A Promising Path Forward in Obesity Pharmacotherapy: Ecnoglutide and the Evolution of GLP-1R Agonist Design
1Stem Cells and Tissue Engineering Manufacture Center, School of Life Science, National & Local Joint Engineering Research Center of High-throughput Drug Screening Technology, Hubei University, Wuhan 430062, China
2Department of Biomedical Engineering, Faculty of Engineering, The Chinese University of Hong Kong, Hong Kong, SAR, China
3Department of Rheumatology and Immunology, Sichuan Provincial People’s Hospital, University of Electronic Science and Technology of China, Chengdu, China
4Department of Biochemistry and Molecular Biology, Institute of Basic Medical Sciences, College of Basic Medicine, Hubei University of Medicine, Shiyan 442000, China
*Correspondence to: Liangbin Zhou, Stem Cells and Tissue Engineering Manufacture Center, School of Life Science, National & Local Joint Engineering Research Center of High-throughput Drug Screening Technology, Hubei University, Wuhan 430062, China and Department of Biomedical Engineering, Faculty of Engineering, The Chinese University of Hong Kong, Hong Kong SAR, China, E-mail: jacobchou@link.cuhk.edu.hk; Dongze Wu, Department of Rheumatology and Immunology, Sichuan Provincial People’s Hospital, University of Electronic Science and Technology of China, Chengdu, China, E-mail: dongze_wu@163.com; Xiangdong Ye, Department of Biochemistry and Molecular Biology, Institute of Basic Medical Sciences, College of Basic Medicine, Hubei University of Medicine, Shiyan 442000, China, E-mail: yexiangdong@hbmu.edu.cn
Published Online: December 31 2025
Cite this paper:
Zhou L, Jin P, Wu D et al. A Promising Path Forward in Obesity Pharmacotherapy: Ecnoglutide and the Evolution of GLP-1R Agonist Design. BIO Integration 2025; 6: 1–4.
DOI: 10.15212/bioi-2025-0129. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
The escalating global prevalence of obesity continues to require innovative, safe, and effective therapeutic strategies. Although glucagon-like peptide-1 receptor agonists (GLP-1RAs) have ushered in a new era in weight management, the understanding of their mechanistic nuances and tolerability profiles continues to evolve. In this context, the phase 3 SLIMMER trial by Ji et al. in China achieved a notable and timely advancement. The trial explored ecnoglutide, a novel cyclic adenosine monophosphate (cAMP)-biased GLP-1RA, in the treatment of overweight and obesity in Chinese adults without diabetes [1].
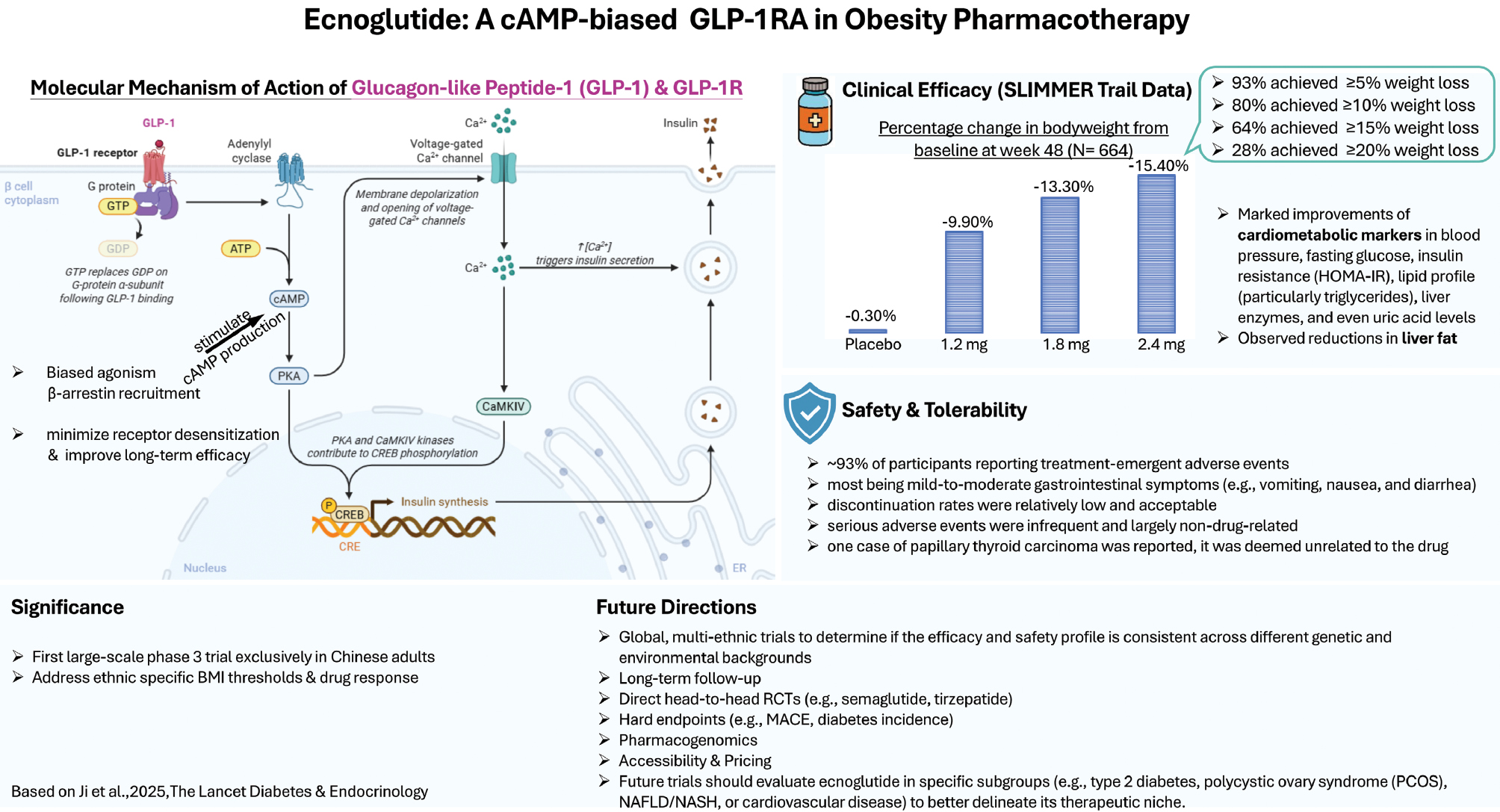
This rigorously conducted multicenter, randomized, double-blind, placebo-controlled study enrolled 664 participants and evaluated three doses of ecnoglutide (1.2, 1.8, and 2.4 mg weekly) over 48 weeks, which achieved robust, dose-dependent, and sustained body weight loss of 9.9%, 13.3%, and 15.4%, respectively, compared with a negligible 0.3% in the placebo arm [1] (Figure 1). These findings match or even surpass the outcomes observed in prior semaglutide and tirzepatide trials in Chinese populations (STEP 7 [2] and SURMOUNT-CN [3]), thus positioning ecnoglutide at the forefront of next-generation weight-loss pharmacotherapy.
Figure 1 Schematic diagram of ecnoglutide’s mechanism of action, clinical efficacy, safety profile, clinical significance, and future directions.
Importance of biased agonism and structural basis in Ecnoglutide’s profile
Ecnoglutide, like the widely used obesity drug semaglutide, is a GLP-1RA analogue that mimics the appetite and blood-sugar regulating hormone GLP-1. However, ecnoglutide uniquely stimulates cAMP production [4]; this distinct mechanistic bias toward cAMP signaling over β-arrestin recruitment might potentially minimize receptor desensitization and enhance long-term efficacy [4] (Figure 1). This biased agonism arises from specific interactions between ecnoglutide and the GLP-1 receptor that favor cAMP pathway activation. The structure-activity relationship between ecnoglutide and the GLP-1 receptor involves key residues that contribute to preferential activation of cAMP signaling. Specifically, ecnoglutide’s binding orientation at the receptor’s extracellular domain stabilizes the conformation that promotes cAMP production while minimizing β-arrestin recruitment. Structural biology findings, including recent X-ray crystallography and cryo-EM data of the GLP-1 receptor in complex with biased agonists, have further supported this mechanism and demonstrated how ecnoglutide selectively induces conformational changes in the receptor. By minimizing β-arrestin recruitment (which is associated with receptor internalization), cAMP-biased agonism might theoretically lead to decreased tachyphylaxis (tolerance), thus potentially sustaining weight loss efficacy over prolonged periods. However, such an advantage remains to be demonstrated in extended clinical studies.
The implications of this pharmacological selectivity are not merely academic: biased agonism provides a targeted strategy to optimize therapeutic benefit while minimizing off-target or desensitization-related adverse effects—issues increasingly recognized in long-term incretin therapy. The potential to decrease receptor desensitization might enhance the drug’s durability of action, thus making it suitable for chronic conditions such as obesity and type 2 diabetes. Although the study did not directly compare ecnoglutide with semaglutide or tirzepatide, a cross-trial comparison suggested at least comparable, if not superior, weight-loss efficacy at the 2.4 mg dose, including a higher proportion of participants achieving >15% and >20% bodyweight loss (64% and 28%, respectively) [1].
Study limitations
Despite its promising results, the SLIMMER trial has several limitations. The homogeneous Chinese cohort limits the generalizability of the findings to other ethnic or racial groups. In addition, the hypotheses generated by the study remain to be tested in global trials in diverse cohorts, to determine whether the efficacy and safety profiles are consistent across various genetic and environmental backgrounds. Moreover, the 48-week duration (short-term follow-up) was insufficient to assess long-term efficacy and safety, and the exclusion of patients with diabetes leaves a notable gap in understanding of the drug’s profile in a key comorbid population. The efficacy claims comparing ecnoglutide to semaglutide or tirzepatide were based on cross-trial observations. Because of the methodological limitations of such indirect comparisons, direct head-to-head randomized controlled trials are warranted in the future. Finally, the study did not include hard endpoints (e.g., MACE or diabetes incidence).
Beyond the scale: cardiometabolic improvements
In obesity pharmacotherapy, weight loss is not an endpoint occurring in isolation. In the SLIMMER trial, ecnoglutide use was accompanied by marked improvements in cardiometabolic markers: blood pressure, fasting glucose, insulin resistance (HOMA-IR), lipid profiles (particularly triglycerides), liver enzymes, and even uric acid levels [1]. The observed decreases in liver fat content (as much as –53.1%) among participants with baseline hepatic steatosis further support ecnoglutide’s utility in addressing metabolic dysfunction-associated steatotic liver disease, which frequently accompanies central obesity [1]. These outcomes suggest that ecnoglutide might eventually serve as a dual-action agent in metabolic and hepatic health, although confirmatory studies in nonalcoholic fatty liver disease/nonalcoholic steatohepatitis (NAFLD/NASH) populations are warranted.
Tolerability and safety profile
A common hurdle in GLP-1RA therapy is gastrointestinal tolerability, which often leads to premature treatment discontinuation [5]. Ecnoglutide’s safety profile mirrored that of other GLP-1RAs: ~93% of participants reported treatment-emergent adverse events, most of which were mild to moderate gastrointestinal symptoms (e.g., vomiting, nausea, and diarrhea), that attenuated over time [1]. Notably, the discontinuation rates were relatively low and were acceptable (ten participants across active arms), and serious adverse events were infrequent and largely non-drug-related [1].
Although one case of papillary thyroid carcinoma was reported, it was deemed unrelated to the drug [1]. Importantly, no instances of pancreatitis or gallbladder-related complications were observed [1], thus providing reassurance, given previous concerns regarding this class of drugs. Nonetheless, longer-term data and post-marketing surveillance will be essential to assess rare but serious events, particularly with long-term use.
Contextualizing in the Chinese population
This trial is particularly important because its recruitment of exclusively Chinese adults filled a notable gap in obesity drug trials, which often underrepresent Asian populations. The inclusion criteria of BMI ≥28 kg/m2 or ≥24 kg/m2 with comorbidities reflected local epidemiologic and clinical realities rather than Western BMI thresholds [1]. Therefore, the results are highly relevant to real-world clinical practice in East Asia and contribute to the emerging discourse regarding ancestry- or ethnicity-specific obesity thresholds and drug responsiveness.
What lies ahead?
Ecnoglutide’s demonstration of high efficacy, manageable tolerability, and cAMP-biased signaling suggests that it might serve as both a first-line and rescue option for patients with inadequate responses to semaglutide or those requiring more aggressive weight reduction. However, several questions remain. (1) A study comparing the clinical effects of a biased GLP-1RA analogue with those of a pharmacokinetically matched but balanced GLP-1RA analogue is necessary. How might ecnoglutide perform in direct head-to-head trials with semaglutide or tirzepatide? (2) Does ecnoglutide’s cAMP bias translate to lower rates of tachyphylaxis or greater durability of effect over the years? (3) What are the effects of ecnoglutide on hard clinical outcomes, such as cardiovascular events or diabetes incidence? (4) How will pricing, accessibility, and regulatory approval shape real-world use of ecnoglutide? Although promising clinical outcomes suggest ecnoglutide’s potential as a novel treatment for obesity and metabolic disorders, its market access might be influenced by factors such as affordability, insurance coverage, and the regulatory approval process. In China, navigating these barriers, including disparities in access to advanced treatments between urban and rural areas, will be crucial in ensuring that ecnoglutide reaches the populations most in need. Addressing these challenges through collaboration among regulatory bodies, healthcare providers, and industry stakeholders will be key to facilitating the broader use of this promising therapeutic option. (5) Future interdisciplinary collaboration should incorporate pharmacogenomics analysis to identify predictive biomarkers of ecnoglutide response. Additionally, future trials could evaluate ecnoglutide in specific subgroups, including individuals with type 2 diabetes, polycystic ovary syndrome, NAFLD/NASH, or cardiovascular disease, to better delineate its therapeutic niche.
Conclusion
The SLIMMER trial marks an important milestone in obesity therapeutics by demonstrating ecnoglutide’s potential, including its high efficacy, metabolic benefits, and tolerable safety profile [1]. The study demonstrated that ecnoglutide is particularly effective for individuals with a BMI ≥28 kg/m2 or those with at least one weight-related comorbidity. Therefore, ecnoglutide is suitable as both a first-line treatment and a second-line option for patients who do not respond sufficiently to lifestyle interventions or other GLP-1RAs. In an era in which obesity is recognized as a complex, multifactorial, and chronic disease with far-reaching health consequences, such pharmacologic innovation is critical. The magnitude of weight loss observed in this trial, particularly the proportions of participants achieving ≥15% and ≥20% weight reductions, is not only statistically significant but also clinically effective, thus indicating this drug’s potential to substantially decrease obesity-related comorbidities. As a cAMP-biased GLP-1RA analogue, ecnoglutide has a novel mechanism of action that preferentially targets intracellular pathways and might enhance efficacy while mitigating the receptor desensitization frequently observed with traditional unbiased agonists. This study offers a compelling proof of concept that precision pharmacology can refine existing drug classes to achieve better patient outcomes. Beyond investigating this mechanistic novelty, the trial’s focus on a Chinese population addresses the pressing need for region-specific data in a country with surging obesity prevalence. Ecnoglutide is mechanistically precise, metabolically holistic, and globally relevant, and therefore exemplifies goals for the next generation of obesity pharmacotherapy.
Conflict of interest
The authors declare that there are no conflicts of interest.
Funding and acknowledgements
No funding or sponsorship was received for this study.
References
- Ji L, Gao L, Xue H, Tian J, Wang K, et al. Efficacy and safety of a biased GLP-1 receptor agonist ecnoglutide in adults with overweight or obesity: a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol 2025;13(9):777-89. [PMID: 40555243 DOI: 10.1016/S2213-8587(25)00141-X]
- Mu Y, Bao X, Eliaschewitz FG, Hansen MR, Kim BT, et al. Efficacy and safety of once weekly semaglutide 2.4 mg for weight management in a predominantly east Asian population with overweight or obesity (STEP 7): a double-blind, multicentre, randomised controlled trial. Lancet Diabetes Endocrinol 2024;12(3):184-95. [PMID: 38330988 DOI: 10.1016/S2213-8587(23)00388-1]
- Zhao L, Cheng Z, Lu Y, Liu M, Chen H, et al. Tirzepatide for weight reduction in Chinese adults with obesity: the SURMOUNT-CN randomized clinical trial. JAMA 2024;332(7):551-60. [PMID: 38819983 DOI: 10.1001/jama.2024.9217]
- Guo W, Xu Z, Zou H, Li F, Li Y, et al. Discovery of ecnoglutide – A novel, long-acting, cAMP-biased glucagon-like peptide-1 (GLP-1) analog. Mol Metab 2023;75:101762. [PMID: 37364710 DOI: 10.1016/j.molmet.2023.101762]
- Bain EK, Bain SC. Recent developments in GLP-1RA therapy: a review of the latest evidence of efficacy and safety and differences within the class. Diabetes Obes Metab 2021;23(suppl 3):30-9. [PMID: 34324260 DOI: 10.1111/dom.14487]