ACE2: A Dilemma in Regulating SARS-CoV-2 Infection and its Metabolic Complications
1Obesity and Metabolic Disease Research Group, Diabetes and Metabolism Division, Garvan Institute of Medical Research, Darlinghurst 2010, Sydney, Australia
2St Vincent’s Clinical School, UNSW Sydney, Australia
*Correspondence to: Yan-Chuan Shi, E-mail: y.shi@garvan.org.au
Received: October 9 2022; Accepted: October 18 2022; Published Online: December 19 2022
Cite this paper:
Shi YC. ACE2: A Dilemma in Regulating SARS-CoV-2 Infection and its Metabolic Complications. BIO Integration 2023; 4(1): 3–6.
DOI: 10.15212/bioi-2022-0026. Available at: https://bio-integration.org/
Download citation
© 2023 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
“This commentary is written on the basis of a recent publication in Cell Metabolism by BIO Integration Editorial Board Member Dr. Sifan Chen and colleagues, entitled “Imatinib and methazolamide ameliorate COVID-19-induced metabolic complications via elevating ACE2 enzymatic activity and inhibiting viral entry.”
Obesity and diabetes are major risk factors for severe SARS-CoV-2 infection, ICU admission and COVID-19-associated death [1]. Increasing evidence also suggests that SARS-CoV-2 infection disrupts glucose and lipid metabolism, promotes insulin resistance and endothelial dysfunction, and eventually may result in long-term metabolic defects and cardiovascular complications [2, 3]. However, the specific mechanisms through which SARS-CoV-2 infection drives these unfavorable metabolic events, and the optimal treatment strategies, remain elusive. After the SARS-CoV-2 virus enters the lungs, the viral spike glycoprotein binds angiotensin converting enzyme 2 (ACE2) on the cell surface, thus allowing the virus to enter the host cells, and subsequently causing an array of damage in the lungs and multiple other organs. Current mainstay COVID-19 treatments focus on decreasing ACE2 levels to limit viral entry via 1) human recombinant soluble ACE2, which neutralizes the virus and limits damage from infection, and 2) ACE2 inhibitors and antibodies, which bind ACE2 and prevent interaction between the virus and the receptor. In Issue 34(3) in Cell Metabolism 2022, Li et al. have identified that elevation of ACE2 enzymatic activity by imatinib and methazolamide ameliorates COVID-19-induced metabolic complications; moreover, the blockade of the virus-ACE2 interaction by these two drugs inhibits viral entry [4].
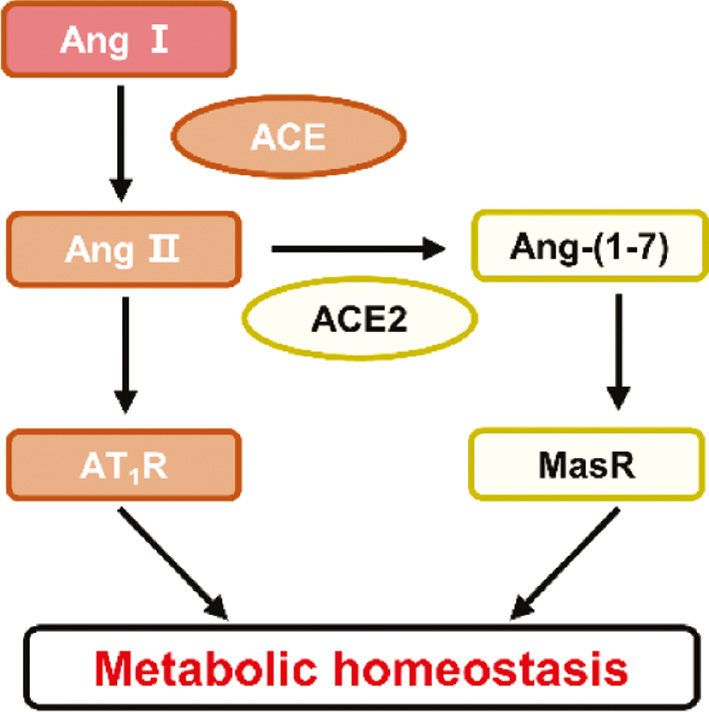
The renin-angiotensin system (RAS) is a crucial system regulating blood pressure and body fluid homeostasis, in which ACE2 plays a key role. Classically, angiotensinogen is converted to angiotensin I (AngI) by renin. ACE subsequently catalyzes the conversion of AngI to angiotensin II (AngII), which in turn exerts effects on various tissues and organs, including the heart, brain and kidneys. ACE2, another central enzyme in the RAS pathway, hydrolyzes AngII into Ang(1–7), which acts on the receptor MasR and counterbalances the effects of AngII in the classical AngII-AT1R axis. Both the ACE-AT1R-AngII and ACE2-Ang(1–7)-MasR axes are important for maintaining fluid balance via the RAS system [5]. However, substantial evidence has indicated that ACE2 is an essential cell entry receptor for SARS-CoV-2 invasion and that elevated ACE2 expression is associated with poor COVID-19 outcomes. For example, Song et al. have demonstrated elevated neuro-invasion of SARS-CoV-2 in mice expressing human ACE2 [6]. Neutralizing antibodies, such as COV2-2196 and COV2-2130, have been found to decrease the risk of infection by blocking the ACE2 receptor-binding domain, and decreasing viral burden and inflammation in the lungs in mice transduced with human ACE2 [7]. These findings strongly indicate that ACE2 plays a major role in SARS-CoV-2 infection, and that decreasing ACE2 levels is beneficial to diminish the risk of SARS-CoV-2 infection, as well as the severity of COVID-19-associated symptoms and complications [8].
Despite aforementioned benefits, however, decreased activity of the ACE2-Ang(1–7)-MasR axis appears to be associated with an increased risk of metabolic disorders. Table 1 lists the roles of this axis in regulating the function of various cells and organs, including pancreatic β-cells [9], the kidneys [10], the liver [11], adipose tissue [12] and the heart [13]. In a meta-analysis including three independent studies on 1471 participants published in 2022, lower ACE2 expression in adipose tissue was associated with adverse cardio-metabolic health indices, such as type 2 diabetes and obesity, as evidenced by higher serum fasting insulin levels and lower serum high-density lipoprotein (HDL) levels [14]. Furthermore, some animal studies have shown that ACE2-knockout mice are susceptible to high-fat-diet-induced pancreatic β-cell dysfunction [15], thus suggesting possible protective effects of ACE2 in metabolic diseases, including diabetes and obesity. Two recent studies further support this notion by showing that ACE2 deficiency impairs islet β-cell function in obese C57BL/6 mice, and accentuates vascular inflammation and atherosclerosis in ApoE-knockout mice [16, 17]. These findings are relevant because people with COVID-19 are at elevated risk of developing metabolic disorders [18, 19]. However, the function of ACE2 remains to be determined in the contexts of COVID-19-induced metabolic complications as well as the subsequent treatment of metabolic disorders [20].
Table 1 Effects of the ACE2-Ang (1–7)-MasR Axis in Various Tissues/Organs
| Tissue/organ | Effects of the ACE2-Ang (1–7)-MasR Axis | Reference |
|---|---|---|
| Pancreas | Ang(1–7) administration attenuates pancreatic β-cell dedifferentiation in mice fed a high-fat diet. | [9] |
| Kidney | Overexpression of human ACE2 in podocytes protects renal function and attenuates the development of diabetic nephropathy in mice. | [10] |
| Liver | Ang(1–7) administration improves glucose and lipid metabolism and mitochondrial function, and decreases fat accumulation in mice fed a high-fat diet. | [11] |
| Adipose tissue | Ang(1–7) treatment increases brown adipose tissue and elevates thermogenesis in subcutaneous white adipose tissue in mice fed a high-fat diet. | [12] |
| Heart | ACE2 overexpression improves left ventricular remodeling and function in diabetic rats. | [13] |
In an exciting breakthrough, Chen’s team has provided key evidence of how ACE2 regulates metabolism, and discovered a new strategy for the treatment of COVID-19 and COVID-19-related metabolic complications. In a previous study, the authors found that COVID-19 can cause new-onset insulin resistance, elevated blood glucose levels and diminished HDL-C levels, all of which persist after viral elimination [21]. The protein expression of ACE2 has been found to be downregulated in lung tissue in hamsters, and vascular endothelial cell function has been found to be impaired after 5 days of intratracheal administration of a pseudovirus expressing the SARS-CoV-2 S protein [22]. In agreement with this observation, Li and colleagues have observed diminished mRNA levels of ACE2 in the kidneys and liver in human ACE2 transgenic mice after a SARS-CoV-2 challenge for 7 days. More importantly, they have reported that overexpression of human ACE2 ameliorates disruptions in glucose and lipid metabolism in diabetic ob/ob mice, as evidenced by 1) improved glucose tolerance and increased insulin sensitivity, as evaluated by the homeostatic model assessment of insulin resistance (HOMA-IR); 2) decreased hepatic steatosis; 3) improved renal function through a decrease in the levels of kidney injury molecule-1 (KIM-1) and rescue of renal glomerular damage; and 4) normalized ventricular ejection fractions and fractional shortening in the heart. Activation of the ACE2-Ang(1–7)-MasR axis may be the most crucial mechanism through which ACE2 activators ameliorate metabolic disorders. Therefore, the authors have proposed that enhancing the enzymatic activity of ACE2 may effectively ameliorate COVID-19-associated metabolic disorders. In search of compounds that potentially activate ACE2 activity, the authors used structure-based high-throughput virtual screening via the Tianhe-2 supercomputer and a connectivity-map database to identify ACE2 enzymatic activators or ACE2 transcriptional activators. After conducting a series of experiments to validate their findings, they discovered that two existing clinical medicines were novel ACE2 enzymatic activators: imatinib and methazolamide. Imatinib is in a class of tyrosine kinase inhibitors used in the treatment of chronic myeloid leukemia [23], and methazolamide is a carbonic anhydrase inhibitor used to treat glaucoma [24]. The proposed mechanisms of action of these two repurposed medications involve 1) activating ACE2 enzymatic activity, thus ameliorating the metabolic complications induced by COVID-19, and 2) altering the conformation of ACE2, thereby decreasing viral invasion and the risk of infection. In addition, because targeting ACE2, a negative regulator of ACE, may have unique advantages in balancing the ACE-AngII-AT1R and ACE2-Ang(1–7)-MasR axes of the RAS system, drug-mediated activation of ACE2 may not only increase Ang(1–7) levels but also decrease AngII levels (Figure 1). This aspect is particularly important, given that a recent study has suggested that decreased activity of the ACE-AngII axis may be metabolically beneficial. In this randomized double-blind study conducted in 96 centers worldwide, irbesartan, an AngII receptor antagonist, has been found to prevent diabetic nephropathy in patients with type 2 diabetes and microalbuminuria independently of its blood-pressure lowering effect [25]. The work from Chen’s team provides evidence for ACE2 as a potential novel target in the treatment of metabolic diseases in which ACE2 expressions is downregulated. More research is required to confirm the benefits of these medications in the context of COVID-19-associated metabolic disorders and to understand the detailed mechanisms.
Figure 1 The ACE-AngII-AT1R axis and ACE2-Ang(1–7)-MasR axis balance each other and maintain metabolic homeostasis.
Of note, concerns exist regarding whether the use of ACE2 agonists (e.g., imatinib and methazolamide) might potentially increase the risk of viral infection or reinfection. Very limited direct evidence is currently available to address this question; however, some indirect evidence from clinical studies indicates that ACE2 agonists might not increase this risk. In a retrospective study, published in 2022, on a national cohort of US veterans, ACEi/ARB medications, which are used to manage hypertension, increased ACE2 expression, but this increase was not associated with elevated COVID-19 severity and mortality [26]. Consequently, COVID-19 treatment guidelines in many countries suggest that patients with hypertension need not discontinue ACEi/ARB medications after SARS-CoV-2 infection. Moreover, Ang(1–7), the downstream product of ACE2, is currently being used to treat COVID-19-induced hyper-inflammation in a clinical trial (NCT04375124), although the results have not been released. However, many key efficacy and safety questions must be addressed before these research findings can be leveraged in clinical applications, and the possibility of using ACE2 agonists to treat metabolic diseases in the general population can be explored. Nevertheless, this study provides a critical basis for further investigation. The identification of ACE2 as a novel therapeutic target holds promise for the prevention and treatment of metabolic diseases, particularly in the context of SARS-CoV-2 infection.
Conflict of Interest
The author has no competing interest to declare.
References
- Sanoudou D, Hill MA, Belanger MJ, Arao K, Mantzoros CS. Editorial: Obesity, metabolic phenotypes and COVID-19. Metabolism 2022;128:155121. [PMID: 35026232 DOI: 10.1016/j.metabol.2021.155121]
- Khunti K, Del Prato S, Mathieu C, Kahn SE, Gabbay RA, et al. COVID-19, Hyperglycemia, and New-Onset Diabetes. Diabetes Care 2021;44:2645-55. [PMID: 34625431 DOI: 10.2337/dc21-1318]
- Perico L, Benigni A, Casiraghi F, Ng LFP, Renia L, et al. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat Rev Nephrol 2021;17:46-64. [PMID: 33077917 DOI: 10.1038/s41581-020-00357-4]
- Li Z, Peng M, Chen P, Liu C, Hu A, et al. Imatinib and methazolamide ameliorate COVID-19-induced metabolic complications via elevating ACE2 enzymatic activity and inhibiting viral entry. Cell Metab 2022;34:424-40.e7. [PMID: 35150639 DOI: 10.1016/j.cmet.2022.01.008]
- Santos RAS, Sampaio WO, Alzamora AC, Motta-Santos D, Alenina N, et al. The ACE2/Angiotensin-(1-7)/MAS Axis of the Renin-Angiotensin System: Focus on Angiotensin-(1-7). Physiol Rev 2018;98:505-53. [PMID: 29351514 DOI: 10.1152/physrev.00023.2016]
- Song E, Zhang C, Israelow B, Lu-Culligan A, Prado AV, et al. Neuroinvasion of SARS-CoV-2 in human and mouse brain. J Exp Med 2021;218:3. [PMID: 33433624 DOI: 10.1084/jem.20202135]
- Zost SJ, Gilchuk P, Case JB, Binshtein E, Chen RE, et al. Potently neutralizing and protective human antibodies against SARS-CoV-2. Nature 2020;584:443-9. [PMID: 32668443 DOI: 10.1038/s41586-020-2548-6]
- Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, et al. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020;181:281-92.e6. [PMID: 32155444 DOI: 10.1016/j.cell.2020.02.058]
- Xuan X, Gao F, Ma X, Huang C, Wang Y, et al. Activation of ACE2/angiotensin (1-7) attenuates pancreatic β cell dedifferentiation in a high-fat-diet mouse model. Metabolism 2018;81:83-96. [PMID: 29225087 DOI: 10.1016/j.metabol.2017.12.003]
- Nadarajah R, Milagres R, Dilauro M, Gutsol A, Xiao F, et al. Podocyte-specific overexpression of human angiotensin-converting enzyme 2 attenuates diabetic nephropathy in mice. Kidney Int 2012;82:292-303. [PMID: 22475818 DOI: 10.1038/ki.2012.83]
- Song LN, Liu JY, Shi TT, Zhang YC, Xin Z, et al. Angiotensin-(1-7), the product of ACE2 ameliorates NAFLD by acting through its receptor Mas to regulate hepatic mitochondrial function and glycolipid metabolism. Faseb J 2020;34:16291-306. [PMID: 33078906 DOI: 10.1096/fj.202001639R]
- Morimoto H, Mori J, Nakajima H, Kawabe Y, Tsuma Y, et al. Angiotensin 1-7 stimulates brown adipose tissue and reduces diet-induced obesity. Am J Physiol Endocrinol Metab 2018;314:E131-E8. [PMID: 29066463 DOI: 10.1152/ajpendo.00192.2017]
- Dong B, Yu QT, Dai HY, Gao YY, Zhou ZL, et al. Angiotensin-converting enzyme-2 overexpression improves left ventricular remodeling and function in a rat model of diabetic cardiomyopathy. J Am Coll Cardiol 2012;59:739-47. [PMID: 22340266 DOI: 10.1016/j.jacc.2011.09.071]
- El-Sayed Moustafa JS, Jackson AU, Brotman SM, Guan L, Villicaña S, et al. ACE2 expression in adipose tissue is associated with cardio-metabolic risk factors and cell type composition-implications for COVID-19. Int J Obes (Lond) 2022;46:1478-86. [PMID: 35589964 DOI: 10.1038/s41366-022-01136-w]
- Lu CL, Wang Y, Yuan L, Li Y, Li XY. The angiotensin-converting enzyme 2/angiotensin (1-7)/Mas axis protects the function of pancreatic β cells by improving the function of islet microvascular endothelial cells. Int J Mol Med 2014;34:1293-300. [PMID: 25175177 DOI: 10.3892/ijmm.2014.1917]
- Shoemaker R, Yiannikouris F, Thatcher S, Cassis L. ACE2 deficiency reduces β-cell mass and impairs β-cell proliferation in obese C57BL/6 mice. Am J Physiol Endocrinol Metab 2015;309:E621-E31. [PMID: 26389599 DOI: 10.1152/ajpendo.00054.2015]
- Thomas MC, Pickering RJ, Tsorotes D, Koitka A, Sheehy K, et al. Genetic Ace2 deficiency accentuates vascular inflammation and atherosclerosis in the ApoE knockout mouse. Circ Res 2010;107:888-97. [PMID: 20671240 DOI: 10.1161/circresaha.110.219279]
- Rathmann W, Kuss O, Kostev K. Incidence of newly diagnosed diabetes after Covid-19. Diabetologia 2022;65:949-54. [PMID: 35292829 DOI: 10.1007/s00125-022-05670-0]
- Xie Y, Al-Aly Z. Risks and burdens of incident diabetes in long COVID: a cohort study. Lancet Diabetes Endocrinol 2022;10:311-21. [PMID: 35325624 DOI: 10.1016/s2213-8587(22)00044-4]
- Guo Z, Jiang S, Li Z, Chen S. Metabolic Syndrome “Interacts” With COVID-19. BIO Integration 2020;1:168-77. [DOI: 10.15212/bioi-2020-0035]
- He X, Liu C, Peng J, Li Z, Li F, et al. COVID-19 induces new-onset insulin resistance and lipid metabolic dysregulation via regulation of secreted metabolic factors. Signal Transduct Target Ther 2021;6:1-427. [PMID: 34916489 DOI: 10.1038/s41392-021-00822-x]
- Lei Y, Zhang J, Schiavon CR, He M, Chen L, et al. SARS-CoV-2 Spike Protein Impairs Endothelial Function via Downregulation of ACE2. bioRxiv 2020. [PMID: 33300001 DOI: 10.1101/2020.12.04.409144]
- Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001;344:1031-7. [PMID: 11287972 DOI: 10.1056/nejm200104053441401]
- Fossati S, Giannoni P, Solesio ME, Cocklin SL, Cabrera E, et al. The carbonic anhydrase inhibitor methazolamide prevents amyloid beta-induced mitochondrial dysfunction and caspase activation protecting neuronal and glial cells in vitro and in the mouse brain. Neurobiol Dis 2016;86:29-40. [PMID: 26581638 DOI: 10.1016/j.nbd.2015.11.006]
- Parving HH, Lehnert H, Bröchner-Mortensen J, Gomis R, Andersen S, et al. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001;345:870-8. [PMID: 11565519 DOI: 10.1056/NEJMoa011489]
- Rizk JG, Wenziger C, Tran D, Hashemi L, Moradi H, et al. Angiotensin-Converting Enzyme Inhibitor and Angiotensin Receptor Blocker Use Associated with Reduced Mortality and Other Disease Outcomes in US Veterans with COVID-19. Drugs 2022;82:43-54. [PMID: 34914085 DOI: 10.1007/s40265-021-01639-2]