting nuclear translocation of NF-κB and activating antioxidant enzymes, thus increasing SOD while decreasing malondialdehyde [82].
Other flavonoids have shown comparable characteristics. For example, kaempferol, found in tea, broccoli, and apples, has demonstrated anti-inflammatory properties in vitro. Kaempferol inhibits the NLRP3 inflammasome cascade (pyroptosis) in RA fibroblasts by binding caspase-1 [83]. By preventing caspase-1 activation, kaempferol treatment decreases RA-FLS migration and proliferation, and markedly decreases the release of IL-1β and IL-18 [84].
Flavones identical to luteolin and apigenin (found in celery and parsley) have also shown effectiveness in RA models. According to preclinical research, these phytochemicals block synoviocyte inflammatory signaling and lower blood TNF-α and IL-6 in arthritic rats. For example, apigenin inhibits dendritic cell migration and maturation, thus delaying the onset of CIA and decreasing serum TNF-α/IL-1β ratios [85]. Similarly, by suppressing NF-κB and modifying Th1/Th17 cytokines (lowering IFN-γ and IL-17), fisetin and hesperidin have been found to decrease arthritis scores in rats. Thus, flavonoids block NF-κB/MAPK pathways and downregulate pro-inflammatory mediators (TNF-α, IL-6, and IL-17) [86]. Additionally, they enhance antioxidant defenses and anti-inflammatory cytokines (IL-10), which together decrease RA pathogenesis [87]. In agreement with the cytokine-driven inflammation described in Section 3.1, flavonoids inhibit TNF-α and IL-6 signaling by blocking NF-κB translocation and NLRP3 activation.
Alkaloids
Alkaloids are plant constituents that contain nitrogen and frequently have potent bioactivities. Remarkably, sinomenine, an alkaloid obtained from Sinomenium acutum, is used to treat RA in traditional Chinese medicine [88]. According to recent research, sinomenine markedly decreases synovial inflammation. Simoenine inhibits crucial signaling and decreases MMP9 expression in LPS-stimulated macrophages [89]. MMP9 has been identified as a sinomenine target through network pharmacology analysis and trials. MMP9 is downregulated, and TNF-α/IL-6 production decreases, when activated RAW264.7 cells are treated with sinomenine. According to these results, sinomenine decreases cytokine-driven damage in RA by obstructing the MMP9/NF-κB axis [90].
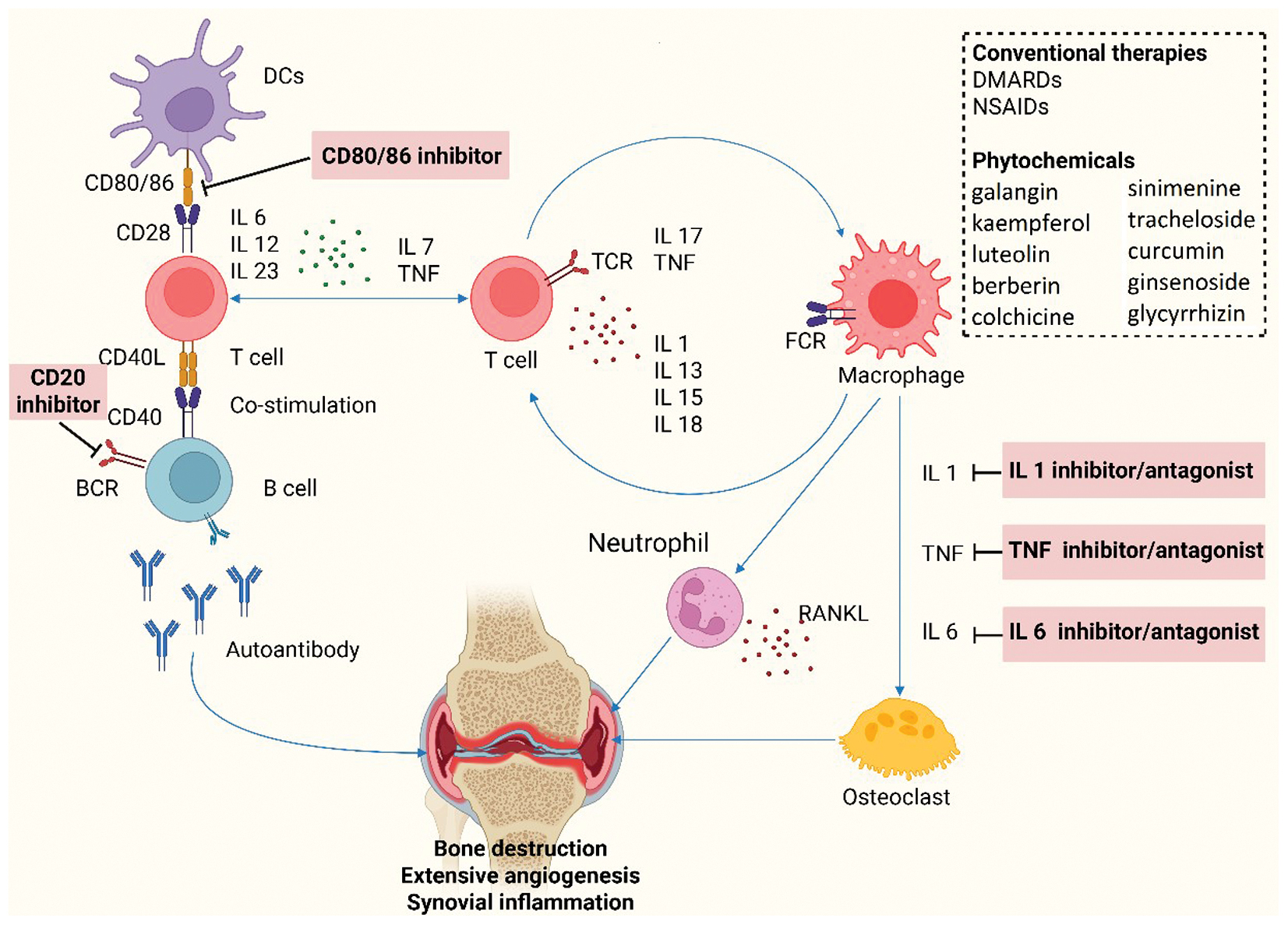
Berberine (found in Coptis and Berberis) is an alkaloid with confirmed immunomodulatory properties. Berberine often decreases IL-1β, TNF-α, and IL-6 in arthritic animals, partly by changing the gut microbiota and modifying TLR4/NF-κB signaling; however, few comprehensive mechanistic investigations have been conducted in RA [91, 92]. Piperine, from black pepper, has demonstrated anti-arthritic synergy by inhibiting TNF-α and IL-1β in synoviocytes and increasing the efficacy of corticosteroids [93]. In rats, CIA has been ameliorated by matrine and oxymatrine (found in Sophora flavescens), which lower synovial TNF-α and IL-17 levels [94, 95]. In addition, the alkaloid colchicine (found in Colchicum spp.) decreases neutrophil-driven inflammation in arthritic models, despite being better known for gout treatment. Alkaloids are attractive immune-targeting drugs, because they often decrease oxidative stress, inhibit NF-κB, and lower inflammatory cytokines and enzymes in RA models [96]. As shown in Figure 3, macrophage-derived TNF-α and IL-1β increase synovial inflammation, whereas alkaloids such as sinomenine directly downregulate these pathways, thus linking phytochemical intervention to RA pathophysiology.
Figure 3 Schematic representation of immune cell interactions and cytokine networks in rheumatoid arthritis pathogenesis, and therapeutic interventions targeting key cytokine pathways (figure created with BioRender.com).
Terpenoids
Terpenoids, a structurally diverse class of natural products (also called secondary or specialized metabolites), have shown notable anti-inflammatory and immunomodulatory effects in preclinical models of RA [97]. Evidence from in vivo studies, such as those using CIA or AIA models, has indicated that triterpenoids such as boswellic acid and triptolide suppress inflammatory mediators through inhibition of the NF-κB and MAPK signaling pathways [98, 99]. For example, boswellic acid inhibits IKKβ activation and decreases TNF-α and IL-6 levels in a CIA rat model, whereas triptolide decreases IL-17 and MMP expression in synovial tissues, thus diminishing joint swelling and cartilage damage [99]. Similarly, in vitro studies in macrophage or synoviocyte cultures have confirmed that terpenoids such as thymoquinone downregulate COX-2 and iNOS expression, thus corroborating their molecular effects detected in animal models [100]. However, these inhibitory properties are compound-specific rather than widespread across all terpenoids, and direct comparative studies between terpenoid subclasses are limited [101, 102].
Phenolic compounds (non-flavonoid phenolics)
Phenolic acids, curcuminoids, and stilbenes have been explored for their antioxidant and anti-inflammatory properties in RA-relevant preclinical systems; however, the potential mechanistic target of their action should be considered with caution [103]. In vivo studies on curcumin and resveratrol have confirmed decreased paw edema and TNF-α and IL-1β expression, as well as restoration of antioxidant enzyme levels via modulation of the NF-κB and Nrf2 pathways [104]. For example, a curcumin treatment in a CIA rat model decreases COX-2 and MMP-9 levels, thus supporting curcumin’s disease-modifying potential under experimental conditions [105]. In in vitro macrophage and synoviocyte assays, rosmarinic acid and caffeic acid have been revealed to downregulate pro-inflammatory enzymes and cytokines, thus providing a molecular justification for their signaling effects [106]. Nonetheless, the pan-class inhibitory report of phenolic compounds on NF-κB or MAPK signaling should be considered a general trend rather than a universal outcome across all compounds [107]. Many reports have used cell-based anti-inflammatory assays rather than disease-specific in vivo RA models, thus limiting the direct traceability of certain mechanistic underlying to RA pathophysiology [108]. Therefore, although evidence supports phenolic compounds as promising modulators of inflammatory cascades, the strength of evidence remains specific to each compound and context, and further standardized studies are warranted to validate comparative efficacy within RA-relevant systems [109].
Saponins
Triterpene or steroidal glycosides, known as saponins, have immunomodulatory properties [110]. Triterpenoid saponins, such as ginsenosides from Panax ginseng and glycyrrhizin from licorice root, have been investigated in RA models. For example, compound K and ginsenoside Rb1 have been found to decrease splenic Th17 cells and joint swelling in a CIA rat model [111]. Glycyrrhizin decreases TNF-α and IL-1β by modulating NF-κB and MAPK [112]. Escin, a saponin derived from horse chestnuts, inhibits the generation of IL-8 and TNF-α in arthritic tissues [113]. Essentially, saponins attenuate cytokine cascades and complement-mediated inflammation by inhibiting pro-inflammatory transcription factors and enzymes. Their amphipathic properties have been found to facilitate phytochemical delivery. Saponin fractions, such as those from Astragalus, are widely known to decrease both IL-6 and paw edema in rat arthritic models, despite a paucity of mechanistic evidence [114].
Tannins
Less research has focused solely on tannins (polymeric phenolic compounds, including proanthocyanidins and hydrolyzable tannins); however, plant extracts high in tannins have been shown to be beneficial [115]. Strong antioxidants are found in green tea polyphenols, primarily catechins, a type of flavonoid. In vitro, epigallocatechin gallate (EGCG) inhibits COX-2 and TNF-α [116]. In synoviocytes, tannic acid inhibits the nuclear translocation of NF-κB [117]. In arthritic cartilage explants, ellagannins (such as pomegranate punicalagin) have been found to decrease the synthesis of MMP and IL-6 [118]. Grape seed extract, which is high in proanthocyanidin, has been observed to decrease CIA severity by inhibiting IL-17 [118]. In conclusion, tannins function primarily as antioxidants and moderate anti-inflammatory agents; chelate metal ions; and scavenge ROS, thereby indirectly decreasing inflammatory signals. Additionally, they block matrix-degrading and COX enzymes. Although few in vivo studies have focused specifically on RA, tannin-rich extracts have been repeatedly demonstrated to decrease joint damage and cytokine levels [118].
Lignans
Herbs, roots, and seeds all contain lignans, a family of phenylpropanoid dimers [119]. A recent study has demonstrated the anti-RA properties of tracheloside, a lignan derived from Trachelospermi Caulis. Tracheloside markedly decreases the release of inflammatory factors and synoviocyte migration in vitro [120]. Tracheloside-treated TNF-α-stimulated synoviocytes specifically display decreased expression of MMP-2/3/9, IL-6, IL-17, and COX-2. Additionally, the inactivity of the important signaling proteins JNK and p38 (MAPKs) has suggested that tracheloside inhibits the IL-17/MAPK pathway [121]. Overall, this lignan decreases several pro-arthritic mediators, thereby limiting synovial inflammation. However, data in RA models are lacking. Other lignans such as sesamin and pinoresinol (from plants and sesame) have been shown to alter NF-κB and arachidonic acid metabolism. However, evidence for tracheloside suggests that lignans target cytokine networks and MAPK in RA pathology [122].
Representative phytochemicals, including their plant origins, experimental paradigms, and documented anti-arthritic activities, are compiled in Table 2. In preclinical tests, each class of phytochemicals has generally been found to produce several molecules that lessen joint erosion, inflammatory cytokines, and paw edema.
Table 2 Anti-rheumatoid Arthritis Phytochemical Compounds from Plant Sources
| Phytochemical Class | Phytochemical | Plant Source | Model/System | Key Effects (Mechanism) | References |
|---|---|---|---|---|---|
| Flavonoid | Galangin | Alpinia officinarum | LPS-stimulated RA fibroblasts (in vitro) | ↓ TNF-α, IL-6, COX-2; inhibits NF-κB/NLRP3 signaling | [81, 82] |
| Kaempferol | Tea, broccoli, apples | RA fibroblasts (in vitro) | ↓ IL-1β, IL-18, caspase-1 activity; inhibits pyroptosis (blocks CASP1) | [83, 84] | |
| Luteolin | Celery, parsley | CIA model (rat) | ↓ IL-17, IFN-γ, ↑ IL-10; suppresses STAT3, NF-κB signaling (CII model) | [85] | |
| Apigenin | Petroselinum crispum | CIA model (rat) | ↓ Serum TNF-α/IL-1β ratios | [85] | |
| Fisetin | Rhus verniciflua | CIA model (rat) | ↓ IL-1β, TNF-α, IL-6/chemokines | [86] | |
| Hesperidin | Citrus sinensis | CIA model (rat) | ↓ TNF-α, IL-6, and IL-17; Block NF-κB/MAPK | [86] | |
| Alkaloid | Sinomenine | Sinomenium acutum | LPS-stimulated RAW264.7 (in vitro) | ↓ MMP9, TNF-α, IL-6; downregulates MMP9/NF-κB pathways | [88] |
| Berberine | Coptis spp., Berberis spp. | CIA model (rat) | ↓ IL-1β, TNF-α; modulates gut microbiota and TLR4/NF-κB | [91, 92] | |
| Piperine | Black pepper | CIA model (rat) | ↓ TNF-α, IL-1β and ↑ effectiveness of corticosteroids | [90] | |
| Matrine | Sophora flavescens | CIA model (rat) | ↓ TNF-α, IL-6, and IL-17; blocks NF-κB/MAPK | [94] | |
| Oxymatrine | Sophora flavescens | CIA model (rat) | ↓ TNF-α, IL-6, and IL-17; blocks NF-κB/MAPK | [95] | |
| Colchicine | Colchicum spp. | CIA model (rat) | ↓ TNF-α, IL-1β; inhibits COX-2, NF-κB | [96] | |
| Terpenoid | β-Boswellic acid | Boswellia serrata | AIA/CIA model (rat) | Binds TLR4/IL-1R; inhibits NF-κB/MAPK; ↓ ROS, IL-6, TNF-α, NLRP3 | [98] |
| Triptolide | Tripterygium wilfordii | AIA/CIA model (rat) | ↓ IL-17 and MMP expression in synovial tissues | [99] | |
| Thymoquinone | Nigella sativa (black cumin) | CIA model (mouse) | ↓ COX-2, PGE2; inhibits NF-κB; decreases TNF-α in synovium | [100] | |
| Phenolic acid and polyphenol | Curcumin | Curcuma longa | CIA model (rat) | ↓ TNF-α, IL-1β; inhibits COX-2, NF-κB; ↑ antioxidants (Nrf2) | [104] |
| Rosmarinic acid | Vitis vinifera | AIA/CIA model (rat) | ↓ IL-17/IL-23 inhibits NF-κB/MAPK | [106] | |
| Caffeic acid | Coffea arabica | AIA/CIA model (rat) | ↓ IL-17/IL-23; inhibits NF-κB/MAPK | [106] | |
| Saponin | Ginsenoside | Panax ginseng | CIA model (rat) | ↓ IL-17/IL-23; TNF-α and IL-1β by modulating NF-κB and MAPK | [112] |
| Glycyrrhizin | Glycyrrhiza glabra | CIA model (rat) | ↓ TNF-α and IL-1β by modulating NF-κB and MAPK | [113] | |
| Astragalus | Astragalus membranaceus | CIA model (rat) | ↓ IL-6, MMPs; inhibits NF-κB/MAPK | [114] | |
| Tannin | Punicalagin | Pomegranate peel | Synoviocyte culture | ↓ IL-6, COX-2; antioxidant scavenging (anti-NF-κB) | [115] |
| Catechins | Epigallocatechin gallate | CIA model (rat) | ↓ TNF-α, IL-1β; antioxidant effect | [117] | |
| Lignan | Tracheloside | Trachelospermi caulis | TNF-α-treated MH7A cells | ↓ IL-6, IL-17, COX-2, MMP-2/3/9; inhibits JNK/p38 (MAPK) | [120, 121] |
| Sesamin | Sesame seeds | AIA model (rat) | ↓ TNF-α, IL-1β; antioxidant effect (Nrf2 activation) | [122] | |
| Pinoresinol | Sesame seeds | AIA model (rat) | ↓ TNF-α and arachidonic acid metabolism | [122] |
Synergistic and multi-target actions of phytochemicals in RA management
RA is a multifactorial and network-driven disease, wherein inflammation, oxidative stress, and immune dysregulation occur simultaneously [123]. Consequently, single-target therapies such as TNF-α or IL-6 inhibitors might improve symptoms but frequently fail to achieve durable remission. In contrast, phytochemicals demonstrate synergistic and multi-target pharmacology, by acting on numerous molecular nodes within the same inflammatory cascade [124].
For example, curcumin simultaneously suppresses NF-κB activation, downregulates COX-2 and TNF-α, and stimulates the Nrf2/HO-1 antioxidant axis, thus achieving dual anti-inflammatory and cytoprotective effects. Resveratrol and quercetin moderate both cytokine expression (TNF-α, IL-6, and IL-17) and intracellular signaling (JAK/STAT and MAPK), while enhancing antioxidant defenses. Similarly, EGCG from green tea decreases B-cell activating factor (BAFF) signaling and inhibits macrophage-derived ROS, thus indicating cross-talk inhibition between adaptive and innate immunity [125].
These combinatorial mechanisms suggest that phytochemicals function not as agents in isolation but as network stabilizers capable of re-balancing immune, oxidative, and metabolic processes. Therefore, phytotherapy is distinct from mono-target methods and aligns with current systems-biology perspectives on chronic inflammatory diseases [126].
Moreover, evidence from co-administration studies has shown that combining phytochemicals (such as curcumin with resveratrol, or quercetin with EGCG) can achieve additive suppression of NF-κB and cytokine yield while decreasing the required dose of each compound. Combining these compounds with low-dose DMARDs might increase efficacy and minimize toxicity, thus suggesting a promising path for future translational research [127].
Critical appraisal and limitations of preclinical evidence
Although preclinical studies have provided a strong basis for understanding the anti-arthritic potential of phytochemicals, numerous important limitations warrant careful consideration. A key problem lies in the lack of consistency in experimental design across studies. Differences in animal strains, induction methods (e.g., AIA or CIA), treatment periods, and dosage regimens have hindered direct comparison of results or establishment of dose-response relationships [128]. Many studies have also included small sample sizes or short treatment durations, thus leading to potential overestimation of therapeutic efficacy, and failure to retain long-term toxicity or disease reduction potential [129].
An additional key limitation is the absence of standardization of herbal extracts and formulations. Phytochemical content changes by plant species, geographic origin, harvesting season, and extraction technique, thus leading to notable inconsistencies in pharmacological effects [130]. Limited studies have used chemically characterized or standardized extracts, and many of the active constituents responsible for the observed effects remain unidentified [131, 132].
Moreover, potential publication bias is evident within the current literature. Positive outcomes are commonly reported, whereas studies yielding negative or inconclusive results remain unpublished, thus prompting excessive optimism regarding phytochemical efficacy. The reproducibility of results is further hindered by inadequate methodological transparency, narrow use of appropriate controls, and partial reporting of statistical analyses [133].
Finally, toxicity and safety valuations are often underexplored. Although phytochemicals are generally considered safe, several classes, including alkaloids and terpenoids, might show hepatotoxic or immunosuppressive effects at high doses. Few studies have included comprehensive toxicological evaluations, such as chronic exposure, genotoxicity, or interaction with standard DMARDs [134, 135].
Mechanisms of action
The mechanisms of phytochemical action in RA closely resemble those of the immune and molecular events outlined in the pathophysiology section (Figure 3). By targeting main pro-inflammatory mediators (TNF-α, IL-6, and IL-1β) and intracellular pathways (NF-κB, MAPK, and JAK/STAT), these natural compounds interfere at multiple stages of RA progression from immune activation to joint destruction [136, 137]. NF-κB signaling is a key pathway. Pro-inflammatory stimuli (TNF-α, IL-1β, and LPS) in RA cause activation of Toll-like and TNF receptors on immunological and synovial cells, and consequently IκB kinase (IKK); degradation of IκBα; and nuclear translocation of NF-κB (p65/p50). NF-κB then stimulates the production of MMPs, COX-2, iNOS, and cytokines (TNF-α and IL-6) [29].
For example, kaempferol and galangin have been found to block synoviocyte NF-κB activity in the presence of LPS [138]. Sinomenine inhibits NF-κB signaling by downregulating MMP9, thereby decreasing inflammation [3]. Through preventing TLR4/IL-1 receptor-mediated IKK activation, boswellic acid specifically inhibits NF-κB [139]. Traditional NF-κB inhibitors, such as curcumin and resveratrol, alleviate IκBα and block nuclear translocation of NF-κB p65. TNF-α, IL-6, IL-1β, COX-2, and MMP transcription are all decreased by NF-κB nuclear translocation, which is a commonly observed molecular mechanism in RA pathogenesis of several phytochemicals [111].
MAPKs, which stimulate cytokine gene expression and synoviocyte proliferation, are triggered by pro-inflammatory cytokines [140]. Lignans such as tracheloside directly inhibit JNK and p38 phosphorylation in RA synoviocytes. For example, the terpenoid thymoquinone blocks MAPKs. Phytochemicals decrease the amplification of IL-17, IL-6, and other mediators in RA by inhibiting MAPKs [141].
Another target is the COX-2/prostaglandin E2 (PGE2) pathway. Several phytochemicals suppress the expression or activity of COX-2. For example, when TNF-α is stimulated, synoviocytes treated with trachelosides display decreased COX-2 protein [121]. COX-2 inhibitors include resveratrol and curcumin. These substances diminish joint pain and vascular inflammation by inhibiting COX-2 and PGE2 [3]. However, these mechanisms do not occur in isolation. Most phytochemicals act on multiple intracellular pathways simultaneously, thus synergistically modulating cytokine, oxidative, and apoptotic networks. This polypharmacological nature contrasts with conservative single-target biologics and reflects the intrinsic complexity of herbal matrices [142].
Cytokine modulation, NLRP3 inflammasomes, and pyroptosis
Flavonoids, alkaloids, and other compounds decrease serum/secreted levels of TNF-α, IL-6, IL-1β, and IL-17 in RA models. For example, sinomenine-treated cells show markedly decreases in IL-6 and TNF-α (via NF-κB/MMP9 suppression) [1]. Kaempferol-bound caspase-1 decreases the release of IL-1β and IL-18 by inhibiting pyroptosis [143]. Galangin markedly decreases IL-1β, TNF-α, IL-18, and IL-6 in synoviocytes [144]. This inhibition prevents the cytokine storm that leads to the development of pannus. Additionally, by upregulating anti-inflammatory cytokines (such as IL-10 and TGF-β), certain phytochemicals alter the Th17/Treg balance. Studies have indicated that flavonoids often increase IL-10 in RA models, thus enhancing inflammation resolution [145], although research in this area is limited.
Oxidative stress and antioxidant effects
Reactive oxygen and nitrogen species, which are found in RA synovium, further deteriorate cartilage. Numerous phytochemicals have strong antioxidant properties [146]. Galangin administration to RA synoviocytes in vitro decreases lipid peroxidation marker malondialdehyde (MDA) and increases SOD activity [138]. SOD, catalase, and glutathione peroxidase are antioxidant enzymes that are made in joints after curcumin and resveratrol activate Nrf2. ROS subsequently activate NF-κB, whereas phytochemicals that deactivate ROS prevent oxidative damage and indirectly lower NF-κB activation. Therefore, their anti-cytokine actions are enhanced by their antioxidant activity [3].
Inhibition of matrix metallo-proteinases
MMPs, particularly MMP-1, MMP-3, and MMP-9, are overproduced by synovial fibroblasts in RA and break down cartilage collagen. MMP expression is normally suppressed by phytochemicals. Sinomenine decreases MMP9 levels in macrophages [147]. Tracheloside decreases the levels of MMP2/3/9 in synoviocytes activated by TNF-α. Inhibiting MMPs helps maintain joint integrity by preventing the breakdown of cartilage and preserving the structural integrity of the joints. Likewise, diminished PGE2-mediated MMP induction results from a decrease in COX-2. Phytochemicals in arthritis preserve the cartilage matrix by inhibiting these proteases [117].
Apoptosis and cell proliferation
The development of pannus is facilitated by the dysregulated proliferation of FLSs. Some phytochemicals shift the balance toward cell death in hyperplastic synovium or induce apoptosis in FLSs. Flavonoids have been shown to cause apoptosis in FLSs by upregulating p53 and caspase-3 [148]. Triptolide and curcumin have been found to halt the growth of FLSs. Moreover, certain alkaloids cause T cells to undergo apoptosis in immune cells [149].
Mechanistic insights regarding representative phytochemicals in RA
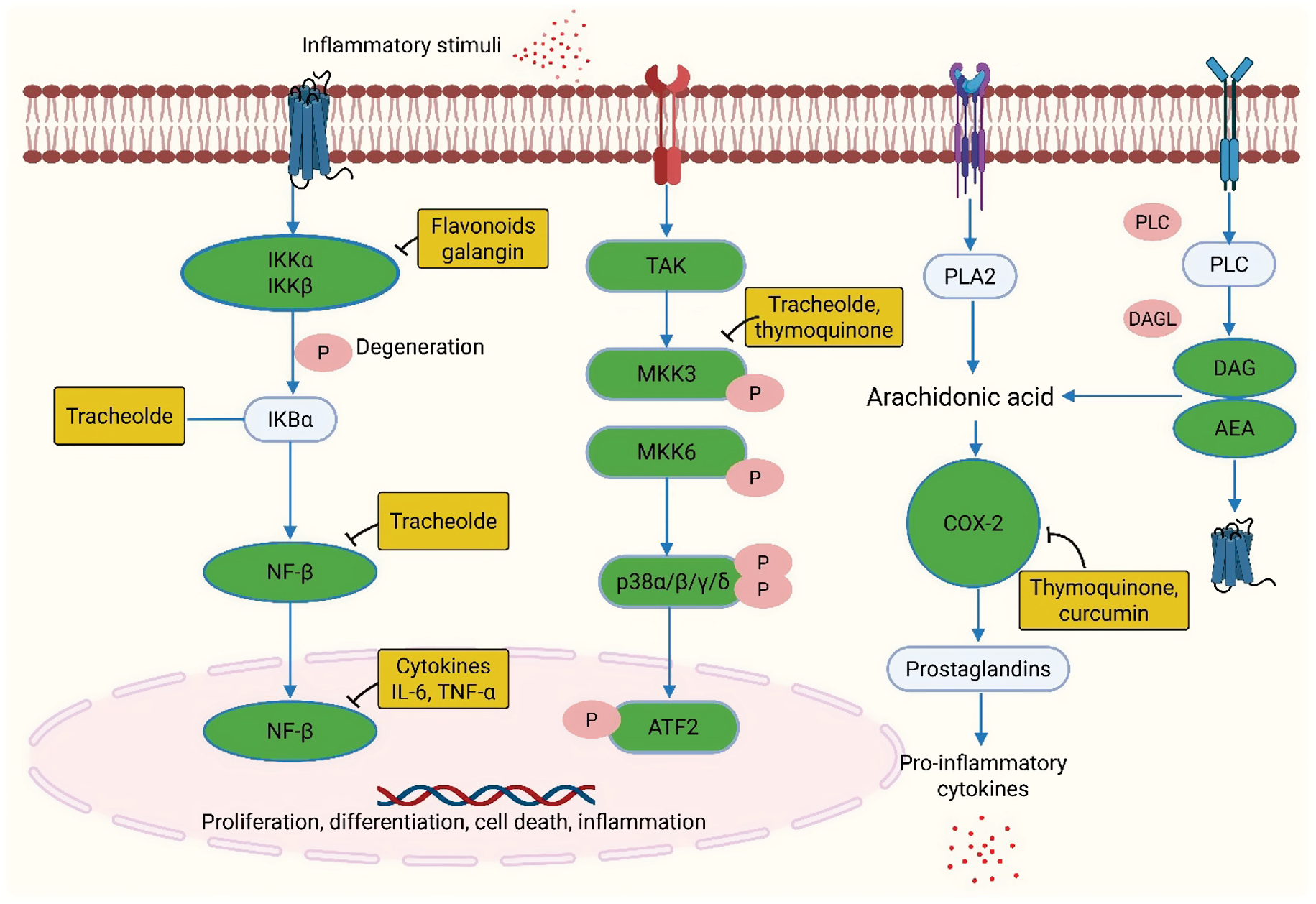
To improve mechanistic understanding, we examined representative compounds from key phytochemical classes to reveal how they modulate RA-relevant molecular targets, such as the NF-κB, MAPK, and JAK/STAT pathways (Figure 4).
Figure 4 The NF-κB signaling pathway and mitogen-activated protein kinase (MAPK) pathways, including JNK, p38, and ERK α/β/γ/δ and cyclooxygenase-2 (COX-2)/prostaglandin E2 (PGE2) pathway, are targets for rheumatoid arthritis (figure created with BioRender.com).
Flavonoids such as quercetin and kaempferol exert anti-arthritic effects by suppressing the activation and nuclear translocation of NF-κB p65 in synoviocytes and macrophages [150]. Quercetin inhibits IκBα phosphorylation and stabilizes cytoplasmic NF-κB, thereby decreasing the transcript levels of pro-inflammatory mediators (TNF-α, IL-1β, COX-2, and iNOS) [151]. Kaempferol further modulates NLRP3 inflammasome and caspase-1 activity, thus decreasing IL-1β and IL-18 release, and controls oxidative stress through Nrf2/HO-1 activation, thus improving SOD and catalase activity. This dual modulation of inflammatory and antioxidant systems is a principal mechanism through which flavonoids restore the redox balance in RA synovium [152].
Among alkaloids, sinomenine and berberine have well-characterized immunomodulatory activity. Sinomenine targets the MMP-9/NF-κB axis in macrophages and FLSs, thereby decreasing the degradation of joint cartilage and increasing protective cytokines [153]. Berberine exerts balancing effects by suppressing JAK2/STAT3 phosphorylation and modulating the gut-immune axis, thus ultimately decreasing Th17 cell differentiation and IL-17 mediated joint inflammation [92].
Terpenoids, primarily boswellic acid and triptolide, strongly inhibit innate immune pathways. Boswellic acid directly inhibits TLR4/IL-1 receptor-mediated signaling, and consequently prevents IKK activation and subsequent NF-κB translocation. This compound also prevents the expression of iNOS and COX-2, thus decreasing PGE2 production and oxidative injury. In contrast, triptolide suppresses JAK/STAT and MAPK signaling in T cells and synoviocytes, and consequently decreases IL-6, IL-17, and MMP-3 expression, while stimulating apoptosis in hyperproliferative synovial fibroblasts [139, 154].
Critical evaluation of preclinical models in rheumatoid arthritis research
Most markers associated with the anti-arthritic potential of phytochemicals have been identified for preclinical investigations using both in vitro and in vivo models; however, these models substantially vary in their strategies, quality, and translational relevance to human RA. In vitro methods such as FLSs (RA-FLSs), RAW 264.7 macrophages, and LPS-stimulated immune cells have provided valuable mechanistic insights into cytokine regulation, oxidative stress, and signaling activation (e.g., NF-κB, MAPK, and JAK/STAT), but they lack the cellular complexity and immune interactions existing in human joints [155]. Among in vivo models, the CIA model most closely mimics autoimmune features of human RA, whereas AIA and Complete Freund’s adjuvant (CFA)-induced arthritis reflect primarily the inflammatory or degenerative aspects of the disease [156]. Nonetheless, contradictions in experimental design, such as differences in dosage, duration, strain susceptibility, and outcome counting, limit comparability and reproducibility across studies. Furthermore, several models neglect major clinical variables such as sex differences, comorbidities, and chronicity, thus limiting their predictive power for human efficacy [157].
Bridging the translational gap: from preclinical promise to clinical application
Although considerable preclinical evidence supports the anti-inflammatory and immunomodulatory potential of phytochemicals in RA, translation into clinical practice remains limited [3]. Few plant-derived compounds have progressed to clinical evaluation, and current studies have indicated both their promise and persistent challenges. For example, curcumin from Curcuma longa has been tested in randomized controlled trials and demonstrated modest improvements in joint pain, swelling, and inflammatory biomarkers when used as an adjunct to methotrexate [217, 218]. In addition, sinomenine, an alkaloid from Sinomenium acutum, is clinically used in China for inflammatory disorders, in which it shows DMARD-like efficacy with a satisfactory safety profile [90, 91]. Paeoniflorin, the primary glycoside of Paeonia lactiflora, has also entered early-phase clinical trials, which have indicated decreases in TNF-α and IL-6 levels comparable to those achieved by low-dose methotrexate; this treatment therefore has potential as a safer adjunctive therapy (Table 3). However, the lack of large-scale, multicenter trials using standardized formulations limits definitive conclusions regarding clinical efficacy from being drawn [158, 159].
Table 3 Clinical Trial Landscape of Phytochemicals with High Potential in RA Treatment
| Phytochemical/Plant Source | Dose | Trial Phase/Design | Sample Size | Key Outcomes | References |
|---|---|---|---|---|---|
| Sinomenine (Sinomenium acutum) | 60–180 mg/day oral | Post-marketing clinical application (China) | n>2500 (registry) | Comparable to low-dose DMARDs; ↓ IL-6/TNF-α; mild GI effects | [90, 91] |
| Paeoniflorin (Paeonia lactiflora) | 120–240 mg/day oral | Phase II; multicenter | n=120 | ↓ TNF-α/IL-6; improved joint mobility; superior safety vs. MTX | [158, 159] |
| Resveratrol (Polygonum cuspidatum) | 1 g/day oral | Phase I/II exploratory | n=30 | ↓ Inflammatory cytokines but limited functional improvement | Exploratory |
| Boswellic acids (Boswellia serrata) | 300–500 mg/day oral | Phase II pilot | n=39 | ↓ Pain and swelling, improved functional scores; well tolerated | Pilot |
A major barrier to translation is poor bioavailability and pharmacokinetic inconsistency. Numerous phytochemicals, including curcumin, quercetin, and resveratrol, have low water solubility, rapid metabolism, and partial systemic absorption [160]. Advanced formulation methods, such as nanoparticles, liposomes, phytosomes, and polymeric micelles, have increased stability and tissue targeting in preclinical studies but require rigorous human validation. In addition, plant extracts are inherently complex mixtures, and a deficiency in standardization in active compound content complicates regulatory approval and reproducibility [161].
An evolving strategy to improve therapeutic outcomes involves synergistic combinations of phytochemicals with conventional DMARDs or biologics [162]. Limited preclinical clinical data suggest that compounds such as curcumin, quercetin, and resveratrol can potentiate the anti-inflammatory effects of methotrexate or TNF-α inhibitors, while mitigating hepatotoxicity and oxidative stress. Such integrative therapeutic regimens have potential to decrease drug resistance, decrease the required doses of synthetic DMARDs, and improve patient tolerability [163].
Key challenges in clinical translation of phytochemicals for RA
Despite promising preclinical outcomes, the clinical advancement of phytochemicals for RA expression faces critical translational barriers that limit their therapeutic use [164]. Major challenges include poor bioavailability and metabolic instability. Many plant-derived compounds, such as curcumin, quercetin, and resveratrol, exhibit low aqueous solubility, poor intestinal absorption, and rapid metabolic degradation, thus leading to insufficient systemic concentrations. Their limited pharmacokinetic profiles often require high oral doses, which might limit compliance and safety. Approaches such as nanoformulation, liposomal encapsulation, and complexation with phospholipids have shown potential to improve absorption and target tissue distribution, yet these technologies require further optimization and validation in clinical settings [165].
Another major problem is the lack of standardization in herbal extracts. Variability in plant species, geographical origins, harvesting seasons, and extraction methods results in varying phytochemical content and biological potency. This heterogeneity hinders reproducibility across studies and complicates dose-response assessments. The use of standardized excerpts, validated reference markers, and Good Manufacturing Practice compliant processing is important in confirming quality and reproducibility [166].
Toxicological and safety concerns also persist but have not been effectively identified. Although phytochemicals are often perceived as naturally safe, several classes including alkaloids and terpenoids can exhibit hepatotoxicity, nephrotoxicity, or immunosuppressive effects at high doses or with prolonged use [167]. Comprehensive toxicity profiling, including acute, sub-chronic, and genotoxic studies, is therefore essential before clinical translation. Moreover, potential herb-drug interactions, primarily with conventional DMARDs or biologics, must be thoroughly evaluated to avoid adverse pharmacodynamics or pharmacokinetic effects [168].
Regulatory challenges continue to hinder clinical development. Differences in classification frameworks, in which herbal products might be considered dietary supplements, traditional medicines, or pharmaceuticals, create contradictions in consent pathways and quality control requirements. Establishing harmonized international standards and integrating pharmacovigilance systems for herbal formulations would aid in confirmation of both efficacy and safety [167, 169].
Comparison of high-potential phytochemicals in RA therapy
Although numerous phytochemicals exhibit anti-arthritic properties, not all demonstrate equivalent mechanistic robustness or translational promise. To provide a more focused perspective, a subset of high-potential phytochemicals has been prioritized according to three key parameters: (i) mechanistic diversity in modulating multiple signaling cascades relevant to RA (e.g., NF-κB, MAPK, JAK/STAT, and Nrf2/HO-1); (ii) experimental demonstration of consistent efficacy across in vitro and in vivo models; and (iii) translational relevance according to clinical or pharmacokinetic studies.
Among flavonoids, quercetin and kaempferol are notable for their dual modulation of inflammatory and oxidative pathways, coupled with reproducible in vivo efficacy in collagen- and adjuvant-induced arthritis models [3]. Curcumin, a polyphenolic compound from Curcuma longa, the most extensively studied natural agent, exhibits broad inhibition of NF-κB and JAK/STAT signaling, strong antioxidant activity via Nrf2 activation, and confirmed clinical benefits as an adjunct to methotrexate [217, 218]. Similarly, sinomenine, an isoquinoline alkaloid, has advanced beyond preclinical testing to clinical use in China, and has demonstrated significant immunosuppressive effects and symptom alleviation in patients with RA [90]. Boswellic acid and triptolide, both terpenoids, are supported by rigorous mechanistic data and ongoing clinical evaluation, and therefore are particularly promising for future drug development. Paeoniflorin, a monoterpene glycoside, also exhibits strong translational value: phase II clinical trials have reported decreases in inflammatory cytokines and improved safety compared with synthetic DMARDs [155].
Translational challenges
Although extensive preclinical research supports the anti-inflammatory and antioxidant potential of phytochemicals in RA, their effective translation into clinical application remains limited. Numerous critical challenges hinder the progression of these promising agents from bench to bedside.
Bioavailability and pharmacokinetics
Several phytochemicals have poor oral bioavailability, because of low aqueous solubility, rapid metabolism, and limited absorption across biological membranes [170]. For example, curcumin, resveratrol, and quercetin display effective in vitro efficacy but exhibit extremely low plasma concentrations after oral administration [171]. Extensive first-pass metabolism and rapid systemic clearance further decrease their therapeutic potential. Advanced formulation approaches such as nanoparticle encapsulation, liposomes, phytosomes, and polymeric micelles have been used to enhance stability, permeability, and sustained release, but their clinical validation remains preliminary [172].
Metabolic instability and standardization issues
Plant-derived preparations often contain multiple bioactive components whose concentrations can vary according to species, geographical source, extraction method, and storage conditions [173]. This lack of standardization introduces variability in pharmacological outcomes and complicates reproducibility. Moreover, the metabolism of phytochemicals can generate active or inactive metabolites, thereby altering efficacy and safety profiles. Establishing standardized extracts and validated analytical markers is therefore essential for consistent dosing and clinical comparability [174].
Safety and toxicological evaluation
Although phytochemicals are often perceived as safe because of their natural origin, long-term safety data remain scarce. Some compounds, including alkaloids and terpenoids, might exhibit hepatotoxic, nephrotoxic, or immunosuppressive effects at high doses or prolonged exposure. Rigorous toxicological evaluation including acute, sub-chronic, and chronic studies is necessary to define safety margins and avoid adverse interactions with standard DMARDs [3, 175].
Translational and regulatory barriers
Few phytochemicals used in preclinical RA studies have advanced to well-designed clinical trials. Regulatory frameworks for herbal medicines vary widely among countries, thus posing challenges in defining them as dietary supplements or pharmacological agents. Moreover, robust randomized controlled trials using standardized preparations, adequate sample sizes, and mechanistic biomarkers are lacking. Integrating pharmacokinetic and pharmacodynamic modeling early in the development process could accelerate the translation of promising compounds [176, 177].
Future directions
Bridging existing gaps will require multidisciplinary collaboration among pharmacologists, formulation scientists, and clinicians. Nanocarrier-based delivery systems, synergistic combinations with DMARDs, and systematic toxicological valuations may help overcome bioavailability and safety challenges. Ultimately, well-designed clinical investigations will be crucial to demonstrate preclinical efficacy and validate phytochemicals as reliable adjuncts or alternatives in RA therapy [178, 179].
Conclusion
This review highlights the notable preclinical potential of phytochemicals as modulators of inflammatory and oxidative pathways involved in RA, including their consistent anti-inflammatory, antioxidant, and immunomodulatory effects in targeting key molecular markers such as NF-κB, MAPK, and JAK/STAT. To translate these promising experimental findings into clinical success, future research should focus on integrating phytochemical discovery using advanced nanocarrier delivery systems to enhance bioavailability and targeted delivery. In addition, exploring combination therapies exploiting synergy between phytochemicals and conventional DMARDs could optimize therapeutic efficacy while potentially avoiding adverse effects. Addressing current limitations, such as heterogeneity in preclinical models, deficiencies in standardized protocols, and inadequate long-term toxicity and pharmacokinetic data, through well-designed, well-controlled clinical trials will be critical to establishing the true therapeutic value and safety of phytochemicals in RA management.
Abbreviations
| RA | Rheumatoid arthritis |
| DMARDs | Disease-modifying antirheumatic drugs |
| NSAIDs | Nonsteroidal anti-inflammatory drugs |
| IL | Interleukin |
| TNF | Tumor necrosis factor |
| MMP | Matrix metalloproteinase |
| NF-κB | Nuclear factor kappa B |
| MAPK | Mitogen-activated protein kinase |
| COX-2 | Cyclooxygenase-2 |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| HO-1 | Heme oxygenase-1 |
| FLSs | Fibroblast-like synoviocytes |
| CIA | Collagen-induced arthritis |
| LPS | Lipopolysaccharide |
| JAK | Janus kinase |
| STAT | Signal transducer and activator of transcription |
| NLRP3 | NOD-, LRR- and pyrin domain-containing protein 3 |
| TLR | Toll-like receptor |
| ACPA | Anti-citrullinated protein antibody |
| RF | Rheumatoid factor |
| PAD | Peptidylarginine deiminase |
| MHC | Major histocompatibility complex |
| DC | Dendritic cell |
| Th | T helper |
| Treg | Regulatory T |
| Tfh | T follicular helper |
| RANKL | Receptor activator of nuclear factor kappa-B ligand |
| AIA | Adjuvant-induced arthritis |
| CFA | Complete Freund’s adjuvant |
Conflict of interest
The authors declare that there are no conflicts of interest.
Funding
No funding or sponsorship was received for this study.
Acknowledgments
We sincerely thank everyone who provided assistance in completing this research. We acknowledge the use of online tools (BioRender) for creating the schematic figures included in this article.
Graphical abstract
Highlights
- Phytochemicals such as flavonoids, alkaloids, and terpenoids target key inflammatory pathways (NF-κB, MAPK, and JAK/STAT) and cytokines (TNF-α and IL-6) to exert anti-inflammatory effects in rheumatoid arthritis models.
- These compounds have synergistic and multi-targeting properties in that they act on both inflammatory and oxidative stress pathways.
- Lack of standardization, poor bioavailability, and insufficient clinical information are some of the key issues involved in clinical translation.
- Promising strategies to expand efficacy involve nanoformulations, combination therapies with DMARDs, and well-designed clinical trials with standardized preparations and pharmacokinetic valuations.
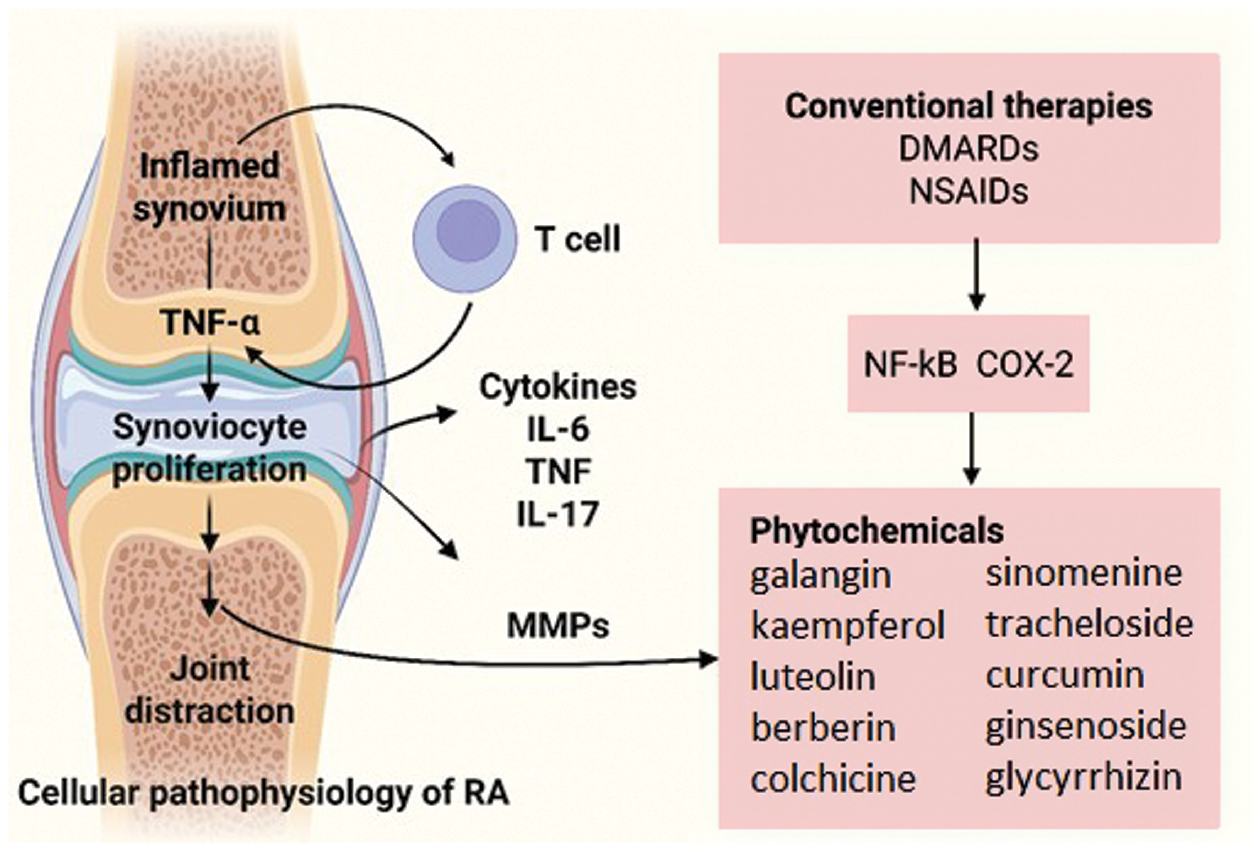
In brief
Graphical representation of cellular pathophysiology of rheumatoid arthritis, highlighting therapeutic intervention points for both conventional therapy and medicinal plant-derived phytochemicals (figure created with BioRender.com).
References
- Luo J, Zhu Y, Yu Y, Chen Y, He K, et al. Sinomenine treats rheumatoid arthritis by inhibiting MMP9 and inflammatory cytokines expression: bioinformatics analysis and experimental validation. Sci Rep 2024;14(1):12786. [PMID: 38834626 DOI: 10.1038/s41598-024-61769-x]
- Wu D, Luo Y, Li T, Zhao X, Lv T, et al. Systemic complications of rheumatoid arthritis: focus on pathogenesis and treatment. Front Immunol 2022;13:1051082. [PMID: 36618407 DOI: 10.3389/fimmu.2022.1051082]
- Kciuk M, Garg A, Rohilla M, Chaudhary R, Dhankhar S, et al. Therapeutic potential of plant-derived compounds and plant extracts in rheumatoid arthritis-comprehensive review. Antioxidants (Basel) 2024;13(7):775. [PMID: 39061843 DOI: 10.3390/antiox13070775]
- van Delft MAM, Huizinga TWJ. An overview of autoantibodies in rheumatoid arthritis. J Autoimmun 2020;110:102392. [PMID: 31911013 DOI: 10.1016/j.jaut.2019.102392]
- Ben Mrid R, Bouchmaa N, Ainani H, El Fatimy R, Malka G, et al. Anti-rheumatoid drugs advancements: new insights into the molecular treatment of rheumatoid arthritis. Biomed Pharmacother 2022;151:113126. [PMID: 35643074 DOI: 10.1016/j.biopha.2022.113126]
- Shams S, Martinez JM, Dawson JRD, Flores J, Gabriel M, et al. The therapeutic landscape of rheumatoid arthritis: current state and future directions. Front Pharmacol 2021;12:680043. [PMID: 34122106 DOI: 10.3389/fphar.2021.680043]
- Nalwa HS, Prasad P, Ganguly NK, Chaturvedi V, Mittal SA. Methotrexate intolerance in rheumatoid arthritis. Transl Med Commun 2023;8(1):10. [DOI: 10.1186/s41231-023-00142-y]
- Handa R. Rheumatoid arthritis. In: Clinical Rheumatology. Singapore: Springer; 2021. pp. 51-65 . [DOI: 10.1007/978-981-33-4885-1_6]
- Kumar A, Nirmal P, Kumar M, Jose A, Tomer V, et al. Major phytochemicals: recent advances in health benefits and extraction method. Molecules 2023;28(2):887. [PMID: 36677944 DOI: 10.3390/molecules28020887]
- Riaz M, Khalid R, Afzal M, Anjum F, Fatima H, et al. Phytobioactive compounds as therapeutic agents for human diseases: a review. Food Sci Nutr 2023;11(6):2500-29. [PMID: 37324906 DOI: 10.1002/fsn3.3308]
- Yu M, Gouvinhas I, Rocha J, Barros AIRNA. Phytochemical and antioxidant analysis of medicinal and food plants towards bioactive food and pharmaceutical resources. Sci Rep 2021;11(1):10041. [PMID: 33976317 DOI: 10.1038/s41598-021-89437-4]
- Kiani HS, Ali A, Zahra S, Hassan ZU, Kubra KT, et al. Phytochemical composition and pharmacological potential of lemongrass (cymbopogon) and impact on gut microbiota. Appl Chem 2022;2(4):229-46. [DOI: 10.3390/appliedchem2040016]
- Ginwala R, Bhavsar R, Chigbu DI, Jain P, Khan ZK. Potential role of flavonoids in treating chronic inflammatory diseases with a special focus on the anti-inflammatory activity of apigenin. Antioxidants (Basel) 2019;8(2):35. [PMID: 30764536 DOI: 10.3390/antiox8020035]
- Nisar A, Jagtap S, Vyavahare S, Deshpande M, Harsulkar A, et al. Phytochemicals in the treatment of inflammation-associated diseases: the journey from preclinical trials to clinical practice. Front Pharmacol 2023;14:1177050. [PMID: 37229273 DOI: 10.3389/fphar.2023.1177050]
- Aravilli RK, Vikram SL, Kohila V. Phytochemicals as potential antidotes for targeting NF-κB in rheumatoid arthritis. 3 Biotech 2017;7(4):253. [PMID: 28721679 DOI: 10.1007/s13205-017-0888-1]
- Mao H, Zhao X, Sun SC. NF-κB in inflammation and cancer. Cell Mol Immunol 2025;22(8):811-39. [PMID: 40562870 DOI: 10.1038/s41423-025-01310-w]
- Zhang J, Zhang M, Huo XK, Ning J, Yu ZL, et al. Macrophage inactivation by small molecule wedelolactone via targeting sEH for the treatment of LPS-induced acute lung injury. ACS Cent Sci 2023;9(3):440-56. [PMID: 36968547 DOI: 10.1021/acscentsci.2c01424]
- Tan SYX, Zhang J, Tee WW. Epigenetic regulation of inflammatory signaling and inflammation-induced cancer. Front Cell Dev Biol 2022;10:931493. [PMID: 35757000 DOI: 10.3389/fcell.2022.931493]
- Ooi SL, Pak SC. Nutraceuticals in immune function. Molecules 2021;26(17):5310. [PMID: 34500743 DOI: 10.3390/molecules26175310]
- Youssef AA, Baron DN. Leucocyte superoxide dismutase in rheumatoid arthritis. Ann Rheum Dis 1983;42(5):558-62. [PMID: 6625704 DOI: 10.1136/ard.42.5.558]
- Noto G, Salvetti F, Demaj A, Altieri P, Chericoni E, et al. Pathogenesis of rheumatoid arthritis: one year in review 2025. Clin Exp Rheumatol 2025;43(9):1533-40. [PMID: 40977292 DOI: 10.55563/clinexprheumatol/53rkan]
- Aswad M, Pechkovsky A, Ghanayiem N, Hamza H, Louria-Hayon I. High CBD extract (CBD-X) modulates inflammation and immune cell activity in rheumatoid arthritis. Front Immunol 2025;16:1599109. [PMID: 40709173 DOI: 10.3389/fimmu.2025.1599109]
- Lv J, Hao P, Zhou Y, Liu T, Wang L, et al. Role of the intestinal flora-immunity axis in the pathogenesis of rheumatoid arthritis-mechanisms regulating short-chain fatty acids and Th17/Treg homeostasis. Mol Biol Rep 2025;52(1):617. [PMID: 40544212 DOI: 10.1007/s11033-025-10714-w]
- Marcucci E, Bartoloni E, Alunno A, Leone MC, Cafaro G, et al. Extra-articular rheumatoid arthritis. Reumatismo 2018;70(4):212-24. [PMID: 30570239 DOI: 10.4081/reumatismo.2018.1106]
- Kasiraja V, Bakar NAA, Suliman NA. Rheumatoid arthritis unmasked: the immune complex as a key driver of disease progression. Explor Immunol 2025;5:1003208. [DOI: 10.37349/ei.2025.1003208]
- Scherer HU, Häupl T, Burmester GR. The etiology of rheumatoid arthritis. J Autoimmun 2020;110:102400. [PMID: 31980337 DOI: 10.1016/j.jaut.2019.102400]
- Degboé Y. Pre-rheumatoid arthritis and ACPA: contribution of ACPAs in the pathogeny of pre-disease stage. Joint Bone Spine 2021;88(3):105098. [PMID: 33157231 DOI: 10.1016/j.jbspin.2020.105098]
- Pertsinidou E, Manivel VA, Westerlind H, Klareskog L, Alfredsson L, et al. Rheumatoid arthritis autoantibodies and their association with age and sex. Clin Exp Rheumatol 2021;39(4):879-82. [PMID: 33822709 DOI: 10.55563/clinexprheumatol/4bcmdb]
- Ding Q, Hu W, Wang R, Yang Q, Zhu M, et al. Signaling pathways in rheumatoid arthritis: implications for targeted therapy. Signal Transduct Target Ther 2023;8(1):68. [PMID: 36797236 DOI: 10.1038/s41392-023-01331-9]
- De Stefano L, D’Onofrio B, Manzo A, Montecucco C, Bugatti S. The genetic, environmental, and immunopathological complexity of autoantibody-negative rheumatoid arthritis. Int J Mol Sci 2021;22(22):12386. [PMID: 34830268 DOI: 10.3390/ijms222212386]
- Pérez-Pérez ME, Nieto-Torres E, Bollain-Y-Goytia JJ, Delgadillo-Ruíz L. Protein citrullination by peptidyl arginine deiminase/arginine deiminase homologs in members of the human microbiota and its recognition by anti-citrullinated protein antibodies. Int J Mol Sci 2024;25(10):5192. [PMID: 38791230 DOI: 10.3390/ijms25105192]
- Chen Y, Teng Y, Xu P, Wang S. The role of citrullination modification in CD4+ T cells in the pathogenesis of immune-related diseases. Biomolecules 2024;14(4):400. [PMID: 38672418 DOI: 10.3390/biom14040400]
- Massarenti L, Enevold C, Damgaard D, Ødum N, Garred P, et al. PADI4 polymorphisms confer risk of anti-CCP-positive rheumatoid arthritis in synergy with HLA-DRB1*04 and smoking. Front Immunol 2021;12:707690. [PMID: 34733271 DOI: 10.3389/fimmu.2021.707690]
- Kurowska W, Kuca-Warnawin EH, Radzikowska A, Maśliński W. The role of anti-citrullinated protein antibodies (ACPA) in the pathogenesis of rheumatoid arthritis. Cent Eur J Immunol 2017;42(4):390-8. [PMID: 29472818 DOI: 10.5114/ceji.2017.72807]
- Petrovská N, Prajzlerová K, Vencovský J, Šenolt L, Filková M. The pre-clinical phase of rheumatoid arthritis: from risk factors to prevention of arthritis. Autoimmun Rev 2021;20(5):102797. [PMID: 33746022 DOI: 10.1016/j.autrev.2021.102797]
- Wu CY, Yang HY, Luo SF, Lai JH . From rheumatoid factor to anti-citrullinated protein antibodies and anti-carbamylated protein antibodies for diagnosis and prognosis prediction in patients with rheumatoid arthritis. Int J Mol Sci 2021;22(2):686. [PMID: 33445768 DOI: 10.3390/ijms22020686]
- Ganeshalingam S, Wilson NJ, Ciciriello S, Antonipillai J, Achuthan AA. Cellular interactions in maintaining an inflammatory microenvironment in rheumatoid arthritis. Mol Immunol 2025;184:112-22. [PMID: 40561673 DOI: 10.1016/j.molimm.2025.06.008]
- Wehr P, Purvis H, Law SC, Thomas R. Dendritic cells, T cells and their interaction in rheumatoid arthritis. Clin Exp Immunol 2019;196(1):12-27. [PMID: 30589082 DOI: 10.1111/cei.13256]
- Okamoto K. Crosstalk between bone and the immune system. J Bone Miner Metab 2024;42(4):470-80. [PMID: 39060500 DOI: 10.1007/s00774-024-01539-x]
- Everts PA, Lana JF, Onishi K, Buford D, Peng J, et al. Angiogenesis and tissue repair depend on platelet dosing and bioformulation strategies following orthobiological platelet-rich plasma procedures: a narrative review. Biomedicines 2023;11(7):1922. [PMID: 37509560 DOI: 10.3390/biomedicines11071922]
- Tsaltskan V, Firestein GS. Targeting fibroblast-like synoviocytes in rheumatoid arthritis. Curr Opin Pharmacol 2022;67:102304. [PMID: 36228471 DOI: 10.1016/j.coph.2022.102304]
- Mousavi MJ, Karami J, Aslani S, Tahmasebi MN, Vaziri AS, et al. Transformation of fibroblast-like synoviocytes in rheumatoid arthritis; from a friend to foe. Auto Immun Highlights 2021;12(1):3. [PMID: 33546769 DOI: 10.1186/s13317-020-00145-x]
- Hu Z, Li Y, Zhang L, Jiang Y, Long C, et al. Metabolic changes in fibroblast-like synoviocytes in rheumatoid arthritis: state of the art review. Front Immunol 2024;15:1250884. [PMID: 38482018 DOI: 10.3389/fimmu.2024.1250884]
- Jang S, Kwon EJ, Lee JJ. Rheumatoid arthritis: pathogenic roles of diverse immune cells. Int J Mol Sci 2022;23(2):905. [PMID: 35055087 DOI: 10.3390/ijms23020905]
- Barnhoorn MC, Hakuno SK, Bruckner RS, Rogler G, Hawinkels LJAC, et al. Stromal cells in the pathogenesis of inflammatory bowel disease. J Crohns Colitis 2020;14(7):995-1009. [PMID: 32160284 DOI: 10.1093/ecco-jcc/jjaa009]
- Honan AM, Chen Z. Stromal cells underlining the paths from autoimmunity, inflammation to cancer with roles beyond structural and nutritional support. Front Cell Dev Biol 2021;9:658984. [PMID: 34113615 DOI: 10.3389/fcell.2021.658984]
- Nygaard G, Firestein GS. Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat Rev Rheumatol 2020;16(6):316-33. [PMID: 32393826 DOI: 10.1038/s41584-020-0413-5]
- Bustamante MF, Garcia-Carbonell R, Whisenant KD, Guma M. Fibroblast-like synoviocyte metabolism in the pathogenesis of rheumatoid arthritis. Arthritis Res Ther 2017;19(1):110. [PMID: 28569176 DOI: 10.1186/s13075-017-1303-3]
- Chen S, Saeed AFUH, Liu Q, Jiang Q, Xu H, et al. Macrophages in immunoregulation and therapeutics. Signal Transduct Target Ther 2023;8(1):207. [PMID: 37211559 DOI: 10.1038/s41392-023-01452-1]
- Abdul-Rahman T, Ghosh S, Badar SM, Nazir A, Bamigbade GB, et al. The paradoxical role of cytokines and chemokines at the tumor microenvironment: a comprehensive review. Eur J Med Res 2024;29(1):124. [PMID: 38360737 DOI: 10.1186/s40001-024-01711-z]
- Ahmadivand S, Fux R, Palić D. Role of T follicular helper cells in viral infections and vaccine design. Cells 2025;14(7):508. [PMID: 40214462 DOI: 10.3390/cells14070508]
- Lu J, Wu J, Xia X, Peng H, Wang S. Follicular helper T cells: potential therapeutic targets in rheumatoid arthritis. Cell Mol Life Sci 2021;78(12):5095-106. [PMID: 33880615 DOI: 10.1007/s00018-021-03839-1]
- Christodoulou M, Moysidou E, Lioulios G, Stai S, Lazarou C, et al. T-follicular helper cells and their role in autoimmune diseases. Life (Basel) 2025;15(4):666. [PMID: 40283219 DOI: 10.3390/life15040666]
- Guo Q, Wang Y, Xu D, Nossent J, Pavlos NJ, et al. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res 2018;6(1):15. [PMID: 29736302 DOI: 10.1038/s41413-018-0016-9]
- Kusiak A, Brady G. Bifurcation of signalling in human innate immune pathways to NF-kB and IRF family activation. Biochem Pharmacol 2022;205:115246. [PMID: 36088989 DOI: 10.1016/j.bcp.2022.115246]
- Zhao Z, Hua Z, Luo X, Li Y, Yu L, et al. Application and pharmacological mechanism of methotrexate in rheumatoid arthritis. Biomed Pharmacother 2022;150:113074. [PMID: 35658215 DOI: 10.1016/j.biopha.2022.113074]
- Jang DI, Lee AH, Shin HY, Song HR, Park JH, et al. The role of tumor necrosis factor alpha (TNF-α) in autoimmune disease and current TNF-α inhibitors in therapeutics. Int J Mol Sci 2021;22(5):2719. [PMID: 33800290 DOI: 10.3390/ijms22052719]
- Yip RML, Yim CW. Role of interleukin 6 inhibitors in the management of rheumatoid arthritis. J Clin Rheumatol 2021;27(8):e516-24. [PMID: 31876844 DOI: 10.1097/RHU.0000000000001293]
- Wang K, Luo H, Liu L, Gao H, Song Y, et al. Blockade of IL-1 family cytokines in the treatment of rheumatoid arthritis. Front Pharmacol 2025;16:1577628. [PMID: 40520203 DOI: 10.3389/fphar.2025.1577628]
- Liang Y, Zha M, Liu Q, Lai Z, Li L, et al. Rheumatoid arthritis therapy based on B cells. Drug Des Devel Ther 2025;19:7837-52. [PMID: 40951695 DOI: 10.2147/DDDT.S527687]
- Mostkowska A, Rousseau G, Raynal NJM. Repurposing of rituximab biosimilars to treat B cell mediated autoimmune diseases. FASEB J 2024;38(5):e23536. [PMID: 38470360 DOI: 10.1096/fj.202302259RR]
- Bastos LL, Mariano D, Lemos RP, Bialves TS, Oliveira CJF, et al. The role of structural bioinformatics in understanding tumor necrosis factor α-interacting protein mechanisms in chronic inflammatory diseases: a review. Immuno 2024;4(1):14-42. [DOI: 10.3390/immuno4010002]
- Kaye AD, Perilloux DM, Hawkins AM, Wester GC, Ragaland AR, et al. Tumor necrosis factor and interleukin modulators for pathologic pain states: a narrative review. Pain Ther 2024;13(3):481-93. [PMID: 38724743 DOI: 10.1007/s40122-024-00603-8]
- Hu L, Liu R, Zhang L. Advance in bone destruction participated by JAK/STAT in rheumatoid arthritis and therapeutic effect of JAK/STAT inhibitors. Int Immunopharmacol 2022;111:109095. [PMID: 35926270 DOI: 10.1016/j.intimp.2022.109095]
- Vallez-Valero L, Gasó-Gago I, Marcos-Fendian Á, Garrido-Alejos G, Riera-Magallón A, et al. Are all JAK inhibitors for the treatment of rheumatoid arthritis equivalent? An adjusted indirect comparison of the efficacy of tofacitinib, baricitinib, upadacitinib, and filgotinib. Clin Rheumatol 2023;42(12):3225-35. [PMID: 37831336 DOI: 10.1007/s10067-023-06787-2]
- Favalli EG, Maioli G, Caporali R. Biologics or Janus kinase inhibitors in rheumatoid arthritis patients who are insufficient responders to conventional anti-rheumatic drugs. Drugs 2024;84(8):877-94. [PMID: 38949688 DOI: 10.1007/s40265-024-02059-8]
- Singh N, Gusain A, Nigam M, Mishra AP. The pharmacological and therapeutic versatility of Allium species: a comprehensive exploration of bioactive constituents and biological activities. Discov ApplSci 2025;7(4):1-29. [DOI: 10.1007/s42452-025-06800-0]
- Xue C, Yao Q, Gu X, Shi Q, Yuan X, et al. Evolving cognition of the JAK-STAT signaling pathway: autoimmune disorders and cancer. Signal Transduct Target Ther 2023;8(1):204. [PMID: 37208335 DOI: 10.1038/s41392-023-01468-7]
- Radu AF, Bungau SG. Management of rheumatoid arthritis: an overview. Cells 2021;10(11):2857. [PMID: 34831081 DOI: 10.3390/cells10112857]
- Hsieh TS, Tsai TF . Combination therapy for psoriasis with methotrexate and other oral disease-modifying antirheumatic drugs: a systematic review. Dermatol Ther (Heidelb) 2023;13(4):891-909. [PMID: 36943580 DOI: 10.1007/s13555-023-00903-5]
- De Souza S, Bansal RK, Galloway J. Managing patients with rheumatoid arthritis. BDJ Team 2017;4(4):17064. [DOI: 10.1038/bdjteam.2017.64]
- Pizano-Martinez O, Mendieta-Condado E, Vázquez-Del Mercado M, Martínez-García EA, Chavarria-Avila E, et al. Anti-drug antibodies in the biological therapy of autoimmune rheumatic diseases. J Clin Med 2023;12(9):3271. [PMID: 37176711 DOI: 10.3390/jcm12093271]
- Laurent MR, Goemaere S, Verroken C, Bergmann P, Body JJ, et al. Prevention and treatment of glucocorticoid-induced osteoporosis in adults: consensus recommendations from the belgian bone club. Front Endocrinol (Lausanne) 2022;13:908727. [PMID: 35757436 DOI: 10.3389/fendo.2022.908727]
- Auriel E, Regev K, Korczyn AD. Nonsteroidal anti-inflammatory drugs exposure and the central nervous system. Handb Clin Neurol 2014;119:577-84. [PMID: 24365321 DOI: 10.1016/B978-0-7020-4086-3.00038-2]
- Jung JY, Lee E, Kim JW, Suh CH, Shin K, et al. Unveiling difficult-to-treat rheumatoid arthritis: long-term impact of biologic or targeted synthetic DMARDs from the KOBIO registry. Arthritis Res Ther 2023;25(1):174. [PMID: 37726808 DOI: 10.1186/s13075-023-03165-w]
- Tornero Molina J, Hernández-Cruz B, Corominas H. Initial treatment with biological therapy in rheumatoid arthritis. J Clin Med 2023;13(1):48. [PMID: 38202055 DOI: 10.3390/jcm13010048]
- Ibrahim F, Scott IC, Scott DL, Ayis SA. Heterogeneity of treatment responses in rheumatoid arthritis using group based trajectory models: secondary analysis of clinical trial data. BMC Rheumatol 2023;7(1):33. [PMID: 37749588 DOI: 10.1186/s41927-023-00348-5]
- Mucke J, Simon HU, Rüdiger Burmester G. The safety of antirheumatic drugs. Dtsch Arztebl Int 2022;119(6):81-7. [DOI: 10.3238/arztebl.m2022.0064]
- Mahajan TD, Mikuls TR. Recent advances in the treatment of rheumatoid arthritis. Curr Opin Rheumatol 2018;30(3):231-7. [PMID: 29461286 DOI: 10.1097/BOR.0000000000000496]
- Al-Khayri JM, Sahana GR, Nagella P, Joseph BV, Alessa FM, et al. Flavonoids as potential anti-inflammatory molecules: a review. Molecules 2022;27(9):2901. [PMID: 35566252 DOI: 10.3390/molecules27092901]
- Khawaja G, El-Orfali Y, Shoujaa A, Abou Najem S. Galangin: a promising flavonoid for the treatment of rheumatoid arthritis-mechanisms, evidence, and therapeutic potential. Pharmaceuticals (Basel) 2024;17(7):963. [PMID: 39065811 DOI: 10.3390/ph17070963]
- Białek W, Czechowicz P, Grzyb J, Sławski J, Collawn JF, et al. Plant flavonoid effects on the non-apoptotic mechanisms of regulated cell death. Phytochem Rev 2025. [DOI: 10.1007/s11101-025-10140-7]
- Alrumaihi F, Almatroodi SA, Alharbi HOA, Alwanian WM, Alharbi FA, et al. Pharmacological potential of kaempferol, a flavonoid in the management of pathogenesis via modulation of inflammation and other biological activities. Molecules 2024;29(9):2007. [PMID: 38731498 DOI: 10.3390/molecules29092007]
- Bellavite P. Neuroprotective potentials of flavonoids: experimental studies and mechanisms of action. Antioxidants (Basel) 2023;12(2):280. [PMID: 36829840 DOI: 10.3390/antiox12020280]
- Chagas MDSS, Behrens MD, Moragas-Tellis CJ, Penedo GXM, Silva AR, et al. Flavonols and flavones as potential anti-inflammatory, antioxidant, and antibacterial compounds. Oxid Med Cell Longev 2022;2022:9966750. [PMID: 36111166 DOI: 10.1155/2022/9966750]
- Ferraz CR, Carvalho TT, Manchope MF, Artero NA, Rasquel-Oliveira FS, et al. Therapeutic potential of flavonoids in pain and inflammation: mechanisms of action, pre-clinical and clinical data, and pharmaceutical development. Molecules 2020;25(3):762. [PMID: 32050623 DOI: 10.3390/molecules25030762]
- Allemailem KS, Almatroudi A, Alharbi HOA, AlSuhaymi N, Alsugoor MH, et al. Apigenin: a bioflavonoid with a promising role in disease prevention and treatment. Biomedicines 2024;12(6):1353. [PMID: 38927560 DOI: 10.3390/biomedicines12061353]
- Ferreira MU. Alkaloids in future drug discovery. Molecules 2022;27(4):1347. [PMID: 35209135 DOI: 10.3390/molecules27041347]
- Lin Y, Yi O, Hu M, Hu S, Su Z, et al. Multifunctional nanoparticles of sinomenine hydrochloride for treat-to-target therapy of rheumatoid arthritis via modulation of proinflammatory cytokines. J Control Release 2022;348:42-56. [PMID: 35569587 DOI: 10.1016/j.jconrel.2022.05.016]
- Zhao XX, Peng C, Zhang H, Qin LP. Sinomenium acutum: a review of chemistry, pharmacology, pharmacokinetics, and clinical use. Pharm Biol 2012;50(8):1053-61. [PMID: 22775422 DOI: 10.3109/13880209.2012.656847]
- Zhang C, Zhang S, Liao J, Gong Z, Chai X, et al. Towards better sinomenine-type drugs to treat rheumatoid arthritis: molecular mechanisms and structural modification. Molecules 2022;27(24):8645. [PMID: 36557779 DOI: 10.3390/molecules27248645]
- Ehteshamfar SM, Akhbari M, Afshari JT, Seyedi M, Nikfar B, et al. Anti-inflammatory and immune-modulatory impacts of berberine on activation of autoreactive T cells in autoimmune inflammation. J Cell Mol Med 2020;24(23):13573-88. [PMID: 33135395 DOI: 10.1111/jcmm.16049]
- Lv T, Zhang C, Hu L, Wang C, Li S, et al. Berberine in sepsis: effects, mechanisms, and therapeutic strategies. J Immunol Res 2023;2023:4452414. [PMID: 36741234 DOI: 10.1155/2023/4452414]
- Bang JS, Oh DH, Choi HM, Sur BJ, Lim SJ, et al. Anti-inflammatory and antiarthritic effects of piperine in human interleukin 1β-stimulated fibroblast-like synoviocytes and in rat arthritis models. Arthritis Res Ther 2009;11(2):R49. [PMID: 19327174 DOI: 10.1186/ar2662]
- Ma A, Yang Y, Wang Q, Wang Y, Wen J, et al. Anti-inflammatory effects of oxymatrine on rheumatoid arthritis in rats via regulating the imbalance between Treg and Th17 cells. Mol Med Rep 2017;15(6):3615-22. [PMID: 28440447 DOI: 10.3892/mmr.2017.6484]
- Pu J, Fang FF, Li XQ, Shu ZH, Jiang YP, et al. Matrine exerts a strong anti-arthritic effect on type II collagen-induced arthritis in rats by inhibiting inflammatory responses. Int J Mol Sci 2016;17(9):1410. [PMID: 27571073 DOI: 10.3390/ijms17091410]
- Chia EW, Grainger R, Harper JL. Colchicine suppresses neutrophil superoxide production in a murine model of gouty arthritis: a rationale for use of low-dose colchicine. Br J Pharmacol 2008;153(6):1288-95. [PMID: 18264123 DOI: 10.1038/bjp.2008.20]
- Masyita A, Mustika Sari R, Dwi Astuti A, Yasir B, Rahma Rumata N, et al. Terpenes and terpenoids as main bioactive compounds of essential oils, their roles in human health and potential application as natural food preservatives. Food Chem X 2022;13:100217. [PMID: 35498985 DOI: 10.1016/j.fochx.2022.100217]
- Behl T, Upadhyay T, Singh S, Chigurupati S, Alsubayiel AM, et al. Polyphenols targeting MAPK mediated oxidative stress and inflammation in rheumatoid arthritis. Molecules 2021;26(21):6570. [PMID: 34770980 DOI: 10.3390/molecules26216570]
- Yeshi K, Turpin G, Jamtsho T, Wangchuk P. Indigenous uses, phytochemical analysis, and anti-inflammatory properties of australian tropical medicinal plants. Molecules 2022;27(12):3849. [PMID: 35744969 DOI: 10.3390/molecules27123849]
- Mantiniotou M, Athanasiadis V, Kalompatsios D, Bozinou E, Lalas SI. Therapeutic capabilities of triterpenes and triterpenoids in immune and inflammatory processes: a review. Compounds 2025;5(1):2. [DOI: 10.3390/compounds5010002]
- Zhao J, Zhang F, Xiao X, Wu Z, Hu Q, et al. Tripterygium hypoglaucum (Lévl.) hutch and its main bioactive components: recent advances in pharmacological activity, pharmacokinetics and potential toxicity. Front Pharmacol 2021;12:715359. [PMID: 34887747 DOI: 10.3389/fphar.2021.715359]
- You L, Cha S, Kim MY, Cho JY. Ginsenosides are active ingredients in Panax ginseng with immunomodulatory properties from cellular to organismal levels. J Ginseng Res 2022;46(6):711-21. [PMID: 36312737 DOI: 10.1016/j.jgr.2021.12.007]
- Sun W, Shahrajabian MH. Therapeutic potential of phenolic compounds in medicinal plants-natural health products for human health. Molecules 2023;28(4):1845. [PMID: 36838831 DOI: 10.3390/molecules28041845]
- Peng Y, Ao M, Dong B, Jiang Y, Yu L, et al. Anti-inflammatory effects of curcumin in the inflammatory diseases: status, limitations and countermeasures. Drug Des Devel Ther 2021;15:4503-25. [PMID: 34754179 DOI: 10.2147/DDDT.S327378]
- Kunnumakkara AB, Hegde M, Parama D, Girisa S, Kumar A, et al. Role of turmeric and curcumin in prevention and treatment of chronic diseases: lessons learned from clinical trials. ACS Pharmacol Transl Sci 2023;6(4):447-518. [PMID: 37082752 DOI: 10.1021/acsptsci.2c00012]
- Riveiro-Naveira RR, Valcárcel-Ares MN, Almonte-Becerril M, Vaamonde-García C, Loureiro J, et al. Resveratrol lowers synovial hyperplasia, inflammatory markers and oxidative damage in an acute antigen-induced arthritis model. Rheumatology (Oxford) 2016;55(10):1889-900. [PMID: 27354682 DOI: 10.1093/rheumatology/kew255]
- Li L, Pan Z, Ning D, Fu Y. Rosmanol and carnosol synergistically alleviate rheumatoid arthritis through inhibiting TLR4/NF-κB/MAPK pathway. Molecules 2022;27(1):78. [PMID: 35011304 DOI: 10.3390/molecules27010078]
- Wu M, Yu Z, Li X, Zhang X, Wang S, et al. Paeonol for the treatment of atherosclerotic cardiovascular disease: a pharmacological and mechanistic overview. Front Cardiovasc Med 2021;8:690116. [PMID: 34368250 DOI: 10.3389/fcvm.2021.690116]
- Direito R, Rocha J, Sepodes B, Eduardo-Figueira M. Phenolic compounds impact on rheumatoid arthritis, inflammatory bowel disease and microbiota modulation. Pharmaceutics 2021;13(2):145. [PMID: 33499333 DOI: 10.3390/pharmaceutics13020145]
- Buhrmann C, Mobasheri A, Busch F, Aldinger C, Stahlmann R, et al. Curcumin modulates nuclear factor κB (NF-κB)-mediated inflammation in human tenocytes in vitro: role of the phosphatidylinositol 3-kinase/Akt pathway. J Biol Chem 2011;286(32):28556-66. [PMID: 21669872 DOI: 10.1074/jbc.M111.256180]
- Lacaille-Dubois MA. Bioactive saponins with cancer related and immunomodulatory activity: recent developments. Stud Nat Prod Chem 2005;32:209-46. [DOI: 10.1016/S1572-5995(05)80057-2]
- Tsutamoto S, Iwasaki Y, Shinohara A, Imamiya R, Samukawa K, et al. Triterpenoid saponin from Panax ginseng increases the sensitivity of methicillin-resistant Staphylococcus aureus to β-lactam and aminoglycoside antibiotics. Microbiol Spectr 2024;12(6):e0322723. [PMID: 38647286 DOI: 10.1128/spectrum.03227-23]
- Elshal M, Hazem SH. Escin suppresses immune cell infiltration and selectively modulates Nrf2/HO-1, TNF-α/JNK, and IL-22/STAT3 signaling pathways in concanavalin A-induced autoimmune hepatitis in mice. Inflammopharmacology 2022;30(6):2317-29. [PMID: 36063304 DOI: 10.1007/s10787-022-01058-z]
- Zheng Q, Wang T, Wang S, Chen Z, Jia X, et al. The anti-inflammatory effects of saponins from natural herbs. Pharmacol Ther 2025;269:108827. [PMID: 40015518 DOI: 10.1016/j.pharmthera.2025.108827]
- Smeriglio A, Barreca D, Bellocco E, Trombetta D. Proanthocyanidins and hydrolysable tannins: occurrence, dietary intake and pharmacological effects. Br J Pharmacol 2017;174(11):1244-62. [PMID: 27646690 DOI: 10.1111/bph.13630]
- Singh BN, Shankar S, Srivastava RK. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochem Pharmacol 2011;82(12):1807-21. [PMID: 21827739 DOI: 10.1016/j.bcp.2011.07.093]
- Piazza S, Fumagalli M, Martinelli G, Pozzoli C, Maranta N, et al. Hydrolyzable tannins in the management of Th1, Th2 and Th17 inflammatory-related diseases. Molecules 2022;27(21):7593. [PMID: 36364420 DOI: 10.3390/molecules27217593]
- Ozogul Y, Ucar Y, Tadesse EE, Rathod N, Kulawik P, et al. Tannins for food preservation and human health: a review of current knowledge. Appl Food Res 2025;5(1):100738. [DOI: 10.1016/j.afres.2025.100738]
- Koyama T, Murata J, Horikawa M, Satake H. Production of beneficial lignans in heterologous host plants. Front Plant Sci 2022;13:1026664. [PMID: 36330251 DOI: 10.3389/fpls.2022.1026664]
- Wang HF, Huang ZH, Zou W, Lai CH, Tan QG. Tracheloside, the main constituent of the total lignan extract from Trachelospermi Caulis, inhibited rheumatoid arthritis via IL-17/MAPK signaling pathway. Fitoterapia 2024;180:106311. [PMID: 39603466 DOI: 10.1016/j.fitote.2024.106311]
- Hadipour E, Emami SA, Tayarani-Najaran N, Tayarani-Najaran Z. Effects of sesame (Sesamum indicum L.) and bioactive compounds (sesamin and sesamolin) on inflammation and atherosclerosis: a review. Food Sci Nutr 2023;11(7):3729-57. [PMID: 37457142 DOI: 10.1002/fsn3.3407]
- Piekarz J, Picheta N, Pobideł J, Daniłowska K, Gil-Kulik P. Phytotherapy as an adjunct to the treatment of rheumatoid arthritis – a systematic review of clinical trials. Phytomedicine 2025;148:157285. [PMID: 41005054 DOI: 10.1016/j.phymed.2025.157285]
- Muheremu A, Aierxiding S, Gao J. Regulation of the innate immune response in rheumatoid arthritis. Front Immunol 2025;16:1545625. [PMID: 41256865 DOI: 10.3389/fimmu.2025.1545625]
- Sahin K, Pala R, Tuzcu M, Ozdemir O, Orhan C, et al. Curcumin prevents muscle damage by regulating NF-κB and Nrf2 pathways and improves performance: an in vivo model. J Inflamm Res 2016;9:147-54. [PMID: 27621662 DOI: 10.2147/JIR.S110873]
- Nakadate K, Ito N, Kawakami K, Yamazaki N. Anti-inflammatory actions of plant-derived compounds and prevention of chronic diseases: from molecular mechanisms to applications. Int J Mol Sci 2025;26(11):1-24. [PMID: 40508016 DOI: 10.3390/ijms26115206]
- Mubeen B, Hasnain A, Naqvi SAH, Hakim F, Naqvi SSH. Phytochemicals as multi-target therapeutic agents for oxidative stress-driven pathologies: mechanisms, synergies, and clinical prospects. Phyton-Int J Exp Bot 2025;94(7):1941-71. [DOI: 10.32604/phyton.2025.064056]
- Mishra SK, Pandey D, Nille OS, Rajmane SJ, Nille GC. Frankincense unveiled: phytochemistry, bioactivity, and future directions. Phytomed Plus 2025;5(4):100898. [DOI: 10.1016/j.phyplu.2025.100898]
- Naik RA, Rajpoot R, Koiri RK, Bhardwaj R, Aldairi AF, et al. Dietary supplementation and the role of phytochemicals against the Alzheimer’s disease: focus on polyphenolic compounds. J Prev Alzheimers Dis 2025;12(1):100004. [PMID: 39800464 DOI: 10.1016/j.tjpad.2024.100004]
- Srisittiratkul P, Limsuvan S, Pattanapholkornsakul S, Akarasereenont P. Optimizing herbal drug use through multivariable standardization and precision approaches. Phytomed Plus 2025;5(4):100907. [DOI: 10.1016/j.phyplu.2025.100907]
- Castelli FA, Rosati G, Moguet C, Fuentes C, Marrugo-Ramírez J, et al. Metabolomics for personalized medicine: the input of analytical chemistry from biomarker discovery to point-of-care tests. Anal Bioanal Chem 2021;414(2):759-89. [PMID: 34432105 DOI: 10.1007/s00216-021-03586-z]
- Klein-Junior LC, De Souza MR, Viaene J, Bresolin TMB, de Gasper AL, et al. Quality control of herbal medicines: from traditional techniques to state-of-the-art approaches. Planta Med 2021;87(12/13):964-88. [PMID: 34412146 DOI: 10.1055/a-1529-8339]
- Fleischhacker W, Schönfeld T. Pioneering ideas for the physical and chemical sciences. 1st ed. New York, NY: Springer; 1997 [DOI: 10.1007/978-1-4899-0268-9]
- Rodríguez-Negrete EV, Morales-González Á, Madrigal-Santillán EO, Sánchez-Reyes K, Álvarez-González I, et al. Phytochemicals and their usefulness in the maintenance of health. Plants (Basel) 2024;13(4):523. [PMID: 38498532 DOI: 10.3390/plants13040523]
- Huang W, Percie du Sert N, Vollert J, Rice ASC. General principles of preclinical study design. Handb Exp Pharmacol 2020;257:55-69. [PMID: 31707471 DOI: 10.1007/164_2019_277]
- Migita K, Komori A, Torigoshi T, Maeda Y, Izumi Y, et al. CP690,550 inhibits oncostatin M-induced JAK/STAT signaling pathway in rheumatoid synoviocytes. Arthritis Res Ther 2011;13(3):R72. [PMID: 21548952 DOI: 10.1186/ar3333]
- Bakrim S, Fessikh ME, Elhrech H, Omari NE, Amanullah M, et al. Targeting inflammation in cancer therapy: from mechanistic insights to emerging therapeutic approaches. J Transl Med 2025;23(1):588. [PMID: 40420174 DOI: 10.1186/s12967-025-06583-3]
- Fu Q, Gao Y, Zhao H, Wang Z, Wang J. Galangin protects human rheumatoid arthritis fibroblast-like synoviocytes via suppression of the NF-κB/NLRP3 pathway. Mol Med Rep 2018;18(4):3619-24. [PMID: 30152847 DOI: 10.3892/mmr.2018.9422]
- Franco-Trepat E, Alonso-Pérez A, Guillán-Fresco M, López-Fagúndez M, Pazos-Pérez A, et al. β Boswellic acid blocks articular innate immune responses: an in silico and in vitro approach to traditional medicine. Antioxidants (Basel) 2023;12(2):371. [PMID: 36829930 DOI: 10.3390/antiox12020371]
- Schett G, Zwerina J, Firestein G. The p38 mitogen-activated protein kinase (MAPK) pathway in rheumatoid arthritis. Ann Rheum Dis 2008;67(7):909-16. [PMID: 17827184 DOI: 10.1136/ard.2007.074278]
- Pal RR, Rajpal V, Singh P, Saraf SA. Recent findings on thymoquinone and its applications as a nanocarrier for the treatment of cancer and rheumatoid arthritis. Pharmaceutics 2021;13(6):775. [PMID: 34067322 DOI: 10.3390/pharmaceutics13060775]
- Majrashi TA, Alshehri SA, Alsayari A, Muhsinah AB, Alrouji M, et al. Insight into the biological roles and mechanisms of phytochemicals in different types of cancer: targeting cancer therapeutics. Nutrients 2023;15(7):1704. [PMID: 37049544 DOI: 10.3390/nu15071704]
- Herrera TES, Tello IPS, Mustafa MA, Jamil NY, Alaraj M, et al. Kaempferol: unveiling its anti-inflammatory properties for therapeutic innovation. Cytokine 2025;186:156846. [PMID: 39754793 DOI: 10.1016/j.cyto.2024.156846]
- Sivasakthi P, Sanmuga Priya E, Senthamil Selvan P. Molecular insights into phytochemicals exhibiting anti-arthritic activity: systematic review. Inflamm Res 2021;70(6):665-85. [PMID: 34031706 DOI: 10.1007/s00011-021-01471-0]
- Nie J, Zhou L, Tian W, Liu X, Yang L, et al. Deep insight into cytokine storm: from pathogenesis to treatment. Signal Transduct Target Ther 2025;10(1):112. [PMID: 40234407 DOI: 10.1038/s41392-025-02178-y]
- Wang X, Fan D, Cao X, Ye Q, Wang Q, et al. The role of reactive oxygen species in the rheumatoid arthritis-associated synovial microenvironment. Antioxidants (Basel) 2022;11(6):1153. [PMID: 35740050 DOI: 10.3390/antiox11061153]
- Xue M, McKelvey K, Shen K, Minhas N, March L, et al. Endogenous MMP-9 and not MMP-2 promotes rheumatoid synovial fibroblast survival, inflammation and cartilage degradation. Rheumatology (Oxford) 2014;53(12):2270-9. [PMID: 24982240 DOI: 10.1093/rheumatology/keu254]
- Preethi V, Min KS, Gowda SVV, Eun HS, Hee KE, et al. Function of flavonoids on different types of programmed cell death and its mechanism: a review. J Biomed Res 2019;33(6):363-70. [DOI: 10.7555/JBR.33.20180126]
- Zhang C, Weng Y, Wang H, Zhan S, Li C, et al. A synergistic effect of triptolide and curcumin on rheumatoid arthritis by improving cell proliferation and inducing cell apoptosis via inhibition of the IL-17/NF-κB signaling pathway. Int Immunopharmacol 2024;142(Pt A):112953. [PMID: 39226828 DOI: 10.1016/j.intimp.2024.112953]
- Ye Y, Zhou J. The protective activity of natural flavonoids against osteoarthritis by targeting NF-κB signaling pathway. Front Endocrinol (Lausanne) 2023;14:1117489. [PMID: 36998478 DOI: 10.3389/fendo.2023.1117489]
- Chekalina N, Burmak Y, Petrov Y, Borisova Z, Manusha Y, et al. Quercetin reduces the transcriptional activity of NF-κB in stable coronary artery disease. Indian Heart J 2018;70(5):593-7. [PMID: 30392493 DOI: 10.1016/j.ihj.2018.04.006]
- Hussain MS, Altamimi ASA, Afzal M, Almalki WH, Kazmi I, et al. Kaempferol: paving the path for advanced treatments in aging-related diseases. Exp Gerontol 2024;188:112389. [PMID: 38432575 DOI: 10.1016/j.exger.2024.112389]
- Zeng MY, Tong QY. Anti-inflammation effects of sinomenine on macrophages through suppressing activated TLR4/NF-κB signaling pathway. Curr Med Sci 2020;40(1):130-7. [PMID: 32166675 DOI: 10.1007/s11596-020-2156-6]
- Li S, Sah DK, Arjunan A, Ameer MY, Lee B, et al. Triptolide suppresses IL-1β-induced expression of interleukin-8 by inhibiting ROS-mediated ERK, AP-1, and NF-κB molecules in human gastric cancer AGS cells. Front Oncol 2025;14:1498213. [PMID: 39950099 DOI: 10.3389/fonc.2024.1498213]
- Bartok B, Firestein GS. Fibroblast-like synoviocytes: key effector cells in rheumatoid arthritis. Immunol Rev 2010;233(1):233-55. [PMID: 20193003 DOI: 10.1111/j.0105-2896.2009.00859.x]
- Babaahmadi M, Makvand Gholipour N, Tayebi B, Pheneger J, Hajizadeh-Saffar E, et al. Clinical evaluation of collagen-induced arthritis in female Lewis rats: a comprehensive analysis of disease progression and severity. Cell J 2023;25(12):854-62. [PMID: 38192256 DOI: 10.22074/cellj.2023.2004504.1326]
- Buoncervello M, Marconi M, Carè A, Piscopo P, Malorni W, et al. Preclinical models in the study of sex differences. Clin Sci (Lond) 2017;131(6):449-69. [PMID: 28265036 DOI: 10.1042/CS20160847]
- Chen H, Dong Y, He X, Li J, Wang J. Paeoniflorin improves cardiac function and decreases adverse postinfarction left ventricular remodeling in a rat model of acute myocardial infarction. Drug Des Devel Ther 2018;12:823-36. [PMID: 29695894 DOI: 10.2147/DDDT.S163405]
- Yang J, Wei Z, Li H, Lv S, Fu Y, et al. Paeoniflorin inhibits the inflammation of rheumatoid arthritis fibroblast-like synoviocytes by downregulating hsa_circ_009012. Heliyon 2024;10(9):e30555. [PMID: 38726183 DOI: 10.1016/j.heliyon.2024.e30555]
- Bertoncini-Silva C, Vlad A, Ricciarelli R, Giacomo Fassini P, Suen VMM, et al. Enhancing the bioavailability and bioactivity of curcumin for disease prevention and treatment. Antioxidants (Basel) 2024;13(3):331. [PMID: 38539864 DOI: 10.3390/antiox13030331]
- Qiu C, Zhang JZ, Wu B, Xu CC, Pang HH, et al. Advanced application of nanotechnology in active constituents of Traditional Chinese Medicines. J Nanobiotechnology 2023;21(1):456. [PMID: 38017573 DOI: 10.1186/s12951-023-02165-x]
- Wang H, Chen Y, Wang L, Liu Q, Yang S, et al. Advancing herbal medicine: enhancing product quality and safety through robust quality control practices. Front Pharmacol 2023;14:1265178. [PMID: 37818188 DOI: 10.3389/fphar.2023.1265178]
- Kour G, Haq SA, Bajaj BK, Gupta PN, Ahmed Z. Phytochemical add-on therapy to DMARDs therapy in rheumatoid arthritis: in vitro and in vivo bases, clinical evidence and future trends. Pharmacol Res 2021;169:105618. [PMID: 33878447 DOI: 10.1016/j.phrs.2021.105618]
- Nikolova-Ganeva KA, Mihaylova NM, Kechidzhieva LA, Ivanova KI, Zarkov AS, et al. The therapeutic potential of phytochemicals unlocks new avenues in the management of rheumatoid arthritis. Int J Mol Sci 2025;26(14):6813. [PMID: 40725063 DOI: 10.3390/ijms26146813]
- Peng Q, Wang J, Li K, Xia C, Yao C, et al. Effects of plant active substances in rheumatoid arthritis—a systematic review and network meta-analysis. Front Pharmacol 2025;16:1536023. [PMID: 39974740 DOI: 10.3389/fphar.2025.1536023]
- Sumbul S, Ahmad MA, Asif M, Akhtar M, Saud I. Physicochemical and phytochemical standardization of berries of Myrtus communis Linn. J Pharm Bioallied Sci 2012;4(4):322-6. [PMID: 23248567 DOI: 10.4103/0975-7406.103266]
- Alfaqeh HH, Idrus RBH, Saim AB, Nordin A. Synergistic effects of natural products and mesenchymal stem cells in osteoarthritis treatment: a narrative review. Curr Issues Mol Biol 2025;47(6):445. [PMID: 40699844 DOI: 10.3390/cimb47060445]
- Abdelfatah S, Naß J, Knorz C, Klauck SM, Küpper JH, et al. Pyrrolizidine alkaloids cause cell cycle and DNA damage repair defects as analyzed by transcriptomics in cytochrome P450 3A4-overexpressing HepG2 clone 9 cells. Cell Biol Toxicol 2022;38(2):325-45. [PMID: 33884520 DOI: 10.1007/s10565-021-09599-9]
- Ng JY, Kim M, Suri A. Exploration of facilitators and barriers to the regulatory frameworks of dietary and herbal supplements: a scoping review. J Pharm Policy Pract 2022;15(1):55. [PMID: 36064655 DOI: 10.1186/s40545-022-00447-7]
- Sohn SI, Priya A, Balasubramaniam B, Muthuramalingam P, Sivasankar C, et al. Biomedical applications and bioavailability of curcumin-an updated overview. Pharmaceutics 2021;13(12):2102. [PMID: 34959384 DOI: 10.3390/pharmaceutics13122102]
- Hegde M, Girisa S, BharathwajChetty B, Vishwa R, Kunnumakkara AB. Curcumin formulations for better bioavailability: what we learned from clinical trials thus far? ACS Omega 2023;8(12):10713-46. [PMID: 37008131 DOI: 10.1021/acsomega.2c07326]
- Parvin N, Aslam M, Joo SW, Mandal TK. Nano-phytomedicine: harnessing plant-derived phytochemicals in nanocarriers for targeted human health applications. Molecules 2025;30(15):3177. [PMID: 40807355 DOI: 10.3390/molecules30153177]
- Estarriaga-Navarro S, Valls T, Plano D, Sanmartín C, Goicoechea N. Potential application of plant by-products in biomedicine: from current knowledge to future opportunities. Antioxidants (Basel) 2025;14(8):942. [PMID: 40867838 DOI: 10.3390/antiox14080942]
- Alum EU, Manjula VS, Uti DE, Echegu DA, Ugwu OPC, et al. Metabolomics-driven standardization of herbal medicine: advances, applications, and sustainability considerations. Nat Prod Commun 2025;20(8):1-16. [DOI: 10.1177/1934578×251367650]
- Tolossa K, Debela E, Athanasiadou S, Tolera A, Ganga G, et al. Ethno-medicinal study of plants used for treatment of human and livestock ailments by traditional healers in South Omo, Southern Ethiopia. J Ethnobiol Ethnomed 2013;9(1):32. [PMID: 23680260 DOI: 10.1186/1746-4269-9-32]
- Parveen A, Parveen B, Parveen R, Ahmad S. Challenges and guidelines for clinical trial of herbal drugs. J Pharm Bioallied Sci 2015;7(4):329-33. [PMID: 26681895 DOI: 10.4103/0975-7406.168035]
- Gamil NM, Elsayed HA, Hamed RM, Salah ET, Ahmed AM, et al. Insights from herb interactions studies: a foundational report for integrative medicine. Futur J Pharm Sci 2025;11(1):1-27. [DOI: 10.1186/s43094-025-00794-7]
- Chihomvu P, Ganesan A, Gibbons S, Woollard K, Hayes MA. Phytochemicals in drug discovery-a confluence of tradition and innovation. Int J Mol Sci 2024;25(16):8792. [PMID: 39201478 DOI: 10.3390/ijms25168792]
- Pushpalatha R, Selvamuthukumar S, Kilimozhi D. Nanocarrier mediated combination drug delivery for chemotherapy – a review. J Drug Deliv Sci Technol 2017;39:362-71. [DOI: 10.1016/j.jddst.2017.04.019]
- Ilic NM, Dey M, Poulev AA, Logendra S, Kuhn PE, et al. Anti-inflammatory activity of grains of paradise (Aframomum melegueta Schum) extract. J Agric Food Chem 2014;62(43):10452-7. [PMID: 25293633 DOI: 10.1021/jf5026086]
- Zhao X, Kim YR, Min Y, Zhao Y, Do K, et al. Natural plant extracts and compounds for rheumatoid arthritis therapy. Medicina (Kaunas) 2021;57(3):266. [PMID: 33803959 DOI: 10.3390/medicina57030266]
- Lekhmici A, Abdelwahab D, Noureddine C, Seddik K, Abderrahmane B. Preventive and curative effect of the methanolic extract of Ajuga iva on collagen induced arthritis in rats. Pharmacognosy Communications. 2013;3(2):17-23. [DOI: 10.5530/pc.2013.2.5]
- Koo HJ, Sung YY, Kim HK. Inhibitory effects of Akebia quinata ethanol extract on TNF-α-mediated vascular inflammation in human aortic smooth muscle cells. Mol Med Rep 2013;7(2):379-83. [PMID: 23175425 DOI: 10.3892/mmr.2012.1193]
- Lee HS, Kwon YJ, Seo EB, Kim SK, Lee H, et al. Anti-inflammatory effects of Allium cepa L. peel extracts via inhibition of JAK-STAT pathway in LPS-stimulated RAW264.7 cells. J Ethnopharmacol 2023;317:116851. [PMID: 37385574 DOI: 10.1016/j.jep.2023.116851]
- Yamasaki M, Yamasaki Y, Furusho R, Kimura H, Kamei I, et al. Onion (Allium cepa L.)-Derived nanoparticles inhibited LPS-induced nitrate production, however, their intracellular incorporation by endocytosis was not involved in this effect on RAW264 cells. Molecules 2021;26(9):2763. [PMID: 34067155 DOI: 10.3390/molecules26092763]
- Pourreza S, Azar PS, Sanaie S, Noshadi N, Jalali S, et al. Therapeutic effects and mechanisms of action of garlic (Allium sativum) on nonalcoholic fatty liver disease: a comprehensive systematic literature review. Evid Based Complement Alternat Med 2022;2022:6960211. [PMID: 37377647 DOI: 10.1155/2022/6960211]
- Pradhan R, Singh S, Singh N, Upadhyay R. Anti-inflammatory & anti-arthritic activity of aqueous extract of aloe vera in wistar albino rats. Int J Pharm Clin Res 2022;14(9):680-9.
- Paul S, Modak D, Chattaraj S, Nandi D, Sarkar A, et al. Aloe vera gel homogenate shows anti-inflammatory activity through lysosomal membrane stabilization and downregulation of TNF-α and Cox-2 gene expressions in inflammatory arthritic animals. Futur J Pharm Sci 2021;7(1):1-8. [DOI: 10.1186/s43094-020-00163-6]
- Deng X, Le H, Wan T, Weng M, Tan Y. Galangin alleviates rheumatoid arthritis in rats by downregulating the phosphatidylinositol 3-kinase/protein kinase B signaling pathway. Bioengineered 2022;13(4):11192-201. [PMID: 35485325 DOI: 10.1080/21655979.2022.2062969]
- Djehiche C, Benzidane N, Djeghim H, Tebboub M, Mebrek S, et al. Ammodaucus leucotrichus seed extract as a potential therapy in animal models of rheumatoid arthritis induced by complete freund adjuvant and chicken cartilage collagen. Appl Biochem Biotechnol 2024;196(11):8214-38. [PMID: 38700618 DOI: 10.1007/s12010-024-04952-0]
- Jadhav RB, Kaje GN, Thore DD, Nikam HD. Comprehensive study of Anacyclus pyrethrum. Int J Pharma Res Appl. 2025;10(1):227-37. [DOI: 10.35629/4494-1001227237]
- Sekkout Z, Youbi AEHE, Boudaia O, Janani S, Radallah D, Amrani NE. Traditional medicinal plants used for rheumatoid arthritis and immune system disorders treatment in the Casablanca-Settat region, Morocco: an ethnopharmacological study. Eur J Med Chem Rep 2024;11:100146. [DOI: 10.1016/j.ejmcr.2024.100146]
- Makbal R, Idrissi FEJ, Ouchbani T, Tastift MA, Kiai H, et al. Anti-inflammatory, antioxidant, chemical characterization, and safety assessment of Argania spinosa fruit shell extract from South-Western morocco. Biomed Res Int 2021;2021:5536030. [PMID: 34395619 DOI: 10.1155/2021/5536030]
- Kim SM, Vetrivel P, Kim HH, Ha SE, Saralamma VVG, et al. Artemisia iwayomogi (Dowijigi) inhibits lipopolysaccharide-induced inflammation in RAW264.7 macrophages by suppressing the NF-κB signaling pathway. Exp Ther Med 2020;19(3):2161-70. [PMID: 32104280 DOI: 10.3892/etm.2020.8472]
- Saleem M, Iftikhar A, Asif M, Hussain K, Alamgeer, et al. Asphodelus tenuifolius extracts arrested inflammation and arthritis through modulation of TNF-α, NF-κB, ILs, and COX-2 activities in in vivo models. Inflammopharmacology 2021;29(2):483-97. [PMID: 33064243 DOI: 10.1007/s10787-020-00761-z]
- Kaur C, Mishra Y, Kumar R, Singh G, Singh S, et al. Pathophysiology, diagnosis, and herbal medicine-based therapeutic implication of rheumatoid arthritis: an overview. Inflammopharmacology 2024;32(3):1705-20. [PMID: 38528307 DOI: 10.1007/s10787-024-01445-8]
- Rathod JD, Pathak NL, Patel RG, Ji vani NP, Patel LD, Chauhan V. Ameliorative effect of Bambusa arundinacea against adjuvant arthritis-with special reference to bone erosion and tropical splenomegaly. J Drug Deliv Ther 2012;2(3):141-5.
- Al-Harbi LN, Pandurangan SB, Al-Dossari AM, Shamlan G, Salamatullah AM, et al. Beta vulgaris rubra L. (Beetroot) peel methanol extract reduces oxidative stress and stimulates cell proliferation via increasing VEGF expression in H2O2 induced oxidative stressed human umbilical vein endothelial cells. Genes (Basel) 2021;12(9):1380. [PMID: 34573361 DOI: 10.3390/genes12091380]
- Van Thai H, Kim E, Kim SC, Jeong D, Yang S, et al. Boerhavia diffusa L. ethanol extract suppresses inflammatory responses via inhibition of Src/Syk/TRAF6. J Funct Foods 2015;17:476-90. [DOI: 10.1016/j.jff.2015.06.004]
- Hamed MA, Aboul Naser AF, Aboutabl ME, Osman AF, Hassan EES, et al. Bioactive compounds and therapeutic role of Brassica oleracea L. seeds in rheumatoid arthritis rats via regulating inflammatory signalling pathways and antagonizing interleukin-1 receptor action. Biomarkers 2021;26(8):788-807. [PMID: 34704882 DOI: 10.1080/1354750X.2021.1999504]
- Zhu H, Qian H, Yang Z, Liu Y. A combined therapy of Brassica rapa L. polysaccharides and dexamethasone in regulation of rheumatoid arthritis and gut microbiome. Food Front 2025;6(6):3024-41. [DOI: 10.1002/fft2.70110]
- Tanwar A, Chawla R, Ansari MM, Neha, Thakur P, et al. In vivo anti-arthritic efficacy of Camellia sinensis (L.) in collagen induced arthritis model. Biomed Pharmacother 2017;87:92-101. [PMID: 28049097 DOI: 10.1016/j.biopha.2016.12.089]
- Patel R, Kadri S, Gohil P, Deshpande S, Shah G. Amelioration of complete Freund’s adjuvant-induced arthritis by Calotropis procera latex in rats. Futur J Pharm Sci 2021;7(1):213. [DOI: 10.1186/s43094-021-00361-w]
- Liu X, Aimaier A, Wang W, Dong Y, Han P, et al. Quality variation and biosynthesis of anti-inflammatory compounds for Capparis spinosa based on the metabolome and transcriptome analysis. Front Plant Sci 2023;14:1224073. [PMID: 37528974 DOI: 10.3389/fpls.2023.1224073]
- He H, Hao M, Luo P, Chen J, An Y, et al. Inhibition peroxiredoxin-2 by capsaicin ameliorates rheumatoid arthritis via ROS-mediated apoptosis in fibroblast-like synoviocytes. MedComm (2020) 2025;6(6):e70209. [PMID: 40443720 DOI: 10.1002/mco2.70209]
- Lü S, Liu Y, Cui J, Yang B, Li G, et al. Mechanism of Caulophyllum robustum Maxim against rheumatoid arthritis using LncRNA-mRNA chip analysis. Gene 2020;722:144105. [PMID: 31521702 DOI: 10.1016/j.gene.2019.144105]
- Xiao S, Yu H, Guo Y, Cheng Y, Yao W. The antibacterial and anti-inflammatory potential of Cinnamomum camphora chvar. Borneol essential oil in vitro. Plants (Basel) 2025;14(12):1880. [PMID: 40573868 DOI: 10.3390/plants14121880]
- Sharma H, Chauhan P, Singh S. Evaluation of the anti-arthritic activity of Cinnamomum cassia bark extract in experimental models. Integr Med Res 2018;7(4):366-73. [PMID: 30591891 DOI: 10.1016/j.imr.2018.08.002]
- Shah BP, Patel NA, Shah SK. Anti-arthritic activity of ethanolic extract of citrus aurantium linn. leaves in complete freund’s adjuvant induced arthritis in rats. Indian Drugs 2023;60(03):81-8. [DOI: 10.53879/id.60.03.13354]
- Zou GS, Li SJ, Zheng SL, Pan X, Huang ZP. Lemon-Peel extract ameliorates rheumatoid arthritis by reducing xanthine oxidase and inflammatory cytokine levels. J Taiwan Inst Chem Eng 2018;93:54-62. [DOI: 10.1016/j.jtice.2018.07.036]
- Rajeshwari CU, Siri S, Andallu B. Antioxidant and antiarthritic potential of coriander (Coriandrum sativum L.) leaves. e-SPEN J 2012;7(6):e223-8. [DOI: 10.1016/j.clnme.2012.09.005]
- Gurisha MS, Rao PVK, Cherupally L, Mbise AJ. A review on therapeutic activities of Coriandrum sativum L for rheumatoid arthritis remedy. Asian J Res Biochem 2024;14(4):95-110. [DOI: 10.9734/ajrb/2024/v14i4298]
- Chrastina M, Dráfi F, Pružinská K, Poništ S, Kamga KS, et al. Crocus sativus L. extract (Saffron) effectively reduces arthritic and inflammatory parameters in monotherapy and in combination with methotrexate in adjuvant arthritis. Nutrients 2023;15(19):4108. [PMID: 37836391 DOI: 10.3390/nu15194108]
- Fonkoua M, Lambou GF, Nguemto GT, Ngassa DN, Youovop J, et al. Aqueous extract of Cucumis sativus Linné fruit attenuates rheumatoid arthritis-associated disorders. Arab J Med Aromat Plants 2022;8(3):131-48.
- Wahid S, Alqahtani A, Alam Khan R. Analgesic and anti-inflammatory effects and safety profile of Cucurbita maxima and Cucumis sativus seeds. Saudi J Biol Sci 2021;28(8):4334-41. [PMID: 34354417 DOI: 10.1016/j.sjbs.2021.04.020]
- Tan S, Xu J, Lai A, Cui R, Bai R, et al. Curculigoside exerts significant anti-arthritic effects in vivo and in vitro via regulation of the JAK/STAT/NF-κB signaling pathway. Mol Med Rep 2019;19(3):2057-64. [PMID: 30664158 DOI: 10.3892/mmr.2019.9854]
- Zeng L, Yang T, Yang K, Yu G, Li J, et al. Efficacy and safety of curcumin and curcuma longa extract in the treatment of arthritis: a systematic review and meta-analysis of randomized controlled trial. Front Immunol 2022;13:891822. [PMID: 35935936 DOI: 10.3389/fimmu.2022.891822]
- Kou H, Huang L, Jin M, He Q, Zhang R, et al. Effect of curcumin on rheumatoid arthritis: a systematic review and meta-analysis. Front Immunol 2023;14:1121655. [PMID: 37325651 DOI: 10.3389/fimmu.2023.1121655]
- Cárdenas Garza GR, Elizondo Luévano JH, Bazaldúa Rodríguez AF, Chávez Montes A, Pérez Hernández RA, et al. Benefits of cardamom (Elettaria cardamomum (L.) Maton) and turmeric (Curcuma longa L.) extracts for their applications as natural anti-inflammatory adjuvants. Plants (Basel) 2021;10(9):1908. [PMID: 34579443 DOI: 10.3390/plants10091908]
- Gohain F, Yachamaneni J, Kallepalli P, Ramesh N, Sarkar M, et al. Ethanolic extract of Equisetum arvense: a potential agent against rheumatoid arthritis in Wistar rats with freund’s complete adjuvant-induced arthritis. Indian J Pharm Educ Res 2024;58(3s):s986-94. [DOI: 10.5530/ijper.58.3s.98]
- Arooj B, Asghar S, Saleem M, Khalid SH, Asif M, et al. Anti-inflammatory mechanisms of eucalyptol rich Eucalyptus globulus essential oil alone and in combination with flurbiprofen. Inflammopharmacology 2023;31(4):1849-62. [PMID: 37179510 DOI: 10.1007/s10787-023-01237-6]
- Grespan R, Paludo M, Lemos Hde P, Barbosa CP, Bersani-Amado CA, et al. Anti-arthritic effect of eugenol on collagen-induced arthritis experimental model. Biol Pharm Bull 2012;35(10):1818-20. [PMID: 23037170 DOI: 10.1248/bpb.b12-00128]
- Rajesh B, Ramasubbualakshmi A, Gopalasatheeskumar K, Bharathi D, Nishanth R. Phytochemical analysis and anti-arthritic activity of ficus carica leaves. Asian J Res Chem 2020;13(2):151. [DOI: 10.5958/0974-4150.2020.00030.9]
- Marrelli M, Amodeo V, Viscardi F, De Luca M, Statti G, et al. Essential oils of foeniculum vulgare subsp. piperitum and their in vitro anti-arthritic potential. Chem Biodivers 2020;17(11):e2000388. [PMID: 33063941 DOI: 10.1002/cbdv.202000388]
- Patel DK. Therapeutic potential of a bioactive flavonoids glycitin from Glycine max: a review on medicinal importance, pharmacological activities and analytical aspects. Curr Tradit Med 2022;9(2):33-42. [DOI: 10.2174/2215083808666220513143957]
- Tu Y, Wang K, Liang Y, Jia X, Wang L, et al. Glycine tabacina ethanol extract ameliorates collagen-induced arthritis in rats via inhibiting pro-inflammatory cytokines and oxidation. J Ethnopharmacol 2019;237:20-7. [PMID: 30880257 DOI: 10.1016/j.jep.2019.03.035]
- Huang QC, Wang MJ, Chen XM, Yu WL, Chu YL, et al. Can active components of licorice, glycyrrhizin and glycyrrhetinic acid, lick rheumatoid arthritis? Oncotarget 2016;7(2):1193-202. [PMID: 26498361 DOI: 10.18632/oncotarget.6200]
- Andersen ML, Santos EHR, Seabra MLV, da Silva AAB, Tufik S. Evaluation of acute and chronic treatments with Harpagophytum procumbens on Freund’s adjuvant-induced arthritis in rats. J Ethnopharmacol 2004;91(2-3):325-30. [PMID: 15120457 DOI: 10.1016/j.jep.2004.01.003]
- Li L, Teng M, Liu Y, Qu Y, Zhang Y, et al. Anti-gouty arthritis and antihyperuricemia effects of sunflower (Helianthus annuus) head extract in gouty and hyperuricemia animal models. Biomed Res Int 2017;2017:5852076. [PMID: 28929115 DOI: 10.1155/2017/5852076]
- Qin M, Chen L, Hou X, Wu W, Liu Y, et al. Ultra-high-performance liquid chromatography-high-definition mass spectrometry-based metabolomics to reveal the potential anti-arthritic effects of Illicium verum in cultured fibroblast-like synoviocytes derived from rheumatoid arthritis. Metabolites 2024;14(10):517. [PMID: 39452898 DOI: 10.3390/metabo14100517]
- Subawa IW, Astawa P, Bakta IM, Astawa INM, Krisna GA. Purple sweet potato (Ipomoea batatas L.) extract effects on levels of inflammatory markers and chondrocyte count in gout arthritis Wistar rat model. Foot Ankle Surg 2023;29(8):611-5. [PMID: 37953101 DOI: 10.1016/j.fas.2023.07.007]
- Mobashar A, Shabbir A, Shahzad M, Gobe G. Preclinical rodent models of arthritis and acute inflammation indicate immunomodulatory and anti-inflammatory properties of Juglans regia extracts. Evid Based Complement Alternat Med 2022;2022:1695701. [PMID: 35422870 DOI: 10.1155/2022/1695701]
- Javed K, Rakha A, Butt MS, Faisal MN, Tariq U, et al. Evaluating the anti-arthritic potential of walnut (Juglans regia L.) in FCA induced Sprague Dawley rats. J Food Biochem 2022;46(10):e14327. [PMID: 35929358 DOI: 10.1111/jfbc.14327]
- Javed K, Rakha A, Butt MS, Faisal MN. Probing the antioxidant potential of Juglans regia (walnut) against arthritis-induced oxidative stress in Sprague Dawley rats. J Food Biochem 2022;46(5):e14082. [PMID: 35067927 DOI: 10.1111/jfbc.14082]
- Seo E, Hsieh YS, Lee JM, Shin YK, Seol GH. Prophylactic application of Lavandula angustifolia Mill. Oil and its active component linalyl acetate alleviate comorbidity and exacerbation risk in rats with collagen-induced arthritis accompanied by chronic nicotine-Induced hypertension. Res Square 2020. [DOI: 10.21203/rs.3.rs-127467/v1]
- Raval Nita D, Ashok BK, Ravishankar B. Evaluation of antiarthritic activity of Lepidium sativum Linn seeds against freund’s adjuvant induced arthritis in rats. Global J Res Med Plants Indigen Med 2013;2(7):532-7.
- Ghumare BC, Karande VV, Patodkar VR, Dhaygude VS, Suryawanshi RV. Evaluation of efficacy of Linum usitatisimum against CFA induced arthritis in rats. J Vet Pharmacol Toxicol 2013;20(2):19-23.
- Singh S, Nair V, Gupta YK. Linseed oil: an investigation of its antiarthritic activity in experimental models. Phytother Res 2012;26(2):246-52. [PMID: 21656600 DOI: 10.1002/ptr.3535]
- Dar E, Mobashar A, Shabbir A, Sharif A, Saleem A, et al. Therapeutic potential of d-limonene in rheumatoid arthritis: modulation of inflammatory, anti-inflammatory cytokines, and prostaglandin E2. Arch Pharm (Weinheim) 2024;357(12):e2400388. [PMID: 39460415 DOI: 10.1002/ardp.202400388]
- Aitbaba A, Kabdy H, Fatimzahra A, Azraida H, Abdoussadeq O, et al. Anti-inflammatory and antiarthritic properties of Marrubium vulgare aqueous extract in an animal model. Nat Prod Res 2025;39(22):6412-20. [PMID: 38949651 DOI: 10.1080/14786419.2024.2372656]
- Yoon CS, Lee H, Liu Z, Dong L, Lee G, et al. Cycloolivil isolated from Nardostachys jatamansi Inhibits TNF-α/IFN-γ-induced chemokine production by blocking NF-κB and JAK/STAT activation in HaCaT keratinocytes. Int J Mol Sci 2024;25(6):3342. [PMID: 38542316 DOI: 10.3390/ijms25063342]
- Sharma A, Goel A, Lin Z. Analysis of anti-rheumatic activity of Nyctanthes arbor-tristis via in vivo and pharmacovigilance approaches. Front Pharmacol 2023;14:1307799. [PMID: 38116080 DOI: 10.3389/fphar.2023.1307799]
- Sharma S, Yadav JP. Nyctanthes arbortristis: in vitro efficacy of anti-oxidant potential and cytotoxicity study of bark extracts on Sw982 rheumatoid arthritis cell line followed by phytochemical profiling by GCMS. J Adv Zool 2022;43(1):1218-29. [DOI: 10.53555/jaz.v43i1.5016]
- Phadtare S, Pandit R, Shinde V, Mahadik KR. comparative phytochemical and pharmacological evaluations of two varieties of Ocimum basilicum for antiarthritic activity. J Pharmacogn Phytochem 2013;2(2):158-67.
- Hong YH, Song C, Shin KK, Choi E, Hwang SH, et al. Tunisian Olea europaea L. leaf extract suppresses Freund’s complete adjuvant-induced rheumatoid arthritis and lipopolysaccharide-induced inflammatory responses. J Ethnopharmacol 2021;268:113602. [PMID: 33246116 DOI: 10.1016/j.jep.2020.113602]
- Abid F, Saleem M, Jamshaid T, Jamshaid U, Youssef FS, et al. Opuntia monacantha: validation of the anti-inflammatory and anti-arthritic activity of its polyphenolic rich extract in silico and in vivo via assessment of pro- and anti-inflammatory cytokines. J Ethnopharmacol 2024;326:117884. [PMID: 38350502 DOI: 10.1016/j.jep.2024.117884]
- Zhang J, Guo X, Bu Q, Shen X, Feng Z. AB0082 anti-inflammatory effects of total saponins of panax japonicus on rheumatoid arthritis. Ann Rheum Dis 2022;81(Suppl 1):1173. [DOI: 10.1136/annrheumdis-2022-eular.3460]
- Ren M, Ma K, Pang X, Liu Y, Song Z, et al. Anti-rheumatoid arthritis effects of total saponins from Rhizoma Panacis Majoris on adjuvant-induced arthritis in rats and rheumatoid arthritis fibroblast-like synoviocytes. Phytomedicine 2023;119:155021. [PMID: 37603974 DOI: 10.1016/j.phymed.2023.155021]
- Akhtar MF, Raza SA, Saleem A, Hamid I, Ashraf Baig MMF, et al. Appraisal of anti-arthritic and anti-inflammatory potential of folkloric medicinal plant Peganum harmala. Endocr Metab Immune Disord Drug Targets 2022;22(1):49-63. [PMID: 33563161 DOI: 10.2174/1871530321666210208211310]
- El-Gendy ZA, Abdelazeem S, Abdel Jaleel GA, Ali ME, Mohamed A, et al. Anti-inflammatory and anti-rheumatic effects of Phoenix dactylifera L. (date palm) seed by controlling cytokines and inhibiting JAK1/STAT3 pathway on CFA-induced arthritis rat and its phytochemical profiling. J Ethnopharmacol 2024;329:118138. [PMID: 38565410 DOI: 10.1016/j.jep.2024.118138]
- Jo HG, Baek CY, Kim D, Kim S, Han Y, et al. Network analysis, in vivo, and in vitro experiments identified the mechanisms by which Piper longum L. [Piperaceae] alleviates cartilage destruction, joint inflammation, and arthritic pain. Front Pharmacol 2024;14:1282943. [PMID: 38328576 DOI: 10.3389/fphar.2023.1282943]
- Das T, Dey YN, Pal T, Ghosh P, Ganguly A, et al. Preclinical assessment of anti-arthritic activity of methanolic extract from processed Plumbago zeylanica roots. Sci Rep 2025;15(1):11454. [PMID: 40181022 DOI: 10.1038/s41598-025-95744-x]
- Gautam RK, Sharma S, Sharma K, Gupta G. Evaluation of antiarthritic activity of butanol fraction of Punica granatum Linn. rind extract against freund’s complete adjuvant-induced arthritis in rats. J Environ Pathol Toxicol Oncol 2018;37(1):53-62. [PMID: 29773000 DOI: 10.1615/JEnvironPatholToxicolOncol.2018025137]
- Hussain A, Aslam B, Muhammad F, Faisal MN, Kousar S, et al. Anti-arthritic activity of Ricinus communis L. and Withania somnifera L. extracts in adjuvant-induced arthritic rats via modulating inflammatory mediators and subsiding oxidative stress. Iran J Basic Med Sci 2021;24(7):951-61. [PMID: 34712426 DOI: 10.22038/ijbms.2021.55145.12355]
- Wei T, Liu Y, Li M. Anti-inflammatory and anti-arthritic activity of rosmarinic acid isolated from Rosmarinus officinalis in an experimental model of arthritis. Indian J Pharm Educ Res 2021;55(2):507-16. [DOI: 10.5530/ijper.55.2.89]
- Ruckmani A, Meti V, Vijayashree R, Arunkumar R, Konda VR, et al. Anti-rheumatoid activity of ethanolic extract of Sesamum indicum seed extract in Freund’s complete adjuvant induced arthritis in Wistar albino rats. J Tradit Complement Med 2017;8(3):377-86. [PMID: 29992108 DOI: 10.1016/j.jtcme.2017.06.003]
- Harrabi S, Ferchichi A, Bacheli A, Fellah H. Policosanol composition, antioxidant and anti-arthritic activities of milk thistle (Silybium marianum L.) oil at different seed maturity stages. Lipids Health Dis 2018;17(1):82. [PMID: 29661192 DOI: 10.1186/s12944-018-0682-z]
- George G, Shyni GL, Mohan S, Abraham B, Nisha P, et al. In vitro and in vivo anti-inflammatory and anti-arthritic effect of Tinospora cordifolia via modulation of JAK/STAT pathway. Inflammopharmacology 2023;31(2):1009-25. [PMID: 36840884 DOI: 10.1007/s10787-023-01155-7]
- Song CY, Xu YG, Lu YQ . Use of Tripterygium wilfordii Hook F for immune-mediated inflammatory diseases: progress and future prospects. J Zhejiang Univ Sci B 2020;21(4):280-90. [PMID: 32253838 DOI: 10.1631/jzus.B1900607]
- Faghfoori Z, Javadivala Z, Khalili Y, Malek Mahdavi A. Effects of Trigonella foenum graecum (fenugreek) on rheumatoid arthritis: a systematic review. Immunopharmacol Immunotoxicol 2023;45(5):626-34. [PMID: 37039783 DOI: 10.1080/08923973.2023.2202298]
- Gajera VG, Meel R, Shah DP. Anti-arthritic activity of various fractions of Trigonella foenum graecum L. leaves in experimental animal models. Int J Pharm Res 2021;13(1):4959-71. [DOI: 10.31838/ijpr/2021.13.01.689]
- Abudoleh N. Anti-Arthritic activity of the methanolic leaf extract of Urtica pilulifera L. on albino rats. Am J Pharm Toxicol 2011;6(1):27-32. [DOI: 10.3844/ajptsp.2011.27.32]
- Saleem A, Saleem M, Akhtar MF, Shahzad M, Jahan S. Polystichum braunii extracts inhibit Complete Freund’s adjuvant-induced arthritis via upregulation of I-κB, IL-4, and IL-10, downregulation of COX-2, PGE2, IL-1β, IL-6, NF-κB, and TNF-α, and subsiding oxidative stress. Inflammopharmacology 2020;28(6):1633-48. [PMID: 32162074 DOI: 10.1007/s10787-020-00688-5]
- Szymczak J, Grygiel-Górniak B, Cielecka-Piontek J. Zingiber officinale roscoe: the antiarthritic potential of a popular spice-preclinical and clinical evidence. Nutrients 2024;16(5):741. [PMID: 38474869 DOI: 10.3390/nu16050741]
- Malhotra H, Garg V, Singh G, Dutt R. Ethanol extract of Ziziphus nummularia ameliorates formaldehyde-induced arthritis in rats by regulating oxidative stress biomarkers and haematological profile. Inflammopharmacology 2024;32(4):2463-76. [PMID: 38739176 DOI: 10.1007/s10787-023-01377-9]