Combined Ultrasound and Microneedle for Enhanced Transdermal Delivery: Synergistic Mechanisms and Therapeutic Advances
1Key Laboratory of Medical Imaging Precision Theranostics and Radiation Protection, College of Hunan Province, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
2Department of Medical Imaging, The Affiliated Changsha Central Hospital, Hengyang Medical School, University of South China, Changsha, China
3Institute of Medical Imaging, Hengyang Medical School, University of South China, Hengyang, China
4Department of Chemistry, University of South Florida, Tampa, Florida 33620, USA
5State Key Laboratory of Bioactive Molecules and Druggability Assessment, Guangdong Basic Research Center of Excellence for Natural Bioactive Molecules and Discovery of Innovative Drugs, College of Pharmacy, Jinan University, Guangzhou 511436, China
aThese authors contributed equally to this work.
*Correspondence to: Jianfeng Cai, E-mail: jianfengcai@usf.edu; Chao Lu, E-mail: chaolu@jnu.edu.cn
Received: May 15 2025; Revised: July 14 2025; Accepted: July 24 2025; Published Online: November 18 2025.
Cite this paper:
Lei L, Peng W, Cai J et al. Combined Ultrasound and Microneedle for Enhanced Transdermal Delivery: Synergistic Mechanisms and Therapeutic Advances. BIO Integration 2025; 1–21.
DOI: 10.15212/bioi-2025-0095. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Transdermal drug delivery (TDD) offers a non-invasive alternative to conventional administration routes, yet the efficacy of TDD is constrained by the impermeable stratum corneum (SC) of the skin, particularly for macromolecules exceeding 500 Da. While microneedle technology addresses this barrier by creating micro-scale channels, the limited penetration depth of microneedles restricts drug delivery to superficial epidermal layers. This review highlights the transformative potential of combining ultrasound and microneedles for enhanced TDD, which synergistically integrate physical disruption and energy-driven permeation enhancement. Unlike material-based or charge/magnetism-dependent strategies, ultrasound leverages multifactorial mechanisms (mechanical stress, cavitation, and thermal effects) to propel drugs through microneedle-generated pathways into deeper tissues. The ultrasound-microneedle (US-MN) system enables spatiotemporally controlled drug release with sonochemical/piezoelectric effects expanding applications in precision medicine. The underlying mechanisms, technological innovations, and clinical translation challenges of the US-MN have been critically evaluated herein, emphasizing the versatility for macromolecules and precision medicine. By bridging mechanisms with translational gaps, this work provides a roadmap for optimizing the US-MN platform, offering researchers actionable strategies to advance TDD for chronic diseases, vaccines, and targeted therapies.
Keywords
Drug delivery, microneedle, transdermal therapeutic strategies, ultrasound, ultrasound-assisted microneedle.
Introduction
Transdermal drug delivery (TDD), an innovative drug delivery system, has emerged as a promising alternative to oral administration and subcutaneous injection [1]. TDD enables non-invasive systemic drug absorption utilizing formulations, such as creams, gels, films, and patches by bypassing hepatic first-pass metabolism and gastrointestinal absorption [2, 3]. These advantages have facilitated the widespread application of TDD in chronic disease management, dermatologic therapies, and novel vaccine development [4, 5]. However, TDD faces persistent challenges due to the inherent barrier properties of the skin. As the largest organ in the human body, the skin exhibits a complex multilayered structure comprising the epidermis, dermis, and hypodermis [6]. The stratum corneum (SC), which is the outermost epidermal layer (10–20 μm thick), serves as a formidable barrier to the penetration of exogenous substances, particularly limiting the effective permeation of macromolecular drugs >500 Da in size, such as gene therapies, proteins, and antigens [7–9]. Overcoming this barrier remains a critical challenge in advancing TDD technologies.
To address this limitation, TDD strategies have evolved significantly. Initial approaches relied on drugs with inherent skin permeability, followed by the incorporation of chemical permeation enhancers to improve the delivery of small molecules. More recently, physical techniques, including thermal ablation, electroporation, ultrasound (US), microneedles (MNs), and combined strategies involving two or more of these techniques, have been developed to enhance the delivery of macromolecules and vaccines [6, 10]. Among the physical enhancement methods, MNs and US have garnered substantial attention in TDD research due to the minimally invasive nature, operational simplicity, and precise local delivery capabilities [11–13]. MNs facilitate drug permeation by creating micron-scale channels that bypass the SC barrier. In addition, US leverages multifactorial biophysical effects, including mechanical, cavitation, and thermal mechanisms, to enhance drug permeation [14].
However, the penetration depth of MNs is typically confined to the epidermal layer, thereby limiting effective drug delivery to deeper dermal or subcutaneous tissues [15, 16]. This limitation stems from the extracellular matrix (ECM), which functions as an additional barrier layer that compromises the efficiency of TDD. Even when drugs successfully traverse the SC, the dense ECM persists as a secondary barrier, hindering access to deeper pathologic regions or subsequent permeation into vascular or lymphatic systems [17].
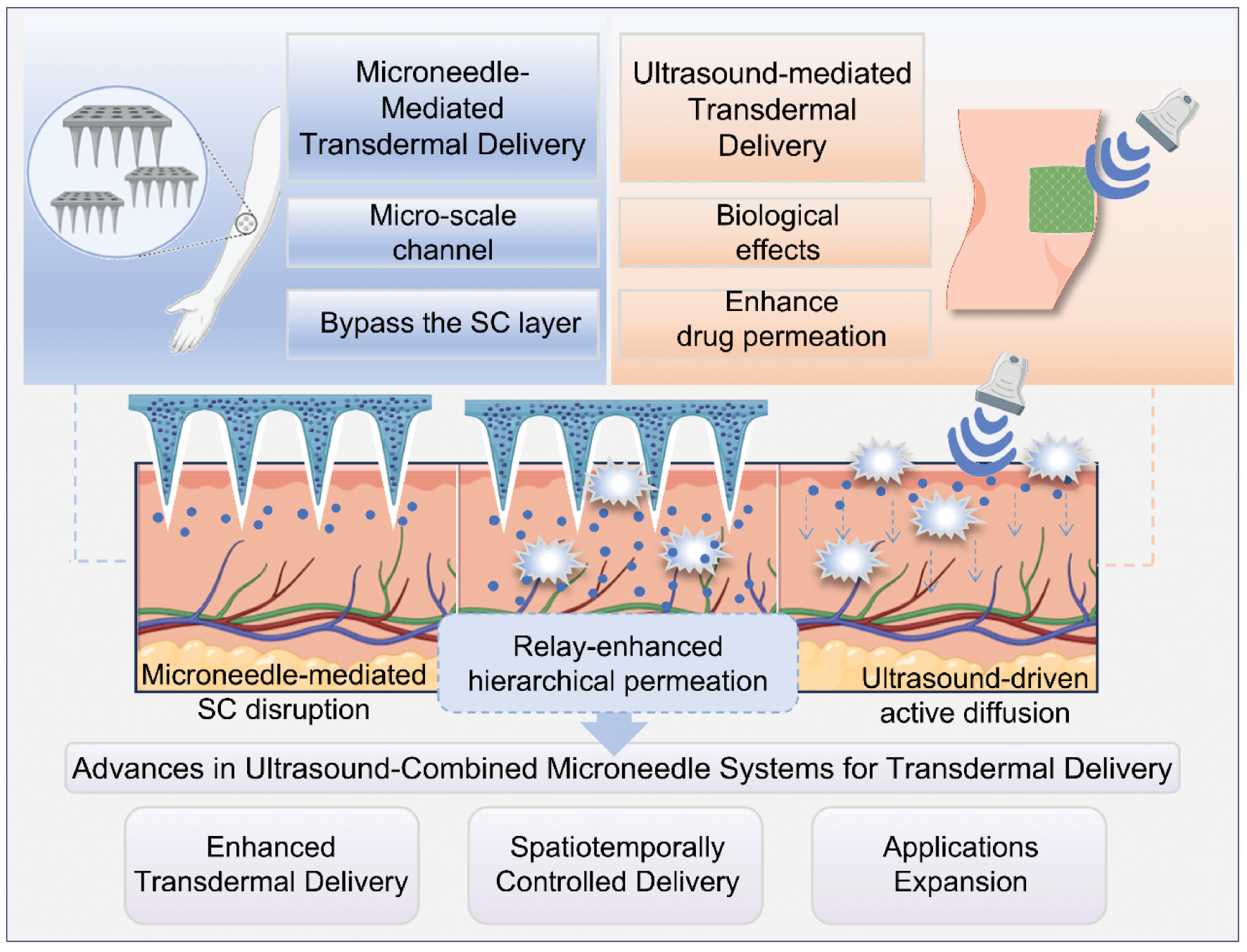
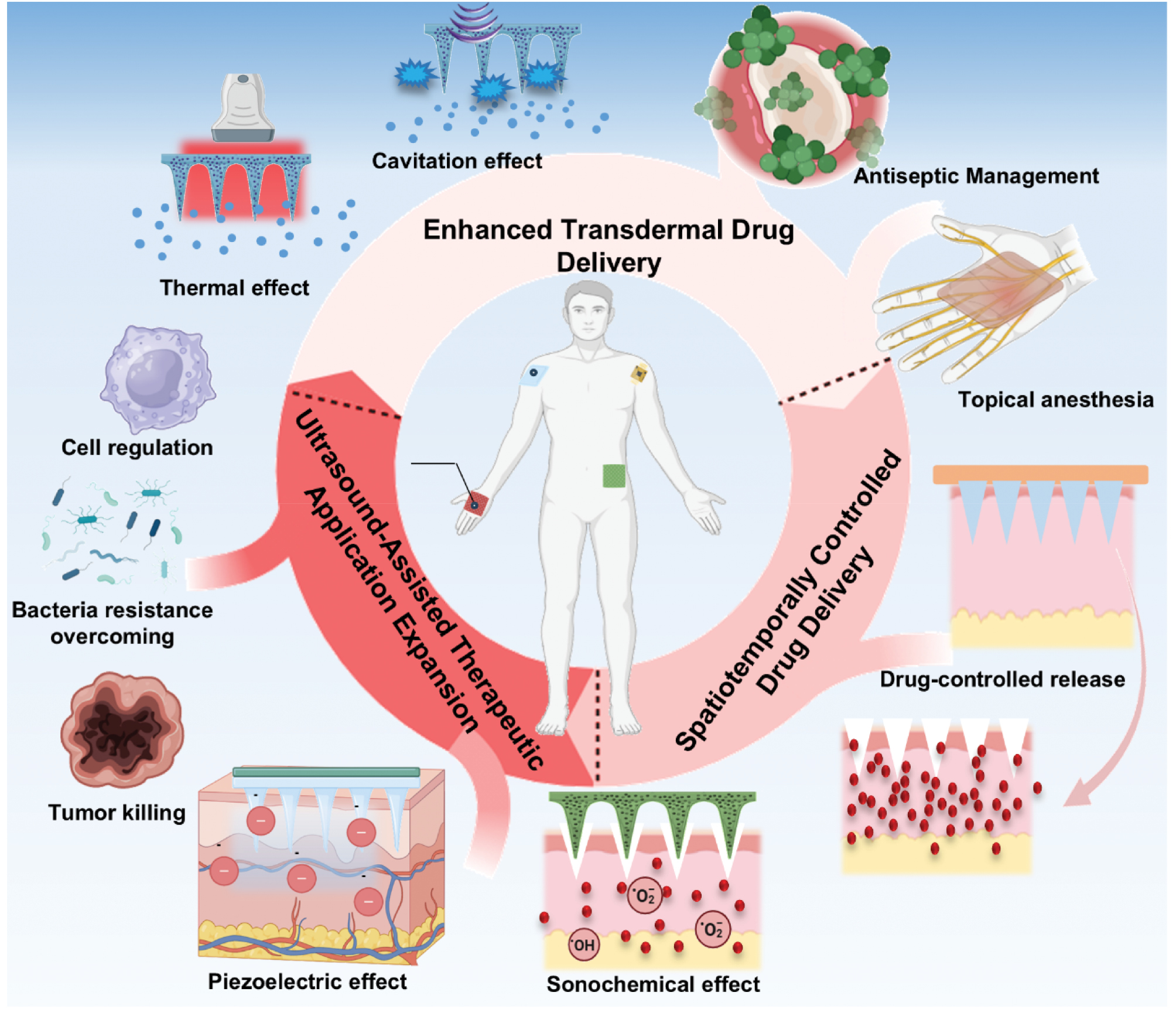
To overcome this constraint, a strategy of integrating external energy-assisted mechanisms has been explored to augment drug permeation. For example, magnetic field-driven drug targeting and iontophoresis (combining MNs with electroosmotic flow) enable directional transport of magnetic or charged drug molecules, respectively [18, 19]. The requirement for specific drug properties (e.g., magnetism or charge) limits broader applicability. Notably, the combination of US and MNs has shown exceptional potential in achieving effective drug delivery to deeper tissues. US enables drugs traversing MN-created channels to actively diffuse into deeper tissues without requiring drug modifications [13, 14]. Moreover, the combination of US and MNs effectively addresses a critical limitation of US-mediated TDD—its characteristically delayed therapeutic onset. A seminal study demonstrated that standalone US application requires a 2h post-irradiation period to achieve significant lidocaine permeation enhancement. In stark contrast, US-MN co-administration accelerated permeation kinetics by >20-fold compared to US alone, establishing immediate therapeutic efficacy [20]. To date, the combination of US and MNs has been successfully applied across diverse diseases. Unlike conventional synergistic approaches based on mechanistic superposition, the US-MN platform achieves superior TDD efficiency via a relay-enhanced hierarchical permeation mechanism that synergistically combines MN-mediated SC disruption with US-driven active diffusion into deeper tissues [21, 22]. Furthermore, the spatiotemporal precision of US allows on-demand drug release from MN reservoirs. Indeed, the combination could accelerate release kinetics to establish early therapeutic windows and inhibit disease progression [23, 24]. The sonochemical and piezoelectric effects of US further expand the therapeutic application in disease management, offering novel opportunities for precision medicine [25] (Figure 1).
Figure 1 Schematic illustration of the ultrasound-combined microneedle systems for enhanced transdermal drug delivery.
This review comprehensively examined the US-MN combination for enhanced TDD, focusing on the synergistic mechanisms and technological innovations. Current challenges in clinical translation were critically evaluated and future directions for this synergistic platform are proposed. By introducing a hierarchical permeation framework, this work aimed to provide new perspectives for advancing efficient drug delivery and precision therapeutics in TDD.
MNs: an emerging powerful platform for TDD
Advances in TDD
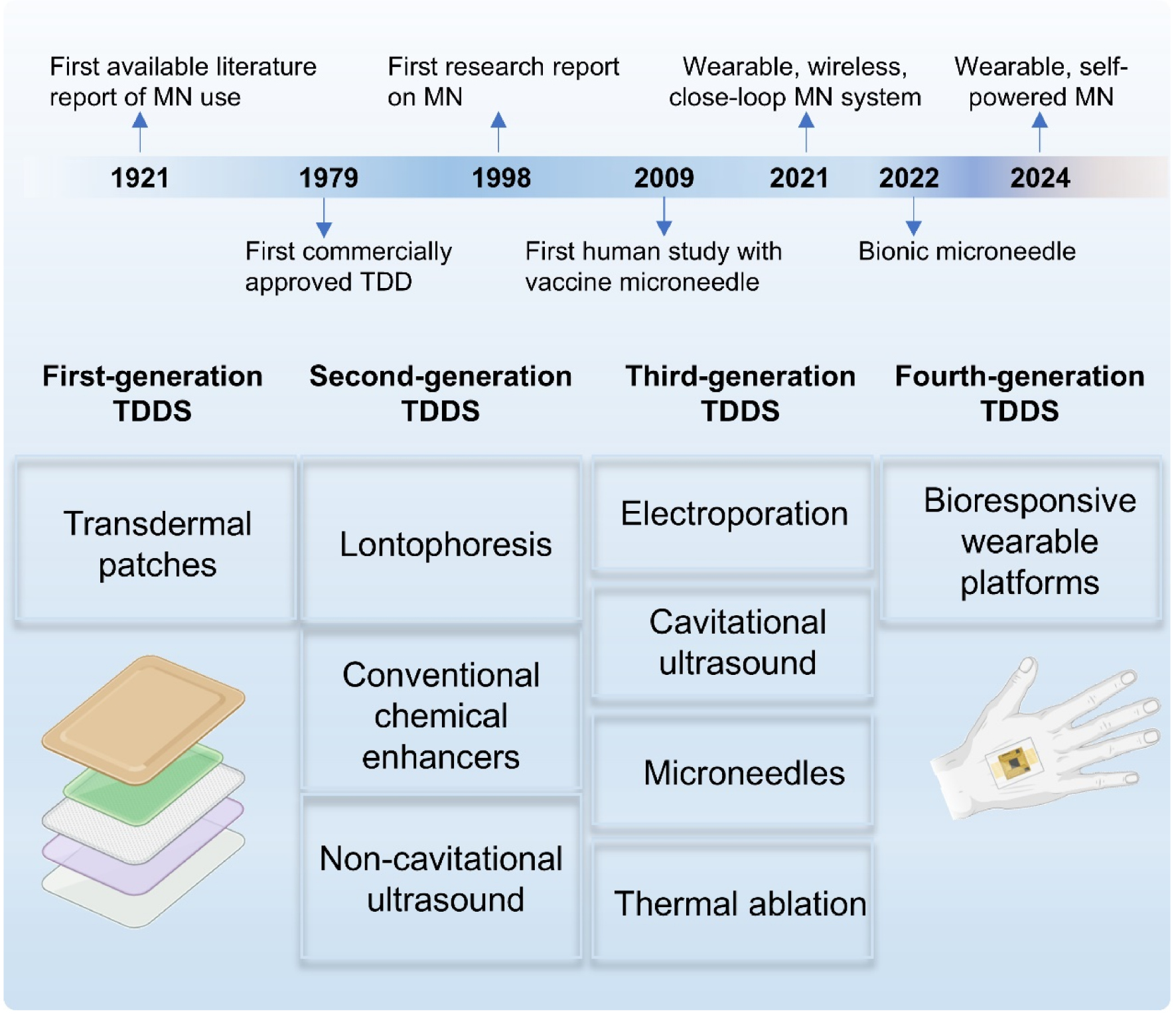
The advent of the first commercially approved transdermal patch (hyoscine hydrobromide adhesive matrix) in 1979 constituted a pivotal advancement in TDD and catalyzed paradigm shifts in non-invasive pharmaceutical administration [26]. TDD facilitates controlled permeation of active pharmaceutical ingredients through the SC barrier by functioning as a non-invasive delivery modality with subsequent sequential diffusion across the epidermal, dermal, and hypodermal layers. Systemic bioavailability is achieved via capillary uptake into the peripheral circulation, enabling both localized therapeutic targeting and systemic pharmacologic effects, while circumventing gastrointestinal degradation and hepatic first-pass metabolism [5, 7]. TDD exhibits superior pharmacokinetic advantages compared to conventional oral or parenteral routes, including the following: (1) sustained zero-order release kinetics minimizing plasma concentration fluctuations; (2) elimination of gastrointestinal irritation and drug-enzyme interactions; (3) enhanced patient adherence through simplified self-administration protocols; and (4) programmable dose titration via patch surface area modulation [27, 28]. These attributes collectively position TDD as a transformative platform for chronic disease management and biologics delivery with growing adoption across neurology, endocrinology, and oncology therapeutics.
The technological evolution of TDD has progressed through four distinct generations, each marked by paradigm-shifting innovations [29]. First-generation TDD systems relied on passive transdermal transport of lipophilic small molecules but was constrained by inherent limitations in delivering macromolecular or polar therapeutics [30]. Second- and third-generation TDD systems addressed these barriers through reversible disruption of the SC or the application of additional driving forces, such as modifying drug properties or using physical methods to create micropores in the SC, thereby enhancing drug delivery efficiency [31]. The current fourth generation TDD systems integrate bioresponsive electronics with wearable platforms, enabling closed-loop drug release control through real-time biomarker monitoring [30]. These advances have led to an increase in the scope and precision of TDD systems.
Current research in TDD primarily focuses on second- and third-generation systems with an aim to improve skin permeability using a range of strategies, including iontophoresis (electro-osmosis), electroporation (using brief high-voltage pulses to create temporary aqueous channels in the skin) [32], thermal ablation (utilizing heat to enhance skin permeability), lasers (which melt the SC), chemical permeation enhancers, and nanocarriers. Among these strategies, chemical permeation enhancers function by altering the conformation of proteins, lipid fluidity, or the solvent properties of the SC, thereby improving drug permeation. These enhancers have been extensively used in the treatment of conditions, such as Alzheimer’s disease and migraines [33, 34]. Nanocarriers, such as solid lipid nanoparticles (SLNs), nanostructured lipid carriers, and liposomes, benefit from a lipidic composition and interactions with the lipid layers of skin, which results in enhanced drug loading capacity and improved permeability [7, 35]. In addition, an emerging platform – MNs which create micron-scale pores in the SC to enhance drug permeation [7], is gaining increasing attention due to its simplicity, low invasiveness, and transdermal ability [36] (Figure 2).
Figure 2 Advances in transdermal drug delivery.
MN-mediated TDD
MNs, an innovative method for TDD, were first conceptualized in the 1970s and officially introduced in 1998 [37]. These systems use micrometer-scale needles to create temporary microchannels in the SC, facilitating efficient drug delivery into the dermal layer. This approach improves drug bioavailability and avoids metabolic processes in the digestive system, significantly reducing patient discomfort compared to traditional injections [38–40].
Types of MN systems
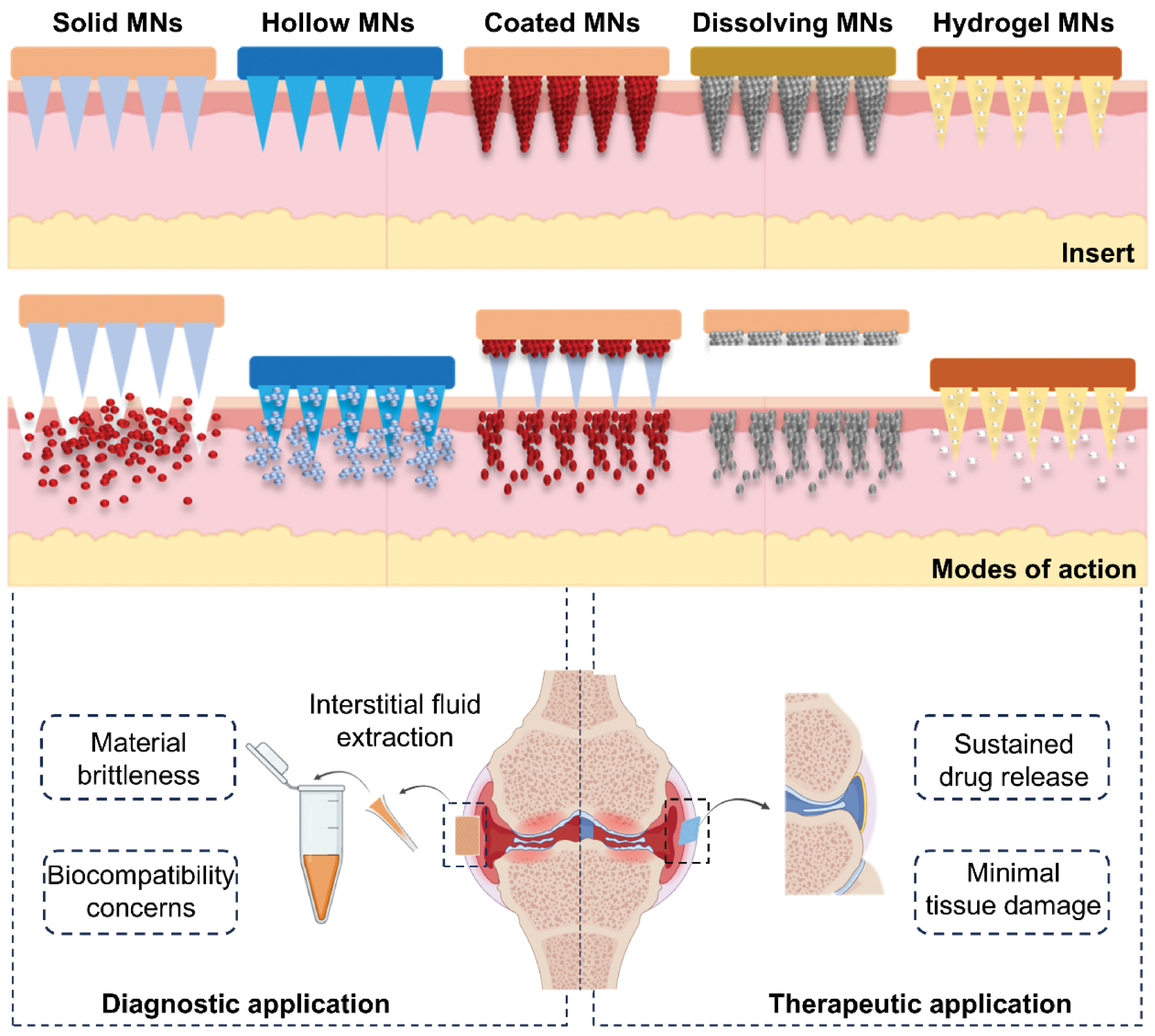
MN systems are categorized into the following five MN types: solid; hollow; coated; dissolving; and hydrogel. Each MN type has distinct mechanisms and materials, making MNs suitable for diverse diagnostic and therapeutic applications (Figure 3). Solid MNs, made from materials (such as stainless steel, silicon, or titanium) create reversible microchannels in the skin, which promotes passive drug diffusion [41]. However, the short duration of action, limited to 24 h due to the natural healing process of the skin, restricts the use of solid MNs for prolonged drug delivery [42]. Coated MNs, in which the drug is applied to the needle surface, provide rapid drug release upon skin penetration [43], although limitations related to coating capacity and uniformity are an issue. Hollow MNs, with microfluidic channels, allow continuous drug delivery and have advanced to clinical trials for vaccine delivery and TDD applications. However, the fragile structure and complex manufacturing process of hollow MNs increase the risk of breakage and limit reliability [44].
Figure 3 Modes of action for different types of microneedles.
Dissolving MNs, which are fabricated from biodegradable polymers through techniques, such as micro-molding, stretch lithography, and 3D printing, have had a significant reduction in cost and an expanding market, which have substantially enhanced patient accessibility [45]. The ability to dissolve within the skin ensures painless administration and precise drug delivery, making dissolving MNs particularly effective for treating deep-seated tumors, including melanoma and breast cancer [46, 47]. Hydrogel MNs represent cutting-edge medical technology and are typically constructed from cross-linked hydrogels, like gelatin methacrylate, hyaluronic acid methacrylate, and PVA-dextran. Upon insertion, these MNs expand, facilitating targeted drug delivery [48, 49]. By modifying the chemical structure of hydrogels, responsive variants can be developed for the treatment of specific diseases [50]. The engineering precision of these MNs allows for remarkable adaptability in therapeutic applications.
Solid and hollow MNs are commonly used for diagnostic purposes in clinical settings, such as interstitial fluid extraction, although material brittleness and biocompatibility are potentially problematic [51]. In contrast, polymer-based dissolving and hydrogel MNs excel in therapeutic applications. Dissolving MNs hold promise for transdermal cell delivery with substantial potential in innovative applications, such as vaccine delivery and chronic disease management [52]. Hydrogels mimic the ECM environment, making hydrogels invaluable in cell culture studies [53]. The microchannel architecture of hydrogels also enables the absorption of interstitial fluid, which offers the potential for biomarker detection and real-time health monitoring [54, 55]. These advances highlight the progress of MN technology towards safer, more adaptable, and clinically versatile applications in precision medicine [37, 47].
Development of MN-mediated TDD
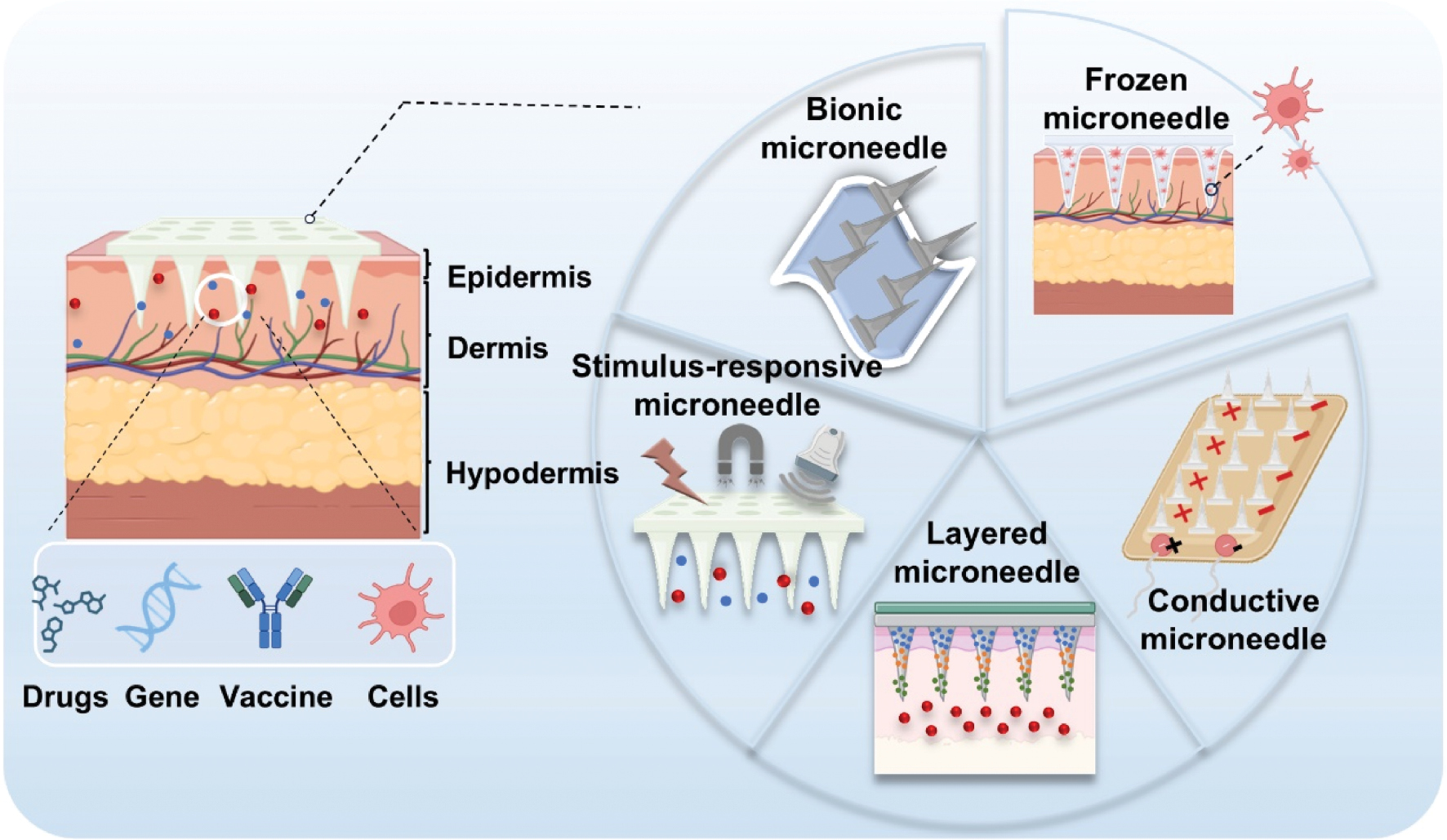
MNs, as a transformative biomedical technology, have shown considerable promise in TDD and disease treatment. However, conventional smooth-tapered MNs face clinical translation barriers due to insufficient mechanical adaptability and limited drug-controlled release capabilities [56, 57]. Recent breakthroughs in smart-responsive systems and bioinspired design strategies offer promising solutions to overcome these challenges. Innovations in MN systems, by leveraging stimuli-responsive mechanisms and biologically inspired architectures, are progressively addressing the limitations of traditional MNs, significantly enhancing drug delivery efficiency and therapeutic outcomes. Moreover, wearable, wireless, and closed-loop MN systems provide patients with more convenient, efficient, and personalized treatment options (Figure 4).
Figure 4 Development of new microneedle osmotic promotion systems for therapeutics.
The integration of smart-responsive systems with MN technology has enabled the development of stimuli-responsive MNs, creating a powerful platform for spatiotemporally controlled drug delivery. Stimuli-responsive MNs enable spatiotemporal drug delivery by recognizing endogenous or exogenous signals to undergo structural transformations, such as degradation, swelling, rupture, or collapse [58]. Exogenous triggers include US, light, electricity, force, magnetic fields, and temperature. For example, a magnetically responsive MN robot guided by external magnetic fields efficiently delivers macromolecular drugs through intestinal barriers, demonstrating blood glucose regulation in preclinical models [59]. Hydrothermal-responsive MNs (HRMAM), which are composed of polycaprolactone, release docetaxel upon exposure to water and generate localized heat for tumor ablation. This system has been reported to achieve 75.11% and 72.29% tumor inhibition in melanoma and breast cancer models, combining hydrothermal therapy with chemotherapy for deep penetration [60]. Drug delivery based on light-responsive MNs can be categorized into photothermal and photochemical responses [16]. Photothermal MNs use photo-absorbers, such as polydopamine, to convert light energy into heat, triggering responsive stimuli through thermosensitive materials [61]. In contrast, photochemical MNs absorb light energy to break the photosensitive covalent bonds in the MN platform, thereby triggering the release of encapsulated drugs [62]. The design of endogenous stimulus-responsive MNs is primarily based on the physiologic changes in disease sites; commonly studied endogenous stimuli include pH [63], glucose [64, 65], and enzymes [66]. Several reviews have summarized the development of these stimulatory probes, so the progress of stimuli-responsive MNs will not be a focus of the current review.
Multilayer MNs offer unique advantages to meet the need for sequential drug delivery. A Christmas tree-like MN patch encapsulating fluorouracil, leucovorin, irinotecan, and oxaliplatin (FOLFIRINOX) chemotherapeutics mimics clinical dosing schedules through layer-by-layer fabrication. The patch adheres to moist tumor surfaces and releases drugs in batches, enhancing spatiotemporal control [67, 68]. It has been shown that electro-stimulated MN patches, which enable rapid drug penetration (>1.0 mm depth within 5 min) under microcurrent stimulation, while activating immune responses against deep fungal infections [69]. Preserving therapeutic efficacy throughout the drug delivery process is of paramount importance. Frozen MNs (cryoMNs) have emerged as a breakthrough in immune cell delivery, enabling the intradermal transfer of viable dendritic cells (DCs), while preserving cellular integrity and inducing antigen-specific immune responses. It has shown that cryoMNs loaded with ovalbumin-pulsed DCs significantly delay tumor progression in melanoma models, outperforming both intravenous and subcutaneous administration methods. This finding highlights the promising potential for use in cancer vaccine applications [52, 70].
Bioinspired innovations have significantly improved the mechanical adaptability of MN systems. For example, a hydrogel MN system combining polydopamine and poly(ethylene glycol) diacrylate-sodium alginate tips mimics octopus suckers, achieving strong adhesion in both dry and wet environments [71]. Similarly, angle-tunable MNs inspired by cat tongue barbs enable dynamic wound closure under high tension [72]. Crab claw- and shark skin-inspired MNs with dual-anchor mechanisms further reduce insertion resistance in joint wounds [73]. These biomimetic designs provide new perspectives for clinical translation of TDD systems. Pomegranate-inspired dual-chamber MNs significantly increase drug payload capacity for drug delivery enhancement, which co-loads free and particle-bound rivastigmine, increasing drug capacity by 40% and sustaining cognitive recovery in Alzheimer’s disease models [74]. Bio-inspired designs also enable multifunctional applications. Specifically, sea cucumber-inspired MNs incorporating piezoelectric zinc oxide nanoparticles and conductive graphene oxide generate microcurrents to promote neural repair, while reducing muscle atrophy [75]. Stem cell niche-inspired MN arrays promote angiogenesis in diabetic wounds by enhancing adipose-derived stem cell proliferation and VEGF secretion [76].
The growing demand for precision and personalized medicine has driven advances in wearable MN-based sensors. Closed-loop wearable systems utilize MN arrays to detect interstitial fluid biomarkers in real time and deliver therapeutics, demonstrating exceptional potential in diabetes management. A self-powered wearable MN patch incorporating a triboelectric nanogenerator (F-TENG) converts mechanical motion into electrical energy to drive deep tissue delivery of drug-loaded nanoparticles. This system enables rapid drug release in acidic tumor microenvironments and achieves superior chemo-photodynamic synergy in melanoma models compared to conventional MNs [77, 78]. While these innovations have addressed traditional limitations in tissue adhesion, drug penetration, and therapeutic precision, further improvements in treatment efficacy and patient compliance are essential to accelerate clinical translation.
Clinical research involving MN-mediated TDD
MNs have rapidly advanced into clinical applications since the formal introduction in 1998. The first clinical trial using MNs explored use in embryo-assisted hatching [79]. Extensive human studies have focused on MN delivery performance and therapeutic applications since 2008 with cosmetic indications making up approximately 50% of these trials. Radiofrequency (RF) microneedling, which combines MN penetration with targeted RF energy, has become a key technique in aesthetic medicine. Insulated needles deliver RF energy to the dermis, stimulating neocollagenesis and tissue remodeling. Initial studies in 2009 showed RF-induced elastin and collagen production with later trials confirming efficacy in skin rejuvenation and acne scar treatment [80].
Recent advances have explored comparative and combined approaches with lasers. Fractional Er:Glass lasers and RF MNs have similar effectiveness in treating facial acne scars, both of which offer good safety profiles [81]. Synergistic effects have been noted when combining RF MNs with non-ablative 1927-nm thulium fiber lasers (TFLs) for wrinkle reduction [82]. RF MNs also outperform non-ablative 1565-nm fractional lasers (NAFLs) in treating nasolabial fold laxity, although RF MNs may cause more discomfort, requiring protocol adjustments for optimal results [83, 84].
In addition to aesthetics, MN applications are expanding into medical fields, such as alopecia areata, psoriasis, verruca vulgaris, and periorbital aging, offering new therapeutic approaches [85–88], For example, the dexmedetomidine hydrochloride MN patch has received clinical approval and is now authorized for use during preoperative sedation in pediatric patients (CXHL2400157). MNs have also revolutionized vaccination strategies. Early-phase trials with influenza vaccines have shown that low-dose intradermal MN delivery achieved immunogenicity equivalent to full-dose intramuscular injections in 180 adults [89]. This success has extended to rabies and poliovirus vaccines [90, 91]. A 2024 Lancet trial confirmed the role of MNs in pediatric immunization, reporting mild local reactions and no serious adverse events from measles-rubella vaccine delivery via MNs patches, proving safety in children [92]. Additionally, MN-based clinical trials for primary axillary hyperhidrosis, diabetes management, dry eye disease, and localized anesthesia highlight MN versatility, as shown in Table 1.
Table 1 Summary of MN-Based Clinical Trials for Primary Axillary Hyperhidrosis, Diabetes Management, Dry Eye Disease, and Anesthesia
| Disease | Application | Year | Delivery/Monitoring | MN | Key Finding | Ref |
|---|---|---|---|---|---|---|
| Intraoperative pain | Anesthesia | 2022 | Lidocaine | Roller Microneedle | Reduced anesthesia onset time and improved pain control with no adverse effects | [136] |
| IV Cannulation pain | Anesthesia | 2022 | Lidocaine | MicronJet600 (NanoPass Technologies Ltd, Israel) | 11-fold pain reduction compared to no pretreatment | [137] |
| Intraoperative pain | Anesthesia | 2016 | Lidocaine + Prilocaine | Fractional Microneedling | Significantly lower pain scores vs. topical anesthesia alone (P < 0.05) | [138] |
| Dental injection pain | Anesthesia | 2021 | Lidocaine | Microneedle Patch | Reduced pain during dental injections and improved patient comfort | [139] |
| Diabetes | Glucose control | 2011, 2012, 2015 | Insulin | 34-gauge 1.5-mm steel microneedle | Faster insulin absorption/onset and reduced glycemic variability over 72 hours. | [140–142] |
| Diabetes | Glucose monitoring | 2014 | Glucose | Hollow microneedle array | 15% MARD accuracy and 72-hour wear feasibility | [143] |
| Diabetes | Glucose control | 2013 | Insulin | Single hollow microneedle | 22-minute faster onset and reduced insertion pain (P = 0.005) | [144] |
| Primary axillary hyperhidrosis | Sweat secretion control | 2015, 2020, 2024 | N/A | Fractional Microneedle Radiofrequency (FMR) | Botulinum toxin-A showed higher efficacy and lower pain. Both treatments significantly reduced HDSS scores. Improved HDSS vs. sham (P < 0.05) | [145–147] |
| Dry eye disease | Tear secretion control | 2022, 2024 | N/A | Biodegradable Microneedle Acupuncture (BMA) | Equivalent efficacy to intradermal acupuncture (improved OSDI, VAS, QoL, and tear secretion; P < 0.05) with no adverse events, demonstrating clinical safety/efficacy for future trials | [148, 149] |
US-mediated TDD
US, a form of mechanical wave energy, offers a non-invasive, cost-effective strategy for drug delivery with the distinct advantage of providing precise spatiotemporal control. Moreover, the deep tissue penetration capability of US enables new possibilities for disease diagnosis and treatment. US energy attenuates via reflection, refraction, scattering, and absorption during tissue propagation. Penetration depth, which is defined as the distance where intensity decays to 1/e (~37%) of the initial value, is inversely correlated with frequency [93, 94]. US reaches a 3–5 cm depth at 1 MHz, which substantially exceeds optical methods [visible light: < 1 mm; NIR: ~1 cm] [95]. This advantage stems from lower absorption in aqueous and biological media versus light waves, permitting non-invasive precision energy delivery to deep targets.
Mechanisms underlying US-mediated TDD
The initial combination of ultrasound with hydrocortisone ointment for enhanced drug delivery in polyarthritis treatment was pioneered in the 1950s [21]. This US-based method was subsequently extended to the treatment of bone and joint diseases as well as bursitis [96]. Extensive research has demonstrated that US enhances skin permeability and promotes efficient TDD through multiple mechanisms, including thermal effects, cavitation effects, and acoustic streaming effects [97]. Each mechanism has a distinct role in enhancing drug diffusion. The thermal effect of US primarily increases the kinetic energy of drug molecules, thereby enhancing diffusion across the skin. In many studies this thermal effect has been combined with iontophoresis (known as sonophoresis), significantly improving TDD [13]. However, some studies have suggested that the US intensity used in sonophoresis is relatively low with no significant increase in temperature. Therefore, the thermal effect may not be a major contributor to enhancing drug diffusion [98].
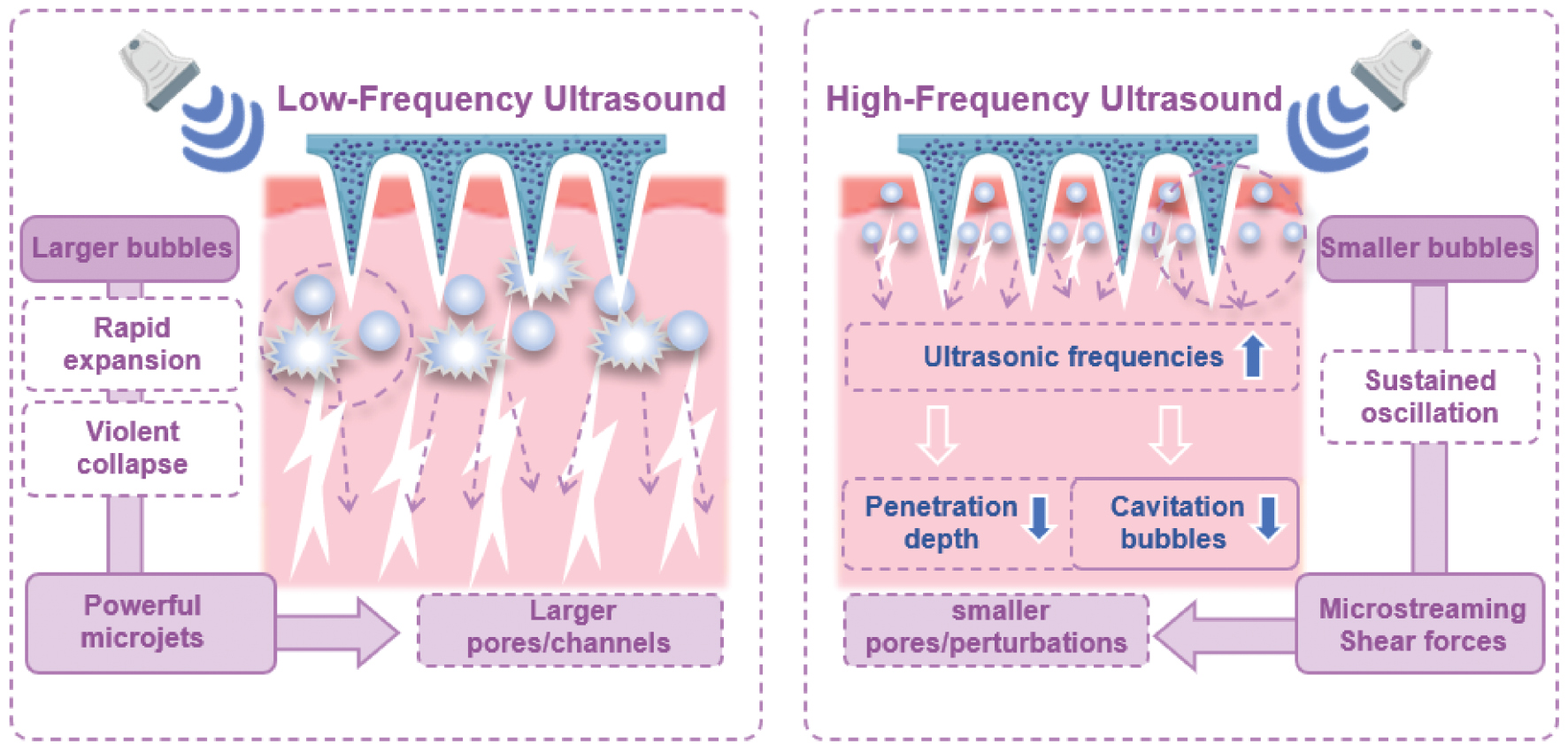
In recent years the cavitation effect has been identified as the primary mechanism underlying US-mediated TDD. Cavitation occurs when US induces compression and expansion of liquid within tissues, reducing local pressure below the vapor pressure and causing bubble formation. Cavitation can be categorized into stable cavitation and inertial cavitation. Stable cavitation involves the oscillation of bubbles at lower intensities, causing periodic contraction and expansion, while inertial cavitation is characterized by rapid bubble expansion and collapse with release of significant energy [99]. Both types of cavitation are influenced by US intensity. Studies have elucidated the role of stable cavitation in TDD Krasovitski et al. suggested that US at intensities below the cavitation threshold induces stable cavitation and leads to the formation of pores in the bilayer membrane, which facilitates material transport [100]. Stable cavitation can also increase drug diffusion by generating microflows. The choice of US frequency has a critical role in this mechanism. It has baren shown that the size of cavitation bubbles is inversely proportional to US frequency [10, 101]. High-frequency US can enhance the diffusion rate of salicylic acid by up to 4-fold under the same treatment conditions, while low-frequency US has minimal effects on diffusion [102]. This finding is due to the smaller bubble size under high-frequency US, which allows for more efficient penetration and diffusion within the skin. We hypothesize that freely diffusing bubbles enhance drug permeation through the skin by interacting with drug molecules and facilitating penetration. However, low-frequency US has also been shown to have significant efficacy in enhancing drug delivery, particularly for the transdermal delivery of insulin in diabetic rats [13]. In fact, low-frequency US has been shown to increase the permeation of ethanol and sucrose up to 1000-fold compared to therapeutic US [1 MHz] [103]. The dominant mechanism in low-frequency US is inertial cavitation, in which larger bubbles rapidly collapse in the coupling medium and generate microjets that disrupt the SC, thereby enhancing skin permeability and drug delivery efficiency [104]. Further research by Ueda et al. demonstrated that lower US frequencies produce stronger cavitation effects [105]. Additionally, the size of the pores formed on the skin surface after US treatment correlates with the frequency of the applied US with lower frequencies resulting in larger pore sizes [106] (Figure 5).
Figure 5 Mechanistic contrast between low- and high-frequency US synergized with microneedles.
Advances in US-mediated TDD
Preclinical studies have substantiated the efficacy of US in enhancing TDD. US has been shown to improve the transdermal delivery of small molecules, such as quantum dots and gold nanoparticles [107, 108]. However, larger molecules, such as peptides and proteins, encounter significant challenges in penetrating the skin barrier due to size. Notably, Park et al. successfully demonstrated the transdermal delivery of insulin using a piezoelectric transducer, showing that this non-invasive method yields a higher effective transdermal delivery of insulin and offering a promising alternative for insulin delivery in diabetic patients [109]. Technological advances have also addressed the limitations of traditional single-frequency US. Recent studies have indicated that dual-frequency US, which combines high and low frequencies, enhances the cavitation effect and leads to a substantial increase in drug delivery efficiency [110, 111]. For example, Polat et al. used a dual-frequency approach with 20 kHz low- and 1 MHz high-frequency US, which resulted in a 3.81- and 13.6-fold increase in the delivery efficiency of inulin and glucose, respectively, across porcine skin [106].
US-mediated transdermal drug delivery has made notable progress in clinical settings. The first clinical applications focused on local anesthesia and in 2004 the FDA approved the SonoPrep® device, which uses US to enhance the skin permeability of local anesthetics, such as lidocaine [13]. This device has been shown to effectively reduce the onset time and enhance the anesthetic effects of lidocaine. The US devices commonly used in clinical trials and the corresponding parameters are summarized in Table 2. US has also facilitated the penetration of dendrimer molecules into human skin. Specifically, dendrimers were detected in the dermis following 10 min of US treatment, whereas the penetration under passive diffusion was minimal [112]. These findings underscore the significant potential of US in clinical TDD.
Table 2 Ultrasound Devices and Parameters Commonly used in Clinical Trials
| Device | Frequency Intensity (W/cm2) | Duty Cycle | Participants (n) | Drug Delivered | Gov Identifier | Year | Ref |
|---|---|---|---|---|---|---|---|
| Langevin-type transducer (Transderma) | 36 kHz, US1: 2.72 (SPTA), US2: 3.50 (SPTA) | US1: 2:5 s US2: 3:5 s |
10 | Histamine | N/A | 2010 | [150] |
| Langevin-type transducer (Transderma) | 36 kHz, 2.72 (SPTA) | 2:5 s | 15 | Betamethasone 17-valerate | N/A | 2010 | [3] |
| Sonopuls 992 (Enraf Nonius) | 1 MHz, 1.0 (SPTA) | Continuous | 22 | Halcinonide | N/A | 2013 | [151] |
| Sonopuls 992 (Enraf Nonius) | 1 MHz, 1.0 (SPTA) | Pulsed (1:4) vs. Continuous | 93 | Lidocaine | NCT01404468 | 2012 | [152] |
| NAVA-01 (Noah Tongzhou) | 40 kHz, 300 mW/cm2 (peak), 100 mW/cm2 (pulse avg) | Pulsed | 110 | CM gel | NCC2015Z-01 | 2019 | [153] |
| Carewear LFUS patch | 45 kHz, 0.075–0.09 (TA) | Continuous | 40 | Dexamethasone sodium phosphate | N/A | 2020 | [154] |
| Chattanooga Vectra Genisys Therapy System | 1 MHz, 1.5 W/cm2 | 50% Duty Cycle | 40 | Dexamethasone sodium phosphate | N/A | 2020 | [154] |
| NAVA-01 (Noah Tongzhou) | 40 kHz, 300 mW/cm2 (peak), 100 mW/cm2 (pulse avg) | Pulsed | 110 | Chinese medicine formula | N/A | 2020 | [155] |
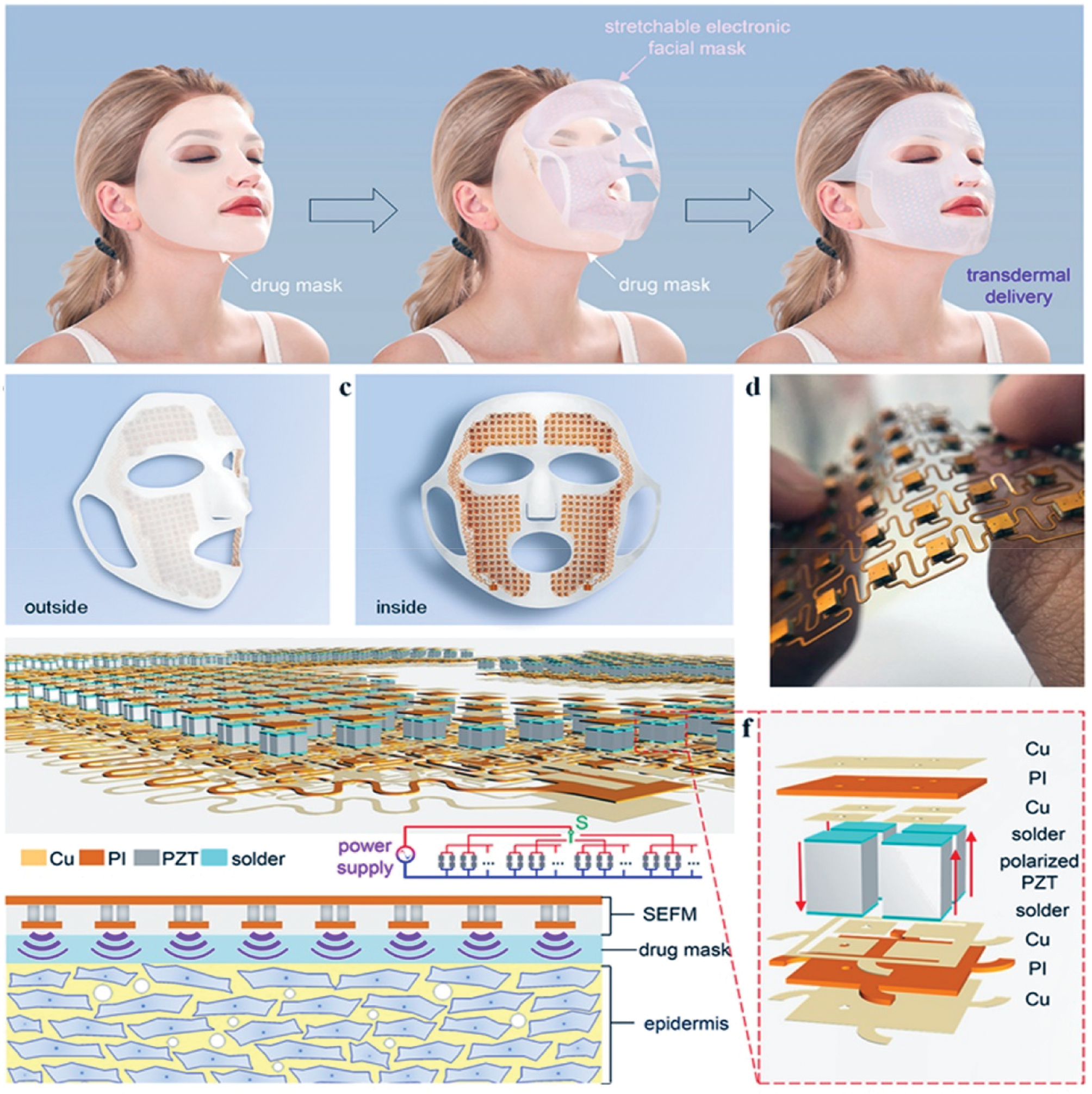
While commercial sonicators have proven effective for US-mediated TDD, the ultrasonic probe or converter from a commercial sonicator can weigh ≥ 1 kg. Therefore, developing lightweight, portable US devices is critical for advancing clinical translation. To address this need, Smith et al. proposed using low-profile, lightweight lanthanum-modified lead zirconate titanate (PZT) array transducers (total weight < 22 g) for transdermal delivery. The miniaturized device generated sufficient acoustic intensity to achieve efficient insulin delivery in hyperglycemic rat and rabbit models [113, 114]. Similarly, PZT transducers have been evaluated for efficacy in enhancing insulin delivery in pigs [109]. In addition, the development of wearable US devices has further increased the clinical applicability of US-mediated drug delivery. A stretchable electronic facial mask (SEFM) was developed to synergize sonophoresis with topical drug masks, integrating real-time sensors and actuators for personalized facial healthcare [115] (Figure 6). At the same time Yu et al. engineered a conformable US patch (cUSP) comprised of 4 PZT elements embedded in a polydimethylsiloxane (PDMS) substrate (5 cm diameter). This wearable platform enhanced transdermal delivery of cosmetic agents (e.g., niacinamide) by 26.2-fold in porcine skin models, outperforming MNs and traditional methods through synergistic reduction of skin impedance and enhanced permeation [10]. Despite device miniaturization, Han et al. developed nanobubble ultrasonic coupling agents to address the limitations of conventional ultrasonic coupling agents in TDD. By amplifying US-induced cavitation effects, these agents disrupt the dual barriers of skin (SC and epidermis), enabling stepwise drug penetration into the dermis and achieving deep, non-invasive TDD [22].
Figure 6 Wearable ultrasound-mediated transdermal delivery systems and nanobubble ultrasonic coupling agents. A stretchable electronic facial mask (SEFM) with an ear-hook design conforming to facial contours. The device integrates stretchable island-bridge mesh circuits and piezoelectric arrays to generate ultrasound, which enhances transdermal drug delivery through acoustic cavitation and thermal effects. Figure 6 was reproduced from Ref. [115] with permission from American Chemical Society. Copyright 2025.
These technological innovations have significantly improved the efficiency of drug delivery. Looking ahead, US-mediated TDD is likely to be integrated with other enhancement techniques, such as electroporation and electro-osmosis, to further optimize delivery efficiency. With continuous advances in device technology, US therapy holds promise as a universal and efficient method for non-invasive drug delivery, particularly in clinical applications.
Combining US and MNs for enhanced TDD
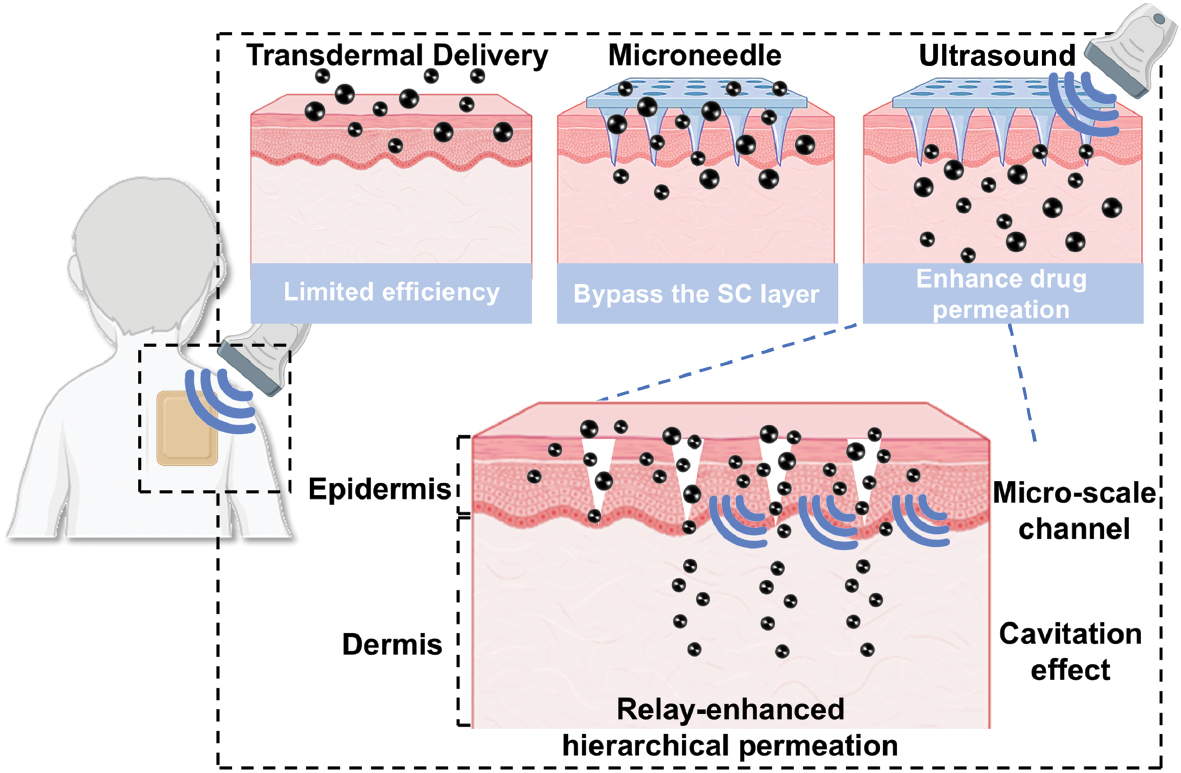
The combination of US and MNs (US-MN) provides an innovative solution to overcome the limitations of conventional TDD. Although MNs effectively penetrate the SC via minimally invasive channels (300–400 μm in depth), the standalone application is constrained by shallow penetration depth and suboptimal drug delivery efficiency to deeper tissues. As a critical modality for enhancing transdermal drug permeation, US solely exhibits limited efficiency in facilitating drug transport across the SC.
The synergistic effects of US-MN enhance therapeutic efficacy through three multidimensional mechanisms: 1) Enhanced drug migration (Microneedles create microchannels through the SC, reducing drug penetration time in US-mediated transdermal delivery. US further enhances drug migration through acoustic streaming and cavitation-induced microjets, improving permeation efficiency.) 2) Relay delivery to deep tissues (The penetrating capability of US enables mechanical forces and thermal effects generated in deeper tissues facilitate post-microchannel drug diffusion, continuously driven drugs to subcutaneous layers.) 3) Spatiotemporal control for intelligent drug release (The spatiotemporal precision of US imparts smart functionality to MNs, allowing on-demand drug release and optimized release kinetics) (Figure 7).
Figure 7 Illustration combining ultrasound and microneedle for enhanced transdermal delivery.
The US-MN synergy transforms MNs from static carriers into an intelligent “relay delivery” system. Additionally, the MN-created microchannels enhance US-mediated therapeutic efficiency within controlled parameter ranges. This integrated platform demonstrates unprecedented versatility, optimizing treatment windows for local anesthesia and infection control, while offering novel strategies for historically challenging issues, such as bacterial antibiotic resistance and subcutaneous tumor therapy. By combining the anatomic precision of MNs with the dynamic controllability of US, this paradigm pioneers new pathways for effective transdermal delivery (Table 3).
Table 3 Summary of the Ultrasound-Assisted Microneedle Platforms for Enhanced Therapeutics
| Synergy Type | Mechanism | Experimental/System Case | Outcome | Ref |
|---|---|---|---|---|
| Enhanced transdermal drug delivery | Thermal effects, cavitation effects, and acoustic streaming enhance drug diffusion and permeation | Hollow MNs + US improve calcein and BSA delivery; solid MNs pretreatment + US for macromolecules | Delivery efficiency increased 9–12×; reduced transdermal time from > 2 hours to 7 minutes (lidocaine) | [20, 21] |
| Enhanced transdermal drug delivery | Cavitation accelerates MN dissolution to release antibacterial materials | HA MNs loaded with sonocatalytic materials triggered by ultrasound | 99.94% antibacterial efficiency (S. aureus) | [120] |
| Applications expansion | Piezoelectric materials convert ultrasound energy to electricity for macrophage polarization modulation | Piezoelectric MNs for localized electrical stimulation | Non-invasive immune cell phenotype regulation | [25] |
| Spatiotemporally controlled delivery | Temperature/ultrasound dual-responsive release; ultrasound-activated targeted delivery | MSNs grafted with copolymers (temperature/ultrasound-responsive) MOF-based MNs for CRISPRa-UCP1 delivery (ultrasound-triggered ROS) |
On-demand drug release; localized fat ablation and white adipose browning | [23, 24] |
| Applications expansion | Fenton/electrolytic reactions generate ROS/gas for combined therapy | Riboflavin MNs + PZY2 electrolysis (shortens ocular axis); Schottky heterostructure MNs (sonodynamic therapy) | Enhanced scleral collagen crosslinking; efficient C. acnes elimination | [127, 128] |
| Applications expansion | Ultrasound-driven cascade reactions (glucose oxidase → H2O2 → OH → CO release) | TiO2–x MNs + MPA–CO prodrug | Controlled CO release for wound healing | [126] |
Combining US and MNs for enhanced TDD
The combination of US and MN enables efficient TDD through multiple mechanisms, involving microchannels, thermal effects, cavitation effects, and acoustic streaming. US-induced acoustic streaming reduces boundary layer resistance in enhancing skin permeability via fluid flow, amplifying drug diffusion through MN-created microchannels that can reduce drug penetration time in SC [20]. Concurrently, cavitation effects generate microjet formation through microbubble (MB) collapse, synergistically improving skin permeability to assist MN-mediated drug penetration. It has been shown that US-enhanced convective flow increased drug permeation through hollow MNs, achieving 9- and 12-fold enhancements in calcein and bovine serum albumin (BSA) delivery efficiency, respectively, compared to passive diffusion [21].
The US and MN combination enables a “relay” for efficient dermal penetration in enhancing drug permeation capabilities. The synergistic effects of US and MNs on TDD are summarized in Table 4. MNs, typically designed at a length of ~1000 μm, exhibit reduced effective insertion depths as a micro-scale transdermal platform due to skin viscoelasticity and insertion force limitations. Nayak et al. reported that MNs, 1100 μm in length, had a penetration depth of only 300–400 μm in ex vivo pig ear skin [20]. Makvandi et al. used optical coherence tomography imaging to evaluate an in situ penetration depth of 850 μm using high MN array patches in ex vivo pig skin and found that the maximum depth only reached ~600 μm [116]. Notably, the peripheral MNs penetrated 27% less than central MNs and further reduction occurred near skin appendages [117].
Table 4 Synergistic Effects of Ultrasound and Microneedles on Transdermal Drug Delivery
| Technology | Penetration Depth | Permeation Enhancement | Clinical Applications | Ref |
|---|---|---|---|---|
| Low-frequency US (20–100 kHz) | >10 cm (in soft tissue at 10 kHz) | Up to 1000-fold for ethanol/sucrose | Betamethasone valerate cream (2010) Histamine (2010) Anesthetic creams (2004) TCM formulations (2020) |
[150, 155–157] |
| High-frequency US (1–3 MHz) | 3–5 cm (at 1 MHz) | Up to 4-fold for salicylic acid | Limited reports | [95] |
| Microneedles | 300–600 μm (< designed height) | 74.7% payload delivery over 6 h | Vaccines (2017, 2022) Betamethasone (2025) Retinoic acid (2013) Insulin (2011, 2016) Nalmefene (2022) Diclofenac (2012) |
[141, 158–164] |
Given that the epidermis ranges from 200–1400 μm and the dermis ranges from 400–2400 μm in thickness, MNs primarily facilitate traversal of the SC within a constrained depth. Current MN systems have moderately improved drug permeation compared to passive diffusion but the diffusion within the dermal layer remains suboptimal and needs further enhancement. The auxiliary role of US addresses this limitation through spatial relay and kinetic enhancement. Moreover, US-generated thermal effects increase molecular kinetic energy, augmenting MN-driven diffusion rates post-SC penetration (Figure 8).
Figure 8 Mechanisms underlying US-MN platforms for enhanced transdermal delivery.
The US-MN platforms have demonstrated enhanced therapeutic effects in various applications. Combining MD pretreatment with US accelerated topical lidocaine anesthesia delivery 23-fold versus passive diffusion, achieving therapeutic concentrations in 7 min versus 30 min for US alone. Moreover, MN-created channels are a prerequisite for US enhancement because US alone fails to overcome SC barriers [20]. Therefore, to realize these synergistic effects in US-MN-based TDD, MNs are typically applied before US treatment. Wu et al. demonstrated that a 5-min MN pretreatment followed by a 10-min US application increased the finasteride penetration depth to 5.01 mm, which was 1.89-fold greater than MNs alone with cavitation-induced dermal fissures confirming the “relay” role of US in deep tissue diffusion [118]. It has been shown that US alone concentrates acoustic energy at the skin surface, forming cavitation-induced pores that hinder deeper propagation. As a result, no porous structures are observed in the deeper dermis, underscoring the necessity of MN pretreatment. A piezoelectric-driven MN platform (PDMN), which is composed of MN arrays, piezoelectric ceramics, and copper electrodes, generates low-frequency US with peak acoustic intensity at the MN tips. This finding enables the formation of cavitation-induced pores from the skin surface down to the needle tips. Unlike US- or MN-only groups, which showed limited diffusion in either direction, the PDMN group achieved enhanced drug diffusion both vertically and horizontally [119].
The US cavitation effects assist hyaluronic acid MNs loaded with TiO2 nanoparticles to dissolve rapidly for open wound infections, enabling deep biofilm penetration. This approach achieved 99.94% antibacterial efficacy against Staphylococcus aureus, rapidly clearing biofilms and promoting healing. These results highlight the dual role of US in enhancing MN-driven drug dispersion and amplifying therapeutic outcomes [120].
Piezocatalytic therapy (PCT) is an emerging dynamic therapeutic strategy that relies on US-activated anisotropic piezocatalysts with built-in electric fields [121]. The efficient delivery of piezoelectric nanoparticles (PNPs) is critical for PCT efficacy. A drug penetration depth of ~4.6 mm was observed in an agarose hydrogel model at 30 min using US combined with dissolving MNs (DMNs) for PNP delivery, doubling the depth achieved in the control group (~2.3 mm). The total diffusion depth reached ~500 μm with US-DMN co-treatment in porcine skin experiments compared to < 400 μm for DMNs alone, confirming that US further enhances deep tissue drug diffusion [122]. US-mediated drug delivery also improves cellular drug uptake via sonoporation. Zandi et al. developed an integrated electrochemical stimulator on a microfabricated silicon MN array coated with zinc-oxide nanowires (ZnONWs) to generate localized MBs for enhanced sonophoresis. While US combined with free drug administration reduced viable cell counts to 80%, the addition of MNs caused a dramatic decrease to ~52%, indicating that MN-generated MBs significantly amplify the anticancer efficacy of US-mediated drug delivery [123].
Combining US and MNs for spatiotemporally controlled drug delivery
Physical stimuli, such as light and US, assist MNs in achieving intelligent, responsive drug release. US supports deeper penetration into skin layers compared to light, reaching deep-seated tumor lesions without causing significant tissue damage, while offering superior controllability [124]. To date, MN platforms assisted by US-responsive materials or gene carriers have enabled spatiotemporally controlled drug delivery and therapeutic applications in preclinical studies. As the core transdermal delivery vehicle, MNs strategically integrated with US demonstrate unparalleled potential for precise disease treatment through spatiotemporal coordination and controlled release.
Anirudhan et al. developed a MN-compatible system based on mesoporous silica nanoparticles (MSNs) grafted with copolymer chains of aminoethyl methacrylate and 2-tetrahydropyranyl methacrylate (2-THPMA), which exhibit dual temperature- and US-responsiveness [23]. The 2-THPMA component contains US-cleavable acetal bonds, transitioning from a hydrophobic to hydrophilic properties under US exposure. These MSNs adopt an open conformation below 4°C for maximal drug loading, collapsing at physiologic temperatures (e.g., skin temperature) to seal pores. US assistance triggers copolymer cleavage, enabling on-demand drug release while enhancing MN-mediated skin permeation.
Building on the unique capabilities of US, Li et al. engineered a MN-dominant platform for spatiotemporally controlled gene therapy targeting obesity. The system encapsulates CRISPRa-UCP1 plasmid-loaded acoustically sensitive nanosystems into hyaluronic acid MNs (MN-MP/aUCP1), creating an US-assisted weight-loss platform. MNs deliver MP/aUCP1 to the subcutaneous fat layer, while US irradiation assists in triggering plasmid release from MNs and enhancing intracellular transfection in adipocytes via sonoporation, a process in which US-induced MB cavitation generates transient pores in cell membranes. This MN-driven strategy, when augmented by US, significantly upregulates UCP1 expression to promote white adipose tissue browning. US simultaneously assists in precision-triggered localized reactive oxygen species (ROS) generation to eliminate excess adipocytes, synergistically improving anti-obesity efficacy [24].
Combining US and MNs for expanding therapeutic applications
The combination of US and MNs extends the therapeutic applications of transdermal delivery. While MNs excel in treating superficial conditions, bacterial-mediated diseases (e.g., infected wounds and acne) face challenges due to antibiotic resistance. To address this finding, MNs establish transdermal pathways for ROS-generating materials, the therapeutic efficacy of which is enhanced by US assistance. Xiang et al. demonstrated that MNs deliver metal-organic frameworks (MOFs) to the skin, while US assists in generating ROS to eradicate Cutibacterium acne for acne treatment [125]. Similarly, Cao et al. developed a MN platform loaded with a sonosensitizer (TiO2–x), glucose oxidase, and an ·OH-responsive CO prodrug (MPA–CO). US assists by triggering Fenton reactions to convert H2O2 into ·OH radicals, which subsequently release CO gas, a cascade mechanism enabling precise diabetic wound therapy [126].
MN systems further benefit from physical properties of US for advanced applications. Hu et al. integrated Schottky heterostructures into MNs, in which US assistance amplifies electron transfer during Fenton reactions, generating singlet oxygen for sonodynamic tumor therapy [127]. Wang et al. engineered MNs with US-assisted charge delivery in piezoelectric applications. Piezoelectric MNs generate electrical signals that polarize macrophages in the dermis when activated by US, offering a non-invasive strategy for lymphedema management [25].
MNs serve as the primary vehicle for sustained riboflavin delivery to strengthen the sclera for ocular disorders, like high myopia. Zhong et al. enhanced this approach by combining riboflavin-loaded MNs with US-electrolyzable PZY2 material. Specifically, US assists in triggering PZY2 electrolysis to generate gas bubbles that dynamically shorten the axial length and powering MN-emitted blue light to crosslink scleral collagen. This MN-driven, US-assisted system achieves dual therapeutic structural reinforcement and dynamic ocular remodeling, providing an innovative solution for myopia-related complications [128].
Challenges and future
The integration of US and MNs has overcome the limitations of conventional TDD in terms of efficiency, penetration depth, and controllability, offering a novel multidisciplinary solution for therapeutic delivery. This synergistic approach not only significantly enhances drug permeation across biological barriers but also expands the application scope of transdermal technologies to complex indications, such as metabolic disorders, infectious diseases, oncology, and ophthalmology through precise spatiotemporal control, thereby advancing transdermal systems from localized therapy to systemic intervention.
However, this combination faces multifaceted challenges in achieving clinical translation. From a drug delivery perspective, the loading characteristics of this platform constrain utility. Conventional approaches favor lipophilic drugs or nanocarriers for enhanced efficiency [129]. Although MNs accommodate diverse payloads (small/large molecules, nucleic acids, nanoparticles, extracellular vesicles, and cells) [130], the interstitial fluid-filled channels create an aqueous microenvironment favoring hydrophilic agents. Hydrophilic nanoparticles exhibit 2.6-fold higher 48h cumulative transdermal permeation than hydrophobic counterparts. Confocal laser scanning microscopy revealed that hydrophilic rhodamine B diffused to a depth of 190 μm, markedly deeper than hydrophobic fluorescein isothiocyanate (FITC), which was confined to 130 μm within the MN channels. This difference is attributed to the covalent binding of FITC to skin proteins [131]. Furthermore, physicochemical properties (e.g., smaller size and negative charge, which leverage the negative surface charge of skin) significantly enhance intradermal diffusion [132]. Mechanically, aligning US parameters (frequency and intensity) with MN geometry is critical to prevent structural compromise or premature drug release, while miniaturizing wearable hybrid devices demands sophisticated engineering to maintain performance within energy-efficient, portable frameworks [21]. The use of Lead-Zirconate-Titanate (PZT) in US transducers raises biocompatibility concerns, which present potential lead leaching risks with adverse implications for human health and environmental safety [133]. Additionally, residual organic solvents, such as dimethyl sulfoxide and chloroform, which are often used during MN fabrication, may cause local tissue irritation and raise concerns about long-term biocompatibility [134]. Although platforms, like the PDMN developed by Chen et al., demonstrate favorable safety profiles (MN channels close within 5 min with skin recovery complete within 30 min at a 114-kHz US frequency). However, the treatment area is limited to 5×5 mm, restricting the ability to cover large or multifocal lesions [119]. High manufacturing costs and limited scalability of precision-engineered systems further hinder widespread adoption [134, 135].
Future advances should prioritize intelligent systems developed through interdisciplinary collaboration, such as real-time biosensors (e.g., pH and glucose) for dynamic adjustment of US intensity and drug release with biodegradable wireless designs for painless chronic disease management. AI-driven personalization of MN geometry and US parameters based on individual skin biomechanics or disease profiles may optimize efficacy. Expanding beyond small molecules to mRNA vaccines and microbiome-modulating probiotics broadens the therapeutic scope, while sustainable manufacturing using 3D/4D printing addresses ecologic concerns. Collectively, these innovations aim to transform US-MN platforms into scalable, patient-centric solutions for precision medicine, bridging engineering, pharmacology, and clinical science to redefine next-generation drug delivery paradigms.
Data availability statement
Data sharing is not applicable to this article because no new data were created or analyzed.
Ethics statement
No direct interactions with human or animal subjects were involved. Therefore, ethical approval and informed consent were not required.
Author contributions
LL: Writing – review & editing, Writing – original draft, Conceptualization, Investigation, Methodology. WP: Writing – review & editing, Writing – original draft, Investigation, Conceptualization, Methodology. JC: Writing –review & editing, Supervision, Resources, Funding acquisition, Project administration. CL: Writing—review & editing, supervision, resources. All the authors read and approved the final manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (Grant nos. 82272028, 81971621, and 82102087), the Key R&D Program of Hunan Province (Grant no. 2021SK2035), the Natural Science Foundation of Hunan Province (Grant nos. 2022JJ30039 and 2022JJ40392), the Natural Science Foundation of Guangdong Province (Grant no. 2021A1515011177), and the Project of Science and Technology Innovation of Hunan Province (Grant no. 2021SK51807).
Acknowledgments
Not applicable.
Conflict of interest
The authors declare that there are no conflicts of interest.
Graphical abstract
Highlights
- Microneedles (MNs) facilitate transdermal drug delivery by creating microchannels through the stratum corneum barrier.
- Ultrasound (US) enhances transdermal delivery through cavitation and thermal mechanisms, enabling deep tissue penetration.
- US-MN integration enables hierarchical drug permeation via relay-enhanced synergy, facilitating modification-free macromolecule/vaccine delivery.
In brief
This review introduced the US-MN platform, which synergistically integrates MN-mediated SC disruption with US-driven active diffusion to overcome extracellular matrix barriers and enable deep tissue delivery. This hierarchical permeation facilitates rapid, modification-free transdermal delivery of macromolecules and vaccines with spatiotemporal control. Despite the therapeutic promise, clinical translation faces material biocompatibility, device miniaturization, and regulatory standardization hurdles.
References
- He J, Zhang Y, Yu X, Xu C. Wearable patches for transdermal drug delivery. Acta Pharm Sin B 2023;13(6):2298-309. [PMID: 37425057 DOI: 10.1016/j.apsb.2023.05.009]
- Jeong WY, Kwon M, Choi HE, Kim KS. Recent advances in transdermal drug delivery systems: a review. Biomater Res 2021;25(1):24. [PMID: 34321111 DOI: 10.1186/s40824-021-00226-6]
- Zhang T, Luo X, Xu K, Zhong W. Peptide-containing nanoformulations: skin barrier penetration and activity contribution. Adv Drug Deliv Rev 2023;203:115139. [PMID: 37951358 DOI: 10.1016/j.addr.2023.115139]
- Xu G, Mao Y, Jiang T, Gao B, He B. Structural design strategies of microneedle-based vaccines for transdermal immunity augmentation. J Control Release 2022;351:907-22. [PMID: 36216175 DOI: 10.1016/j.jconrel.2022.10.008]
- Prausnitz MR, Langer R. Transdermal drug delivery. Nat Biotechnol 2008;26(11):1261-8. [PMID: 18997767 DOI: 10.1038/nbt.1504]
- Nguyen HX, Banga AK. Advanced transdermal drug delivery system: a comprehensive review of microneedle technologies, novel designs, diverse applications, and critical challenges. Int J Pharm 2025;670:125118. [PMID: 39710310 DOI: 10.1016/j.ijpharm.2024.125118]
- Phatale V, Vaiphei KK, Jha S, Patil D, Agrawal M, et al. Overcoming skin barriers through advanced transdermal drug delivery approaches. J Control Release 2022;351:361-80. [PMID: 36169040 DOI: 10.1016/j.jconrel.2022.09.025]
- Harris-Tryon TA, Grice EA. Microbiota and maintenance of skin barrier function. Science 2022;376(6596):940-5. [PMID: 35617415 DOI: 10.1126/science.abo0693]
- Zhang W, Jiao Y, Zhang Z, Zhang Y, Yu J, et al. Transdermal gene delivery. J Control Release 2024;371:516-29. [PMID: 38849095 DOI: 10.1016/j.jconrel.2024.06.013]
- Yu C-C, Shah A, Amiri N, Marcus C, Nayeem MOG, et al. A conformable ultrasound patch for cavitation-enhanced transdermal cosmeceutical delivery. Adv Mater 2023;35(23):e2300066. [PMID: 36934314 DOI: 10.1002/adma.202300066]
- Li Z, Wang Y, Zhang R, Liu Z, Chang Z, et al. Microneedles-based theranostic platform: from the past to the future. ACS Nano 2024;18(35):23876-93. [PMID: 39177073 DOI: 10.1021/acsnano.4c04277]
- Zhi D, Yang T, Zhang T, Yang M, Zhang S, et al. Microneedles for gene and drug delivery in skin cancer therapy. J Control Release 2021;335:158-77. [PMID: 33984344 DOI: 10.1016/j.jconrel.2021.05.009]
- Azagury A, Khoury L, Enden G, Kost J. Ultrasound mediated transdermal drug delivery. Adv Drug Deliv Rev 2014;72:127-43. [PMID: 24463344 DOI: 10.1016/j.addr.2014.01.007]
- Wang R, Bian Q, Xu Y, Xu D, Gao J. Recent advances in mechanical force-assisted transdermal delivery of macromolecular drugs. Int J Pharm 2021;602:120598. [PMID: 33862129 DOI: 10.1016/j.ijpharm.2021.120598]
- Alkilani AZ, McCrudden MTC, Donnelly RF. Transdermal drug delivery: innovative pharmaceutical developments based on disruption of the barrier properties of the stratum corneum. Pharmaceutics 2015;7(4):438-70. [PMID: 26506371 DOI: 10.3390/pharmaceutics7040438]
- Chu H, Xue J, Yang Y, Zheng H, Luo D, et al. Advances of smart stimulus-responsive microneedles in cancer treatment. Small Methods 2024;8(9):e2301455. [PMID: 38148309 DOI: 10.1002/smtd.202301455]
- Zhao J, Chen J, Li C, Xiang H, Miao X. Hyaluronidase overcomes the extracellular matrix barrier to enhance local drug delivery. Eur J Pharm Biopharm 2024;203:114474. [PMID: 39191305 DOI: 10.1016/j.ejpb.2024.114474]
- Li J, Zhang Y, Tian J, Ling G, Zhang P. Advances in magnetic microneedles: from fabrications to applications. Biomaterials 2025;318:123143. [PMID: 40032442 DOI: 10.1016/j.biomaterials.2025.123143]
- Subedi RK, Oh SY, Chun M-K, Choi H-K. Recent advances in transdermal drug delivery. Arch Pharm Res 2010;33(3):339-51. [PMID: 20361297 DOI: 10.1007/s12272-010-0301-7]
- Nayak A, Babla H, Han T, Das DB. Lidocaine carboxymethylcellulose with gelatine co-polymer hydrogel delivery by combined microneedle and ultrasound. Drug Deliv 2016;23(2):658-69. [PMID: 25034877 DOI: 10.3109/10717544.2014.935985]
- Han T, Das DB. Potential of combined ultrasound and microneedles for enhanced transdermal drug permeation: a review. Eur J Pharm Biopharm 2015;89:312-28. [PMID: 25541440 DOI: 10.1016/j.ejpb.2014.12.020]
- Han X, Wang F, Shen J, Chen S, Xiao P, et al. Ultrasound nanobubble coupling agent for effective noninvasive deep-layer drug delivery. Adv Mater 2024;36(3):e2306993. [PMID: 37851922 DOI: 10.1002/adma.202306993]
- Anirudhan TS, Nair AS. Temperature and ultrasound sensitive gatekeepers for the controlled release of chemotherapeutic drugs from mesoporous silica nanoparticles. J Mater Chem B 2018;6(3):428-39. [PMID: 32254522 DOI: 10.1039/c7tb02292a]
- Li S, Yu J, Shen Y, Xiong B, Zhao D, et al. Transdermal microneedle-assisted ultrasound-enhanced CRISPRa system to enable sono-gene therapy for obesity. Nat Commun 2025;16(1):1499. [PMID: 39929846 DOI: 10.1038/s41467-025-56755-4]
- Wang X, Han C, Xia J, Cui C, Min P, et al. Ultrasound-mediated piezoelectric microneedles regulating macrophage polarization and remodeling pathological microenvironment for lymphedema improvement. ACS Nano 2025;19(1):1447-62. [PMID: 39810381 DOI: 10.1021/acsnano.4c14292]
- Sharma G, Alle M, Chakraborty C, Kim J-C. Strategies for transdermal drug delivery against bone disorders: a preclinical and clinical update. J Control Release 2021;336:375-95. [PMID: 34175368 DOI: 10.1016/j.jconrel.2021.06.035]
- Du M, Wang T, Feng R, Zeng P, Chen Z. Establishment of ultrasound-responsive SonoBacteriaBot for targeted drug delivery and controlled release. Front Bioeng Biotechnol 2023;11:1144963. [PMID: 36911192 DOI: 10.3389/fbioe.2023.1144963]
- Qin W, Quan G, Sun Y, Chen M, Yang P, et al. Dissolving microneedles with spatiotemporally controlled pulsatile release nanosystem for synergistic chemo-photothermal therapy of melanoma. Theranostics 2020;10(18):8179-96. [PMID: 32724465 DOI: 10.7150/thno.44194]
- Xu Y, Zhao M, Cao J, Fang T, Zhang J, et al. Applications and recent advances in transdermal drug delivery systems for the treatment of rheumatoid arthritis. Acta Pharm Sin B 2023;13(11):4417-41. [PMID: 37969725 DOI: 10.1016/j.apsb.2023.05.025]
- Lee H, Song C, Baik S, Kim D, Hyeon T, et al. Device-assisted transdermal drug delivery. Adv Drug Deliv Rev 2018;127:35-45. [PMID: 28867296 DOI: 10.1016/j.addr.2017.08.009]
- Dixon RV, Skaria E, Lau WM, Manning P, Birch-Machin MA, et al. Microneedle-based devices for point-of-care infectious disease diagnostics. Acta Pharm Sin B 2021;11(8):2344-61. [PMID: 34150486 DOI: 10.1016/j.apsb.2021.02.010]
- Jadoul A, Bouwstra J, Préat VV. Effects of iontophoresis and electroporation on the stratum corneum. Review of the biophysical studies. Adv Drug Deliv Rev 1999;35(1):89-105. [PMID: 10837691 DOI: 10.1016/s0169-409x(98)00065-9]
- Mueller J, Trapp M, Neubert RHH. The effect of hydrophilic penetration/diffusion enhancer on stratum corneum lipid models: Part II*: DMSO. Chem Phys Lipids 2019;225:104816. [PMID: 31525381 DOI: 10.1016/j.chemphyslip.2019.104816]
- Liu C, Guan Y, Tian Q, Shi X, Fang L. Transdermal enhancement strategy of ketoprofen and teriflunomide: the effect of enhanced drug-drug intermolecular interaction by permeation enhancer on drug release of compound transdermal patch. Int J Pharm 2019;572:118800. [PMID: 31678378 DOI: 10.1016/j.ijpharm.2019.118800]
- Yu Z, Meng X, Zhang S, Chen Y, Zhang Z, et al. Recent progress in transdermal nanocarriers and their surface modifications. Molecules 2021;26(11):3093. [PMID: 34064297 DOI: 10.3390/molecules26113093]
- Kováčik A, Kopečná M, Vávrová K. Permeation enhancers in transdermal drug delivery: benefits and limitations. Expert Opin Drug Deliv 2020;17(2):145-55. [PMID: 31910342 DOI: 10.1080/17425247.2020.1713087]
- Ganeson K, Alias AH, Murugaiyah V, Amirul A-A, Ramakrishna S, et al. Microneedles for efficient and precise drug delivery in cancer therapy. Pharmaceutics 2023;15(3):744. [PMID: 36986606 DOI: 10.3390/pharmaceutics15030744]
- Zhang R, Miao Q, Deng D, Wu J, Miao Y, et al. Research progress of advanced microneedle drug delivery system and its application in biomedicine. Colloids Surf B Biointerfaces 2023;226:113302. [PMID: 37086686 DOI: 10.1016/j.colsurfb.2023113302]
- Permana AD, Nainu F, Moffatt K, Larrañeta E, Donnelly RF. Recent advances in combination of microneedles and nanomedicines for lymphatic targeted drug delivery. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2021;13(3):e1690. [PMID: 33401339 DOI: 10.1002/wnan.1690]
- McAlister E, Kirkby M, Domínguez-Robles J, Paredes AJ, Anjani QK, et al. The role of microneedle arrays in drug delivery and patient monitoring to prevent diabetes induced fibrosis. Adv Drug Deliv Rev 2021;175:113825. [PMID: 34111467 DOI: 10.1016/j.addr.2021.06.002]
- Zahraie N, Perota G, Dehdari Vais R, Sattarahmady N. Simultaneous chemotherapy/sonodynamic therapy of the melanoma cancer cells using a gold-paclitaxel nanostructure. Photodiagnosis Photodyn Ther 2022;39:102991. [PMID: 35779857 DOI: 10.1016/j.pdpdt.2022.102991]
- Sartawi Z, Blackshields C, Faisal W. Dissolving microneedles: applications and growing therapeutic potential. J Control Release 2022;348:186-205. [PMID: 35662577 DOI: 10.1016/j.jconrel.2022.05.045]
- Shakya AK, Lee CH, Gill HS. Coated microneedle-based cutaneous immunotherapy prevents Der p 1-induced airway allergy in mice. J Allergy Clin Immunol 2018;142(6):2007-2011.e3. [PMID: 30171871 DOI: 10.1016/j.jaci.2018.08.017]
- Cárcamo-Martínez Á, Mallon B, Domínguez-Robles J, Vora LK, Anjani QK, et al. Hollow microneedles: a perspective in biomedical applications. Int J Pharm 2021;599:120455. [PMID: 33676993 DOI: 10.1016/j.ijpharm.2021.120455]
- Wu C, Yu Q, Huang C, Li F, Zhang L, et al. Microneedles as transdermal drug delivery system for enhancing skin disease treatment. Acta Pharm Sin B 2024;14(12):5161-80. [PMID: 39807331 DOI: 10.1016/j.apsb.2024.08.013]
- Yang J, Liu X, Fu Y, Song Y. Recent advances of microneedles for biomedical applications: drug delivery and beyond. Acta Pharm Sin B 2019;9(3):469-83. [PMID: 31193810 DOI: 10.1016/j.apsb.2019.03.007]
- Bok M, Zhao Z-J, Jeon S, Jeong J-H, Lim E. Ultrasonically and iontophoretically enhanced drug-delivery system based on dissolving microneedle patches. Sci Rep 2020;10(1):2027. [PMID: 32029808 DOI: 10.1038/s41598-020-58822-w]
- Filho D, Guerrero M, Pariguana M, Marican A, Durán-Lara EF. Hydrogel-based microneedle as a drug delivery system. Pharmaceutics 2023;15(10):2444. [PMID: 37896204 DOI: 10.3390/pharmaceutics15102444]
- Yao W, Li D, Zhao Y, Zhan Z, Jin G, et al. 3D Printed multi-functional hydrogel microneedles based on high-precision digital light processing. Micromachines (Basel) 2019;11(1):17. [PMID: 31877987 DOI: 10.3390/mi11010017]
- Martínez-Navarrete M, Pérez-López A, Guillot AJ, Cordeiro AS, Melero A, et al. Latest advances in glucose-responsive microneedle-based systems for transdermal insulin delivery. Int J Biol Macromol 2024;263(Pt 2):130301. [PMID: 38382776 DOI: 10.1016/j.ijbiomac.2024.130301]
- Kim Y-C, Quan F-S, Compans RW, Kang S-M, Prausnitz MR. Formulation and coating of microneedles with inactivated influenza virus to improve vaccine stability and immunogenicity. J Control Release 2010;142(2):187-95. [PMID: 19840825 DOI: 10.1016/j.jconrel.2009.10.013]
- Chang H, Chew SWT, Zheng M, Lio DCS, Wiraja C, et al. Cryomicroneedles for transdermal cell delivery. Nat Biomed Eng 2021;5(9):1008-18. [PMID: 33941895 DOI: 10.1038/s41551-021-00720-1]
- Gao Z, Sheng T, Zhang W, Feng H, Yu J, et al. Microneedle-mediated cell therapy. Adv Sci (Weinh) 2024;11(8):e2304124. [PMID: 37899686 DOI: 10.1002/advs.202304124]
- Ma S, Li J, Pei L, Feng N, Zhang Y. Microneedle-based interstitial fluid extraction for drug analysis: advances, challenges, and prospects. J Pharm Anal 2023;13(2):111-26. [PMID: 36908860 DOI: 10.1016/j.jpha.2022.12.004]
- Himawan A, Vora LK, Permana AD, Sudir S, Nurdin AR, et al. Where microneedle meets biomarkers: futuristic application for diagnosing and monitoring localized external organ diseases. Adv Healthc Mater 2023;12(5):e2202066. [PMID: 36414019 DOI: 10.1002/adhm.202202066]
- Ren Y, Fan L, Alkildani S, Liu L, Emmert S, et al. Barrier membranes for guided bone regeneration (GBR): a focus on recent advances in collagen membranes. Int J Mol Sci 2022;23(23):14987. [PMID: 36499315 DOI: 10.3390/ijms232314987]
- Mao X, Cheng R, Zhang H, Bae J, Cheng L, et al. Self-healing and injectable hydrogel for matching skin flap regeneration. Adv Sci (Weinh) 2018;6(3):1801555. [PMID: 30775235 DOI: 10.1002/advs.201801555]
- Makvandi P, Jamaledin R, Chen G, Baghbantaraghdari Z, Zare EN, et al. Stimuli-responsive transdermal microneedle patches. Mater Today (Kidlington) 2021;47:206-22. [PMID: 36338772 DOI: 10.1016/j.mattod.2021.03.012]
- Zhang X, Chen G, Fu X, Wang Y, Zhao Y. Magneto-responsive microneedle robots for intestinal macromolecule delivery. Adv Mater 2021;33(44):e2104932. [PMID: 34532914 DOI: 10.1002/adma.202104932]
- Xu R, Guo H, Chen X, Xu J, Gong Y, et al. Smart hydrothermally responsive microneedle for topical tumor treatment. J Control Release 2023;358:566-78. [PMID: 37182803 DOI: 10.1016/j.jconrel.2023.05.008]
- Meng Q, Xie E, Sun H, Wang H, Li J, et al. High-strength smart microneedles with “offensive and defensive” effects for intervertebral disc repair. Adv Mater 2024;36(2):e2305468. [PMID: 37681640 DOI: 10.1002/adma.202305468]
- Hardy JG, Larrañeta E, Donnelly RF, McGoldrick N, Migalska K, et al. Hydrogel-forming microneedle arrays made from light-responsive materials for on-demand transdermal drug delivery. Mol Pharm 2016;13(3):907-14. [PMID: 26795883 DOI: 10.1021/acs.molpharmaceut.5b00807]
- Zhang A, Zeng Y, Xiong B, Jiang X, Jin Y, et al. A pH-responsive core-shell microneedle patch with self-monitoring capability for local long-lasting analgesia. Adv Funct Mater 2023;34(12):2314048. [DOI: 10.1002/adfm.202314048]
- Yang C, Sheng T, Hou W, Zhang J, Cheng L, et al. Glucose-responsive microneedle patch for closed-loop dual-hormone delivery in mice and pigs. Sci Adv 2022;8(48):eadd3197. [PMID: 36449622 DOI: 10.1126/sciadv.add3197]
- Wang S, Yang C, Zhang W, Zhao S, You J, et al. Glucose-responsive microneedle patch with high insulin loading capacity for prolonged glycemic control in mice and minipigs. ACS Nano 2024;18(38):26056-65. [PMID: 39259604 DOI: 10.1021/acsnano.4c05562]
- Zhang Y, Yu J, Wang J, Hanne NJ, Cui Z, et al. Thrombin-responsive transcutaneous patch for auto-anticoagulant regulation. Adv Mater 2017;29(4):10.1002/adma.201604043. [PMID: 27885722 DOI: 10.1002/adma.201604043]
- Zhang X, Zhou C, Chen T, Jiang Z, Lu C, et al. State-of-the-art strategies to enhance the mechanical properties of microneedles. Int J Pharm 2024;663:124547. [PMID: 39097155 DOI: 10.1016/j.ijpharm.2024.124547]
- Huang D, Fu X, Zhang X, Zhao Y. Christmas tree-shaped microneedles as FOLFIRINOX spatiotemporal delivery system for pancreatic cancer treatment. Research (Wash D C) 2022;2022:9809417. [PMID: 36340504 DOI: 10.34133/2022/9809417]
- Ghosh S, Zheng M, He J, Wu Y, Zhang Y, et al. Electrically-driven drug delivery into deep cutaneous tissue by conductive microneedles for fungal infection eradication and protective immunity. Biomaterials 2025;314:122908. [PMID: 39454504 DOI: 10.1016/j.biomaterials.2024.122908]
- Pawar K. Recent updates in vaccine delivery through microneedles. Adv Pharm Bull 2023;13(1):1-4. [PMID: 36721802 DOI: 10.34172/apb.2023.001]
- Zhang X, Chen G, Yu Y, Sun L, Zhao Y. Bioinspired adhesive and antibacterial microneedles for versatile transdermal drug delivery. Research (Wash D C) 2020;2020:3672120. [PMID: 32490376 DOI: 10.34133/2020/3672120]
- Gan N, Li X, Wei M, Li Z, Zhou S, et al. Tongue prick bionic angularly adjustable microneedles for enhanced scarless wound healing. Adv Funct Mater 2025;35(22):2422602. [DOI: 10.1002/adfm.202422602]
- Zhou L, Zeng Z, Liu J, Zhang F, Bian X, et al. Double bionic deformable DNA hydrogel microneedles loaded with extracellular vesicles to guide tissue regeneration of diabetes ulcer wound. Adv Funct Mater 2023;34(14):2312499. [DOI: 10.1002/adfm.202312499]
- Zhang M, Yang B, Ren T, Wang X, Chen H, et al. Dual engine-driven bionic microneedles for early intervention and prolonged treatment of Alzheimer’s disease. J Control Release 2024;367:184-96. [PMID: 38242212 DOI: 10.1016/j.jconrel.2024.01.030]
- Hu C, Liu B, Huang X, Wang Z, Qin K, et al. Sea cucumber-inspired microneedle nerve guidance conduit for synergistically inhibiting muscle atrophy and promoting nerve regeneration. ACS Nano 2024;18(22):14427-40. [PMID: 38776414 DOI: 10.1021/acsnano.4c00794]
- Aliakbar Ahovan Z, Esmaeili Z, Eftekhari BS, Khosravimelal S, Alehosseini M, et al. Antibacterial smart hydrogels: new hope for infectious wound management. Mater Today Bio 2022;17:100499. [PMID: 36466959 DOI: 10.1016/j.mtbio.2022.100499]
- Yang Y, Chen BZ, Zhang XP, Zheng H, Li Z, et al. Conductive microneedle patch with electricity-triggered drug release performance for atopic dermatitis treatment. ACS Appl Mater Interfaces 2022;14(28):31645-54. [PMID: 35790212 DOI: 10.1021/acsami.2c05952]
- Wang C, He G, Zhao H, Lu Y, Jiang P, et al. Enhancing deep-seated melanoma therapy through wearable self-powered microneedle patch. Adv Mater 2024;36(11):e2311246. [PMID: 38123765 DOI: 10.1002/adma.202311246]
- Cieslak J, Ivakhnenko V, Wolf G, Sheleg S, Verlinsky Y. Three-dimensional partial zona dissection for preimplantation genetic diagnosis and assisted hatching. Fertil Steril 1999;71(2):308-13. [PMID: 9988403 DOI: 10.1016/s0015-0282(98)00452-x]
- Hantash BM, Ubeid AA, Chang H, Kafi R, Renton B. Bipolar fractional radiofrequency treatment induces neoelastogenesis and neocollagenesis. Lasers Surg Med 2009;41(1):1-9. [PMID: 19143021 DOI: 10.1002/lsm.20731]
- Chae WS, Seong JY, Jung HN, Kong SH, Kim MH, et al. Comparative study on efficacy and safety of 1550 nm Er:Glass fractional laser and fractional radiofrequency microneedle device for facial atrophic acne scar. J Cosmet Dermatol 2015;14(2):100-6. [PMID: 25810322 DOI: 10.1111/jocd.12139]
- Park M-Y, Hwang S, Chun SI, Kim SM, Almurayshid A, et al. A prospective, split-face, comparative study of combined treatment with fractional microneedle radiofrequency and nonablative 1927-nm fractional thulium fiber laser for wrinkle treatment. Dermatol Surg 2021;47(3):e101-5. [PMID: 32804892 DOI: 10.1097/DSS.0000000000002672]
- Hendel K, Karmisholt K, Hedelund L, Haedersdal M. Fractional CO2-laser versus microneedle radiofrequency for acne scars: a randomized, single treatment, split-face trial. Lasers Surg Med 2023;55(4):335-43. [PMID: 36934435 DOI: 10.1002/lsm.23655]
- Tang Z, Wen S, Liu T, Yu A, Li Y. Comparative study of treatment for striae alba stage striae gravidarum: 1565-nm non-ablative fractional laser versus fractional microneedle radiofrequency. Lasers Med Sci 2021;36(9):1823-30. [PMID: 33411130 DOI: 10.1007/s10103-020-03203-y]
- Qiao R, Zhu J, Fang J, Shi H, Zhang Z, et al. Microneedle transdermal delivery of compound betamethasone in alopecia areata-a randomized controlled trial. J Am Acad Dermatol 2025;92(2):269-75. [PMID: 39393548 DOI: 10.1016/j.jaad.2024.09.059]
- Yew YW, Phuan CZY, Zhao X, Tan EST, Chong WS, et al. Novel transdermal device for delivery of triamcinolone for nail psoriasis treatment. Ann Acad Med Singap 2022;51(1):16-23. [PMID: 35091727 DOI: 10.47102/annals-acadmedsg.2021380]
- Ryu HR, Jeong H-R, Seon-Woo H-S, Kim JS, Lee SK, et al. Efficacy of a bleomycin microneedle patch for the treatment of warts. Drug Deliv Transl Res 2018;8(1):273-80. [PMID: 29204924 DOI: 10.1007/s13346-017-0458-4]
- Gao Y, Che Q, He Q, Wang W, Luo W, et al. Treatment of periorbital aging with negative pressure fractional microneedle radiofrequency: a self-controlled clinical trial. J Cosmet Dermatol 2024;23(4):1269-76. [PMID: 38013586 DOI: 10.1111/jocd.16091]
- Van Damme P, Oosterhuis-Kafeja F, Van der Wielen M, Almagor Y, Sharon O, et al. Safety and efficacy of a novel microneedle device for dose sparing intradermal influenza vaccination in healthy adults. Vaccine 2009;27(3):454-9. [PMID: 19022318 DOI: 10.1016/j.vaccine.2008.10.077]
- Laurent PE, Bourhy H, Fantino M, Alchas P, Mikszta JA. Safety and efficacy of novel dermal and epidermal microneedle delivery systems for rabies vaccination in healthy adults. Vaccine 2010;28(36):5850-6. [PMID: 20600481 DOI: 10.1016/j.vaccine.2010.06.062]
- Anand A, Zaman K, Estívariz CF, Yunus M, Gary HE, et al. Early priming with inactivated poliovirus vaccine (IPV) and intradermal fractional dose IPV administered by a microneedle device: a randomized controlled trial. Vaccine 2015;33(48):6816-22. [PMID: 26476367 DOI: 10.1016/j.vaccine.2015.09.039]
- Adigweme I, Yisa M, Ooko M, Akpalu E, Bruce A, et al. A measles and rubella vaccine microneedle patch in The Gambia: a phase 1/2, double-blind, double-dummy, randomised, active-controlled, age de-escalation trial. Lancet 2024;403(10439):1879-92. [PMID: 38697170 DOI: 10.1016/S0140-6736(24)00532-4]
- Zhu P, Simon I, Kokalari I, Kohane DS, Rwei AY. Miniaturized therapeutic systems for ultrasound-modulated drug delivery to the central and peripheral nervous system. Adv Drug Deliv Rev 2024;208:115275. [PMID: 38442747 DOI: 10.1016/j.addr.2024.115275]
- Wells PN. Review: absorption and dispersion of ultrasound in biological tissue. Ultrasound Med Biol 1975;1(4):369-76. [PMID: 1098249 DOI: 10.1016/0301-5629(75)90124-6]
- Speed CA. Therapeutic ultrasound in soft tissue lesions. Rheumatology (Oxford) 2001;40(12):1331-6. [PMID: 11752501 DOI: 10.1093/rheumatology/40.12.1331]
- Newman MK, Kill M, Frampton G. Effects of ultrasound alone and combined with hydrocortisone injections by needle or hypo-spray. Am J Phys Med 1958;37(4):206-9. [PMID: 13559414]
- Tao W, Lai Y, Zhou X, Yang G, Wu P, et al. A narrative review: ultrasound-assisted drug delivery: improving treatments via multiple mechanisms. Ultrasonics 2025;151:107611. [PMID: 40068411 DOI: 10.1016/j.ultras.2025.107611]
- Levy D, Kost J, Meshulam Y, Langer R. Effect of ultrasound on transdermal drug delivery to rats and guinea pigs. J Clin Invest 1989;83(6):2074-8. [PMID: 2498396 DOI: 10.1172/JCI114119]
- Polat BE, Hart D, Langer R, Blankschtein D. Ultrasound-mediated transdermal drug delivery: mechanisms, scope, and emerging trends. J Control Release 2011;152(3):330-48. [PMID: 21238514 DOI: 10.1016/j.jconrel.2011.01.006]
- Krasovitski B, Frenkel V, Shoham S, Kimmel E. Intramembrane cavitation as a unifying mechanism for ultrasound-induced bioeffects. Proc Natl Acad Sci U S A 2011;108(8):3258-63. [PMID: 21300891 DOI: 10.1073/pnas.1015771108]
- Gaertner W. Frequency dependence of ultrasonic cavitation. J Acoust Soc Am 1954;26(6):977-80. [DOI: 10.1121/1.1907464]
- Bommannan D, Okuyama H, Stauffer P, Guy RH. Sonophoresis. I. The use of high-frequency ultrasound to enhance transdermal drug delivery. Pharm Res 1992;9(4):559-64. [PMID: 1495903 DOI: 10.1023/a:1015808917491]
- Mitragotri S, Blankschtein D, Langer R. Transdermal drug delivery using low-frequency sonophoresis. Pharm Res 1996;13(3):411-20. [PMID: 8692734 DOI: 10.1023/a:1016096626810]
- Alvarez-Román R, Merino G, Kalia YN, Naik A, Guy RH. Skin permeability enhancement by low frequency sonophoresis: lipid extraction and transport pathways. J Pharm Sci 2003;92(6):1138-46. [PMID: 12761803 DOI: 10.1002/jps.10370]
- Ueda H, Mutoh M, Seki T, Kobayashi D, Morimoto Y. Acoustic cavitation as an enhancing mechanism of low-frequency sonophoresis for transdermal drug delivery. Biol Pharm Bull 2009;32(5):916-20. [PMID: 19420764 DOI: 10.1248/bpb.32.916]
- Polat BE, Deen WM, Langer R, Blankschtein D. A physical mechanism to explain the delivery of chemical penetration enhancers into skin during transdermal sonophoresis – insight into the observed synergism. J Control Release 2012;158(2):250-60. [PMID: 22100440 DOI: 10.1016/j.jconrel.2011.11.008]
- Lopez RFV, Seto JE, Blankschtein D, Langer R. Enhancing the transdermal delivery of rigid nanoparticles using the simultaneous application of ultrasound and sodium lauryl sulfate. Biomaterials 2011;32(3):933-41. [PMID: 20971504 DOI: 10.1016/j.biomaterials.2010.09.060]
- Seto JE, Polat BE, Lopez RFV, Blankschtein D, Langer R. Effects of ultrasound and sodium lauryl sulfate on the transdermal delivery of hydrophilic permeants: comparative in vitro studies with full-thickness and split-thickness pig and human skin. J Control Release 2010;145(1):26-32. [PMID: 20346994 DOI: 10.1016/j.jconrel.2010.03.013]
- Park EJ, Werner J, Smith NB. Ultrasound mediated transdermal insulin delivery in pigs using a lightweight transducer. Pharm Res 2007;24(7):1396-401. [PMID: 17443398 DOI: 10.1007/s11095-007-9306-4]
- Schoellhammer CM, Polat BE, Mendenhall J, Maa R, Jones B, et al. Rapid skin permeabilization by the simultaneous application of dual-frequency, high-intensity ultrasound. J Control Release 2012;163(2):154-60. [PMID: 22940128 DOI: 10.1016/j.jconrel.2012.08.019]
- Liu H-L, Hsieh C-M. Single-transducer dual-frequency ultrasound generation to enhance acoustic cavitation. Ultrason Sonochem 2009;16(3):431-8. [PMID: 18951828 DOI: 10.1016/j.ultsonch.2008.08.009]
- Rao R, Nanda S, Nair AB. Influence of ultrasound mediated transdermal delivery of losartan potassium. Drug Deliv Lett 2012;2(1):46-53. [DOI: 10.2174/2210304×11202010046]
- Smith NB, Lee S, Maione E, Roy RB, McElligott S, et al. Ultrasound-mediated transdermal transport of insulin in vitro through human skin using novel transducer designs. Ultrasound Med Biol 2003;29(2):311-7. [PMID: 12659919 DOI: 10.1016/s0301-5629(02)00706-8]
- Smith NB, Lee S, Shung KK. Ultrasound-mediated transdermal in vivo transport of insulin with low-profile cymbal arrays. Ultrasound Med Biol 2003;29(8):1205-10. [PMID: 12946523 DOI: 10.1016/s0301-5629(03)00908-6]
- Li S, Xu J, Li R, Wang Y, Zhang M, et al. Stretchable electronic facial masks for sonophoresis. ACS Nano 2022;16(4):5961-74. [PMID: 35363481 DOI: 10.1021/acsnano.1c11181]
- Makvandi P, Shabani M, Rabiee N, Anjani QK, Maleki A, et al. Engineering and development of a tissue model for the evaluation of microneedle penetration ability, drug diffusion, photothermal activity, and ultrasound imaging: a promising surrogate to ex vivo and in vivo tissues. Adv Mater 2023;35(18):e2210034. [PMID: 36739591 DOI: 10.1002/adma.202210034]
- Shu W, Heimark H, Bertollo N, Tobin DJ, O’Cearbhaill ED, et al. Insights into the mechanics of solid conical microneedle array insertion into skin using the finite element method. Acta Biomater 2021;135:403-13. [PMID: 34492370 DOI: 10.1016/j.actbio.2021.08.045]
- Wu H, Chen Z, Jiang Z, Chen M, Shan H, et al. Ultrasonic hollow microneedle array (USHM) for androgenetic alopecia treatment through modulating the expression of hair-growth-associated genes. ACS Appl Mater Interfaces 2025;17(14):21419-30. [PMID: 40145651 DOI: 10.1021/acsami.5c00379]
- Chen Z, Liu X, Jiang Z, Wu H, Yang T, et al. A piezoelectric-driven microneedle platform for skin disease therapy. Innovation (Camb) 2024;5(3):100621. [PMID: 38680817 DOI: 10.1016/j.xinn.2024.100621]
- Ouyang Q, Zeng Y, Yu Y, Tan L, Liu X, et al. Ultrasound-responsive microneedles eradicate deep-layered wound biofilm based on TiO2 crystal phase engineering. Small 2023;19(3):e2205292. [PMID: 36408892 DOI: 10.1002/smll.202205292]
- Wang K, Han C, Li J, Qiu J, Sunarso J, et al. The mechanism of piezocatalysis: energy band theory or screening charge effect? Angew Chem Int Ed Engl 2022;61(6):e202110429. [PMID: 34612568 DOI: 10.1002/anie.202110429]
- Xue H, Jin J, Huang X, Tan Z, Zeng Y, et al. Wearable flexible ultrasound microneedle patch for cancer immunotherapy. Nat Commun 2025;16(1):2650. [PMID: 40102412 DOI: 10.1038/s41467-025-58075-z]
- Zandi A, Khayamian MA, Saghafi M, Shalileh S, Katebi P, et al. Microneedle-based generation of microbubbles in cancer tumors to improve ultrasound-assisted drug delivery. Adv Healthc Mater 2019;8(17):e1900613. [PMID: 31328442 DOI: 10.1002/adhm.201900613]
- Yan P, Liu L-H, Wang P. Sonodynamic therapy (SDT) for cancer treatment: advanced sensitizers by ultrasound activation to injury tumor. ACS Appl Bio Mater 2020;3(6):3456-75. [PMID: 35025218 DOI: 10.1021/acsabm.0c00156]
- Xiang Y, Lu J, Mao C, Zhu Y, Wang C, et al. Ultrasound-triggered interfacial engineering-based microneedle for bacterial infection acne treatment. Sci Adv 2023;9(10):eadf0854. [PMID: 36888703 DOI: 10.1126/sciadv.adf0854]
- Cao L, Liu C, Mu C, Li Q, Wu M, et al. Ultrasound-responsive carbon monoxide microneedle for enhanced healing of infected diabetic wounds. Adv Healthc Mater 2025;14(3):e2402910. [PMID: 39573876 DOI: 10.1002/adhm.202402910]
- Hu T, Jia L, Li H, Yang C, Yan Y, et al. An intelligent and soluble microneedle composed of Bi/BiVO4 Schottky heterojunction for tumor Ct imaging and starvation/gas therapy-promoted synergistic cancer treatment. Adv Healthc Mater 2024;13(8):e2303147. [PMID: 38206853 DOI: 10.1002/adhm.202303147]
- Zhong T, Yi H, Gou J, Li J, Liu M, et al. A wireless battery-free eye modulation patch for high myopia therapy. Nat Commun 2024;15(1):1766. [PMID: 38409083 DOI: 10.1038/s41467-024-46049-6]
- Dong W, Ye J, Wang W, Yang Y, Wang H, et al. Self-assembled lecithin/chitosan nanoparticles based on phospholipid complex: a feasible strategy to improve entrapment efficiency and transdermal delivery of poorly lipophilic drug. Int J Nanomedicine 2020;15:5629-43. [PMID: 32801706 DOI: 10.2147/IJN.S261162]
- Lyu S, Dong Z, Xu X, Bei H-P, Yuen HY, et al. Going below and beyond the surface: microneedle structure, materials, drugs, fabrication, and applications for wound healing and tissue regeneration. Bioact Mater 2023;27:303-26. [PMID: 37122902 DOI: 10.1016/j.bioactmat.2023.04.003]
- Gomaa YA, Garland MJ, McInnes FJ, Donnelly RF, El-Khordagui LK, et al. Microneedle/nanoencapsulation-mediated transdermal delivery: mechanistic insights. Eur J Pharm Biopharm 2014;86(2):145-55. [PMID: 23461860 DOI: 10.1016/j.ejpb.2013.01.026]
- Dragicevic N, Maibach H. Combined use of nanocarriers and physical methods for percutaneous penetration enhancement. Adv Drug Deliv Rev 2018;127:58-84. [PMID: 29425769 DOI: 10.1016/j.addr.2018.02.003]
- Yuan H, Han P, Tao K, Liu S, Gazit E, et al. Piezoelectric peptide and metabolite materials. Research (Wash D C) 2019;2019:9025939. [PMID: 31912048 DOI: 10.34133/2019/9025939]
- Zeng Q, Li G, Chen W. Ultrasound-activatable and skin-associated minimally invasive microdevices for smart drug delivery and diagnosis. Adv Drug Deliv Rev 2023;203:115133. [PMID: 37925075 DOI: 10.1016/j.addr.2023.115133]
- Han W, Liu F, Liu G, Li H, Xu Y, et al. Research progress of physical transdermal enhancement techniques in tumor therapy. Chem Commun (Camb) 2023;59(23):3339-59. [PMID: 36815500 DOI: 10.1039/d2cc06219d]
- Hu H, Guo L, He W, Zhang H, Xiang Y, et al. Roller microneedles transdermal delivery of compound lidocaine cream for enhancing the analgesic effect: a randomized self-controlled trial. J Cosmet Dermatol 2022;21(11):5825-36. [PMID: 35620951 DOI: 10.1111/jocd.15114]
- Rzhevskiy A, Popov A, Pavlov C, Anissimov Y, Zvyagin A, et al. Intradermal injection of lidocaine with a microneedle device to provide rapid local anaesthesia for peripheral intravenous cannulation: a randomised open-label placebo-controlled clinical trial. PLoS One 2022;17(1):e0261641. [PMID: 35100279 DOI: 10.1371/journal.pone.0261641]
- El-Fakahany H, Medhat W, Abdallah F, Abdel-Raouf H, Abdelhakeem M. Fractional microneedling: a novel method for enhancement of topical anesthesia before skin aesthetic procedures. Dermatol Surg 2016;42(1):50-5. [PMID: 26671202 DOI: 10.1097/DSS.0000000000000580]
- Daly S, Claydon NCA, Newcombe RG, Seong J, Addy M, et al. Randomised controlled trial of a microneedle patch with a topical anaesthetic for relieving the pain of dental injections. J Dent 2021;107:103617. [PMID: 33636242 DOI: 10.1016/j.jdent.2021.103617]
- McVey E, Hirsch L, Sutter DE, Kapitza C, Dellweg S, et al. Pharmacokinetics and postprandial glycemic excursions following insulin lispro delivered by intradermal microneedle or subcutaneous infusion. J Diabetes Sci Technol 2012;6(4):743-54. [PMID: 22920798 DOI: 10.1177/193229681200600403]
- Pettis RJ, Hirsch L, Kapitza C, Nosek L, Hövelmann U, et al. Microneedle-based intradermal versus subcutaneous administration of regular human insulin or insulin lispro: pharmacokinetics and postprandial glycemic excursions in patients with type 1 diabetes. Diabetes Technol Ther 2011;13(4):443-50. [PMID: 21355716 DOI: 10.1089/dia.2010.0183]
- Rini CJ, McVey E, Sutter D, Keith S, Kurth H-J, et al. Intradermal insulin infusion achieves faster insulin action than subcutaneous infusion for 3-day wear. Drug Deliv Transl Res 2015;5(4):332-45. [PMID: 26037035 DOI: 10.1007/s13346-015-0239-x]
- Jina A, Tierney MJ, Tamada JA, McGill S, Desai S, et al. Design, development, and evaluation of a novel microneedle array-based continuous glucose monitor. J Diabetes Sci Technol 2014;8(3):483-7. [PMID: 24876610 DOI: 10.1177/1932296814526191]
- Norman JJ, Brown MR, Raviele NA, Prausnitz MR, Felner EI. Faster pharmacokinetics and increased patient acceptance of intradermal insulin delivery using a single hollow microneedle in children and adolescents with type 1 diabetes. Pediatr Diabetes 2013;14(6):459-65. [PMID: 23517449 DOI: 10.1111/pedi.12031]
- Eid RO, Shaarawi E, Hegazy RA, Hafez V. Long-term efficacy of fractional microneedle radiofrequency versus botulinum toxin-a in primary axillary hyperhidrosis: a randomized controlled trial. Lasers Med Sci 2024;39(1):177. [PMID: 38981914 DOI: 10.1007/s10103-024-04115-x]
- Fatemi Naeini F, Abtahi-Naeini B, Pourazizi M, Nilforoushzadeh MA, Mirmohammadkhani M. Fractionated microneedle radiofrequency for treatment of primary axillary hyperhidrosis: a sham control study. Australas J Dermatol 2015;56(4):279-84. [PMID: 25496000 DOI: 10.1111/ajd.12260]
- Rummaneethorn P, Chalermchai T. A comparative study between intradermal botulinum toxin A and fractional microneedle radiofrequency (FMR) for the treatment of primary axillary hyperhidrosis. Lasers Med Sci 2020;35(5):1179-84. [PMID: 31939036 DOI: 10.1007/s10103-020-02958-8]
- Song J-H, Park S-Y. Biodegradable microneedle acupuncture has equivalent efficacy to traditional intradermal acupuncture for dry eye disease: a pilot single-blinded, randomized controlled trial. Medicine (Baltimore) 2024;103(2):e36864. [PMID: 38215150 DOI: 10.1097/MD.0000000000036864]
- Song J-H, Park S-Y. A comparative investigator-initiated pilot study on the efficacy and safety of biodegradable microneedle acupuncture and conventional intradermal acupuncture for dry eye patient: a superiority, assessor-blinded, randomized controlled trial. Medicine (Baltimore) 2022;101(47):e31468. [PMID: 36451509 DOI: 10.1097/MD.0000000000031468]
- Maruani A, Vierron E, Machet L, Giraudeau B, Boucaud A. Efficiency of low-frequency ultrasound sonophoresis in skin penetration of histamine: a randomized study in humans. Int J Pharm 2010;385(1-2):37-41. [PMID: 19837146 DOI: 10.1016/j.ijpharm.2009.10.024]
- Clijsen R, Baeyens JP, Barel AO, Clarys P. Influence of the timing of ultrasound application on the penetration of corticosteroids. Skin Res Technol 2013;19(1):e279-82. [PMID: 22712560 DOI: 10.1111/j.1600-0846.2012.00639.x]
- Ebrahimi S, Abbasnia K, Motealleh A, Kooroshfard N, Kamali F, et al. Effect of lidocaine phonophoresis on sensory blockade: pulsed or continuous mode of therapeutic ultrasound? Physiotherapy 2012;98(1):57-63. [PMID: 22265386 DOI: 10.1016/j.physio.2011.01.009]
- Tian A-P, Yin Y-K, Yu L, Yang B-Y, Li N, et al. Low-frequency sonophoresis of chinese medicine formula improves efficacy of malignant pleural effusion treatment. Chin J Integr Med 2020;26(4):263-9. [PMID: 31444669 DOI: 10.1007/s11655-019-3167-7]
- Rigby JH, Hagan AM, Kelcher AR, Ji C. Dexamethasone sodium phosphate penetration during phonophoresis at 2 ultrasound frequencies. J Athl Train 2020;55(6):628-35. [PMID: 32320285 DOI: 10.4085/1062-6050-556-18]
- Tian A-P, Yin Y-K, Yu L, Yang B-Y, Li N, et al. Topical delivery of modified Da-Cheng-Qi Decoction () using low-frequency ultrasound sonophoresis for refractory metastatic malignant bowel obstruction: an open-label single-arm clinical trial. Chin J Integr Med 2020;26(5):382-7. [PMID: 31134466 DOI: 10.1007/s11655-019-3041-7]
- Maruani A, Boucaud A, Perrodeau E, Gendre D, Giraudeau B, et al. Low-frequency ultrasound sonophoresis to increase the efficiency of topical steroids: a pilot randomized study of humans. Int J Pharm 2010;395(1-2):84-90. [PMID: 20472045 DOI: 10.1016/j.ijpharm.2010.05.010]
- Katz NP, Shapiro DE, Herrmann TE, Kost J, Custer LM. Rapid onset of cutaneous anesthesia with EMLA cream after pretreatment with a new ultrasound-emitting device. Anesth Analg 2004;98(2):371-6. [PMID: 14742372 DOI: 10.1213/01.ANE.0000099716.02783.C4]
- Iwata H, Kakita K, Imafuku K, Takashima S, Haga N, et al. Safety and dose-sparing effect of Japanese encephalitis vaccine administered by microneedle patch in uninfected, healthy adults (MNA-J): a randomised, partly blinded, active-controlled, phase 1 trial. Lancet Microbe 2022;3(2):e96-e104. [PMID: 35544051 DOI: 10.1016/S2666-5247(21)00269-X]
- Ono A, Azukizawa H, Ito S, Nakamura Y, Asada H, et al. Development of novel double-decker microneedle patches for transcutaneous vaccine delivery. Int J Pharm 2017;532(1):374-83. [PMID: 28855138 DOI: 10.1016/j.ijpharm.2017.08.110]
- Hiraishi Y, Hirobe S, Iioka H, Quan Y-S, Kamiyama F, et al. Development of a novel therapeutic approach using a retinoic acid-loaded microneedle patch for seborrheic keratosis treatment and safety study in humans. J Control Release 2013;171(2):93-103. [PMID: 23791975 DOI: 10.1016/j.jconrel.2013.06.008]
- Kochba E, Levin Y, Raz I, Cahn A. Improved insulin pharmacokinetics using a novel microneedle device for intradermal delivery in patients with type 2 diabetes. Diabetes Technol Ther 2016;18(9):525-31. [PMID: 27500713 DOI: 10.1089/dia.2016.0156]
- Papich MG, Narayan RJ. Naloxone and nalmefene absorption delivered by hollow microneedles compared to intramuscular injection. Drug Deliv Transl Res 2022;12(2):376-83. [PMID: 34817831 DOI: 10.1007/s13346-021-01096-0]
- Brogden NK, Milewski M, Ghosh P, Hardi L, Crofford LJ, et al. Diclofenac delays micropore closure following microneedle treatment in human subjects. J Control Release 2012;163(2):220-9. [PMID: 22929967 DOI: 10.1016/j.jconrel.2012.08.015]
- Cheng Z, Lin H, Wang Z, Yang X, Zhang M, et al. Preparation and characterization of dissolving hyaluronic acid composite microneedles loaded micelles for delivery of curcumin. Drug Deliv Transl Res 2020;10(5):1520-30. [PMID: 32100266 DOI: 10.1007/s13346-020-00735-2]