Exogenous Extracellular Vesicles as Emerging Platforms in Translational Medicine
1Institute for Engineering Medicine, Kunming Medical University, Kunming 510631, China
2Department of Hepatobiliary Surgery, Jining Public Health Medical Center, Jining 272100, China
3Department of Molecular, Cell, and Cancer Biology, University of Massachusetts Chan Medical School, Worcester, MA, USA
4Thayer School of Engineering, Dartmouth College, Hanover, NH 03755, USA
5Department of Biological Sciences, University of North Carolina at Charlotte, Charlotte, NC 28223, USA
aThese authors contributed equally.
*Correspondence to: Jian Ma, E-mail: majian7@mail3.sysu.edu.cn; Lei Huang, E-mail: lei.huang@umassmed.edu; Zhijin Fan, E-mail: fanzhijin@m.scnu.edu.cn
Received: July 3 2025; Revised: August 11 2025; Accepted: August 27 2025; Published Online: September 19 2025
Cite this paper:
Bai M, Li Z, Shi T et al. Exogenous Extracellular Vesicles as Emerging Platforms in Translational Medicine. BIO Integration 2025; 6: 1–25.
DOI: 10.15212/bioi-2025-0122. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Extracellular vesicles (EVs) are nanoscale, membrane-bound carriers that are naturally secreted by cells and capable of transporting proteins, lipids, and nucleic acids across biological barriers. As key mediators of intercellular communication, EVs participate in immune regulation, tissue repair, and disease progression. The structural integrity of EVs, which is conferred by a lipid bilayer, protects cargo from degradation, while surface molecules facilitate targeted cell interactions. With increasing interest in their therapeutic utility, EVs have emerged as promising candidates in diagnostics, drug delivery, and regenerative medicine. EVs derived from milk, plants, and microbes exhibit distinct bioactivities, broadening the applicability in translational research. The intrinsic biocompatibility and low immunogenicity of EVs further enhance their clinical relevance. This review highlights recent advances in the understanding and biomedical utilization of exogenous EVs. The structural features, mechanisms of cellular uptake, and functional roles in modulating disease pathways are discussed. Furthermore, the unique advantages and challenges of leveraging exogenous EVs for clinical translation are explored, including standardization, loading efficiency, and targeting specificity. With continuous innovation at the interface of nanotechnology, synthetic biology, and biomedicine, exogenous EVs are poised to become next-generation platforms for precision therapy and regenerative strategies.
Keywords
Extracellular vesicles, Immunogenicity, Translational medicine, Plant-derived extracellular vesicles, Outer membrane vesicles.
Introduction
Cell-based therapies have emerged as transformative tools in regenerative medicine, immuno-oncology, and tissue engineering [1]. However, despite the significant therapeutic potential of cell-based therapies, these approaches face critical limitations, including immune rejection, tumorigenicity, limited engraftment, and complex manufacturing and regulatory hurdles [2]. These challenges have spurred growing interest in cell-free therapeutic strategies that aim to replicate the benefits of living cells, while addressing safety and scalability concerns [3, 4].
Among such strategies, extracellular vesicles (EVs), which are nano-sized, membrane-enclosed vesicles secreted by virtually all cell types, have garnered significant interest as next-generation therapeutic vectors [5]. As natural mediators of intercellular communication, EVs are enriched with diverse bioactive cargos, including proteins, nucleic acids, lipids, and metabolites, which are capable of modulating immune responses, promoting tissue repair, and influencing disease progression [6]. Compared to whole-cell therapies, EV-based approaches offer the following distinct advantages: lower immunogenicity; reduced toxicity; enhanced stability under physiologic conditions; and the intrinsic ability to traverse biological barriers, such as the blood-brain barrier, thereby broadening their clinical utility [7].
Of particular translational interest are exogenous EVs, which are derived from non-autologous sources, such as bovine milk, edible plants, and engineered microorganisms [8]. These vesicles offer notable practical and clinical advantages. Specifically, EVs can be produced at scale through cost-effective pipelines, are amenable to non-invasive delivery routes (e.g., oral or intravenous), and can be engineered to improve targeting or carry customized therapeutic cargos [9]. Additionally, the unique biomolecular signatures of EVs enable innovative applications in diagnostics and prognostics, particularly in cancer, neurodegenerative diseases, and inflammatory conditions [10].
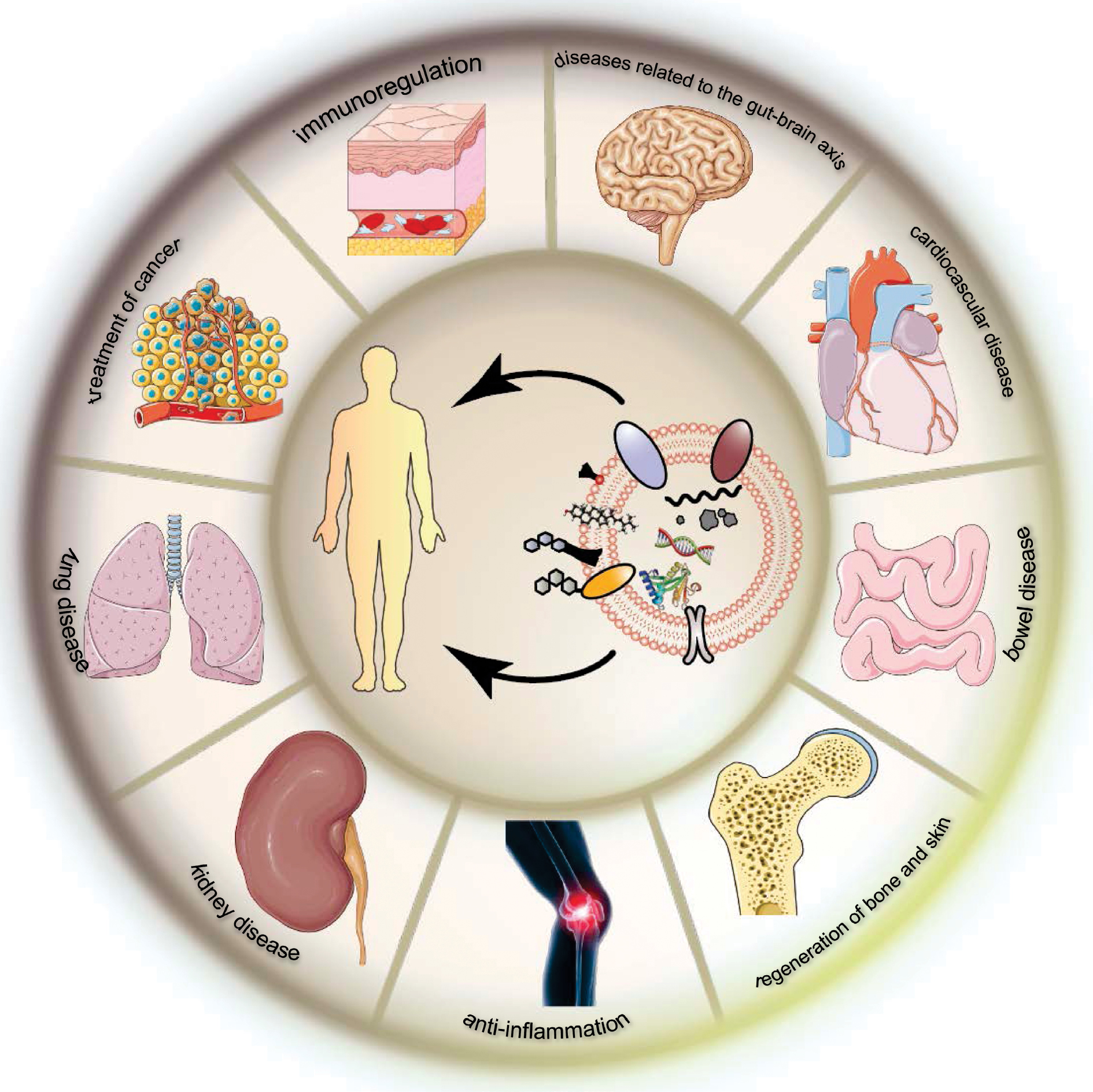
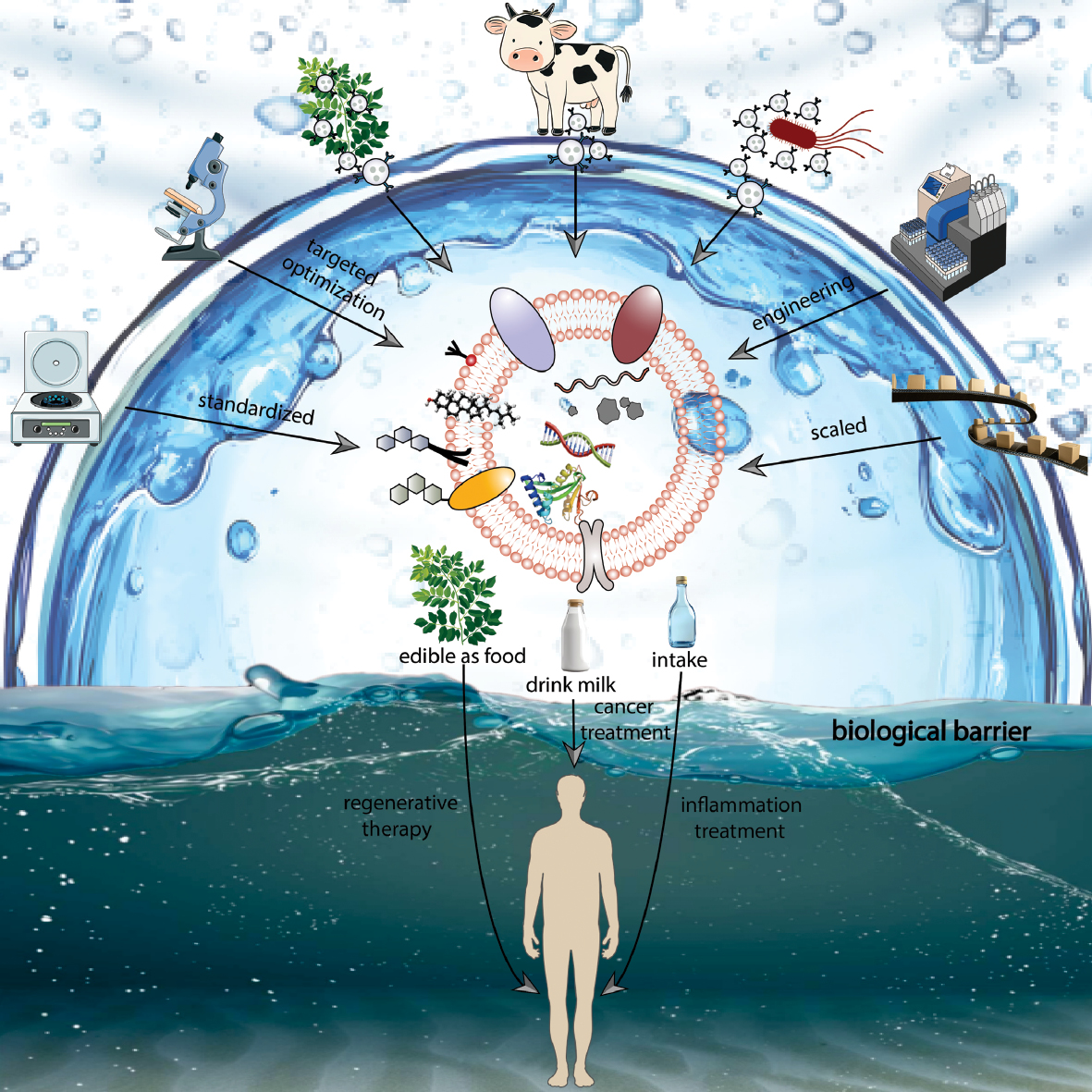
This review explores the emerging landscape of exogenous EVs in translational medicine (Scheme 1) with a focus on their roles in intercellular communication, immune modulation, and therapeutic innovation. The recent advances in the application of EVs in cancer therapy, regenerative medicine, and inflammatory disease management are critically examined, while highlighting key challenges requiring resolution to unlock the full clinical potential (standardization, bioengineering, and regulatory oversight) [11]. Exogenous EVs represent a promising, safe, and scalable alternative to traditional cell-based therapies and are poised to drive the next generation of precision medicine [5].
Scheme 1 Exogenous extracellular vesicles as emerging platforms in translational medicine. Created with the Adobe Illustrator.
Overview of EVs
Definition and structure of EVs
As research on EVs continues to advance, the essential roles of EVs in intercellular communication, immune modulation, and various physiologic and pathologic processes have become increasingly recognized. EVs are broadly defined as lipid bilayer-enclosed vesicles secreted by cells into the extracellular space [1]. While extensively studied in mammalian systems, EVs are also released by plants and bacteria, highlighting the evolutionary conservation and universal role in cross-kingdom communication. EVs encompass a diverse range of vesicles in multicellular organisms that are secreted by nearly all cell types [12]. EVs are commonly categorized into exosomes (30–150 nm), microvesicles (100 nm–1 μm), and apoptotic bodies (50–5000 nm) based on size, origin, secretion mechanisms, surface markers, and function (Figure 1) [1, 11, 13]. Even larger vesicles (i.e., large oncosomes, 1–10 μm in size) can bud directly from the plasma membrane in cancer [14]. Due to overlapping features and dynamic release patterns, precise classification of EVs remains a challenge [15].
Figure 1 The origin, structure, and function of extracellular vesicles. Created with the Adobe Illustrator.
EVs are enclosed by a lipid bilayer that recapitulates the asymmetric architecture of the parent cell membrane, which are enriched in cholesterol, sphingolipids, and glycosphingolipids. This specialized composition preserves membrane integrity, maintains vesicle morphology, and safeguards the encapsulated cargo from extracellular enzymatic degradation [16]. Key membrane proteins, such as tetraspanins (CD9, CD63, and CD81), serve as markers for EV subtypes and mediate interactions with recipient cells [17]. The presence of ceramides and specific phospholipids also implicates lipid rafts in cargo sorting during vesicle formation [18]. EVs carry a variety of bioactive molecules, including mRNA, microRNAs, DNA, proteins, and metabolites, that reflect the physiologic state of the source cells. These cargos are selectively packaged via endosomal sorting complex required for transport (ESCRT)-dependent and -independent mechanisms, enabling EVs to modulate gene expression and signaling pathways in recipient cells [6, 19, 20]. Similarly, plant-derived EVs have been shown to transport small RNAs that regulate microbial gene expression, while bacterial outer membrane vesicles (OMVs), which are equivalent to EVs, carry virulence factors, signaling molecules, and immunomodulatory compounds involved in host-pathogen and microbe–microbe interactions.
Importantly, the composition of EVs is dynamically influenced by the physiologic or pathologic state of the producing cells. Stress or transformation can alter the lipid or protein profiles of EVs, making EVs promising biomarkers for cancer, neurodegeneration, and inflammatory diseases [21]. This structural and functional plasticity also supports the growing interest in engineered EVs for therapeutic delivery with the potential for high targeting specificity and low immunogenicity [22]. In particular, plant- and bacteria-derived EVs offer unique advantages, such as oral bioavailability and immune compatibility, expanding the toolkit for drug and gene delivery. In summary, EVs are structurally organized, membrane-bound carriers that mediate intercellular communication across diverse biological kingdoms. Despite the heterogeneity in size, origin, and cargo, EVs share a defining phospholipid bilayer enriched with functional biomolecules, endowing EVs with stability, targeting capacity, and the ability to deliver bioactive contents to recipient cells [17, 19].
Discovery process and research history of EVs
The study of EVs can be traced back to the mid-20th century, when researchers first identified particulate matter in plasma with biological activity. In the 1940s, Chargaff and West observed that these sedimentable particles could accelerate blood coagulation, implying a functional role [13]. However, due to the limited resolution of early electron microscopy, these vesicles were initially mistaken for cellular debris. Indeed, these structures were initially dismissed as metabolic waste. A pivotal turning point came in the early 1980s, when Pan et al. reported that maturing reticulocytes actively secrete transferrin receptors via small vesicles [23]. This finding provided strong evidence that vesicle release is a regulated process, not merely a byproduct of cell damage [13, 24]. Around the same time, the term, “exosome,” was introduced by Trams et al. [25] to describe vesicles shed from the plasma membrane. Later, exosomes as vesicles were found to form within multivesicular bodies (MVBs) and release through fusion with the plasma membrane [24, 26]. In contrast, microvesicles are generated by direct outward budding from the plasma membrane. Although this biogenetic distinction is widely accepted, significant overlap in vesicle size and content has continued to challenge clear classification [2]. In the late 1980s and early 1990s, research expanded beyond erythroid cells. Immune cells, such as B lymphocytes and dendritic cells, were shown to release EVs capable of antigen presentation and intercellular molecular transfer [6]. These discoveries highlighted the role of EVs in immune regulation and laid the groundwork for future exploration in therapeutic and diagnostic contexts [27].
Despite growing interest in EVs, researchers struggled with the inconsistent terminology. Terms, like “platelet dust,” “microparticles,” and “exosome-like vesicles,” were used interchangeably, reflecting the lack of a unified classification system [13]. To resolve this dilemma, the International Society for Extracellular Vesicles (ISEV) recommended the umbrella term, “extracellular vesicles,” to encompass all membrane-bound vesicles with subclassification based on size and molecular markers (e.g., small EVs vs. large EVs) [9, 28]. A major step forward came with the launch of EVpedia in 2012, a public database aggregating EV composition data, homologous molecule identification, and bioinformatics tools [29]. This initiative, along with the release of the minimal information for studies of extracellular vesicles (MISEV) guidelines by the ISEV, helped standardize EV isolation, characterization, and experimental reporting, all of which were key to improving reproducibility and comparability across studies [13, 28]. Taken together, these milestones illustrate the evolving understanding of EVs from overlooked cellular byproducts-to-critical mediators of intercellular communication and potential tools for clinical application. The historical trajectory of EV research reflects a broader shift in cell biology, from static structural views-to-dynamic, communicative systems with therapeutic potential.
Role and mechanism underlying exogenous EVs in biological communication
EVs derived from allogeneic sources, such as different individuals of the same species or from distinct animal species, exhibit highly conserved mechanisms of biogenesis and functional characteristics. This evolutionary conservation suggests that EVs have developed a relatively universal regulatory system across diverse organisms [1]. The formation of EVs involves a series of conserved molecular pathways, including ESCRT-dependent membrane budding, the organization of membrane microdomains by tetraspanin family proteins, and the regulation of vesicle trafficking and secretion by Ras-like proteins from rat brain (Rab) GTPases. These shared mechanisms result in notable similarities among EVs from different sources in terms of structural features, membrane protein expression profiles, and cargo composition, including RNAs and proteins [24]. Moreover, ligand–receptor interactions on the EV surface are not strictly specific. Some surface molecules, such as integrins and tetraspanins, exhibit broad binding affinities, enabling EVs to recognize and be internalized by heterologous recipient cells. This cross-species targeting capability provides a strong theoretical basis for the application of EVs in interspecies communication and therapeutic interventions [1, 12].
In recent years, growing attention has been directed toward EVs derived from animals, plants, and even microorganisms for applications in disease treatment, drug delivery, and inflammation modulation. Owing to the low toxicity, minimal immunogenicity, and effective cross-species functionality of EVs, these naturally sourced EVs are emerging as promising candidates for next-generation biotherapeutic delivery systems.
Milk-derived EVs (milk EVs)
Milk EVs are primarily secreted by mammary epithelial cells with contributions from immune cells, stem cells, and other specialized cells [7]. A defining feature of these EVs is a robust lipid bilayer, which preserves the bioactive cargo under harsh conditions, such as digestive enzymes, low pH, high temperatures, and freezing. This stability ensures functional delivery during milk processing, storage, and gastrointestinal transit [30, 31]. In addition to nutritional roles, milk EVs serve as intercellular messengers, modulating immune development, gut homeostasis, and systemic responses [32, 33]. Notably, EV functions extend beyond infancy, potentially mediating interspecies communication and affecting health in adult dairy consumers [31].
Milk EVs are enriched with functional proteins, including classic exosomal markers (CD63, CD9, and Alix) that identify EVs and mediate cell signaling [34, 35]. Milk EVs also transport growth factors, cytokines, and enzymes involved in metabolism, inflammation, and tissue repair [30, 36]. The lipid composition of EVs, rich in phospholipids, cholesterol, and sphingolipids, facilitates membrane fusion and intracellular delivery. Some lipids further activate signaling pathways, such as ERK/MAPK, which promotes cell survival and proliferation [35, 37]. A key component of milk EVs is the RNA cargo, including thousands of mRNAs and regulatory non-coding RNAs (miRNAs, lncRNAs, and circRNAs). Dominant miRNAs, such as miR-148a, miR-21, and miR-146a, regulate immune responses, cell proliferation, and metabolism by modulating post-transcription gene expression [30, 34]. These RNAs act as epigenetic regulators, influencing chromatin structure and transcriptional activity, thereby affecting processes, like differentiation, development, and stress adaptation [33, 36].
Milk EVs interact with target cells through receptor binding or endocytic uptake [31, 32]. Once internalized, milk EVs release cargo into the cytoplasm, modulating signaling pathways, such as NF-κB, Wnt/β-catenin, PI3K/AKT, and mTORC1. These pathways govern inflammation, tissue repair, and epithelial integrity [31, 36]. Uptake efficiency is affected by membrane composition and environmental cues, like calcium, which also stabilize EVs during digestion [31, 38]. Milk EVs carry TGF-β and immune-regulatory miRNAs that promote regulatory T cell differentiation and modulate innate and adaptive immune cells [31, 34]. These effects contribute to neonatal immune maturation and tolerance [32, 33]. Moreover, milk EVs may exert long-term immunomodulatory effects, with implications in autoimmune diseases, allergies, and even cancer [35, 39].
Milk EVs deliver mRNAs and proteins that stimulate proliferation, enhance stem cell activity, and repair damaged tissues in tissue regeneration [30, 36]. Milk EVs support epithelial integrity and regeneration in the gut via activation of PI3K/AKT and mTORC1 signaling, which is aided by EV cargo (growth factors and cytokines) [7, 31, 40, 41]. Milk EVs also influence epigenetic programming, especially in early life. EV miRNAs can downregulate DNA methyltransferases, which alters gene methylation patterns that shape immune tolerance, metabolism, and neurodevelopmental changes with lasting health consequences [31, 33, 34, 42]. Milk EVs uniquely facilitate cross-species communication. The structural resilience of milk EVs allows milk EVs to survive digestion and potentially affect gene expression in non-native species, including humans consuming bovine milk [31, 33]. Milk EVs have attracted interest as natural drug delivery vehicles based on milk EV stability, biocompatibility, and low immunogenicity. Milk EVs can encapsulate and protect therapeutic agents, including chemotherapeutics and siRNAs, which enhances bioavailability while minimizing toxicity [31, 35]. For example, paclitaxel-loaded milk EVs demonstrate improved delivery and reduced side effects [38, 43]. Furthermore, milk EV composition reflects the health status of the lactating organism, making milk EVs promising non-invasive biomarkers for conditions, like metabolic disorders and cancer [30, 39].
In conclusion, milk EVs represent a powerful natural system for intercellular and interspecies communication with roles in immune regulation, tissue repair, developmental programming, and therapeutic delivery [7, 34, 42]. As research progresses, milk EVs hold great potential for clinical translation in diagnostics and targeted therapies [30, 31, 36].
Plant-derived EVs
The cross-kingdom communication capacity of plant-derived EVs holds profound implications for agriculture and medicine [44]. These EVs can penetrate rigid plant cell walls and be taken up not only by neighboring plant cells but also by microbial pathogens, symbionts, and even animal cells. EVs transport small RNAs (sRNAs) and defense proteins to pathogenic fungi, such as Botrytis cinerea and Verticillium dahliae, effectively silencing key virulence genes. This cross-kingdom RNA interference (RNAi) mechanism illustrates how plants utilize EV-mediated RNA transfer as an adaptive defense strategy to inhibit pathogen proliferation [45, 46].
In addition to pathogen defense, plant EVs mediate broader interspecies communication. During mutualistic symbiosis with arbuscular mycorrhizal (AM) fungi, EVs likely facilitate molecular signaling at the symbiotic interface, promoting nutrient exchange and modulating fungal metabolism and colonization to benefit both partners [47]. Similarly, pollen-derived EVs have critical roles in plant reproductive biology by facilitating molecular dialogues with the pistil that is essential for fertilization [46].
The antifungal function of plant EVs is well-supported by experimental evidence. For example, EVs isolated from sunflower seedling apoplastic fluid significantly inhibit fungal spore germination and induce morphologic abnormalities, confirming the presence of antifungal molecules that disrupt fungal cell integrity. Proteomic analyses further reveal that these EVs deliver enzymes involved in cell wall remodeling and defense, underscoring a multilayered antifungal strategy [48].
In parallel, plant-derived EVs are emerging as highly promising platforms for nanomedicine, owing to the biocompatibility, ability to cross biological barriers, and therapeutic versatility. EVs isolated from fruits and vegetables, such as grapes, grapefruit, bitter melon, and ginger, have demonstrated anti-inflammatory, anticancer, and gut-modulatory properties [49, 50]. Notably, folate-modified ginger-derived EVs have shown efficacy in remodeling the immune microenvironment, offering targeted therapeutic benefits for rheumatoid arthritis [51].
Plant-derived EVs also contribute to the modernization of traditional Chinese medicine. For example, exosome-like nanoparticles from Platycodon grandiflorus (“Jiegeng”) are rich in bioactive proteins and RNAs, exhibit low toxicity and excellent biocompatibility, and can significantly alleviate lung inflammation in animal models by modulating macrophage metabolism and polarization to suppress inflammatory cascades [52]. Similarly, ginseng-derived EV-like nanoparticles (GENs) alleviate colitis by modulating the gut microbiota and immune responses, helping to maintain intestinal homeostasis and prevent bacterial dysbiosis, which highlights the promise of GENs as novel therapeutics for inflammatory bowel disease (IBD) [53].
Furthermore, yam-derived nanovesicles (YNVs) enhance osteoblast differentiation and mineralization, showing significant therapeutic effects in an ovariectomy (OVX)-induced mouse model of osteoporosis [53]. Nanovesicles derived from goji berries (GqDNVs) promote MC3T3-E1 cell proliferation and differentiation, while accelerating bone fracture healing in mouse models, effects that are closely associated with activation of the PI3K/Akt/mTOR/p70S6K/4EBP1 signaling pathway [54]. The inherent robustness and ability of nanovesicles to traverse biological barriers, such as the gastrointestinal tract, position plant-derived EVs as ideal carriers for the oral delivery of therapeutics [49].
The uptake of plant EVs by recipient cells occurs through multiple mechanisms, including receptor-mediated endocytosis and direct membrane fusion. Intestinal epithelial cells in mammals internalize plant EVs via endocytosis, enabling plant EV cargo to modulate gene expression and immune responses [49]. Clathrin-mediated endocytosis appears to have a role in fungal EV uptake, although further investigation is needed to fully elucidate these pathways. Lipidomic profiling has revealed that plant EV membranes are enriched with bioactive lipids, such as phosphatidic acid, which may facilitate targeted membrane fusion [55].
In conclusion, plant-derived EVs represent sophisticated, dynamic carriers that reflect the complexity of cellular regulation and enable intra- and cross-kingdom communication. The ability of plant-derived EVs to transport functional RNAs and proteins across cellular barriers allows plants to coordinate defense responses, adapt to environmental challenges, and interact within diverse ecosystems [46, 48]. Continued advances in plant-derived EV science promise to unlock the potential of plant-derived EVs for enhancing crop resistance, developing innovative agricultural solutions, modernizing traditional medicine, and pioneering new nanomedicine applications [52, 56, 57].
Microbial-derived EVs
Microbial-derived EVs serve as fundamental communication tools bridging the microbial world and their hosts. One of the core functions is modulation of the host immune system. EVs from symbiotic and probiotic microbes have a crucial role in maintaining immune homeostasis and promoting tolerance, thereby preventing excessive inflammatory responses in the gut [58, 59]. These vesicles carry effector molecules capable of stimulating pro- or anti-inflammatory cytokine production. For example, EVs from probiotic strains, such as Escherichia coli Nissle 1917, promote the secretion of regulatory cytokines and induce T regulatory (Treg) cells, which are essential for balancing protective and tolerant immune responses [60, 61]. In contrast, EVs from pathogenic bacteria may carry virulence factors that trigger strong pro-inflammatory reactions by activating immune pathways, such as inflammasomes, contributing to disease pathogenesis, including IBD [62, 63]. Thus, microbial EVs have a dual function in immune regulation: maintaining intestinal barrier integrity and immune tolerance; and potentially exacerbating inflammation and tissue damage when derived from pathogens [58].
In addition to immune modulation, microbial EVs are vital for maintaining gut barrier function and preventing systemic dissemination of pathogens and toxins. Microbial EVs influence the expression of tight junction proteins, such as occludin, claudins, and zonula occludens, thereby enhancing barrier integrity [60, 64]. This regulation of tight junctions is not only critical for local gut health but also impacts systemic metabolism. For example, EVs from Akkermansia muciniphila improve intestinal permeability, which in turn regulates glucose metabolism and insulin sensitivity, possibly underlying the protective effects against obesity and metabolic disorders [63, 65]. Additionally, these vesicles often carry enzymes that degrade dietary polysaccharides, which aid in the production of beneficial metabolites, like short-chain fatty acids (SCFAs) that serve as energy substrates for colonocytes and have significant roles in host metabolic regulation [60, 66].
Moreover, recent studies have shown that the concentration of specific types of EVs is closely correlated with the progression of some diseases. For example, patients with IBD exhibit a significant reduction in Lactobacillus plantarum within the gut microbiota. Supplementation with L. plantarum or L. plantarum-derived EVs can alleviate IBD symptoms in mouse models [61]. Further investigations demonstrated that colonization by L. plantarum and L. plantarum-derived EVs exerts therapeutic effects on IBD severity by inhibiting intestinal epithelial cell apoptosis through the mPTP–CytC–caspase-9–caspase-3 signaling pathway [67].
Microbial EVs are key factors in bacterial communication in addition to mediating microbe-host interactions. By packaging and transferring genetic materials, such as DNA and antibiotic resistance genes, microbial EVs facilitate horizontal gene transfer, which enhances bacterial adaptability and enabling the spread of traits, like drug resistance and virulence within microbial communities [68]. EV-mediated communication also involves the transfer of quorum-sensing molecules and other signaling compounds, and coordinating collective behaviors, such as biofilm formation and phage defense mechanisms [63, 69].
Emerging research indicates that microbial EVs participate in the gut-brain axis, a novel field linking gastrointestinal microbiota composition with central nervous system function. EVs can cross the intestinal barrier and potentially the blood-brain barrier, delivering neuroactive compounds, including neurotransmitters, neuromodulators, and RNAs, that influence neuronal activity and behavior [69, 70]. For example, EVs derived from probiotic lactobacilli have been shown to regulate brain-derived neurotrophic factor (BDNF) and serotonin levels, producing antidepressant-like effects and affecting cognitive functions. These effects may result from direct neuronal uptake of EVs or indirect modulation through immune signaling and endocrine responses [45, 71]. Furthermore, the ability of EVs to deliver specific RNAs and regulatory molecules suggests a bidirectional communication pathway, whereby gut microbes may influence brain gene expression, neuroinflammation, and neurodegenerative disease processes [63, 72].
Microbial EVs primarily affect host cell function through RNA cargo. Bacterial- and host-derived RNAs, including mRNAs, small non-coding RNAs, and miRNA-like molecules, are delivered to target cells, where the RNAs regulate gene expression by modulating transcriptional and post-transcriptional processes [69, 72]. These regulatory RNAs can interact with host innate immune receptors, such as RIG-I and NOD1, triggering interferon production and other inflammatory responses [59, 73]. Conversely, host miRNAs can enter bacteria via EVs, potentially affecting bacterial gene expression related to biofilm formation and antibiotic resistance. This bidirectional RNA-mediated communication underscores the complexity of microbe-host interactions [63, 73].
In conclusion, microbial EVs are vital biological messengers that influence a broad spectrum of host functions through precise cargo delivery and receptor-mediated signaling. The roles of microbial EVs in immune regulation, barrier maintenance, metabolic homeostasis, and even brain function highlight the indispensability of microbial EVs in the complex ecosystem of host-microbiota interactions. With ongoing advances in research and technology, these vesicles are poised to further illuminate the molecular dialogues underpinning health and disease, ushering in a new era of microbiome-based clinical applications [64, 69].
Comparison of EVs from different sources
Milk EVs, particularly milk-derived EVs from bovine sources, offer the highest natural yield (up to 1012 particles/mL) and benefit from mature industrial supply chains, making them attractive for large-scale and GMP-compliant production [74]. Milk EV membranes are enriched with tetraspanins (CD9, CD63, and CD81) and ligands, such as MFG-E8 and integrins, which confer selective cellular targeting [75, 76]. These vesicles efficiently carry miRNAs, proteins, and other bioactive molecules, which have strong stability, efficient uptake, and low toxicity, although downstream purification from complex milk matrices still poses a cost challenge [77, 78].
In contrast, plant-derived EVs have intrinsically lower yields and face heterogeneity in raw materials and isolation protocols, resulting in limited scalability [79, 80]. Despite these limitations, plant EVs exhibit distinct molecular characteristics, including plant-specific tetraspanins (TET8/9) and unique lipid components, such as glycosyl inositol phosphoryl ceramides, which influence receptor recognition and vesicle stability [45, 77, 77]. With low immunogenicity and oral bioavailability, plant EVs hold promise for nutraceutical and therapeutic applications once standardization and large-scale manufacturing are established.
Microorganism-derived EVs present a cost-effective and scalable alternative due to the ease of bioreactor cultivation, theoretically producing 1017–1018 vesicles per batch. However, purification requires stringent removal of endotoxins and microbial contaminants. Unlike milk or plant EVs, microbial EVs rely on broad host-pathogen recognition mechanisms through pattern-recognition molecules and receptors rather than precise ligand–receptor interactions. These properties enable strong immunomodulatory effects but also necessitate thorough safety evaluation before clinical use [81, 82].
Immunogenicity of exogenous EVs
The immunogenicity of exogenous EVs largely depends on the parental cell origin and molecular composition. Compared to autologous EVs, allogeneic or xenogeneic EVs are more prone to recognition and clearance by innate immune cells, such as mononuclear phagocytes, and adaptive immunity, especially with repeated administration. This property is partly due to the major histocompatibility complex (MHC) molecules on EV surfaces, which are significantly enriched relative to the source cells and can activate T cells via antigen presentation, thus triggering adaptive immune responses [83].
When utilized as therapeutic delivery vehicles, EV immunogenicity is influenced by the parental cell type and the nucleic acid or protein cargo that is carried. Despite concerns that EVs from human immortalized cell lines (e.g., HEK293T) might harbor oncogenic or toxic elements, studies reveal that wild-type and engineered HEK293T-derived EVs, administered to mice over 3 weeks, induce no significant toxicity or immune activation [84]. Assessments covering blood biochemistry, immune cell phenotyping, and histopathology demonstrated no inflammatory infiltration, tissue damage, or functional abnormalities. Minor fluctuations in immune markers, such as IP-10 and VCAM-1, only occur in engineered EV groups and remain within physiologic limits, indicating that engineered EVs with exogenous RNAs or protein tags maintain low immunogenicity, supporting clinical translation safety [85].
Mononuclear phagocytes discriminate self from non-self EVs through polymorphic ligand-receptor interactions. Therefore, xenogeneic and allogeneic EVs may be rapidly cleared even without adaptive immune involvement [86, 87]. For example, SIRPα receptor polymorphisms in mice increase macrophage phagocytosis of xenogeneic EVs, resembling xenograft clearance. Despite this finding, clinical trials report no evident immunotoxicity from autologous or allogeneic exosomes and plasma transfusions containing native EVs do not provoke significant adverse reactions, which underscores the high biocompatibility [88].
A central challenge in cross-individual EV immunogenicity arises from immune memory responses triggered by repeated dosing. Donor-recipient genetic differences can elevate the immunogenic risk of allogeneic or xenogeneic EVs. Nevertheless, studies show minimal immune responses in mice exposed to human-derived HEK293T EVs, with no significant changes in splenic T, B, or NK cell populations or systemic inflammatory cytokines (e.g., IL-6 and TNF-α) [85]. In non-human primates, repeated intravenous allogeneic EV injections cause accelerated blood clearance (ABC), which is marked by a shortened EV circulation half-life and increased EV-specific IgG antibodies, indicating antibody-mediated immune memory that is likely driven by EV surface MHC molecules [89]. In contrast, engineered EV surface tags, such as FLAG or PC94 peptide, do not elicit anti-drug antibodies, suggesting inherent immune “silencing” properties of EVs. Future studies should confirm if such immune evasion applies broadly across diverse EV types and animal models [85].
EVs often acquire biomolecular coronas (adsorbed plasma proteins, including complement factors and immunoglobulins) in vivo that can exacerbate immune recognition by activating complement cascades or Fc receptor-mediated phagocytosis. Gene-editing techniques knocking out B2M in EV-producing cells effectively lower MHC-I expression to reduce cross-individual immunogenicity, which significantly diminishes allogeneic EV immunogenicity [89]. Additionally, mesenchymal stem cell (MSC)-derived EVs naturally exhibit low immunogenicity and immunosuppressive properties. For example, MSC-EVs modulate ischemia-reperfusion injury by downregulating pro-inflammatory cytokines and promoting anti-inflammatory macrophage polarization, which favors allogeneic application [1]. Genetic engineering to overexpress immune checkpoint molecules, like PD-L1 on EV surfaces, further enhances the ability to suppress T cell activation and evade adaptive immunity [90].
The cross-species immunogenicity of EVs is primarily dictated by cellular origin and immune recognition. Compared to autologous EVs, xenogeneic and allogeneic EVs display greater susceptibility to innate and adaptive immune clearance, especially under repeated administration. This characteristic arises from foreign antigenic molecules on EV surfaces, especially elevated MHC-I levels in xenogeneic EVs, that interact with T cell receptors to activate adaptive immunity. Innate immune cells also recognize polymorphic differences in molecules, like CD47 through SIRPα receptors, which triggers phagocytosis [86]. Nevertheless, preclinical studies reveal low immunogenicity of human EVs in mouse and non-human primate models. For example, repeated injections of human Expi293F EVs do not cause significant toxicity or systemic inflammation, although circulation times decrease due to ABC, which correlates with EV-specific IgG production [89]. These findings suggest that EV immunogenicity is context-dependent and influenced by origin, dosage, and host immune status.
Exogenous EVs are promising therapeutic carriers given the low immunogenicity. Multiple studies have confirmed the safety of human-derived EVs. Zhu et al. demonstrated that wild-type and engineered HEK293T EVs did not induce toxicity in mice after 3 weeks with only minor immune marker changes in the engineered EV groups [85]. Similarly, Saleh et al. reported that Expi293F-derived EVs did not impair HepG2 cell viability in vitro or provoke systemic immune responses in BALB/c mice, further supporting human EV safety [89].
Engineering approaches can enhance EV immune compatibility and expand clinical utility. Genetic modifications or chemical conjugations embedding chimeric proteins or pre-miR-199a-3p reduce immune marker fluctuations, improving biosafety. Zhu et al. reported minimal VCAM-1 changes from engineered EVs without significant toxicity differences versus controls, demonstrating a balance of functionality and safety [85]. Species differences in EV surface proteins, like MHC, may influence immune responses in cross-species applications. However, murine tolerance to human EVs is encouraging for preclinical research.
Clinical interest focuses on EVs from regenerative cells. Cardiovascular progenitor cell-derived EVs for non-ischemic dilated cardiomyopathy (trial NCT05774509) are administered repeatedly to elicit reparative immunomodulation without harmful immune activation. Early data showed low immunogenicity and safety in repeated dosing. MSC-derived EVs, leveraging the parent cell intrinsic immunosuppressive properties, are under clinical evaluation (trial NCT05787288). These EVs shift immune phenotypes from pro- to anti-inflammatory and induce regulatory T cell expansion. The low immunogenicity is due to minimal MHC-II expression and immunoregulatory cargo (microRNAs and proteins) that reprogram recipient immunity. Preclinical studies have confirmed that MSC-EVs suppress allogeneic peripheral blood mononuclear cell activation without cytokine storm or adverse events.
Intravenous infusion of bone marrow MSC-derived EVs (ExoFlo) in COVID-19-associated ARDS patients (trial NCT04493242) showed no treatment-related immunogenic adverse events despite extensive immune monitoring. Other trials (NCT06002841, NCT06242379, and NCT06825572) evaluating MSC-EVs in liver failure post-transplant patients revealed no notable immune rejection or pathologic inflammation when combined with standard immunosuppression.
In contrast, pathogen-derived EVs (pEVs) possess marked immunogenicity due to cargo, including lipopolysaccharides, peptidoglycans, and microbial nucleic acids (pathogen-associated molecular patterns [PAMPs] [91]. These PAMPs are recognized by host pattern recognition receptors (PRRs), such as toll-like receptors (TLRs), NOD-like receptors, and STING, all of which activate potent innate immune responses. pEVs also carry toxins and virulence factors that can amplify inflammation or suppress immunity to favor pathogen survival [92]. This finding illustrates the inherent capacity of exogenous EVs to carry antigenic material triggering innate and adaptive immunity [93]. In addition to pathogen recognition, EVs regulate allogeneic immune responses, especially in transplantation. Donor EVs can transfer intact MHC molecules to recipient cells (“cross-dressing”), enabling recipient antigen-presenting cells to activate allogeneic T cells. Some EV subtypes also deliver immunosuppressive factors, such as Fas ligand, TGF-β, and microRNAs, that induce T cell anergy or regulatory T cell expansion, potentially fostering graft tolerance over rejection [94].
Placenta-derived EVs promote immune tolerance, which is crucial in pregnancy. By modulating maternal immunity, placenta-derived EVs prevent rejection of the semi-allogeneic fetus, balancing pathogen defense with fetal tolerance [95]. This mechanism offers valuable paradigms for the safe clinical use of EVs in transplantation and regenerative medicine, highlighting the therapeutic potential in immune regulation without provoking harmful immunogenicity [96].
In conclusion, the immunogenicity of exogenous EVs depends strongly on cellular origin, cargo, and surface molecules. Regenerative cell-derived EVs have low immunogenicity and potent immunomodulatory effects, supporting the therapeutic potential (e.g., clinical trials NCT05774509 and NCT05787288). Conversely, pathogen- or tumor-derived EVs exhibit high immunogenicity, triggering robust innate and adaptive immune responses [91, 93]. This dual nature positions EVs as versatile immunotherapeutics, for which rational engineering and dosing strategies are essential to optimize safety and efficacy [96, 97].
Application of exogenous EVs in biomedicine
Building upon the intrinsic properties and source-specific advantages of exogenous EVs, the exploitation in biomedicine has gained significant momentum (Table 1), particularly in areas with high unmet clinical needs and where traditional therapies face limitations [98]. The ability of EVs to navigate biological barriers, deliver bioactive cargo with precision, and modulate cellular functions, often with inherent low immunogenicity and favorable safety profiles, makes EVs uniquely suited for therapeutic intervention [22]. This section delves into the burgeoning applications of exogenous EVs across three key translational domains: cancer therapy (leveraging targeting, drug delivery, and immunomodulation); regenerative medicine (capitalizing on pro-regenerative signaling and biomaterial integration); and inflammatory diseases (exploiting anti-inflammatory and immunoregulatory actions).
Table 1 Application of Exogenous Extracellular Vesicles in Translational Medicine
| Source | Vesicle Type and Size (diameter/nm) | Active Molecules | Isolation Method | Engineering Strategy | Disease Model | Therapeutic Effect | Advantages & Disadvantages | Ref | |
|---|---|---|---|---|---|---|---|---|---|
| Milk-derived EVs | Human | Breast milk exosomes (30–150) | Lipids | Ultracentrifugation | None | Necrotizing enterocolitis (NEC) | Administration of full-term and preterm milk exosomes significantly enhanced in vitro epithelial proliferation and migration, and improved the severity of NEC in vivo | Disadvantage: Low lipid content extracted | [8] |
| Human | Breast milk exosomes (30–150) | CD36 | Ultracentrifugation | None | Non-alcoholic fatty liver disease (NAFLD) | Inhibited lipogenesis and increased lipolysis, effectively alleviating high-fat diet-induced hepatic steatosis and insulin resistance in NAFLD mice | Limitation: The composition of bioactive compounds in breast milk, including EV concentration, varies across lactation stages; conclusions only apply to EVs from human milk administered intraperitoneally, not orally | [51] | |
| Human | Human colostrum and derived extracellular vesicles (50–200) | MFG-E8 | ExoQuick purification | Co-incubation | Resistance to rotavirus and respiratory syncytial virus | Intrinsic antiviral activity of human colostrum against rotavirus and respiratory syncytial virus | Contributes to revealing new mechanisms of the functional role of breast milk as a protective and therapeutic agent for preterm infants; ExoQuick purification is a method with “high recovery rate and low specificity,” cannot exclude the presence of protein contaminants or antiviral molecules | [132] | |
| Human | Human milk exosomes (30–150) | Growth Factors | Ultracentrifugation | None | Murine Necrotizing Enterocolitis (NEC) | Reduced hypoxia, inflammation formation, and intestinal damage | Not mentioned | [133] | |
| Pig | Porcine milk exosomes (40–100) | miR-4334, miR-219, and miR-338 | Ultracentrifugation | None | LPS-induced intestinal inflammation and apoptosis | Inhibited cellular inflammation and prevented apoptosis, thereby protecting intestinal epithelial cells from LPS-induced damage | Porcine milk exosomes contain a large amount of miRNAs, whose target genes are rich in immunity, and miRNAs encapsulated in exosomes can survive gastrointestinal conditions | [134] | |
| Pig | Porcine milk exosomes (40–100) | miRNAs, mRNAs, and proteins | Centrifugation | None | Murine intestinal proliferation | Promoted intestinal cell proliferation and intestinal development | Not mentioned | [135] | |
| Cow | Bovine milk exosomes (30–200) | Active proteins | Ultracentrifugation | None | Hair regeneration | Milk-exo promoted the proliferation of hair dermal papilla cells and rescued dihydrotestosterone (DHT, androgen)-induced hair follicle developmental arrest | Compared to animal cell-derived exosomes, milk-derived exosomes have very high yields, which have economic advantages in drug commercialization | [136] | |
| Cow | Bovine milk exosomes (30–201) | miRNA-2478 | Centrifugation | Co-incubation | Melanin formation | Bovine milk exosomes reduced melanin content, tyrosinase activity, and expression of melanogenesis-related genes in melanoma cells and melanocytes, and bovine-specific miR-2478 inhibited melanin production | Contributes to the development of functional whitening cosmetics with low cytotoxicity | [137] | |
| Cow | Bovine milk-derived EVs [BMEVs] (30–200) | Not specifically mentioned | Centrifugation | None | Murine osteogenesis | Increased the number of osteoblasts and woven bone formation, contributing to altered osteoblast differentiation and bone matrix formation | BMEV seems to contribute to the development and dominance of osteoblasts in the immature stage, with faster differentiation into osteoblasts in the later stage | [138] | |
| Plant-derived EVs | Grape | Grape exosome-like nanoparticles (EPDEN) (380.5 ± 37.47) | Lipids | Centrifugation | Sucrose step gradient | Murine colitis | Not only regulates intestinal tissue renewal processes but also participates in the remodeling in response to pathological triggers |
|
[138] |
| Ginger | Ginger EPDENs (0–250) | Heme oxygenase-1 (HO-1) and anti-inflammatory cytokine IL-10 | Centrifugation | Sucrose step gradient | Murine in vitro experiments | Ginger EPDENs preferentially induced the expression of the antioxidant gene, heme oxygenase-1 (HO-1), and the anti-inflammatory cytokine, IL-10. | [139] | ||
| Ginger | Ginger EPDENs (0–250) | Nuclear factor erythroid 2-related factor 2 (Nrf2) | Centrifugation | Sucrose step gradient | Murine liver injury | Mediated activation of Nrf2 leading to the expression of a set of liver detoxification/antioxidant genes and inhibition of reactive oxygen species production. | [140] | ||
| Grapefruit | Grapefruit-derived nanovesicles (0–250) | Heme oxygenase-1 (HO-1) | Centrifugation | Sucrose step gradient | Murine colitis | Vesicles were selectively taken up by intestinal macrophages and improved murine colitis | [141] | ||
| Lemon | Citrus limon-derived nanovesicles (50–70) | TRAIL | Ultracentrifugation | Sucrose step gradient | Murine in vitro tumor model | Inhibited cancer cell proliferation in different tumor cell lines by activating TRAIL-mediated apoptotic cell death | [142] | ||
| Carrot | Carrot EPDEN | Nuclear factor (e rythroid-derived 2)-like 2 (Nrf2) | Centrifugation | Sucrose step gradient | Murine inflammation model | Carrot EPDEN increased nuclear translocation of Nrf2 | [139] | ||
| Grapefruit | Grapefruit-derived nanovesicles (0–250) | Not mentioned | Gradient centrifugation | Vortex mixing and assembly | In vitro cell culture model and murine tumor model | Excellent candidates for delivering various types of therapeutic agents | [143] | ||
| Arabidopsis | Exosome-like extracellular vesicles | Small RNAs (sRNAs) | None | None | Arabidopsis fungus | Silenced genes related to fungal pathogenicity | [126] | ||
| Ginger | Folic acid-modified ginger-derived extracellular vesicles (268) | TNF-α 和 IL-1β | Combined electrophoretic technique with 300 kDa cut-off dialysis bag (named ELD) | Conjugating GDEVs with folic acid (FA) | Mouse arthritis model | Demonstrated significant ability to alleviate RA symptoms in mouse models | [51] | ||
| Microbial-derived EVs | Pediococcus pentosaceus | Extracellular membrane vesicles (235–245) | IL-10, arginase-1, PD-L1 | Ultracentrifugation | None | Mouse models of liver fibrosis, peritonitis, and colitis | Reduced pathologic processes with potent immunoregulatory effects | Not mentioned | [144] |
| Propionibacterium freudenreichii CIRM-BIA 129 | Extracellular Vesicles (84.80 ± 2.34) | SlpB | Centrifugation | None | HT-29/kb-seap-25 cell inflammation model | Attenuated OMV-driven neuroinflammation by inhibiting LPS-induced NF-κB signaling pathway | Whether P. freudenreichii EVs exert immunoregulatory effects in vivo is unknown | [145] | |
| Lactobacillus paracasei | L. paracasei-derived extracellular vesicles (34.22 ± 6.876) | ER stress-related proteins, IL-10, and TGFβ | Ultracentrifugation | None | LPS-induced inflammation in HT29 human colorectal cancer cells and DSS-induced colitis in C57BL/6 mice | Alleviated LPS-induced intestinal inflammation through ER stress activation | Not mentioned | [146] | |
| E. coli Nissle 1917 | Outer membrane vesicles (OMVs) [size not mentioned] | tight junction proteins ZO-1 and claudin-14, and IL-22 activation | Ultracentrifugation | Genetic engineering | Human intestinal epithelial barrier model | OMVs contributed to enhancing epithelial barrier integrity directly through transcriptional regulation of TJ proteins and indirectly through modulation of IL-22 | Not mentioned | [147] | |
| Akkermansia muciniphila | A. muciniphila-derived extracellular vesicles (AmEVs) [40–60] | occludin | Ultracentrifugation | None | Diabetic mouse model | AmEVs may function as a component controlling intestinal permeability, and the regulation of intestinal barrier integrity can improve metabolic functions in HFD-fed mice | The results do not verify whether EVs remain intact after systemic travel | [148] | |
| L. plantarum APsulloc 331261 | L. plantarum-derived EVs (LEVs) [104 ± 42.4] | IL-10, IL-1B, and GM-CSF | Ultracentrifugation | Density gradient | Human inflammatory skin disease cell model | LEVs can trigger M2 macrophage polarization in vitro and induce anti-inflammatory phenomena in human skin | Low-cost production; specific EV factors responsible for effects are unknown | [149] | |
| L. plantarum | L-EVs (particle size not mentioned) | Neurotrophic factors (Bdnf1/4 and Nt4/5) | Ultracentrifugation | None | HT22 cell model treated with the stress hormone glucocorticoid (GC; corticosterone), depressive-like mouse model | Can alter the expression of neurotrophic factors in the hippocampus and provide antidepressant-like effects in mice with stress-induced depression | Not mentioned | [150] | |
| B. thetaiotaomicron | Bt OMV (a mean size of 237) | KGF-2 | Centrifugation | Genetic engineering | DSS colitis model | Reduced disease severity and promoted intestinal epithelial repair | Safe, relatively inexpensive and easy to produce, highly suitable for use in vaccines, can also overcome limitations associated with traditional OMV-related toxins, reducing the risk of side effects | [151] |
Tumor therapy
In recent years heterologous EVs have attracted significant attention in the field of cancer therapy. Among the heterologous EVs, animal- and plant-derived EVs have emerged as promising candidates for next-generation drug delivery vehicles due to unique biological properties and therapeutic potential. Specifically, milk EVs have gained widespread interest owing to excellent biocompatibility, intrinsic targeting ability, and scalability for large-scale production.
Samuel et al. [38] demonstrated that bovine milk EVs effectively induce cellular senescence and apoptosis in primary colorectal cancer cell lines (LIM1215 and SW620) through a series of in vitro and in vivo experiments (P < 0.01). Notably, milk EVs also uncovered a bidirectional regulatory effect of these EVs on tumor metastasis. While promoting the metastatic potential of pancreatic and breast cancer cells in the presence of a primary tumor, the EVs significantly suppress metastasis after surgical removal of the primary lesion. This discovery highlights the context-dependent functionality of milk EVs within the tumor microenvironment and offers new insights for designing postoperative strategies for metastatic cancer patients.
Wang et al. [99] developed an innovative milk EV-based carrier platform (i.e., the KME system) that enables the co-delivery of celastrol (CEL) and PD-L1 siRNA, aiming to synergize chemotherapy and immunotherapy. In a colorectal cancer mouse model, this system led to a 68.3% ± 5.7% reduction in tumor volume, demonstrating remarkable therapeutic efficacy and laying a solid foundation for the application of milk EVs in combined chemo-immunotherapy.
In addition to animal-derived vesicles, plant-derived EVs have also gained momentum as promising delivery vectors due to the abundant sources, oral bioavailability, and low immunogenicity. A recent study [100] reported that aloe vera–derived exosome-like vesicles treated with indocyanine green (ICG)-induced hypoosmotic stress exhibited an approximately 40% increase in mechanical strength, as measured by atomic force microscopy, significantly improving drug stability and bioactivity retention during transport. Furthermore, these vesicles showed high tumor-targeting efficiency in vivo, reaching 85.2% ± 3.1%, highlighting the potential of natural plant EVs for tumor-specific drug delivery.
In another study Yan et al. [101] (Figure 2A) successfully isolated exosome-like nanovesicles (BF-Exos) from the fruit of the traditional Chinese medicinal plant, Brucea javanica, for the first time. miRNA sequencing revealed that these vesicles are enriched in multiple anticancer bioactive components. In a triple-negative breast cancer mouse model, BF-Exos achieved a tumor growth inhibition rate of 72.4% ± 6.3% and reduced angiogenesis by 65.8% ± 5.2%, demonstrating potent antitumor and anti-angiogenic properties, and providing robust preclinical evidence for the therapeutic application of herbal EVs.
Figure 2 The application of exogenous EVs in tumor therapy. A. Brucea javanica-derived exosome-like nanovesicles deliver miRNAs for cancer therapy [101]. Copyright 2024, Elsevier B.V. All rights reserved. B. Platycodon grandiflorum-derived EVs suppress triple-negative breast cancer growth by reversing the immunosuppressive tumor microenvironment and modulating the gut microbiota [102]. Copyright 2025, The authors. C. Hybrid Ginseng-derived EVs-Like Particles with Autologous Tumor Cell Membrane for Personalized Vaccination to Inhibit Tumor Recurrence and Metastasis [103]. Copyright 2024, The authors. D. Enhanced tumor self-targeting of lemon-derived EVs by embedding homotypic cancer cell membranes for efficient drug delivery [104]. Copyright 2025, The authors. E. Bacterial protoplast-derived nanovesicles carrying CRISPR-Cas9 tools re-educate tumor-associated macrophages for enhanced cancer immunotherapy [105]. Copyright 2024, The author(s). F. Gut commensal Bifidobacterium-derived EVs modulate the therapeutic effects of anti-PD-1 in lung cancer [106]. Copyright 2025, The author(s).
Platycodon grandiflorum-derived EVs (PGEVs) have been shown to promote the polarization of tumor-associated macrophages (TAMs) toward the M1 phenotype, enhance the infiltration of cytotoxic T lymphocytes (CTLs) into tumor tissues, suppress PD-1 expression within the tumor microenvironment, and boost CTL-mediated cytotoxicity against tumor cells, thereby inhibiting tumor growth (Figure 2B) [102]. Moreover, PGEVs can improve gut microbiota composition by increasing microbial diversity and abundance in tumor-bearing mice, contributing to therapeutic outcomes by alleviating dysbiosis. Similarly, functional hybrid nanovesicles (HM-NPs), formed by fusing ginseng-derived extracellular vesicle-like particles (G-EVLPs) with membranes extracted from resected autologous tumor tissue, have been reported to enhance antigen-specific immune responses, effectively suppressing tumor recurrence and metastasis in subcutaneous and orthotopic tumor models (Figure 2C). [103]. In addition, vaccination with HM-NPs also induces long-term immune protection, leading to prolonged survival, while maintaining excellent biocompatibility. This personalized hybrid tumor vaccine platform based on G-EVLPs offers a promising strategy to prevent postoperative tumor recurrence and metastasis [103].
In another example, a novel nanoplatform (LEVBD) was developed by encapsulating membrane fragments from breast cancer cells within a scaffold of lemon-derived nanovesicles (LEVs). LEVBD demonstrates efficient and selective targeting to homologous tumor sites, even in the presence of competing heterologous tumors, significantly suppressing tumor growth without any observable systemic toxicity (Figure 2D) [104]. Furthermore, researchers have engineered an in vivo gene editing system that used bacteria-derived nanovesicles (NVs) to efficiently deliver CRISPR-Cas9 components [105]. This approach enables effective reprogramming of endogenous TAMs, producing notable anticancer effects without triggering systemic immune responses (Figure 2E).
Microbial-derived EVs also exhibit distinct advantages in modulating the tumor immune microenvironment. Recent findings showed that EVs from Bifidobacterium (Bif.BEVs) [106] can modulate the lung tumor microenvironment via the gut–lung axis (Figure 2F), enhancing the response rate to anti–PD-1 therapy from 35% to 62% (P < 0.001). This work provides valuable insight into the long-range regulatory mechanisms linking the gut microbiota to tumor immunity and paves the way for integrating microbiome-targeted interventions into cancer immunotherapy. Regulation of the tumor microenvironment has been widely studied and confirmed to be related to the overall condition of patients, such as muscle integrity and inflammation, which profoundly affects the prognosis and quality of life of patients. In addition, these studies have also opened up therapeutic targets for EVs, broadening the perspective for the exploration of EVs [107, 108].
Additionally, Qin et al. [109] engineered a probiotic-based delivery system (ENZC) that used dual nanobody technology to specifically recognize and bind tumor-derived exosomes (TDEs) via PD-L1 and CD9 antigens. In vivo experiments showed that this system could enhance CD8+ T cell infiltration by 3.2-fold and inhibit tumor metastasis with an efficiency of 78.5% ± 4.3%, demonstrating great promise for targeting tumor immune evasion mechanisms.
The above-mentioned examples vividly illustrate the potent utility of exogenous EVs in oncology, primarily leveraging the capabilities for targeted drug co-delivery, modulation of the immunosuppressive tumor microenvironment, and induction of anti-tumor immunity. In addition to the transformative impact in cancer, the inherent ability of EVs to shuttle bioactive molecules, such as growth factors, regulatory RNAs, and immunomodulatory signals, between cells positions EVs as equally compelling candidates for regenerative medicine. In this domain the focus shifts from combating malignant growth to actively promoting tissue repair, functional restoration, and constructive immune remodeling at sites of injury or degeneration.
Regenerative medicine
Exogenous EVs, due to efficient delivery of bioactive molecules, are emerging as promising therapeutic carriers in regenerative medicine. EVs orchestrate cellular proliferation, differentiation, and immune microenvironment remodeling at injury sites acting through the intercellular transfer of functional cargos, such as miRNAs, mRNAs, and cytokines, thereby promoting multidimensional tissue repair and regeneration.
Mesenchymal stem cell (MSC)-derived EVs, with potent osteoinductive properties, represent a highly promising acellular approach for bone defect repair [12]. Wang et al. engineered a biohybrid scaffold by combining decellularized fish scale scaffolds with osteogenically induced bone marrow MSC-EVs, achieving sustained EV release and promoting BMSC differentiation [110]. Similarly, Kang et al. developed an EV-functionalized scaffold composed of human MSC-EVs, magnesium ions, and gallic acid (GA) with a nanoscale interface that significantly enhanced osteogenesis [111]. Moreover, neutrophil-derived EVs have also been shown to accelerate bone regeneration by promoting BMSC osteogenic differentiation. Based on this finding, Wang et al. proposed the integration of these EVs with BMSC-based cell sheets to further enhance bone repair [112].
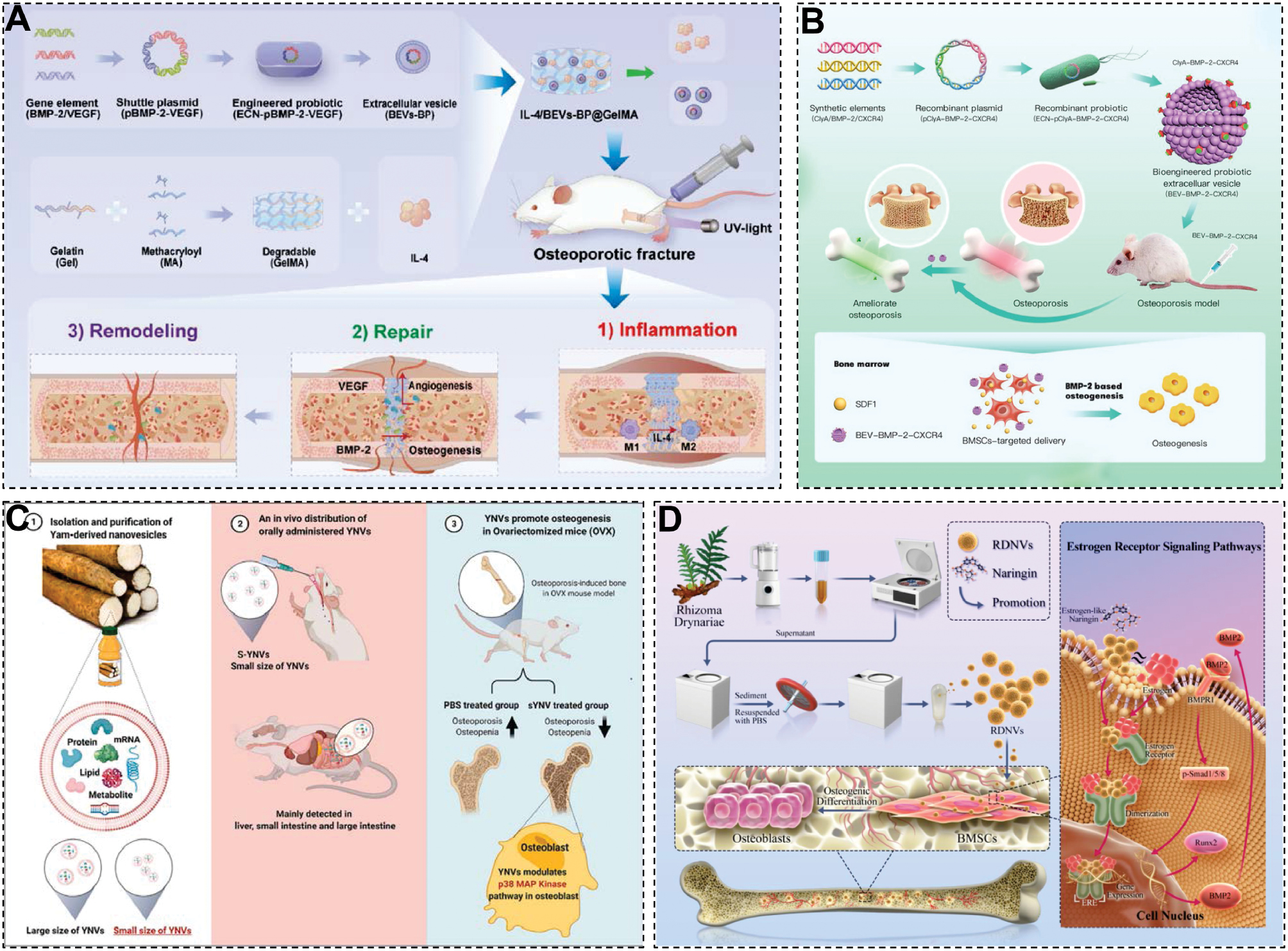
Another innovative approach involved encapsulating bacterial EVs loaded with BMP-2 and VEGF (BEVspBMP-2-VEGF) with IL-4 into GelMA hydrogels to create a multifunctional composite (IL-4/BEVspBMP-2-VEGF@GelMA) [113]. IL-4 reshaped the immune microenvironment by promoting macrophage polarization from pro-inflammatory M1 to reparative M2 phenotypes, while BEVspBMP-2-VEGF boosted BMP-2 and VEGF expression at fracture sites, thereby accelerating osteogenesis and angiogenesis. In vitro studies using RAW264.7 cells, BMSCs, and vascular endothelial cells (VECs) confirmed the composite anti-inflammatory, osteogenic, and pro-angiogenic effects. Furthermore, bone organoid models and in vivo studies in osteoporotic fracture (OPF) mouse models demonstrated that the composite significantly enhanced fracture healing by increasing M2 macrophage recruitment, osteogenic protein expression, and type-H vessel formation (Figure 3A) [113]. In parallel, an engineered bacterial EV platform was developed by displaying BMP-2 and CXCR4 proteins on the surface of E. coli, yielding BEVs-BC vesicles with excellent bone-targeting properties [114]. These engineered vesicles significantly enhanced BMSC osteogenic differentiation and effectively inhibited bone loss in ovariectomized (OVX) mouse models, thereby reversing osteoporosis progression (Figure 3B) [114].
Figure 3 The application of exogenous EVs in regenerative medicine. A. Synthetically engineered bacterial EVs and IL-4-encapsulated hydrogels sequentially promote osteoporotic fractures repair [113]. Copyright 2025, American Chemical Society. B. Synthetic biology-based bacterial EVs displaying BMP-2 and CXCR4 to ameliorate osteoporosis [114]. Copyright 2024 The authors. C. Yam-derived exosome-like nanovesicles stimulate osteoblast formation and prevent osteoporosis in mice [53]. Copyright 2023 Elsevier B.V. All rights reserved. D. Rhizoma drynariae-derived nanovesicles reverse osteoporosis by potentiating osteogenic differentiation of human bone marrow mesenchymal stem cells via targeting ERα signaling [115]. Copyright 2024 The authors.
Traditional Chinese medicine (TCM)-derived NVs are gaining attention in bone regeneration. For example [53], yam-derived exosome-like nanovesicles (YNVs) promote osteoblast growth, differentiation, and mineralization, mitigating bone fragility and improving bone regeneration in OVX-induced osteoporotic mice (Figure 3C) [53]. Similarly, RDNVs, NVs isolated from Drynaria fortunei root juice, exhibit strong bone-targeting activity and anti-osteoporotic effects in OVX mouse models [115]. RDNVs are efficiently internalized by human BMSCs (hBMSCs), which enhances the proliferation, ERα expression, osteogenic differentiation, and bone formation (Figure 3D) [115].
To summarize, regenerative medicine integrates biological and engineering strategies to repair or reconstruct damaged tissues and organs [116]. In addition to the differentiation potential, MSCs contribute significantly through the secretion of EVs, which deliver bioactive molecules that regulate tissue regeneration [117]. With continued advances in EV engineering and a deeper understanding of EV biology, tissue engineering and clinical therapy applications are expected to expand, accelerating the development and clinical translation of next-generation acellular regenerative therapies [118].
Inflammatory diseases
Inflammation is a key driver in diseases, like IBD, psoriasis, osteoarthritis (OA), and rheumatoid arthritis (RA). Chronic or excessive inflammation causes tissue damage and immune imbalance, making suppression of inflammation essential for therapy. Recently, EVs from animals, plants, and microbes have gained attention due to the biocompatibility, low immunogenicity, and efficient delivery of bioactive molecules.
IBD, including ulcerative colitis (UC) and Crohn’s disease (CD), is marked by impaired intestinal barriers and systemic release of pro-inflammatory cytokines that can trigger distant organ inflammation. Plant- and microbe-derived EVs have strong anti-inflammatory effects and restore gut homeostasis. For example, Lu et al. [109] isolated exosome-like NVs (PLDENs) from Pueraria lobata, which significantly reduced TNF-α, IL-6, and IL-1β (P < 0.01), restored barrier integrity, and suppressed lung inflammation in DSS-induced colitis. Hwang et al. [53] developed YNVs that inhibited TIMP2 signaling, decreased pro-inflammatory cytokines, and promoted beneficial gut bacteria, which lowered disease severity (P < 0.05). Wu et al. [67] demonstrated that Lactobacillus plantarum EVs improved epithelial cell survival, enhanced tight junction proteins, and reduced colonic inflammation by 60%, suppressing cytokine release.
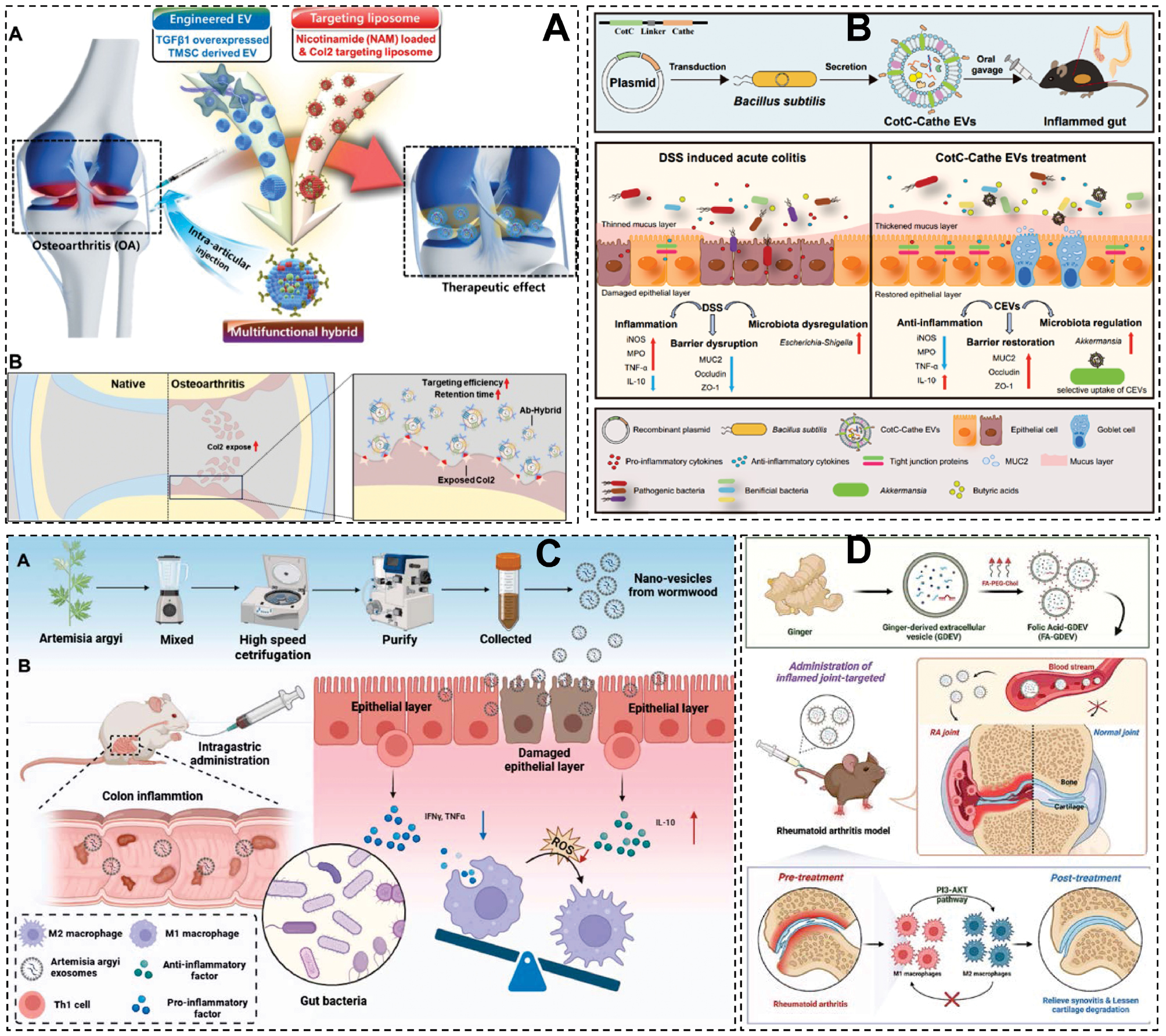
A research team has achieved a stable and efficient engineering method by hybridizing ethanol-mediated EVs with functionalized lipid nanoparticles (LNPs) containing polymeric denatured lipid components. During the hybridization process, the internal bioactive factors and targeted parts maintain the characteristics of LNPs and EVs. An Ab-hybrid was successfully synthesized by hybridization with nicotinamide encapsulated Col2A1 antibody-modified liposomes and EVs overexpressing transforming growth factor (TGF)-β1). The Ab-hybrid was used for surgical instability of the medial meniscus in rats induced by osteoarthritis (OA). Ultimately, the Ab-hybrid demonstrated excellent cartilage protection and anti-inflammatory effects in OA lesions with targeting and persistence (Figure 4A) [119]. In another innovative approach, researchers engineered Bacillus subtilis as a chassis organism to express the broad-spectrum antimicrobial peptide, cathelicidin-BF (Cathe), which was derived from the banded krait (Bungarus fasciatus; Figure 4B) [120]. Cathe was fused with the spore coat protein, CotC, and subsequently presented within B. subtilis-secreted EVs (CEVs) using surface display technology. These engineered CEVs demonstrated significant therapeutic efficacy in alleviating dextran sulfate sodium (DSS)-induced acute colitis. Folium Artemisiae Argyi-derived exosome-like NVs (FAELNs) have demonstrated promising therapeutic potential for UC (Figure 4C) [121] FAELNs reached peak fluorescence intensity in the mouse intestine within 2 h following oral administration and remained detectable for up to 24 h, predominantly localizing to the gastrointestinal tract and selectively accumulating at inflamed sites. In vitro studies revealed that FAELNs are internalized by cells through multiple complex pathways. FAELNs effectively reduce pro-inflammatory cytokine levels, promote macrophage polarization toward the M2 phenotype, scavenge reactive oxygen species (ROS), and modulate apoptosis. FAELNs significantly improved clinical indicators in UC mouse models, such as body weight, colon length, and disease activity index (DAI), while alleviating tissue inflammation and injury. Moreover, FAELNs are able to restore gut microbiota balance and exhibit no apparent cytotoxicity or systemic toxicity [121].
Figure 4 The application of exogenous EVs in Inflammatory diseases. A. The cartilage protection and anti-inflammatory effects of hybrid EVs in OA lesions [119] Copyright 2024, The authors. B. Genetically engineered Bacillus subtilis-derived EVs Alleviates Ulcerative Colitis by Restoring Intestinal Barrier and Regulating Gut Microbiota [120]. Copyright 2025, Elsevier B.V. C. Oral administration of Folium artemisiae Argyi-derived exosome-like nanovesicles can improve ulcerative colitis by regulating intestinal microorganisms [121]. Copyright 2025, Elsevier GmbH. D. Folic acid-modified ginger-derived EVs for targeted treatment of rheumatoid arthritis by remodeling immune microenvironment via the PI3K-AKT pathway [51]. Copyright 2025, The authors.
Psoriasis involves keratinocyte overgrowth and Th17 activation. MSC-derived small EVs (sEVs) have immunomodulatory effects but are costly. Plant- and microbe-derived EVs offer affordable alternatives. Huang et al. [122] fused grapefruit EVs with gingival MSC membranes to create CCR6-targeting NVs carrying CX5461. These vesicles cut epidermal thickness by 70% and reduced inflammation in a psoriasis mouse model (P < 0.001). The same group used Parabacteroides goldsteinii OMVs to inhibit the IL-23/Th17 axis, improving PASI scores without side effects [100].
Non-animal EVs also show promise in joint diseases. Spirulina platensis EVs (SP-EVs), which are rich in antioxidants, were loaded into a hydrogel (Rh Gel@SP-EVs) that scavenged ROS and boosted chondrocyte metabolism. This system halved cartilage degradation and reduced IL-1β in an OA rat model (P < 0.01) [123]. Another research team discovered that folic acid-modified ginger-derived extracellular vesicles (FA-GDEVs) selectively target M1 macrophages in inflamed joints via folate receptors (FRs). FA-GDEVs promote the polarization of macrophages toward a reparative M2 phenotype by modulating the PI3K-AKT signaling pathway in vitro. In vivo studies further demonstrated that FA-GDEVs not only effectively accumulate in affected joints but also significantly alleviate RA symptoms. Based on these findings, the authors proposed that FA-GDEVs represent a promising therapeutic candidate for RA treatment (Figure 4D) [51].
Exogenous EVs combine natural targeting, low toxicity, and scalability but face challenges, including standardized production, long-term safety assessment, and mass manufacturing. With ongoing advances, these EVs could revolutionize inflammation-targeted therapies and precision medicine [124].
Challenges of clinical transformation
EVs have demonstrated broad application prospects in various biomedical fields. EVs have emerged as a promising platform in precision medicine research due to excellent biocompatibility, low immunogenicity, and ability to traverse biological barriers. Nevertheless, the translation of EVs from basic research to clinical application is hindered by several critical challenges.
Currently, several challenges dominate the landscape of EV research and application. First, there is limited yield [12]. Existing EV isolation and extraction techniques, such as ultracentrifugation and size-exclusion chromatography, are inefficient and fail to meet the demands of large-scale clinical production. Second, maintenance of bioactivity and in vivo stability [12]. EVs are rapidly cleared by the mononuclear phagocyte system, particularly in the liver and lungs, resulting in a short circulation half-life. Moreover, potential structural damage during storage or delivery may compromise the functionality and the underlying mechanisms remain poorly understood, which adds further uncertainty to the therapeutic use. Third, source heterogeneity and purity control [1], especially analytical gaps in non-mammalian EV purity. EVs derived from various biological fluids exhibit significant variability in composition and function. Current separation technologies often struggle to achieve high purity and uniformity, which impedes the development of standardized EV-based drug delivery systems. Critical deficiencies exist in verifying plant-derived EVs purity, particularly in discriminating vesicles from plant-specific contaminants. Although protocols for high-purity Arabidopsis EV isolation exist [125], most studies fail to exclude contaminants, obscuring functional interpretations. Standardized validation methods targeting phyto-contaminants are urgently needed [48, 126]. Fourth, while several guidelines, such as the updated MISEV and platforms, like EV-TRACK, have been developed to facilitate reproducibility, these protocols frequently neglect the detailed characterization of immunogenic profiles, which is of crucial importance given that subtle alterations in EV composition could significantly influence immune recognition and clearance [105, 127, 128]. Fifth, although EVs are generally regarded as having low immunogenicity, repeated dosing in preclinical models has been associated with the development of anti-EV IgG antibodies. These antibodies, once present, can opsonize subsequently administered EVs and enhance clearance from the circulation, thereby reducing therapeutic efficacy over time. In view of the above challenges, standardization of isolation, loading, and characterization protocols is necessary to progress toward clinical translation.
Despite these limitations, EVs continue to demonstrate unique advantages as highly efficient and biocompatible drug carriers. Preclinical studies have reported negligible toxicity of EVs in in vitro and in vivo environments (Table 2), reinforcing the safety profile for therapeutic applications [89]. With ongoing advancements in EV-related research and continuous refinement of associated technologies, there is increasing optimism about the future clinical translation of EVs.
Table 2 Clinical Trials of Exogenous EVs for Disease Treatment.a
| ID | Disease | Mode | Intervention | Enrollment | Sponsor | Phase |
|---|---|---|---|---|---|---|
| NCT04493242 | ARDS | Treatment | Biological: ExoFlo Other: Intravenous normal saline |
102 | Direct Biologics, LLC | II |
| NCT05774509 | Non-ischemic Cardiomyopathies | Treatment | Biological: Extracellular vesicle-enriched secretome of cardiovascular progenitor cells differentiated from induced pluripotent stem cells | 12 | Assistance Pulique-Hôpitaux de Paris | I |
| NCT05787288 | COVID-19 | Treatment | Biological: Extracellular vesicles from mesenchymal stem cells | 240 | First Affiliated Hospital of Wenzhou Medical University | I |
| NCT06002841 | Acute respiratory failure | Treatment | Biological: intravenous treatment with EVs Biological: intravenous treatment with placebo solution |
15 | D’Or Institute for Research and Education | I II |
| NCT06242379 | Retinitis pigmentosa | Treatment | Biological: GMP compliant-BM-MSC derived sEVs | 15 | Mahidol University | I II |
| NCT06825572 | Acute/acute-on-chronic Liver failure after liver transplantation | Treatment | Biological: MSC-EVs | 30 | Third Affiliated Hospital, Sun Yat-Sen University | I |
aAll data from NIH (website: https://clinicaltrials.gov/). ARDS: acute respiratory distress syndrome.
Summary and outlook
EVs, as natural carriers of intercellular communication, offer a novel paradigm for disease treatment and regenerative medicine owing to diverse biological origins and unique structural features [21]. Studies have demonstrated that EVs can mediate inter-species and -organ communication by transporting bioactive molecules, such as proteins, lipids, and nucleic acids. For example, milk EVs modulate gut immune homeostasis via microRNA signaling, plant-derived EVs deliver flavonoids and other metabolites to host cells and facilitate antioxidant responses, and microbe-derived EVs can present antigens to activate immune responses within host–microbiota interactions [2]. These findings provide a theoretical foundation for developing EV-based therapeutics from non-mammalian sources.
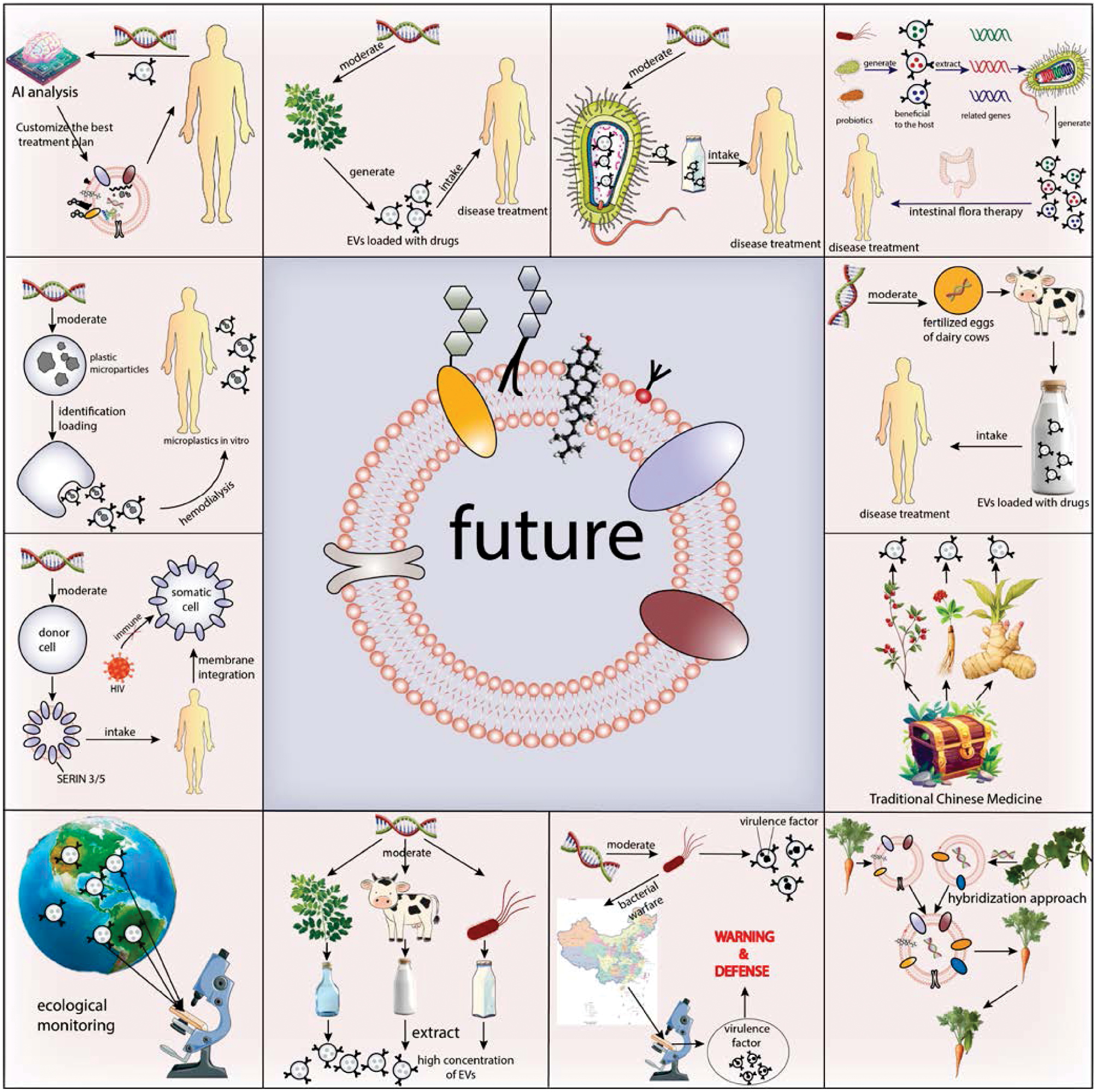
Notably, plant factories and in vitro tissue culture systems are emerging as promising platforms for the large-scale production of plant-derived EVs. These systems offer standardized growth conditions and EV-inducing strategies, enabling sustainable production of EVs with high purity and low cost [129]. In parallel, the rapid advancement of synthetic biology is providing new technological avenues for the functional design and controlled release of EVs. By engineering donor cells, it is now possible to precisely regulate EV biosynthesis pathways by introducing synthetic regulatory elements to enhance the encapsulation of specific miRNAs or by modulating EV secretion via artificial signaling circuits with spatiotemporal specificity [130]. Artificial intelligence (AI) and machine learning are also reshaping EV research by enabling high-throughput data integration, molecular “fingerprint” identification, disease-associated pattern recognition, and personalized EV design. These computational tools will accelerate the translation of laboratory discoveries into clinically viable products [131].
In the future, the application of exogenous EVs is expected to be revolutionized by the integration of AI and synthetic biology (Figure 5). By leveraging AI to analyze a patient’s genomic information and EV membrane architecture, it will be possible to identify the optimal lipid composition, membrane proteins, and bioactive cargo, thereby enabling the design of fully customized, high-performance therapeutic EVs for truly personalized medicine [9]. Furthermore, by introducing specifically engineered DNA into plants or microbial cell factories, these organisms could be programmed to naturally produce medicinally active EVs during growth or metabolism. Such an approach would allow patients to achieve therapeutic effects through simple ingestion or administration, dramatically reducing production costs and improving accessibility. In addition, EVs hold great promise as sensitive and highly specific biomarkers for the early diagnosis and risk stratification of major diseases, paving the way for a paradigm shift in healthcare-from treatment to a comprehensive “prediction–prevention–intervention” model.
Figure 5 The outlook of exogenous EVs in translational medicine. Created with the Adobe Illustrator. Map Review Number for the Map of China: GS (2022) No. 4308.
Author contributions
Muran Bai, Zhongshi Li, and Tianren Shi contributed equally. They are contributors in writing the manuscript and responsible for the visualization. Xiangling Li, Jia Li, Jian Ma, and Lei Huang are responsible for review the manuscript. Zhijin Fan is responsible for conceptualization and review the manuscript. All authors read and approved the final manuscript.
Acknowledgment
This work was supported by the National Natural Science Foundation of China (82002253). Scheme 1 and Figures 1 & 5 were created using the Adobe Illustrator software.
Conflict of interest
Zhijin Fan is an Editorial Board Member of BIO Integration. He was not involved in the peer-review or handling of the manuscript. The other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Graphical abstract
Highlights
- This review summarizes exogenous extracellular vesicles (EVs) from milk, plants, and microbes, covering their structure, uptake, and roles in intercellular communication.
- Their unique advantages (low immunogenicity, biocompatibility) and challenges (standardization, loading efficiency) for clinical use are analyzed.
- It also details applications in cancer therapy, regenerative medicine, and inflammatory diseases with preclinical/clinical evidence.
- Future directions via AI and synthetic biology for personalized EV-based precision therapy are discussed.
In brief
This review focuses on exogenous EVs as translational medicine platforms. It explores their sources, biological properties, and roles in disease modulation, highlights applications in oncology, regeneration, and inflammation, and addresses clinical translation challenges, envisioning AI-driven personalized EV therapies.
References
- Gurunathan S, Kang M-H, Jeyaraj M, Qasim M, Kim J-H. Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes. Cells 2019;8(4):307. [PMID: 30987213 DOI: 10.3390/cells8040307]
- Li X, Corbett AL, Taatizadeh E, Tasnim N, Little JP, et al. Challenges and opportunities in exosome research—perspectives from biology, engineering, and cancer therapy. APL Bioeng 2019;3(1):011503. [PMID: 31069333 DOI: 10.1063/1.5087122]
- Dooley K, McConnell RE, Xu K, Lewis ND, Haupt S, et al. A versatile platform for generating engineered extracellular vesicles with defined therapeutic properties. Mol Ther 2021;29(5):1729-43. [PMID: 33484965 DOI: 10.1016/j.ymthe.2021.01.020]
- Ye Y, Liu Y, Ma S, Li X, Wang W, et al. Multifunctional DNA hydrogels with light-triggered gas-therapy and controlled G-Exos release for infected wound healing. Bioact Mater 2025;52:422-37. [PMID: 40585387 DOI: 10.1016/j.bioactmat.2025.06.004]
- Dad HA, Gu T-W, Zhu A-Q, Huang L-Q, Peng L-H. Plant exosome-like nanovesicles: emerging therapeutics and drug delivery nanoplatforms. Mol Ther 2021;29(1):13-31. [PMID: 33278566 DOI: 10.1016/j.ymthe.2020.11.030]
- Cheng L, Hill AF. Therapeutically harnessing extracellular vesicles. Nat Rev Drug Discov 2022;21(5):379-99. [PMID: 35236964 DOI: 10.1038/s41573-022-00410-w]
- Ong SL, Blenkiron C, Haines S, Acevedo-Fani A, Leite JAS, et al. Ruminant milk-derived extracellular vesicles: a nutritional and therapeutic opportunity? Nutrients 2021;13(8):2505. [PMID: 34444665 DOI: 10.3390/nu13082505]
- Chen W, Chen X, Qian Y, Wang X, Zhou Y, et al. Lipidomic profiling of human milk derived exosomes and their emerging roles in the prevention of necrotizing enterocolitis. Mol Nutr Food Res 2021;65(10):e2000845. [PMID: 33715285 DOI: 10.1002/mnfr.202000845]
- Lucchetti D, Fattorossi A, Sgambato A. Extracellular vesicles in oncology: progress and pitfalls in the methods of isolation and analysis. Biotechnol J 2019;14(1):e1700716. [PMID: 29878510 DOI: 10.1002/biot.201700716]
- Hoshino A, Kim HS, Bojmar L, Gyan KE, Cioffi M, et al. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell 2020;182(4):1044-61.e18. [PMID: 32795414 DOI: 10.1016/j.cell.2020.07.009]
- Liu S, Chen L, Guo M, Li Y, Liu Q, et al. Targeted delivery of engineered RVG-BDNF-exosomes: a novel neurobiological approach for ameliorating depression and regulating neurogenesis. Research 2024;7:0402. [DOI: 10.34133/research.0402]
- Yang C, Xue Y, Duan Y, Mao C, Wan M. Extracellular vesicles and their engineering strategies, delivery systems, and biomedical applications. J Control Release 2024;365:1089-123. [PMID: 38065416 DOI: 10.1016/j.jconrel.2023.11.057]
- Couch Y, Buzàs EI, Di Vizio D, Gho YS, Harrison P, et al. A brief history of nearly EV-erything – the rise and rise of extracellular vesicles. J Extracell Vesicles 2021;10(14):e12144. [PMID: 34919343 DOI: 10.1002/jev2.12144]
- Maas SLN, Breakefield XO, Weaver AM. Extracellular vesicles: unique intercellular delivery vehicles. Trends Cell Biol 2017;27(3):172-88. [PMID: 27979573 DOI: 10.1016/j.tcb.2016.11.003]
- Gandham S, Su X, Wood J, Nocera AL, Alli SC, et al. Technologies and standardization in research on extracellular vesicles. Trends Biotechnol 2020;38(10):1066-98. [PMID: 32564882 DOI: 10.1016/j.tibtech.2020.05.012]
- van der Koog L, Gandek TB, Nagelkerke A. Liposomes and extracellular vesicles as drug delivery systems: a comparison of composition, pharmacokinetics, and functionalization. Adv Healthc Mater 2022;11(5):e2100639. [PMID: 34165909 DOI: 10.1002/adhm.202100639]
- Greening DW, Simpson RJ. Understanding extracellular vesicle diversity – current status. Expert Rev Proteomics 2018;15(11):887-910. [PMID: 30326765 DOI: 10.1080/14789450.2018.1537788]
- Kalra H, Drummen GPC, Mathivanan S. Focus on extracellular vesicles: introducing the next small big thing. Int J Mol Sci 2016;17(2):170. [PMID: 26861301 DOI: 10.3390/ijms17020170]
- Abels ER, Breakefield XO. Introduction to extracellular vesicles: biogenesis, RNA cargo selection, content, release, and uptake. Cell Mol Neurobiol 2016;36(3):301-12. [PMID: 27053351 DOI: 10.1007/s10571-016-0366-z]
- Greening DW, Xu R, Gopal SK, Rai A, Simpson RJ. Proteomic insights into extracellular vesicle biology – defining exosomes and shed microvesicles. Expert Rev Proteomics 2017;14(1):69-95. [PMID: 27838931 DOI: 10.1080/14789450.2017.1260450]
- Di Bella MA. Overview and update on extracellular vesicles: considerations on exosomes and their application in modern medicine. Biology (Basel) 2022;11(6):804. [PMID: 35741325 DOI: 10.3390/biology11060804]
- Lener T, Gimona M, Aigner L, Börger V, Buzas E, et al. Applying extracellular vesicles based therapeutics in clinical trials – an ISEV position paper. J Extracell Vesicles 2015;4:30087. [PMID: 26725829 DOI: 10.3402/jev.v4.30087]
- Pan BT, Johnstone RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell 1983;33(3):967-78. [PMID: 6307529 DOI: 10.1016/0092-8674(83)90040-5]
- Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 2013;200(4):373-83. [PMID: 23420871 DOI: 10.1083/jcb.201211138]
- Trams EG, Lauter CJ, Salem N Jr, Heine U. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim Biophys Acta 1981;6;645(1):63-70. [PMID: 6266476 DOI: 10.1016/0005-2736(81)90512-5.
- Yan H, Li Y, Cheng S, Zeng Y. Advances in analytical technologies for extracellular vesicles. Anal Chem 2021;93(11):4739-74. [PMID: 33635060 DOI: 10.1021/acs.analchem.1c00693]
- Gangadaran P, Ahn B-C. Extracellular vesicle- and extracellular vesicle mimetics-based drug delivery systems: new perspectives, challenges, and clinical developments. Pharmaceutics 2020;12(5):442. [PMID: 32403320 DOI: 10.3390/pharmaceutics12050442]
- De Sousa KP, Rossi I, Abdullahi M, Ramirez MI, Stratton D, et al. Isolation and characterization of extracellular vesicles and future directions in diagnosis and therapy. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2023;15(1):e1835. [PMID: 35898167 DOI: 10.1002/wnan.1835]
- Kim DK, Lee J, Kim SR, Choi DS, Yoon YJ, et al. EVpedia: a community web portal for extracellular vesicles research. Bioinformatics 2014;31(6):933-9. DOI: 10.1093/bioinformatics/btu741
- Barathan M, Ng SL, Lokanathan Y, Ng MH, Law JX. Milk-derived extracellular vesicles: a novel perspective on comparative therapeutics and targeted nanocarrier application. Vaccines (Basel) 2024;12(11):1282. [PMID: 39591185 DOI: 10.3390/vaccines12111282]
- Sanwlani R, Fonseka P, Chitti SV, Mathivanan S. Milk-derived extracellular vesicles in inter-organism, cross-species communication and drug delivery. Proteomes 2020;8(2):11. [PMID: 32414045 DOI: 10.3390/proteomes8020011]
- Galley JD, Besner GE. The therapeutic potential of breast milk-derived extracellular vesicles. Nutrients 2020;12(3):745. [PMID: 32168961 DOI: 10.3390/nu12030745]
- Jiang X, You L, Zhang Z, Cui X, Zhong H, et al. Biological properties of milk-derived extracellular vesicles and their physiological functions in infant. Front Cell Dev Biol 2021;9:693534. [PMID: 34249944 DOI: 10.3389/fcell.2021.693534]
- Chutipongtanate S, Morrow AL, Newburg DS. Human milk extracellular vesicles: a biological system with clinical implications. Cells 2022;11(15):2345. [PMID: 35954189 DOI: 10.3390/cells11152345]
- Kim N-H, Kim J, Lee J-Y, Bae H-A, Kim CY. Application of milk exosomes for musculoskeletal health: talking points in recent outcomes. Nutrients 2023;15(21):4645. [PMID: 37960298 DOI: 10.3390/nu15214645]
- Di S, Huang Y, Qiao W, Zhang X, Wang Y, et al. Advances in the isolation and characterization of milk-derived extracellular vesicles and their functions. Front Nutr 2024;11:1512939. [PMID: 39742102 DOI: 10.3389/fnut.2024.1512939]
- Feng X, Chen X, Zheng X, Zhu H, Qi Q, et al. Latest trend of milk derived exosomes: cargos, functions, and applications. Front Nutr 2021;8:747294. [PMID: 34778341 DOI: 10.3389/fnut.2021.747294]
- Samuel M, Fonseka P, Sanwlani R, Gangoda L, Chee SH, et al. Oral administration of bovine milk-derived extracellular vesicles induces senescence in the primary tumor but accelerates cancer metastasis. Nat Commun 2021;12(1):3950. [PMID: 34168137 DOI: 10.1038/s41467-021-24273-8]
- Melnik BC, Schmitz G. Exosomes of pasteurized milk: potential pathogens of Western diseases. J Transl Med 2019;17(1):3. [PMID: 30602375 DOI: 10.1186/s12967-018-1760-8]
- Liu W, Du C, Nan L, Li C, Wang H, et al. The difference of milk-derived extracellular vesicles from cow colostrum and mature milk on miRNAs expression and protecting intestinal epithelial cells against lipopolysaccharide damage. Int J Mol Sci 2024;25(7):3880. [PMID: 38612689 DOI: 10.3390/ijms25073880]
- Melnik BC, Stremmel W, Weiskirchen R, John SM, Schmitz G. Exosome-derived MicroRNAs of human milk and their effects on infant health and development. Biomolecules 2021;11(6):851. [PMID: 34200323 DOI: 10.3390/biom11060851]
- Papakonstantinou E, Dragoumani K, Mitsis T, Chrousos GP, Vlachakis D. Milk exosomes and a new way of communication between mother and child. EMBnet J 2024;29:e1050. [PMID: 38845751 DOI: 10.14806/ej.29.0.1050]
- Prasadani M, Kodithuwakku S, Pennarossa G, Fazeli A, Brevini TAL. Therapeutic potential of bovine milk-derived extracellular vesicles. Int J Mol Sci 2024;25(10):5543. [PMID: 38791583 DOI: 10.3390/ijms25105543]
- Yang C, Zhang W, Bai M, Luo Q, Zheng Q, et al. Edible plant-derived extracellular vesicles serve as promising therapeutic systems. Nano TransMed 2023;2(2-3):100004. [DOI: 10.1016/j.ntm.2023.100004]
- Cai Q, Halilovic L, Shi T, Chen A, He B, et al. Extracellular vesicles: cross-organismal RNA trafficking in plants, microbes, and mammalian cells. Extracell Vesicles Circ Nucl Acids 2023;4(2):262-82. [PMID: 37575974 DOI: 10.20517/evcna.2023.10]
- Cui Y, Gao J, He Y, Jiang L. Plant extracellular vesicles. Protoplasma 2020;257(1):3-12. [PMID: 31468195 DOI: 10.1007/s00709-019-01435-6]
- Samuel M, Bleackley M, Anderson M, Mathivanan S. Extracellular vesicles including exosomes in cross kingdom regulation: a viewpoint from plant-fungal interactions. Front Plant Sci 2015;6:766. [PMID: 26442078 DOI: 10.3389/fpls.2015.00766]
- Regente M, Pinedo M, San Clemente H, Balliau T, Jamet E, et al. Plant extracellular vesicles are incorporated by a fungal pathogen and inhibit its growth. J Exp Bot 2017;68(20):5485-95. [PMID: 29145622 DOI: 10.1093/jxb/erx355]
- Urzì O, Gasparro R, Ganji NR, Alessandro R, Raimondo S. Plant-RNA in extracellular vesicles: the secret of cross-kingdom communication. Membranes (Basel) 2022;12(4):352. [PMID: 35448322 DOI: 10.3390/membranes12040352]
- Gao B, Huang X, Fu J, Chen L, Deng Z, et al. Oral administration of Momordica charantia-derived extracellular vesicles alleviates ulcerative colitis through comprehensive renovation of the intestinal microenvironment. J Nanobiotechnology 2025;23(1):261. [PMID: 40170075 DOI: 10.1186/s12951-025-03346-6]
- Han R, Zhou D, Ji N, Yin Z, Wang J, et al. Folic acid-modified ginger-derived extracellular vesicles for targeted treatment of rheumatoid arthritis by remodeling immune microenvironment via the PI3K-AKT pathway. J Nanobiotechnology 2025;23(1):41. [PMID: 39849554 DOI: 10.1186/s12951-025-03096-5]
- Fu J, Liu Z, Feng Z, Huang J, Shi J, et al. Platycodon grandiflorum exosome-like nanoparticles: the material basis of fresh platycodon grandiflorum optimality and its mechanism in regulating acute lung injury. J Nanobiotechnology 2025;23(1):270. [PMID: 40186259 DOI: 10.1186/s12951-025-03331-z]
- Hwang J-H, Park Y-S, Kim H-S, Kim D-H, Lee S-H, et al. Yam-derived exosome-like nanovesicles stimulate osteoblast formation and prevent osteoporosis in mice. J Control Release 2023;355:184-98. [PMID: 36736431 DOI: 10.1016/j.jconrel.2023.01.071]
- Xu S, Zhang Z, Zhou X, Liao Y, Peng Z, et al. Gouqi-derived Nanovesicles (GqDNVs) promoted MC3T3-E1 cells proliferation and improve fracture healing. Phytomedicine 2025;142:156755. [PMID: 40252435 DOI: 10.1016/j.phymed.2025.156755]
- Woith E, Fuhrmann G, Melzig MF. Extracellular vesicles-connecting kingdoms. Int J Mol Sci 2019;20(22):5695. [PMID: 31739393 DOI: 10.3390/ijms20225695]
- Jimenez-Jimenez S, Hashimoto K, Santana O, Aguirre J, Kuchitsu K, et al. Emerging roles of tetraspanins in plant inter-cellular and inter-kingdom communication. Plant Signal Behav 2019;14(4):e1581559. [PMID: 30829110 DOI: 10.1080/15592324.2019.1581559]
- Liu G, Kang G, Wang S, Huang Y, Cai Q. Extracellular vesicles: emerging players in plant defense against pathogens. Front Plant Sci 2021;12:757925. [PMID: 34659325 DOI: 10.3389/fpls.2021.757925]
- Cuesta CM, Guerri C, Ureña J, Pascual M. Role of microbiota-derived extracellular vesicles in gut-brain communication. Int J Mol Sci 2021;22(8):4235. [PMID: 33921831 DOI: 10.3390/ijms22084235]
- Diaz-Garrido N, Badia J, Baldomà L. Modulation of dendritic cells by microbiota extracellular vesicles influences the cytokine profile and exosome cargo. Nutrients 2022;14(2):344. [PMID: 35057528 DOI: 10.3390/nu14020344]
- Badia J, Baldomà L. Membrane vesicles from the gut microbiota and their interactions with the host. In: Kaparakis-Liaskos M, Kufer TA, editors. Bacterial membrane vesicles: biogenesis, functions and applications. Cham: Springer International Publishing; 2020. pp. 189-217.
- Díaz-Garrido N, Badia J, Baldomà L. Microbiota-derived extracellular vesicles in interkingdom communication in the gut. J Extracell Vesicles 2021;10(13):e12161. [PMID: 34738337 DOI: 10.1002/jev2.12161]
- Chang X, Wang S-L, Zhao S-B, Shi Y-H, Pan P, et al. Extracellular vesicles with possible roles in gut intestinal tract homeostasis and IBD. Mediators Inflamm 2020;2020:1945832. [PMID: 32410847 DOI: 10.1155/2020/1945832]
- Ñahui Palomino RA, Vanpouille C, Costantini PE, Margolis L. Microbiota-host communications: bacterial extracellular vesicles as a common language. PLoS Pathog 2021;17(5):e1009508. [PMID: 33984071 DOI: 10.1371/journal.ppat.1009508]
- Macia L, Nanan R, Hosseini-Beheshti E, Grau GE. Host- and microbiota-derived extracellular vesicles, immune function, and disease development. Int J Mol Sci 2019;21(1):107. [PMID: 31877909 DOI: 10.3390/ijms21010107]
- Díez-Sainz E, Milagro FI, Riezu-Boj JI, Lorente-Cebrián S. Effects of gut microbiota-derived extracellular vesicles on obesity and diabetes and their potential modulation through diet. J Physiol Biochem 2022;78(2):485-99. [PMID: 34472032 DOI: 10.1007/s13105-021-00837-6]
- Liang X, Dai N, Sheng K, Lu H, Wang J, et al. Gut bacterial extracellular vesicles: important players in regulating intestinal microenvironment. Gut Microbes 2022;14(1):2134689. [PMID: 36242585 DOI: 10.1080/19490976.2022.2134689]
- Wu Y, Huang X, Li Q, Yang C, Huang X, et al. Reducing severity of inflammatory bowel disease through colonization of Lactiplantibacillus plantarum and its extracellular vesicles release. J Nanobiotechnology 2025;23(1):227. [PMID: 40114208 DOI: 10.1186/s12951-025-03280-7]
- Sartorio MG, Pardue EJ, Feldman MF, Haurat MF. Bacterial outer membrane vesicles: from discovery to applications. Annu Rev Microbiol 2021;75:609-30. [PMID: 34351789 DOI: 10.1146/annurev-micro-052821-031444]
- Sultan S, Mottawea W, Yeo J, Hammami R. Gut microbiota extracellular vesicles as signaling molecules mediating host-microbiota communications. Int J Mol Sci 2021;22(23):13166. [PMID: 34884969 DOI: 10.3390/ijms222313166]
- Pirolli NH, Bentley WE, Jay SM. Bacterial extracellular vesicles and the gut-microbiota brain axis: emerging roles in communication and potential as therapeutics. Adv Biol (Weinh) 2021;5(7):e2000540. [PMID: 33857347 DOI: 10.1002/adbi.202000540]
- Marquez-Paradas E, Torrecillas-Lopez M, Barrera-Chamorro L, Del Rio-Vazquez JL, Gonzalez-de la Rosa T, et al. Microbiota-derived extracellular vesicles: current knowledge, gaps, and challenges in precision nutrition. Front Immunol 2025;16:1514726. [PMID: 40051622 DOI: 10.3389/fimmu.2025.1514726]
- Tsatsaronis JA, Franch-Arroyo S, Resch U, Charpentier E. Extracellular vesicle RNA: a universal mediator of microbial communication? Trends Microbiol 2018;26(5):401-10. [PMID: 29548832 DOI: 10.1016/j.tim.2018.02.009]
- Stanton BA. Extracellular vesicles and host-pathogen interactions: a review of inter-kingdom signaling by small noncoding RNA. Genes (Basel) 2021;12(7):1010. [PMID: 34208860 DOI: 10.3390/genes12071010]
- Marsh SR, Beard CE, Gourdie RG. Milk extracellular vesicles: a burgeoning new presence in nutraceuticals and drug delivery. Bioeng Transl Med 2025;10(3):e10756. [PMID: 40385535 DOI: 10.1002/btm2.10756]
- Babaker MA, Aljoud FA, Alkhilaiwi F, Algarni A, Ahmed A, et al. The therapeutic potential of milk extracellular vesicles on colorectal cancer. Int J Mol Sci 2022;23(12):6812. [PMID: 35743255 DOI: 10.3390/ijms23126812]
- Turner N, Abeysinghe P, Peiris H, Vaswani K, Sadowski P, et al. Proteomic profiling of plasma-derived small extracellular vesicles: a novel tool for understanding the systemic effects of tick burden in cattle. J Anim Sci 2022;100(2):skac015. [PMID: 35045163 DOI: 10.1093/jas/skac015]
- Çelik E, Cemali Ö, Şahin TÖ, Deveci G, Biçer NÇ, et al. Human breast milk exosomes: affecting factors, their possible health outcomes, and future directions in dietetics. Nutrients 2024;16(20):3519. [PMID: 39458514 DOI: 10.3390/nu16203519]
- Ginini L, Billan S, Fridman E, Gil Z. Insight into extracellular vesicle-cell communication: from cell recognition to intracellular fate. Cells 2022;11(9):1375. [PMID: 35563681 DOI: 10.3390/cells11091375]
- Marl A. Developing a scalable isolation process for extracellular vesicles from human milk bacteria and food derived sources. Paris-Lodron: University of Salzburg; 2024.
- Alzahrani FA, Khan MI, Kameli N, Alsahafi E, Riza YM. Plant-derived extracellular vesicles and their exciting potential as the future of next-generation drug delivery. Biomolecules 2023;13(5):839. [PMID: 37238708 DOI: 10.3390/biom13050839]
- Thakur A, Rai D. Global requirements for manufacturing and validation of clinical grade extracellular vesicles. J Liq Biopsy 2024;6:100278. [PMID: 40027307 DOI: 10.1016/j.jlb.2024.100278]
- Lai JJ, Hill JJ, Huang CY, Lee GC, Mai KW, et al. Unveiling the complex world of extracellular vesicles: novel characterization techniques and manufacturing considerations. Chonnam Med J 2024;60(1):1-12. [PMID: 38304124 DOI: 10.4068/cmj.2024.60.1.1]
- Malsy M, Bitzinger D, Graf B, Bundscherer A. Staurosporine induces apoptosis in pancreatic carcinoma cells PaTu 8988t and Panc-1 via the intrinsic signaling pathway. Eur J Med Res 2019;24(1):5. [PMID: 30686270 DOI: 10.1186/s40001-019-0365-x]
- Jurgielewicz BJ, Yao Y, Stice SL. Kinetics and specificity of HEK293T extracellular vesicle uptake using imaging flow cytometry. Nanoscale Res Lett 2020;15(1):170. [PMID: 32833066 DOI: 10.1186/s11671-020-03399-6]
- Zhu X, Badawi M, Pomeroy S, Sutaria DS, Xie Z, et al. Comprehensive toxicity and immunogenicity studies reveal minimal effects in mice following sustained dosing of extracellular vesicles derived from HEK293T cells. J Extracell Vesicles 2017;6(1):1324730. [PMID: 28717420 DOI: 10.1080/20013078.2017.1324730]
- Nairz M, Weiss G. Iron in infection and immunity. Mol Aspects Med 2020;75:100864. [PMID: 32461004 DOI: 10.1016/j.mam.2020.100864]
- Ganz T, Nemeth E. Iron homeostasis in host defence and inflammation. Nat Rev Immunol 2015;15(8):500-10. [PMID: 26160612 DOI: 10.1038/nri3863]
- Pinheiro da Silva F, Nizet V. Cell death during sepsis: integration of disintegration in the inflammatory response to overwhelming infection. Apoptosis 2009;14(4):509-21. [PMID: 19199035 DOI: 10.1007/s10495-009-0320-3]
- Saleh AF, Lázaro-Ibáñez E, Forsgard M-M, Shatnyeva O, Osteikoetxea X, et al. Extracellular vesicles induce minimal hepatotoxicity and immunogenicity. Nanoscale 2019;11(14):6990-7001. [PMID: 30916672 DOI: 10.1039/c8nr08720b]
- Olsson KS, Weinfeld A. Availability of iron dextran for hemoglobin synthesis as studied with phlebotomy. Acta Med Scand 1972;192(6):543-9. [PMID: 4565624 DOI: 10.1111/j.0954-6820.1972.tb04864.x]
- Kuipers ME, Hokke CH, Smits HH, Nolte-’t Hoen ENM. Pathogen-derived extracellular vesicle-associated molecules that affect the host immune system: an overview. Front Microbiol 2018;9:2182. [PMID: 30258429 DOI: 10.3389/fmicb.2018.02182]
- Buzas EI. The roles of extracellular vesicles in the immune system. Nat Rev Immunol 2023;23(4):236-50. [PMID: 35927511 DOI: 10.1038/s41577-022-00763-8]
- Schorey JS, Cheng Y, Singh PP, Smith VL. Exosomes and other extracellular vesicles in host-pathogen interactions. EMBO Rep 2015;16(1):24-43. [PMID: 25488940 DOI: 10.15252/embr.201439363]
- Benichou G, Wang M, Ahrens K, Madsen JC. Extracellular vesicles in allograft rejection and tolerance. Cell Immunol 2020;349:104063. [PMID: 32087929 DOI: 10.1016/j.cellimm.2020.104063]
- Morelli AE, Sadovsky Y. Extracellular vesicles and immune response during pregnancy: a balancing act. Immunol Rev 2022;308(1):105-22. [PMID: 35199366 DOI: 10.1111/imr.13074]
- Aloi N, Drago G, Ruggieri S, Cibella F, Colombo P, et al. Extracellular vesicles and immunity: at the crossroads of cell communication. Int J Mol Sci 2024;25(2):1205. [PMID: 38256278 DOI: 10.3390/ijms25021205]
- Zhang Y, Wu D, Zhou C, Bai M, Wan Y, et al. Engineered extracellular vesicles for tissue repair and regeneration. Burns Trauma 2024;12:tkae062. [PMID: 39439545 DOI: 10.1093/burnst/tkae062]
- Hu Z, Wang W, Lin Y, Guo H, Chen Y, et al. Extracellular vesicle-inspired therapeutic strategies for the COVID-19. Adv Healthc Mater 2024;13(29):e2402103. [PMID: 38923772 DOI: 10.1002/adhm.202402103]
- Wang J, Zhang Z, Zhuo Y, Zhang Z, Chen R, et al. Endoplasmic reticulum-targeted delivery of celastrol and PD-L1 siRNA for reinforcing immunogenic cell death and potentiating cancer immunotherapy. Acta Pharm Sin B 2024;14(8):3643-60. [PMID: 39234613 DOI: 10.1016/j.apsb.2024.04.010]
- Zeng L, Shi W, Chen K, Wang K, Dai Y, et al. Indocyanine green aggregation-induced hypotonic stress to remodel aloe exosome-like vesicles for enhanced tumor penetration and phototherapy. ACS Nano 2025;19(16):15425-43. [PMID: 40243994 DOI: 10.1021/acsnano.4c15440]
- Yan G, Xiao Q, Zhao J, Chen H, Xu Y, et al. Brucea javanica derived exosome-like nanovesicles deliver miRNAs for cancer therapy. J Control Release 2024;367:425-40. [PMID: 38295998 DOI: 10.1016/j.jconrel.2024.01.060]
- Yang M, Guo J, Li J, Wang S, Sun Y, et al. Platycodon grandiflorum-derived extracellular vesicles suppress triple-negative breast cancer growth by reversing the immunosuppressive tumor microenvironment and modulating the gut microbiota. J Nanobiotechnology 2025;23(1):92. [PMID: 39920791 DOI: 10.1186/s12951-025-03139-x]
- Wang H, Mu J, Chen Y, Liu Y, Li X, et al. Hybrid ginseng-derived extracellular vesicles-like particles with autologous tumor cell membrane for personalized vaccination to inhibit tumor recurrence and metastasis. Adv Sci (Weinh) 2024;11(17):e2308235. [PMID: 38353384 DOI: 10.1002/advs.202308235]
- Yang L-Y, Liang G-W, Cai B-J, Xu K-M, Zhao Y-J, et al. Enhanced tumor self-targeting of lemon-derived extracellular vesicles by embedding homotypic cancer cell membranes for efficient drug delivery. J Nanobiotechnology 2025;23(1):74. [PMID: 39893448 DOI: 10.1186/s12951-025-03161-z]
- Zhao M, Cheng X, Shao P, Dong Y, Wu Y, et al. Bacterial protoplast-derived nanovesicles carrying CRISPR-Cas9 tools re-educate tumor-associated macrophages for enhanced cancer immunotherapy. Nat Commun 2024;15(1):950. [PMID: 38296939 DOI: 10.1038/s41467-024-44941-9]
- Preet R, Islam MA, Shim J, Rajendran G, Mitra A, et al. Gut commensal Bifidobacterium-derived extracellular vesicles modulate the therapeutic effects of anti-PD-1 in lung cancer. Nat Commun 2025;16(1):3500. [PMID: 40221398 DOI: 10.1038/s41467-025-58553-4]
- Ma S, Zhao G, Sui S, Chen X, Wu L, et al. Tumor microenvironment and immune-related myositis: addressing muscle wasting in cancer immunotherapy. Front Immunol 2025;16:1580108. [PMID: 40386783 DOI: 10.3389/fimmu.2025.1580108]
- Ma S, Lu Y, Sui S, Yang J-S, Fu B-B, et al. Unraveling the triad of immunotherapy, tumor microenvironment, and skeletal muscle biomechanics in oncology. Front Immunol 2025;16:1572821. [PMID: 40242775 DOI: 10.3389/fimmu.2025.1572821]
- Lu Y, Xu J, Tang R, Zeng P, Li Z, You J, et al. Edible pueraria lobata-derived exosome-like nanovesicles ameliorate dextran sulfate sodium-induced colitis associated lung inflammation through modulating macrophage polarization. Biomed Pharmacother 2024;170:116098. [PMID: 38154276 DOI: 110.1016/j.biopha.2023.116098]
- Wang Y, Kong B, Chen X, Liu R, Zhao Y, et al. BMSC exosome-enriched acellular fish scale scaffolds promote bone regeneration. J Nanobiotechnology 2022;20(1):444. [PMID: 36224596 DOI: 10.1186/s12951-022-01646-9]
- Kang Y, Xu C, Meng L, Dong X, Qi M, et al. Exosome-functionalized magnesium-organic framework-based scaffolds with osteogenic, angiogenic and anti-inflammatory properties for accelerated bone regeneration. Bioact Mater 2022;18:26-41. [PMID: 35387167 DOI: 10.1016/j.bioactmat.2022.02.012]
- Wang L, Zhang G, Gao Y, Dai T, Yu J, et al. Extracellular vesicles derived from neutrophils accelerate bone regeneration by promoting osteogenic differentiation of BMSCs. ACS Biomater Sci Eng 2024;10(6):3868-82. [PMID: 38703236 DOI: 10.1021/acsbiomaterials.4c00106]
- Zhou G, Zhou Q, Li R, Sheng S, Gao Q, et al. Synthetically engineered bacterial extracellular vesicles and IL-4-encapsulated hydrogels sequentially promote osteoporotic fracture repair. ACS Nano 2025;19(16):16064-83. [PMID: 40237831 DOI: 10.1021/acsnano.5c03106]
- Liu H, Song P, Zhang H, Zhou F, Ji N, et al. Synthetic biology-based bacterial extracellular vesicles displaying BMP-2 and CXCR4 to ameliorate osteoporosis. J Extracell Vesicles 2024;13(4):e12429. [PMID: 38576241 DOI: 10.1002/jev2.12429]
- Zhao Q, Feng J, Liu F, Liang Q, Xie M, et al. Rhizoma Drynariae-derived nanovesicles reverse osteoporosis by potentiating osteogenic differentiation of human bone marrow mesenchymal stem cells via targeting ER α signaling. Acta Pharm Sin B 2024;14(5):2210-27. [PMID: 38799625 DOI: 10.1016/j.apsb.2024.02.005]
- Zhang L, Luo Z, Chen H, Wu X, Zhao Y. Glycyrrhizic acid hydrogel microparticles encapsulated with mesenchymal stem cell exosomes for wound healing. Research (Wash D C) 2024;7:0496. [PMID: 39403257 DOI: 10.34133/research.0496]
- Keshtkar S, Azarpira N, Ghahremani MH. Mesenchymal stem cell-derived extracellular vesicles: novel frontiers in regenerative medicine. Stem Cell Res Ther 2018;9(1):63. [PMID: 29523213 DOI: 10.1186/s13287-018-0791-7]
- Zheng L, Hu B, Zhao D, Liu W, Liu Q, et al. Recent progresses of exosome–liposome fusions in drug delivery. Chinese Chemical Letters 2024;35(2):108647. [DOI: 10.1016/j.cclet.2023.108647]
- Kim JY, Rhim W-K, Lee SY, Park JM, Song DH, et al. Hybrid nanoparticle engineered with transforming growth factor -β1-overexpressed extracellular vesicle and cartilage-targeted anti-inflammatory liposome for osteoarthritis. ACS Nano 2024;18(50):33937-52. [PMID: 39648484 DOI: 10.1021/acsnano.4c07992]
- Chen J, Wei C, Pan C, Wei Y, Li B, et al. Genetically engineered Bacillus subtilis-derived extracellular vesicles Alleviates Ulcerative colitis by restoring intestinal barrier and regulating gut microbiota. Chem Eng J 2025;512:162727. [DOI: 10.1016/j.cej.2025.162727]
- Li Y, Shao S, Zhou Y, Wang Y, Zheng W, et al. Oral administration of Folium Artemisiae Argyi-derived exosome-like nanovesicles can improve ulcerative colitis by regulating intestinal microorganisms. Phytomedicine 2025;137:156376. [PMID: 39813847 DOI: 10.1016/j.phymed.2025.156376]
- Huang R, Jia B, Su D, Li M, Xu Z, et al. Plant exosomes fused with engineered mesenchymal stem cell-derived nanovesicles for synergistic therapy of autoimmune skin disorders. J Extracell Vesicles 2023;12(10):e12361. [PMID: 37859568 DOI: 10.1002/jev2.12361]
- Liang F, Zheng Y, Zhao C, Li L, Hu Y, et al. Microalgae-derived extracellular vesicles synergize with herbal hydrogel for energy homeostasis in osteoarthritis treatment. ACS Nano 2025;19(8):8040-57. [PMID: 39982764 DOI: 10.1021/acsnano.4c16085]
- Ren X, Xu R, Xu C, Su J. Harnessing exosomes for targeted therapy: strategy and application. Biomater Transl 2024;5(1):46-58. [PMID: 39220669 DOI: 10.12336/biomatertransl.2024.01.005]
- Chen A, He B, Jin H. Isolation of extracellular vesicles from arabidopsis. Curr Protoc 2022;2(1):e352. [PMID: 35030291 DOI: 10.1002/cpz1.352]
- Cai Q, Qiao L, Wang M, He B, Lin F-M, et al. Plants send small RNAs in extracellular vesicles to fungal pathogen to silence virulence genes. Science 2018;360(6393):1126-9. [PMID: 29773668 DOI: 10.1126/science.aar4142]
- Herrmann IK, Wood MJA, Fuhrmann G. Extracellular vesicles as a next-generation drug delivery platform. Nat Nanotechnol 2021;16(7):748-59. [PMID: 34211166 DOI: 10.1038/s41565-021-00931-2]
- Claridge B, Lozano J, Poh QH, Greening DW. Development of extracellular vesicle therapeutics: challenges, considerations, and opportunities. Front Cell Dev Biol 2021;9:734720. [PMID: 34616741 DOI: 10.3389/fcell.2021.734720]
- Wang H, Yang Z, Ai S, Xiao J. Updated methods of extracellular vesicles isolation. Adv Exp Med Biol 2023;1418:3-14. [PMID: 37603269 DOI: 10.1007/978-981-99-1443-2_1]
- Sproviero D, Gagliardi S, Zucca S, Arigoni M, Giannini M, et al. Different miRNA profiles in plasma derived small and large extracellular vesicles from patients with neurodegenerative diseases. Int J Mol Sci 2021;22(5):2737. [PMID: 33800495 DOI: 10.3390/ijms22052737]
- Kushawaha B, Rem TT, Pelosi E. Harnessing microbiome, bacterial extracellular vesicle, and artificial intelligence for polycystic ovary syndrome diagnosis and management. Biomolecules 2025;15(6):834. [PMID: 40563474 DOI: 10.3390/biom15060834]
- Civra A, Francese R, Donalisio M, Tonetto P, Coscia A, et al. Human colostrum and derived extracellular vesicles prevent infection by human rotavirus and respiratory syncytial virus in vitro. J Hum Lact 2021;37(1):122-34. [PMID: 33534629 DOI: 10.1177/0890334420988239]
- Miyake H, Lee C, Chusilp S, Bhalla M, Li B, et al. Human breast milk exosomes attenuate intestinal damage. Pediatr Surg Int 2020;36(2):155-63. [PMID: 31713717 DOI: 10.1007/s00383-019-04599-7]
- Xie M-Y, Hou L-J, Sun J-J, Zeng B, Xi Q-Y, et al. Porcine milk exosome MiRNAs attenuate LPS-induced apoptosis through inhibiting TLR4/NF-κB and p53 pathways in intestinal epithelial cells. J Agric Food Chem 2019;67(34):9477-91. [PMID: 31429552 DOI: 10.1021/acs.jafc.9b02925]
- Chen T, Xie M-Y, Sun J-J, Ye R-S, Cheng X, et al. Porcine milk-derived exosomes promote proliferation of intestinal epithelial cells. Sci Rep 2016;6:33862. [PMID: 27646050 DOI: 10.1038/srep33862]
- Kim H, Jang Y, Kim EH, Jang H, Cho H, et al. Potential of colostrum-derived exosomes for promoting hair regeneration through the transition from telogen to anagen phase. Front Cell Dev Biol 2022;10:815205. [PMID: 35359449 DOI: 10.3389/fcell.2022.815205]
- Bae I-S, Kim SH. Milk exosome-derived MicroRNA-2478 suppresses melanogenesis through the Akt-GSK3β pathway. Cells 2021;10(11):2848. [PMID: 34831071 DOI: 10.3390/cells10112848]
- Oliveira MC, Arntz OJ, Blaney Davidson EN, van Lent PLEM, Koenders MI, et al. Milk extracellular vesicles accelerate osteoblastogenesis but impair bone matrix formation. J Nutr Biochem 2016;30:74-84. [PMID: 27012623 DOI: 10.1016/j.jnutbio.2015.11.017]
- Mu J, Zhuang X, Wang Q, Jiang H, Deng Z-B, et al. Interspecies communication between plant and mouse gut host cells through edible plant derived exosome-like nanoparticles. Mol Nutr Food Res 2014;58(7):1561-73. [PMID: 24842810 DOI: 10.1002/mnfr.201300729]
- Zhuang X, Deng Z-B, Mu J, Zhang L, Yan J, et al. Ginger-derived nanoparticles protect against alcohol-induced liver damage. J Extracell Vesicles 2015;4:28713. [PMID: 26610593 DOI: 10.3402/jev.v4.28713]
- Wang B, Zhuang X, Deng Z-B, Jiang H, Mu J, et al. Targeted drug delivery to intestinal macrophages by bioactive nanovesicles released from grapefruit. Mol Ther 2014;22(3):522-34. [PMID: 23939022 DOI: 10.1038/mt.2013.190]
- Raimondo S, Naselli F, Fontana S, Monteleone F, Lo Dico A, et al. Citrus limon-derived nanovesicles inhibit cancer cell proliferation and suppress CML xenograft growth by inducing TRAIL-mediated cell death. Oncotarget 2015;6(23):19514-27. [PMID: 26098775 DOI: 10.18632/oncotarget.4004]
- Wang Q, Zhuang X, Mu J, Deng Z-B, Jiang H, et al. Delivery of therapeutic agents by nanoparticles made of grapefruit-derived lipids. Nat Commun 2013;4:1867. [PMID: 23695661 DOI: 10.1038/ncomms2886]
- Alpdundar Bulut E, Bayyurt Kocabas B, Yazar V, Aykut G, Guler U, et al. Human gut commensal membrane vesicles modulate inflammation by generating M2-like macrophages and myeloid-derived suppressor cells. J Immunol 2020;205(10):2707-18. [PMID: 33028617 DOI: 10.4049/jimmunol.2000731]
- Rodovalho VR, da Luz BSR, Rabah H, do Carmo FLR, Folador EL, et al. Extracellular vesicles produced by the probiotic Propionibacterium freudenreichii CIRM-BIA 129 mitigate inflammation by modulating the NF-κB pathway. Front Microbiol 2020;11:1544. [PMID: 32733422 DOI: 10.3389/fmicb.2020.01544]
- Choi JH, Moon CM, Shin T-S, Kim EK, McDowell A, et al. Lactobacillus paracasei-derived extracellular vesicles attenuate the intestinal inflammatory response by augmenting the endoplasmic reticulum stress pathway. Exp Mol Med 2020;52(3):423-37. [PMID: 32123288 DOI: 10.1038/s12276-019-0359-3]
- Alvarez C-S, Badia J, Bosch M, Giménez R, Baldomà L. Outer membrane vesicles and soluble factors released by probiotic Escherichia coli Nissle 1917 and commensal ECOR63 enhance barrier function by regulating expression of tight junction proteins in intestinal epithelial cells. Front Microbiol 2016;7:1981. [PMID: 28018313 DOI: 10.3389/fmicb.2016.01981]
- Chelakkot C, Choi Y, Kim D-K, Park HT, Ghim J, et al. Akkermansia muciniphila-derived extracellular vesicles influence gut permeability through the regulation of tight junctions. Exp Mol Med 2018;50(2):e450. [PMID: 29472701 DOI: 10.1038/emm.2017.282]
- Kim W, Lee EJ, Bae I-H, Myoung K, Kim ST, et al. Lactobacillus plantarum-derived extracellular vesicles induce anti-inflammatory M2 macrophage polarization in vitro. J Extracell Vesicles 2020;9(1):1793514. [PMID: 32944181 DOI: 10.1080/20013078.2020.1793514]
- Choi J, Kim Y-K, Han P-L. Extracellular vesicles derived from Lactobacillus plantarum increase BDNF expression in cultured hippocampal neurons and produce antidepressant-like effects in mice. Exp Neurobiol 2019;28(2):158-71. [PMID: 31138987 DOI: 10.5607/en.2019.28.2.158]
- Carvalho AL, Fonseca S, Miquel-Clopés A, Cross K, Kok K-S, et al. Bioengineering commensal bacteria-derived outer membrane vesicles for delivery of biologics to the gastrointestinal and respiratory tract. J Extracell Vesicles 2019;8(1):1632100. [PMID: 31275534 DOI: 10.1080/20013078.2019.1632100]