Mechanistic Insights and Therapeutic Potential of Quercetin in Neuroprotection: A Comprehensive Review of Pathways and Clinical Perspectives
1Department of Pharmaceutical Chemistry, Bengal College of Pharmaceutical Sciences & Research, Durgapur, West Bengal-713212, India
2Department of Pharmaceutical Chemistry, Bengal College of Pharmaceutical Technology, Dubrajpur, West Bengal-731123, India
3Department of Pharmaceutics, Bengal College of Pharmaceutical Technology, Dubrajpur, West Bengal-731123, India
4Department of Pharmaceutical Science, Guru Nanak Institute of Pharmaceutical Science and Technology, Kolkata-700114, West Bengal, India
5Department of Pharmaceutical Chemistry, Dr. B. C. Roy College of Pharmacy and Allied Health Sciences, Durgapur, West Bengal-713212, India
6Department of Pharmacology, Gupta College of Technological Sciences, Asansol, West Bengal-713301, India
*Correspondence to: Sumit Nandi, Department of Pharmacology, Gupta College of Technological Sciences, Asansol, West Bengal-713301, India. E-mail: sumitrx27@gmail.com
Received: April 21 2025; Revised: May 3 2025; Accepted: July 22 2025; Published Online: August 18 2025
Cite this paper:
Debnath I, Ghosh S, Jha SK et al. Mechanistic Insights and Therapeutic Potential of Quercetin in Neuroprotection: A Comprehensive Review of Pathways and Clinical Perspectives. BIO Integration 2025; 6: 1–31.
DOI: 10.15212/bioi-2025-0073. Available at: https://bio-integration.org/
Download citation
© 2025 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Abstract
Quercetin, a bioactive flavonoid abundant in diverse plant species, has been extensively investigated for its neuroprotective properties against neurodegenerative diseases (NDDs), such as Alzheimer’s, Parkinson’s, and Huntington’s diseases. This review systematically explored the multifaceted therapeutic potential of quercetin, emphasizing the mechanisms of action, pharmacologic efficacy, and translational significance in modern neurotherapeutics. Quercetin demonstrated potent antioxidant effects by scavenging reactive oxygen species and modulating the Nrf2-ARE pathway, thereby mitigating oxidative stress, a hallmark of NDDs associated with mitochondrial dysfunction, protein aggregation, and neuronal apoptosis. Furthermore, the ability of quercetin to regulate the PI3K/Akt pathway promoted mitochondrial biogenesis and preserved neuronal integrity by stabilizing membrane potential. Anti-inflammatory effects were evident vis-a-vis inhibition of the NF-κB and MAPK pathways, suppression of microglial activation, and cytokine release. In addition, quercetin disrupted tau hyperphosphorylation via GSK3β inhibition and attenuated amyloid-beta toxicity, offering cognitive protection. Preclinical studies highlighted the ability of quercetin to modulate excitotoxicity and enhance neuroplasticity, while emerging evidence support synergy of quercetin with existing pharmacologic agents. Genetic variations influencing key pathways, including Nrf2 and PI3K, underscore the necessity for personalized therapeutic approaches. Advances in drug delivery systems, scaffold modelling, and CRISPR-mediated interventions revealed the potential for optimizing the bioavailability and specificity of quercetin. This review bridges critical knowledge gaps by integrating mechanistic insights with clinical perspectives, advocating for translating quercetin-based therapies into precision medicine. By addressing challenges in bioavailability and exploring innovative strategies, this article underscores the promise of quercetin as a cornerstone for neuroprotective interventions in NDDs.
Keywords
Inflammatory pathways, neurodegenerative diseases, neuroprotection, oxidative stress, quercetin, therapeutic potential.
Introduction
Quercetin, a bioactive flavonoid, has emerged as a significant neuroprotective agent with multifaceted therapeutic potential in combating neurodegenerative diseases (NDDs), such as Alzheimer’s (AD), Parkinson’s (PD), and Huntington’s diseases (HD) [1]. These progressive disorders, driven by mechanisms like oxidative stress, mitochondrial dysfunction, neuro-inflammation, and apoptotic pathways, represent a growing global health burden with limited curative options [2, 3]. This review aims to comprehensively elucidate the role of quercetin in mitigating these pathologic processes, emphasizing the underlying molecular mechanisms, pharmacologic applications, and future clinical perspectives. Drawing upon extensive findings, including the findings in the provided documents, the review sought to bridge knowledge gaps and underscore the relevance of quercetin in modern neurotherapeutics. The therapeutic potential of quercetin lies in robust antioxidant activity of quercetin, mediated by an ability to scavenge reactive oxygen species (ROS) and enhance the nuclear factor erythroid 2-related factor 2 (Nrf2)-ARE pathway [4]. This mechanism is pivotal in reducing oxidative stress, a hallmark of NDDs that exacerbates protein aggregation, mitochondrial damage, and neuronal apoptosis [5]. The structural components of quercetin, including hydroxyl groups and conjugated systems, further augment the efficacy of quercetin in targeting ROS and inhibiting lipid peroxidation. These properties have been validated in vitro and in vivo, demonstrating protection against amyloid-beta (Aβ) accumulation in AD models and α-synuclein fibrillization in PD [6, 7]. Mitochondrial dysfunction, another critical factor in NDDs, is effectively countered by quercetin [3]. Quercetin enhances mitochondrial biogenesis and maintains membrane potential by modulating the PI3K/Akt pathway, thereby reducing apoptosis and energy deficits [8]. Studies highlight the ability of quercetin to restore mitochondrial function in HD models, mitigating oxidative damage and preserving neuronal integrity. Furthermore, the ability of quercetin to inhibit glycogen synthase kinase-3β (GSK3β) disrupts tau (τ) hyperphosphorylation and Aβ toxicity, offering neuroprotection against cognitive decline in AD [9]. The anti-inflammatory properties of quercetin suppress microglial activation and cytokine release, primarily through the NF-κB and mitogen-activated protein kinase (MAPK) pathways, thereby minimizing neuroinflammation, a driving force behind neuronal damage in AD and PD [10]. Additionally, the interaction of quercetin with the paraoxonase-2 (PON2) pathway enhances antioxidant capacity, further protecting neurons from inflammatory and oxidative insults [11]. Quercetin mitigates excitotoxicity in HD by modulating glutamate receptor activity, which provides neuroprotection against excitatory amino acid-induced damage [12]. Emerging studies underscore the potential of quercetin in precision medicine. Genetic polymorphisms influencing the efficacy of the Nrf2 and PI3K pathways necessitate personalized approaches to optimize quercetin therapy [13]. The integration of quercetin with existing pharmacologic agents, such as cholinesterase inhibitors in AD, exemplifies the synergistic potential of quercetin [14]. Moreover, the ability of quercetin to target gene expression, as occurs with CRISPR-modulated SIRT1 activation, opens avenues for combination therapies addressing the multifactorial nature of NDDs [15]. The structural analogs of quercetin (scaffold modelling), derived through rational design and computational modelling, promise enhanced specificity and potency against neurodegenerative targets [16]. The inclusion of marine-derived flavonoids and other polyphenols in quercetin-based research broadens the scope for novel therapeutics [17]. This review evaluated current knowledge and highlighted future directions, emphasizing the need for innovative strategies to enhance the clinical translation of quercetin. The integration of advanced drug delivery systems, personalized medicine, scaffold-based drug development, and computational modelling enhances specificity and potency against neurodegenerative targets.
Sources of quercetin from nature
The term, quercetin, originated from the Latin word, quercetum, in 1857 and refers to oak, reflecting the natural prevalence of quercetin in plant ecosystems [18]. Quercetin is commonly found as glycosides, such as rutin, isoquercetin, and hyperin. Quercetin is a potent plant antioxidant that protects the human body from oxidative damage and offers numerous health benefits [19]. Quercetin is abundant in fruits (apples, grapes, and berries) and vegetables (onions, garlic, and tomatoes). In addition, quercetin is present in medicinal plants and extracted from the leaves, fruits, or flowering tops with these sources being particularly rich in quercetin [20, 21]. Table 1 describes the biological sources of quercetin (quercetin is extracted from parts of plants under different families).
Table 1 Sources of Quercetin From Nature
| Serial No. | Family | Scientific Names | Common Names | Quercetin Presence in Plant Parts | References |
|---|---|---|---|---|---|
| 1. | Amaryllidaceae | Allium cepa | Onion (red) | Bulb | [195] |
| Allium sativum L. | Garlic | Leaves, cloves | |||
| 2. | Moraceae | Morus alba | White mulberry | Leaves | [196] |
| 3. | Rosaceae | Cydonia oblonga | Quince | Fruits, leaves | [197] |
| Malus domestica | Apple | Fruits | [198] | ||
| Prunus avium | Sweet cherry | Leaves, fruits, flowers, stems | [199] | ||
| Prunus domestica | European plum, Prune plum, Bullace, Damson, Dawson, Gage, Gardalu, and Greengage | Fruit | [200] | ||
| 4. | Moringaceae | Moringa oleifera | Drumstick tree | Leaves, flowers | [201] |
| 5. | Lythraceae | Punica granatum | Pomegranate | Fruits | [202] |
| 6. | Sapotaceae | Achras sapota | Sapodilla | Fruits | [3] |
| 7. | Lamiaceae | Mentha piperita L. | Peppermint | Leaves, herbs | [203] |
| Mentha spicata | Spearmint | Leaves, herbs | [204] | ||
| 8. | Rhamnaceae | Rhamnus alaternus | Buckthorn | Bark | [205] |
| 9. | Asteraceae | Achillea millefolium L. | Yarrow | Flowering top | [206] |
| Lactuca sativa | Lettuce | Leaves | [207] | ||
| 10. | Ginkgoaceae | Ginkgo biloba | Maidenhair tree | Leaves | [208] |
| 11. | Anacardiaceae | Mangifera indica | Mango | Fruits | [209] |
| 12. | Compositae | Cichorium intybus | Chicory | Leaves | [210] |
| 13. | Ericacea | Vaccinium myrtillus | Blueberry, bilberry | Fruits | [211] |
| Vaccinium oxycoccus | Cranberry | Fruits, leaves | [212] | ||
| 14. | Theaceae | Camellia sinensis | Green tea | Leaves | [213] |
| 15. | Brassicaceae | Brassica oleracea var. Italica | Broccoli | Leaves, flower | [214] |
| Brassica oleracea var. Sabellica | Kale | Leaves | [215] | ||
| Nasturtium officinale | Watercress | Flowers, leaves, stem | [216] | ||
| 16. | Hypericaceae | Hypericum perforatum L | St. John’s wort | Aerial parts (leaves, flowers) | [217, 218] |
| 17. | Apiaceae | Centella asiatica | Indian pennywort, spade leaf, coin worth, or gotu kola | Leaves | [219] |
| Apium graveolens L. | Celery | Seeds | [220] | ||
| Coriandum sativum | Cilantro, Chinese parsley | Leaves, seeds | [221] | ||
| 18. | Chenopodiaceae | Spinacia oleracea | Spinach | Leaves | [222] |
| 19. | Maleacae | Azadirachta indica | Neem | Leaves, bark | [223] |
| 20. | Fabaceae | Mimosa pudica | “Touch me not” or “Sensitive plant” | Whole plant | [224] |
| Sesbania grandiflora | Agati, Corkwood tree, and West Indian pea | Leaves | [225] | ||
| Delonix regia | Gulmohar | Leaves | [226] | ||
| 21. | Cannabaceae | Cannabis sativa | Hemp, marijuana | Leaves | [227] |
| 22. | Acanthaceae | Andrographis paniculata | Kalmegh | Leaves | [228] |
| Justicia adhatoda | Basak | Leaves | [229] | ||
| 23. | Libiatae | Ocimum sanctum | Tulshi | Leaves | [230] |
| 24. | Musaceae | Musa acuminata | Colla, banana | Fruit pulp | [231] |
| 25. | Solanaceae | Solanum lycopersicum | Tomato | Fruit, seeds | [232, 233] |
Dose-dependent effects, administration regimens, and toxicologic considerations
Dose-dependent biphasic effects of quercetin
Quercetin exhibits a biphasic dose-response curve (hormesis), i.e., low-to-moderate doses confer neuroprotection and high doses induce cytotoxicity. In vitro studies indicated that concentrations < 10–20 μM are generally neuroprotective via antioxidant and anti-inflammatory effects [6]. However, a quercetin concentration >50 μM may result in mitochondrial membrane disruption, DNA damage, and pro-oxidant activity [22]. Animal models have revealed that chronic administration > 50 mg/kg/day may impair hepatic antioxidant defenses and trigger apoptotic pathways in non-target cells [23].
Pharmacokinetics and bioavailability challenges
Oral bioavailability of quercetin is low (< 10%) due to poor water solubility, gastrointestinal degradation, and rapid phase II metabolism [24]. Quercetin undergoes extensive glucuronidation, sulfation, and methylation in the liver and intestines, limiting the concentration in the systemic circulation. Peak plasma concentrations of quercetin following a typical dietary intake (~100 mg/day) rarely exceed 1–2 μM, which is significantly lower than doses used in in vitro models [25, 26].
Routes of administration and novel delivery systems
Novel delivery strategies, nanoparticles, liposomes, solid lipid carriers, and PEGylated formulations have been developed to overcome poor oral absorption, enhance CNS penetration, and protect the compound from rapid metabolism. Intranasal administration has emerged as a promising non-invasive route for direct delivery to the brain, bypassing the blood-brain barrier (BBB) [27, 28].
Clinical considerations and therapeutic index
The therapeutic window for quercetin is narrow and may vary based on individual metabolic capacity, genetic polymorphisms (e.g., UGT and COMT), and co-morbid diseases. There is an urgent need for standardized clinical trials evaluating dose escalation, long-term safety, and pharmacokinetic-pharmacodynamic (PK-PD) correlations. Personalized medicine approaches may be required to tailor quercetin dosages, especially in patients with neurodegenerative disorders who may have altered hepatic or renal function [29].
Molecular mechanisms of quercetin-induced cellular damage at high concentrations
Mitochondrial membrane disruption
Quercetin accumulates within mitochondria at high concentrations, destabilizing the mitochondrial membrane potential. This effect occurs due to the interaction of quercetin with inner mitochondrial membrane phospholipids, especially cardiolipin, leading to permeabilization. Mitochondrial permeability transition pores (mPTPs) open, causing cytochrome c release, calcium dysregulation, ATP depletion, and ultimately initiating intrinsic apoptosis [30].
Pro-oxidant activity
Quercetin exhibits dual redox properties. Quercetin scavenges ROS at physiologic concentrations. However, quercetin undergoes autoxidation at supra-physiologic levels, forming semiquinone radicals and quercetin-quinone (QQ) species. These intermediates deplete intracellular glutathione (GSH), elevate hydrogen peroxide (H2O2), and catalyze Fenton reactions with transition metals (Fe2+ and Cu2+), producing hydroxyl radicals and exerting oxidative stress [31].
DNA damage and genotoxicity
Quercetin-quinone metabolites can covalently bind to DNA and nucleophilic cellular proteins, leading to strand breaks and chromatin damage. Studies using the Comet assay and micronucleus formation in vitro have revealed that high-dose quercetin (≥ 100 μM) causes double-stranded DNA breaks, chromosomal aberrations, and p53 pathway activation, potentially inducing cell cycle arrest or apoptosis [32].
Interference with cellular enzymes
At cytotoxic levels, quercetin inhibits topoisomerase II, impairs telomerase function, and alters redox-sensitive transcription factors (NF-κB and AP-1) in a deleterious manner. This suppression disrupts normal cellular repair and survival processes, contributing to carcinogenic risk under chronic high-dose exposure [33].
These findings emphasize the importance of careful dose calibration and duration of administration. While quercetin is generally regarded as safe at dietary levels, high-dose, long-term use, especially in the supplement form, requires caution and additional pharmacovigilance.
Description of chemical structure and pharmacokinetics of quercetin for the multifaceted neuroprotective potential in NDDs
Chemical structure
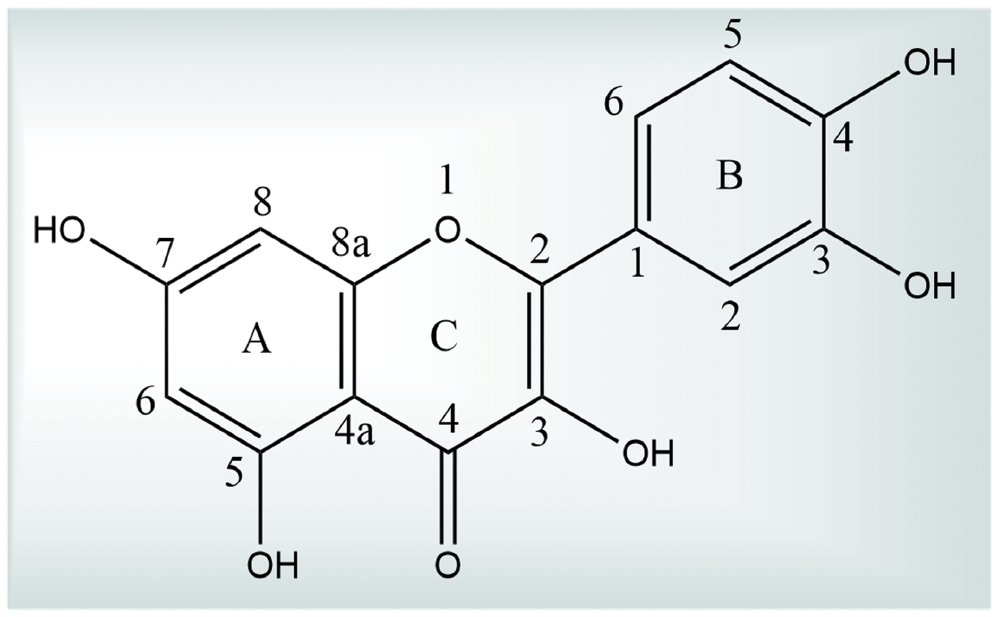
Quercetin, a prominent flavonoid belonging to the flavonol subclass, has the following molecular formula: C15H10O7 [34]. Structurally, quercetin consists of two aromatic rings (A and B) connected by a heterocyclic pyrone (C ring) with five hydroxyl groups at positions 3, 3’, 4’, 5, and 7 [35]. This polyphenolic configuration allows quercetin to scavenge free radicals, making quercetin one of the most potent antioxidants among dietary polyphenols. Quercetin also exists as glycosides, in which a glycosyl group attaches to the hydroxyl group (typically at position 3), which enhances solubility in aqueous media and reduces the free radical scavenging potential (Figure 1) [35, 36].
Figure 1 Chemical structure of quercetin.
Pharmacokinetics
Quercetin undergoes passive diffusion and is absorbed in the glycosidic and aglycone forms via organic anion-transporting polypeptides (OATPs) in the small intestine. Quercetin is rapidly metabolized inside the liver, intestines, and kidneys upon absorption and forms conjugated forms, such as glucuronides, sulfates, and methylated derivatives. The bioavailability of quercetin, which ranges from 0–50%, is considered poor due to extensive first-pass metabolism and rapid clearance with a half-life of 1–2 h. Strategies to overcome these limitations include the development of nano-formulations, such as nanoparticles, micelles, and mucoadhesive emulsions, which enhance bioavailability and brain penetration [3, 37].
Quercetin undergoes extensive and rapid metabolism after dietary absorption, which explains why the pharmacologic effects are more prominent in in vitro studies compared to in vivo conditions [38, 39]. The limitations of quercetin, including poor bioavailability, aqueous solubility, permeability, and instability, have restricted the application of quercetin for neurodegenerative diseases. Several studies have focused on structural modifications to address these challenges, improve these properties, and enhance the neuroprotective potential [3, 40]. Innovative formulations, such as quercetin-loaded gels, nanoparticles, polymeric micelles, and mucoadhesive nanoemulsions, have been developed to boost brain bioavailability [41]. Structural alterations, such as conjugation, have also improved the scavenging and antioxidant activities of quercetin [42].
Structure-activity relationship (SAR) of quercetin with the neuroprotective mechanism
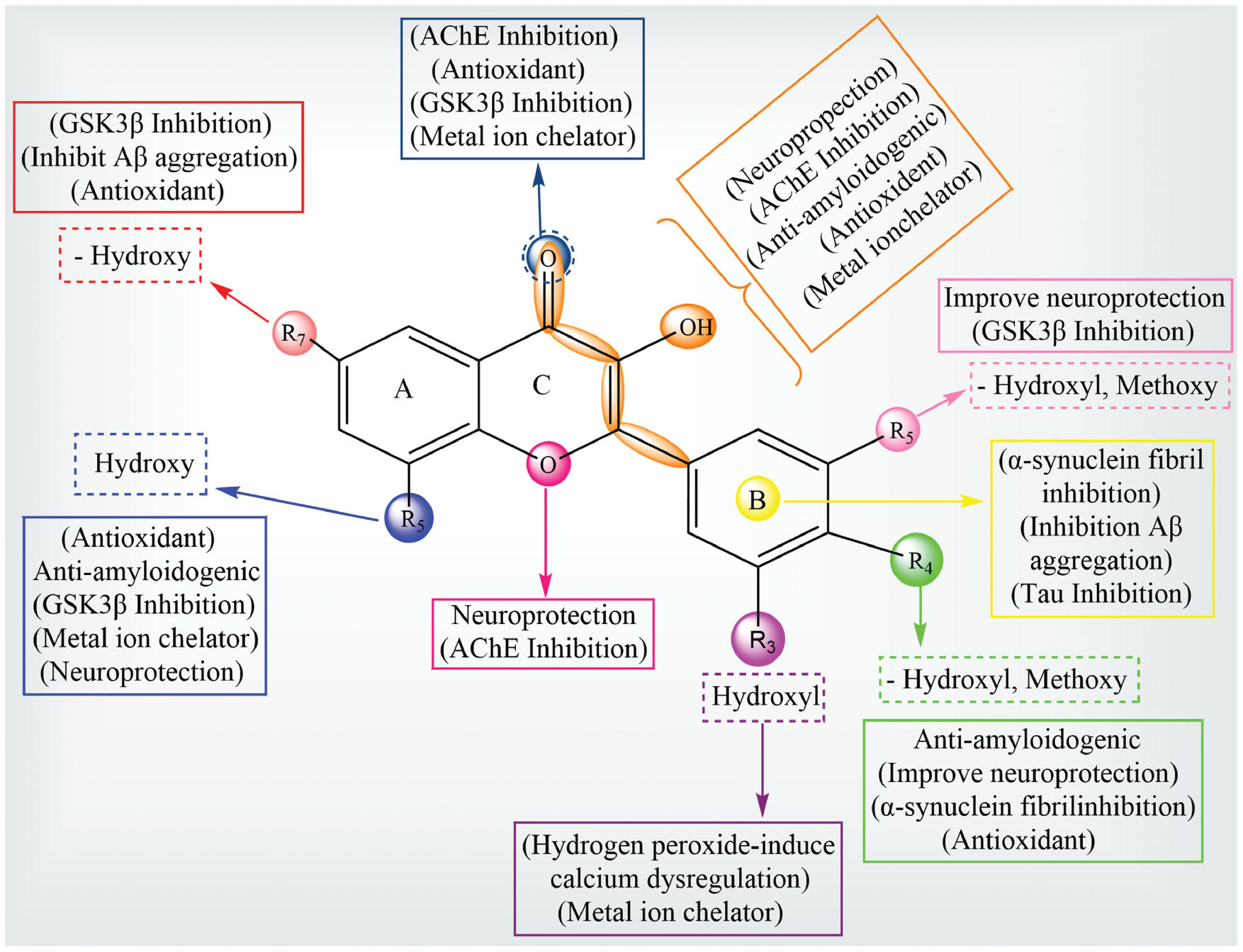
The neuroprotective mechanisms underlying quercetin are intricately linked to the SAR because the polyphenolic structure governs interactions with cellular targets and molecular pathways critical in combating neurodegeneration (Figure 2). Below is a detailed correlation between the SAR of quercetin and the neuroprotective mechanisms.
Figure 2 Quercetin SAR in relation to neuroprotective targets.
Antioxidant activity and free radical scavenging
SAR insight
The −OH groups at positions 3, 5, and 7 (A and C rings) and the ortho-dihydroxy structure in ring B significantly enhance the radical scavenging ability of quercetin. These groups stabilize ROS by donating hydrogen atoms, which neutralizes free radicals [43, 44].
Mechanistic correlation
Quercetin mitigates oxidative stress by scavenging ROS and reactive nitrogen species (RNS), which are the most significant contributors to neuronal damage in neurodegenerative diseases [45]. Quercetin stimulates the Nrf2/ARE pathway due to the radical-scavenging functional groups, which increases the expression of endogenous antioxidants, such as GSH, superoxide dismutase (SOD), and catalase (CAT) [18, 46].
Metal chelation
SAR insight
The ‘oxo’ group at the 4 position of ring C and the catechol group in the B ring form chelates with neurotoxic metal ions, such as Fe2+ and Cu2+. This chelation prevents Fenton reactions, which produce harmful ROS [47]. In addition, quercetin inhibits HIF-prolyl hydroxylase (HPH) by chelating iron, especially through hydroxyl (−OH) groups at the 3 position on the C ring and the 3’,4’ positions on the B ring, which further helps regulate the cellular response [48].
Mechanistic correlation
Excessive metal ions (Fe2+ and Cu2+) catalyze the production of ROS in AD, exacerbating oxidative stress and promoting Aβ aggregation. The chelation properties of quercetin neutralize these metal ions, reducing the formation of neurotoxic aggregates and protecting neuronal integrity [49].
Inhibition of protein aggregation
SAR insight
The catechol group in ring B and the conjugated π-electron system in the C ring interact with amyloidogenic proteins through hydrogen bonding and hydrophobic interactions [50].
Mechanistic correlation
Quercetin inhibits the aggregation of misfolded proteins, including Aβ and α-synuclein in AD and PD, respectively. Quercetin disrupts fibril formation and oligomerization by stabilizing proteins in non-toxic forms, thereby reducing synaptic dysfunction and neuronal death. This mechanism performs a crucial role in NDDs, which are characterized by toxic protein aggregates [51].
Anti-inflammatory activity
SAR insight
The A and B ring hydroxyl groups allow quercetin to inhibit enzymes and signalling pathways associated with inflammation, like the cyclooxygenase (COX) and lipoxygenase (LOX) pathways. Additionally, the C ring conjugated system enhances the interaction with inflammatory mediators [52].
Mechanistic correlation
Quercetin lowers the expression of pro-inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α), interleukin (IL)-6, and IL-1β, by blocking NF-κB pathway activation. Additionally, quercetin inhibits the activity of inflammatory enzymes (LOX and COX) [53]. This anti-inflammatory action prevents microglial activation and reduces neuroinflammation, which are hallmark features of multiple sclerosis (MS) and amyotrophic lateral sclerosis (ALS) [54].
Regulation of mitochondrial function
SAR insight
The hydroxyl group at position 3 and the catechol group of the B ring interact with mitochondrial enzymes and ion channels, preserving mitochondrial integrity [52, 55].
Mechanistic correlation
Quercetin enhances mitochondrial bioenergetics by stabilizing the mitochondrial membrane potential and preventing calcium overload. Quercetin reduces mitochondrial ROS production, thereby preventing mitochondrial DNA and protein damage due to oxidative stress [56]. Additionally, quercetin stimulates the PI3K/Akt signalling pathway, promoting mitochondrial biogenesis and improving energy metabolism, which is critical for neuronal survival in conditions, like HD and PD [15, 57].
Inhibition of apoptotic pathways
SAR insight
The conjugated system increases the binding affinity of quercetin to enzymes like caspases and kinases, while the −OH groups create hydrogen bonds with important residues in apoptotic proteins [58].
Mechanistic correlation
Quercetin suppresses apoptotic cascades by inhibiting the stimulation of pro-apoptotic proteins like caspase-3 and -9. Quercetin also downregulates the MAPK pathway, particularly the JNK and p38 branches, which are activated in response to cellular stress [59, 60]. By preventing neuronal apoptosis, quercetin preserves neural networks and cognitive function in diseases, like AD and HD [61].
Cholinesterase inhibition
SAR insight
The hydroxyl groups, particularly at position 3, interact with the catalytic active site of acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) through hydrogen bonding and hydrophobic interactions [62].
Mechanistic correlation
Quercetin inhibits AChE activity, increasing acetylcholine (ACh) levels in the synaptic cleft. This action enhances synaptic transmission and alleviates cognitive deficits associated with AD. BChE inhibition further supports the role of quercetin in preserving cholinergic function [63, 64].
Gene modulation
SAR insight
The conjugated polyphenolic system, including hydroxyl groups and the B ring catechol structure, allows interaction with transcription factors like Nrf2 and cyclic AMP response element binding protein (CREB) [65].
Mechanistic correlation
Quercetin increases the synthesis of antioxidant enzymes by stimulating the Nrf2/ARE pathway, which controls gene expression. Additionally, quercetin promotes neurogenesis and synaptic repair by increasing the expression of brain-derived neurotrophic factor (BDNF). These effects contribute to the neuroprotective potential of quercetin in NDDs, like ALS and HD [66, 67].
Quercetin demonstrates strong inhibition of GSK3β, binding with high affinity and forming four hydrogen bonds with essential amino acids (Asp200, Arg141, and Val135) [3, 68]. Neuroprotective activity is further improved by specific substitutions in the structure of quercetin, such as hydroxyl groups at carbon positions 5, 7, 3, and OCH3 or H substitutions at the carbon 3 and 4 positions. The modifications have been confirmed through in vitro studies with neuron cell cultures. Important structural elements that have been demonstrated to lessen H2O2-induced Ca2+ imbalance and oxidative stress include polyphenolic structures, like a 2,3-double bond conjugated with the 4-oxo group on ring C and the −OH group at the 3′,4′-hydroxyl group of the B ring [3, 6, 69]. Moreover, hydroxyl substitutions on the A ring, a 4-carbonyl group in the C ring, and ortho-dihydroxy substitution in the B ring are responsible for the metal ion-chelating action of quercetin. The ortho-dihydroxyl group also inhibits α-synuclein fibril formation, underscoring the therapeutic potential of quercetin in managing PD [70].
The structural basis for the neuroprotective activity of quercetin is highly dependent on specific functional groups and substitutions. Various functional elements, such as hydroxyl (−OH), carbonyl (=O), and methoxy (−OCH3) groups, are linked to specific biological effects (e.g., antioxidant, metal chelation, anti-aggregation, and anti-inflammatory activities) to facilitate a clearer understanding of these structural-functional relationships of the quercetin SAR. Figure 2 illustrates the quercetin SAR, highlighting the various biological effects associated with different functional groups (mainly hydroxyl and methoxy) at specific positions on the flavonoid skeleton. The substitutions at R3, R4, R5, R5’, and R7 significantly contribute to neuroprotection through diverse mechanisms. Hydroxyl or methoxy groups at R3, R4, and R5’ enhance antioxidant activity, anti-amyloidogenic effects, α-synuclein fibril inhibition, and improve neuroprotection. Hydroxyl groups at R5 and R7 are particularly important for inhibition of GSK3β, inhibition of Aβ aggregation, and metal ion chelation mechanisms linked to AD pathology. The central C ring, especially the C2=C3 double bond and 4-oxo group, has a vital role in AChE inhibition and antioxidant action and further supports metal ion chelation. The overall structure suggests that multiple substitutions contribute synergistically to neuroprotection by targeting oxidative stress, protein aggregation (Aβ and τ), and dysregulated kinase activity (GSK3β), while also modulating calcium signalling and metal ion toxicity. This comprehensive mapping shows how specific functional groups on flavonoids can be tailored to enhance therapeutic efficacy against NDDs.
Pathophysiologic mechanism of NDDs
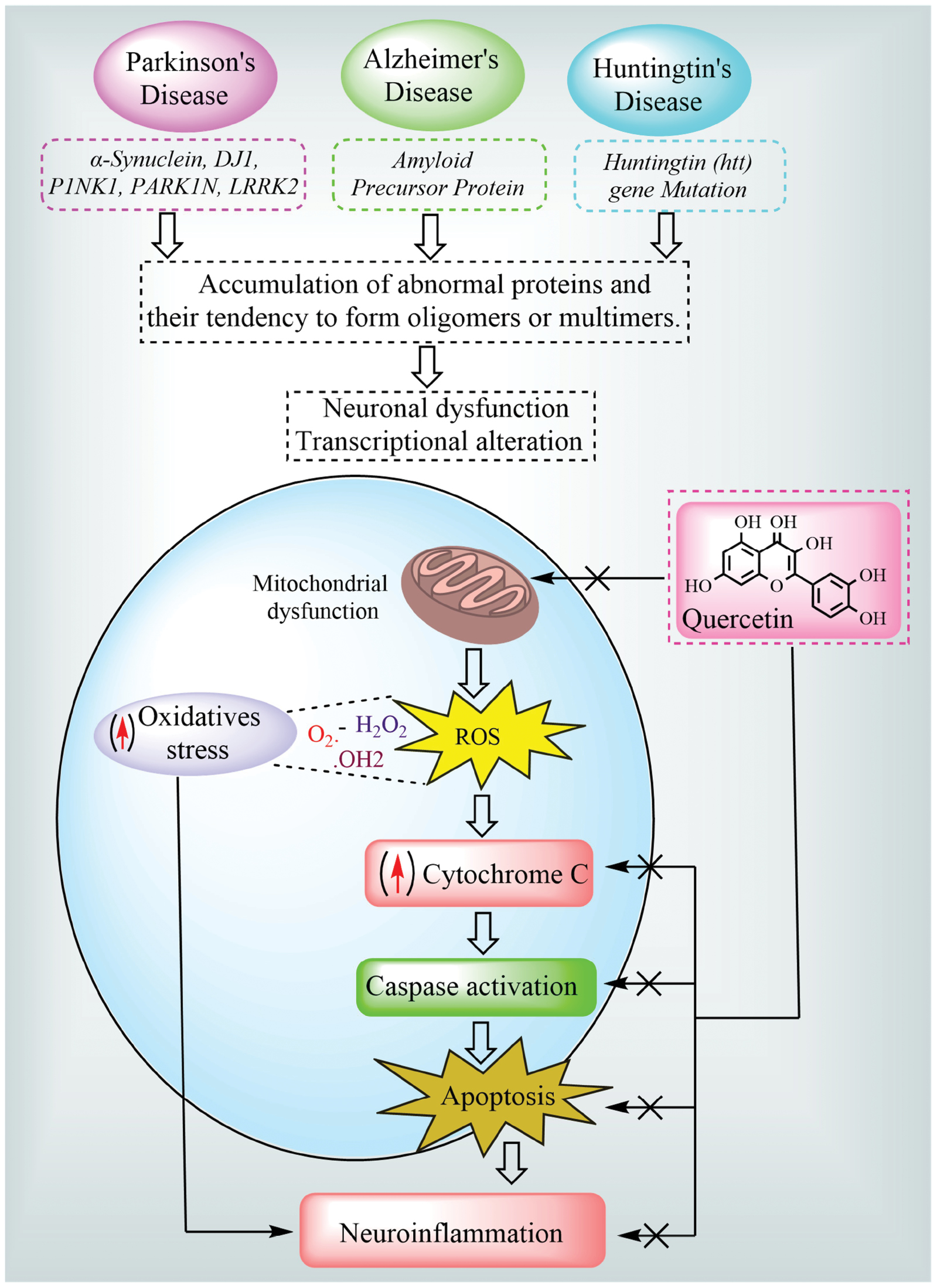
NDDs (AD, PD, and HD) are described by gradual neuronal loss, often with age-dependent onset. These conditions are marked by cognitive decline, motor dysfunction, and behavioral changes, underpinned by complex pathophysiologic processes, like protein misfolding and aggregation, oxidative stress, mitochondrial dysfunction, and inflammation [71]. Each disease has distinct pathologic hallmarks, as follows: Aβ plaques and neurofibrillary tangles in AD; α-synuclein aggregation in PD; and mutant huntingtin proteins in HD. These misfolded proteins destroy cellular function and cause the death of neurons in specific brain regions in major NDDs [72].
Mechanisms in AD
Amyloid plaque formation
The formation of Aβ plaques is the hallmark of AD. Aβ plaques are caused by beta (β) and gamma (γ)-secretases cleaving the amyloid precursor protein (APP). The toxic Aβ oligomers accumulate extracellularly, disrupting synaptic function and triggering neuronal death [73]. Drugs, like aducanumab, a monoclonal antibody targeting Aβ, aim to reduce amyloid plaques, although the efficacy remains controversial [74]. BACE1 inhibitors (e.g., verubecestat) also target amyloid plaque formation by inhibiting the beta-secretase enzyme [75].
Tau hyperphosphorylation and tangle formation
The τ protein, essential for microtubule stabilization in neurons, becomes hyperphosphorylated in AD, forming neurofibrillary tangles that interfere with axonal transport. This accumulation impairs intracellular transport and cell viability [76]. Investigational sigma-1 receptor agonists, like ANAVEX2-73 (blarcamesine), are being studied for the ability to modify τ phosphorylation [77]. Tau aggregation inhibitors, like TRx0237 (LMTX), target τ pathology although clinical success has been limited [78].
Neuroinflammation
Neuroinflammatory responses, primarily mediators, like activated microglia and astrocytes, are prominent in AD pathology. These cells release cytokines, such as IL-1β and TNF-α, exacerbating Aβ-induced neurotoxicity [79]. Anti-inflammatory agents (e.g., ibuprofen) and immunomodulatory drugs, like sargramostim (leukine, a colony-stimulating factor), are explored for the potential to reduce neuroinflammation, although results vary across clinical trials [80].
Cholinergic deficiency
The resulting cholinergic neuron death from depletion of ACh is a characteristic of AD and causes cognitive impairments [81]. Donepezil, rivastigmine, and galantamine are examples of cholinesterase inhibitors that temporarily improve cognitive and memory abilities in mild-to-moderate stages of AD by inhibiting acetylcholinesterase while preserving ACh levels [82].
Mechanisms in PD
α-Synuclein aggregation and Lewy body formation
The misfolding and accumulation of α-synuclein protein in PD lead to the formation of Lewy bodies in dopaminergic neurons. These inclusions disrupt cellular homeostasis and promote neurotoxicity [83]. Clinical trial therapies that reduce alpha-synuclein accumulation include NPT200-11 and PRX002/RG7935, which are alpha-synuclein aggregation inhibitors and anti-alpha-synuclein monoclonal antibodies, respectively [84, 85].
Dopaminergic neuronal loss in the substantia nigra
The primary cause leading to the pathogenesis of PD involves substantia nigra dopaminergic nerve cell degradation, which leads to dopamine insufficiency [86]. Levodopa, often combined with carbidopa, remains the gold standard for symptomatic treatment by replenishing dopamine levels [87]. The dopamine agonists, ramipexole and ropinirole, are also used to stimulate dopamine receptors directly [88].
Mitochondrial dysfunction and oxidative stress
Mitochondrial complex I dysfunction in PD increases ROS production, which contributes to oxidative stress and neuronal death. Antioxidants, like coenzyme Q10 and N-acetylcysteine, protect against mitochondrial damage, while drugs, like rasagiline (a monoamine oxidase-B inhibitor), provide neuroprotection by reducing dopamine metabolism-induced ROS [89, 90].
Neuroinflammation
Chronic inflammation in PD, which is driven by activated microglia, releases cytokines that exacerbate dopaminergic neuron loss. Inhibitors of microglial activation, such as minocycline (an antibiotic with anti-inflammatory effects), are under investigation [91]. Ibuprofen has been shown to have the potential to reduce neuroinflammation in preclinical PD models, although the clinical efficacy in PD remains uncertain [92].
Mechanisms in HD
Mutant huntingtin (mHTT) aggregation
HD occurs due to the amplification of the huntingtin (HTT) gene of cytosine, adenine, and guanine (CAG) repeats, which results in mutant HTT aggregation. This aggregation disrupts cellular processes, which causes neuronal loss [93, 94]. Branaplam, a small molecule splicing modulator, and tominersen, an antisense oligonucleotide (ASO), are in development to reduce HTT production, targeting the root cause of HD. Branaplam is presently being tested in phase II HD research (NCT05111249) [94, 95].
The clinical trial (NCT02519036) with tominersen involving 46 HD patients was carried out for 28 weeks (including a phase I/II trial) and showed a dramatic reduction in the levels of mutant huntingtin in cerebrospinal fluid, which raised hopes for the medication [96]. However, Roche halted a clinical trial (NCT03761849) in March 2021 with 899 HD disease patients. Data review indicated that the potential benefits of tominersen did not justify the associated risks after 2 years in a phase III trial. The trial evaluated two dosing regimens of tominersen (120 mg administered every 8 weeks and 120 mg every 16 weeks). Patients receiving a higher dosage (120 mg every 8 weeks) had more motor and cognitive deterioration after 69 weeks than patients receiving a placebo. However, patients receiving the lesser dosage (120 mg per 16 weeks) did not exhibit any significant benefits above the placebo group. Additionally, both tominersen treatment groups exhibited significantly increased brain ventricle volumes relative to the placebo group. Although tominersen decreased the synthesis of the mutant huntingtin protein, tominersen also inhibited the creation of healthy wild-type protein, which may have had neurotoxic effects. Shortly after terminating the trial, Wave Life Sciences announced ceasing the formation of two ASOs that were in phase I/II trials for HD [97, 98].
Mitochondrial dysfunction and energy deficiency
mHTT impairs mitochondrial function, resulting in ATP depletion and enhanced ROS production [99]. Therapies, like creatine, enhance mitochondrial function, although clinical trials have yielded mixed results [100]. Coenzyme Q10 has also been investigated for improving mitochondrial bioenergetics in HD, although larger studies are needed [101].
Excitotoxicity and glutamate toxicity
HD is marked by excessive glutamate release, causing excitotoxicity and neuronal death. Memantine, which is an antagonist of NMDA receptors, is used experimentally to block glutamate receptors and protect against excitotoxic damage [102]. Riluzole, another glutamate antagonist, has shown neuroprotective effects in HD models [103].
Dysregulated apoptosis and cell death
Increased pro-apoptotic signalling, partly due to mHTT toxicity, results in neuronal apoptosis. Drugs, like minocycline, show potential in inhibiting caspase activity and reducing apoptosis in HD models. Minocycline exhibits neuroprotective effects by suppressing hypoxia-induced activation of rodent microglia, inhibiting caspase-1 and -3 expression in an HD transgenic mouse model, and facilitating mitochondrial permeability-transition-mediated cytochrome c release from mitochondria in an ALS animal model [104]. Additionally, pifithrin-α, an experimental drug targeting p53-mediated apoptosis, has been explored for neuroprotective effects in HD [105].
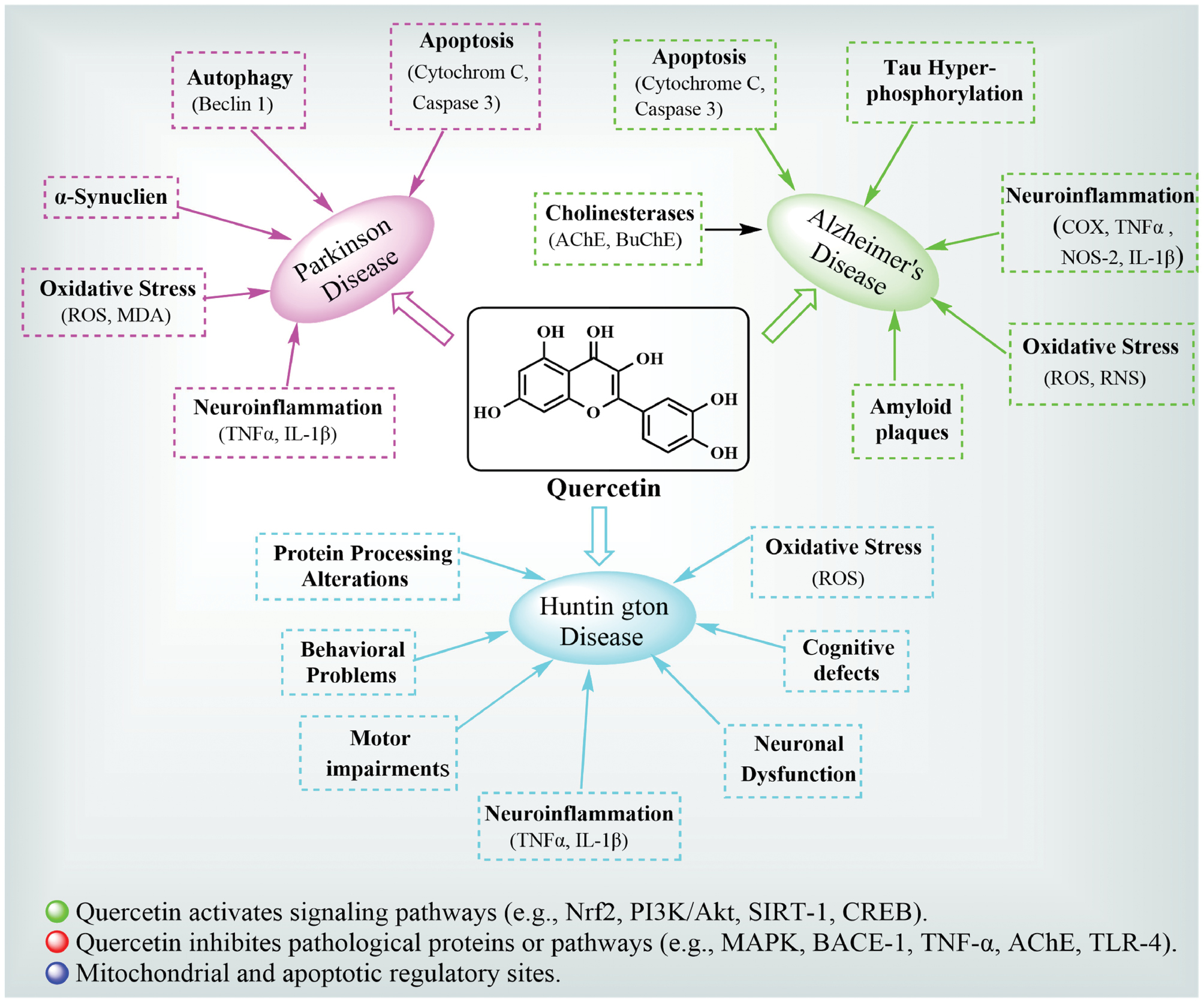
Effectiveness of quercetin in NDDs
Quercetin in AD
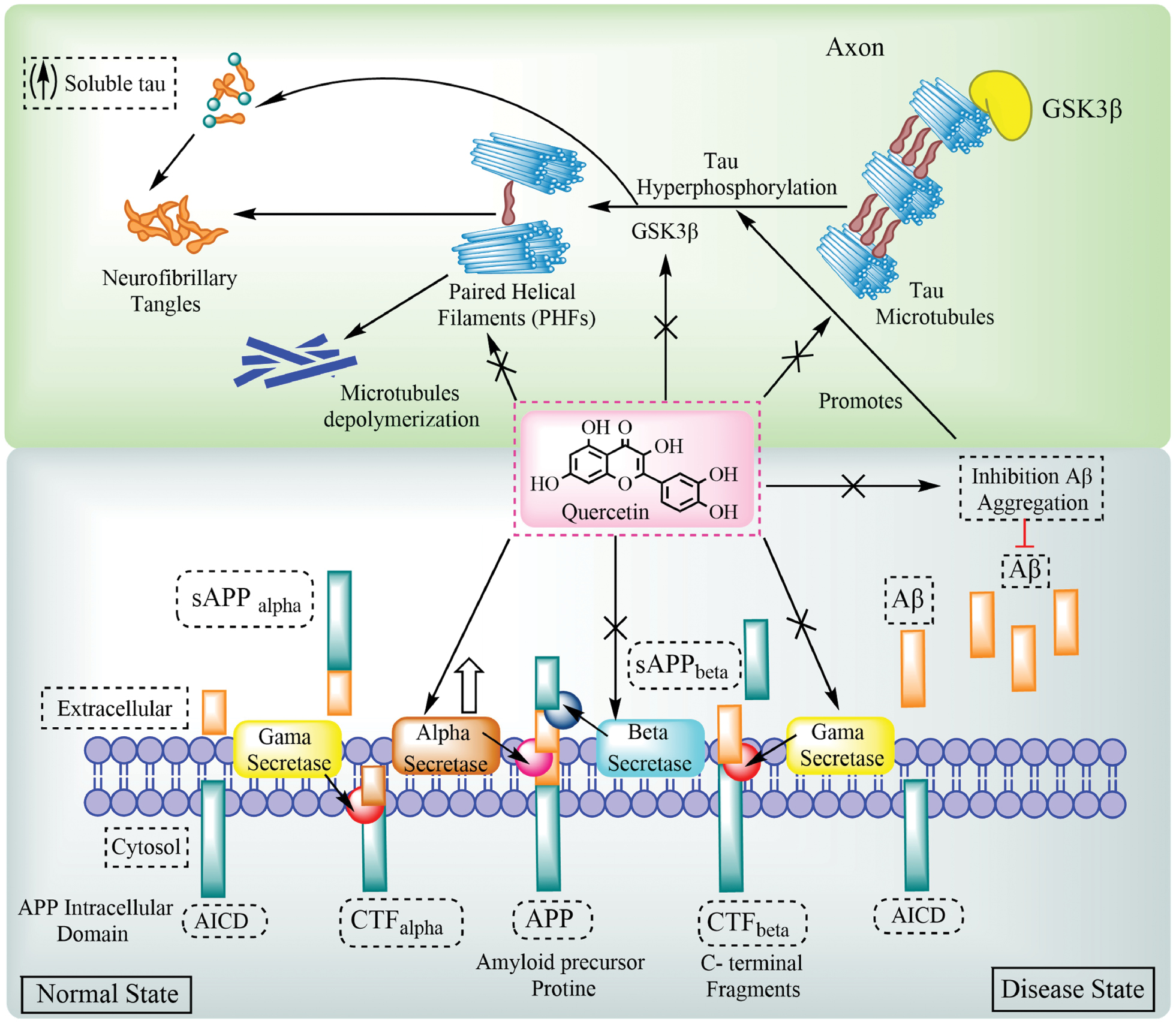
Quercetin exhibits profound neuroprotective effects in AD primarily through the capacity to counteract Aβ aggregation, τ hyperphosphorylation, and neuroinflammation [6, 57]. Research has shown that quercetin reduces amyloidogenic processing of the APP by inhibiting the activities of β- and γ-secretase, two important enzymes that cause Aβ plaque formation. Additionally, quercetin modulates τ protein, decreasing hyperphosphorylation and preventing neurofibrillary tangle formation, which are crucial pathologic hallmarks of AD. The strong antioxidant qualities of quercetin are achieved by activation of the Nrf2-ARE pathway, which increases the expression of endogenous antioxidant enzymes, like GSH peroxidase and SOD. Additionally, quercetin inhibits the NF-κB signaling pathway, which lowers the production of pro-inflammatory cytokines, like IL-6, TNF-α, and IL-1β, which attenuate neuroinflammatory cascades [106, 107]. Quercetin has been shown to improve cognitive deficits and memory function in aged triple-transgenic AD experimental rats by reversing extracellular amyloidosis, astrogliosis, and microgliosis. The ability of quercetin to inhibit AChE also elevates ACh levels, restoring synaptic function and cognitive abilities [108]. Structural modifications and delivery enhancements, such as nanoparticle-based formulations, further enhance the bioavailability and effectiveness against AD, paving the way for advanced clinical applications. Figure 3 depicts an integrated visual representation of the neuroprotective mechanisms of quercetin in AD. Quercetin reduces Aβ accumulation by inhibiting β- and γ-secretase activity and interferes with the hyperphosphorylation of τ proteins through GSK-3β inhibition. Quercetin enhances antioxidant defenses by activating the Nrf2-ARE pathway and increasing endogenous antioxidant enzyme levels, such as SOD, CAT, and GSH. The diagram also shows the role of quercetin in suppressing inflammatory cascades via NF-κB inhibition and downregulation of cytokines, like TNF-α, IL-6, and IL-1β. Moreover, quercetin boosts cholinergic neurotransmission by inhibiting AChE, thereby increasing ACh levels in the synaptic cleft [109] (Figure 3; Table 2).
Figure 3 Mechanism and role of quercetin in the pathogenesis of Alzheimer’s disease.
Table 2 Biological Activities of Quercetin and the Neuroprotective Outcomes, as Reported in AD
| Serial No. | Biological Activities of Quercetin | Outcomes (Effect Produced) | References |
|---|---|---|---|
| 1. | Antioxidant, anti-inflammatory | Decrease taupathy, microgliosis, and astrogliosis | [234] |
| 2. | Antioxidant | Preventing tau protein hyperphosphorylation through PI3K/Akt/GSK3β signalling pathways and MAPKs | [153] |
| Decrease amyloid precursor protein (APP) maturation, altering Aβ production and accumulation | [3] | ||
| Decrease the AChE level | [3] | ||
| 3. | Anti-inflammatory | Reduce the cytotoxicity and apoptosis caused by Aβ(1-42) | [6] |
| Suppress the expression of the iNOS gene in microglia | [3] | ||
| Lowering inflammatory cytokines in activated macrophages, such as TNF-α, IFN-γ, interleukin (IL)-1β, IL-6, IL-12, and COX-2 | [235] | ||
| 4. | Inhibiting neuronal apoptosis | Improving calcium homeostasis, growth factor signalling, and neuroplasticity | [236] |
| 5. | Antioxidant, Attenuating oxidative stress | Ameliorate cellular oxidative stress and inhibit the production of nitric oxide (NO) | [237] |
| Modulating cell antioxidant pathways, like Nrf-2-ARE and paraoxonase 2 (PON2) pathways | [11] |
Quercetin in PD
A neurodegenerative disease linked to aging (PD) is typified by the death of dopaminergic neurons in the substantia nigra pars compacta (SNpc). Clinical signs and symptoms include stiffness, tremors, and bradykinesia [110]. According to Grewal AK et al., isoquercetin prevents α-synuclein fibrillization in PD and enhanced motor function [3]. Quercetin in PD targets multiple pathogenic mechanisms, including oxidative stress, mitochondrial dysfunction, α-synuclein aggregation, and neuroinflammation. Through the activation of the Nrf2 pathway, quercetin improves cellular antioxidant defenses, reducing oxidative damage in substantia nigra dopaminergic neurons. Quercetin even stabilizes mitochondrial membranes and improves mitochondrial bioenergetics, which is crucial for neuronal survival. Quercetin inhibits alpha-synuclein aggregation and the synthesis of Lewy bodies, which are toxic to neuronal cells. Furthermore, quercetin reduces neuroinflammatory responses by suppressing microglial activation and decreasing levels of pro-inflammatory mediators. Quercetin preserved dopamine levels, decreased lipid peroxidation, and restored the amount of GSH in the striatum, exhibiting neuroprotective effects in animal studies. Quercetin also ameliorates motor deficits and behavioral impairments in 6-OHDA-induced PD models [2, 111]. Isoquercetin, a glycoside of quercetin, has shown improved bioavailability and efficacy in PD by reducing α-synuclein fibrillization and enhancing motor performance. These results highlight the promise of quercetin as a PD treatment candidate. The practical applicability of quercetin is further enhanced by sophisticated delivery methods, such as liposomal encapsulation. The neuroprotective benefits of quercetin were investigated by Zhang et al. in PC12 cells and zebrafish. Sehar et al. and Zhang et al. reported that quercetin suppresses pro-inflammatory gene upregulation (IL-1s, COX-2, and TNF-α) in zebrafish while inhibiting excessive NO generation and iNOS overexpression in PC12 cells (Figure 4; Table 3) [112, 113].
Figure 4 The mechanism and function of quercetin in the pathophysiology of brain ischemia, Parkinson’s disease, and Huntington’s disease.
Table 3 Biological Activities of Quercetin and Neuroprotective Outcomes, as Reported in Parkinson’s Disease
| Serial No. | Outcomes (Produced Effects) | Biological Actions of Quercetin | References |
|---|---|---|---|
| 1. | Inhibit mitochondrial activity | Reduce the rate of dopaminergic degeneration | [238] |
| 2. | Mitochondrial function | Increase mitophagy | [239] |
| 3. | Antioxidant | Improve cognitive impairment | [240] |
| Increase in striatal dopamine concentrations | [241] | ||
| Reduce oxidative stress and induce autophagy. | [242] | ||
| 4. | Anti-inflammatory | Increase in production of NO and overexpression of iONS | [243] |
| Reduced elevated expression of the p53, TNF-α, IκKB, NF-κB, and IL-1β genes | [244] | ||
| 5. | Reduces cellular apoptosis | Attenuates nuclear condensation | [245] |
Figure 4 illustrates the multifaceted roles of quercetin in mitigating the pathogenesis of PD, brain ischemia, and HD. Quercetin not only inhibits α-synuclein aggregation and preserves dopaminergic neurons in PD but also exerts significant anti-ischemic and mitochondrial-protective actions. Figure 4 highlights how quercetin improves mitochondrial dynamics by enhancing mitophagy and reducing mitochondrial-derived ROS. Quercetin also modulates neuroinflammation by suppressing microglial activation and the expression of pro-inflammatory mediators, such as TNF-α and iNOS. Furthermore, the ability of quercetin to stabilize the BBB and protect neuronal integrity under ischemic conditions underscores the vasculoneuroprotective potential, which is particularly relevant in PD, in which vascular dysfunction exacerbates neurodegeneration [114].
Quercetin and HD
Quercetin targets the primary pathogenic mechanisms of neuroinflammation, oxidative stress, excitotoxicity, and mitochondrial dysfunction in HD. Quercetin restores ATP synthesis and lessens oxidative damage by preventing mitochondrial swelling and lipid peroxidation [115]. Quercetin reduces oxidative stress in neuronal cells by boosting the activity of antioxidant enzymes, including CAT and SOD. The neuroprotective effects of quercetin extend to reducing excitotoxicity by modulating NMDA receptor activity and restoring calcium homeostasis [116]. Quercetin supplementation improves motor coordination, reduced neurobehavioral deficits, and diminished astrogliosis in the striatum using animal models. Histopathologic studies have revealed reduced neuronal apoptosis and pyknotic nuclei in HD models treated with quercetin. Quercetin also attenuates quinolinic acid-induced neurotoxicity by decreasing TNF-α levels and preserving neurotransmitter levels, including dopamine and serotonin. Advanced formulations, such as quercetin-loaded nanoparticles, have been developed to enhance brain bioavailability, further validating the therapeutic potential in HD. The ability of quercetin to modulate gene expression and signalling pathways implicated in HD highlights the promise of quercetin for disease modification and symptom management [117–119]. Kuhad and colleagues conducted another study to assess the protective properties of quercetin against quinolinic acid (QA)-induced neurotoxicity. QA exposure decreased dopamine, serotonin, and norepinephrine levels in the rat forebrain and markedly increased TNF-α levels, which are indicative of neuroinflammatory injury. Due to the strong anti-inflammatory qualities of quercetin, these behavioral, biochemical, and neurochemical abnormalities were successfully reduced. Table 4 describes the biological activities of Quercetin and neuroprotective outcome as reported in Huntington disease.
Table 4 Biological Activities of Quercetin and Neuroprotective Outcomes, as Reported in Huntington’s Disease
| Serial No. | Outcomes (Effect Formed) | Biological Actions of Quercetin | References |
|---|---|---|---|
| 1. | Antioxidant | Within the mitochondria, quercetin increases the total thiol level in 3-3-nitropropionic acid (NP)-treated animals | [246] |
| Lower lipid peroxidation in mitochondria also inhibits the formation of malondialdehyde (MDA) inside the brain | [246] | ||
| Diminish astrogliosis and pyknotic nuclei | [246] | ||
| Enhancing MnSOD levels to decrease mitochondrial oxidative stress | [247] | ||
| Reduces the anxiety, poor motor coordination, and gait despair brought on by 3-NP | [248] | ||
| 2. | Anti-inflammatory | Decreased microglial proliferation | [249] |
| Reduces changes in behavior, biochemistry, and neurochemistry | [250] |
Figure 5 provides a holistic overview of the neuroprotective role of quercetin in AD, PD, and HD by highlighting the shared pathologic events. The mutant huntingtin protein in HD promotes abnormal protein aggregation, which leads to mitochondrial dysfunction and increased oxidative stress through excessive ROS production. This cascade results in cytochrome C release, caspase activation, apoptosis, and neuroinflammation. Figure 5 emphasizes the role of quercetin in intercepting this pathologic progression at multiple stages, as follows: quercetin neutralizes ROS; stabilizes mitochondrial membranes; inhibits cytochrome C leakage; suppresses apoptosis by downregulating caspase signalling; and reduces neuroinflammation. These protective mechanisms align with preclinical findings; specifically, quercetin preserved mitochondrial integrity, lowered TNF-α levels, and improved motor coordination in HD models. Quercetin offers disease-modifying potential across a spectrum of NDDs by acting on these convergent targets, reinforcing the relevance as a multi-targeted therapeutic candidate [120, 121].
Figure 5 Neuroprotective properties of quercetin in a spectrum of neurodegenerative diseases. Quercetin directly protects neurons through a range of molecular neuroprotective mechanisms in these diseases, such as the downstream control of oxidative stress, neuroinflammation, apoptosis, and cell death.
Role of quercetin in vascular endothelial function and neurovascular protection
Clinical application of quercetin in Russia: Capilar formulation
Quercetin is available in Russia under the commercial name, Capilar, which contains 10 mg of quercetin per tablet and is primarily used among the aging population for improving capillary function and preventing venous insufficiency. Capilar is marketed as a vasoprotective supplement intended to improve microcirculation and support cardiovascular and peripheral vascular health. The long-standing clinical use of quercetin in this formulation underscores the systemic protective effects, including potential benefits on cerebral microvasculature integrity, which is closely linked to neuroprotection in elderly individuals [122].
Neurovascular unit and BBB integrity
The neurovascular unit (NVU), which is comprised of endothelial cells, astrocytes, neurons, and pericytes, is essential for maintaining brain homeostasis [123]. Quercetin has been shown to enhance the integrity of the NVU by reducing oxidative stress and inflammation in endothelial cells, preserving tight junction proteins, such as claudin-5 and occludin. These effects mitigate BBB disruption, a common pathologic feature in NDDs, such as AD and vascular dementia [124, 125].
Antioxidant effects on cerebral microcirculation
The antioxidant actions of quercetin reduce oxidative damage to endothelial cells and improve NO bioavailability. This effect promotes vasodilation, reduces leukocyte adhesion, and enhances cerebral perfusion [126]. Quercetin administration restores cerebrovascular tone and reduces infarct volume in rodent models of ischemia-reperfusion injury, indicating that the vascular protective role of quercetin extends to cerebral blood flow regulation and ischemic neuroprotection [127].
Anti-inflammatory modulation of endothelial dysfunction
Endothelial dysfunction is a key contributor to chronic neuroinflammation. Quercetin exerts anti-inflammatory effects by inhibiting endothelial NF-κB activation, which reduces the expression of vascular adhesion molecules (e.g., ICAM-1 and VCAM-1) and limits immune cell infiltration into neural tissues. Such modulation reduces the initiation and propagation of neuroinflammatory cascades, further supporting cognitive health and neuronal survival [128, 129].
Evidence from clinical and nutraceutical use
The success of Capilar in clinical practice in Russia offers indirect yet valuable insight into the tolerability and utility of quercetin for vascular-related disorders in aging populations [122]. Although primarily used for peripheral circulation, the systemic effects of quercetin suggest a plausible benefit in supporting cerebral perfusion and preventing age-associated cognitive decline. These observations advocate for further clinical studies targeting the role of quercetin in maintaining neurovascular health and delaying neurodegeneration [130].
Synergistic neurovascular and neuronal mechanisms
Dual modulation of vascular and neuronal pathways by quercetin is through the PI3K/Akt, Nrf2-ARE, and MAPK cascades and facilitates cross-talk between endothelial and neural cells [125]. This integrated mechanism protects against ischemia-induced neuronal apoptosis and may explain the cognitive benefits observed in aging populations consuming quercetin-rich supplements [130].
Comparative neuroprotective efficacy of quercetin and other flavonoids
Quercetin exhibits multi-targeted neuroprotective actions and is superior to many flavonoids due to the SAR involving catechol and hydroxyl groups in the B and C rings. These functional groups enable quercetin to scavenge ROS, chelate metal ions, and inhibit protein aggregation. Quercetin has higher potency in modulating Nrf2-ARE and PI3K/Akt pathways compared to kaempferol and rutin but is limited by poor bioavailability. EGCG, though potent in inhibiting aggregation, lacks metabolic stability in vivo. Luteolin and baicalein show strong anti-inflammatory and antioxidant actions but are not as well-characterized across multiple models as quercetin. The integration of quercetin signalling and mitochondrial and epigenetic regulation mechanisms makes quercetin a versatile candidate in precision neurotherapeutics. Table 5 describes the comparative analysis of quercetin neuroprotective efficacy with other flavonoids.
Table 5 Comparative Evaluation of Quercetin with other Neuroprotective Flavonoids
| Flavonoid | Disease Model | Mechanism | Unique Advantage | Limitation | Reference |
|---|---|---|---|---|---|
| Quercetin | AD, PD, HD, ischemia | Nrf2-ARE, PI3K/Akt, MAPK, HIF-PHD, and PON2 | Strong multi-target action (oxidative stress, inflammation, apoptosis, metal chelation, and mitochondrial support) | Poor oral bioavailability, rapid metabolism | [251] |
| Kaempferol | AD, PD | Anti-inflammatory (NF-κB), antioxidant | Moderate AChE inhibition and improved cognitive function | Lower ROS scavenging than quercetin | [252, 253] |
| Luteolin | AD, ALS | Inhibition of NF-κB, MAPK, and microglial activation | High BBB permeability and neurogenesis induction | Limited clinical validation | [254] |
| EGCG (Epigallocatechin gallate) | AD, PD | α-synuclein and Aβ aggregation inhibition, mitochondrial protection | Potent metal chelator and proteostasis modulator | Instability at physiologic pH, GI degradation | [255, 256] |
| Rutin (quercetin-3-rutinoside) | AD, PD | Antioxidative, mild anti-inflammatory, AChE inhibition | Better water solubility, glycosylation improves absorption | Lower receptor binding affinity vs. quercetin aglycone | [257] |
| Baicalein | PD | Dopamine preservation, ROS inhibition, GSK-3β modulation | Potent mitochondrial protector, strong ROS inhibition | Less studied in human trials | [258, 259] |
Targeting signalling pathways by quercetin for neuroprotection
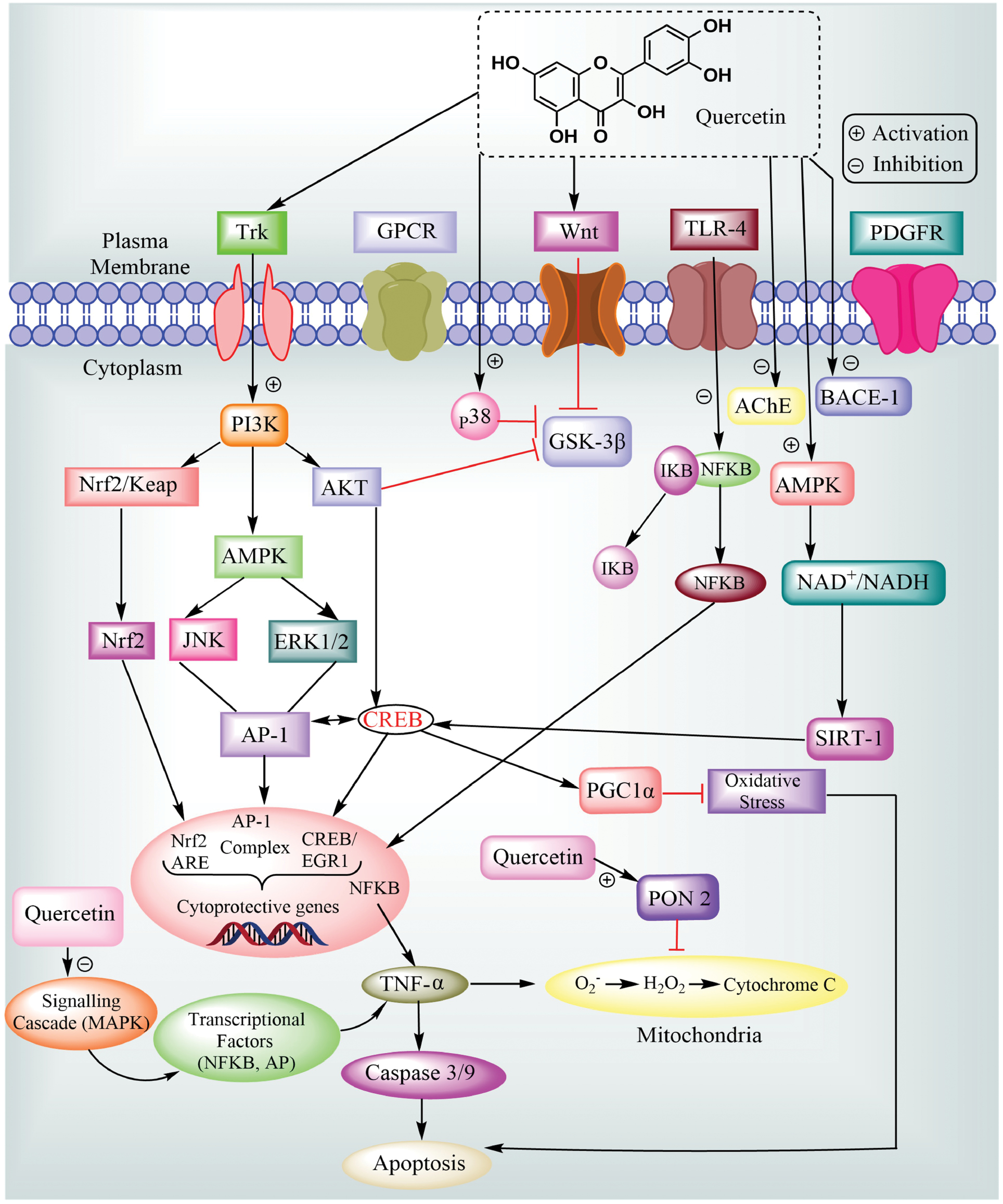
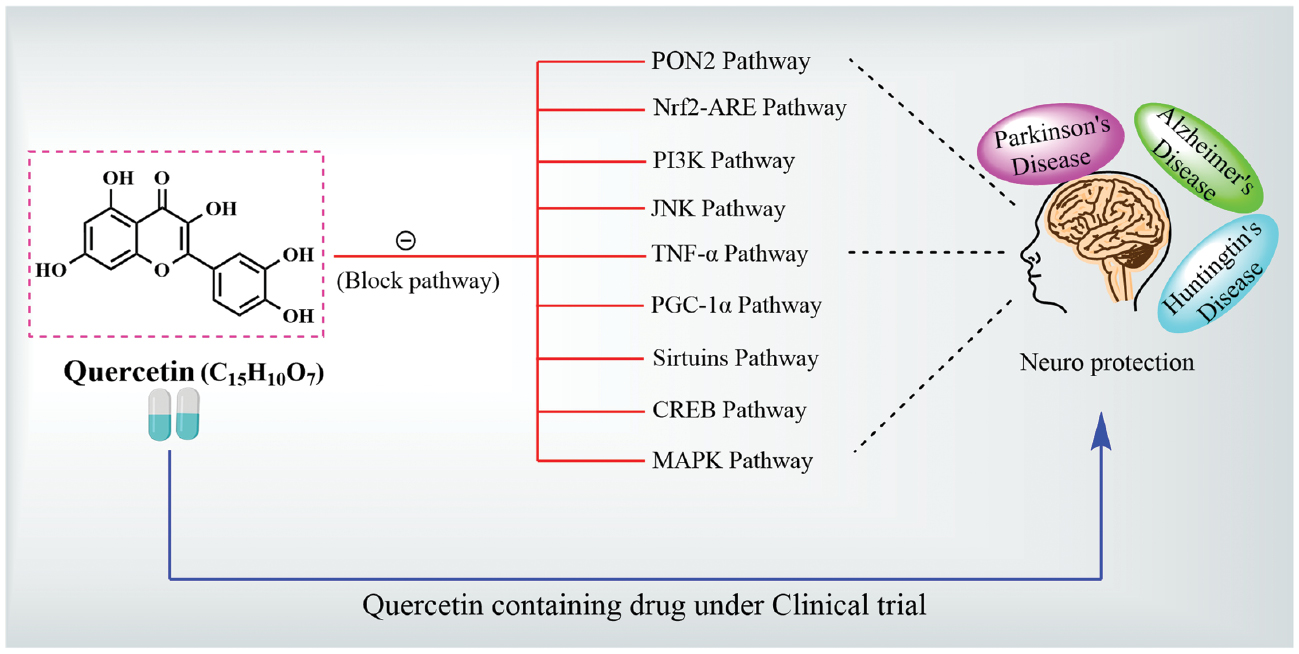
Quercetin targets several critical signalling pathways that are included in the mechanism underlying NDDs, including AD and PD. Neurodegeneration is identified by complex cascades of oxidative damage, inflammatory responses, and apoptotic mechanisms, which progressively contribute to neuronal dysfunction and loss. This review systematically examined the downstream signalling pathways modulated by quercetin, including the PON2, Nrf2-ARE, and PI3K pathways, JNK mechanism, TNF-α formation and inhibition, PGC-1α, sirtuin, CREB, and MAPK pathways, by elucidating the roles in mitigating oxidative stress, reducing neuroinflammation, and enhancing cell survival. Quercetin is a promising therapeutic approach for slowing neurodegenerative processes and improving neuronal resilience by targeting these key mechanisms.
PON2 pathway
The eldest of the PON family consists of 3 genes (PON1, PON2, and PON3) close to the longer branch of human chromosome 7q21-22. PON2 is found in macrophages and endothelial cells and serves as an antioxidant in humans [131]. PON2 is mostly found in mitochondria based on subcellular research, where PON2 is vital in reducing oxidative stress brought on by free radicals. The location of PON2 in the mitochondria is crucial for protecting cells from oxidative damage and a lack of PON2 results in mitochondrial dysfunction [132]. It has been demonstrated that PON2 selectively reduces the release of superoxide from the inner mitochondrial membrane in human endothelial cells, while not affecting other reactive species, like hydrogen peroxide (H2O2) or peroxynitrite [133]. Unlike PON1, which primarily associates with HDL and circulates in the bloodstream, PON2 functions exclusively as an intracellular antioxidant [134].
PON2 mRNA is detected in both mouse and human brains, while PON2 protein is found in mouse, rat, human, and monkey brains, with the highest concentrations observed in dopaminergic regions of the mouse brain [135]. There is a gender-based differential in PON2 levels with female mice showing higher levels than humans and other rodent and non-rodent species. The PON2 antioxidant qualities are essential for atherosclerosis prevention and neuroprotection. PON2-deficient murine cells are much more cytotoxic when exposed to oxidants, such as H2O2 and 2,3-dimethoxy-1,4-naphthoquinone (DMNQ) [136]. Similarly, compared to female cells, male mouse neurons and striatal astrocytes are far more susceptible to oxidative stress and cytotoxicity driven by these oxidants because male mouse neurons and striatal astrocytes express lower levels of PON2 [137]. PON2-deficient mice also show elevated lipid hydroperoxide levels in tissues and increased macrophage migration into arterial walls compared to wild-type controls [138].
Although quercetin has been shown to increase PON1 expression, it is still unknown what precise biochemical mechanisms flavonoids use to affect PON2 gene expression [131]. It has been demonstrated that pomegranate flavonoids modify the DNA-binding properties of the transcription factor, AP-1, which is found in the promoter region of the PON2 gene and may regulate expression [139]. Superoxide anion radicals are produced by the quercetin molecular target, NADPH oxidase, which controls AP-1 DNA binding [140]. Additionally, new research suggests that glucocorticoid-glucocorticoid receptor complexes directly mediate PON2 gene transcriptional activation, which may have an impact on AP-1 transactivation. According to in vitro research, quercetin treatment increases the expression of the PON2 protein in striatal astrocytes, macrophages, and mouse neurons. It is hypothesized that quercetin induces a mild oxidative stress response, which in turn increases PON2 expression. Additionally, in striatal astrocytes, quercetin eliminates ROS levels and mitigates cytotoxicity driven by H2O2 and DMNQ (Figure 6) [139, 141].
Figure 6 Quercetin modulates molecular processes involved in neuronal survival. Quercetin eventually caused the cytoprotective genes to be activated by activating the PI3K-Akt, AMPK, Wnt, p38, PON-2, and related signalling pathways, including Nrf2, MAPK, CREB, and SIRT-1, which promotes neuronal survival. However, for neuroprotective action, quercetin inhibits TLR-4, BACE-1, AChE, TNF-α, and the MAPK signalling cascade.
Nrf2-ARE pathway
Nuclear factor erythroid 2-related factor 2 (Nrf2) is a transcription factor that is essential for controlling oxidative stress brought on by free radicals. Kelch-like ECH-associated protein 1 (KEAP-1) anchors Nrf2 in the cytoplasm under physiologic settings through Cullin 3-based E3 ligase, facilitating ubiquitination and subsequent proteasomal destruction [142]. Antioxidant response element (Nrf2-ARE) signalling pathway activation offers neuroprotection against oxidative stress and cellular death. According to recent studies, the NRF2-ARE pathway also controls the development of misfolded protein aggregates, which are frequently seen in NDDs, like AD, PD, and HD. Moreover, γ-glutamyl-cysteine synthetase (GCS), an essential enzyme in the production of the antioxidant, GSH, is stimulated to appear when Nrf2-ARE is activated [143].
Quercetin has been shown to activate the Nrf2-ARE pathway, thereby reducing cellular damage caused by oxidative stress. Studies using tert-butylhydroquinone, a well-known Nrf2 inducer, have highlighted the neuroprotective benefits of Nrf2-ARE activation against neurotoxicity, particularly the benefits brought on by Aβ [144]. Similar protective effects have been observed with dihydroquercetin [145]. Additionally, nutraceutical compounds, such as kaempferol and pterostilbene, exhibit synergistic effects when combined with quercetin. According to Ayyalasomayajula et al. and Cheng et al., quercetin is a neuro-hormetic phytochemical that activates Nrf2 through the ERK and JNK pathway [146, 147]. Several in vitro investigations on cerebral cortex tissues and neuronal cell lines have demonstrated that quercetin improves cellular resistance to oxidative damage brought on by neurotoxic agents, like Aβ peptide or oxidants, like hydroperoxide (Figure 6)
Phosphoinositide 3-kinase (PI3K) pathway
Age-associated NDDs and other severe brain disorders are significantly influenced by PI3K. Akt, a crucial enzyme that controls neuronal survival and plasticity, regulates the PI3K/Akt pathway. Phosphatidylinositol 3,4,5-trisphosphate (PIP3) can be activated by a variety of stimuli, including integrins, receptor tyrosine kinases (RTK), cytokines, B and T cell receptors, and G-protein coupled receptors (GPCRs). This activation leads to Akt phosphorylation, which ultimately influences the activation or inhibition of several target proteins, hence regulating important physiologic processes, such as the cell cycle, development, metabolism, growth, protein synthesis, and apoptosis [148, 149].
Animal models of NDDs have shown that a high-fat diet (HFD) impairs learning and memory, primarily due to oxidative stress. Such diets are rich in saturated fats and involve carbonyls and ROS and decrease hippocampal gene expression, especially of PI3K/Akt and Nrf2 pathways [150]. Quercetin supplementation in conjunction with HFD enhances antioxidant capacity and mitigates learning and memory deficits. Quercetin administration dramatically decreased oxidative stress (i.e., increased levels of cytochrome c, GSH, GSH peroxidase, and glutaredoxin, as well as decreased lipid peroxidation in the cortex and striatum), while fostering anti-apoptotic and antioxidant pathways in ischemic rat brains [18, 151].
The PI3K/Akt blocker, LY294002, elevated the Bax:Bcl-2 ratio and highlighted the pathway participation in the antioxidant actions of quercetin and exercise by negating the combined effects [152]. Additionally, quercetin demonstrated neuroprotective benefits in NDDs, such as AD and PD, by inhibiting oxidative stress-induced τ protein phosphorylation, a crucial downstream target of this pathway. Nearly the same outcomes were shown in PC12 pheochromocytoma cells, in which quercetin restored GSH and SOD function while lowering lactate dehydrogenase, ROS, and malondialdehyde levels to prevent H2O2-induced death [153] (Figure 6).
c-Jun N-terminal kinase (JNK) pathway
The JNK pathway plays a vital part as a key signal in the MAPK downstream signalling pathway. This kinase is a member of the threonine protein kinase family, which is encoded by three different genes (JNK1, JNK2, and JNK3) that together result in 10 variations [154]. While JNK1 and JNK2 are widely distributed throughout many tissues and play a major role in obesity-induced insulin resistance, JNK3 is mostly found in the central nervous system (CNS) and may have therapeutic uses in NDDs and other disorders involving the CNS [155]. Threonine and tyrosine residue phosphorylation controls JNK activation, whereas MAPK phosphatases drive a negative feedback process that deactivates JNK [156].
In a mouse model of CaCl2-induced abdominal aortic aneurysm, quercetin was reported to decrease the production of ROS by inhibiting activation of the transcription factor, AP-1, and downregulating the expression of JNK and the phosphorylated version [157]. Additionally, quercetin mitigates H2O2-induced apoptosis in mesangial cells by modulating the AP-1 pathway. H2O2-induced rapid phosphorylation of ERK and JNK kinases is also reduced with quercetin pre-treatment. Additionally, it has been shown that JNK and the JNK substrate, c-Jun, mediate apoptosis in a variety of cell types [157, 158]. Quercetin prevents RL34 cell JNK activation when 4-hydroxy-2-nonenal, a byproduct of lipid peroxidation, is present [159]. The inhibitory effect of quercetin in stress-related signalling pathways, particularly involving the enzyme PKC, was suggested as the underlying mechanism in HNE-induced pathways. These results show that the JNK signalling pathway, which has been shown in macrophages, mediates the anti-inflammatory and protective effects of quercetin (Figure 6) [160].
TNF-α pathway
TNF is an inflammatory cytokine released by macrophages and monocytes during acute inflammation. TNF sets off several signalling cascades within cells that lead to apoptosis and necrosis. TNF-α and TNF-β are the two forms of TNF. The anti-inflammatory qualities of quercetin have been demonstrated by several in vitro and in vivo studies [161]. For example, by blocking the TNF-α pathway, quercetin reduces inflammation brought on by 6-hydroxydopamine (6-OHDA) toxicity in PC12 cells and zebrafish models. Additionally, by reducing the LPS-induced synthesis of TNF and IL-1 mRNA in glial cells and astrocytes, quercetin reduces microglial-mediated neuronal cell death in co-cultures of microglia and PC12 cells [162]. Through TLR4 regulation and TNF-α pathway suppression, quercetin-loaded nanoparticles had notable anti-inflammatory effects by reducing the stress caused by oxysterol in human neuroblastoma cells, SH-SY5Y [163]. Additionally, microglial cells are crucial in starting the inflammatory cascade-mediated neuronal death by producing inflammatory and neurotoxic mediators, like TNF-α, IL-1, IL-6, and NO [164]. In studies using astrocyte cells, quercetin reduced the number of apoptotic cells in rat glioma cells (C6 cells) by decreasing the formation of ROS and shielding the cells from toxicity from tertiary-butyl hydroperoxide and H2O2 [165]. Additionally, quercetin increased the survival of rat oligodendrocytes (OLN-93), which are essential for the health of neurons [166]. The anti-inflammatory properties are probably due to the capacity to suppress TNF-α expression by modulating NF-κB (Figure 6).
Peroxisome proliferator-activated receptor gamma co-activator 1-alpha (PGC-1α) pathway
PGC-1α has a critical role in the CNS by activating various nuclear receptors and influencing their functions. Research has shown that nuclear PGC-1α levels rise significantly after traumatic brain injury (TBI), particularly in the cortical neurons, suggesting involvement in post-TBI neurobiologic changes. A study investigated the neuroprotective properties of quercetin and the effect on PGC-1α signalling pathways [113]. Mice administered quercetin showed less brain water content and less neuronal degeneration in processed brain tissues, which suggested that quercetin reduces cerebral edema after TBI. Following quercetin administration, alterations in downstream PGC-1α signalling pathways verified the pathway significance in preventing cerebral cortex mitochondrial dysfunction [167]. Furthermore, in the AD model mouse, learning and memory were improved by upregulating PGC-1α and SIRT1 levels. PGC-1α and SIRT1 deacetylation and activation promoted mitochondrial biogenesis and reduced AD mitochondrial dysfunction [168]. Furthermore, suppression of BACE1, a crucial transcriptional regulator, is linked to SIRT1-PGC-1α activation (Figure 6) [169].
Sirtuins pathway
Sirtuins, particularly SIRT1, perform a pivotal role in cellular stress resistance and longevity. Quercetin activation of SIRT1 results in deacetylation of transcription factors involved in anti-apoptotic and anti-inflammatory responses. Through SIRT1 activation, quercetin enhances mitochondrial health, supporting cellular resilience against neurodegenerative stressors, hence supporting the preservation of neuronal integrity and function (Figure 6) [170].
CREB pathway
CREB, a key regulator of neuronal plasticity and memory formation, is activated by quercetin and promotes the expression of neuroprotective genes. Quercetin-induced CREB activation fosters neurogenesis, cognitive enhancement, and long-term survival of neurons. This pathway is particularly relevant in memory preservation and learning, providing therapeutic insights for conditions, like AD (Figure 6) [171].
MAPK pathway
The MAPK pathway, which includes ERK, JNK, and p38, governs cellular responses to oxidative and inflammatory stress. Quercetin modulates MAPK signalling by downregulating p38 and JNK, thereby decreasing pro-inflammatory responses and reducing apoptosis. This pathway modulation by quercetin promotes neuronal survival and homeostasis, making downregulation of p38 and JNK a pivotal aspect of neuroprotective action (Figure 6) [172].
Crosstalk between quercetin-regulated signalling pathways in neuroprotection
PI3K/Akt and Nrf2-ARE pathway interaction
Quercetin activation of the PI3K/Akt pathway promotes neuronal survival and stimulates Nrf2 nuclear translocation, which enhances the expression of downstream antioxidant enzymes, such as HO-1 and SOD. Akt-mediated phosphorylation of Nrf2 prevents degradation by Keap1, thereby facilitating oxidative defense mechanisms. This crosstalk is crucial for amplifying neuroprotective responses under oxidative stress [173].
MAPK, JNK, and Nrf2 crosstalk
MAPK family members, such as ERK and p38, can modulate Nrf2 phosphorylation, affecting nuclear localization. Quercetin inhibition of JNK and p38 pathways reduces inflammation and synergizes with Nrf2-driven transcription of antioxidant genes. Thus, suppression of stress-activated MAPKs enhances the efficiency of Nrf2 signalling [174].
Sirtuin and PGC-1α axis integration
SIRT1, activated by quercetin, deacetylates PGC-1α, which enhances mitochondrial biogenesis and energy metabolism. This axis also enhances PI3K/Akt signalling, suggesting triangular feedback that integrates energy homeostasis, anti-apoptotic mechanisms, and oxidative stress regulation [175].
NF-κB suppression and anti-inflammatory feedback
PI3K/Akt activation inhibits NF-κB translocation via phosphorylation of IκBα, while quercetin direct inhibition of MAPK-JNK and TNF-α further suppresses inflammatory cytokine release. This orchestrated inhibition across pathways diminishes neuroinflammation in diseases, like AD and PD [176, 177].
PON2 and mitochondrial signalling coupling
PON2 enhances mitochondrial stability and mitigates superoxide generation. PON2 expression, increased by quercetin, may be regulated through indirect modulation of JNK and Akt pathways, as these govern mitochondrial membrane potential and apoptotic thresholds [178].
CREB and PI3K/Akt synergy in neurogenesis
The PI3K/Akt-CREB pathway supports BDNF expression and synaptic plasticity. Quercetin enhances CREB phosphorylation downstream of Akt activation, promoting memory consolidation and neuronal differentiation, key elements inhibit neurodegeneration [179]. The integration of these signalling cascades enables a multi-targeted effect, in which quercetin orchestrates a finely tuned balance between antioxidant defense, inflammation control, mitochondrial stabilization, and neuronal repair. Understanding this interplay is crucial for optimizing quercetin-based therapies in complex NDDs.
Quercetin as a direct inhibitor of hypoxia-inducible factor prolyl hydroxylase (HIF-PHD): an anti-ischemic mechanism
HIF prolyl hydroxylase enzyme: a target for neuroprotection
Prolyl hydroxylases (PHD1, PHD2, and PHD3) function as oxygen sensors and catalyze the hydroxylation of HIF-1α at proline residues, marking prolyl hydroxylases for ubiquitin-mediated proteasomal degradation. Inhibition of HIF-PHDs under hypoxia allows HIF-1α accumulation and translocation to the nucleus, initiating transcription of protective genes, like VEGF, which collectively confer neurovascular repair and metabolic adaptation [48, 180].
Evidence supporting the anti-ischemic effects of quercetin via HIF stabilization
Several preclinical studies have confirmed that quercetin mimics hypoxic preconditioning by stabilizing HIF-1α in vitro and in vivo. For example, in cerebral ischemia models, quercetin administration significantly increases HIF-1α levels and VEGF expression, enhancing angiogenesis and reducing infarct size. Moreover, the anti-ischemic activity of quercetin has been shown to be independent of classical kinase signalling, highlighting HIF-PHD inhibition as a distinct, direct mechanism [181].
Therapeutic implications in neurologic disorders
By stabilizing HIF-1α through direct PHD inhibition, quercetin may offer substantial therapeutic advantage in treating stroke, TBI, and NDDs exacerbated by hypoxic stress. This mechanism is particularly relevant in patients in which ischemia-induced cell death and vascular damage contribute to disease progression, such as occurs in AD and PD. Importantly, the dual role of quercetin as an antioxidant and HIF stabilizer may synergistically improve neurologic outcomes [182].
Clinical relevance and pharmacologic outlook
Although most HIF-PHD inhibitors under clinical evaluation (e.g., roxadustat) are synthetic, quercetin offers a natural, bioavailable alternative with minimal toxicity. The HIF-stabilizing property of quercetin supports ongoing interest in phytochemicals for ischemic preconditioning strategies in neurology. Thus, inclusion of this mechanism not only expands the pharmacodynamic spectrum of quercetin but also underscores the translational value in ischemia-related neurotherapy [183, 184].
Disease-specific relevance of HIF-PHD inhibition by quercetin
Quercetin in ischemic stroke and cerebral ischemia
Quercetin treatment significantly reduces infarct volume and neurologic deficits in animal models of middle cerebral artery occlusion (MCAO). These effects were attributed to HIF-1α stabilization through HIF-PHD inhibition, leading to upregulation of VEGF and angiogenesis and promoting neurovascular repair and oxygen homeostasis. Quercetin preconditioning mimics hypoxia-induced neuroprotection, suggesting potential for stroke management [185].
Application in TBI
Quercetin prevents cerebral edema and improves BBB integrity in rodent TBI models. These effects are partly mediated through activation of the HIF-1α pathway, indicating neuroprotective adaptation to hypoxia via PHD inhibition. Combined with the anti-inflammatory action, HIF-PHD targeting by quercetin contributes to better functional recovery post-TBI [186].
Relevance in PD
PD is characterized by dopaminergic neuronal death that is often exacerbated by mitochondrial dysfunction and localized hypoxia. Studies have shown that quercetin-induced HIF-1α activation improves mitochondrial bioenergetics and enhances expression of glycolytic enzymes in PD models, potentially offering a compensatory metabolic mechanism under hypoxic stress [187].
AD and neurovascular deficits
AD pathology includes neurovascular degeneration and chronic hypoperfusion. By stabilizing HIF-1α, quercetin helps restore VEGF levels and improve cerebral perfusion, mitigating hypoxia-related neuronal damage and enhancing memory performance in AD mouse models [147].
Integrative summary of quercetin neuroprotective mechanisms
Multitargeted modulation of key pathways
Quercetin exerts neuroprotective activity by regulating interconnected signalling networks, including Nrf2-ARE, PI3K/Akt, MAPK, CREB, and SIRT1, which collectively enhance antioxidant defenses, promote neurogenesis, and suppress inflammation and apoptosis.
Protection against oxidative stress and mitochondrial dysfunction
Through direct ROS scavenging and stimulation of endogenous antioxidant enzymes, like SOD and CAT, quercetin maintains mitochondrial integrity and supports ATP synthesis. Quercetin also prevents mitochondrial swelling and lipid peroxidation, especially in AD, PD, and HD models.
Anti-inflammatory effects across neural tissues
By suppressing the NF-κB and TNF-α pathways, quercetin limits microglial activation, cytokine release, and endothelial adhesion molecule expression, thereby preserving BBB integrity and reducing chronic neuroinflammation.
Modulation of protein aggregation
Quercetin inhibits amyloid-beta (Aβ) aggregation, τ hyperphosphorylation, and α-synuclein fibrillization, reducing synaptic dysfunction and neuronal toxicity across NDD models.
Enhancement of neuroplasticity and cognitive function
Quercetin stimulates BDNF- and CREB-mediated transcription, promotes dendritic growth, and enhances learning and memory by preserving ACh levels through AChE and BChE inhibition.
Neurovascular protection and BBB integrity
By stabilizing tight junction proteins (claudin-5 and occludin) and promoting NO bioavailability, quercetin supports cerebral perfusion and reduces leukocyte infiltration in neurovascular units.
Gene modulation and epigenetic influence
Emerging evidence suggests that quercetin affects the transcription of genes related to neuroprotection through epigenetic modulation of key enzymes and transcription factors, such as PGC-1α, SIRT1, and HIF-1α, indicating potential for long-term neuronal resilience.
Clinical and translational promise
The synergy of these molecular effects has been validated in multiple in vitro, in vivo, and clinical trials, affirming the potential of quercetin as an integrative therapeutic agent in NDD management.
In vitro characterization and cell viability studies related to quercetin
Quercetin has been extensively evaluated in various in vitro models to assess cytoprotective, antioxidant, and anti-inflammatory properties. While neural cell lines, such as SH-SY5Y, PC12, and BV-2 microglial cells, are commonly used for direct neuroprotective investigations, HepG2 cells derived from human hepatocellular carcinoma are frequently used in complementary studies to evaluate cytotoxicity, metabolic stability, and cellular bioavailability of quercetin and its derivatives. These hepatocyte-like cells possess active phase I and II metabolic enzymes and provide valuable insights into the hepatic metabolism and safety profile of phytochemicals prior to neurotherapeutic translation [188, 189].
Several studies have utilized HepG2 cells for MTT and LDH assays to quantify the effect of quercetin on cell viability and oxidative stress. For example, quercetin showed dose-dependent cytoprotection against H2O2-induced oxidative damage in HepG2 cells by upregulating antioxidant defense enzymes, such as GSH peroxidase and SOD. Moreover, these cells have served as a robust model for evaluating the hepatocellular impact of quercetin conjugates, including glycosidic and methylated forms, which are biologically relevant metabolites after oral administration. However, it is important to clarify that the current review compiles and analyses data from in vitro studies without performing any new experimentation involving HepG2 or other cell lines. The inclusion of HepG2-related data in this context aims to provide a holistic view of the safety, pharmacokinetics, and suitability of quercetin for neurologic applications [190, 191].
Clinical trials of quercetin focusing on therapeutic potential and outcomes in NDDs
Quercetin research as a possible treatment for NDDs has increased significantly in the last several years. The use of quercetin as a possible treatment for NDDs has grown significantly. Progressive neuronal degeneration, which results in cognitive and motor impairments, is a hallmark of NDDs. Additionally, quercetin or quercetin combination therapy has shown encouraging results in neuroprotection or NDDs in clinical trials. A large number of clinical trials have been conducted to evaluate the mechanisms, safety, and efficacy of these drugs as the focus on drug development increases (Table 6) provides an updated overview of quercetin and other medications presently undergoing clinical trials along with the results in treating NDDs.
Table 6 Clinical Trials of Quercetin and Outcomes (US National Library of Medicine. Available online: https://clinicaltrials.gov)
| Serial No. | Title | Intervention or Treatment | Clinical Trial Number (Current Requirement Status) | Present Clinical Trial Phase | Clinical Outcome |
|---|---|---|---|---|---|
| 1. | Senolytic treatment to regulate the progression of AD | Dasatinib + quercetin (AD) | NCT04063124 (completed) | Phase 1 and Phase 2 | In preclinical models, the combination of the two medicines has been shown to efficiently eliminate senescent cells and treat a variety of chronic medical diseases, such as hepatic stenosis, vascular stiffness, muscle weakness, osteoporosis, and physical frailty |
| 2. | ALSENLITE: Senolytics for Alzheimer’s disease | 1. Dasatinib 2. Quercetin (mild cognitive impairment, AD) | NCT04785300 (enrolling by invitation) | Phase 1 and Phase 2 | The number of participants who experienced hypersensitive responses and adverse events (AEs), including serious adverse events (SAEs) |
| 3. | Dasatinib with quercetin safety and viability in adults at risk for AD | 1. Dasatinib 2. Quercetin (aging) | NCT05422885 (completed) | Phase 1 and Phase 2 | Using transcranial Doppler ultrasound, the cerebral blood flow changes during N-back cognitive activity |
| 4. | RQC for the prevention of AD and retinal amyloid-β | 1. Resveratrol, quercetin, and curcumin (RQC) 2. Curcumin (Alzheimer disease, mild cognitive impairment, cognitive decline) |
NCT06470061 (not recruiting) | Phase 2 | Change in retinal amyloid-β |
| 5. | Senolytic therapy to modulate the progression of AD (SToMP-AD) study (SToMP-AD) | 1. Dasatinib + quercetin 2. Placebo capsules (AD, early onset mild cognitive impairment) |
NCT04685590 (active, not recruiting) | Phase 2 | Primary: The treatment group had more adverse events (AEs) and serious adverse events (SAEs) compared to the placebo group. Secondary:
|
| 6. | Short-term efficacy and safety of peri-spinal administration of etanercept in mild-to-moderate AD | 1. Etanercept 2. Curcum.luteol.theaflav.lip.acid, fish oil, quercetin, resveratrol (AD) |
NCT01716637 (completed) | Phase 1 | Primary: The Mini-mental Status Examination (MMSE) score was affected differently after 6 weeks of treatment with etanercept and nutritional supplements compared to nutritional supplements alone. Secondary:
|
Clinical evidence of quercetin in neuroprotection
Quercetin, a flavonoid found in many fruits, vegetables, and other plant-based foods, has shown significant neuroprotective effects in preclinical and clinical studies primarily by antioxidant, anti-inflammatory, and anti-apoptotic characteristics. The ability to scavenge ROS and modulate neuroinflammatory pathways is pivotal in mitigating the neurodegeneration seen in conditions, like AD and PD. Clinical evidence underscores the potential of quercetin in reducing neurotoxicity. For example, a study involving patients with ischemic stroke demonstrated that quercetin inhibits the secretion of inflammatory cytokines and reduces platelet aggregation, thereby minimizing inflammatory thrombosis and protecting neuronal integrity. This outcome was attributed to quercetin modulation of ROS and inhibition of neuroinflammation-associated pathways, which are critical for reducing ischemic damage.
Quercetin administration in patients with mild cognitive impairment (MCI) was the subject of one noteworthy case study. Daily oral administration of quercetin for 6 months significantly improved memory recall and executive function compared to the placebo group. The improvement correlated with reduced biomarkers of oxidative stress and inflammation in cerebrospinal fluid, suggesting the direct involvement of quercetin in neuronal health and plasticity [61, 192].
One example involves the application of quercetin in AD models, where quercetin reduced amyloid-beta toxicity and oxidative damage. In a recent animal study, quercetin inhibited the aggregation of Aβ plaques while activating the NRF2/HO1 pathway, leading to neuroprotective effects and improved memory in rodents. This finding illustrates the capability of quercetin to modify the disease process rather than merely alleviating symptoms, warranting further trials for integration into therapeutic regimens for AD and related disorders [193].
Another case study focused on patients with PD, in which adjunctive therapy with quercetin alongside standard dopamine agonists improved motor symptoms and slowed disease progression. This benefit was hypothesized to arise from the ability of quercetin to stabilize mitochondrial function, reduce neuroinflammatory markers, and enhance dopaminergic neuron survival [194].
Future perspective
The therapeutic promise of quercetin in neuroprotection calls for further research is focused on overcoming bioavailability challenges and exploring advanced delivery systems, such as nanoformulations and conjugates. Future investigations could prioritize developing encapsulated quercetin delivery systems to enhance BBB permeability and sustained release, which are critical for treating NDDs. Additionally, understanding the interaction of quercetin with molecular pathways, including NF-κB, HO-1, and TLR4/MyD88, will deepen insights into the mechanistic roles in mitigating oxidative stress, neuroinflammation, and neuronal apoptosis. Expanding clinical trials with diverse patient populations and long-term studies will be essential to establish optimal dosing regimens, side effect profiles, and therapeutic efficacy. Collaborative efforts between researchers and clinicians are crucial for translating the neuroprotective properties of quercetin into viable therapeutic interventions for diseases, like AD, PD, and HD. Furthermore, genetic and epigenetic studies exploring the impact of quercetin on neuroprotective gene expression could unlock personalized medicinal approaches.
Conclusion
In conclusion, quercetin, as a naturally occurring flavonoid, has demonstrated extensive neuroprotective properties through mechanisms, like antioxidant activity, anti-inflammatory effects, mitochondrial stabilization, and modulation of signalling pathways (Nrf2-ARE and PI3K/Akt). These actions target key processes implicated in NDDs, including AD, PD, and HD. Despite promising preclinical and clinical data, challenges, such as poor bioavailability and metabolic instability, limit therapeutic application. Recent advancements in quercetin delivery systems, including nanoparticles and nanoemulsions, have significantly enhanced its pharmacokinetics, presenting a strong case for its integration into neurodegenerative treatment protocols. Future therapeutic interventions may capitalize on combining quercetin with complementary neuroprotective agents, enhancing the synergistic effects while minimizing side effects. For example, clinical studies have shown the efficacy of quercetin in reducing amyloid-beta aggregation and oxidative damage in AD, highlighting the potential as an adjunct therapy. Further exploration of the role of quercetin in regulating neuroinflammatory and apoptotic pathways could extend the application to other CNS-related disorders, including ischemic strokes and multiple sclerosis. Large-scale, long-term clinical trials are essential to determine the ideal dosages to confirm the effectiveness, as well as address safety concerns. Quercetin represents a promising natural compound, paving the way for innovative and effective strategies in managing NDDs.
Ethics statement
No direct interactions with human or animal subjects were involved. Therefore, ethical approval and informed consent were not required.
Author contributions
Conceptualization, Ishita Debnath; Suman Ghosh; and Sobhanjan Bhunia; writing- original draft preparation, Ishita Debnath; Suman Ghosh; and Sobhanjan Bhunia; writing- review and editing, Aditi Nayak; Souvik Basak; Sumit Nandi; Soumik Bhattacharjee; Sajal Kumar Jha. All authors have read and agreed to the published version of the manuscript.
Funding
No funding or sponsorship was received for this study.
Conflicts of interest
The authors declare that there are no conflicts of interest.
Abbreviations
AD, Alzheimer’s disease
PD, Parkinson’s disease
HD, Huntington’s disease
NDDs, Neurodegenerative diseases
ROS, Reactive oxygen species
RNS, Reactive nitrogen species
GSH, Glutathione
SOD, Superoxide dismutase
Aβ, Amyloid-beta
BBB, Blood-brain barrier
APP, Amyloid precursor protein
BDNF, Brain-derived neurotrophic factor
CREB, cAMP response element binding protein
GSK3β, Glycogen synthase kinase-3β
MAPK, Mitogen-activated protein kinase
NF-κB, Nuclear factor kappa-light-chain-enhancer of activated B cells
PON2, Paraoxonase 2
HIF, Hypoxia-inducible factor
PHD, Prolyl hydroxylase domain
VEGF, Vascular endothelial growth factor
TNF-α, Tumor necrosis factor-alpha
IL, Interleukin
SIRT1, Sirtuin 1
PGC-1α, Peroxisome proliferator-activated receptor gamma coactivator 1-alpha
mPTP, Mitochondrial permeability transition pore
OATPs, Organic anion-transporting polypeptides
AChE, Acetylcholinesterase
BChE, Butyrylcholinesterase
ASO, Antisense oligonucleotide
CRISPR, Clustered regularly interspaced short palindromic repeats
SAR, Structure–activity relationship
NO, Nitric oxide
ERK, Extracellular signal-regulated kinase
JNK, c-Jun N-terminal kinase
COX, Cyclooxygenase
LOX, Lipoxygenase
ICAM-1, Intercellular adhesion molecule-1
VCAM-1, Vascular cell adhesion molecule-1
Supplementary materials
Supplementary Material can be downloaded from https://bio-integration.org/wp-content/uploads/2025/08/bioi20250073_Supplemental.pdf.
Figure authorship and originality statement
All figures (Figures 1–6) in the current version of the manuscript, including the graphical abstract and structure-activity relationship (SAR) diagrams, have been professionally re-drawn using the ChemDraw Ultra 22.2.0 tool. Each image was exported in 600 DPI high-resolution, suitable for print and digital publication. Special attention was paid to symbol standardization, labelling clarity, structural accuracy, and pathway annotations to ensure that molecular interactions, therapeutic targets, and signalling cascades are clearly communicated to both specialist and interdisciplinary readers. Color coding and iconography were integrated to improve readability and scientific value, aligning with the visual communication standards of Bio-Integration.
Graphical abstract
Highlights
- This review explores quercetin’s multi-targeted neuroprotective mechanisms in AD, PD, and HD.
- It describes the modulation of Nrf2-ARE, PI3K/Akt, MAPK, and PON2 pathways.
- It also demonstrates SAR-based insights linking quercetin’s structure to its therapeutic actions.
- Novel delivery strategies to overcome bioavailability challenges are emphasized.
- It advocates for quercetin’s role in precision medicine and translational neurotherapeutics.
In brief
This review critically examines quercetin’s multifaceted neuroprotective effects across Alzheimer’s, Parkinson’s, and Huntington’s diseases. Emphasis is placed on mechanistic pathways, including oxidative stress, inflammation, mitochondrial dysfunction, and protein aggregation, and translational relevance. Integration of SAR, pharmacokinetics, and delivery systems highlights quercetin’s promise as a multi-targeted therapeutic in neurodegenerative disease management and precision neurotherapeutics.
References
- Amanzadeh E, Esmaeili A, Rahgozar S, Nourbakhshnia M. Application of quercetin in neurological disorders: from nutrition to nanomedicine. Rev Neurosci 2019;30(5):555-72. [PMID: 30753166 DOI: 10.1515/revneuro-2018-0080]
- Islam MS, Quispe C, Hossain R, Islam MT, Al-Harrasi A, et al. Neuropharmacological effects of quercetin: a literature-based review. Front Pharmacol 2021;12:665031. [PMID: 34220504 DOI: 10.3389/fphar.2021.665031]
- Grewal AK, Singh TG, Sharma D, Sharma V, Singh M, et al. Mechanistic insights and perspectives involved in neuroprotective action of quercetin. Biomed Pharmacother 2021;140:111729. [PMID: 34044274 DOI: 10.1016/j.biopha.2021.111729]
- Zhang L, Xu L-Y, Tang F, Liu D, Zhao X-L, et al. New perspectives on the therapeutic potential of quercetin in non-communicable diseases: targeting Nrf2 to counteract oxidative stress and inflammation. J Pharm Anal 2024;14(6):100930. [PMID: 39005843 DOI: 10.1016/j.jpha.2023.12.020]
- Singh S, Nagalakshmi D, Sharma KK, Ravichandiran V. Natural antioxidants for neuroinflammatory disorders and possible involvement of Nrf2 pathway: a review. Heliyon 2021;7(2):e06216. [PMID: 33659743 DOI: 10.1016/j.heliyon.2021.e06216]
- Khan H, Ullah H, Aschner M, Cheang WS, Akkol EK. Neuroprotective effects of quercetin in Alzheimer’s disease. Biomolecules 2019;10(1):59. [PMID: 31905923 DOI: 10.3390/biom10010059]
- Jomova K, Raptova R, Alomar SY, Alwasel SH, Nepovimova E, et al. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging. Arch Toxicol 2023;97(10):2499-574. [PMID: 37597078 DOI: 10.1007/s00204-023-03562-9]
- Ho C-L, Kao N-J, Lin C-I, Cross T-WL, Lin S-H. Quercetin increases mitochondrial biogenesis and reduces free radicals in neuronal SH-SY5Y cells. Nutrients 2022;14(16):3310. [PMID: 36014814 DOI: 10.3390/nu14163310]
- Wang J, Zhang J, Yu Z-L, Chung SK, Xu B. The roles of dietary polyphenols at crosstalk between type 2 diabetes and Alzheimer’s disease in ameliorating oxidative stress and mitochondrial dysfunction via PI3K/Akt signaling pathways. Ageing Res Rev 2024;99:102416. [PMID: 39002644 DOI: 10.1016/j.arr.2024.102416]
- Jazvinšćak Jembrek M, Oršolić N, Mandić L, Sadžak A, Šegota S. Anti-oxidative, anti-inflammatory and anti-apoptotic effects of flavonols: targeting Nrf2, NF-κB and p53 pathways in neurodegeneration. Antioxidants (Basel) 2021;10(10):1628. [PMID: 34679762 DOI: 10.3390/antiox10101628]
- Costa LG, Garrick JM, Roquè PJ, Pellacani C. Mechanisms of neuroprotection by quercetin: counteracting oxidative stress and more. Oxid Med Cell Longev 2016;2016:2986796. [PMID: 26904161 DOI: 10.1155/2016/2986796]
- Jakaria M, Park S-Y, Haque ME, Karthivashan G, Kim I-S, et al. Neurotoxic agent-induced injury in neurodegenerative disease model: focus on involvement of glutamate receptors. Front Mol Neurosci 2018;11:307. [PMID: 30210294 DOI: 10.3389/fnmol.2018.00307]
- Almatroodi SA, Alsahli MA, Almatroudi A, Verma AK, Aloliqi A, et al. Potential therapeutic targets of quercetin, a plant flavonol, and its role in the therapy of various types of cancer through the modulation of various cell signaling pathways. Molecules 2021;26(5):1315. [PMID: 33804548 DOI: 10.3390/molecules26051315]
- Orhan IE. Cholinesterase inhibitory potential of quercetin towards Alzheimer’s disease – a promising natural molecule or fashion of the day? A narrowed review. Curr Neuropharmacol 2021;19(12):2205-13. [PMID: 33213346 DOI: 10.2174/1570159X18666201119153807]
- Cui Z, Zhao X, Amevor FK, Du X, Wang Y, et al. Therapeutic application of quercetin in aging-related diseases: SIRT1 as a potential mechanism. Front Immunol 2022;13:943321. [PMID: 35935939 DOI: 10.3389/fimmu.2022.943321]
- McKay TB, Emmitte KA, German C, Karamichos D. Quercetin and related analogs as therapeutics to promote tissue repair. Bioengineering (Basel) 2023;10(10):1127. [PMID: 37892857 DOI: 10.3390/bioengineering10101127]
- Pereira L, Cotas J. Therapeutic potential of polyphenols and other micronutrients of marine origin. Mar Drugs 2023;21(6):323. [PMID: 37367648 DOI: 10.3390/md21060323]
- Qi W, Qi W, Xiong D, Long M. Quercetin: its antioxidant mechanism, antibacterial properties and potential application in prevention and control of toxipathy. Molecules 2022;27(19):6545. [PMID: 36235082 DOI: 10.3390/molecules27196545]
- Patel K, Kumar V, Rahman M, Verma A, Patel DK. New insights into the medicinal importance, physiological functions and bioanalytical aspects of an important bioactive compound of foods ‘Hyperin’: health benefits of the past, the present, the future. Beni Suef Univ J Basic Appl Sci 2018;7(1):31-42. [DOI: 10.1016/j.bjbas.2017.05.009]
- Rana A, Samtiya M, Dhewa T, Mishra V, Aluko RE. Health benefits of polyphenols: a concise review. J Food Biochem 2022;46(10):e14264. [PMID: 35694805 DOI: 10.1111/jfbc.14264]
- Montané X, Kowalczyk O, Reig-Vano B, Bajek A, Roszkowski K, et al. Current perspectives of the applications of polyphenols and flavonoids in cancer therapy. Molecules 2020;25(15):3342. [PMID: 32717865 DOI: 10.3390/molecules25153342]
- Sabiu S, Madende M, Ajao AA, Aladodo RA, Nurain IO, et al. The genus Allium (Amaryllidaceae: Alloideae): features, phytoconstituents, and mechanisms of antidiabetic potential of Allium cepa and Allium sativum. In: Watson RR, Preedy VR, editors. Bioactive food as dietary interventions for diabetes. Elsevier; 2019. pp. 137-54. [DOI: 10.1016/B978-0-12-813822-9.00009-6]
- Chan EWC, Wong SK, Tangah J, Inoue T, Chan HT. Phenolic constituents and anticancer properties of Morus alba (white mulberry) leaves. J Integr Med 2020;18(3):189-95. [PMID: 32115383 DOI: 10.1016/j.joim.2020.02.006]
- Al-Zughbi I, Krayem M. Quince fruit Cydonia oblonga Mill nutritional composition, antioxidative properties, health benefits and consumers preferences towards some industrial quince products: a review. Food Chem 2022;393:133362. [PMID: 35661598 DOI: 10.1016/j.foodchem.2022.133362]
- Patocka J, Bhardwaj K, Klimova B, Nepovimova E, Wu Q, et al. Malus domestica: a review on nutritional features, chemical composition, traditional and medicinal value. Plants (Basel) 2020;9(11):1408. [PMID: 33105724 DOI: 10.3390/plants9111408]
- Nunes AR, Gonçalves AC, Falcão A, Alves G, Silva LR. Prunus avium L. (Sweet Cherry) by-products: a source of phenolic compounds with antioxidant and anti-hyperglycemic properties—a review. Appl Sci 2021;11(18):8516. [DOI: 10.3390/app11188516]
- Navarro M, Moreira I, Arnaez E, Quesada S, Azofeifa G, et al. Polyphenolic characterization and antioxidant activity of malus domestica and prunus domestica cultivars from Costa Rica. Foods 2018;7(2):15. [PMID: 29385709 DOI: 10.3390/foods7020015]
- Klimek-Szczykutowicz M, Gaweł-Bęben K, Rutka A, Blicharska E, Tatarczak-Michalewska M, et al. Moringa oleifera (drumstick tree)-nutraceutical, cosmetological and medicinal importance: a review. Front Pharmacol 2024;15:1288382. [PMID: 38370483 DOI: 10.3389/fphar.2024.1288382]
- Ranjha MMAN, Shafique B, Wang L, Irfan S, Safdar MN, et al. A comprehensive review on phytochemistry, bioactivity and medicinal value of bioactive compounds of pomegranate (Punica granatum). Adv Tradit Med 2023;23(1):37-57. [DOI: 10.1007/s13596-021-00566-7]
- Hudz N, Kobylinska L, Pokajewicz K, Horčinová Sedláčková V, Fedin R, et al. Mentha piperita: essential oil and extracts, their biological activities, and perspectives on the development of new medicinal and cosmetic products. Molecules 2023;28(21):7444. [PMID: 37959863 DOI: 10.3390/molecules28217444]
- Mahendran G, Verma SK, Rahman L-U. The traditional uses, phytochemistry and pharmacology of spearmint (Mentha spicata L.): a review. J Ethnopharmacol 2021;278:114266. [PMID: 34087400 DOI: 10.1016/j.jep.2021.114266]
- Nekkaa A, Benaissa A, Mutelet F, Canabady-Rochelle L. Rhamnusalaternus plant: extraction of bioactive fractions and evaluation of their pharmacological and phytochemical properties. Antioxidants (Basel) 2021;10(2):300. [PMID: 33669348 DOI: 10.3390/antiox10020300]
- Applequist WL, Moerman DE. Yarrow (Achillea millefolium L.): a neglected panacea? A review of ethnobotany, bioactivity, and biomedical research. Econ Bot 2011;65(2):209-25 [DOI: 10.1007/s12231-011-9154-3]
- Assefa AD, Choi S, Lee J-E, Sung J-S, Hur O-S, et al. Identification and quantification of selected metabolites in differently pigmented leaves of lettuce (Lactuca sativa L.) cultivars harvested at mature and bolting stages. BMC Chem 2019;13(1):56. [PMID: 31384804 DOI: 10.1186/s13065-019-0570-2]
- Guo J, Wang Y, Li J, Zhang J, Wu Y, et al. Overview and recent progress on the biosynthesis and regulation of flavonoids in Ginkgo biloba L. Int J Mol Sci 2023;24(19):14604. [PMID: 37834050 DOI: 10.3390/ijms241914604]
- Kumar M, Saurabh V, Tomar M, Hasan M, Changan S, et al. Mango (Mangifera indica L.) leaves: nutritional composition, phytochemical profile, and health-promoting bioactivities. Antioxidants (Basel) 2021;10(2):299. [PMID: 33669341 DOI: 10.3390/antiox10020299]
- Perović J, Tumbas Šaponjac V, Kojić J, Krulj J, Moreno DA, et al. Chicory (Cichorium intybus L.) as a food ingredient – nutritional composition, bioactivity, safety, and health claims: a review. Food Chem 2021;336:127676. [PMID: 32768902 DOI: 10.1016/j.foodchem.2020.127676]
- Vrhovsek U, Masuero D, Palmieri L, Mattivi F. Identification and quantification of flavonol glycosides in cultivated blueberry cultivars. J Food Compos Anal 2012;25(1):9-16. [DOI: 10.1016/j.jfca.2011.04.015]
- Tundis R, Tenuta MC, Loizzo MR, Bonesi M, Finetti F, et al. Vaccinium species (Ericaceae): from chemical composition to bio-functional activities. Appl Sci 2021;11(12):5655. [DOI: 10.3390/app11125655]
- Kingori SM, Ochanda SO, Koech RK. Variation in levels of flavonols myricetin, quercetin and kaempferol—in Kenyan tea (Camellia sinensis L.) with processed tea types and geographic location. Open J Appl Sci 2021;11(6):736-49. [DOI: 10.4236/ojapps.2021.116054]
- Gudiño I, Martín A, Casquete R, Prieto MH, Ayuso MC, et al. Evaluation of broccoli (Brassica oleracea var. italica) crop by-products as sources of bioactive compounds. Sci Hortic 2022;304:111284. [DOI: 10.1016/j.scienta.2022.111284]
- Zietz M, Weckmüller A, Schmidt S, Rohn S, Schreiner M, et al. Genotypic and climatic influence on the antioxidant activity of flavonoids in Kale (Brassica oleracea var. sabellica). J Agric Food Chem 2010;58(4):2123-30. [PMID: 20095605 DOI: 10.1021/jf9033909]
- Kyriakou S, Michailidou K, Amery T, Stewart K, Winyard PG, et al. Polyphenolics, glucosinolates and isothiocyanates profiling of aerial parts of Nasturtium officinale (Watercress). Front Plant Sci 2022;13:998755. [PMID: 36457522 DOI: 10.3389/fpls.2022.998755]
- Galeotti N. Hypericum perforatum (St John’s wort) beyond depression: a therapeutic perspective for pain conditions. J Ethnopharmacol 2017;200:136-46. [PMID: 28216196 DOI: 10.1016/j.jep.2017.02.016]
- Rusalepp L, Raal A, Püssa T, Mäeorg U. Comparison of chemical composition of Hypericum perforatum and H. maculatum in Estonia. Biochem Syst Ecol 2017;73:41-6. [DOI: 10.1016/j.bse.2017.06.004]
- Mohammad Azmin SNH, Mat Nor MS. Chemical fingerprint of Centella Asiatica’s bioactive compounds in the ethanolic and aqueous extracts. Adv Biomark Sci Technol 2020;2:35-44. [DOI: 10.1016/j.abst.2020.10.001]
- Minaiyan M, Ghanadian S-M, Hossaini M. Protective effect of Apium graveolens L. (Celery) seeds extracts and luteolin on acetic acid-induced colitis in rats. Int J Prev Med 2021;12:100. [PMID: 34729134 DOI: 10.4103/ijpvm.IJPVM_651_20]
- Saygi KO. Quantification of phenolics from Coriandrum sativum vulgare and Coriandrum sativum microcarpum by HPLC–DAD. Iran J Sci Technol Trans A Sci 2021;45(4):1319-26. [DOI: 10.1007/s40995-021-01132-1]
- Udaya Rajesh R, Dhanaraj S. A critical review on quercetin bioflavonoid and its derivatives: scope, synthesis, and biological applications with future prospects. Arab J Chem 2023;16(8):104881. [DOI: 10.1016/j.arabjc.2023.104881]
- Sarkar S, Singh RP, Bhattacharya G. Exploring the role of Azadirachta indica (neem) and its active compounds in the regulation of biological pathways: an update on molecular approach. 3 Biotech 2021;11(4):178. [PMID: 33927969 DOI: 10.1007/s13205-021-02745-4]
- Adurosakin OE, Iweala EJ, Otike JO, Dike ED, Uche ME, et al. Ethnomedicinal uses, phytochemistry, pharmacological activities and toxicological effects of Mimosa pudica– a review. Pharmacol Res Mod Chin Med 2023;7:100241. [DOI: 10.1016/j.prmcm.2023.100241]
- Thissera B, Visvanathan R, Khanfar MA, Qader MM, Hassan MHA, et al. Sesbania grandiflora L. Poir leaves: a dietary supplement to alleviate type 2 diabetes through metabolic enzymes inhibition. S Afr J Bot 2020;130:282-99. [DOI: 10.1016/j.sajb.2020.01.011]
- Azab SS, Abdel-Daim M, Eldahshan OA. Phytochemical, cytotoxic, hepatoprotective and antioxidant properties of Delonix regia leaves extract. Med Chem Re 2013;22(9):4269-77. [DOI: 10.1007/s00044-012-0420-4]
- Izzo L, Castaldo L, Narváez A, Graziani G, Gaspari A, et al. Analysis of phenolic compounds in commercial Cannabis sativa L. inflorescences using UHPLC-Q-Orbitrap HRMS. Molecules 2020;25(3):631. [PMID: 32024009 DOI: 10.3390/molecules25030631]
- Okhuarobo A, Falodun JE, Erharuyi O, Imieje V, Falodun A, et al. Harnessing the medicinal properties of Andrographis paniculata for diseases and beyond: a review of its phytochemistry and pharmacology. Asian Pac J Trop Dis 2014;4(3):213-22. [DOI: 10.1016/S2222-1808(14)60509-0]
- Basit A, Shutian T, Khan A, Khan SM, Shahzad R, et al. Anti-inflammatory and analgesic potential of leaf extract of Justicia adhatoda L. (Acanthaceae) in Carrageenan and Formalin-induced models by targeting oxidative stress. Biomed Pharmacother 2022;153:113322. [PMID: 35763968 DOI: 10.1016/j.biopha.2022.113322]
- Jain S, Mehata MS. Medicinal plant leaf extract and pure flavonoid mediated green synthesis of silver nanoparticles and their enhanced antibacterial property. Sci Rep 2017;7(1):15867. [PMID: 29158537 DOI: 10.1038/s41598-017-15724-8]
- Dong C, Hu H, Hu Y, Xie J. Metabolism of flavonoids in novel banana germplasm during fruit development. Front Plant Sci 2016;7:1291. [PMID: 27625665 DOI: 10.3389/fpls.2016.01291]
- Aghofack-Nguemezi J, Schwab W. Differential accumulation of flavonoids by tomato (Solanum lycopersicum) fruits tissues during maturation and ripening. J Appl Biosci 2015;84(1):7682. [DOI: 10.4314/jab.v84i1.2]
- Alhaithloul HAS, Galal FH, Seufi AM. Effect of extreme temperature changes on phenolic, flavonoid contents and antioxidant activity of tomato seedlings (Solanum lycopersicum L.). PeerJ 2021;9:e11193. [PMID: 34026345 DOI: 10.7717/peerj.11193]
- Jing Z, Wang Z, Li X, Li X, Cao T, et al. Protective effect of quercetin on posttraumatic cardiac injury. Sci Rep 2016;6:30812. [PMID: 27470932 DOI: 10.1038/srep30812]
- Xiong F, Zhang Y, Li T, Tang Y, Song S-Y, et al. A detailed overview of quercetin: implications for cell death and liver fibrosis mechanisms. Front Pharmacol 2024;15:1389179. [PMID: 38855739 DOI: 10.3389/fphar.2024.1389179]
- Kandemir K, Tomas M, McClements DJ, Capanoglu E. Recent advances on the improvement of quercetin bioavailability. Trends Food Sci Technol 2022;119:192-200. [DOI: 10.1016/j.tifs.2021.11.032]
- Wang W, Sun C, Mao L, Ma P, Liu F, et al. The biological activities, chemical stability, metabolism and delivery systems of quercetin: a review. Trends Food Sci Technol 2016;56:21-38. [DOI: 10.1016/j.tifs.2016.07.004]
- Scalbert A, Williamson G. Dietary intake and bioavailability of polyphenols. J Nutr 2000;130(8S Suppl):2073S-85S. [PMID: 10917926 DOI: 10.1093/jn/130.8.2073S]
- Tomou E-M, Papakyriakopoulou P, Saitani E-M, Valsami G, Pippa N, et al. Recent advances in nanoformulations for quercetin delivery. Pharmaceutics 2023;15(6):1656. [PMID: 37376104 DOI: 10.3390/pharmaceutics15061656]
- Koo J, Lim C, Oh KT. Recent advances in intranasal administration for brain-targeting delivery: a comprehensive review of lipid-based nanoparticles and stimuli-responsive gel formulations. Int J Nanomedicine 2024;19:1767-807. [PMID: 38414526 DOI: 10.2147/IJN.S439181]
- Michala A-S, Pritsa A. Quercetin: a molecule of great biochemical and clinical value and its beneficial effect on diabetes and cancer. Diseases 2022;10(3):37. [PMID: 35892731 DOI: 10.3390/diseases10030037]
- De Marchi U, Biasutto L, Garbisa S, Toninello A, Zoratti M. Quercetin can act either as an inhibitor or an inducer of the mitochondrial permeability transition pore: a demonstration of the ambivalent redox character of polyphenols. Biochim Biophys Acta 2009;1787(12):1425-32. [PMID: 19523917 DOI: 10.1016/j.bbabio.2009.06.002]
- Kim G-N, Jang H-D. Protective mechanism of quercetin and rutin using glutathione metabolism on HO-induced oxidative stress in HepG2 cells. Ann N Y Acad Sci 2009;1171:530-7. [PMID: 19723100 DOI: 10.1111/j.1749-6632.2009.04690.x]
- Someya Y, Saito S, Takeda S, Adachi N, Kurosawa A. Quercetin exhibits cytotoxicity in cancer cells by inducing two-ended DNA double-strand breaks. Biochem Biophys Res Commun 2024;739:150977. [PMID: 39549336 DOI: 10.1016/j.bbrc.2024.150977]
- Fakhri KU, Sharma D, Fatma H, Yasin D, Alam M, et al. The dual role of dietary phytochemicals in oxidative stress: implications for oncogenesis, cancer chemoprevention, and ncRNA regulation. Antioxidants (Basel) 2025;14(6):620. [PMID: 40563255 DOI: 10.3390/antiox14060620]
- Bardestani A, Ebrahimpour S, Esmaeili A, Esmaeili A. Quercetin attenuates neurotoxicity induced by iron oxide nanoparticles. J Nanobiotechnology 2021;19(1):327. [PMID: 34663344 DOI: 10.1186/s12951-021-01059-0]
- Vollmannová A, Bojňanská T, Musilová J, Lidiková J, Cifrová M. Quercetin as one of the most abundant represented biological valuable plant components with remarkable chemoprotective effects – a review. Heliyon 2024;10(12):e33342. [PMID: 39021910 DOI: 10.1016/j.heliyon.2024.e33342]
- Aghababaei F, Hadidi M. Recent advances in potential health benefits of quercetin. Pharmaceuticals (Basel) 2023;16(7):1020. [PMID: 37513932 DOI: 10.3390/ph16071020]
- Nam J-S, Sharma AR, Nguyen LT, Chakraborty C, Sharma G, et al. Application of bioactive quercetin in oncotherapy: from nutrition to nanomedicine. Molecules 2016;21(1):E108. [PMID: 26797598 DOI: 10.3390/molecules21010108]
- Kawabata K, Mukai R, Ishisaka A. Quercetin and related polyphenols: new insights and implications for their bioactivity and bioavailability. Food Funct 2015;6(5):1399-417. [PMID: 25761771 DOI: 10.1039/c4fo01178c]
- Hai Y, Zhang Y, Liang Y, Ma X, Qi X, et al. Advance on the absorption, metabolism, and efficacy exertion of quercetin and its important derivatives. Food Front 2020;1(4):420-34. [DOI: 10.1002/fft2.50]
- Chen X, Yin OQP, Zuo Z, Chow MSS. Pharmacokinetics and modeling of quercetin and metabolites. Pharm Res 2005;22(6):892-901. [PMID: 15948033 DOI: 10.1007/s11095-005-4584-1]
- Kyriakoudi A, Spanidi E, Mourtzinos I, Gardikis K. Innovative delivery systems loaded with plant bioactive ingredients: formulation approaches and applications. Plants (Basel) 2021;10(6):1238. [PMID: 34207139 DOI: 10.3390/plants10061238]
- Sun J, Jing H, Liu T, Dong S, Obadi M, et al. Evaluation of antioxidant modification on the functional and structural properties of EWP conjugates. RSC Adv 2020;10(18):10666-72. [PMID: 35492916 DOI: 10.1039/d0ra00023j]
- Veiko AG, Lapshina EA, Zavodnik IB. Comparative analysis of molecular properties and reactions with oxidants for quercetin, catechin, and naringenin. Mol Cell Biochem 2021;476(12):4287-99. [PMID: 34406575 DOI: 10.1007/s11010-021-04243-w]
- Chiorcea-Paquim A-M. Electrochemistry of flavonoids: a comprehensive review. Int J Mol Sci 2023;24(21):15667. [PMID: 37958651 DOI: 10.3390/ijms242115667]
- Evans JA, Mendonca P, Soliman KFA. Neuroprotective effects and therapeutic potential of the citrus flavonoid hesperetin in neurodegenerative diseases. Nutrients 2022;14(11):2228. [PMID: 35684025 DOI: 10.3390/nu14112228]
- Xu D, Hu M-J, Wang Y-Q, Cui Y-L. Antioxidant activities of quercetin and its complexes for medicinal application. Molecules 2019;24(6):1123. [PMID: 30901869 DOI: 10.3390/molecules24061123]
- Nam G, Hong M, Lee J, Lee HJ, Ji Y, et al. Multiple reactivities of flavonoids towards pathological elements in Alzheimer’s disease: structure-activity relationship. Chem Sci 2020;11(37):10243-54. [PMID: 34094290 DOI: 10.1039/d0sc02046j]
- Jeon H, Kim H, Choi D, Kim D, Park S-Y, et al. Quercetin activates an angiogenic pathway, hypoxia inducible factor (HIF)-1-vascular endothelial growth factor, by inhibiting HIF-prolyl hydroxylase: a structural analysis of quercetin for inhibiting HIF-prolyl hydroxylase. Mol Pharmacol 2007;71(6):1676-84. [PMID: 17377063 DOI: 10.1124/mol.107.034041]
- Cheignon C, Tomas M, Bonnefont-Rousselot D, Faller P, Hureau C, et al. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Biol 2018;14:450-64. [PMID: 29080524 DOI: 10.1016/j.redox.2017.10.014]
- Dini I, Grumetto L. Recent advances in natural polyphenol research. Molecules 2022;27(24):8777. [PMID: 36557912 DOI: 10.3390/molecules27248777]
- Yang K, Lv Z, Zhao W, Lai G, Zheng C, et al. The potential of natural products to inhibit abnormal aggregation of α-Synuclein in the treatment of Parkinson’s disease. Front Pharmacol 2024;15:1468850. [PMID: 39508052 DOI: 10.3389/fphar.2024.1468850]
- Ribeiro D, Freitas M, Lima JLFC, Fernandes E. Proinflammatory pathways: the modulation by flavonoids. Med Res Rev 2015;35(5):877-936. [PMID: 25926332 DOI: 10.1002/med.21347]
- Aleebrahim-Dehkordi E, Soveyzi F, Arian AS, Hamedanchi NF, Hasanpour-Dehkordi A, et al. Quercetin and its role in reducing the expression of pro-inflammatory cytokines in osteoarthritis. Antiinflamm Antiallergy Agents Med Chem 2023;21(3):153-65. [PMID: 36518039 DOI: 10.2174/1871523022666221213155905]
- Spagnuolo C, Moccia S, Russo GL. Anti-inflammatory effects of flavonoids in neurodegenerative disorders. Eur J Med Chem 2018;153:105-15. [PMID: 28923363 DOI: 10.1016/j.ejmech.2017.09.001]
- Dorta DJ, Pigoso AA, Mingatto FE, Rodrigues T, Prado IMR, et al. The interaction of flavonoids with mitochondria: effects on energetic processes. Chem Biol Interact 2005;152(2-3):67-78. [PMID: 15840381 DOI: 10.1016/j.cbi.2005.02.004]
- Koklesova L, Liskova A, Samec M, Zhai K, Al-Ishaq RK, et al. Protective effects of flavonoids against mitochondriopathies and associated pathologies: focus on the predictive approach and personalized prevention. Int J Mol Sci 2021;22(16):8649. [PMID: 34445360 DOI: 10.3390/ijms22168649]
- Chiang M-C, Tsai T-Y, Wang C-J. The potential benefits of quercetin for brain health: a review of anti-inflammatory and neuroprotective mechanisms. Int J Mol Sci 2023;24(7):6328. [PMID: 37047299 DOI: 10.3390/ijms24076328]
- Massi A, Bortolini O, Ragno D, Bernardi T, Sacchetti G, et al. Research progress in the modification of quercetin leading to anticancer agents. Molecules 2017;22(8):1270. [PMID: 28758919 DOI: 10.3390/molecules22081270]
- Duan N, Hu X, Zhou R, Li Y, Wu W, et al. A review on dietary flavonoids as modulators of the tumor microenvironment. Mol Nutr Food Res 2023;67(7):e2200435. [PMID: 36698331 DOI: 10.1002/mnfr.202200435]
- Eisvand F, Tajbakhsh A, Seidel V, Zirak MR, Tabeshpour J, et al. Quercetin and its role in modulating endoplasmic reticulum stress: a review. Phytother Res 2022;36(1):73-84. [PMID: 34528309 DOI: 10.1002/ptr.7283]
- Jakaria M, Azam S, Jo S-H, Kim I-S, Dash R, et al. Potential therapeutic targets of quercetin and its derivatives: its role in the therapy of cognitive impairment. J Clin Med 2019;8(11):1789. [PMID: 31717708 DOI: 10.3390/jcm8111789]
- Jabir NR, Khan FR, Tabrez S. Cholinesterase targeting by polyphenols: a therapeutic approach for the treatment of Alzheimer’s disease. CNS Neurosci Ther 2018;24(9):753-62. [PMID: 29770579 DOI: 10.1111/cns.12971]
- Breijyeh Z, Karaman R. Comprehensive review on Alzheimer’s disease: causes and treatment. Molecules 2020;25(24):5789. [PMID: 33302541 DOI: 10.3390/molecules25245789]
- Walczak-Nowicka ŁJ, Herbet M. Acetylcholinesterase inhibitors in the treatment of neurodegenerative diseases and the role of acetylcholinesterase in their pathogenesis. Int J Mol Sci 2021;22(17):9290. [PMID: 34502198 DOI: 10.3390/ijms22179290]
- Andrés CMC, Pérez de la Lastra JM, Bustamante Munguira E, Juan CA, Plou FJ, et al. Electrophilic compounds in the human diet and their role in the induction of the transcription factor NRF2. Int J Mol Sci 2024;25(6):3521. [PMID: 38542492 DOI: 10.3390/ijms25063521]
- Moosavi F, Hosseini R, Saso L, Firuzi O. Modulation of neurotrophic signaling pathways by polyphenols. Drug Des Devel Ther 2015;10:23-42. [PMID: 26730179 DOI: 10.2147/DDDT.S96936]
- Naoi M, Inaba-Hasegawa K, Shamoto-Nagai M, Maruyama W. Neurotrophic function of phytochemicals for neuroprotection in aging and neurodegenerative disorders: modulation of intracellular signaling and gene expression. J Neural Transm (Vienna) 2017;124(12):1515-27. [PMID: 29030688 DOI: 10.1007/s00702-017-1797-5]
- Gómez-Ganau S, de Julián-Ortiz JV, Gozalbes R. Recent advances in computational approaches for designing potential anti-Alzheimer’s agents. In: Roy K, editor. Computational modeling of drugs against Alzheimer’s disease. New York, NY: Humana Press; 2018. pp. 25-59. [DOI: 10.1007/978-1-4939-7404-7_2]
- Fakhri S, Gravandi MM, Abdian S, Moradi SZ, Echeverría J. Quercetin derivatives in combating spinal cord injury: a mechanistic and systematic review. Life (Basel) 2022;12(12):1960. [PMID: 36556325 DOI: 10.3390/life12121960]
- Liu Y, Guo M. Studies on transition metal-quercetin complexes using electrospray ionization tandem mass spectrometry. Molecules 2015;20(5):8583-94. [PMID: 25985359 DOI: 10.3390/molecules20058583]
- Lamptey RNL, Chaulagain B, Trivedi R, Gothwal A, Layek B, et al. A review of the common neurodegenerative disorders: current therapeutic approaches and the potential role of nanotherapeutics. Int J Mol Sci 2022;23(3):1851. [PMID: 35163773 DOI: 10.3390/ijms23031851]
- Toader C, Tataru CP, Munteanu O, Serban M, Covache-Busuioc R-A, et al. Decoding neurodegeneration: a review of molecular mechanisms and therapeutic advances in Alzheimer’s, Parkinson’s, and ALS. Int J Mol Sci 2024;25(23):12613. [PMID: 39684324 DOI: 10.3390/ijms252312613]
- Sehar U, Rawat P, Reddy AP, Kopel J, Reddy PH. Amyloid beta in aging and Alzheimer’s disease. Int J Mol Sci 2022;23(21):12924. [PMID: 36361714 DOI: 10.3390/ijms232112924]
- Zhang ZJ, Cheang LCV, Wang MW, Lee SM-Y. Quercetin exerts a neuroprotective effect through inhibition of the iNOS/NO system and pro-inflammation gene expression in PC12 cells and in zebrafish. Int J Mol Med 2011;27(2):195-203. [PMID: 21132259 DOI: 10.3892/ijmm.2010.571]
- Vaz M, Silva V, Monteiro C, Silvestre S. Role of aducanumab in the treatment of Alzheimer’s disease: challenges and opportunities. Clin Interv Aging 2022;17:797-810. [PMID: 35611326 DOI: 10.2147/CIA.S325026]
- Das B, Yan R. A close look at BACE1 inhibitors for Alzheimer’s disease treatment. CNS Drugs 2019;33(3):251-63. [PMID: 30830576 DOI: 10.1007/s40263-019-00613-7]
- Reddy PH. Abnormal tau, mitochondrial dysfunction, impaired axonal transport of mitochondria, and synaptic deprivation in Alzheimer’s disease. Brain Res 2011;1415:136-48. [PMID: 21872849 DOI: 10.1016/j.brainres.2011.07.052]
- Reyes ST, Deacon RMJ, Guo SG, Altimiras FJ, Castillo JB, et al. Effects of the sigma-1 receptor agonist blarcamesine in a murine model of fragile X syndrome: neurobehavioral phenotypes and receptor occupancy. Sci Rep 2021;11(1):17150. [PMID: 34433831 DOI: 10.1038/s41598-021-94079-7]
- Huang L-K, Chao S-P, Hu C-J. Clinical trials of new drugs for Alzheimer disease. J Biomed Sci 2020;27(1):18. [PMID: 31906949 DOI: 10.1186/s12929-019-0609-7]
- Miao J, Ma H, Yang Y, Liao Y, Lin C, et al. Microglia in Alzheimer’s disease: pathogenesis, mechanisms, and therapeutic potentials. Front Aging Neurosci 2023;15:1201982. [PMID: 37396657 DOI: 10.3389/fnagi.2023.1201982]
- Jung YJ, Tweedie D, Scerba MT, Kim DS, Palmas MF, et al. Repurposing immunomodulatory imide drugs (IMiDs) in neuropsychiatric and neurodegenerative disorders. Front Neurosci 2021;15:656921. [PMID: 33854417 DOI: 10.3389/fnins.2021.656921]
- Hampel H, Mesulam M-M, Cuello AC, Farlow MR, Giacobini E, et al. The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain 2018;141(7):1917-33. [PMID: 29850777 DOI: 10.1093/brain/awy132]
- Marucci G, Buccioni M, Ben DD, Lambertucci C, Volpini R, et al. Efficacy of acetylcholinesterase inhibitors in Alzheimer’s disease. Neuropharmacology 2021;190:108352. [PMID: 33035532 DOI: 10.1016/j.neuropharm.2020.108352]
- Negi S, Khurana N, Duggal N. The misfolding mystery: α-synuclein and the pathogenesis of Parkinson’s disease. Neurochem Int 2024;177:105760. [PMID: 38723900 DOI: 10.1016/j.neuint.2024.105760]
- Savitt D, Jankovic J. Targeting α-synuclein in Parkinson’s disease: progress towards the development of disease-modifying therapeutics. Drugs 2019;79(8):797-810. [PMID: 30982161 DOI: 10.1007/s40265-019-01104-1]
- Gouda NA, Elkamhawy A, Cho J. Emerging therapeutic strategies for Parkinson’s disease and future prospects: a 2021 update. Biomedicines 2022;10(2):371. [PMID: 35203580 DOI: 10.3390/biomedicines10020371]
- Masato A, Plotegher N, Boassa D, Bubacco L. Impaired dopamine metabolism in Parkinson’s disease pathogenesis. Mol Neurodegener 2019;14(1):35. [PMID: 31488222 DOI: 10.1186/s13024-019-0332-6]
- Kwon DK, Kwatra M, Wang J, Ko HS. Levodopa-induced dyskinesia in Parkinson’s disease: pathogenesis and emerging treatment strategies. Cells 2022;11(23):3736. [PMID: 36496996 DOI: 10.3390/cells11233736]
- Ferraiolo M, Hermans E. The complex molecular pharmacology of the dopamine D2 receptor: implications for pramipexole, ropinirole, and rotigotine. Pharmacol Ther 2023;245:108392. [PMID: 36958527 DOI: 10.1016/j.pharmthera.2023.108392]
- Mazzio EA, Close F, Soliman KFA. The biochemical and cellular basis for nutraceutical strategies to attenuate neurodegeneration in Parkinson’s disease. Int J Mol Sci 2011;12(1):506-69. [PMID: 21340000 DOI: 10.3390/ijms12010506]
- Yang N, Guan Q-W, Chen F-H, Xia Q-X, Yin X-X, et al. Antioxidants targeting mitochondrial oxidative stress: promising neuroprotectants for epilepsy. Oxid Med Cell Longev 2020;2020:6687185. [PMID: 33299529 DOI: 10.1155/2020/6687185]
- Kobayashi K, Imagama S, Ohgomori T, Hirano K, Uchimura K, et al. Minocycline selectively inhibits M1 polarization of microglia. Cell Death Dis 2013;4(3):e525. [PMID: 23470532 DOI: 10.1038/cddis.2013.54]
- Wixey JA, Sukumar KR, Pretorius R, Lee KM, Colditz PB, et al. Ibuprofen treatment reduces the neuroinflammatory response and associated neuronal and white matter impairment in the growth restricted newborn. Front Physiol 2019;10:541. [PMID: 31133875 DOI: 10.3389/fphys.2019.00541]
- Bhat AA, Moglad E, Afzal M, Thapa R, Almalki WH, et al. Therapeutic approaches targeting aging and cellular senescence in Huntington’s disease. CNS Neurosci Ther 2024;30(10):e70053. [PMID: 39428700 DOI: 10.1111/cns.70053]
- Tong H, Yang T, Xu S, Li X, Liu L, et al. Huntington’s disease: complex pathogenesis and therapeutic strategies. Int J Mol Sci 2024;25(7):3845. [PMID: 38612657 DOI: 10.3390/ijms25073845]
- Sampaio C. Huntington disease – update on ongoing therapeutic developments and a look toward the future. Parkinsonism Relat Disord 2024;122:106049. [PMID: 38418319 DOI: 10.1016/j.parkreldis.2024.106049]
- Tabrizi SJ, Leavitt BR, Landwehrmeyer GB, Wild EJ, Saft C, et al. Targeting huntingtin expression in patients with Huntington’s disease. N Engl J Med 2019;380(24):2307-16. [PMID: 31059641 DOI: 10.1056/NEJMoa1900907]
- Imbimbo BP, Triaca V, Imbimbo C, Nisticò R. Investigational treatments for neurodegenerative diseases caused by inheritance of gene mutations: lessons from recent clinical trials. Neural Regen Res 2023;18(8):1679-83. [PMID: 36751779 DOI: 10.4103/1673-5374.363185]
- Kwon D. Failure of genetic therapies for Huntington’s devastates community. Nature 2021;593(7858):180. [PMID: 33963316 DOI: 10.1038/d41586-021-01177-7]
- Jodeiri Farshbaf M, Ghaedi K. Huntington’s disease and mitochondria. Neurotox Res 2017;32(3):518-29. [PMID: 28639241 DOI: 10.1007/s12640-017-9766-1]
- Beal MF. Neuroprotective effects of creatine. Amino Acids 2011;40(5):1305-13. [PMID: 21448659 DOI: 10.1007/s00726-011-0851-0]
- Orsucci D, Mancuso M, Ienco EC, LoGerfo A, Siciliano G. Targeting mitochondrial dysfunction and neurodegeneration by means of coenzyme Q10 and its analogues. Curr Med Chem 2011;18(26):4053-64. [PMID: 21824087 DOI: 10.2174/092986711796957257]
- Anitha M, Nandhu MS, Anju TR, Jes P, Paulose CS. Targeting glutamate mediated excitotoxicity in Huntington’s disease: neural progenitors and partial glutamate antagonist–memantine. Med Hypotheses 2011;76(1):138-40. [PMID: 20943326 DOI: 10.1016/j.mehy.2010.09.003]
- Singh K, Jain D, Sethi P, Gupta JK, Tripathi AK, et al. Emerging pharmacological approaches for Huntington’s disease. Eur J Pharmacol 2024;980:176873. [PMID: 39117264 DOI: 10.1016/j.ejphar.2024.176873]
- Choi Y, Kim H-S, Shin KY, Kim E-M, Kim M, et al. Minocycline attenuates neuronal cell death and improves cognitive impairment in Alzheimer’s disease models. Neuropsychopharmacology 2007;32(11):2393-404. [PMID: 17406652 DOI: 10.1038/sj.npp.1301377]
- Talebi M, Talebi M, Kakouri E, Farkhondeh T, Pourbagher-Shahri AM, et al. Tantalizing role of p53 molecular pathways and its coherent medications in neurodegenerative diseases. Int J Biol Macromol 2021;172:93-103. [PMID: 33440210 DOI: 10.1016/j.ijbiomac.2021.01.042]
- Ayyalasomayajula N, Suresh C. Mechanistic comparison of current pharmacological treatments and novel phytochemicals to target amyloid peptides in Alzheimer’s and neurodegenerative diseases. Nutr Neurosci 2018;21(10):682-94. [PMID: 28683598 DOI: 10.1080/1028415X.2017.1345425]
- Cheng M, Yuan C, Ju Y, Liu Y, Shi B, et al. Quercetin attenuates oxidative stress and apoptosis in brain tissue of APP/PS1 double transgenic AD mice by regulating Keap1/Nrf2/HO-1 pathway to improve cognitive impairment. Behav Neurol 2024;2024:5698119. [PMID: 39233848 DOI: 10.1155/2024/5698119]
- Cheng S-C, Huang W-C, Pang J-HS, Wu Y-H, Cheng C-Y. Quercetin inhibits the production of IL-1β-induced inflammatory cytokines and chemokines in ARPE-19 cells via the MAPK and NF-κB signaling pathways. Int J Mol Sci 2019;20(12):2957. [PMID: 31212975 DOI: 10.3390/ijms20122957]
- Ebrahimpour S, Zakeri M, Esmaeili A. Crosstalk between obesity, diabetes, and Alzheimer’s disease: introducing quercetin as an effective triple herbal medicine. Ageing Res Rev 2020;62:101095. [PMID: 32535272 DOI: 10.1016/j.arr.2020.101095]
- Chakraborty S, Vishwas S, Harish V, Gupta G, Paudel KR, et al. Exploring nanoparticular platform in delivery of repurposed drug for Alzheimer’s disease: current approaches and future perspectives. Expert Opin Drug Deliv 2024;21(12):1771-92. [PMID: 39397403 DOI: 10.1080/17425247.2024.2414768]
- Sabogal-Guáqueta AM, Muñoz-Manco JI, Ramírez-Pineda JR, Lamprea-Rodriguez M, Osorio E, et al. The flavonoid quercetin ameliorates Alzheimer’s disease pathology and protects cognitive and emotional function in aged triple transgenic Alzheimer’s disease model mice. Neuropharmacology 2015;93:134-45. [PMID: 25666032 DOI: 10.1016/j.neuropharm.2015.01.027]
- Jiang W, Luo T, Li S, Zhou Y, Shen X-Y, et al. Quercetin protects against okadaic acid-induced injury via MAPK and PI3K/Akt/GSK3β signaling pathways in HT22 hippocampal neurons. PLoS One 2016;11(4):e0152371. [PMID: 27050422 DOI: 10.1371/journal.pone.0152371]
- Saeedi-Boroujeni A, Mahmoudian-Sani M-R. Anti-inflammatory potential of Quercetin in COVID-19 treatment. J Inflamm (Lond) 2021;18(1):3. [PMID: 33509217 DOI: 10.1186/s12950-021-00268-6]
- Zu G, Sun K, Li L, Zu X, Han T, et al. Mechanism of quercetin therapeutic targets for Alzheimer disease and type 2 diabetes mellitus. Sci Rep 2021;11(1):22959. [PMID: 34824300 DOI: 10.1038/s41598-021-02248-5]
- Rao AV, Balachandran B. Role of oxidative stress and antioxidants in neurodegenerative diseases. Nutr Neurosci 2002;5(5):291-309. [PMID: 12385592 DOI: 10.1080/1028415021000033767]
- Vila M. Neuromelanin, aging, and neuronal vulnerability in Parkinson’s disease. Mov Disord 2019;34(10):1440-51. [PMID: 31251435 DOI: 10.1002/mds.27776]
- Saha S, Buttari B, Profumo E, Tucci P, Saso L. A perspective on Nrf2 signaling pathway for neuroinflammation: a potential therapeutic target in Alzheimer’s and Parkinson’s diseases. Front Cell Neurosci 2022;15:787258. [PMID: 35126058 DOI: 10.3389/fncel.2021.787258]
- Subramaniam SR, Chesselet M-F. Mitochondrial dysfunction and oxidative stress in Parkinson’s disease. Prog Neurobiol 2013;106-107:17-32. [PMID: 23643800 DOI: 10.1016/j.pneurobio.2013.04.004]
- Wang W-W, Han R, He H-J, Li J, Chen S-Y, et al. Administration of quercetin improves mitochondria quality control and protects the neurons in 6-OHDA-lesioned Parkinson’s disease models. Aging (Albany NY) 2021;13(8):11738-51. [PMID: 33878030 DOI: 10.18632/aging.202868]
- Sriraksa N, Wattanathorn J, Muchimapura S, Tiamkao S, Brown K, et al. Cognitive-enhancing effect of quercetin in a rat model of Parkinson’s disease induced by 6-hydroxydopamine. Evid Based Complement Alternat Med 2012;2012:823206. [PMID: 21792372 DOI: 10.1155/2012/823206]
- Khan MM, Raza SS, Javed H, Ahmad A, Khan A, et al. Rutin protects dopaminergic neurons from oxidative stress in an animal model of Parkinson’s disease. Neurotox Res 2012;22(1):1-15. [PMID: 22194158 DOI: 10.1007/s12640-011-9295-2]
- Acıkara OB, Karatoprak GŞ, Yücel Ç, Akkol EK, Sobarzo-Sánchez E, et al. A critical analysis of quercetin as the attractive target for the treatment of Parkinson’s disease. CNS Neurol Disord Drug Targets 2022;21(9):795-817. [PMID: 34872486 DOI: 10.2174/1871527320666211206122407]
- Iova O-M, Marin G-E, Lazar I, Stanescu I, Dogaru G, et al. Nitric oxide/nitric oxide synthase system in the pathogenesis of neurodegenerative disorders-an overview. Antioxidants (Basel) 2023;12(3):753. [PMID: 36979000 DOI: 10.3390/antiox12030753]
- Josiah SS, Famusiwa CD, Crown OO, Lawal AO, Olaleye MT, et al. Neuroprotective effects of catechin and quercetin in experimental Parkinsonism through modulation of dopamine metabolism and expression of IL-1β, TNF-α, NF-κB, IκKB, and p53 genes in male Wistar rats. Neurotoxicology 2022;90:158-71. [PMID: 35337893 DOI: 10.1016/j.neuro.2022.03.004]
- Henríquez G, Gomez A, Guerrero E, Narayan M. Potential role of natural polyphenols against protein aggregation toxicity: in vitro, in vivo, and clinical studies. ACS Chem Neurosci 2020;11(19):2915-34. [PMID: 32822152 DOI: 10.1021/acschemneuro.0c00381]
- Tan S, Caparros-Martin JA, Matthews VB, Koch H, O’Gara F, et al. Isoquercetin and inulin synergistically modulate the gut microbiome to prevent development of the metabolic syndrome in mice fed a high fat diet. Sci Rep 2018;8(1):10100. [PMID: 29973701 DOI: 10.1038/s41598-018-28521-8]
- Lum PT, Sekar M, Gan SH, Bonam SR, Shaikh MF. Protective effect of natural products against Huntington’s disease: an overview of scientific evidence and understanding their mechanism of action. ACS Chem Neurosci 2021;12(3):391-418. [PMID: 33475334 DOI: 10.1021/acschemneuro.0c00824]
- Riche K, Lenard NR. Quercetin’s effects on glutamate cytotoxicity. Molecules 2022;27(21):7620. [PMID: 36364448 DOI: 10.3390/molecules27217620]
- Silvestro S, Bramanti P, Mazzon E. Role of quercetin in depressive-like behaviors: findings from animal models. Appl Sci 2021;11(15):7116. [DOI: 10.3390/app11157116]
- Vishwas S, Kumar R, Khursheed R, Ramanunny AK, Kumar R, et al. Expanding arsenal against neurodegenerative diseases using quercetin based nanoformulations: breakthroughs and bottlenecks. Curr Neuropharmacol 2023;21(7):1558-74. [PMID: 35950245 DOI: 10.2174/1570159X20666220810105421]
- Ryu H, Rosas HD, Hersch SM, Ferrante RJ. The therapeutic role of creatine in Huntington’s disease. Pharmacol Ther 2005;108(2):193-207. [PMID: 16055197 DOI: 10.1016/j.pharmthera.2005.04.008]
- Kuhad A, Singla S, Arora V, Chopra K. Neuroprotective effect of sesamol and quercetin against QA induced neurotoxicity: an experimental paradigm of Huntington’s disease. J Neurol Sci 2013;333:e149-50. [DOI: 10.1016/j.jns.2013.07.498]
- Tayab MA, Islam MN, Chowdhury KAA, Tasnim FM. Targeting neuroinflammation by polyphenols: a promising therapeutic approach against inflammation-associated depression. Biomed Pharmacother 2022;147:112668. [PMID: 35104696 DOI: 10.1016/j.biopha.2022.112668]
- Sandhir R, Mehrotra A. Quercetin supplementation is effective in improving mitochondrial dysfunctions induced by 3-nitropropionic acid: implications in Huntington’s disease. Biochim Biophys Acta 2013;1832(3):421-30. [PMID: 23220257 DOI: 10.1016/j.bbadis.2012.11.018]
- Kumar A, Ratan RR. Oxidative stress and Huntington’s disease: the good, the bad, and the ugly. J Huntingtons Dis 2016;5(3):217-37. [PMID: 27662334 DOI: 10.3233/JHD-160205]
- Khan H, Ullah H, Tundis R, Belwal T, Devkota HR, et al. Dietary flavonoids in the management of Huntington’s disease: mechanism and clinical perspective eFood 2020;1(1):38-52. [DOI: 10.2991/efood.k.200203.001]
- Gao C, Jiang J, Tan Y, Chen S. Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal Transduct Target Ther 2023;8(1):359. [PMID: 37735487 DOI: 10.1038/s41392-023-01588-0]
- Kalonia H, Mishra J, Kumar A. Targeting neuro-inflammatory cytokines and oxidative stress by minocycline attenuates quinolinic-acid-induced Huntington’s disease-like symptoms in rats. Neurotox Res 2012;22(4):310-20. [PMID: 22392362 DOI: 10.1007/s12640-012-9315-x]
- Moré MI. Taxifolin from dahurian larch – application for the approval as novel food. Amur District, Russia; 2010.
- Yu X, Ji C, Shao A. Neurovascular unit dysfunction and neurodegenerative disorders. Front Neurosci 2020;14:334. [PMID: 32410936 DOI: 10.3389/fnins.2020.00334]
- Liu R, Zhang TT, Zhou D, Bai XY, Zhou WL, et al. Quercetin protects against the Aβ(25-35)-induced amnesic injury: involvement of inactivation of rage-mediated pathway and conservation of the NVU. Neuropharmacology 2013;67:419-31. [PMID: 23231807 DOI: 10.1016/j.neuropharm.2012.11.018]
- Sun P, Yang Y, Yang L, Qian Y, Liang M, et al. Quercetin protects blood-brain barrier integrity via the PI3K/Akt/Erk signaling pathway in a mouse model of meningitis induced by Glaesserella parasuis. Biomolecules 2024;14(6):696. [PMID: 38927100 DOI: 10.3390/biom14060696]
- Dagher O, Mury P, Thorin-Trescases N, Noly PE, Thorin E, et al. Therapeutic potential of quercetin to alleviate endothelial dysfunction in age-related cardiovascular diseases. Front Cardiovasc Med 2021;8:658400. [PMID: 33860002 DOI: 10.3389/fcvm.2021.658400]
- Li L, Jiang W, Yu B, Liang H, Mao S, et al. Quercetin improves cerebral ischemia/reperfusion injury by promoting microglia/macrophages M2 polarization via regulating PI3K/Akt/NF-κB signaling pathway. Biomed Pharmacother 2023;168:115653. [PMID: 37812891 DOI: 10.1016/j.biopha.2023.115653]
- Kobuchi H, Roy S, Sen CK, Nguyen HG, Packer L. Quercetin inhibits inducible ICAM-1 expression in human endothelial cells through the JNK pathway. Am J Physiol 1999;277(3):C403-11. [PMID: 10484327 DOI: 10.1152/ajpcell.1999.277.3.C403]
- Chen T, Zhang X, Zhu G, Liu H, Chen J, et al. Quercetin inhibits TNF-α induced HUVECs apoptosis and inflammation via downregulating NF-kB and AP-1 signaling pathway in vitro. Medicine (Baltimore) 2020;99(38):e22241. [PMID: 32957369 DOI: 10.1097/MD.0000000000022241]
- Zhang Y, Fu K, Wang C, Ma C, Gong L, et al. Protective effects of dietary quercetin on cerebral ischemic injury: pharmacology, pharmacokinetics and bioavailability-enhancing nanoformulations. Food Funct 2023;14(10):4470-89. [PMID: 37067239 DOI: 10.1039/d2fo03122a]
- Silva Dos Santos J, Gonçalves Cirino JP, de Oliveira Carvalho P, Ortega MM. The pharmacological action of kaempferol in central nervous system diseases: a review. Front Pharmacol 2021;11:565700. [PMID: 33519431 DOI: 10.3389/fphar.2020.565700]
- Alexander C, Parsaee A, Vasefi M. Polyherbal and multimodal treatments: kaempferol- and quercetin-rich herbs alleviate symptoms of Alzheimer’s disease. Biology (Basel) 2023;12(11):1453. [PMID: 37998052 DOI: 10.3390/biology12111453]
- Zhang J-X, Xing J-G, Wang L-L, Jiang H-L, Guo S-L, et al. Luteolin inhibits fibrillary β-amyloid1-40-induced inflammation in a human blood-brain barrier model by suppressing the p38 MAPK-mediated NF-κB signaling pathways. Molecules 2017;22(3):334. [PMID: 28245546 DOI: 10.3390/molecules22030334]
- Gonçalves PB, Sodero ACR, Cordeiro Y. Green tea epigallocatechin-3-gallate (EGCG) targeting protein misfolding in drug discovery for neurodegenerative diseases. Biomolecules 2021;11(5):767. [PMID: 34065606 DOI: 10.3390/biom11050767]
- Nargeh H, Aliabadi F, Ajami M, Pazoki-Toroudi H. Role of polyphenols on gut microbiota and the ubiquitin-proteasome system in neurodegenerative diseases. J Agric Food Chem 2021;69(22):6119-44. [PMID: 34038102 DOI: 10.1021/acs.jafc.1c00923]
- Maan G, Sikdar B, Kumar A, Shukla R, Mishra A. Role of flavonoids in neurodegenerative diseases: limitations and future perspectives. Curr Top Med Chem 2020;20(13):1169-94. [PMID: 32297582 DOI: 10.2174/1568026620666200416085330]
- Liang H, Ma Z, Zhong W, Liu J, Sugimoto K, et al. Regulation of mitophagy and mitochondrial function: natural compounds as potential therapeutic strategies for Parkinson’s disease. Phytother Res 2024;38(4):1838-62. [PMID: 38356178 DOI: 10.1002/ptr.8156]
- Li Y, Zhao J, Hölscher C. Therapeutic potential of baicalein in Alzheimer’s disease and Parkinson’s disease. CNS Drugs 2017;31(8):639-52. [PMID: 28634902 DOI: 10.1007/s40263-017-0451-y]
- Kunachowicz D, Ściskalska M, Kepinska M. Modulatory effect of lifestyle-related, environmental and genetic factors on paraoxonase-1 activity: a review. Int J Environ Res Public Health 2023;20(4):2813. [PMID: 36833509 DOI: 10.3390/ijerph20042813]
- Sreekumar PG, Su F, Spee C, Hong E, Komirisetty R, et al. Paraoxonase 2 deficiency causes mitochondrial dysfunction in retinal pigment epithelial cells and retinal degeneration in mice. Antioxidants (Basel) 2023;12(10):1820. [PMID: 37891899 DOI: 10.3390/antiox12101820]
- Altenhöfer S, Witte I, Teiber JF, Wilgenbus P, Pautz A, et al. One enzyme, two functions: PON2 prevents mitochondrial superoxide formation and apoptosis independent from its lactonase activity. J Biol Chem 2010;285(32):24398-403. [PMID: 20530481 DOI: 10.1074/jbc.M110.118604]
- Devarajan A, Su F, Grijalva V, Yalamanchi M, Yalamanchi A, et al. Paraoxonase 2 overexpression inhibits tumor development in a mouse model of ovarian cancer. Cell Death Dis 2018;9(3):392. [PMID: 29531225 DOI: 10.1038/s41419-018-0395-2]
- Garrick JM, Dao K, Costa LG, Marsillach J, Furlong CE. Examining the role of paraoxonase 2 in the dopaminergic system of the mouse brain. BMC Neurosci 2022;23(1):52. [PMID: 36056313 DOI: 10.1186/s12868-022-00738-4]
- Giordano G, Cole TB, Furlong CE, Costa LG. Paraoxonase 2 (PON2) in the mouse central nervous system: a neuroprotective role? Toxicol Appl Pharmacol 2011;256(3):369-78. [PMID: 21354197 DOI: 10.1016/j.taap.2011.02.014]
- Costa LG, de Laat R, Dao K, Pellacani C, Cole TB, et al. Paraoxonase-2 (PON2) in brain and its potential role in neuroprotection. Neurotoxicology 2014;43:3-9. [PMID: 24012887 DOI: 10.1016/j.neuro.2013.08.011]
- Farid AS, Horii Y. Modulation of paraoxonases during infectious diseases and its potential impact on atherosclerosis. Lipids Health Dis 2012;11:92. [PMID: 22824324 DOI: 10.1186/1476-511X-11-92]
- Parween F, Gupta RD. Insights into the role of paraoxonase 2 in human pathophysiology. J Biosci 2022;47(1):4. [PMID: 35092416 DOI: 10.1007/s12038-021-00234-7]
- Romero M, Jiménez R, Sánchez M, López-Sepúlveda R, Zarzuelo MJ, et al. Quercetin inhibits vascular superoxide production induced by endothelin-1: role of NADPH oxidase, uncoupled eNOS and PKC. Atherosclerosis 2009;202(1):58-67. [PMID: 18436224 DOI: 10.1016/j.atherosclerosis.2008.03.007]
- Oakley RH, Cidlowski JA. The biology of the glucocorticoid receptor: new signaling mechanisms in health and disease. J Allergy Clin Immunol 2013;132(5):1033-44. [PMID: 24084075 DOI: 10.1016/j.jaci.2013.09.007]
- Loboda A, Damulewicz M, Pyza E, Jozkowicz A, Dulak J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell Mol Life Sci 2016;73(17):3221-47. [PMID: 27100828 DOI: 10.1007/s00018-016-2223-0]
- Rai SR, Bhattacharyya C, Sarkar A, Chakraborty S, Sircar E, et al. Glutathione: role in oxidative/nitrosative stress, antioxidant defense, and treatments. ChemistrySelect 2021;6(18):4566-90. [DOI: 10.1002/slct.202100773]
- Kanninen K, Malm TM, Jyrkkänen H-K, Goldsteins G, Keksa-Goldsteine V, et al. Nuclear factor erythroid 2-related factor 2 protects against beta amyloid. Mol Cell Neurosci 2008;39(3):302-13. [PMID: 18706502 DOI: 10.1016/j.mcn.2008.07.010]
- Liang L, Gao C, Luo M, Wang W, Zhao C, et al. Dihydroquercetin (DHQ) induced HO-1 and NQO1 expression against oxidative stress through the Nrf2-dependent antioxidant pathway. J Agric Food Chem 2013;61(11):2755-61. [PMID: 23419114 DOI: 10.1021/jf304768p]
- Saw CLL, Guo Y, Yang AY, Paredes-Gonzalez X, Ramirez C, et al. The berry constituents quercetin, kaempferol, and pterostilbene synergistically attenuate reactive oxygen species: involvement of the Nrf2-ARE signaling pathway. Food Chem Toxicol 2014;72:303-11. [PMID: 25111660 DOI: 10.1016/j.fct.2014.07.038]
- Dalle S, Abderrahmani A. Receptors and signaling pathways controlling beta-cell function and survival as targets for anti-diabetic therapeutic strategies. Cells 2024;13(15):1244. [PMID: 39120275 DOI: 10.3390/cells13151244]
- Danielsen SA, Eide PW, Nesbakken A, Guren T, Leithe E, et al. Portrait of the PI3K/AKT pathway in colorectal cancer. Biochim Biophys Acta 2015;1855(1):104-21. [PMID: 25450577 DOI: 10.1016/j.bbcan.2014.09.008]
- Penna E, Pizzella A, Cimmino F, Trinchese G, Cavaliere G, et al. Neurodevelopmental disorders: effect of high-fat diet on synaptic plasticity and mitochondrial functions. Brain Sci 2020;10(11):805. [PMID: 33142719 DOI: 10.3390/brainsci10110805]
- Dong YS, Wang JL, Feng DY, Qin HZ, Wen H, et al. Protective effect of quercetin against oxidative stress and brain edema in an experimental rat model of subarachnoid hemorrhage. Int J Med Sci 2014;11(3):282-90. [PMID: 24516353 DOI: 10.7150/ijms.7634]
- da Silva MF, Lins AA, Gomes MC, Marinho WPJ, de Araújo RSA, et al. Anticancer drug discovery from natural compounds targeting PI3K/AKT/mTOR signaling pathway. Curr Med Chem 2024. [PMID: 39390838 DOI: 10.2174/0109298673325229240928040758]
- Yan H, He L, Lv D, Yang J, Yuan Z. The role of the dysregulated JNK signaling pathway in the pathogenesis of human diseases and its potential therapeutic strategies: a comprehensive review. Biomolecules 2024;14(2):243. [PMID: 38397480 DOI: 10.3390/biom14020243]
- Garg R, Kumariya S, Katekar R, Verma S, Goand UK, et al. JNK signaling pathway in metabolic disorders: an emerging therapeutic target. Eur J Pharmacol 2021;901:174079. [PMID: 33812885 DOI: 10.1016/j.ejphar.2021.174079]
- Ha J, Kang E, Seo J, Cho S. Phosphorylation dynamics of JNK signaling: effects of dual-specificity phosphatases (DUSPs) on the JNK pathway. Int J Mol Sci 2019;20(24):6157. [PMID: 31817617 DOI: 10.3390/ijms20246157]
- Zaplatic E, Bule M, Shah SZA, Uddin MS, Niaz K. Molecular mechanisms underlying protective role of quercetin in attenuating Alzheimer’s disease. Life Sci 2019;224:109-19. [PMID: 30914316 DOI: 10.1016/j.lfs.2019.03.055]
- Dhanasekaran DN, Reddy EP. JNK signaling in apoptosis. Oncogene 2008;27(48):6245-51. [PMID: 18931691 DOI: 10.1038/onc.2008.301]
- Uchida K, Shiraishi M, Naito Y, Torii Y, Nakamura Y, et al. Activation of stress signaling pathways by the end product of lipid peroxidation. 4-hydroxy-2-nonenal is a potential inducer of intracellular peroxide production. J Biol Chem 1999;274(4):2234-42. [PMID: 9890986 DOI: 10.1074/jbc.274.4.2234]
- Merecz-Sadowska A, Sitarek P, Śliwiński T, Zajdel R. Anti-inflammatory activity of extracts and pure compounds derived from plants via modulation of signaling pathways, especially PI3K/AKT in macrophages. Int J Mol Sci 2020;21(24):9605. [PMID: 33339446 DOI: 10.3390/ijms21249605]
- Megha KB, Joseph X, Akhil V, Mohanan PV. Cascade of immune mechanism and consequences of inflammatory disorders. Phytomedicine 2021;91:153712. [PMID: 34511264 DOI: 10.1016/j.phymed.2021.153712]
- Abidar S, Hritcu L, Nhiri M. The natural neuroprotective compounds used in the 6-hydroxydopamine- induced Parkinson’s disease in zebrafish: the current applications and perspectives. CNS Neurol Disord Drug Targets 2023;22(10):1472-83. [PMID: 36306449 DOI: 10.2174/1871527322666221028152600]
- Suganthy N, Devi KP, Nabavi SF, Braidy N, Nabavi SM. Bioactive effects of quercetin in the central nervous system: focusing on the mechanisms of actions. Biomed Pharmacother 2016;84:892-908. [PMID: 27756054 DOI: 10.1016/j.biopha.2016.10.011]
- Isik S, Yeman Kiyak B, Akbayir R, Seyhali R, Arpaci T. Microglia mediated neuroinflammation in Parkinson’s disease. Cells 2023;12(7):1012. [PMID: 37048085 DOI: 10.3390/cells12071012]
- Zamin LL, Filippi-Chiela EC, Dillenburg-Pilla P, Horn F, Salbego C, et al. Resveratrol and quercetin cooperate to induce senescence-like growth arrest in C6 rat glioma cells. Cancer Sci 2009;100(9):1655-62. [PMID: 19496785 DOI: 10.1111/j.1349-7006.2009.01215.x]
- Benameur T, Soleti R, Porro C. The potential neuroprotective role of free and encapsulated quercetin mediated by miRNA against neurological diseases. Nutrients 2021;13(4):1318. [PMID: 33923599 DOI: 10.3390/nu13041318]
- Qian L, Zhu Y, Deng C, Liang Z, Chen J, et al. Peroxisome proliferator-activated receptor gamma coactivator-1 (PGC-1) family in physiological and pathophysiological process and diseases. Signal Transduct Target Ther 2024;9(1):50. [PMID: 38424050 DOI: 10.1038/s41392-024-01756-w]
- Song J, Du G, Wu H, Gao X, Yang Z, et al. Protective effects of quercetin on traumatic brain injury induced inflammation and oxidative stress in cortex through activating Nrf2/HO-1 pathway. Restor Neurol Neurosci 2021;39(1):73-84. [PMID: 33612499 DOI: 10.3233/RNN-201119]
- Dong W, Quo W, Wang F, Li C, Xie Y, et al. Electroacupuncture upregulates SIRT1-dependent PGC-1α expression in SAMP8 mice. Med Sci Monit 2015;21:3356-62. [PMID: 26530101 DOI: 10.12659/msm.894864]
- Wang R, Li JJ, Diao S, Kwak Y-D, Liu L, et al. Metabolic stress modulates Alzheimer’s β-secretase gene transcription via SIRT1-PPARγ-PGC-1 in neurons. Cell Metab 2013;17(5):685-94. [PMID: 23663737 DOI: 10.1016/j.cmet.2013.03.016]
- Kratz EM, Sołkiewicz K, Kubis-Kubiak A, Piwowar A. Sirtuins as important factors in pathological states and the role of their molecular activity modulators. Int J Mol Sci 2021;22(2):630. [PMID: 33435263 DOI: 10.3390/ijms22020630]
- Nakagawa T, Ohta K. Quercetin regulates the integrated stress response to improve memory. Int J Mol Sci 2019;20(11):2761. [PMID: 31195662 DOI: 10.3390/ijms20112761]
- Yue J, López JM. Understanding MAPK signaling pathways in apoptosis. Int J Mol Sci 2020;21(7):2346. [PMID: 32231094 DOI: 10.3390/ijms21072346]
- Moratilla-Rivera I, Sánchez M, Valdés-González JA, Gómez-Serranillos MP. Natural products as modulators of Nrf2 signaling pathway in neuroprotection. Int J Mol Sci 2023;24(4):3748. [PMID: 36835155 DOI: 10.3390/ijms24043748]
- Islam MA, Medha MM, Nahar AU, Al Fahad MA, Siraj MA, et al. Cancer protective role of selected dietary polyphenols via modulating Keap1/Nrf2/ARE and interconnected signaling pathways. Nutr Cancer 2023;75(4):1065-102. [PMID: 37078744 DOI: 10.1080/01635581.2023.2183546]
- Gravandi MM, Fakhri S, Zarneshan SN, Yarmohammadi A, Khan H. Flavonoids modulate AMPK/PGC-1α and interconnected pathways toward potential neuroprotective activities. Metab Brain Dis 2021;36(7):1501-21. [PMID: 33988807 DOI: 10.1007/s11011-021-00750-3]
- Rullah K, Shamsudin NF, Koeberle A, Tham CL, Fasihi Mohd Aluwi MF, et al. Flavonoid diversity and roles in the lipopolysaccharide-mediated inflammatory response of monocytes and macrophages. Future Med Chem 2024;16(1):75-99. [PMID: 38205612 DOI: 10.4155/fmc-2023-0174]
- Habtemariam S. Anti-inflammatory therapeutic mechanisms of natural products: insight from rosemary diterpenes, carnosic acid and carnosol. Biomedicines 2023;11(2):545. [PMID: 36831081 DOI: 10.3390/biomedicines11020545]
- Islam MR, Al-Imran MIK, Zehravi M, Sweilam SH, Mortuza MR, et al. Targeting signaling pathways in neurodegenerative diseases: quercetin’s cellular and molecular mechanisms for neuroprotection. Animal Model Exp Med 2025;8(5):798-818. [PMID: 39843406 DOI: 10.1002/ame2.12551]
- Zarneshan SN, Fakhri S, Khan H. Targeting Akt/CREB/BDNF signaling pathway by ginsenosides in neurodegenerative diseases: a mechanistic approach. Pharmacol Res 2022;177:106099. [PMID: 35092819 DOI: 10.1016/j.phrs.2022.106099]
- Wolf D, Muralidharan A, Mohan S. Role of prolyl hydroxylase domain proteins in bone metabolism. Osteoporos Sarcopenia 2022;8(1):1-10. [PMID: 35415275 DOI: 10.1016/j.afos.2022.03.001]
- Cai H, Yang J, Wang J-N, Gong Y, Dai H-Z, Chen G-J, et al. Quercetin alleviates cerebral ischemia/reperfusion injury by regulating Tnf-1a/Hif-1α activation of Vegfa/Vegfr/Akt/Pi3k pathway. 2024. [DOI: 10.2139/ssrn.4997549]
- Zhang L, Ma J, Yang F, Li S, Ma W, et al. Neuroprotective effects of quercetin on ischemic stroke: a literature review. Front Pharmacol 2022;13:854249. [PMID: 35662707 DOI: 10.3389/fphar.2022.854249]
- Su K, Li Z, Yu Y, Zhang X. The prolyl hydroxylase inhibitor roxadustat: paradigm in drug discovery and prospects for clinical application beyond anemia. Drug Discov Today 2020;25(7):1262-9. [PMID: 32380083 DOI: 10.1016/j.drudis.2020.04.017]
- Miao M, Wu M, Li Y, Zhang L, Jin Q, et al. Clinical potential of hypoxia inducible factors prolyl hydroxylase inhibitors in treating nonanemic diseases. Front Pharmacol 2022;13:837249. [PMID: 35281917 DOI: 10.3389/fphar.2022.837249]
- Patil N, Bhatt LK. Targeting Acyl-CoA synthetase long-chain family member 4: a potential approach for the treatment of cerebral ischemia/reperfusion injury. Metab Brain Dis 2025;40(5):212. [PMID: 40418418 DOI: 10.1007/s11011-025-01638-2]
- Alqahtani F, Mohamed Ali YS, Almutairi MM, Alotaibi AF, Imran I, et al. Therapeutic benefits of quercetin in traumatic brain injury model exposed to cigarette smoke. Saudi Pharm J 2024;32(1):101895. [PMID: 38226352 DOI: 10.1016/j.jsps.2023.101895]
- Gao Y, Zhang J, Tang T, Liu Z. Hypoxia pathways in Parkinson’s disease: from pathogenesis to therapeutic targets. Int J Mol Sci 2024;25(19):10484. [PMID: 39408813 DOI: 10.3390/ijms251910484]
- Jha NK, Jha SK, Sharma R, Kumar D, Ambasta RK, et al. Hypoxia-induced signaling activation in neurodegenerative diseases: targets for new therapeutic strategies. J Alzheimers Dis 2018;62(1):15-38. [PMID: 29439330 DOI: 10.3233/JAD-170589]
- Ossola B, Kääriäinen TM, Männistö PT. The multiple faces of quercetin in neuroprotection. Expert Opin Drug Saf 2009;8(4):397-409. [PMID: 19538101 DOI: 10.1517/14740330903026944]
- Hashemzaei M, Delarami Far A, Yari A, Heravi RE, Tabrizian K, et al. Anticancer and apoptosis-inducing effects of quercetin in vitro and in vivo. Oncol Rep 2017;38(2):819-28. [PMID: 28677813 DOI: 10.3892/or.2017.5766]
- Zhao P, Mao J-M, Zhang S-Y, Zhou Z-Q, Tan Y, et al. Quercetin induces HepG2 cell apoptosis by inhibiting fatty acid biosynthesis. Oncol Lett 2014;8(2):765-9. [PMID: 25009654 DOI: 10.3892/ol.2014.2159]
- Sonmez E, Cacciatore I, Bakan F, Turkez H, Mohtar YI, et al. Toxicity assessment of hydroxyapatite nanoparticles in rat liver cell model in vitro. Hum Exp Toxicol 2016;35(10):1073-83. [PMID: 26655636 DOI: 10.1177/0960327115619770]
- Zhang X-Y, Li Y-Q, Yin Z-H, Bao Q-N, Xia M-Z, et al. Supplements for cognitive ability in patients with mild cognitive impairment or Alzheimer’s disease: a protocol for systematic review and network meta-analysis of randomised controlled trials. BMJ Open 2024;14(4):e077623. [PMID: 38569691 DOI: 10.1136/bmjopen-2023-077623]
- Bayazid AB, Lim BO. Quercetin is an active agent in berries against neurodegenerative diseases progression through modulation of Nrf2/HO1. Nutrients 2022;14(23):5132. [PMID: 36501161 DOI: 10.3390/nu14235132]
- Lama A, Pirozzi C, Avagliano C, Annunziata C, Mollica MP, et al. Nutraceuticals: an integrative approach to starve Parkinson’s disease. Brain Behav Immun Health 2020;2:100037. [PMID: 34589828 DOI: 10.1016/j.bbih.2020.100037]