Unlocking the Signal Transduction between Glioma-initiating Cells of Tumor Edge
1Department of Neurosurgery, Nanfang Hospital, Southern Medical University, Guangzhou Dadao Bei Street, Guangzhou City 510515, Guangdong Province, China
2Department of Neurosurgery, The First Affiliated Hospital of Shantou University Medical College, No. 57, Changping Road, Shantou City 515000, Guangdong Province, China
3Department of Stomatology, The Seventh Affiliated Hospital of Sun Yat-sen University, No. 628, Zhenyuan Road, Guangming (New) District, Shenzhen City 518107, Guangdong Province, China
aThese authors made equal contribution to the study.
*Correspondence to Shanqiang Qu, E-mail: qushq3@163.com
Received: March 16 2021; Revised: April 19 2021; Accepted: April 21 2021; Published Online: May 20 2021
Cite this paper:
Zihan Wang, Jing Huang and Shanqiang Qu. Unlocking the Signal Transduction between Glioma-initiating Cells of Tumor Edge. BIO Integration 2021; 2(3): 130–132.
DOI: 10.15212/bioi-2021-0010. Available at: https://bio-integration.org/
Download citation
© 2021 The Authors. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See https://bio-integration.org/copyright-and-permissions/
Tumor heterogeneity is a common feature in tumor evolution and plays an important role in tumorigenesis and progression [1]. Previous studies mainly focus on the intra-tumor heterogeneity within the resectable region of glioblastoma multiforme (GBM). GBM cells often infiltrate the surrounding normal brain tissue, which is thought to have a certain biological function and is preserved during the operation. Thus, only a few studies focus on the peritumoral edge lesion. Clinically, almost all patients with GBM relapse in situ, and this part of the tumor cells infiltrating the peritumoral edge lesion is the source of GBM recurrence.
Jin et al. [2] analyzed the heterogeneity of GBM by transcriptional subtyping and found that the expression of proneural genes was significantly increased in the peripheral portion of GBM, whereas mesenchymal genes were significantly expressed in the central portion of GBM. Different populations of GBM cell not only coexist but also influence each other through cell-to-cell communication, promoting tumor malignant behavior together [3]. Therefore, tumor heterogeneity is also an important reason for refractory tumor. However, the interaction patterns between spatially distinct GBM cells and their potential molecular mechanisms remain unclear.
In the current issue of Nature Communications, Bastola et al. [4] focused on the tumor initiation cells in the peritumoral edge lesion, and further confirmed that tumor core cells could promote the proliferation and radiation resistance of glioma-initiating cells at tumor edge in vitro and in vivo. Based on previous studies, Bastola et al. used stereotactic survey to biopsy the lesions with T1-weighted magnetic resonance imaging as core tissue and with non-T1-weighted and high-flair signal magnetic resonance imaging as edge tissue. The core and edge glioma sphere models derived from core and edge tissue were constructed, respectively. There were significant differences not only in histopathological features but also in immunohistochemistry and transcriptional profiling between core tissues and edge tissues. For instance, the core tissue of GBM exhibits high expression of core-marker genes, including CD44, MYC, HIF1α, VIM, ANXA1, CDK6, and JAG1, whereas the edge tissue exhibits high expression edge-marker genes, including OLIG1, TC2, SRRM2, ERBB3, PHGDH, and RAP1GAP. Notably, marker genes related to proneural and mesenchymal subtypes can both be expressed in both edge and core tissues, which suggests that edge tumor cells are not solely composed of proneural (PN) cells and that, similarly, core cells are a mixture of multiple tumor subtypes. However, the distinct molecular properties between core and edge GBM tissues result from the proportion of normal or reactive non-tumorigenic cells. Bastola et al. used edge and core GBM tissues to construct the edge- and core-derived <discrhypen>glioma sphere models, respectively, and then injected these glioma sphere into the brains of mice. The results of the in vivo experiments showed that the core-derived cells were almost completely located in the tumor, whereas the edge-<discrhypen>derived tumor cells existed in a wider part of the mouse brain, and it was further verified that the difference in distribution is associated with the inherent properties of core and edge GBM cells, but not with GBM cell invasion. These findings support the hypothesis that regionally specified GBM sphere models phenocopy intra-tumor spatial identities in vivo.
Clinically, Bastola et al. further found that patients that did not undergo complete GBM resection had poorer prognosis. Small residual tumors have poor prognosis, and this phenomenon is not easy to explain by the real superposition effect of residual core tumors. Bastola et al. speculates that the residual tumor may play an important role in promoting the peripheral infiltrating tumor cells. To verify the conjecture, they used conditioned media (CM) of core-like GBM cells to stimulate edge-like cells and observed that the CM could significantly promote the proliferation and radiation resistance of edge-like cells. The results were also verified in nude mice. Notably, the CM of edge-like cells had no effect on stimulating core-like cells. However, the molecular mechanism of the effect of core-like cells on the malignant phenotype of edge-like cells is still unclear.
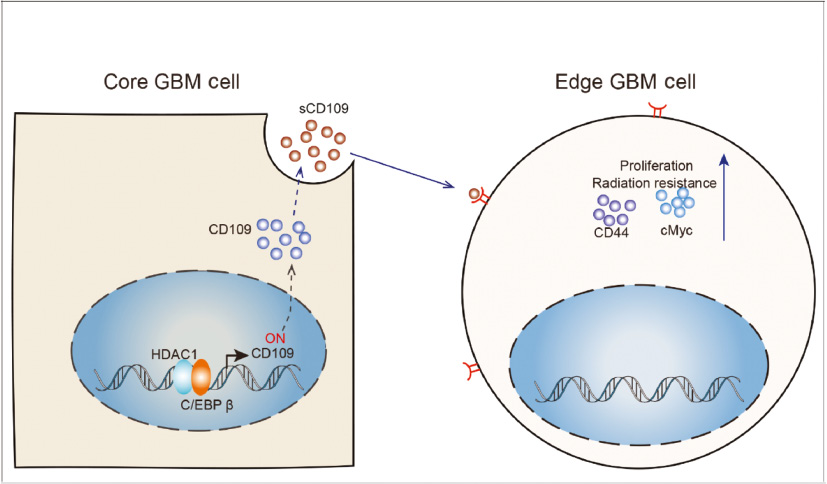
To identify the key gene, a core- and edge-like GBM cell sphere co-culture system was set up in vitro, and 12 compounds with inhibitory effects were identified from more than 30,000 small molecular compounds. Interestingly, among them, half were histone deacetylase (HDAC) inhibitors. A previous study reported that HDAC is a class of molecules related to signal transduction between tumor cells and that changes the spectrum of soluble proteins secreted by tumor cells [5]. However, whether it plays a role in intercellular crosstalk from core to edge GBM cells is unclear. Thus, Bastola et al. further used a lentiviral vector to knockdown HDAC1 in core-like GBM sphere and found that the CM of core-like GBM sphere could not promote the proliferation, radiation resistance, or CD44 expression of edge-like sphere any further. This suggests that the HDAC1 gene plays a key role in the core–edge axis. However, what are the signal molecules of the core–edge axis? Bastola et al. pretreated CM with proteinase K and found that CM pretreated with proteinase K could eliminate the growth-promoting effects of edge-like cells. This suggests that the molecular signal from core-like cells to edge-like cells is likely to be soluble proteins rather than lipids or exosome. The CM of core-like cells was also identified by liquid chromatography mass spectrometry. Finally, four soluble proteins, including CD109, TGFB36, COL12A1, and SRPX37, was identified. It was further confirmed that HDAC1 can regulate the secretion of CD109 in core-like cells. Notably, the ability of core-like cells to mediate the proliferation of edge-like cells could be restored with knock-down of CD109 of core-like cells, confirming that sCD109 is secreted by core-like cells and mediates the proliferation of edge-like cells. However, how does HDAC1 regulate the expression of sCD109 in core-like cells? In view of the fact that HDAC1 can regulate the mRNA and protein levels of CD109, it is suggested that HDAC1 can affect the transcriptional levels of CD109. Chromatin immunoprecipitation assay confirmed that HDAC1 could bind to the promoter of CD109 gene. Bastola et al. previously found that C/EBPβ is a key transcription factor in the expression of CD109 [6]. After HDAC1 was knocked down in core-like cells, chromatin immunoprecipitation assay confirmed that the C/EBPβ protein showed greatly reduced binding to the CD109 promoter. Therefore, HDAC1 positively regulates CD109 in a C/EBPβ-dependent manner in core GBM cells.
Increasing studies showed that patients with GBM could benefit from surgery [7]. Although neurosurgeons have precision equipment such as fluorescence microscopes, it is very difficult to completely remove GBM cells from the brain. The seed cells of tumor recurrence mainly exist in the peritumoral edge lesion. This study reveals a novel mechanism that neighboring residual core cells can mediate the growth and radiotherapy resistance of glioma-initiating cells at tumor edge through HDAC1–CD109 signal axis (Figure 1), thus providing a new potential target for glioma patients with residual tumors. Although this study is the first to reveal intercellular communication between core and edge GBM cells, the protein receptor of sCD109 and how it promotes malignant proliferation and radiotherapy resistance of edge cells remain unclear.
Figure 1 Cell-to-Cell Communications.
Conflict of interests
The authors declare that they have no competing interests.
References
- Easwaran H, Tsai HC, Baylin SB. Cancer epigenetics: tumor heterogeneity, plasticity of stem-like states, and drug resistance. Molecular Cell 2014;54:716-27. [PMID: 24905005 DOI: 10.1016/j.molcel.2014.05.015]
- Jin X, Kim LJY, Wu Q, Wallace LC, Prager BC, et al. Targeting glioma stem cells through combined BMI1 and EZH2 inhibition. Nat Med 2017;23:1352-61. [PMID: 29035367 DOI: 10.1038/nm.4415]
- Jeon HM, Kim SH, Jin X, Park JB, Kim SH, et al. Crosstalk between glioma-initiating cells and endothelial cells drives tumor progression. Cancer Res 2014;74:4482-92. [PMID: 24962027 DOI: 10.1158/0008-5472.CAN-13-1597]
- Bastola S, Pavlyukov MS, Yamashita D, Ghosh S, Cho H, et al. Glioma-initiating cells at tumor edge gain signals from tumor core cells to promote their malignancy. Nat Commun 2020;11:4660. [PMID: 32938908 DOI: 10.1038/s41467-020-18189-y]
- Carta S, Tassi S, Semino C, Fossati G, Mascagni P, et al. Histone deacetylase inhibitors prevent exocytosis of interleukin-1beta-containing secretory lysosomes: role of microtubules. Blood 2006;108:1618-26. [PMID: 16684958 DOI: 10.1182/blood-2006-03-014126]
- Minata M, Audia A, Shi J, Lu S, Bernstock J, et al. Phenotypic plasticity of invasive edge glioma stem-like cells in response to ionizing radiation. Cell Rep 2019;26:1893-905.e7. [PMID: 30759398 DOI: 10.1016/j.celrep.2019.01.076]
- Cajigas I, Mahavadi AK, Shah AH, Borowy V, Abitbol N, et al. Analysis of intra-operative variables as predictors of 30-day readmission in patients undergoing glioma surgery at a single center. J Neurooncol 2019;145:509-18. [PMID: 31642024 DOI: 10.1007/s11060-019-03317-6]