Advances in the Application of Living Cells in Tumor Radiosensitization
1Key Laboratory of Medical Imaging Precision Theranostics and Radiation Protection, University of South China, College of Hunan Province, Changsha, Hunan 410004, China
2Department of Medical Imaging, Hengyang Medical School, The Affiliated Changsha Central Hospital, University of South China, Changsha, Hunan 410004, China
3Institute of Medical Imaging, Hengyang Medical School, University of South China, Hengyang, Hunan 421001, China
4School of Public Health, University of South China, Hengyang, Hunan 421001, China
5The Seventh Affiliated Hospital, Hunan Veterans Administration Hospital, Hengyang Medical School, University of South China, Changsha, Hunan, China
6Department of Biochemistry, University of Cambridge, Cambridge, CB2 1QW, UK
*Correspondence to: Hengrui Liu, Department of Biochemistry, University of Cambridge. E-mail: hl546@cam.ac.uk
Received: May 17 2025; Revised: July 14 2025; Accepted: August 13 2025; Published Online: December 4 2025
Cite this paper:
Xu H, Feng R, Liu H et al. Advances in the Application of Living Cells in Tumor Radiosensitization. BIO Integration 2025; 6: 1–16.
DOI: 10.15212/bioi-2025-0097. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Radiation therapy (RT) has a critical role in cancer treatment, yet the efficacy is often limited by tumor resistance mechanisms, such as cellular DNA repair activation, heterogeneous cell cycle, hypoxia, and an immunosuppressive microenvironment. Conventional radiosensitization strategies face significant challenges, which are caused by insufficient efficiency, a short treatment window, and off-target toxicities. In contrast, living cells offer a novel strategy to overcome these limitations by leveraging innate characteristics, including tumor chemotaxis and radiosensitization factor secretion. Living cells have recently been applied in radiotherapy to amplify tumor cell killing effects and avoid healthy tissue damage. This review systematically summarizes recent advances in living cell-based radiosensitization strategies, emphasizing the dual roles as tumor targeting carriers and dynamic microenvironment modulators. The radiosensitization mechanisms underlying diverse cell types are analyzed. For example, stem cells enhance radiotherapy via STAT3-mediated DNA repair inhibition, while immune cells and bacteria use immunogenic cell death synergy to induce immune activation. Finally, the challenges and prospects of living cells in achieving radiosensitization are highlighted.
Keywords
Living cells, radiosensitization, tumor microenvironment, tumor radiotherapy, tumor targeting delivery.
Introduction
Radiation therapy (RT) is an important tumor treatment modality. RT directly damages DNA to kill tumor cells and simultaneously induces cells to produce reactive oxygen species (ROS), thereby inducing cell death [1]. Nevertheless, clinical efficacy is significantly constrained by multiple resistance mechanisms arising from tumor cell intrinsic adaptations and extrinsic microenvironment barriers [2]. Hypoxia is a hallmark of solid tumors that diminishes oxygen-dependent free radical formation and subsequent DNA damage during irradiation [3]. DNA repair pathways, including homologous recombination (HR) and non-homologous end joining (NHEJ), rapidly restore gene integrity following radiation-induced double-strand breaks (DSBs) [4]. Concurrently, the immunosuppressive tumor microenvironment (TME) causes immune evasion by Treg infiltration and immune checkpoint ligand upregulation after irradiation. Moreover, heterogeneous cell cycle distribution across tumor cells introduces additional therapeutic challenges because different radiosensitivities among tumor cells mediate tumor recurrence [5].
Current research has developed physical and chemical radiosensitization strategies to address these challenges in radiotherapy [6]. Conventional strategies enhance ROS generation and DNA damage by regulating intracellular signaling pathways or remodeling the TME [7]. However, these strategies face limitations in clinical practice, including insufficient efficacy, transient effects, and off-target toxicity [8]. Biological radiosensitization strategies utilizing living cells (e.g., stem cells, immune cells, and bacteria) have emerged as novel solutions [9–11]. For example, living cells deliver radiosensitizers into tumor tissues through chemotactic movement and microenvironment-responsive payload release [12]. Living cell-based radiosensitization demonstrates superior efficacy and safety compared to physical/chemical approaches and relies on localized cytokine storm instead of conventional systemic toxicities and enhanced specificity through tumor targeting. In contrast, living cells utilize intrinsic properties to dynamically regulate the TME and achieve precision modulation radioresistance challenges [13]. Living cells overcome the limitations of conventional strategies and achieve effective radiosensitization by leveraging inherent tumor chemotaxis and functional activity [14].
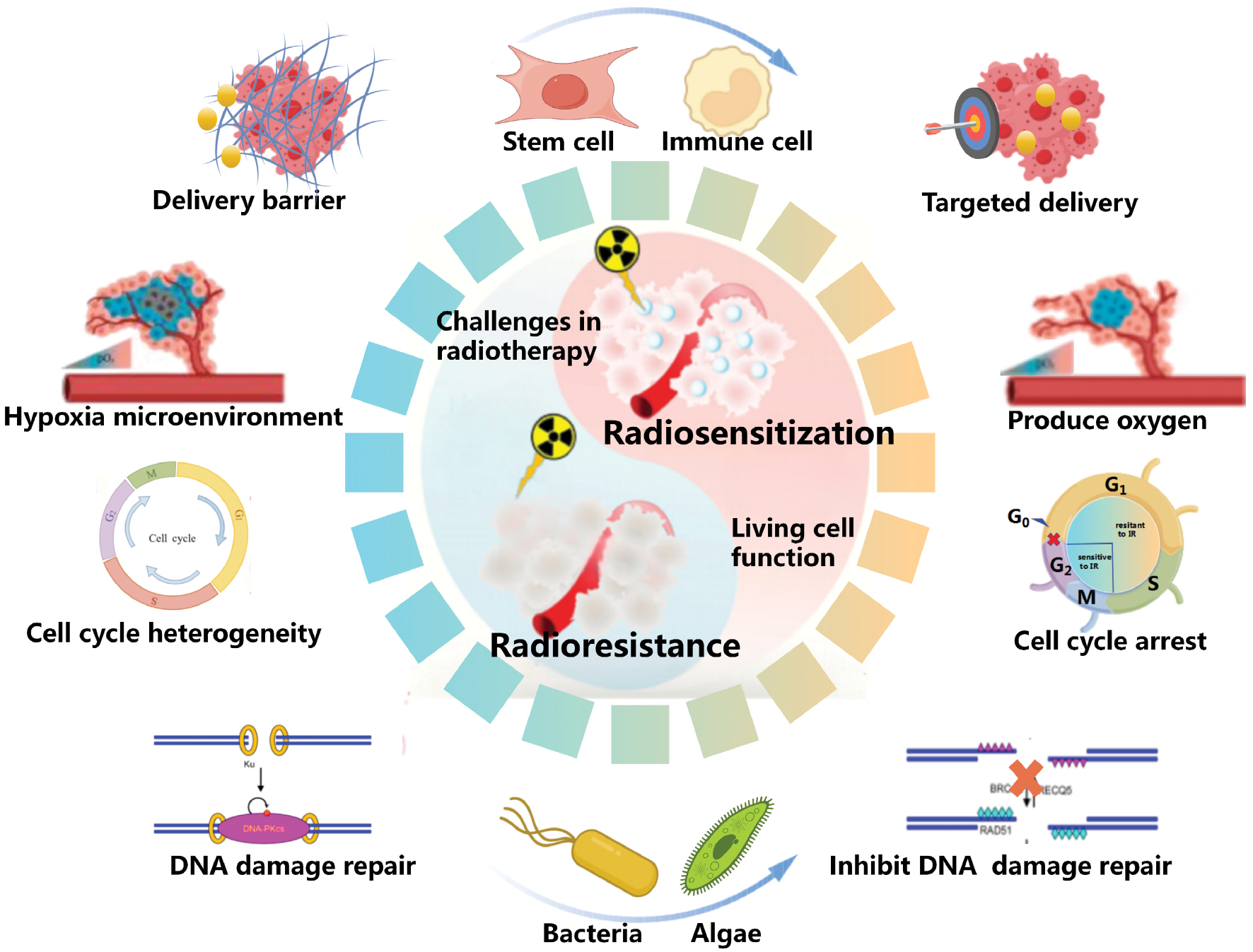
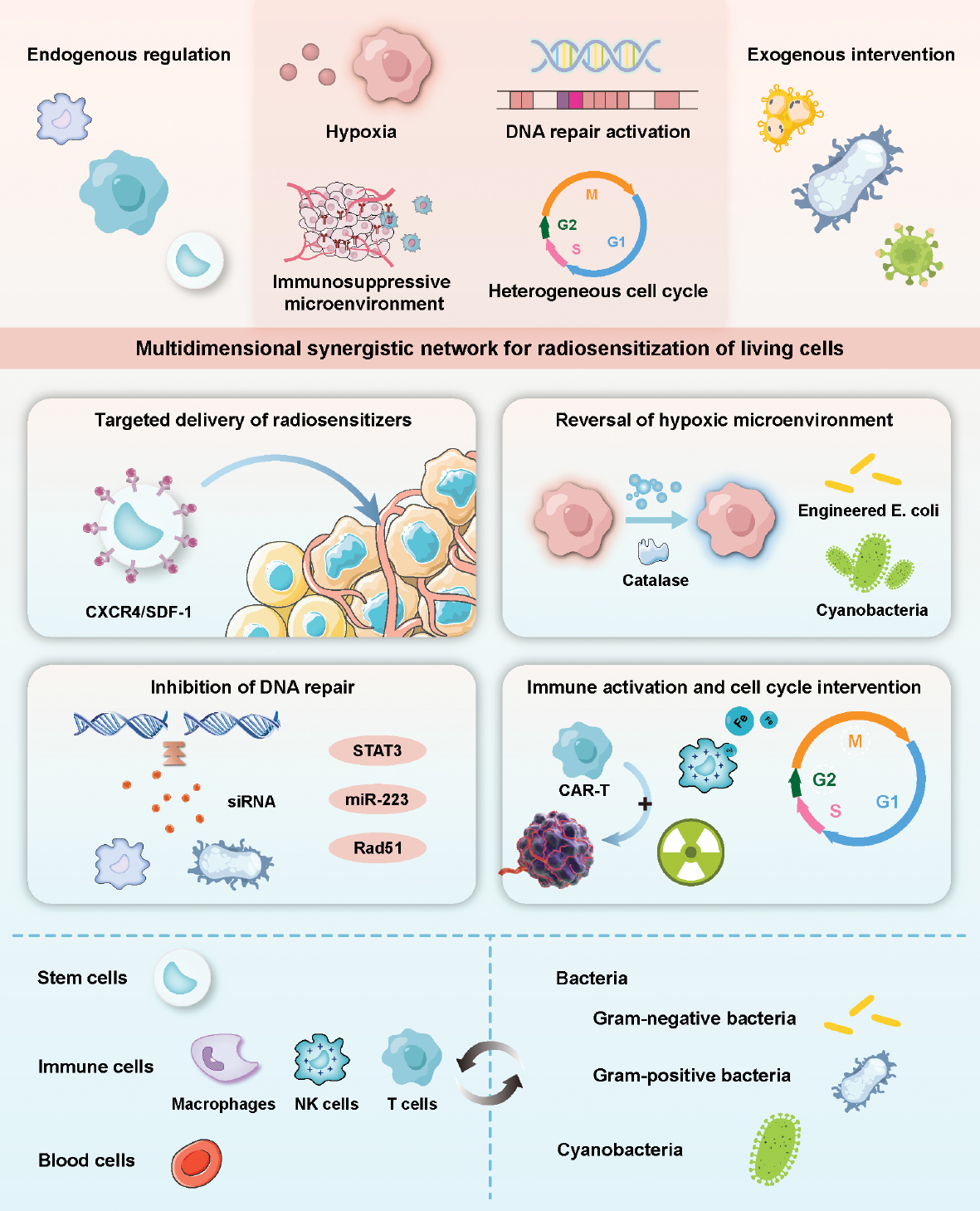
RT resistance is not driven by isolated factors. Indeed, the interconnected resistance pathways collectively undermine radiotherapy outcomes [15]. Resistance pathways form a self-reinforcing network through HIF-1α/NF-κB-mediated crosstalk, through which hypoxia stabilizes HIF-1α to upregulate DNA repair enzymes and immunosuppressive ligands. Radiation-induced DNA damage activates NF-κB to amplify PD-L1 expression and Treg infiltration and the resultant immunosuppression maintains hypoxia by impairing vascular normalization. However, conventional strategies often fail to achieve effective tumor suppression by neglecting cross-talk between resistance pathways [16]. Recent advances in understanding radiosensitization mechanisms and living cell biological capabilities have propelled the development of multifunctional living cells [17]. Living cells integrate capabilities, such as hypoxia alleviation, DNA repair inhibition, and immune activation, shifting from single-function interventions to synergistic multi-target modulation [18, 19]. In this review recent advances in the application of living cells in tumor radiosensitization are summarized and various radiosensitization strategies of different living cell types are analyzed. Finally, challenges and future directions for living cell-based radiosensitization are discussed (Figure 1).
Figure 1 Schematic illustration of living cells in tumor radiosensitization application. This figure illustrates the key factors mediating radioresistance and the mechanisms by which living cell-based platforms synergistically enhance radiotherapy efficacy through targeted delivery, hypoxia modulation, DNA repair inhibition, and cell cycle intervention.
This review used a systematic search strategy in PubMed, Web of Science, and Scopus (January 2015–June 2025). Keywords included ‘living cells,’ ‘radiosensitization,’ ‘bacteria radiotherapy,’ ‘stem cell radiotherapy,’ ‘immune cell radiotherapy,’ and ‘tumor microenvironment.’ The inclusion criteria were as follows: (i) original studies reporting living cell-based radiosensitization; (ii) mechanistic investigations of radioresistance modulation; and (iii) clinical trials (any phase). The exclusion criteria were as follows: (i) non-English publications; (ii) non-cancer applications; and (iii) reviews without primary data. Of the 537 studies retrieved, 35 met the eligibility criteria.
Potential role of living cells in tumor radiosensitization
Increasing the amount of radiation-induced damage
Amplifying radiation-induced damage represents a radiosensitization strategy to overcome tumor heterogeneity, transcend dose limitations, and improve the clinical prognosis. Radiosensitizers are compounds or materials designed to increase tumor vulnerability to irradiation through dual mechanisms [amplifying initial radiation damage (e.g., ROS/DNA radicals) or suppressing DNA repair pathways] [20]. In recent years radiosensitizers have been extensively investigated in preclinical research, predominantly through mechanisms involving direct DNA damage increasing or indirect ROS amplification to enhance therapeutic efficacy [21].
Despite promising preclinical outcomes, the clinical translation of radiosensitizers is constrained by ruleless biodistribution and systemic toxicity [22]. Moreover, radiotherapy activates the immune system by inducing the release of cellular damage-associated molecular patterns (DAMPs) and tumor-associated antigens from cancer cells [12]. However, the immune activation impact of radiotherapy is frequently diminished by various mechanisms, including regulatory T cell (Treg) infiltration and programmed death-ligand 1 (PD-L1) upregulation [23]. Traditional immune activation strategies (e.g., immune checkpoint inhibitor therapy and systemic cytokine administration) are limited by system toxicity and incomplete immunosuppression recovery [19].
Living cells address these limitations by utilizing innate biological functionalities, including tumor targeting and microenvironment responsiveness. For example, stem cells are designed to actively transport high-Z element nanoparticles to tumor sites through chemotactic movement [24], while macrophages are programmed to release cytotoxic payloads in response to tumor-associated inflammatory signals [25]. Living cells induce tumor damage and avoid systemic toxicity by the targeted delivery of radiosensitizers. Additionally, radiation exerts the most potent cytotoxic effect on cells during the G2/M phase of the cell cycle. Tumor cell cycle heterogeneity drives radiotherapy resistance by reducing the proportion of radiosensitive G2/M-phase cells [26]. Living cells enable constrained regulation of tumor cell cycle dynamics, thereby expanding the therapeutic window for radiation-induced mitotic disruption [27]. Furthermore, Salmonella arrest tumor-specific cell cycle with apoptosis induction through caspase pathway activation [28]. Therefore, living cells efficiently overcome radioresistance by increasing the amount of radiation-induced damage.
Decreasing the efficiency of post-radiation DNA repair
Increasing radiation-induced damage corresponds to an increase in radiation dose. As a result, a preferable method for enhancing irradiation efficacy is achieved by targeting DNA repair mechanisms [20]. DNA repair inhibition represents a core radiosensitization strategy that is complementary to damage amplification. Tumor cells efficiently repair radiation-induced DNA double-strand breaks (DSBs) through HR and NHEJ pathways, significantly diminishing radiotherapy efficacy [29]. Conventional DNA repair inhibition strategies, including PARP inhibitors and glutathione (GSH)-depleting agents, suppress DNA repair pathways and damage gene integrity in healthy cells, while increasing risks of secondary malignancies [30]. Living cells overcome these limitations via tumor-targeted precision. For example, engineered neutrophils selectively deliver BRCA1-targeting siRNA to tumors and restrict HR pathway silencing, while bacteria secrete redox-modulating factors to diminish local tumor cell gene integrity [1, 31]. Living cells can easily achieve DNA damage repair inhibition and preserve systemic gene stability compared to systemic inhibitors.
Furthermore, oxygen serves as the fuel to stabilize DNA damage caused by radiation and prevents the DNA self-repair process [32]. However, the efficacy of radiotherapy has been severely limited due to the hypoxia status in most solid tumors [33]. Conventional approaches, including hyperbaric oxygen therapy and vascular normalization strategies, are constrained by insufficient oxygen production and transient therapeutic effects [34]. In contrast, living cells are able to sustain a sufficient tumor oxygen level through consistent oxygen generation [35]. For example, engineered bacteria stably express oxygen-generating enzymes to achieve prolonged oxygen generation compared to small-molecule oxygen carriers [11]. Similarly, photosynthetic cyanobacteria leverage light-triggered oxygen generation to dynamically regulate tumor oxygen levels [36]. In conclusion, living cells mediate stable inhibition of DNA repair, offering a potential solution for enhancing radiotherapy efficacy.
Targeting manipulation of two or more individual pathways
The interdependence of radioresistance pathways (DNA repair enables immunosuppression and hypoxia sustains cell cycle heterogeneity, and vice versa) forms a self-reinforcing network that single-target interventions cannot disrupt. Building on this framework, simultaneous targeting of two or more pathways becomes essential to overcome cross-pathway compensation [37]. Living cells address these challenges through synergistic radioresistance modulation via multiple functional integration compared to conventional approaches. For example, engineered T cells co-deliver PD-1 antibodies and interleukin-12 (IL-12) within tumor lesions, simultaneously depleting Treg populations and recovering cytotoxic T lymphocyte activity [38].
Furthermore, living cells integrate mechanisms, including immune system activation, metabolic reprogramming, and extracellular matrix remodeling, to achieve coordinated optimization of tumor radiosensitization and systemic biosafety [39]. Unlike traditional combined therapies that are limited by off-target drug interactions and cumulative toxicities, living cells serve as tumor targeting carriers and dynamic microenvironment modulators that disrupt radioresistance networks, while preserving physiologic tissue homeostasis [40].
Advances in eukaryotic cells for tumor radiosensitization
Stem cells
Stem cells represent a unique cellular population characterized by self-renewal capacity and differentiation potential [41]. The distinctive biological properties of stem cells exploit chemotaxis, immunomodulation, and microenvironment sensing to overcome radioresistance. [42]. Stem cells actively migrate toward tumor sites via chemokines (e.g., SDF-1 and CXCR4). This targeting capability offers stem cells dual advantages for enhancing localized radiation cytotoxicity, while reducing systemic toxicity [43]. Stem cells are simply divided into three different varieties (adult stem cells, embryonic stem cells, and induced pluripotent stem cells [iPSCs]). Embryonic stem cells have a high probability of immune rejection and ethical dilemmas and iPSCs have genetic instability. Therefore, embryonic stem cells have not been widely used in current research [44].
Mesenchymal stem cells (MSCs) are the most widely utilized stem cell type. MSCs can be engineered to deliver radiosensitizers through surface modifications, internalization strategies, or gene transfection approaches [45]. Yun et al. reported that engineered MSCs selectively accumulate in tumor tissues in colon cancer models, significantly improving therapeutic efficacy by increasing radiation energy deposition [46]. Notably, stem cell intrinsic protective mechanisms (e.g., antioxidant stress resistance) preserve migratory activity and cellular viability. MSCs loaded with high atomic number (high-Z) nanoparticles achieved a 20-fold increase in tumor-specific drug concentration in non-small cell lung cancer (NSCLC) models, markedly suppressing orthotopic tumor growth [47]. Furthermore, MSCs serve as gene delivery vectors for tumor radiosensitization. Wang et al. demonstrated that an MSC-expressed sodium iodide symporter efficiently improves radiotherapy outcomes [48]. MSCs exhibit limited targeting efficiency for brain tumors restricted by the blood-brain barrier, whereas neural stem cells (NSCs) leverage intrinsic glioma chemotaxis to pass through delivery barriers, enabling extensive intratumor distribution of radiosensitizers in glioblastomas [49].
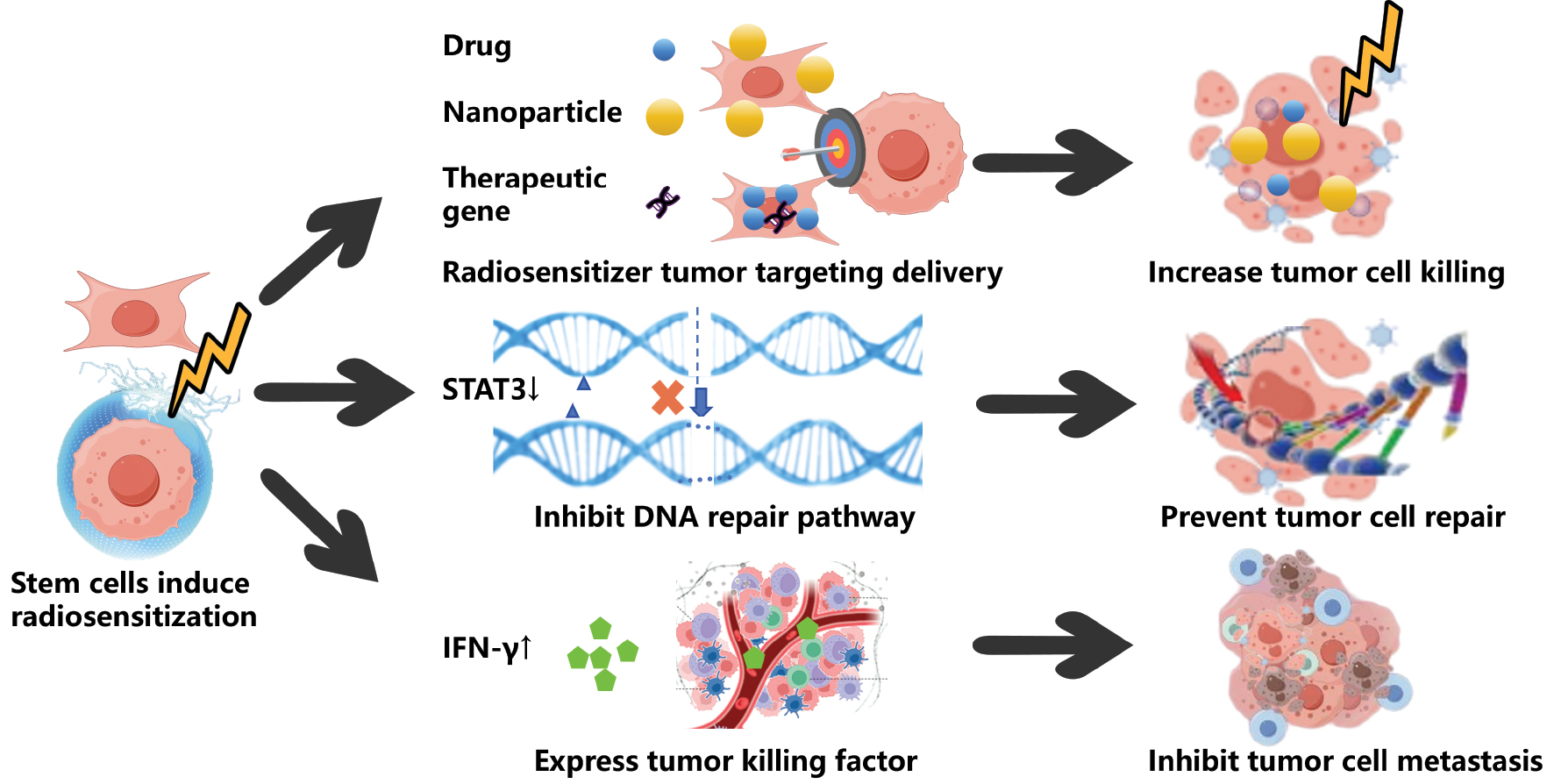
In addition to radiosensitizer delivery, stem cells exhibit a remarkable potential in modulating tumor biology [50]. Studies have shown that low-dose radiation activates MSCs to secrete antitumor cytokines, inhibiting proliferation and inducing cytotoxic synergy. Combining non-irradiated MSCs with radiation in vivo amplifies therapeutic effects through enhanced bystander effects on both primary and distant lesions, which is attributed to increased tumor cell loss and reduced proliferation [51]. Mechanistically, MSCs impair tumor DNA repair by suppressing STAT3 activation. STAT3 is a key regulator of the damage response, while amplifying radiation-induced cytotoxicity in preclinical models. In addition, MSCs reduce ALDH+ cancer stem cell populations, modulate stemness markers, and inhibit tumor metastasis [52]. Complementary research involving verified STAT3 downregulation as a therapeutic mechanism identified IFN-γ-induced transmembrane protein 1 (IFITM1) activation to be critical for enhancing radiotherapy suppression of hepatocellular carcinoma invasion [53] but is limited by inherent tumorigenic risks and prohibitive manufacturing costs. Future directions require synergistic optimization through gene editing technologies to enhance cellular safety profiles and advanced material engineering approaches to improve drug-loading efficiency. These innovative strategies will collectively pave the way for the clinical realization of precision stem cell-based radiosensitization therapies. Furthermore, MSC-based strategies carry tumorigenicity risks. MSCs can paradoxically promote tumor growth via STAT3-mediated secretion of VEGF/IL-6 [52, 54]. Mitigation strategies include the following: (i) genetic insertion of suicide genes (HSV-TK); (ii) pre-treatment irradiation to halt proliferation; and (iii) stringent in vivo tumorigenicity screening (e.g., oncogene expression profiling). These approaches preserve radiosensitization function, while minimizing oncogenic potential (Figure 2).
Figure 2 Application of stem cells for tumor radiosensitization. This figure exhibits the progress in the application of stem cells in radiotherapy sensitization, which can inhibit tumor growth, recurrence, and apoptosis in terms of targeted delivery of a radiotherapy sensitizer, inhibition of DNA repair, and expression of functional factors.
Immune cells
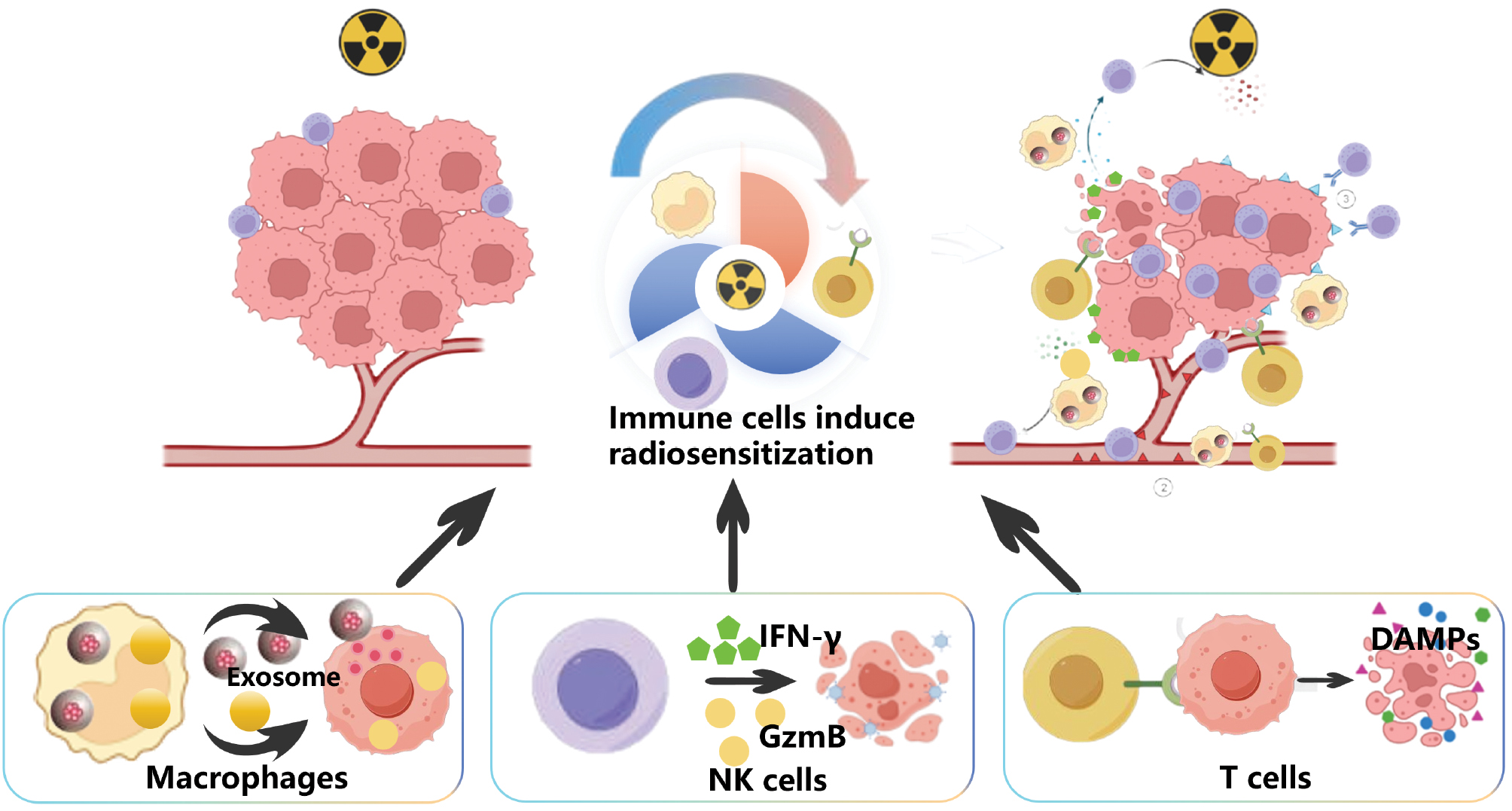
Immune cells, comprising innate and adaptive immunity components, are specialized cellular entities endowed with pathogen recognition, antigen presentation, and cytotoxic effector functions [55]. The unique radiosensitization properties of immune cells come from the following three main attributes: (i) intrinsic tumor tropism via chemokine receptor signaling (e.g., the CCR5/CXCR3 axis); (ii) dynamic crosstalk with irradiated tumor cells; and (iii) capacity to convert a “cold” immunosuppressive TME into “hot” immune responsive states [56]. These characteristics of immune cells give facilitate dual functions for targeted radiosensitizer delivery and systemic immune activation [57]. The latest research showed the inherent immunotherapy function of various immune cells on activation of immunosuppressive microenvironment after radiotherapy. Each cell type also has its own unique characteristics adapted to the radiosensitization mechanism [55] (Figure 3).
Figure 3 Application of immune cells for tumor radiosensitization. This figure introduces the unique biological characteristics of three different immune cells (macrophages, NK cells, and T cells), and the different mechanisms in the application of radiotherapy sensitization.
Among different types of immune cells, macrophages are the most widely used carrier type. Macrophages can efficiently load nanoparticles based on phagocytosis effects without affecting normal function. Li et al. designed a radiosensitizer delivery strategy, known as the “Trojan horse,” and successfully delivered gold nanorods to tumor areas [58]. With further study on the secretion characteristics of macrophages, Fu et al. reported that macrophages promote drug release through exosome-mediated cellular communication, thus significantly increasing the local drug concentration of tumors. Based on this feature Fu et al. realized the precise controlled release and local accumulation of doxorubicin (DOX), a classical radiosensitized agent, at the tumor site [59]. Macrophages deliver antigen and have a critical role in RT remote effects. Macrophages easily mediate radioresensitizer secretion and orchestrate antitumor immune responses within the tumor microenvironment, thereby contributing to effective tumor regression.
Natural killer (NK) cells with rapid killing, immunomodulatory, and antigen-independent characteristics enhance tumor radiotherapy sensitivity through multiple mechanisms involving synergistic killing and metabolic regulation, especially through the secretion of cytokines and cytotoxic molecules [60]. A key mechanism underlying tumor radiotherapy sensitivity involves IFN-γ-driven ferroptosis, an iron-dependent form of regulated cell death, which has emerged as a critical pathway to overcome radioresistance in solid tumors. NK cell-derived IFN-γ triggers lipid peroxidation and disrupts redox homeostasis in tumor cells n prostate cancer-bearing mice, which promotes ferroptosis and amplifies radiation-induced damage. In addition to cytokine-mediated effects, NK cells directly execute tumor cell killing through the release of granzymes (notably granzyme B [GzmB]), which induces DNA fragmentation and apoptosis [61]. Up-regulated tumor cell-intrinsic GzmB expression has been shown to significantly enhance radiosensitivity independent of immune cell activity, highlighting the dual role of GzmB in direct cytotoxicity and metabolic regulation. In conclusion, these findings illustrate the intricate interplay between NK cell-secreted factors and tumor cell responses. Indeed, IFN-γ-mediated ferroptosis and GzmB-driven DNA damage are promising targets for improving tumor radiotherapy efficacy [62]. Chimeric antigen receptor natural killer (CAR-NK) cells are genetically engineered immune cells that combine the inherent cytotoxicity of NK cells with the antigen-targeting capability of CAR technology. CAR-NK cells enhance antitumor efficacy in radiosensitization through multi-mechanism synergy [63]. For example, CAR-mediated recognition of tumor-specific antigens enables precise elimination of radiation-surviving cells. In addition, CAR-NK cells deplete immunosuppressive cells (e.g., Tregs and tumor-associated macrophages [TAMs]) to enhance CD8+ T-cell infiltration. Xia et al. confirmed in an in situ pancreatic tumor model that Robo1-CAR-NK cells combined with radiotherapy synergistically reduce the tumor load and prolong survival, highlighting the clinical transformation potential [64].
The combination of chimeric antigen receptor T (CAR-T) cell therapy with radiotherapy has emerged as an innovative strategy to enhance tumor radiosensitivity by promoting immunogenic cell death (ICD) [65]. Radiation facilitates ICD through the release of DAMPs [66]. These molecules synergistically activate dendritic cells and initiate tumor-specific T-cell responses. CAR-T cells further amplify this mechanism by enhancing antigen presentation and establishing a pro-inflammatory TME [67]. Preclinical studies have demonstrated that CAR-T cells effectively target radiosensitive parental tumor cell lines. CD98hc-targeted CAR-T cells combined with localized radiotherapy enhanced calreticulin (CRT) exposure and promoted CD8+ T-cell infiltration in head and neck squamous cell carcinoma (HNSCC)-bearing mice, leading to improved tumor control effect [68]. Notably, redirected UniCAR-T cells exhibited significant growth inhibition of radioresistant cancer cells even in immunodeficient mice models [69]. These collective findings substantiate the potential of CAR-T cells to overcome radioresistance. Currently, the clinical translation of CAR-T and radiotherapy combined therapy is progressing rapidly. Six patients with hepatic metastases received CAR-T and RT combined therapy in a phase 1b trial. The outcomes have shown that intra-arterial delivery of cytokine-armored CAR-T cells synergizes with radiation-induced ICD, generating sustained antitumor responses [70]. Moreover, the tumor marker levels were decreased in the test patients and prolonged the survival time. A phase II clinical trial testing the combined therapy efficacy was subsequently conducted in non-Hodgkin lymphoma patients [71]. Six patients showed a higher overall response rate after treatment and one patient achieved complete remission. Nevertheless, there are some challenges associated with combined therapy. A CAR-T cell-based radiosensitization strategy requires a suitable radiation dose to balance the ICD induction effect with CAR-T cell viability. In addition, the manufacturing cost of CAR-T cells is expensive and it is difficult to promote the application of CAR-T cells.
Blood cells
Blood cells are widely utilized in drug delivery due to abundant availability, inherent biocompatibility, and changeability [72–75]. Among the blood cells, red blood cells (RBCs) possess unique biological advantages as the most abundant blood cells in humans. RBCs have a long circulation time because of CD47-mediated evasion of the mononuclear phagocyte system. In addition, the biconcave morphology provides exceptional gas exchange capacity through optimized surface-to-volume characteristics [76]. These intrinsic properties position RBCs as natural oxygen carriers with therapeutic potential. Hypoxia in solid tumors, which is driven by abnormal vasculature and excessive hypoxia-inducible factor secretion, significantly diminishes oxygen-dependent radiation damage [77]. RBCs have been engineered into oxygen-delivery platforms to reprogram the hypoxia TME by capitalizing on the hemoglobin-rich composition. RBC-based systems triple tumor oxygenation levels in glioblastoma models by directly enhancing radiation-induced DNA damage, while suppressing HIF-1α-mediated resistance pathways [78]. Zhou et al. further designed a two-stage oxygen delivery system combining perfluorocarbon nanoparticles with RBCs. This innovative design enhanced tumor penetration and achieved targeted oxygen release in hypoxic regions through precise physical activation [79]. With advances in technology, RBCs are transformed from passive carriers to active oxygen modulators.
In addition to RBC-mediated strategies, platelets have unique roles in radiosensitization. Platelets are anucleate cytoplasmic fragments derived from megakaryocytes that circulate for 8–10 days, providing circulatory stability for radiosensitizer delivery. Platelets naturally accumulate at irradiated tumor sites due to vascular injury with engineered platelets observed within tumors 24 h post-injection. Recent studies highlight platelets against radioresistance through distinctive biological properties. Xia et al. designed platelet-based hybrids combining hemoglobin and gold nanoparticles (AuNPs) to simultaneously address hypoxia and DNA repair [80]. Platelets leverage the wound accumulation property to deposit oxygen-releasing hemoglobin in tumor cores and secrete miR-223, which silence a key homologous recombination protein (Rad51) to inhibit DNA damage repair [81]. This strategy suppressed tumors and distant lung metastases in breast cancer models, demonstrating the ability of platelets to simultaneously target microenvironmental and molecular resistance. Blood cells offer advantages in biocompatibility and circulatory stability, yet a short functional lifespan and passive targeting dependency remain as barriers to clinical translation. Future efforts require optimized design through targeted signal peptide modification to address these barriers (Table 1).
Table 1 Application of Eukaryotic Cells in Tumor Radiosensitization
| Type of Living Cells | Radiosensitization Strategy | Function | Study Type | Tumor Type | Ref. |
|---|---|---|---|---|---|
| MSC | Targeted deliver of radiosensitizers (Au/Bi2Se3 NPs) | Amplification of radiation energy deposition (ROS ↑, damaged DNA ↑) | Preclinical in mice | Colon/lung tumor (HT-29/A549 cells) | [46, 47] |
| Targeted delivery of radiosensitive genes | Delivery of the sodium iodide symporter gene (ROS ↑,) | Preclinical in mice | Hypopharyngeal carcinoma | [48] | |
| Improvement in the immunosuppressive environment | Secrete anti-tumor cytokines (TNF-α ↑, IFN-γ ↑) | Preclinical at the cellular level | Melanoma (A375 and G361 cells) | [51] | |
| Inhibit DNA damage repair | Downregulated expression of signal transducers (IFITM1 ↓, STAT3 ↓) | Preclinical in mice and at the cellular level | Breast/liver tumor (MDA-MB-231/Huh7 cells) | [52, 53] | |
| NSC | Targeted delivery of radiosensitizers | Accumulate radiation energy absorption (ROS ↑, Tumor volume ↓) | Preclinical in mice | Glioblastoma (U87-MG cells) | [49] |
| Macrophage | Targeted delivery of radiosensitizers | Increase tumor apoptosis (ROS ↑, damaged DNA ↑) | Preclinical in mice | Breast/liver tumor (MDA-MB-231/HepG2 cells) | [58, 59] |
| NK cell | Improvement in the immunosuppressive environment | Secrete anti-tumor cytokines (IFN-γ ↑) | Preclinical in mice | Prostate tumor (PC3 cells) | [61] |
| Arrest the tumor cell cycle | Secrete cell metabolism regulation protein (granzyme B ↑) | Preclinical in mice | Lung tumor (A549 cells) | [62] | |
| Improvement in the immunosuppressive environment | Deplete immunosuppressive cells (IL-10 ↓, TGF-β ↓) | Preclinical in mice | Pancreatic tumor | [64] | |
| T cell | Improvement in the immunosuppressive environment | Mediate tumor cell lysis (tumor-associated antigens ↑) | Preclinical in mice and at the cellular level | HNSCC | [68, 69] |
| Improvement in the immunosuppressive environment | Mediate tumor cell lysis (tumor-associated antigens ↑) | Clinical trials in humans (NCT03196830) | Lymphoma | [71] | |
| Improvement in the immunosuppressive environment | Mediate tumor cell lysis (tumor-associated antigens ↑) | Clinical trials in humans (NCT02416466) | Liver tumor | [70] | |
| RBC | Targeted delivery of radiosensitizers | Increase tumor apoptosis (ROS ↑, damaged DNA ↑) | Preclinical in mice and at the cellular level | Colon/breast tumor (HT-29/MDA-MB-231 cells) | [72, 75] |
| Overcome tumor hypoxia limitation | Release oxygen in tumors and precise antiangiogenesis (oxygen level ↑) | Preclinical in mice | Melanoma/colon tumor (A375/CT26 cells) | [78, 79] | |
| PLT | Overcome tumor hypoxia limitation and inhibit DNA damage repair | Achieving dual modulation (oxygen level ↑, damaged DNA ↑) | Preclinical in mice | Cervical tumor (ID8 cells) | [80] |
| Inhibit DNA damage repair | Secrete DNA damage repair inhibition cytokine (miR-223 ↑) | Preclinical at the cellular level | Breast tumor (MDA-MB-231 cells) | [81] |
Advances in prokaryotic cells for tumor radiosensitization
Gram-negative bacteria
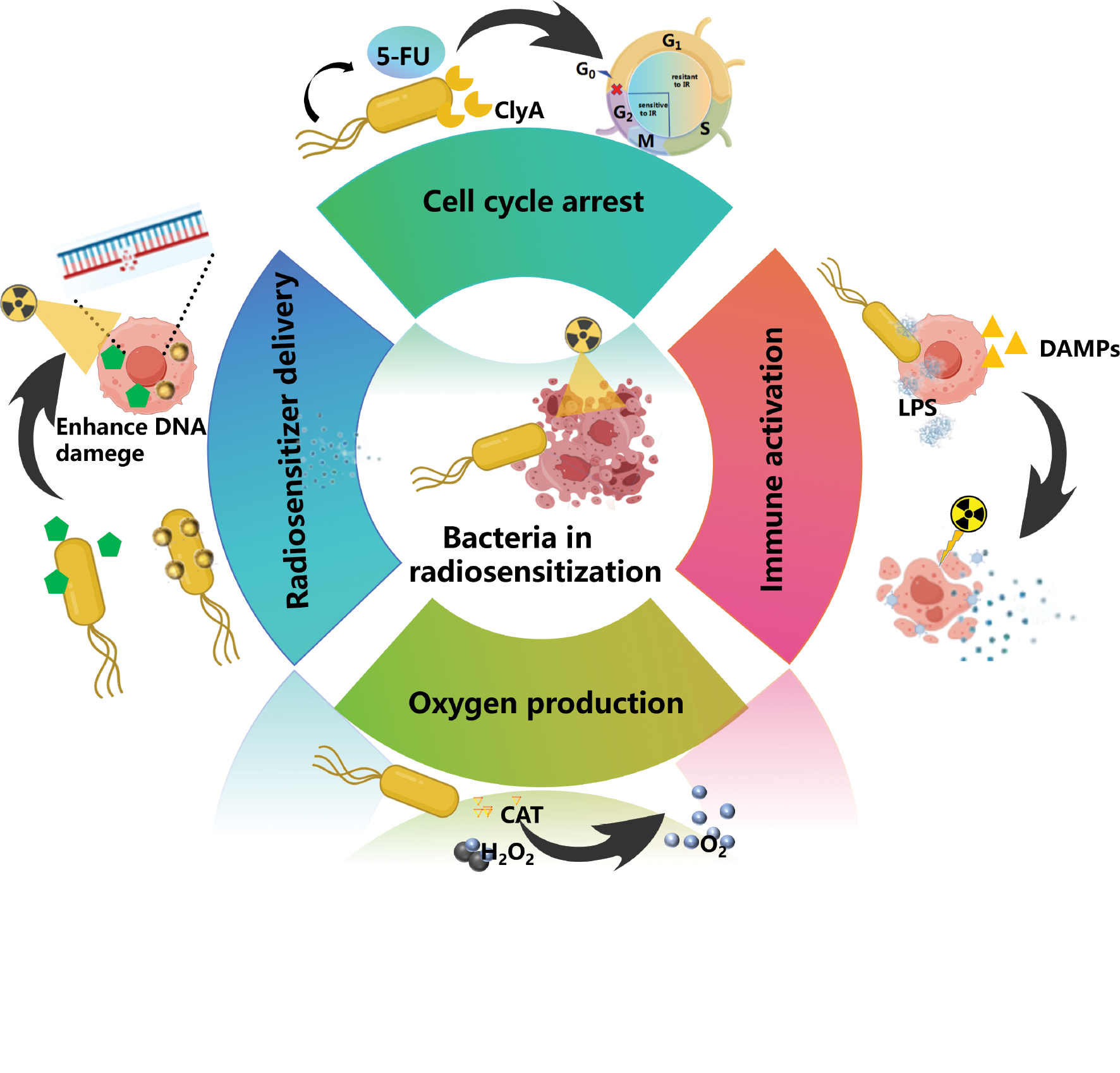
Gram-negative bacteria exhibit unique advantages in tumor radiosensitization due to inherent biological properties [11]. Rich lipopolysaccharides (LPSs) in the outer membrane composition enable immune activation through TLR4 signaling, while surface flagella facilitate autonomous motility and stromal penetration for tumor specific colonization [82]. Gram-negative bacteria, like Escherichia coli and Salmonella, demonstrate exceptional toward radioresistant hypoxic regions. Genetic engineering further extend the radiosensitization function. For example, engineered LPS modification reduces systemic toxicity, while preserving immunogenicity. Inducible promoters enable tumor-localized expression of radiosensitizing enzymes or conversion of prodrugs [83]. Notably, the periplasmic space of Gram-negative bacteria serves as a natural compartment for nanomaterial encapsulation. Recent advances integrate bacterial vectors with high-Z element nanosystems [84]. In a representative study Duo et al. used Salmonella to transport AuNPs through tumor the ECM via flagellum-driven chemotaxis. Salmonella achieved higher intratumoral AuNP retention compared to passive diffusion [85].
Engineered Gram-negative bacteria have been widely used in radiosensitization. Catalase (CAT)-mediated decomposition of intratumoral hydrogen peroxide into oxygen represents a promising strategy to ameliorate hypoxia and potentiate radiotherapy efficacy. However, conventional CAT delivery systems are hindered by inefficient tumor targeting and poor spatiotemporal controllability. To address these limitations, engineered bacteria have emerged as precision platforms for hypoxia modulation. Huang et al. pioneered engineered E. coli Nissle 1917 (ECN) with tumor-localized CAT expression, achieving sustained hypoxia alleviation through enzymatic oxygen generation [86]. Building on this finding, Liu et al. developed ultrasound-responsive bacteria (UEB) featuring stimulus-dependent CAT activation, which minimized oxidative stress in healthy tissues by restricting oxygen production to sonicated tumor regions [87]. Lei et al. further enhanced therapeutic synergy by integrating UEB with AuNPs, coupling enzymatic oxygenation with radiation dose amplification through high-Z element-mediated energy deposition [88]. In addition to enzymatic oxygenation, bacterial systems concurrently address hypoxia-exacerbating microenvironmental barriers. Zhang et al. engineered Salmonella to secrete nattokinase, which degraded the fibronectin-rich ECM and suppressed CAF activity, thereby reducing tumor stiffness and improving oxygen perfusion [89].
Engineered bacteria efficiently regulate tumor cell cycles into radiosensitive phases. Wang et al. developed a prodrug system using bacteria to deliver 5-fluorouracil (5-FU) in response to tumor-specific high GSH signals. The system is able to control the release of 5-FU, shifting tumor cells from the radioresistant S-phase to radiosensitive G2/M-phase [90]. To further expand functionality, Pan et al. engineered bacteria to co-deliver high-Z materials nanoparticles and express cytolytic toxin A, simultaneously enhancing radiation energy deposition and inducing tumor cell cycle arrest [91]. Moreover, Gram-negative bacteria can also self-arrest the cell cycle by the regulated factor and enhance the radiosensitization effect. Cytolethal distending toxin (CDT) is one of the most widely studied bacterial toxins produced by Gram-negative bacteria. CDT has been reported to arrest the cell cycle in the most radiosensitive phase, thus continuously enhancing ionizing radiation (IR)-induced DSBs [92].
Gram-positive bacteria
In contrast to Gram-negative bacteria, Gram-positive species exhibit less efficient secretion systems and lower yields of heterologous protein expression. The capacity of Gram-positive bacteria to enhance radiotherapy efficacy primarily relies on non-engineered mechanisms, such as pattern recognition receptor-mediated immune activation and hypoxia-targeted selective spore germination [54]. Gram-positive bacteria exhibit a high frequency of the immunostimulatory CpG gene sequence with potent activation of human Toll-like receptor 9 (TLR9). CpG gene sequences in Gram-positive bacteria specifically activate plasmacytoid dendritic cells and TLR9-expressing B cells, enhancing co-stimulatory molecule expression with favorable safety profiles [93]. Mason et al. established this approach and demonstrated that CpG synergize with radiotherapy in fibrosarcoma models increasing tumor antigen release, dendritic cell activation, and cytotoxic T-cell infiltration, while inducing necrotic cell death and inflammatory remodeling [94]. Subsequent studies revealed that CpG not only amplify radiation-induced abscopal effects in immunologically “cold” tumors but also establish systemic antitumor memory. However, the mechanistic interplay between CpG and radiation remains underexplored, warranting deeper investigation into radiation-immune crosstalk [95].
Obligate anaerobic bacteria (e.g., Bifidobacterium and Clostridium novyi-NT) exhibit unique tumor-targeting capabilities by exploiting hypoxic regions within the TME by complementing conventional immune activation strategies. Gram-positive bacteria selectively grow and release spores in hypoxic regions, where the proliferative activity physically disrupts the structural integrity of tumor cores, degrades fibrotic stroma, and restores vascular perfusion to alleviate hypoxia [96, 97]. Wang et al. demonstrated that Bifidobacterium significantly augments the abscopal effect of radiotherapy in immunologically “cold” tumors. Wang et al. revealed that Bifidobacterium-mediated vascular normalization increased tumor oxygenation using a murine model of triple-negative breast cancer. Concurrently, bacterial colonization triggeres localized ICD, releasing tumor-associated antigens that synergize with radiation to boost CD8+ T-cell infiltration. These findings highlight the transformative role of obligate anaerobes as dual functional agents capable of simultaneously modulating TME biophysics and immunogenicity, offering a novel strategy in radiosensitization [98]. Various bacteria demonstrate potential in hypoxia modulation and localized toxin delivery. However, spore dissemination and potential germination in healthy organs are concerning. Preclinical toxicity studies in immunocompetent mice revealed that intravenously administered C. novyi-NT spores primarily germinate in hypoxic tumor regions with minimal off-target effects. Furthermore, genetic insertion of auxotrophic markers enables pharmacologic control of bacterial proliferation. Future efforts require optimized design through synthetic gene circuits and immune-evasion coatings to solve these problems (Figure 4).
Figure 4 Application of bacteria for tumor radiosensitization. This figure introduces the strategies and specific application examples of engineered bacteria in radiotherapy sensitization, such as delivery of a radiotherapy sensitizer, improvement in the anaerobic environment, regulation of the cell cycle, and activation of the immune response.
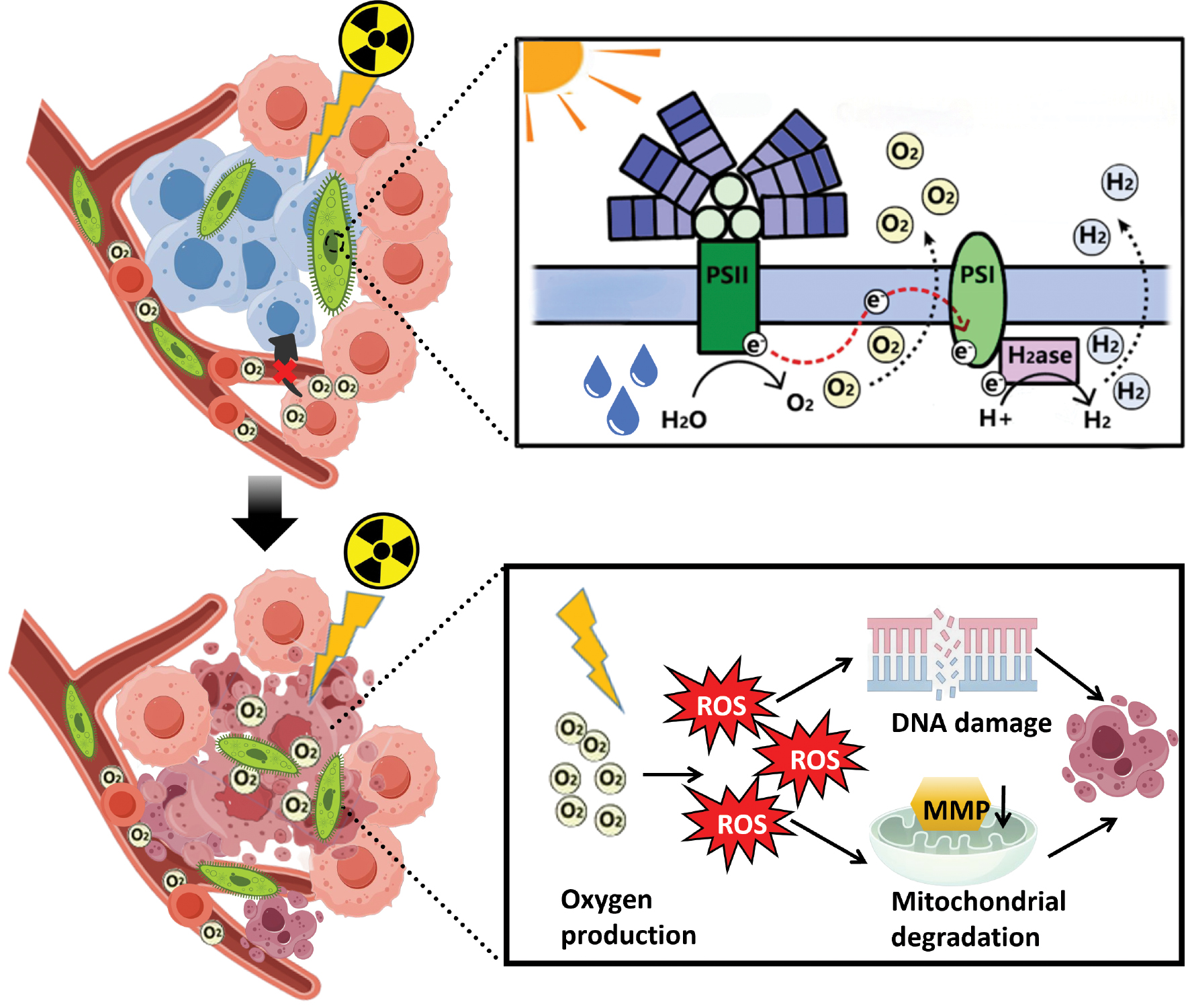
Cyanobacteria
Cyanobacteria are the first autotrophs found on earth with photosynthesis capability. Researchers have widely used cyanobacteria to improve the TME and induce radiosentisization based on cyanobacteria features (Figure 5). In addition to chemical strategies, generating oxygen in situ can be achieved by a natural photosynthetic system [99]. Qiao et al. showed that modifying the algal surface and delivering RBC membrane (RBCM)-algae to the tumor increases tumor oxygenation. RBCM-algae generated O2 in situ and alleviated tumor hypoxia with red light-induced photosynthesis, which further enhanced the radiotherapy effect [100]. Given the potential risks associated with the application of cyanobacteria in vivo, Li et al. further reported a biohybrid microalgae system using a biomineralization approach to improve biocompatibility, while keeping the living activities for radiotherapy in breast cancer. The significant suppression of tumor growth and good safety in mice bearing the 4T1 tumor successfully demonstrated the promising anti-tumor effect of the biomineralization algae-mediated radiotherapy [101].
Figure 5 Application of cyanobacteria for tumor radiosensitization. This figure shows the tumor hypoxia microenvironment and the principle and sensitization effect of cyanobacteria oxygen production through photosynthesis.
Cyanobacteria have been strategically integrated into hybrid platforms combining photosynthetic oxygen generation with high-Z element-mediated radiosensitization to further augment radiosensitization efficiency. A representative example is the development of a two-dimensional bismuthene-cyanobacteria system, which synergistically leverages the sustained photosynthetic hypoxia alleviation capability of cyanobacteria and the high-Z radiosensitization properties of bismuth [102]. Expanding this paradigm, Hua et al. engineered a whole-cell inorganic-biohybrid system by conjugating spirulina with gold nanoclusters. This platform operates through a cascade photocatalytic-radiosensitization mechanism, as follows: (i) light-driven oxygen production to reverse tumor hypoxia; (ii) partial conversion of oxygen to cytotoxic superoxide anions via electron transfer at the biohybrid interface; and (iii) oxidation of intracellular GSH to disrupt redox homeostasis [103]. These advances illustrate the evolution of cyanobacteria-based radiosensitization strategies from single-function oxygenators to multi-function therapeutic platforms. However, the depth dependency of light penetration restricts efficacy in solid tumors and scalable manufacturing of biohybrid systems poses engineering challenges. Future efforts should explore up-conversion NP (UCNP)-mediated deep-tissue photoactivation and microfluidic bioreactor technologies to standardize hybrid NP-cell assembly. To address depth limitations, UCNPs convert near-infrared light (980 nm) to visible wavelengths (400–650 nm), activating cyanobacteria in orthotopic pancreatic models [102]. Implantable fiber-optic diffusers (e.g., cylindrical diffusers at 630 nm) enable precise light delivery, achieving 5-cm depth coverage in clinical prototypes [104]. Combining UCNPs with magnetically guided biohybrids may further enhance deep-tissue activation (Table 2).
Table 2 Application of Prokaryotic Cells in Tumor Radiosensitization
| Type of Living Cells | Radiosensitization Strategy | Function | Anti-Tumor Efficacy | Tumor Type | Ref. |
|---|---|---|---|---|---|
| Escherichia coli | Targeted delivery of radiosensitizers | Delivery of bismuth-based nanomaterials and enhance radiation energy absorption (ROS ↑) | 85% ↓ | Colon/Breast tumor (CT26/4T1 cells) | [83] |
| Targeted delivery of radiosensitizers | Activate prodrug and enhance cytotoxic drug level in tumor (TNF-α ↑, IFN-γ ↑) | 75% ↓ | Colon tumor (HT29 cells) | [84] | |
| Overcome tumor hypoxia limitation | Express catalase to decompose hydrogen peroxide (oxygen level ↑) | 81% ↓ | Breast tumor (MDA-MB-231/4T1 cells) | [86, 87] | |
| Targeted delivery of radiosensitizers and overcome tumor hypoxia limitation | Express catalase and AuNPs (oxygen level ↑, damged DNA ↑) | 92% ↓ | Breast tumor (4T1 cells) | [88] | |
| Regulate tumor cell cycle | Arrest tumor cell cycle in the radiosensitive G2/M phase (damged DNA ↑) | 68% ↓ | Colon tumor (HT29 cells) | [90, 91] | |
| Salmonella | Targeted delivery of radiosensitizers | Deliver AuNPs into tumor cells (ROS ↑) | 77% ↓ | Pancreatic tumor (PANC1 cells) | [85] |
| Overcome tumor hypoxia limitation | Express nattokinase to degrade ECM (oxygen level ↑) | 79% ↓ | Colon tumor (CT26 cells) | [89] | |
| Inhibit DNA damage repair | Sequestering DNA damage repair proteins (damaged DNA ↑) | 75% ↓ | Breast tumor (MDA-MB-231 cells) | [92] | |
| Clostridium | Improvement in the immunosuppressive environment | Activating immune effect and conferring immune memory (IL-10 ↓, TGF-β ↓) | 84% ↓ | Fibrosarcoma/Breast tumor (C57/MDA-MB-231 cells) | [94, 95] |
| Overcome tumor hypoxia limitation | Release spores and destruct tumor core hypoxia region (oxygen level ↑, TNF-α ↑) | 85% ↓ | Bladder tumor | [96] | |
| Bifidobacterium | Overcome tumor hypoxia limitation | Release spores and destruct tumor core hypoxia region (oxygen level ↑, TNF-α ↑) | 74% ↓ | Fibrosarcoma (C57 cells) | [97] |
| Improvement in the immunosuppressive environment | Release immune active factors and induce ICD (TNF-α ↑, IFN-γ ↑) | 76% ↓ | Colon/Breast tumor (CT26/4T1 cells) | [98] | |
| Nostoc | Overcome tumor hypoxia limitation | Alleviate hypoxia by photosynthesis (oxygen level ↑) | 69% ↓ | Breast tumor/fibrosarcoma (MDA-MB-231/C57 cells) | [100, 101] |
| Spirulina | Overcome tumor hypoxia limitation and targeted delivery of radiosensitizers | Alleviate hypoxia by photosynthesis and delivery of Bi/Au-based nanomaterials (oxygen level ↑, damaged DNA ↑) | 84% ↓ | Melanoma/breast tumor (A375/4T1 cells) | [102, 103] |
Challenges and prospects
Living cell-based radiosensitization strategies, which harness dynamic targeting, microenvironment remodeling, and multifunctional synergy of eukaryotic and prokaryotic cells, represent a novel strategy overcoming conventional radiotherapy limitations. Emerging clinical trials also demonstrate the viability of living cell-radiotherapy combinations, promising preclinical efficacy in achieving localized radiosensitizer delivery and tumor TME reprogramming. CAR-T cells combined with radiotherapy induced sustained responses in lymphoma/liver metastasis patients by enhancing ICD in phase I/II trials [70, 71]. Intravenous Salmonella VNP20009 (NCT00004216) improved tumor targeting in metastatic melanoma [105]. However, the clinical translation faces many challenges. Specifically, current studies predominantly rely on animal models, which fail to fully explore living cell-tumor interactions under physiologic conditions. Humanized organ platforms, including 3D bioprinted tumor models and patient-derived organs, are needed to elucidate the specific radiosensitization mechanism of living cells. Advanced biomanufacturing platforms are critical for standardization. Microfluidic systems achieve > 95% loading efficiency for cell-nanomaterial assemblies [106]. 3D-bioprinted tumor organoids facilitate high-throughput screening of living cell radiosensitization under physiologic hypoxia [107]. Scalable hollow-fiber bioreactors support GMP-compliant expansion of engineered immune cells for clinical use [108]. In contrast, some living cells (e.g., anaerobic bacteria and stem cells) require a rigorous cultivation environment to ensure viability. It is necessary for advanced strategies (e.g., quality control system and dynamic feeding technology) to further improve the production process. Moreover, conventional radiosensitizer-loading methods often impair cellular viability or motility. Therefore, new loading techniques, like outer membrane vesicle fusion or CRISPR-based gene transformation, should be explored (Table 3).
Table 3 Advantages and Disadvantages of Various Living Cells in Radiosensitization
| Cell type | Radiosensitization Mechanisms | Advantages | Disadvantages |
|---|---|---|---|
| Stem cells | |||
| MSCs | STAT3-mediated DNA repair inhibition | Precise identification of tumor cells; long cycle duration; immune memory | Surface modification instability; limited efficacy in brain tumors |
| NSCs | BBB-penetrating radiosensitizer delivery | Natural tendency and selective tumor targeting; ability to penetrate the blood-brain barrier | Short duration of treatment; limited application outside brain tumors |
| Immune cells | |||
| Macrophages | “Trojan horse” nanoparticle delivery | Chemotaxis; phagocytosis; intercellular communication gradients | Nanoparticle loading may impair functionality |
| NK cells | IFN-γ-induced ferroptosis + GzmB DNA damage | Intercellular communication gradients; direct DNA fragmentation via granzyme B | Tumor heterogeneity reduces efficacy |
| T cells | Radiation-enhanced immune activation + DAMP release | Enhance ICD via DAMPs release; amplify CD8+ T-cell infiltration | Risk of cytokine release syndrome; dose optimization challenges |
| Blood cells | |||
| Red blood cells | Oxygen delivery via hemoglobin | Broad availability; good innate biocompatibility and deformability | Limited drug-loading efficiency; hypoxia modulation requires sustained delivery |
| Platelets | Hypoxia-responsive miR-223 delivery | Hypoxia-responsive trafficking; deliver oxygen and suppress DNA repair | Short lifespan (8–10 days); complex engineering requirements |
| Gram-negative bacteria | |||
| Escherichia coli | Redox-modulating factor secretion | Good tumor targeting; low bacterial toxicity; easy to modify and express gene | Potential risk of infection of normal tissues |
| Salmonella | Flagellum-driven tumor penetration + CDT toxin | Good tumor targeting and penetration; intracellular delivery function | Virulence of bacteria; potential risk in normal tissues infection |
| Gram-positive bacteria | |||
| Clostridium | Hypoxia-triggered spore germination | Hypoxia tropism; colonization; immune response and destroy tumor hypoxic areas | Difficulty to culture in normal environment; high risk of wound infection |
| Bifidobacterium | Hypoxia-triggered spore germination | Natural tendency and selective tumor targeting; ability to penetrate the blood-brain barrier | Difficulty to culture in normal environment; genetic modification complexity |
| Cyanobacteria | |||
| Nostoc | Light-triggered superoxide generation | Light-driven oxygen production to reverse tumor hypoxia; rapid metabolic degradation | Depth dependency of light penetration; engineering challenges in surface modification |
| Spirulina | Light-triggered superoxide generation | Good tumor targeting; improving hypoxic microenvironment | Biosafety and potential toxicity in humans has not been established |
Biosafety concerns of living cells, including uncontrolled proliferation and immunogenicity, further cause potential toxic risks in clinical application. Genetic “self-exploding” circuits and radiolabeled imaging probes offer real-time monitoring and regulated strategies to ensure living cell radiosensitization efficiency and biosafety. These circuits typically use radiation/hypoxia-activated promoters (RecA/HREs) to drive caspase-9 or cytosine deaminase expression. For example, engineered E. coli with X-ray-triggered RecA promoters achieved > 90% bacterial lysis within post-irradiation in murine colon tumors [109]. While validated preclinically, immune clearance and long-term circuit stability require optimization. Progress will not stop here and the living cell-based radiosensitization for enhancing the radiotherapy effect on tumors is still a question warranting further investigation. Clinical translation also faces significant regulatory hurdles. Living cells are classified as advanced therapy medicinal products (ATMPs), which require the following: (i) GMP-compliant manufacturing; (ii) potency assays; and (iii) stability profiling. Autologous therapies incur high costs, necessitating allogeneic ‘off-the-shelf’ solutions. Key challenges include the following: (i) batch variability in cell functionality (e.g., MSC chemotaxis); (ii) cryopreservation recovery < 60% for bacteria; and (iii) endotoxin control in Gram-negative systems. FDA/EMA guidelines mandate 18-month tumorigenicity/immunogenicity monitoring (e.g., anti-CAR antibodies) [110]. As the developments of synthetic biology methods and precision manufacturing technology, living cells are promising to serve as a high-efficacy radiosensitization strategy and widely utilized in clinical cancer treatment.
Data availability statement
The data are available from the corresponding author upon reasonable request.
Author contributions
We acknowledge the contributions of Hengrui Liu, who conceived and presented the idea, and Haonan Xu who performed the literature research and wrote the manuscript. Renjie Feng was involved in drawing pictures and participated in the revision. All authors read and approved the final manuscript.
Funding
No funding or sponsorship was received for this study.
Conflict of interest
The authors declare that there are no conflicts of interest.
Graphical abstract
Highlights
- Dual-function living cells: Eukaryotic (stem/immune/blood cells) and prokaryotic (bacteria/cyanobacteria) systems serve as tumor-targeting carriers and dynamic TME modulators, enabling spatiotemporally preciseradiosensitization.
- Mechanism diversity: Stem cells inhibit DNA repair via STAT3 downregulation; bacteria reverse hypoxia through catalase/nattokinase expression; NK cells induce ferroptosis via IFN-γ; cyanobacteria generate O2 via photosynthesis.
- Clinical synergy: CAR-T/radiotherapy combinations demonstrate sustained responses in lymphoma/liver metastasis patients (phase I/II trials), while engineered Salmonella enhances tumor targeting in melanoma.
In brief
This review systematically summarizes the latest research progress in utilizing living cells (eukaryotic and prokaryotic) for tumor radiosensitization. Dual roles as targeted carriers for radiosensitizer delivery and dynamic modulators of the tumor microenvironment (TME) are highlighted. Key mechanisms include the following: (1) stem cell-mediated STAT3/DNA repair inhibition; (2) immune cell-driven immunogenic cell death (ICD) synergy; (3) bacterial enzymatic hypoxia alleviation; and (4) cyanobacterial photosynthetic oxygen generation. The integration of these approaches overcomes conventional radiotherapy limitations by synergistically amplifying radiation-induced damage, while suppressing resistance pathways. We further discuss translational challenges in biosafety, manufacturing scalability, and clinical trial design, providing a roadmap for future development.
References
- Goldstein M, Kastan MB. The DNA damage response: implications for tumor responses to radiation and chemotherapy. Annu Rev Med 2015;66:129-43. [PMID: 25423595 DOI: 10.1146/annurev-med-081313-121208]
- Jarosz-Biej M, Smolarczyk R, Cichoń T, Kułach N. Tumor microenvironment as a “Game Changer” in cancer radiotherapy. Int J Mol Sci 2019;20(13):3212. [PMID: 31261963 DOI: 10.3390/ijms20133212]
- McLaughlin M, Patin EC, Pedersen M, Wilkins A, Dillon MT, et al. Inflammatory microenvironment remodelling by tumour cells after radiotherapy. Nat Rev Cancer 2020;20(4):203-17. [PMID: 32161398 DOI: 10.1038/s41568-020-0246-1]
- Price JM, Prabhakaran A, West CML. Predicting tumour radiosensitivity to deliver precision radiotherapy. Nat Rev Clin Oncol 2023;20(2):83-98. [PMID: 36477705 DOI: 10.1038/s41571-022-00709-y]
- Wang H, Mu X, He H, Zhang X-D. Cancer radiosensitizers. Trends Pharmacol Sci 2018;39(1):24-48. [PMID: 29224916 DOI: 10.1016/j.tips.2017.11.003]
- Higgins GS, O’Cathail SM, Muschel RJ, McKenna WG. Drug radiotherapy combinations: review of previous failures and reasons for future optimism. Cancer Treat Rev 2015;41(2):105-13. [PMID: 25579753 DOI: 10.1016/j.ctrv.2014.12.012]
- Yu W, Liu R, Zhou Y, Gao H. Size-tunable strategies for a tumor targeted drug delivery system. ACS Cent Sci 2020;6(2):100-16. [PMID: 32123729 DOI: 10.1021/acscentsci.9b01139]
- Sheibani M, Azizi Y, Shayan M, Nezamoleslami S, Eslami F, et al. Doxorubicin-induced cardiotoxicity: an overview on pre-clinical therapeutic approaches. Cardiovasc Toxicol 2022;22(4):292-310. [PMID: 35061218 DOI: 10.1007/s12012-022-09721-1]
- Nicolay NH, Lopez Perez R, Debus J, Huber PE. Mesenchymal stem cells – a new hope for radiotherapy-induced tissue damage? Cancer Lett 2015;366(2):133-40. [PMID: 26166559 DOI: 10.1016/j.canlet.2015.06.012]
- Sadeghzadeh M, Bornehdeli S, Mohahammadrezakhani H, Abolghasemi M, Poursaei E, et al. Dendritic cell therapy in cancer treatment; the state-of-the-art. Life Sci 2020;254:117580. [PMID: 32205087 DOI: 10.1016/j.lfs.2020.117580]
- Kouhsari E, Ghadimi-Daresajini A, Abdollahi H, Amirmozafari N, Mahdavi SR, et al. The potential roles of bacteria to improve radiation treatment outcome. Clin Transl Oncol 2018;20(2):127-39. [PMID: 28623514 DOI: 10.1007/s12094-017-1701-7]
- Zhu M, Yang M, Zhang J, Yin Y, Fan X, et al. Immunogenic cell death induction by ionizing radiation. Front Immunol 2021;12:705361. [PMID: 34489957 DOI: 10.3389/fimmu.2021.705361]
- Xia D, Li J, Feng L, Gao Z, Liu J, et al. Advances in targeting drug biological carriers for enhancing tumor therapy efficacy. Macromol Biosci 2023;23(12):e2300178. [PMID: 37466216 DOI: 10.1002/mabi.202300178]
- Shirvani SM, Huntzinger CJ, Melcher T, Olcott PD, Voronenko Y, et al. Biology-guided radiotherapy: redefining the role of radiotherapy in metastatic cancer. Br J Radiol 2021;94(1117):20200873. [PMID: 33112685 DOI: 10.1259/bjr.20200873]
- Zhang T, Lin R, Wu H, Jiang X, Gao J. Mesenchymal stem cells: a living carrier for active tumor-targeted delivery. Adv Drug Deliv Rev 2022;185:114300. [PMID: 35447165 DOI: 10.1016/j.addr.2022.114300]
- Lin Z, Wu Y, Xu Y, Li G, Li Z, et al. Mesenchymal stem cell-derived exosomes in cancer therapy resistance: recent advances and therapeutic potential. Mol Cancer 2022;21(1):179. [PMID: 36100944 DOI: 10.1186/s12943-022-01650-5]
- Zhou B, Szymanski CM, Baylink A. Bacterial chemotaxis in human diseases. Trends Microbiol 2023;31(5):453-67. [PMID: 36411201 DOI: 10.1016/j.tim.2022.10.007]
- Krishnan N, Fang RH, Zhang L. Engineering of stimuli-responsive self-assembled biomimetic nanoparticles. Adv Drug Deliv Rev 2021;179:114006. [PMID: 34655662 DOI: 10.1016/j.addr.2021.114006]
- Deutsch E, Chargari C, Galluzzi L, Kroemer G. Optimising efficacy and reducing toxicity of anticancer radioimmunotherapy. Lancet Oncol 2019;20(8):e452-63. [PMID: 31364597 DOI: 10.1016/S1470-2045(19)30171-8]
- Huang R-X, Zhou P-K. DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer. Signal Transduct Target Ther 2020;5(1):60. [PMID: 32355263 DOI: 10.1038/s41392-020-0150-x]
- Guo S, Yao Y, Tang Y, Xin Z, Wu D, et al. Radiation-induced tumor immune microenvironments and potential targets for combination therapy. Signal Transduct Target Ther 2023;8(1):205. [PMID: 37208386 DOI: 10.1038/s41392-023-01462-z]
- Wang Y, Liu Z-G, Yuan H, Deng W, Li J, et al. The reciprocity between radiotherapy and cancer immunotherapy. Clin Cancer Res 2019;25(6):1709-17. [PMID: 30413527 DOI: 10.1158/1078-0432.CCR-18-2581]
- Shi LZ, Bonner JA. Bridging radiotherapy to immunotherapy: the IFN-JAK-STAT axis. Int J Mol Sci 2021;22(22):12295. [PMID: 34830176 DOI: 10.3390/ijms222212295]
- Wu H-H, Zhou Y, Tabata Y, Gao J-Q. Mesenchymal stem cell-based drug delivery strategy: from cells to biomimetic. J Control Release 2019;294:102-13. [PMID: 30553849 DOI: 10.1016/j.jconrel.2018.12.019]
- Shah Z, Tian L, Li Z, Jin L, Zhang J, et al. Human anti-PSCA CAR macrophages possess potent antitumor activity against pancreatic cancer. Cell Stem Cell 2024;31(6):803-17.e6. [PMID: 38663406 DOI: 10.1016/j.stem.2024.03.018]
- Pauwels B, Wouters A, Peeters M, Vermorken JB, Lardon F. Role of cell cycle perturbations in the combination therapy of chemotherapeutic agents and radiation. Future Oncol 2010;6(9):1485-96. [PMID: 20919831 DOI: 10.2217/fon.10.104]
- Langroudi L, Jamshidi-Adegani F, Shafiee A, Rad SMAH, Keramati F, et al. MiR-371-373 cluster acts as a tumor-suppressor-miR and promotes cell cycle arrest in unrestricted somatic stem cells. Tumour Biol 2015;36(10):7765-74. [PMID: 25941115 DOI: 10.1007/s13277-015-3519-7]
- Cheng P-T, Cheng Y-C, Oner M, Li Y-H, Chen M-C, et al. Antrodia salmonea extract inhibits cell proliferation through regulating cell cycle arrest and apoptosis in prostate cancer cell lines. Chin J Physiol 2022;65(4):209-14. [PMID: 36073569 DOI: 10.4103/cjp.cjp_78_21]
- Heylmann D, Ponath V, Kindler T, Kaina B. Comparison of DNA repair and radiosensitivity of different blood cell populations. Sci Rep 2021;11(1):2478. [PMID: 33510180 DOI: 10.1038/s41598-021-81058-1]
- Huang Z, Guo H, Lin L, Li S, Yang Y, et al. Application of oncolytic virus in tumor therapy. J Med Virol 2023;95(4):e28729. [PMID: 37185868 DOI: 10.1002/jmv.28729]
- Mei J, Wang R, Xia D, Yang X, Zhou W, et al. BRCA1 is a novel prognostic indicator and associates with immune cell infiltration in hepatocellular carcinoma. DNA Cell Biol 2020;39(10):1838-49. [PMID: 32876480 DOI: 10.1089/dna.2020.5644]
- Forster JC, Douglass MJJ, Phillips WM, Bezak E. Monte Carlo simulation of the oxygen effect in DNA damage induction by ionizing radiation. Radiat Res 2018;190(3):248-61. [PMID: 29953346 DOI: 10.1667/RR15050.1]
- Caputo F, De Nicola M, Ghibelli L. Pharmacological potential of bioactive engineered nanomaterials. Biochem Pharmacol 2014;92(1):112-30. [PMID: 25175739 DOI: 10.1016/j.bcp.2014.08.015]
- Fliervoet LAL, Mastrobattista E. Drug delivery with living cells. Adv Drug Deliv Rev 2016;106(Pt A):63-72. [PMID: 27129442 DOI: 10.1016/j.addr.2016.04.021]
- Ye M, Liu T, Miao L, Ji H, Xu Z, et al. Cisplatin-encapsulated TRAIL-engineered exosomes from human chorion-derived MSCs for targeted cervical cancer therapy. Stem Cell Res Ther 2024;15(1):396. [PMID: 39497209 DOI: 10.1186/s13287-024-04006-6]
- Huo M, Wang L, Zhang L, Wei C, Chen Y, et al. Photosynthetic tumor oxygenation by photosensitizer-containing cyanobacteria for enhanced photodynamic therapy. Angew Chem Int Ed Engl 2020;59(5):1906-13. [PMID: 31721383 DOI: 10.1002/anie.201912824]
- Xing JL, Stea B. Molecular mechanisms of sensitivity and resistance to radiotherapy. Clin Exp Metastasis 2024;41(4):517-24. [PMID: 38231337 DOI: 10.1007/s10585-023-10260-4]
- Qin Y, You S-H, Zhang Y, Venu A, Hong Y, et al. Genetic programming by nitric oxide-sensing gene switch system in tumor-targeting bacteria. Biosensors (Basel) 2023;13(2):266. [PMID: 36832032 DOI: 10.3390/bios13020266]
- Bhat SA, Kumar V, Dhanjal DS, Gandhi Y, Mishra SK, et al. Biogenic nanoparticles: pioneering a new era in breast cancer therapeutics-a comprehensive review. Discov Nano 2024;19(1):121. [PMID: 39096427 DOI: 10.1186/s11671-024-04072-y]
- Dillon MT, Good JS, Harrington KJ. Selective targeting of the G2/M cell cycle checkpoint to improve the therapeutic index of radiotherapy. Clin Oncol (R Coll Radiol) 2014;26(5):257-65. [PMID: 24581946 DOI: 10.1016/j.clon.2014.01.009]
- Tian Z, Yu T, Liu J, Wang T, Higuchi A. Introduction to stem cells. Prog Mol Biol Transl Sci 2023;199:3-32. [PMID: 37678976 DOI: 10.1016/bs.pmbts.2023.02.012]
- Liu W, Cheng G, Cui H, Tian Z, Li B, et al. Theoretical basis, state and challenges of living cell-based drug delivery systems. Theranostics 2024;14(13):5152-83. [PMID: 39267776 DOI: 10.7150/thno.99257]
- Bianchi ME, Mezzapelle R. The chemokine receptor CXCR4 in cell proliferation and tissue regeneration. Front Immunol 2020;11:2109. [PMID: 32983169 DOI: 10.3389/fimmu.2020.02109]
- Gopalarethinam J, Nair AP, Iyer M, Vellingiri B, Subramaniam MD. Advantages of mesenchymal stem cell over the other stem cells. Acta Histochem 2023;125(4):152041. [PMID: 37167794 DOI: 10.1016/j.acthis.2023.152041]
- Sun X, Li K, Zha R, Liu S, Fan Y, et al. Erratum: Preventing tumor progression to the bone by induced tumor-suppressing MSCs: Erratum. Theranostics 2022;12(14):6155-6. [PMID: 36168625 DOI: 10.7150/thno.77186]
- Yun WS, Shim MK, Lim S, Song S, Kim J, et al. Mesenchymal stem cell-mediated deep tumor delivery of gold nanorod for photothermal therapy. Nanomaterials (Basel) 2022;12(19):3410. [PMID: 36234538 DOI: 10.3390/nano12193410]
- Xiao J, Zeng L, Ding S, Chen Y, Zhang X, et al. Tumor-tropic adipose-derived mesenchymal stromal cell mediated Bi2 Se3 nano-radiosensitizers delivery for targeted radiotherapy of non-small cell lung cancer. Adv Healthc Mater 2022;11(8):e2200143. [PMID: 35195958 DOI: 10.1002/adhm.202200143]
- Wang J, Kong D, Zhu L, Wang S, Sun X. Human bone marrow mesenchymal stem cells modified hybrid baculovirus-adeno-associated viral vectors targeting 131I therapy of hypopharyngeal carcinoma. Hum Gene Ther 2020;31(23-24):1300-11. [PMID: 32940055 DOI: 10.1089/hum.2020.081]
- Sababathy M, Ramanathan G, Tan SC. Targeted delivery of gold nanoparticles by neural stem cells to glioblastoma for enhanced radiation therapy: a review. AIMS Neurosci 2022;9(3):303-19. [PMID: 36329899 DOI: 10.3934/Neuroscience.2022017]
- Busato F, Khouzai BE, Mognato M. Biological mechanisms to reduce radioresistance and increase the efficacy of radiotherapy: state of the art. Int J Mol Sci 2022;23(18):10211. [PMID: 36142122 DOI: 10.3390/ijms231810211]
- de Araújo Farias V, O’Valle F, Lerma BA, Ruiz de Almodóvar C, López-Peñalver JJ, et al. Human mesenchymal stem cells enhance the systemic effects of radiotherapy. Oncotarget 2015;6(31):31164-80. [PMID: 26378036 DOI: 10.18632/oncotarget.5216]
- He N, Kong Y, Lei X, Liu Y, Wang J, et al. MSCs inhibit tumor progression and enhance radiosensitivity of breast cancer cells by down-regulating Stat3 signaling pathway. Cell Death Dis 2018;9(10):1026. [PMID: 30297887 DOI: 10.1038/s41419-018-0949-3]
- Wu L, Tang Q, Yin X, Yan D, Tang M, et al. The therapeutic potential of adipose tissue-derived mesenchymal stem cells to enhance radiotherapy effects on hepatocellular carcinoma. Front Cell Dev Biol 2019;7:267. [PMID: 31781559 DOI: 10.3389/fcell.2019.00267]
- Mason KA, Neal R, Hunter N, Ariga H, Ang K, et al. CpG oligodeoxynucleotides are potent enhancers of radio- and chemoresponses of murine tumors. Radiother Oncol 2006;80(2):192-8. [PMID: 16905212 DOI: 10.1016/j.radonc.2006.07.024]
- Xia Y, Rao L, Yao H, Wang Z, Ning P, et al. Engineering macrophages for cancer immunotherapy and drug delivery. Adv Mater 2020;32(40):e2002054. [PMID: 32856350 DOI: 10.1002/adma.202002054]
- Gaspar N, Zambito G, Löwik CMWG, Mezzanotte L. Active nano-targeting of macrophages. Curr Pharm Des 2019;25(17):1951-61. [PMID: 31291874 DOI: 10.2174/1381612825666190710114108]
- Sun Z, Chen J, Chen G, Zhang C, Li C. Recent advances of engineered and artificial drug delivery system towards solid tumor based on immune cells. Biomed Mater 2022;17(2). [PMID: 35042206 DOI: 10.1088/1748-605X/ac4c8b]
- Li Z, Huang H, Tang S, Li Y, Yu X-F, et al. Small gold nanorods laden macrophages for enhanced tumor coverage in photothermal therapy. Biomaterials 2016;74:144-54. [PMID: 26454052 DOI: 10.1016/j.biomaterials.2015.09.038]
- Fu J, Wang D, Mei D, Zhang H, Wang Z, et al. Macrophage mediated biomimetic delivery system for the treatment of lung metastasis of breast cancer. J Control Release 2015;204:11-9. [PMID: 25646783 DOI: 10.1016/j.jconrel.2015.01.039]
- Zheng W, Ling S, Cao Y, Shao C, Sun X. Combined use of NK cells and radiotherapy in the treatment of solid tumors. Front Immunol 2024;14:1306534. [PMID: 38264648 DOI: 10.3389/fimmu.2023.1306534]
- Kim K-S, Choi B, Choi H, Ko MJ, Kim D-H, et al. Enhanced natural killer cell anti-tumor activity with nanoparticles mediated ferroptosis and potential therapeutic application in prostate cancer. J Nanobiotechnology 2022;20(1):428. [PMID: 36175895 DOI: 10.1186/s12951-022-01635-y]
- Zhuang M, Jiang S, Gu A, Chen X, Mingyan E. Radiosensitizing effect of gold nanoparticle loaded with small interfering RNA-SP1 on lung cancer: AuNPs-si-SP1 regulates GZMB for radiosensitivity. Transl Oncol 2021;14(12):101210. [PMID: 34517158 DOI: 10.1016/j.tranon.2021.101210]
- He J, Yan Y, Zhang J, Wei Z, Li H, et al. Synergistic treatment strategy: combining CAR-NK cell therapy and radiotherapy to combat solid tumors. Front Immunol 2023;14:1298683. [PMID: 38162672 DOI: 10.3389/fimmu.2023.1298683]
- Xia N, Haopeng P, Gong JU, Lu J, Chen Z, et al. Robo1-specific CAR-NK immunotherapy enhances efficacy of 125I seed brachytherapy in an orthotopic mouse model of human pancreatic carcinoma. Anticancer Res 2019;39(11):5919-25. [PMID: 31704816 DOI: 10.21873/anticanres.13796]
- Fan J, Adams A, Sieg N, Heger J-M, Gödel P, et al. Potential synergy between radiotherapy and CAR T-cells – a multicentric analysis of the role of radiotherapy in the combination of CAR T cell therapy. Radiother Oncol 2023;183:109580. [PMID: 36842663 DOI: 10.1016/j.radonc.2023.109580]
- Zhou Z, Mai Y, Zhang G, Wang Y, Sun P, et al. Emerging role of immunogenic cell death in cancer immunotherapy: advancing next-generation CAR-T cell immunotherapy by combination. Cancer Lett 2024;598:217079. [PMID: 38936505 DOI: 10.1016/j.canlet.2024.217079]
- Zhong L, Li Y, Muluh TA, Wang Y. Combination of CAR-T cell therapy and radiotherapy: opportunities and challenges in solid tumors (Review). Oncol Lett 2023;26(1):281. [PMID: 37274466 DOI: 10.3892/ol.2023.13867]
- Liu S, Yuan S, Liu M, Liu J, Fu S, et al. In situ tumor cell engineering reverses immune escape to enhance immunotherapy effect. Acta Pharm Sin B 2025;15(1):627-41. [PMID: 40041911 DOI: 10.1016/j.apsb.2024.08.028]
- Arndt C, Loureiro LR, Feldmann A, Jureczek J, Bergmann R, et al. UniCAR T cell immunotherapy enables efficient elimination of radioresistant cancer cells. Oncoimmunology 2020;9(1):1743036. [PMID: 32426176 DOI: 10.1080/2162402X.2020.1743036]
- Katz SC, Hardaway J, Prince E, Guha P, Cunetta M, et al. HITM-SIR: phase Ib trial of intraarterial chimeric antigen receptor T-cell therapy and selective internal radiation therapy for CEA+ liver metastases. Cancer Gene Ther 2020;27(5):341-55. [PMID: 31155611 DOI: 10.1038/s41417-019-0104-z]
- Qu C, Ping N, Kang L, Liu H, Qin S, et al. Radiation priming chimeric antigen receptor T-cell therapy in relapsed/refractory diffuse large B-cell lymphoma with high tumor burden. J Immunother 2020;43(1):32-7. [PMID: 31219975 DOI: 10.1097/CJI.0000000000000284]
- Lucas A, Lam D, Cabrales P. Doxorubicin-loaded red blood cells reduced cardiac toxicity and preserved anticancer activity. Drug Deliv 2019;26(1):433-42. [PMID: 30929538 DOI: 10.1080/10717544.2019.1591544]
- Dong H, Xu X, Wang L, Mo R. Advances in living cell-based anticancer therapeutics. Biomater Sci 2020;8(9):2344-65. [PMID: 32254876 DOI: 10.1039/d0bm00036a]
- Wang C, Huang J, Zhang Y, Jia H, Chen B. Construction and evaluation of red blood cells-based drug delivery system for chemo-photothermal therapy. Colloids Surf B Biointerfaces 2021;204:111789. [PMID: 33932889 DOI: 10.1016/j.colsurfb.2021.111789]
- Ahn S, Jung SY, Seo E, Lee SJ. Gold nanoparticle-incorporated human red blood cells (RBCs) for X-ray dynamic imaging. Biomaterials 2011;32(29):7191-9. [PMID: 21777977 DOI: 10.1016/j.biomaterials.2011.05.023]
- Chiangjong W, Netsirisawan P, Hongeng S, Chutipongtanate S. Red blood cell extracellular vesicle-based drug delivery: challenges and opportunities. Front Med (Lausanne) 2021;8:761362. [PMID: 35004730 DOI: 10.3389/fmed.2021.761362]
- Siemann DW, Horsman MR. Modulation of the tumor vasculature and oxygenation to improve therapy. Pharmacol Ther 2015;153:107-24. [PMID: 26073310 DOI: 10.1016/j.pharmthera.2015.06.006]
- Liu T, Shi C, Duan L, Zhang Z, Luo L, et al. A highly hemocompatible erythrocyte membrane-coated ultrasmall selenium nanosystem for simultaneous cancer radiosensitization and precise antiangiogenesis. J Mater Chem B 2018;6(29):4756-64. [PMID: 30450208 DOI: 10.1039/C8TB01398E]
- Zhou Z, Zhang B, Wang H, Yuan A, Hu Y, et al. Erratum: Two-stage oxygen delivery for enhanced radiotherapy by perfluorocarbon nanoparticles: Erratum. Theranostics 2020;10(7):2943. [PMID: 32194846 DOI: 10.7150/thno.42420]
- Xia D, Hang D, Li Y, Jiang W, Zhu J, et al. Au-hemoglobin loaded platelet alleviating tumor hypoxia and enhancing the radiotherapy effect with low-dose X-ray. ACS Nano 2020;14(11):15654-68. [PMID: 33108152 DOI: 10.1021/acsnano.0c06541]
- Askari S, Goldfinger LE. Roles of miR-223 in platelet function and high on-treatment platelet reactivity: a brief report and review. Genes (Basel) 2025;16(3):312. [PMID: 40149463 DOI: 10.3390/genes16030312]
- Vass SO, Jarrom D, Wilson WR, Hyde EI, Searle PF. E. coli NfsA: an alternative nitroreductase for prodrug activation gene therapy in combination with CB1954. Br J Cancer 2009;100(12):1903-11. [PMID: 19455141 DOI: 10.1038/sj.bjc.6605094]
- Liu Y, Niu L, Li N, Wang Y, Liu M, et al. Bacterial-mediated tumor therapy: old treatment in a new context. Adv Sci (Weinh) 2023;10(12):e2205641. [PMID: 36908053 DOI: 10.1002/advs.202205641]
- Wu X, Zhang J, Deng Z, Sun X, Zhang Y, et al. Bacteria-based biohybrids for remodeling adenosine-mediated immunosuppression to boost radiotherapy-triggered antitumor immune response. Biomaterials 2025;316:123000. [PMID: 39674101 DOI: 10.1016/j.biomaterials.2024.123000]
- Duo Y, Chen Z, Li Z, Li X, Yao Y, et al. Combination of bacterial-targeted delivery of gold-based AIEgen radiosensitizer for fluorescence-image-guided enhanced radio-immunotherapy against advanced cancer. Bioact Mater 2023;30:200-13. [PMID: 37663305 DOI: 10.1016/j.bioactmat.2023.05.010]
- Huang C, Wang F-B, Liu L, Jiang W, Liu W, et al. Hypoxic tumor radiosensitization using engineered probiotics. Adv Healthc Mater 2021;10(10):e2002207. [PMID: 33645010 DOI: 10.1002/adhm.202002207]
- Liu Z, Lei L, Zhang Z, Du M, Chen Z. Ultrasound-responsive engineered bacteria mediated specific controlled expression of catalase and efficient radiotherapy. Mater Today Bio 2025;31:101620. [PMID: 40104641 DOI: 10.1016/j.mtbio.2025.101620]
- Lei L, Xu H, Li M, Du M, Chen Z. Dual-pathway tumor radiosensitization strategy based on engineered bacteria capable of targeted delivery of AuNPs and specific hypoxia alleviation. J Nanobiotechnology 2025;23(1):254. [PMID: 40155884 DOI: 10.1186/s12951-025-03329-7]
- Zhang Y, Liu Y, Li T, Yang X, Lang S, et al. Engineered bacteria breach tumor physical barriers to enhance radio-immunotherapy. J Control Release 2024;373:867-78. [PMID: 39097194 DOI: 10.1016/j.jconrel.2024.07.076]
- Shi J, Ma Z, Pan H, Liu Y, Chu Y, Wang J. Biofilm-encapsulated nano drug delivery system for the treatment of colon cancer. J Microencapsul 2020;37(7):481-491. [PMID: 32700606 DOI: 10.1080/02652048.2020.1797914]
- Pan P, Dong X, Chen Y, Zeng X, Zhang X-Z. Engineered bacteria for enhanced radiotherapy against breast carcinoma. ACS Nano 2022;16(1):801-12. [PMID: 35029367 DOI: 10.1021/acsnano.1c08350]
- Zhang Y, Huang R, Jiang Y, Shen W, Pei H, et al. The role of bacteria and its derived biomaterials in cancer radiotherapy. Acta Pharm Sin B 2023;13(10):4149-71. [PMID: 37799393 DOI: 10.1016/j.apsb.2022.10.013]
- Milas L, Mason KA, Ariga H, Hunter N, Neal R, et al. CpG oligodeoxynucleotide enhances tumor response to radiation. Cancer Res 2004;64(15):5074-7. [PMID: 15289307 DOI: 10.1158/0008-5472.CAN-04-0926]
- Mason KA, Ariga H, Neal R, Valdecanas D, Hunter N, et al. Targeting toll-like receptor 9 with CpG oligodeoxynucleotides enhances tumor response to fractionated radiotherapy. Clin Cancer Res 2005;11(1):361-9. [PMID: 15671567]
- Krieg AM, Yi AK, Matson S, Waldschmidt TJ, Bishop GA, et al. CpG motifs in bacterial DNA trigger direct B-cell activation. Nature 1995;374(6522):546-9. [PMID: 7700380 DOI: 10.1038/374546a0]
- Prack Mc Cormick B, Belgorosky D, Langle Y, Balarino N, Sandes E, et al. Bacillus Calmette-Guerin improves local and systemic response to radiotherapy in invasive bladder cancer. Nitric Oxide 2017;64:22-30. [PMID: 28126499 DOI: 10.1016/j.niox.2017.01.008]
- Yang J, Wu Z, Chen Y, Hu C, Li D, et al. Pre-treatment with Bifidobacterium infantis and its specific antibodies enhance targeted radiosensitization in a murine model for lung cancer. J Cancer Res Clin Oncol 2021;147(2):411-22. [PMID: 33130941 DOI: 10.1007/s00432-020-03434-0]
- Wang W, Zheng Y, Wu Z, Wu M, Chen Y, et al. Antibody targeting of anaerobic bacteria warms cold tumors and improves the abscopal effect of radiotherapy. J Transl Med 2024;22(1):657. [PMID: 39010088 DOI: 10.1186/s12967-024-05469-0]
- Li X, Wang H, Li Z, Tao F, Wu J, et al. Oxygen switches: refueling for cancer radiotherapy. Front Oncol 2023;12:1085432. [PMID: 36873299 DOI: 10.3389/fonc.2022.1085432]
- Qiao Y, Yang F, Xie T, Du Z, Zhong D, et al. Engineered algae: a novel oxygen-generating system for effective treatment of hypoxic cancer. Sci Adv 2020;6(21):eaba5996. [PMID: 32490207 DOI: 10.1126/sciadv.aba5996]
- Li W, Zhong D, Hua S, Du Z, Zhou M. Biomineralized biohybrid algae for tumor hypoxia modulation and cascade radio-photodynamic therapy. ACS Appl Mater Interfaces 2020;12(40):44541-53. [PMID: 32935973 DOI: 10.1021/acsami.0c14400]
- Chai R, Yu L, Dong C, Yin Y, Wang S, et al. Oxygen-evolving photosynthetic cyanobacteria for 2D bismuthene radiosensitizer-enhanced cancer radiotherapy. Bioact Mater 2022;17:276-88. [PMID: 35386463 DOI: 10.1016/j.bioactmat.2022.01.014]
- Hua S, Zhao J, Li L, Liu C, Zhou L, et al. Photosynthetic bacteria-based whole-cell inorganic-biohybrid system for multimodal enhanced tumor radiotherapy. J Nanobiotechnology 2024;22(1):379. [PMID: 38943158 DOI: 10.1186/s12951-024-02654-7]
- Kosoglu MA, Hood RL, Rossmeisl JH Jr, Grant DC, Xu Y, et al. Fiberoptic microneedles: novel optical diffusers for interstitial delivery of therapeutic light. Lasers Surg Med 2011;43(9):914-20. [PMID: 22006734 DOI: 10.1002/lsm.21129]
- Liang K, Liu Q, Li P, Luo H, Wang H, Kong Q. Genetically engineered Salmonella Typhimurium: recent advances in cancer therapy. Cancer Lett 2019;448:168-81. [PMID: 30753837 DOI: 10.1016/j.canlet.2019.01.037]
- Alonso-Roman R, Mosig AS, Figge MT, Papenfort K, Eggeling C, et al. Organ-on-chip models for infectious disease research. Nat Microbiol 2024;9(4):891-904. [PMID: 38528150 DOI: 10.1038/s41564-024-01645-6]
- Branco F, Cunha J, Mendes M, Sousa JJ, Vitorino C. 3D Bioprinting models for glioblastoma: from scaffold design to therapeutic application. Adv Mater 2025;37(18):e2501994. [PMID: 40116532 DOI: 10.1002/adma.202501994]
- Nie M, Shima A, Yamamoto M, Takeuchi S. Scalable tissue biofabrication via perfusable hollow fiber arrays for cultured meat applications. Trends Biotechnol 2025;43(8):1938-60. [PMID: 40246628 DOI: 10.1016/j.tibtech.2025.02.022]
- Ishag HZA, Xiong Q, Liu M, Feng Z, Shao G. E. coli recA gene improves gene targeted homologous recombination in Mycoplasma hyorhinis. J Microbiol Methods 2017;136:49-56. [PMID: 28285864 DOI: 10.1016/j.mimet.2017.03.004]
- Abrams K, Aiuti A, Eichler H-G, Ziegler A. Considerations driving the choice in clinical trial design of cell and gene therapy products: weighing convenience versus necessity. Cytotherapy 2025;S1465-3249(25)00738-8. [PMID: 40542809 DOI: 10.1016/j.jcyt.2025.06.001]